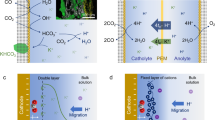
Abstract
Creating and maintaining a favorable microenvironment for electrocatalytic CO2 reduction reaction (eCO2RR) is challenging due to the vigorous interactions with both gas and electrolyte solution during the electrocatalysis. Herein, to boost the performance of eCO2RR, a unique synthetic method that deploys the in situ reduction of precoated precursors is developed to produce activated Ag nanoparticles (NPs) within the gas diffusion layer (GDL), where the thus-obtained Ag NPs-Skeleton can block direct contact between the active Ag sites and electrolyte. Specifically, compared to the conventional surface loading mode in the acidic media, our freestanding and binder free electrode can achieve obvious higher CO selectivity of 94%, CO production rate of 23.3 mol g−1 h−1, single-pass CO2 conversion of 58.6%, and enhanced long-term stability of 8 hours. Our study shows that delivering catalysts within the GDL does not only gain the desired physical protection from GDL skeleton to achieve a superior local microenvironment for more efficient pH-universal eCO2RR, but also manifests the pore structures to effectively address gas accumulation and flood issues.
Similar content being viewed by others
Introduction
The electrocatalytic CO2 reduction reaction (eCO2RR), when powered by renewable electricity, provides a sustainable and appealing pathway to convert the greenhouse gas CO2 into value-added chemicals1,2,3,4. To date, significant efforts have been directed towards the conversion of CO2 to carbon monoxide (CO) due to its economic viability and extensive industrial applications5,6,7,8. Silver (Ag)-based catalysts have emerged as superior candidates for CO production, showing promising prospects for market feasibility based on techno-economic assessments9,10,11. High CO Faradaic efficiencies (FE) of up to 90% has been achieved over various Ag-based catalysts in neutral or alkali media12,13. However, in acidic media, the hydrogen evolution reaction (HER) is normally predominant, resulting in poor catalytic activity. Considering that acidic media have been proved useful with high carbon utilization14,15,16, developing catalysts that work well over a wide pH range is therefore important for providing more opportunities to directly integrate with the preceding carbon capture technology, and following functional reactions17,18,19.
From a technical point of view, constructing a gas/solid/liquid interface in a flow cell, where a powdery Ag-based catalyst deposited on a gas diffusion layer (GDL) has generally served as the cathode, is crucial for achieving industrial-level reaction rates20,21. To improve the eCO2RR performance of Ag-based catalysts at high current densities, extensive attention has been given to strategies for modulating the microenvironment around the electrocatalytic center22,23,24. For examples, the surface modification of Ag catalyst with functional molecule25,26,27 or polymer binders28, could increase the CO2/H2O ratio, effectively inhibiting the HER and driving the eCO2RR. However, these additives may reduce conductivity and impede mass transport within active sites. Moreover, the thus-coated catalysts are prone to detach from the GDL under the impact of gas and electrolyte flush during electrocatalysis because of weak interfacial adhesion29.
Previous successful examples have demonstrated that the construction of metal sandwich electrodes30 and self-supported metal species-decorated carbon membranes31 can greatly enhance eCO2RR activity and stability in alkaline and neutral media. However, such three-dimensional (3D) skeleton-type electrodes have rarely been employed to optimize electrocatalytic eCO2RR behavior in acidic media, primarily due to the high concentration of protons and indispensable cations. In addition, the in-depth understanding of the electrocatalytic mechanism involving the active sites and microenvironment of those electrodes remains elusive, challenged by the encapsulated state of the active sites within the 3D skeleton32,33,34. Therefore, achieving targeted delivery of activated Ag NPs to a superior microenvironment that offers a safe shelter to prevent direct contact with the electrolyte and buffer gas flush, while preserving accessibility to the Ag active sites, is highly desirable for practical eCO2RR applications.
In this work, activated Ag NPs catalysts were in situ generated within GDL skeleton, resulting in a binder free, self-supported electrode. Referred to as Ag NPs-Skeleton, this electrode utilizes a layer of carbon nanoparticles within the GDL to prevent direct contact between the Ag NPs and electrolyte, and buffer gas flush. Therefore, the Ag NPs-Skeleton exhibit a high local CO2/H2O ratio and a high concentration of K+, favoring the formation of a highly active and stable solid-liquid-gas interfacial microenvironment. The Ag NPs-Skeleton exhibit a high CO FE of over 94% for the eCO2RR at a current density of 400 mA cm−2 in pH-universal (acidic, neutral and alkaline) electrolytes over a wide potential range. Impressively, high durability was also achieved for this binder-free electrode, with a remarkably high current density of 100 mA cm−2 for nearly 8 h in acidic media. In situ bubble observation by an optical microscope and COMSOL Multiphysics simulation revealed that the Ag NPs-Skeleton exposed more mass transporting pores and thus equalized the interfacial gas pressure and decreased the gas flow rate of each pore, which can efficiently address gas accumulation and flood issues, and stabilize the catalyst surface. Furthermore, density functional theory (DFT) calculations demonstrated that defect-rich Ag NPs with a high local CO2/H2O ratio promoted H2 suppression and CO production.
Results and Discussion
Preparation of the Ag NPs-Skeleton
The Ag NPs-Skeleton were prepared by a simple in situ electrodeposition method. The silver nitrate (AgNO3) precursor (6 mg mL−1) was sonicated in ethanol for dispersion and drop-casting in the GDL and then in situ electro-reduced at a constant current density of 25 mA cm−2 in a flow cell (Fig. S1). The cross-sectional field emission scanning electron microscopy (FESEM) image and corresponding X-ray energy-dispersive (EDS) mapping images in Fig. 1a-c show that the bright Ag NPs are uniformly distributed within the three-dimensional GDL skeleton, heavily overlapping with the microporous layer (MPL) of carbon nanoparticles with traces in the carbon fiber substrate (CFS) of the GDL (Fig. S2). A negligible amount of Ag NPs was located on the surface of the GDL, as shown in Fig. S3a-b, which shows no difference from the surface of the bare GDL in Fig. S3c. Transmission electron microscopy image (TEM, Fig. 1d) and size distribution plot (Fig. S4) of the Ag NPs-Skeleton illustrate the distribution of Ag NPs with an average diameter of 60 nm. Importantly, high-resolution TEM (HRTEM) images of the Ag NPs (Fig. 1e, Fig. S5) show the presence of a high density of structural defects along the Ag(111) plane35, as evidenced by the periodic stacking sequence in Fig. 1f, g. Moreover, the electron paramagnetic resonance (EPR) spectrum of the Ag NPs-Skeleton as presented in Fig. S6 exhibits a very strong signal at g = 2.003 as arisen from the surface oxygen vacancy, further confirming the existence of defects along the Ag NPs-Skeleton36. The formation of structural defects in Ag NPs-Skeleton could be caused by the disruption of the surface atomic assembly under electroreduction conditions, which is beneficial for improving the adsorption energy and catalytic activity of the intermediates for the eCO2RR37,38.
a Cross-sectional FESEM image and (b) the corresponding EDS mapping images for Ag and C. c Cross-sectional FESEM image at high magnification. d TEM image. e–f HRTEM images of the structural defects in different regions. g Line profiles of the red line in f. h XRD patterns of I: MPL surface (magenta), II: MPL skeleton (gray), III: MPL back (red), and IV: CFS surface (blue), the insert describes the position of I ~ IV in GDL. i Ag 3 d XPS spectrum.
The crystal structures of the Ag NPs-Skeleton in the GDL, including the MPL surface, MPL skeleton, MPL back, and CFS surface, were identified by powder X-ray diffraction (XRD) (Fig. 1h). All samples show diffraction peaks consistent with the face-centered cubic structure of Ag (JCPDs#04-0783)12,39, with the intensity of the Ag signal following the order of MPL skeleton > MPL surface > MPL back > CFS surface, indicating that the Ag NPs are mainly distributed and seamlessly loaded within the MPL of the GDL. Ag NPs-Skeleton also exhibit a characteristic peak with the typical (002) plane feature of graphitic carbon (JCPDSs#41-1487) at 26.6°40,41,42,43. This intense and sharp peak indicates the highly crystalline nature of graphene in the GDL, which provides decent conductivity for electron transfer during electrolysis. The observed shift for the (002) plane of the carbon fiber (curve IV) could be caused by its different crystalline feature and graphitization degree from the top porous carbon sphere in the GDL. In addition, the chemical state of the Ag NPs-Skeleton was examined by X-ray photoelectron spectroscopy (XPS), where the peaks at 374.3 and 368.3 eV (Fig. 1i) could be assigned to the Ag 3d5/2 and Ag 3d3/2 peaks, respectively, suggesting the metallic Ag phase39. In addition, the Ag signal intensity also follows the sequence of MPL skeleton > MPL surface > MPL back > CFS surface (Fig. S7), which is consistent with the XRD results. Thus, these results underscore the effectiveness of a simple method involving drop coating silver precursors and in situ electroreduction to obtain evenly distributed Ag NPs-Skeleton with abundant structural defects located in the porous carbon nanoparticle layer of the GDL, enabling direct utilization as an electrode without any extra use of organic binder.
eCO2RR performance in a flow cell
The electrochemical performance for the eCO2RR was firstly studied in acidic media by performing bulk electrolysis in 1 M KCl with the pH adjusted to 1.42 ± 0.03 by 20 mM H2SO4. A high concentration of K+ cations can make the electrode-electrolyte interface alkaline when the current density increases, effectively suppressing the HER in acidic electrolytes6,20,44. The eCO2RR performances of the bare and prepared GDL used in our work were evaluated with H2 Faradaic efficiency (FE) of almost 100% (Fig. S8), excluding the electrocatalytic contribution of GDL substrate. The products of the Ag NPs-Skeleton were CO (94%) and H2 (6%), as evidenced by nuclear magnetic resonance (NMR) spectroscopy results with no signal from liquid products which confirmed the good eCO2RR performance (Fig. 2a, Fig. S9). To exclude the possibility of over-compensation of iR, particularly at high current densities, the resistances were measured and non-compensate potentials were compared among these samples (Fig. S10, Fig. S11), and the same trends of potentials were observed to the 100% iR-corrected one (Fig. 2, Fig. 3). Previous work from Fan et al.6 reported that the FE for CO of commercial gold nanoparticles that were sprayed onto the surface of a GDL at pH = 2 was 40% at current densities below 50 mA cm−2. Similar results were also obtained for a Ag-PTFE GDL with 53% FECO33. The low FEs for CO production can be understood because the corresponding production of OH– is not sufficient to neutralize the protons diffusing from the bulk acidic media toward the catalyst surface, thus favors the HER. In our case, the in situ formed OH– during the eCO2RR process can rapidly neutralize the local acidic environment because they are far away from the bulk electrolyte. As a result, the Ag NPs-Skeleton displayed superior selectivity for CO over a wide range of current densities from 25 mA cm−2 to 400 mA cm−2 (Fig. 2a). The Ag NPs-Skeleton electrode can maintain nearly 85% of the FECO during the 8 h stability test (Fig. 2b). Both the morphology and composition of the Ag NPs-Skeleton electrode remained stable, as indicated by the post-reaction XRD and TEM measurements (Fig. S12a-e). Moreover, the Ag content was determined before and after stability testing with 0.27 wt% and 0.26 wt%, respectively, using Inductively Coupled Plasma Atomic Emission Spectroscopy (ICP-AES), confirming the decent stability of the self-supported Ag NPs-Skeleton electrode (Fig. S12f). However, over the extended time, due to the gradual variation in hydrophobicity and the presence of too many cracks on the GDL surface (Fig. S2), the electrolyte gradually permeates into the gas layer as electrolysis proceeds, eventually leading to flooding. Future systematic research can focus on optimizing the GDL properties to further improve the eCO2RR stability.
a Potentials and product FEs of Ag NPs-Skeleton at different current densities. b potential and FECO variation of Ag NPs-Skeleton during long-term electrolysis at 100 mA cm−2 in 20 mM H2SO4 + 1 M KCl. (c) Product FEs and potentials of Ag NPs-Skeleton with various electrolytes. d Product FEs and potentials of Ag NPs-Skeleton, Ag NPs, and Ag MP GDL in 20 mM H2SO4 + 1 M KCl at 100 mA cm−2. e SPCE and CO yield rate of Ag NPs-Skeleton, Ag NPs and Ag MPs at 100 mA cm−2. f Comparison of the eCO2RR performance on the current density, CO product FE, and pH of the bulk electrolyte for Ag NPs-Skeleton with those of commercial Ag NPs, Ag MPs and other state-of-the-art electrocatalysts. This comparison is limited to reports with a pH ≤ 4.0 or with a total current density > 10 mA cm−2. All the eCO2RR tests here were conducted in a flow cell of 15 sccm CO2 flow rate at 25 °C. For panels (a), (c) and (d), 100% iR correction was applied. Error bars in (a), (c), (d) and (e) represent the standard deviation based on at least two or three separate measurements. Relevant source data are provided as a Source Data file.
Potentials and product FEs of (a) Ag NPs-Skeleton covered by MWCNTs and (b) Ag NPs-Skeleton washed with electrolyte for 1 h in 20 mM H2SO4 + 1 M KCl at various current densities. c FESEM and EDS mapping images for K of the Ag NPs-Skeleton after eCO2RR at 100 mA cm−2 for 20 min. d Humidity variation curves of the flow cell with/without electrolysis at a current density of 25 mA cm−2, (e) contact angles, and (f) FECO of Ag NPs-Skeleton and Ag NPs-Skeleton-Ot. All the eCO2RR tests here were conducted in a flow cell of 15 sccm CO2 flow rate at 25 °C. For panels (a) and (b), 100% iR correction was applied. Error bars in (a), (b), (d) and (f) represent the standard deviation based on at least two or three separate measurements. Relevant source data are provided as a Source Data file.
Subsequently, we investigated the loading effect of the Ag NPs-Skeleton under the same conditions by conducting the eCO2RR under a constant current density of 100 mA cm−2 with different catalyst loadings, and the results were summarized in Fig. S13a. No significant changes in CO selectivity were observed, but at a loading of 0.3 mg cm−2, the electrode exhibits the lowest overpotential. As the loading increases to 0.3 mg cm−2, additional Ag NPs within the skeleton enhance the electron transport and lower the electrode resistance, enabling higher current densities at lower potentials. But further increasing loading would occur the aggregating of Ag nanoparticles on GDL surface (Fig. S14), causing bubble formation in the electrolyte and increasing interface resistance, and thus resulting in the voltage raise. Therefore, we chose a catalyst loading of 0.3 mg cm−2 for the subsequent investigations, which led us to achieve high CO yield rates of 6.3 mol g−1 h−1 at 100 m A cm−2 and 23.3 mol g−1 h−1 at 400 m A cm−2 (Fig. S13b). Moreover, we examined the eCO2RR product distribution of Ag NPs-Skeleton in 20 mM H2SO4 with different concentrations of K+ at a current density of 100 mA cm−2, which was expected to shed light on the key role of K+ in suppressing the HER45. Fig. S13c shows that CO2 can be converted with a high selectivity even at a low concentration of K+ (0.01 M), and a higher concentration of K+ in the electrolyte results in a lower resistance and cell potential33,46. These results illustrate that the presence of K+ is truly important for the selective eCO2RR in acidic media, and the local cation concentration of the Ag NPs-Skeleton is sufficient even at a low concentration of K+ in the bulk electrolyte for the Ag NPs-Skeleton, which is beneficial for alleviating the salt deposition issue. Considering the applied potential, a K+ concentration of 1.0 M was chosen for the following investigation. As mentioned above, the challenge of carbonate formation in the alkaline eCO2RR can be addressed by improving the carbon utilization efficiency by using acidic media14. To evaluate the carbon utilization efficiency of the Ag NPs-Skeleton in acidic electrolytes, the single-pass CO2 conversion efficiency (SPCE) for the eCO2RR was measured at 100 mA cm−2 with different CO2 flow rates, as presented in Fig. S13d. At a higher flow rate, the SPCE was relatively lower since the CO2 input greatly exceeded the consumption. By gradually decreasing the flow rate of CO2 to 3 sccm, the SPCE reached 58.6% for CO production, reflecting the high carbon utilization efficiency of the Ag NPs-Skeleton.
Notably, Ag NPs-Skeleton exhibit versatility in a wide range of pH environments, including acidic, buffered, neutral and alkaline electrolytes, with all CO selectivity above 90% at a current density of 100 mA cm−2 (Fig. 2c), making them promising candidates for the eCO2RR in different practical applications or for coupling with various anodic reactions. To make an intuitive comparison, the control samples, commercial silver nanoparticles (Ag NPs, 50-100 nm) and silver macroparticles (Ag MPs, 1-3 µm) coated on GDLs by the drop-casting method with the use of Nafion, were tested for the eCO2RR in acidic media. The commercial Ag NPs disperse along the porous carbon nanoparticle layer of the GDL as Ag NPs-Skeleton but are more retained on the surface of the GDL (Fig. S15a-c). The commercial Ag MPs are uniformly distributed on the GDL surface without entering the skeleton due to their large size (Fig. S15d-f). XRD patterns of these control samples are shown in Fig. S15g to identify the material phases, which are identical to Ag NPs-Skeleton with metallic Ag phase. Furthermore, N2 adsorption-desorption measurements (Fig. S16) indicate that the Brunauer-Emmett-Teller specific surface areas of Ag NPs-skeleton, Ag NPs, and Ag MPs are all approximately to be 13 m2 g−1. Despite the similar specific surface areas of Ag NPs-skeleton, Ag NPs, and Ag MPs and close particle sizes for Ag NPs-skeleton and Ag NPs, the three samples display distinct eCO2RR performances. Obviously, Ag NPs and Ag MPs show much lower FEs, SPCEs and CO yield rates at the same current density as Ag NPs-Skeleton (Fig. 2d and Fig. S15h, i), such as CO FEs of 58% and 47%, SPCEs of 35% and 29%, and CO yield rates of 3.4 and 3.0 mol g−1 h−1 at 100 mA cm−2, respectively. The results show that the specific surface area and the particle size have little influence on the catalytic performance. Compared with recently reported CO production electrocatalysts and the control powdery electrocatalysts used in this work, the Ag NPs-Skeleton show a higher current density, greater CO product FE, and lower pH of the bulk electrolyte, making their performance among the best in acidic media (Fig. 2f, Table S1)6,24,32,33,47,48,49,50,51,52,53,54,55.
More importantly, the synthesis method can be extended to other soluble Ag and Cu salts, such as silver tetrafluoroborate (AgBF4) and copper nitrate (Cu(NO3)2). The structural characteristics and eCO2RR performance for Ag NPs-Skeleton with AgBF4 as a precursor are shown in Fig. S17 and are like these prepared with AgNO3. Similarly, Cu NPs-Skeleton using Cu(NO3)2 as a precursor (Fig. S18) could achieve a significantly higher eCO2RR product FE of 75% than that of the commercial Cu nanoparticles (Cu NPs) coated on GDLs using the drop-casting method (FE 27%) at 300 mA cm−2 (Fig. S19). These results suggest that the targeted delivery of active sites to the porous carbon nanoparticle layer of the GDL give a great university for creating a favorable microenvironment for the electrocatalytic reaction sites, which is significant for promoting eCO2RR performance. However, the performance of eCO2RR in acidic media of the Cu NPs-Skeleton was not as satisfactory as the Ag NPs-Skeleton. To enhance its activity in acidic media, strategies such as alloying with other metals to tune electronic structure44,56, and modified with cationic organic polyelectrolyte to improve Faradaic efficiency can be future employed57.
The role of the microenvironment in the eCO2RR
Due to the isolation effect of the Ag NPs-Skeleton, most Ag sites are separated from the bulk electrolyte, raising questions of whether the Ag sites located in the skeleton could contribute to the eCO2RR. To address this inquiry, the surface of the Ag NPs-Skeleton was covered with multiwalled carbon nanotubes (MWCNTs, Fig. S20a), which show no electrocatalytic activity for eCO2RR40, or washed with electrolyte for 1 h to dissolve the surface soluble AgNO3 salt before in situ electroreduction (Fig. S20b), respectively. Both methods are aimed at eliminating the direct contact between the Ag NPs and the electrolyte. The XRD patterns of these control samples revealed that the intensity of the typical Ag signal was much weaker than that of Ag NPs-Skeleton (Fig. S20c, Fig. 1h). The thus-treated Ag NPs-Skeleton could still maintain a high CO production selectivity with an FECO > 90% at current densities ranging from 25 to 400 mA cm−2 (Fig. 3a, b, Fig. S20d). The outcome suggested that the modifications implemented had effectively altered the surface characteristics of the Ag NPs-Skeleton while still maintain good eCO2RR performance, indicating that the Ag NPs within the 3D carbon skeleton could act as the main active sites for the eCO2RR, which can provide more active sites than that of the traditional surface coated method. To smoothly promote the eCO2RR, not only are electrocatalytic active sites needed but also a suitable microenvironment is indispensable. First, the cation (K+) presence of 1.6 at% is verified by SEM-EDS mapping images of the Ag NPs-Skeleton electrode during the eCO2RR (Fig. 3c), which shows that a large amount of K+ disperses homogeneously across the carbon skeleton. This could help increase the local pH and stabilize key intermediates of the eCO2RR58.
Afterwards, the critical proton provider, water59, was checked by connecting a buffering gas container equipped with a humidity meter at the gas chamber exit of the flow cell (Fig. 3d). Due to the transpiration effect of CO2 gas, the water could evaporate and pass through the GDL into the gas chamber even without the electrolysis process, resulting a humidity of 60% after 1 h. After applying the potential, the water evaporation became more severe at a humidity of 74% after 1 h, which possibly resulted from the heating effect during the eCO2RR. These results show that the Ag NPs-Skeleton could reach the available water to support the eCO2RR, and the amount of water vapor around the Ag active sites was much less than that in the surface liquid water, resulting in a comparatively high local CO2/H2O ratio, which is quite important for suppressing the HER and promoting the eCO2RR. To confirm the important role of the local CO2/H2O ratio for the eCO2RR on Ag NPs-Skeleton electrode, an electro-oxidation treatment of the GDL by a bias voltage was conducted to increase the surface hydrophilicity60. The water contact angle of the GDL decreased from 133.7° to 123.3° after applying 2 V vs Ag/AgCl for 1000 s (Fig. S21), thus providing a lower water contact angle for the electro-oxidation-treated GDL loaded Ag NPs-Skeleton (Ag NPs-Skeleton-Ot) than for the plain GDL loaded Ag NPs-Skeleton (Fig. 3e). The decreased hydrophobicity could be ascribed to the detachment of the polytetrafluoroethylene along with the carbon corrosion. As expected, the Ag NPs-Skeleton-Ot show more water evaporation with a humidity of 84% after 1 h (Fig. 3d) and an evidently lower FECO from 25 to 200 mA cm−2 in acidic media (Fig. 3f). Furthermore, the local pH during the eCO2RR could be tested by pH test strips as they were pasted on the back of the cathode GDL because of water evaporation. Fig. S22 shows that the Ag NPs-Skeleton electrode has a pH of ~9, which is higher than that of the Skeleton Ag NP-Ot electrode (pH = ~8). This suggests that the continuously generated OH− during eCO2RR on Ag NPs-Skeleton electrode could thoroughly neutralize the local protons carried by the water evaporation. A partial of the OH− would react with the rinsed CO2 forming soluble (bi)carbonate species to maintain a local micro-alkaline microenvironment for suppressing the HER14,16. The residual OH−, and formed (bi)carbonate species might permeate into the gas chamber by the water vapor or get into the bulk electrolyte by the CO2 blowing to guarantee a continuous and stable electrocatalytic process. These results illustrate that the space confinement of active Ag NPs within the hydrophobic GDL skeleton can construct a favorable microenvironment through water evaporation, such as a high concentration of local cations and hydroxides and an increased CO2/H2O ratio, for the eCO2RR.
Bubble management at the reaction interface
The evolution and transport of bubbles significantly complicate the gas/solid/liquid three-phase electrocatalytic reaction interface61,62, and an unambiguous understanding of bubble behaviors and their impacts on eCO2RR performance has been scarce. An in situ flow cell for the observation of gas bubble formation through an optical microscope was designed and is shown in Fig. S23. With no electrolysis, the top views of the catalyst surface of Ag NPs-Skeleton, Ag NPs and Ag MPs and the electrolyte surface all showed no bubble formation. Once the potential was applied, the Ag NPs-Skeleton provided a static electrolyte surface with no bubble inflation, while a few small gas bubbles and large bubbles on the Ag NPs and Ag MPs were observed at the same current density of 100 mA cm−2 (Fig. 4). Therefore, the appearance of bubbles is highly dependent on the location of the Ag active sites. The permeability experiment can illustrate the effect of Ag site positions on the electrode structure63,64. As presented in Fig. S24, the CO2 and water vapor permeability within Ag NPs-Skeleton achieves the value of 294 L m−2 day−1 and 2.72 kg m−2 day−1, respectively, significantly higher than those of Ag NPs (159 L m−2 day−1 and 2.64 kg m−2 day−1) and Ag MPs (7.0 L m−2 day−1 and 2.62 kg m−2 day−1). This reflects that the Ag sites within the skeleton do not block the pores of the GDL and provide a favorable gas-transferring path to achieve a high ratio of CO2/H2O. Moreover, for the Ag NPs-Skeleton, the Ag sites are mainly located within the framework of the GDL (Fig. S3a) without blocking the pores in the electrode and do not directly contact the electrolyte; thus, the bubbles generated in the skeleton dissipate within the framework and do not enter the electrolyte layer. Conversely, Ag MPs are mainly located on the surface of the GDL (Fig. S15d) blocking the pores and directly contact the electrolyte. The generation and dissolution of bubbles during the eCO2RR occurred in the electrolyte layer, which caused a turbulent three-phase interface and poor electrocatalytic stability. Ag NPs are located both on the surface and within the framework of the GDL blocking the pores partially (Fig. S15a). Hence, a portion of the generated bubbles dissipate within the framework, while others enter the electrolyte layer to destabilize the three-phase interface. As a result, the existence of vast bubbles hindered ion transport and led to voltage fluctuations (Fig. S25), resulting in much worse stability and lower CO FEs of Ag NPs and Ag MPs than those of Ag NPs-Skeleton (Fig. 2b). These results reveal that the skeleton loading mode of Ag NPs within the hydrophobic GDL skeleton can efficiently control bubble behaviors, such as decreasing bubble coverage, and preventing gas accumulation in the flow cell. These factors are highly important for improving the mass transport, catalyst utilization, and stability of the eCO2RR.
The bubble evolution process ascribed to CO exsolution produced on the electrodes of (a) Ag NPs-Skeleton, (b) Ag NPs and (c) Ag MPs.
To further elucidate the intrinsic mechanism behind bubble evolution and transport in the eCO2RR, COMSOL Multiphysics simulation65,66,67,68 was employed to study the motion characteristics of bubbles generated during the transport process of mixed gases CO2, CO, and H2 under the effect of electric field force (Fig. 5). Results obtained from Hg porosimetry showed that the pore size distributions of the Ag NPs-Skeleton, Ag NPs and Ag MPs are close to each other within 5 nm-100 µm, while within the macropore range (100-500 µm), the pore volume intensity of the Ag NPs-Skeleton is evidently higher than those of the contrasts (Fig. S26), depicting its enhanced available pores for gas transferring, which is consistent with the CO2 and water vapor permeability results (Fig. S24). Therefore, the gas inlet velocity of the pores on the GDL surface follows the sequence: Ag NPs-Skeleton <Ag NPs <Ag MPs, the pore quantity on the GDL surface follows the sequence: Ag NPs-Skeleton > Ag NPs > Ag MPs, and the pore separation distance on the GDL surface follows the sequence: Ag NPs-Skeleton <Ag NPs <Ag MPs (Fig. S27). These factors are all contributing factors to the formation of bubbles. At a gas inlet velocity of 0.1 m s−1, no bubble was able to penetrate the pores and form a gas film within the computational time. When the inlet velocity was increased to 0.115 m s−1, only one bubble appeared and was able to enter the liquid phase (Fig. S28a), confirming that the critical velocity was approximately 0.1 m s−1 to avoid the appearance of bubbles. Furthermore, the influence of velocities ranging from 0.5 to 1.5 m s−1 on the gas film length and bubble quantity was investigated (Fig. 5a-b). It is evident that elevated velocities lead to more pronounced expansion of the gas film, rendering it more susceptible to shear forces and resulting in an increased frequency of bubble growth, which is applicable for the Ag MPs sample. The increase in the number of pores results in fewer bubbles, as revealed by the shorter gas film in the Ag NPs-Skeleton sample (Fig. 5c).
a Gas spreading film length curve at various gas velocities and (b) their corresponding velocity clouds. c Velocity cloud of the gas spreading film length with various pores. d Gas spreading film length between pore 2 and pore 3. Velocity cloud of (e) a single gas inlet and (f) multiple independent inlets at a 175 μm pore separation distance. Gas area (red), Liquid area (blue) and bubble (yellow).
Furthermore, the quantity of bubbles follows a volcano-shaped curve in response to variations in the pore distance. In our model, three pores with separation distances ranging from 125 to 500 μm were simulated based on the Hg porosimetry results (Fig. 5d, Fig. S26 and Fig. S28b). When the pore spacing separation distance decreases from 500 to 250 μm, the gas film at pore 1 rapidly ruptures upon reaching 250 μm due to the disruptive influence of gas film growth at pore 2, making bubble formation easier (Fig. S28c). However, as the pore separation distance is further decreased to 125 μm, the growth process of the gas film completely covers both pore 1 and pore 2. At this point, the gas film growth and bubble detachment processes resemble those of a single pore (Fig. S28d and Fig. S29). Therefore, a comparatively shorter pore separation distance of approximately 175 μm is conducive to preventing bubble formation, in good accordance with the result that the Ag NPs-Skeleton has enhanced pores of 175 μm (Fig. S26). Additionally, considering that the Ag NPs-Skeleton could homogenize the gas pressure of the pores on the GDL surface, we changed the single gas inlet of the traditional flow cell (Fig. 5e) to multiple independent inlets in our simulation and observed the obvious suppression of liquid backfilling into the gas chamber, i.e., flooding (Fig. 5f). Consequently, microenvironment regulation to achieve a relatively stable fluid field through smart catalyst loading and reactor design contributes to the long-term stability and high CO Faradaic efficiency of the eCO2RR at industrial current densities.
Density functional theory (DFT) calculations
DFT calculations, widely used to understand eCO2RR reaction mechanisms69,70, were conducted to elucidate the enhanced selectivity and stability of the Ag NPs-Skeleton. The assumption is that the dramatically enhanced activity can be related to the specific crystalline structure, namely, evident vacancy defects on the Ag NPs-Skeleton (Fig. 1e, Fig. S6). Two simulation models, i.e., the Ag(111) surface and Ag(111) surface with vacancies (denoted as Ag111-v, Fig. 6a and Fig. S30), are employed to evaluate the CO formation pathway. As shown in Fig. 6b, the potential-determining step energy (PDS), *COOH formation, of Ag111-v is ~0.2 eV, lower than that of Ag(111), which facilitates CO formation, demonstrating that the existence of structural defects are responsible in promoting eCO2RR activity. In addition, we further evaluated the effect of the CO2/H2O ratio on the activity of Ag NPs-Skeleton (Fig. 6c). The constructed CO2/H2O ratios were set as 10, and 8.33×10−2 for the Ag(111)-v-CO2 rich, and Ag(111)-v-H2O rich, respectively. With the high coverage of water on the Ag surface, the abundant Ag(111)-H2O provides a highly favorable energy for the HER. Compared to the Ag(111)-H2O-rich sample, the Ag(111)-CO2-rich sample evidently has a higher energy barrier for the HER. Coupled with the introduction of vacancies, the Ag(111)-v-CO2-rich catalyst further achieves an increased energy barrier for the HER in contrast to the bare Ag(111)-CO2-rich catalyst and Ag(111)-v. The concentration of CO2 are much higher on Ag(111)-v-CO2 rich surface, that is, the coverage of CO2 on active sites is obviously enhanced, which can be facilitated to CO2 activation and reduction while suppress HER, as indicated by the energetics calculation (Fig. 6b, c). These results are in line with experimental observations and highlight the importance of interface microenvironment modulation.
a Top view of Ag(111)-v, Ag(111)-v-CO2 rich, Ag(111)-v-H2O rich, and Ag(111)-CO2 rich configurations. b Calculated Gibbs free energy of the eCO2RR intermediates for CO formation on Ag(111) and Ag(111)-v surfaces. c Calculated Gibbs free energy of the HER on Ag(111)-v-, Ag(111)-v-CO2-rich, Ag(111)-v-H2O-rich, and Ag(111)-CO2-rich electrodes. d Schematic diagrams of the eCO2RR using skeleton loading mode compared with the surface loading mode. Raw computational and simulation data are provided as Source Data and Supplementary Data 1 files.
Overall, as illustrated in Fig. 6d, the underlying reason for the increase in the eCO2RR performance of the Ag NPs-Skeleton is based on the skeleton loading mode used for the delivery of the active Ag sites to the superior microenvironment. Specifically, compared to the conventional surface loading mode, the skeleton loading mode can embed active sites into the GDL framework and construct a beneficial local microenvironment for eCO2RR with a high CO2/H2O ratio, a low concentration of protons, and a static fluid field with a suppressed bubbles generation. Consequently, the Ag NPs-Skeleton show significantly enhanced CO FEs, yields and SPECs over a wide range of potentials, pH ranges, and decent long-term stabilities.
In summary, a freestanding, binder-free electrode composed of activated Ag NPs catalysts located uniformly within GDL skeleton was successfully designed. The in situ reduction procedure ensures the intimate interactions between the catalysts and skeleton, and therefore promoting good electronic conductivity during the eCO2RR. The GDL skeleton effectively protects Ag NPs from direct erosion caused by the physical impacts of electrolyte flow and gas bubble evolution, leading to a greatly improved stability. As supported by the in situ observation of the optical microscope and COMSOL Multiphysics simulation, delivering catalysts within the GDL also manifests the pore structures, leading to the reduction of bubble formation/coverage and the alleviation of gas accumulation issues, and thereby enhancing the eCO2RR performance. DFT calculations further demonstrate that the regulation of the catalyst structure (vacancy defect) and local microenvironment (CO2/H2O ratio) is responsible for improving the eCO2RR activity. The achievement of wide potential and wide pH adaptability for CO production allows for the coupling of the eCO2RR with other anodic reactions beyond the OER or tandem cathodic reactions to directly employ the in situ formed CO for higher reaction efficiency and more value-added products.
Methods
Chemicals and reagents
All chemicals were used as received without further treatments. Silver nitrate (AgNO3, ≥99.8%, Aladdin), silver tetrafluoroborate (AgBF4) (HISPEC3000, 99.9%, Johnson Matthey), copper nitrate hydrate (Cu(NO3)2·3H2O, 99.9%, Aladdin), Ag nanoparticles (Ag NPs, 99.5%, Meryer company, 60-120 nm), Ag microparticles (Ag MPs, 99.9%, thermos scientific company, 1-3 µm), Cu nanoparticles (Cu NPs, 99.5%, Xiya chemical Technology, 40 nm), iridium oxide (IrO2, 99.9%, Xiya Agent), Titanium mesh (Ti-mesh, ≥99.6%, Shang Te company), Nafion (10 wt% aqueous solution, Fuel Cell Store) multiwalled carbon nanotubes (MWCNTs) ( > 98%, Sigma-Aldrich), were used as received. The commercial gas diffusion layer (GDL, Sigracet 29 BC, Fuel Cell Store) contains a microporous layer (MPL) blending with ~20% polytetrafluoroethylene coating onto a macroporous carbon fiber substrate (CFS) (Fig. S2a). The X-ray powder diffraction (XRD) result shows the GDL with typical (002) plane feature of a graphitic carbon (JCPDS card no. 41-1487) (Fig. S2b). The prepared GDL was made by blending carbon sphere (Aladdin, 99.5%, 30 nm) and ~20% polytetrafluoroethylene (Aladdin, 60 wt% dispersion) together, which was then coated onto a pristine carbon fiber paper (Fuel Cell Store). Ethanol (EA, 99.7%), Isopropyl alcohol (IPA, 99.7%), and potassium hydroxide (KOH) were purchased from Sinopharm Chemical Reagent Co., Ltd. Potassium hydroxide (KOH, 95%), Potassium bicarbonate (KHCO3, 99%), and potassium chloride (KCl, 99.8%) were purchased from Innochem. High-purity carbon dioxide (CO2, 99.993%) was purchased from Wenzhou Haichang gas company.
Preparation of Ag NPs-Skeleton electrode
AgNO3 (24 mg) was dissolved in EA (4 mL). The solution was stirred using a magnetic stir bar until there was no visible powder. AgNO3 ink was then directly coated on GDL with an area of 1 cm × 2 cm to get a loading of 0.5 mg cm−2 as the working electrode. The GDL used in this work contains a microporous layer (MPL) of carbon nanoparticles blending with ~20% polytetrafluoroethylene coated onto a macroporous carbon fiber substrate (CFS) to create a hydrophobic interface and facilitate gas diffusion to the catalytic surface71. The Ag NPs-Skeleton were then obtained by an in situ electro-reduced at a constant current density of 25 mA cm−2 in a flow cell to get Ag nanoparticles with a loading of 0.3 mg cm−2. The GDL was weighed before and after drop-casting to record its actual catalyst loading.
Preparation of commercial Ag NPs electrode
To prepare the catalyst ink, 25 mg of Ag NPs was ultrasonically dispersed in the mixture of 3 mL of EA and 20 µL of Nafion (10 wt% aqueous solution, Fuel Cell Store, America). Then, the mixture was sonicated for 30 min. The ink was then drop-casted onto the GDL to get a loading of 0.3 mg cm−2. The GDL was weighed before and after drop-casting to record its actual catalyst loading.
Preparation of commercial Ag MPs electrode
To prepare the catalyst ink, 25 mg of Ag MPs was ultrasonically dispersed in the mixture of 3 mL of EA and 20 µL of Nafion (10 wt% aqueous solution, Fuel Cell Store, America). Then, the mixture was sonicated for 30 min. The ink was then drop-casted onto the GDL to get a loading of 0.3 mg cm−2. The GDL was weighed before and after drop-casting to record its actual catalyst loading.
Preparation of Ag NPs-Skeleton covered by MWCNTs electrode
MWCNTs solution (5 mg MWCNTs dispersed in 5 mL tetrahydrofuran (THF)) was dropcasted onto the surface of the Ag NPs-Skeleton electrode with a loading of 0.3 mg cm−2 to get the Ag NPs-Skeleton covered by MWCNTs electrode. The GDL was weighed before and after drop-casting to record its actual MWCNTs loading.
Preparation of Ag NPs-Skeleton washed with electrolytes electrode
Ag NPs-Skeleton electrode was washed with electrolyte for 1 h to dissolve the surface soluble AgNO3 salt before in situ electroreduction. The Ag NPs-Skeleton washed with electrolytes electrode were then obtained by an in situ electro-reduced at a constant current density of 25 mA cm−2.
Preparation of Ag NPs-Skeleton-Ot electrode
The GDL was firstly conducted by a bias voltage of 2 V vs Ag/AgCl for 1000 s. AgNO3 ink was then directly coated on the treated GDL with an area of 1 cm × 2 cm to get a loading of 0.5 mg cm−2 as the working electrode. The treated GDL was weighed before and after drop-casting to record its actual catalyst loading.
Preparation of Skeleton Ag NPs with AgBF4 as a precursor electrode
AgBF4 (24 mg) was dissolved in EA (4 mL). The solution was stirred using a magnetic stir bar until there was no visible powder. AgBF4 ink was then directly coated on GDL with an area of 1 cm×2 cm to get a loading of 1.1 mg cm−2 as the working electrode. The Ag NPs-Skeleton were then obtained by an in situ electro-reduced at a constant current density of 25 mA cm−2 in a flow cell to get Ag nanoparticles with a loading of 0.3 mg cm−2. The GDL was weighed before and after drop-casting to record its actual catalyst loading.
Preparation of Cu NPs-Skeleton electrode
Cu(NO3)2·3H2O (24 mg) was dissolved in EA (4 mL). The solution was stirred using a magnetic stir bar until there was no visible powder. The ink was then directly coated on GDL with an area of 1 cm × 2 cm to get a loading of 2.2 mg cm−2 as the working electrode. The Ag NPs-Skeleton were then obtained by an in situ electro-reduced at a constant current density of 25 mA cm−2 in a flow cell to get Cu nanoparticles with a loading of 0.3 mg cm−2. The GDL was weighed before and after drop-casting to record its actual catalyst loading
Preparation of commercial surface Cu NPs electrode
To prepare the catalyst ink, 25 mg of Cu NPs was ultrasonically dispersed in the mixture of 3 mL of EA and 20 µL of Nafion. Then, the mixture was sonicated for 30 min. The ink was then drop-casted onto the GDL to get a loading of 0.3 mg cm−2. The GDL was weighed before and after drop-casting to record its actual catalyst loading.
Structure characterization
Surface morphology and atomic ratio of the samples was characterized using a Tescan Mira LMS scanning electron microscopy (SEM) operated at 20.0 kV and the energy dispersive spectrometer (EDS) attached to the SEM, respectively. The transmission electron microscopy (TEM), and high-resolution TEM (HRTEM) using a JEM-2100F TEM operating at 200 kV. X-ray diffraction (XRD) patterns (Bruker D8 Advanced) were obtained to determine the crystal structures of the samples. X-ray photoelectron spectroscopy (XPS) experiment was carried out using Thermo Scientific K-Alpha. All spectra were calibrated according to the C 1 s binding energy at 284.8 eV. Contact angle measurements were tested on a Contact Angle Measuring Device (SDC-100S, Dynetech) with a drop of aqueous electrolyte on the samples, which were thoroughly cleaned and dried as before measure. The gas bubble formation was characterized by an in-situ cell through an optical microscope (OLYMPUS, DSX1000). Macropore pore size distribution was analyzed by the mercury intrusion porosimetry. The analysis was conducted on a Micromeritics porosimeter (Micromeritics Autopore V 9620) with Hg contact angle of 140.9 degrees and Hg surface tension of 485 dynes cm−1. The Hg intrusion pressure was set from 0.2 to 33,000 psia. The EPR measurement was performed on an Endor spectrometer (Bruker EMXplus-9.5/12). The g factor was obtained by taking the signal of manganese as a standard. Micromeritics ASAP 2460 was used for measuring the specific Brunauer-Emmett-Teller (BET) surface area. Ag content was measured by Inductively coupled plasma atomic emission spectrometry (ICP-AES, 720ES) after mineralization at 200 °C for 35 min under pressure (Multiwave 3000, Anton Paar) using 69% HNO3 (TraceSELECT, Fluka Analytical) and subsequent dilution by 2% HNO3. CO2 and water vapor permeability were measured with the MOCON 430 tester and Lan Guang C360M tester, respectively. pH meter (PHSJ-3F, Zici, Shanghai) was used to test the pH of the electrolyte.
Cell assembly
Custom-built electrochemical flow cell assembly
The flow cell, depicted in Fig. S1, comprises a gas chamber, a catholyte chamber located ≈ 5 cm from the cathode (housing an Ag/AgCl reference electrode, saturated KCl, CHI111), an anolyte chamber, and sealing plates. After assembling, the catholyte and anolyte chambers are separated by an ion exchange membrane. The working electrode is positioned between the gas chamber and the catholyte chamber, while the counter electrode (IrO2-loaded Ti mesh) is placed between the anolyte chamber and the anodic plate. The dimension of the three compartments is 2 × 0.5 × 0.2 cm3. The electrode area is 1 cm2 and the electrode-to-membrane distance is 2 mm.
In situ flow cell assembly
The flow cell, depicted in Fig. S23, comprises a gas chamber, a catholyte chamber located ≈5 cm from the cathode (housing an Ag/AgCl reference electrode, saturated KCl, CHI111), an anolyte chamber, and sealing plates. After assembling, the catholyte and anolyte chambers are separated by an ion exchange membrane. The working electrode is positioned between the gas chamber and the catholyte chamber, while the counter electrode (IrO2-loaded Ti mesh) is placed between the anolyte chamber and the anodic plate. The dimension of the three compartments is 10 × 2.5 × 0.2 cm3. The electrode area is 25 cm2 and the electrode-to-membrane distance is 2 mm.
The device was fabricated from acrylic plastic and contained a gas compartment for flowing CO2, the anode and cathode channels for flowing electrolyte, the membrane for separating the anode and cathode, and solid end pieces. Poly(tetrafluoroethylene) (PTFE) gaskets were placed between each component for sealing and the device was tightened using six bolts. The gas flow rate into the flow cell was set via a mass flow controller (Alicat Scientific), and the CO2 flow rate was set at 15 sccm if no otherwise described. The catholyte flow rates were controlled via a peristaltic pump (YZ1515x, Baoding Chuangrui Precision Pump CO., LTD), with the catholyte flow rate ranging from 0.1 to 1 mL min−1 depending on the current densities (lower flow rates were used at lower current densities to allow for sufficient accumulation of liquid products). To prepare the above electrolyte, a calculated amount of salt (KOH, KCl, and KHCO3) and H2SO4 was weighed and dissolved in 500 mL of ultra-pure water, which was contained in a sealed Polypropylene bottle. The corresponding solution pH was measured using a benchtop pH Meter. The pH values for 20 mM H2SO4 in 1 M KCl, 1 M KCl, 1 M KHCO3 and 1 M KOH are 1.42 ± 0.03, 6.26 ± 0.07, 8.06 ± 0.06 and 13.66 ± 0.13, respectively. The electrolytes are prepared when testing and stored under the normal temperature and pressure of 25 °C and 1 atm. An anion exchange membrane (FAA-3, Fumatech, Length×Width×Thickness: 4 cm × 3 cm × 0.1 mm) was used in 1 M KOH, 1 M KCl and 1 M KHCO3 electrolyte, and a proton exchange membrane (Nafion, Fumatech, Length×Width×Thickness: 4 cm× 3 cm×0.1 mm) was used in 20 mM H2SO4 and 1 M KCl electrolyte. All membranes are immersed in the relevant electrolyte for 24 hours prior to use. The anolyte flow rate was 2 mL min−1. The backpressure of the gas in the flow cell was tuned to atmospheric pressure as controlled by a backpressure controller (Alicat Scientific). Each reported data point reflected the mean of at least two or three measurements, where the error bars were the standard deviation.
Electrochemical measurements
Electrochemical measurements were conducted on an Autolab potentiostat (PG128N) in a three-electrode system. All potential measurements were converted to the reversible hydrogen electrode (RHE) based on the following formula: ERHE = EAg/AgCl + EθAg/AgCl + 0.0591 × pH (in volts), with 100% iR compensation unless otherwise stated. The current interrupt (i-Interrupt) measurement was conducted on the Autolab Electrochemical Workstation to determine the uncompensated resistance. During a current interrupt measurement, a constant potential is applied on the cell before the current interrupt circuit triggered. This circuit interrupts the current flow in the cell and measures the potential decay. From the measured potential decay, the uncompensated resistance value is determined, using a linear regression.
The Ag/AgCl reference was calibrated using a reversible hydrogen electrode (RHE) under controlled conditions. The calibration was performed in a high-purity H2-saturated electrolyte (0.5 M H2SO4, pH = 0) with a Pt wire as the working electrode, a graphite rod as the counter electrode, and the Ag/AgCl electrode as the reference. CVs were run at a scan speed of 1 mV s−1. The average value of the two potentials at which the current equals 0 was recognized as the thermodynamic potential for the hydrogen evolution reaction (E = 0 V vs. RHE). The measured value for RHE is −0.197 V vs. Ag/AgCl, therefore the calibrated potential for E (vs. Ag/AgCl) is 0.197 V.
Gas products were quantified on a Multigas #5 gas chromatography (GC, SRI Instruments) equipped with a HayeSep D and Molsieve 5 A columns connected to a thermal conductivity detector (TCD) and a HayeSep D column connected to a flame ionization detector (FID).
Liquid products were quantified using a Bruker AVIII 500 MHz NMR spectrometer. Typically, 500 μL of collected catholyte exiting the reactor was mixed with 100 μL D2O containing 50 ppm (m/m) dimethyl sulphoxide ( ≥ 99.9%; Alfa) as the internal standard. The one-dimensional 1HNMR spectrum was measured with water suppression using a pre-saturation method.
Faradaic efficiency (FE) measurements
Based on the definition of Faradaic efficiency: FE = QCO /Qtotal = (Z × n × F)/Qtotal, where Z is the number of electrons transferred (Z = 2 for CO and H2 production), n is the number of moles for a given product, F is the Faraday’s constant (96 485 C mol-1), and Q is the total charge passed throughout the electrolysis process (measured by calculating the curve area of current density vs. time plot). CO and H2 mole fractions of injected samples were calculated based on the GC calibration curve.
The single-pass CO2 conversion efficiency (SPCE) is calculated as follows:
where n is the number of transferred electrons of the product, F is the Faraday constant, R is the gas constant, T is the reaction temperature, P is the reaction pressure, and N is the number of C in products.
COMSOL Multiphysics simulations
COMSOL Multiphysics finite element model was employed to investigate the characteristics of the interfacial liquid and gas fluid field65,66,67,71 This model mainly focuses on studying the dynamic characteristics of bubbles generated during the transportation of mixed gases (CO2, CO and H2) under the application of electric field force. The phase field method was used to track the gas/liquid interface. The electric field force source term was added to the N-S equation as the volume force by the Maxwell equation. The flow characteristics of the gas film, pressure and other physical features were obtained through simulation calculations of the two-phase flow process during the eCO2RR process. The corresponding control equation is as follows:
Electric field equations
Bubbles exhibit behavioral characteristics such as movement, deformation, oscillation, coalescence, fragmentation, and splitting under the electric field. Therefore, it is crucial to accurately describe the electric field force’s effect on fluid movement and morphological changes. In electrohydrodynamics, due to the small dynamic current, the magnetic induction effect can be neglected, and the electric field intensity is non-rotational (\(\nabla\)×E = 0). The relationship between volume charge density and electric field intensity is established based on the differential form of Gauss’s law, where D represents the electric flux density, \({\rho }_{v}\) represents the volume free charge density of the fluid, ε0 represents the vacuum permittivity (8.85e−12 F/m), \({\varepsilon }_{0}\) represents the relative permittivity, and the electric field intensity E is equal to the negative gradient of the electric potential.
For two-phase flow, the relative permittivity of the fluid needs to be distinguished through a phase function to obtain the relative permittivity of fluid 1, fluid 2, and at the fluid-phase interface. The formula is as follows. The subscripts 1 and 2 represent the parameters of fluid 1 (CO2, CO and H2) and fluid 2 (liquid), respectively.
Based on the calculated charge density and electric field intensity distribution, the electric field force of the liquid near the interface can be determined by solving the divergence of the Maxwell stress tensor (\({T}^{M}\)). By adding the electric field force as a source term of volume force into the N-S equation, the fluid is subject to force to produce motion and deformation. The motion of the fluid and the change of the phase interface will cause the redistribution of charges, and the redistribution of charges will in turn change the electric field force, further affecting the motion process of the fluid. Therefore, the electric field and fluid field are a bidirectional coupling process.
Flow control equations
The mass conservation equation (continuity equation) reflects the relationship between fluid movement and fluid mass distribution:
Where \(\rho\) is the fluid density, \(t\) is the flow time and \({u}\) is the flow velocity.
The momentum conservation equation (N-S equation), which reflects the basic mechanical law of viscous fluid flow, indicates that the increment of fluid momentum in the control body per unit time is equal to the sum of the external forces acting on it per unit time.
where g is the gravitational acceleration. \(F\) is the volumetric force, \({F}_{{st}}\) represents the surface tension term, and \({Vf}\) is the volume fraction.
Phase field method interface tracking
In the phase field, the value of the phase variable \(\phi\) is between (-1, 1), \(\phi\) = -1 represents fluid 1 (CO2, CO and H2), \(\phi\) = 1 represents fluid 2 (liquid), and -1 <\(\phi\) < 1 represents the gas/liquid interface.
\(\psi\) is the phase field auxiliary variable, \(\lambda\) is the mixture energy, \(\gamma\) is the migration rate, \(\sigma\) is the surface tension coefficient, and \(\chi\) is the migration adjustment parameter of 1 m ∙ s/kg. The interface thickness parameter (\({\varepsilon }_{{pf}}\)) defaults to half the maximum grid size, so a reasonable interface thickness parameter can be obtained by refining the grid.
Surface tension calculation
A continuous surface tension (CSF) model was proposed to determine the surface tension. The surface tension jump conditions were transformed into equivalent singular volume forces and then added to the N-S equation. The gradient of the phase fraction (\(\phi\)) was used to calculate the interface normal vector (\({{{{\bf{n}}}}}_{{{{\bf{s}}}}}\)). At present, the chemical potential (G) and the interface normal vector are widely used in the phase field method to calculate the surface tension and are added to the N-S equation as the volume force source term.
DFT calculations
The ab initio quantum mechanical (QM) calculations were performed using the Vienna Ab initio Simulation Package (VASP) version 5.4.4 with the projector augmented wave (PAW) method and a plane wave basis set. The method used was DFT with generalized gradient approximations (GGA) of the Perdew-Burke-Ernzerhof (PBE) functional72. A dispersion correction, the DFT-D3 method with Becke–Jonson damping73, was included in the calculations. The energy cutoff was set to 400 eV. A larger energy cutoff does not produce more accurate predictions based on our benchmark calculations. The reciprocal space was sampled by the Γ-centered Monkhorst-Pack scheme with a 3×3×1 grid. A finer K spacing does not produce a more accurate prediction basin in our benchmark calculation. The partial occupancies for each orbital are set with the first-order Methfessel-Paxton scheme at a smearing width of 0.2 eV. The dipole moment corrections for the total energy are considered in the direction normal to the surface. A self-consistent electronic step is considered to converge when the change in the total energy and the change in the eigenvalues between the two steps are both smaller than 1 e−6 eV. A conjugate-gradient algorithm is used to relax the ions in energy minimization. The minimization was considered converged when all the atomic forces were smaller than 0.01 eV/Å, where convergence to an energy minimum was confirmed in all cases with frequency calculations to verify that all imaginary frequencies were eliminated. The frequencies and normal modes are determined from the Hessian matrix. To calculate the Hessian matrix, finite differences are used. Each ion is displaced ± 0.04 Å in the direction of each Cartesian coordinate, and from the forces, the Hessian matrix is determined.
A p (4×4) supercell including 3 atomic layers was used, and the vacuum thickness was set to 15 Å. In our calculations, the bottom atomic layer of Ag(111) was fixed at its original atomic position in the bulk structure, while all remaining layers together with the adsorbates were fully relaxed during structural optimization. Ag(111)-v was constructed by simply removing one of the surface Ag atoms within other settings, similar to Ag(111). The temperature is 298 K, and the pressure is 1 bar. The CO2/H2O ratio is estimated based on the CO2 solubility in the 20 mM H2SO4 solution and the measured humidity data. The free energies for all the structures were computed using standard statistical mechanics formulas that account for translational, rotational, vibrational, and electronic degrees of freedom69,70. Translational and rotational contributions to the free energy were omitted for all surface-adsorbed species. The partition function under the harmonic oscillator approximation is as follows:
where k is a force constant and ν is the vibrational frequency. After substituting the partition function qvib, the internal energy correction is as follows:
The first term is the contribution of zero-point energy (ZPE), and the second term is the contribution of internal energy correction from 0 K to 298 K. The correction of entropy (S) is as follows:
Here, the first term exactly cancels with the second term of the internal energy shown above. Therefore, only the second term of the entropy needs to be corrected. For adsorbed species, the six degrees of freedom of translation and rotation are frustrated and considered vibrations. This approximation fails when the vibration is low, which significantly contributes to the correction. To avoid such overestimation, the contributions of frequencies below 50 cm−1 are all considered to be 50 cm−1.
The zero-point energy (ZPE) in thermoenergy correction is as follows:
The Gibbs free energy G can be derived as follows:
The above correction is obtained by using the VASPKIT code for postprocessing of the VASP calculated data74.
Data availability
All data supporting the findings of this study are available in the article and its Supplementary Information. Source data are provided with this paper.
Code availability
All codes supporting the findings of this study are available from the corresponding author upon request.
References
Tan, X. et al. Recent advances in innovative strategies for the CO2 electroreduction reaction. Energy Environ. Sci. 14, 765–780 (2021).
Yao, Z. et al. Electrochemical Carbon Dioxide Reduction in Acidic Media. Energy Rev. 7, 8 (2024).
Yan, Z. et al. Improving the efficiency of CO2 electrolysis by using a bipolar membrane with a weak-acid cation exchange layer. Nat. Chem. 13, 33–40 (2021).
Wang, K. et al. Atomic-level insight of sulfidation-engineered Aurivillius-related Bi2O2SiO3 nanosheets enabling visible light low-concentration CO2 conversion. Carbon Energy 5, e264 (2023).
Chen, J. et al. Selective and stable CO2 electroreduction at high rates via control of local H2O/CO2 ratio. Nat. Commun. 15, 5893 (2024).
Monteiro, M. C. O. et al. Efficiency and selectivity of CO2 reduction to CO on gold gas diffusion electrodes in acidic media. Nat. Commun. 12, 4943 (2021).
Liu, X. & Koper, M. T. M. Tuning the interfacial reaction environment for CO2 electroreduction to CO in mildly acidic media. J. Am. Chem. Soc. 146, 5242–5251 (2024).
Guan, M. et al. Engineering of oxygen vacancy and bismuth cluster assisted ultrathin Bi12O17Cl2 nanosheets with efficient and selective photoreduction of CO2 to CO. Carbon Energy 6, e420 (2024).
Shin, H. et al. Techno-economic assessment of low-temperature carbon dioxide electrolysis. Nat. Sustain 4, 911–919 (2021).
Hong, J. et al. Ag/C composite catalysts derived from spray pyrolysis for efficient electrochemical CO2 reduction. Chem. Eng. J. 431, 133384 (2022).
Hiragond, C. B. et al. Surface-modified Ag@Ru-P25 for photocatalytic CO2 conversion with high selectivity over CH4 formation at the solid–gas interface. Carbon Energy 6, e386 (2024).
Li, S. et al. Hierarchical micro/nanostructured silver hollow fiber boosts electroreduction of carbon dioxide. Nat. Commun. 13, 3080 (2022).
Abdinejad, M. et al. CO2 Electrolysis via Surface-Engineering Electrografted Pyridines on Silver Catalysts. ACS Catal. 12, 7862–7876 (2022).
Huang, J. E. et al. CO2 electrolysis to multicarbon products in strong acid. Science 372, 1074–1078 (2021).
Ringe, S. et al. Double layer charging driven carbon dioxide adsorption limits the rate of electrochemical carbon dioxide reduction on Gold. Nat. Commun. 11, 33 (2020).
Bondue, C. J. et al. Suppression of hydrogen evolution in acidic electrolytes by electrochemical CO2 reduction. J. Am. Chem. Soc. 143, 279–285 (2021).
Chen, Z. et al. Reconstruction of pH-universal atomic FeNC catalysts towards oxygen reduction reaction. J. Colloid Interface Sci. 582, 1033–1040 (2021).
Qiao, J., Lin, R., Li, B., Ma, J. & Liu, J. Kinetics and electrocatalytic activity of nanostructured Ir–V/C for oxygen reduction reaction. Electrochim. Acta 55, 8490–8497 (2010).
Li, B., Qiao, J., Yang, D., Wang, H. & Ma, J. New Ir-V-W Electro-Catalyst Exceeding Pt for the Anode of Fuel Cells. ECS Trans. 33, 343 (2010).
Qiao, Y. et al. Engineering the local microenvironment over Bi nanosheets for highly selective electrocatalytic conversion of CO2 to HCOOH in strong acid. ACS Catal. 12, 2357–2364 (2022).
Burdyny, T. et al. CO2 reduction on gas-diffusion electrodes and why catalytic performance must be assessed at commercially-relevant conditions. Energy Environ. Sci. 12, 1442–1453 (2019).
Kim, C. et al. Tailored catalyst microenvironments for CO2 electroreduction to multicarbon products on copper using bilayer ionomer coatings. Nat. Energy 6, 1026–1034 (2021).
Li, M. et al. The role of electrode wettability in electrochemical reduction of carbon dioxide. J. Mater. Chem. A 9, 19369–19409 (2021).
Pan, B. et al. Close to 90% single-pass conversion efficiency for CO2 electroreduction in an acid-fed membrane electrode assembly. ACS Energy Lett. 7, 4224–4231 (2022).
Buckley, A. K. et al. Approaching 100% Selectivity at Low Potential on Ag for Electrochemical CO2 Reduction to CO Using a Surface Additive. ACS Catal. 11, 9034–9042 (2021).
Kim, C. et al. Insight into Electrochemical CO2 Reduction on Surface-Molecule-Mediated Ag Nanoparticles. ACS Catal. 7, 779–785 (2017).
Koshy, D. M. et al. Chemical Modifications of Ag Catalyst Surfaces with Imidazolium Ionomers Modulate H2 Evolution Rates during Electrochemical CO2 Reduction. J. Am. Chem. Soc. 143, 14712–14725 (2021).
Wang, R. et al. Maximizing Ag Utilization in High-Rate CO2 Electrochemical Reduction with a Coordination Polymer-Mediated Gas Diffusion Electrode. ACS Energy Lett. 4, 2024–2031 (2019).
Li, X. et al. Recent advances in metal-based electrocatalysts with hetero-interfaces for CO2 reduction reaction. Chem. Catal. 2, 262–291 (2022).
Dinh, C.-T. et al. CO2 electroreduction to ethylene via hydroxide-mediated copper catalysis at an abrupt interface. Science 360, 783–787 (2018).
Yang, H. et al. Carbon dioxide electroreduction on single-atom nickel decorated carbon membranes with industry compatible current densities. Nat. Commun. 11, 593 (2020).
Shen, J. et al. Electrocatalytic reduction of carbon dioxide to carbon monoxide and methane at an immobilized cobalt protoporphyrin. Nat. Commun. 6, 8177 (2015).
Xu, Z. et al. Steering the selectivity of electrochemical CO2 reduction in acidic media. ChemCatChem 14, e202200052 (2022).
Pu, Y. et al. Surface coating combined with in situ cyclic voltammetry to enhance the stability of gas diffusion electrodes for electrochemical CO2 reduction. Sci. Total Environ. 918, 170758 (2024).
Wang, C. et al. Creation of controllable high-density defects in silver nanowires for enhanced catalytic property. Nano Lett. 16, 5669–5674 (2016).
Nakamura, I. et al. Role of oxygen vacancy in the plasma-treated TiO2 photocatalyst with visible light activity for NO removal. J. Mol. Catal. A: Chem. 161, 205–212 (2000).
Li, Z. et al. A silver catalyst activated by stacking faults for the hydrogen evolution reaction. Nat. Catal. 2, 1107–1114 (2019).
Kang, W.-J. et al. Engineering Dense Stacking Faults in Silver Nanoparticles for Boosting the Oxygen Reduction Reaction. ACS Energy Lett. 8, 3512–3519 (2023).
Ding, J. et al. Facile decoration of carbon fibers with Ag nanoparticles for adsorption and photocatalytic reduction of CO2. Appl Catal. B: Environ. 202, 314–325 (2017).
Lv, J. J. et al. A Highly Porous Copper Electrocatalyst for Carbon Dioxide Reduction. Adv. Mater. 30, e1803111 (2018).
Xu, T. et al. Fullerene micro/nanostructures: controlled synthesis and energy applications. Mater. Today Nano 11, 100081 (2020).
Tsang, A. C. H. et al. Ink-printed metal/graphene aerogel for glucose electro-oxidation. Battery Energy 1, 20220004 (2022).
Yuan, R. et al. Graphene nanotube array assists all-wood supercapacitors to access high energy density and stability. Battery Energy 2, 20220055 (2023).
Xie, Y. et al. High carbon utilization in CO2 reduction to multi-carbon products in acidic media. Nat. Catal. 5, 564–570 (2022).
Monteiro, M. C. O. et al. Absence of CO2 electroreduction on copper, gold and silver electrodes without metal cations in solution. Nat. Catal. 4, 654–662 (2021).
Li, J.-J. et al. K+ enhanced electrocatalytic CO2 reduction to multicarbon products in strong acid. Rare Met. 41, 723–725 (2021).
Fan, Q. et al. Iron nanoparticles tuned to catalyze CO2 electroreduction in acidic solutions through chemical microenvironment engineering. ACS Catal. 12, 7517–7523 (2022).
Liu, Z. et al. Acidic electrocatalytic CO2 reduction using space-confined nanoreactors. ACS Appl Mater. Interfaces 14, 7900–7908 (2022).
Sheng, X. et al. Engineering the Ni-N-C catalyst microenvironment enabling CO2 electroreduction with nearly 100% CO selectivity in acid. Adv. Mater. 34, e2201295 (2022).
Gu, J. et al. Modulating electric field distribution by alkali cations for CO2 electroreduction in strongly acidic medium. Nat. Catal. 5, 268–276 (2022).
Fujinuma, N. et al. Highly efficient electrochemical CO2 reduction reaction to CO with one-pot synthesized co-pyridine-derived catalyst incorporated in a nafion-based membrane electrode assembly. Adv. Energy Mater. 10, 2001645 (2020).
Moreno-García, P. et al. Toward CO2 electroreduction under controlled mass flow conditions: A combined inverted RDE and gas chromatography approach. Anal. Chem. 92, 4301–4308 (2020).
Liu, L.-Y. et al. Boosting oxygen-resistant CO2 electroreduction reaction in acidic media over conjugated frameworks. J. Mater. Chem. A 12, 9486–9493 (2024).
Zhang, Y. et al. In situ formed copper nanoparticles via strong electronic interaction with organic skeleton for pH-universal electrocatalytic CO2 reduction. J. Mater. Chem. A 11, 22992–23000 (2023).
Wang, Z. et al. Acidic electrochemical reduction of CO2 using nickel nitride on multiwalled carbon nanotube as selective catalyst. ACS Sustain Chem. Eng. 7, 6106–6112 (2019).
Zhang, J. et al. Accelerating electrochemical CO2 reduction to multi-carbon products via asymmetric intermediate binding at confined nanointerfaces. Nat. Commun. 14, 1298 (2023).
Li, W. et al. Bifunctional ionomers for efficient co-electrolysis of CO2 and pure water towards ethylene production at industrial-scale current densities. Nat. Energy 7, 835–843 (2022).
Zou, X. et al. Strategies for efficient CO2 electroreduction in acidic conditions. Chin. J. Catal. 52, 14–31 (2023).
Masel, R. I. et al. An industrial perspective on catalysts for low-temperature CO2 electrolysis. Nat. Nanotechnol. 16, 118–128 (2021).
Xu, T. et al. Monitoring of anodic corrosion on carbon-based gas diffusion layer in a flow cell. Carbon 205, 207–213 (2023).
Yuan, S. et al. Bubble management in PEM water electrolysis via imprinting patterned grooves on catalyst layer. Int J. Heat. Mass Transf. 212, 124249 (2023).
Yuan, S. et al. Bubble evolution and transport in PEM water electrolysis: Mechanism, impact, and management. Prog. Energy Combust. Sci. 96, 101075 (2023).
Wei, J. et al. High-performance carbon-capture membranes developed by (non)solvent-induced nanostructural rearrangement in Nafion. J. Mater. Chem. A 11, 18146–18157 (2023).
Kim, Y. et al. Permeability Matters When Reducing CO2 in an Electrochemical Flow Cell. ACS Energy Lett. 7, 2382–2387 (2022).
Wen, G. et al. Continuous CO2 electrolysis using a CO2 exsolution-induced flow cell. Nat. Energy 7, 978–988 (2022).
Zhou, L. et al. Two-dimensional Cu plates with steady fluid fields for high-rate nitrate electroreduction to ammonia and efficient Zn-nitrate batteries. Angew. Chem. Int Ed. 63, e202401924 (2024).
Liu, W. et al. Electrochemical CO2 reduction to ethylene by ultrathin CuO nanoplate arrays. Nat. Commun. 13, 1877 (2022).
Zhou, L. et al. Synergistic regulation of hydrophobicity and basicity for copper hydroxide-derived copper to promote the CO2 electroreduction reaction. Carbon Energy 5, e328 (2023).
Chen, L. D. et al. Electric field effects in electrochemical CO2 reduction. ACS Catal. 6, 7133–7139 (2016).
Lejaeghere, K. et al. Reproducibility in density functional theory calculations of solids. Science 351, aad3000 (2016).
Schwartz, N. et al. Effects of electrode support structure on electrode microstructure, transport properties, and gas diffusion within the gas diffusion layer. ACS Omega 7, 29832–29839 (2022).
Grimme, S. et al. Effect of the damping function in dispersion corrected density functional theory. J. Comput Chem. 32, 1456–1465 (2011).
Perdew, J. P. et al. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Wang, V. et al. A user-friendly interface facilitating high-throughput computing and analysis using VASP code. Comput Phys. Commun. 267, 108033 (2021).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (52201227, 52272088, 52331009, 22103054, 22173066, 52401244), Chinese Education Ministry’s Chunhui program (202200767), Zhejiang Provincial Natural Science Foundation of China (LQ23B030001, Q24B020025), the open research fund of Songshan Lake Materials Laboratory (2023SLABFN09), and the Natural Science Foundation of Jiangsu Province (BK20230065). This work was partly supported by Suzhou Key Laboratory of Functional Nano & Soft Materials, the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), the Collaborative Innovation Center of Suzhou Nano Science & Technology, the 111 Project and Joint International Research Laboratory of Carbon-Based Functional Materials and Devices. The authors thank Tao Cheng (Soochow University) for assistance in DFT calculation and Enyu Guo (Dalian University of Technology) for assistance in COMSOL Multiphysics simulations.
Author information
Authors and Affiliations
Contributions
T.X. fabricated and characterized the catalysts. H.Y. did the DFT calculation. T.L., R.Z., S.Z. and M.Z. performed the morphology characterization and analysis; R.Z. and Y.Y. performed the optical microscope; Z.J.W. and H.J. helped to conduct the supplementary experiments; J.J.L., X.W., T.C., and S.W. supervised and directed this project; T.X. and J.J.L. wrote the original manuscript. J.L., S.P., J.W. revised the manuscript. All authors commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Zhuo Xing, Lei Wang, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xu, T., Yang, H., Lu, T. et al. Microenvironment engineering by targeted delivery of Ag nanoparticles for boosting electrocatalytic CO2 reduction reaction. Nat Commun 16, 977 (2025). https://doi.org/10.1038/s41467-025-56039-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-56039-x
This article is cited by
-
Silver-doped TiO2 catalysts for efficient methylene blue removal
Journal of Materials Science: Materials in Engineering (2026)
-
Cobalt-Based Electrocatalysts for Sustainable Nitrate Conversion: Structural Design and Mechanistic Advancements
Nano-Micro Letters (2026)