Abstract
Developing acid-stable and active ruthenium dioxide (RuO2) catalysts for the oxygen evolution reaction (OER) is crucial for facilitating the large-scale applications of proton exchange membrane water electrolysis (PEMWE) for hydrogen production. Here, we propose a strain heterogeneity engineering strategy to simultaneously enhance the OER stability and activity of RuO2 electrocatalysts by introducing single-atom platinum (Pt). In a PEM water electrolyzer, the resultant Pt-RuO2 catalyst archives 3 A cm−2 at a low voltage of 1.791 V and maintains a stable performance for over 500 h at 500 mA cm−2. These performance metrics highlight its potential for practical applications. Experiments and calculations analyses confirm that the bulk tensile strain effectively stabilizes the entire structure of electrocatalysts, while the regions of compressive strain are identified as highly active catalytic sites, where the weakened binding energy of oxo-intermediates improves the catalytic activity.
Similar content being viewed by others
Introduction
Water electrolysis powered by renewable electricity supplies is a promising technology for sustainable hydrogen (H2) production1,2,3. Proton exchange membrane water electrolysis (PEMWE) currently stands out as the technology capable of effectively harnessing fluctuating renewable energy sources like wind or solar to generate H2, owing to its instantaneous current response4,5,6. Electrocatalysts are the key components of PEMWE, determining the energy efficiency and durability of the electrolyzers7,8,9,10. Iridium oxide (IrO2) is commonly used as a commercial OER electrocatalyst in PEMWE systems due to its superior stability under acidic conditions11, but its high cost significantly limits the widespread application of PEMWE. Ru-based electrocatalysts, particularly ruthenium dioxide (RuO2), are a promising alternative owing to their higher intrinsic acidic OER activity and relatively lower costs. Nevertheless, their application is hindered by poor stability under harsh acidic and oxidative conditions, primarily due to Ru dissolution as RuO4 species and the release of lattice oxygen during the OER, limiting their application in PEMWE12,13,14,15,16.
Tensile strain engineering has emerged as an effective strategy to stabilize Ru-based catalysts by weakening Ru–O bond covalency, thereby mitigating Ru dissolution and lattice oxygen release.17,18,19,20. However, tensile strain often compromises catalytic activity. Positioned on the left of the acidic OER volcano plot21, Ru-based catalysts bind OER intermediates too strongly, limiting their activity22,23. According to d-band theory, for later transition metals such as Ru with a more than half-filled d-band, tensile strain typically shifts the d-band center upward, strengthening intermediate adsorption and further reducing OER activity24,25. This necessitates a higher voltage to achieve the required current density, accelerating the leaching of Ru species. Therefore, it is imperative to propose a strategy that enhances the stability of RuO2 without compromising its activity.
Heteroatom substitution engineering, which exploits differences in atom radii, serves as a primary method for inducing strain. However, conventional descriptions of strain are generally limited to the volume average of materials, neglecting local heterogeneity in strain within the electrocatalyst26. In the case of Gorilla glass, the substitution of the larger potassium ion (K+) for the smaller sodium ion (Na+) via ion exchange resulted in the creation of a local compressive stress layer on the surface, which improved the glass’s resistance to damage27. Inspired by this, we hypothesize that strain engineering induced by heteroatom doping may lead to strain heterogeneity within catalysts. The implications of such strain heterogeneity on the activity and stability of OER catalysts are not yet understood and warrant further investigation.
Herein, we investigated an unconventional heterogeneous strained structure for the rational design of high-performance electrocatalysts. Unlike traditional alloys or epitaxially grown heterostructures that typically exhibit either compressive or tensile strain exclusively on the catalyst surface, we have introduced a concept of a heterogeneously strained structure featuring bulk tensile and localized compressive strains. Specifically, introducing a larger Pt atom leads to lattice expansion of the RuO2 crystal, simultaneously pushing away the surrounding smaller Ru atoms outward, creating compressive strain in the Pt-doped region. Experiments and density functional theory (DFT) studies confirmed that the bulk tensile strain can enhance the thermodynamic stability of lattice oxygen and stabilize the entire structure of electrocatalysts during OER progress, while the Ru site with compressive strain became more active due to the reduced binding energies of oxo-intermediate, thus identifying the regions of compressive strain as highly active catalytic area. Additionally, the ligand effect of Pt atoms suppressed the participation of lattice oxygen and stabilized Ru sites within the Pt-doped region. The Pt-doped RuO2 catalyst (Pt-RuO2) demonstrated stable operation for over 1500 h at 10 mA cm−2, providing a significant improvement compared to the undoped counterparts (less than 200 h) and the most recently reported acidic OER catalysts. Impressively, when utilized in a PEM electrolyzer, the Pt-RuO2 catalyst achieved a long-term water electrolysis performance of over 500 h at 500 mA cm−2 and exhibited high activity with a low full cell voltage of 1.791 V at 3 A cm−2, surpassing US Department of Energy (DOE) performance target for 2025 (1.9 V at 3 A cm−2). When applying this strategy to other large-radius atoms (Re, Cu, Au, and Ag) doping, the heterogeneous strained structure can be observed within Re- and Cu-RuO2 electrocatalysts, which also displayed enhanced OER performance compared with the RuO2 electrode. This work introduces a universal strategy of strain heterogeneity for the development of advanced Ru-based electrocatalysts for PEMWE with both long-term stability and high activity.
Results
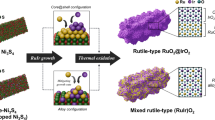
Concept of the strain heterogeneity in RuO2
Unlike conventional descriptions that generally focus on the bulk average strain in materials, we investigate a heterogeneous strain structure in RuO2 induced by heteroatom subrogation, simultaneously enhancing the activity and stability of RuO2 electrocatalyst. The ion radii of Pt (Pt2+ 0.86 Å; Pt4+ 0.63 Å) is larger than that of Ru (Ru4+ 0.62 Å). Introducing Pt atoms expands the RuO2 crystal volume but simultaneously exerts a repulsive force on the surrounding smaller Ru atoms, creating compressive stress in the Pt-doped region (Fig. 1)28. Additionally, Pt doping enables electron transfer from Ru to Pt, as the higher electronegativity of Pt (2.28) compared to Ru (2.2) according to the Pauling scale, which increases the oxidation state of Ru while reducing that of Pt. The resulting smaller size of high-valence Ru cations and larger size of reduced Pt cations further compress Ru-O bonds.
The calculated dissociation energy of lattice oxygen in RuO2 crystal cell as a function of applied strain (left). Compressive stress and the corresponding d-band center shifts in the doped region (right). EF, Fermi level, ɛd, d-band center.
DFT calculation is adopted to investigate the role of strain on stabilizing RuO2 and improving its catalytic properties. First, we constructed RuO2 with different strain levels (−5%, −3%, −1%, 0%, +1%, +3%, and +5%) by modifying the lattice parameters (a, b, c). Our calculations of the enthalpy change during the lattice oxygen loss process (Supplementary Note 1 and Table 1) show that a 1% tensile strain in the RuO2 structure results in the highest enthalpy change of 88.06 KJ mol–1. This finding indicates that bulk tensile strain in the RuO2 crystal enhances the thermodynamic stability of lattice oxygen during the OER, ensuring the overall stability of the oxygen framework (Fig. 1, left). In the compressive strained regions, the downshift of Ru d-band center weakens the adsorption of oxo-intermediates, optimizing the adsorption energy of reaction intermediates on Ru sites. Therefore, compressively strained regions are identified as highly active catalytic area (Fig. 1, right). Although compressive strain results in the thermodynamic instability of lattice oxygen, the ligand effect of Pt atoms improves the kinetic stability of lattice oxygen, that can suppress the participation of lattice oxygen and stabilize Ru sites within Pt-doped region, as evidenced by subsequent calculations and experiments.
Material synthesis and structure characterizations
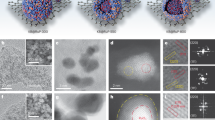
To verify the rationality of constructing a heterogeneous strained structure, DFT calculations were conducted to investigate the geometry modification and strain distribution caused by the Pt substitution (Fig. 2a). The RuO2 unit cell model was used to simulate the bulk structure of electrocatalysts. The lattice paraments are expanded after introducing Pt ions, indicating the bulk tensile strain in the Pt-RuO2 crystal (Fig. 2a, left). Given that the (110) surface is the dominant exposed surface for RuO229, Pt-RuO2 (110) was used as the model to simulate the strain distribution in the local doped region. The color-coded strain distribution for Pt-RuO2 (110) (Fig. 2a, right and Supplementary Note 2) shows compressive strain within Pt-doped region. The DFT results suggest that the incorporation of Pt single atoms into RuO2 crystal induces the heterogeneous strain.
a Theoretical analyses of the RuO2 and Pt-RuO2. Crystalline structure models and corresponding lattice paraments of RuO2 and Pt-RuO2 (left). Atomistic structure of the surface of Pt-RuO2 (110) and the corresponding color-coded strain distribution (right). The Pt atom is given as the red dot. b TEM image, c HAADF STEM image, and d colored HAADF STEM image with a temperature scale of Pt-RuO2. e The line profiles for atomic imaging intensity analysis labeled with a red box in (d). arb. units, arbitrary units. f XRD patterns of RuO2 and Pt-RuO2. Inset, magnified XRD patterns of RuO2 and Pt-RuO2 for (110) peaks. g Colored HAADF STEM image with a temperature scale of Pt-RuO2. h The line profiles for atomic imaging intensity analysis labeled with red and blue boxes in (g). i Lattice strain (εxx) measured from GPA of HRTEM images for RuO2 (up) and Pt-RuO2 (down).
In line with our rational design, the Pt-RuO2 catalyst was experimentally synthesized through a liquid cation exchange strategy guided by the Hard-Soft-Acid-Base (HSAB) principle (Supplementary Note 3 and Supplementary Fig. 1). The atomic ratio of Pt is 0.94 at%, while the surface atomic ratio of Pt was calculated as 19.57 at% (Supplementary Tables 2-3), indicating a mainly distribution of Pt over the surface of RuO230. Microscopy analysis was utilized to characterize the morphology and composition distributions of catalysts. Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) images exhibited that the average size of Pt-RuO2 nanoparticles ranged between 15 and 19 nm, and the well-defined lattice fringes of 0.316 nm can be ascribed to the (110) planes of RuO2 (Fig. 2b and Supplementary Fig. 5). Then, the atomic status and distributions of Pt were probed by aberration-corrected high-angle annular dark-field scanning TEM (HAADF-STEM) (Fig. 2c and Supplementary Fig. 7). Because the atomic number of elements is proportional to the intensity of STEM signal31, the brighter dots marked by red circles in Fig. 2c can be recognized as heavier Pt species, mainly distributed on the surface of Pt-RuO2 in the form of single atoms or few atom clusters. The HAADF-STEM images, colored with a temperature scale (Fig. 2d) and corresponding intensity profile (Fig. 2e), further confirmed that partial Ru atoms were replaced by Pt atoms, facilitating the formation of multiple atomic-scale Ru-O-Pt local motifs.
To probe the changes in geometric structure after incorporating atomic Pt into RuO2, we used powder X-ray diffraction (XRD), HADDF-STEM, and geometric phase analysis (GPA) to investigate the crystal and local structure. As shown in Fig. 2f, the diffraction peaks of RuO2 and Pt-RuO2 can be indexed to rutile RuO2 (JCPDS #880286), the absence of Pt diffraction peaks indicated well-dispersed Pt atoms. Additionally, the dominant (110) peak at ~27° in Pt-RuO2 shifted to lower angle side compared to that of RuO2, with the shift intensifying as Pt concentration increased (Supplementary Fig. 8). Combined with XRD Rietveld refinement results (Supplementary Fig. 9 and Table 4), the introduction of larger Pt atoms could induce bulk tensile strain. HAADF signal analysis provided insights into the local structure of Pt-doped region in catalysts (Fig. 2g). Intensity profiles of HAADF STEM images (Fig. 2h) exhibited that the interatomic distance of Ru-Ru surrounded by Pt atoms was compressed to 1.66 Å, compared to arrays of Ru atom with less Pt atoms (1.75 Å) and homemade RuO2 (1.79 Å), which matched with DFT simulations (Supplementary Fig. 10). The compressive distance of Ru-Ru can be attributed that larger Pt atoms pushed away the surrounding smaller Ru atoms, validated by the much longer interatomic distance of Pt-Ru (1.85 Å). Figure 2i illustrated the strain distributions calculated from the corresponding HRTEM images. Unlike relatively flat GPA mapping of RuO2, the intercalation of larger Pt atoms induced varying degrees of local compressive strain in the Pt-RuO2 crystal. We furtherly conducted an in-depth statistical analysis of heterogenous strain induced by different Pt doping contents (Supplementary Note 4). Both theoretical and experimental results consistently showed that bulk tensile and local compressive strains increased with Pt single atom concentration. However, when Pt concentrations exceed a certain threshold, the compressive strain starts to reduce due to stress relaxation mechanisms. Specifically, higher Pt concentrations distribute the local stress more evenly, thereby diminishing the compression effect of individual Pt atoms on surrounding Ru atoms. (Supplementary Figs. 11–13). Similar to Pt-RuO2 catalysts, RuO2 doped with Re and Cu can be fabricated through a cation exchange strategy (Supplementary Note 3 and Table 7). Heterogenous strained structures in Re- and Cu-RuO2 electrocatalysts are evidenced by both theoretical and experimental results, indicating that such strain is a universal characteristic (Supplementary Figs. 14–18).
The electronic state and local configurations of Pt-RuO2 catalyst were further probed by X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure spectroscopy (EXAFS) measurements (Supplementary Fig. 19). In the XANES spectra for Pt L3-edge (Fig. 3a), the white-line peak intensity of Pt-RuO2 is close to that of PtO2 and significantly higher than that of Pt foil, suggesting that the chemical state of Pt is close to +4, which distinguishes it from nanoparticles or supported single atoms32. The Pt L3-edge EXAFS spectra exhibited three characteristic peaks in R space (Fig. 3b). Based on the fitting results of Pt L3-edge EXAFS (Supplementary Table 8), the first peak was ascribed to the first-shell Pt-O bond, while the second and the third peaks can be attributed to Pt-Ru coordination with distinct bond distances. The presence of Pt-O-Ru structures in the Pt-RuO2 crystal confirmed that Pt species exist and are incorporated as single atoms into the Ru sites of RuO2. Wavelet transform (WT)-EXAFS analysis further verified the local atomic environment around Pt, revealing two distinct regions: the first shell domain corresponding to Pt-O scattering and the second shell domain for Pt-Ru scattering (Fig. 3c). The absence of Pt-Pt coordination indicated the atomic dispersion of Pt species in the RuO2 host.
a Pt L3-edge XANES spectra (inset: enlarged white line peak profiles) and b corresponding Fourier transform of EXAFS spectra (k weight = 3) and EXAFS fitting of Pt L3-edge of Pt-RuO2 catalyst. Inset, the structure of Pt-RuO2 (110) surface. c Pt L3-edge WT-EXAFS spectra of PtO2, Pt foil, and Pt-RuO2. d Ru K-edge XANES spectra (inset: enlarged pre-edge profiles) and e corresponding Fourier transform of EXAFS spectra (k weight = 3). R, bond length, FT, Fourier transform; k, wave vector.
In the XANES spectra at the Ru K-edge (Fig. 3d), the spectrum of Pt-RuO2 exhibited a shape and position similar to those of the homemade RuO2, a magnified XANES spectrum revealed Pt-RuO2 showing a slightly higher near-edge absorption energy compared to homemade RuO2 (inset of Fig. 3d), suggesting a relatively higher Ru oxidation state in Pt-RuO2, which is consistent with the findings from XPS analysis (Supplementary Fig. 20). The higher shift can be ascribed to the electron-withdrawing effect of Pt (IV), as the higher electronegativity of Pt (2.28) compared to Ru (2.2) according to the Pauling scale. The local configurations around Ru atoms were revealed by Ru K-edge EXAFS (Fig. 3e). The Ru-O bond intensity in Pt-RuO2 was slightly weaker than that in RuO2, suggesting an unsaturated coordination environment for Ru, possibly due to oxygen vacancies formation33. This formation was further evidenced by Raman spectroscopy16, electron paramagnetic resonance (EPR), and O 1 s XPS results (Supplementary Figs. 21–22, 34).
Electrochemical performances and bulk structural stability
The OER performance of the prepared Pt-RuO2 catalyst was evaluated in a standard threeelectrode configuration with 0.5 M H2SO4 electrolyte. As shown in Fig. 4a, the Pt-RuO2 catalyst exhibited an overpotential of 215 mV at the current density of 10 mA cm−2, much lower than that of undoped RuO2 (250 mV) and commercial RuO2 (270 mV). Pt-RuO2 also exhibited the lowest Tafel slope of 63.89 mV dec−1(Fig. 4b), indicating enhanced kinetics for OER34. The EIS data were fitted using the equivalent circuit model, which showed a decrease in the charge transfer resistance (Rct) value for Pt-RuO2, confirming that the charge transfer process is more efficient compared to RuO2 (Supplementary Fig. 24). Electrochemical surface area (ECSA) of catalysts is proportional to their double-layer capacitance (Cdl) calculated from cyclic voltammetry (CV) method with different scan rates. Pt-RuO2 exhibited a higher Cdl, corresponding to a larger ECSA, suggesting that it can expose more active sites for the OER (Supplementary Fig. 25).
a LSV curves and b Tafel plots for Pt-RuO2, RuO2, and commercial RuO2 in 0.5 M H2SO4. c Chronopotentiometry stability at the current density of 10 mA cm−2 for Pt-RuO2, RuO2 coated on CP. Inset, chronopotentiometry stability at the current density of 10 mA cm−2 for Pt-RuO2, RuO2 coated on GCE. d LSV curves of Pt-RuO2 and RuO2 and their spent samples after 10,000th cycling test. All the electrochemical performance were measured without iR-drop compensation (R was 3.8 ± 0.05 Ω). e Time-dependent Ru ion dissolution of Pt-RuO2 and RuO2 during OER.
Stability is a crucial factor in evaluating the practical application potential of the catalysts35. First, a chronopotentiometry test at a constant current density of 10 mA cm−2 was carried out to test the electrochemical stability of Pt-RuO2. As shown in Fig. 4c, the overpotential of Pt-RuO2 coated on carbon paper (CP) with a loading of 2.5 mg cm−2 only exhibited an increase of 40 mV during a 1500 h test for OER. The degradation rate in activity was approximately 27 μV h−1, a magnitude slower than that of the undoped RuO2 catalyst (750 μV h−1). The stability of the as-prepared Pt-RuO2 catalyst outperformed most reported electrocatalysts for acidic OER (Supplementary Fig. 27 and Table 10). The overpotential at 10 mA cm−2 was increased by only 12 mV after a 10,000-cycle CV test (Fig. 4d), much smaller than that of RuO2 (38 mV). The above results demonstrated the superior OER stability in the acidic electrolyte after Pt doping.
In-situ Raman spectroscopy revealed that the Ru-O bonding structure in Pt-RuO2 remained stable during OER, whereas RuO2 exhibited a positive peak shift of ~10 cm−1, indicating significant damage to the Ru-O bonding (Supplementary Fig. 28)36. We further characterized the structure and composition of catalysts after a 5,000-cycle CV test. TEM images after the OER test showed that there was no aggregation observed in the morphology and lattice spacing of Pt-RuO2 nanoparticles (Supplementary Fig. 29). XPS results revealed the valence and peak intensity of Ru in the Pt-RuO2 catalyst remained almost unchanged after OER process (Supplementary Fig. 30a)15. Whereas the positive shift and alleviated peak intensities of Ru 3p XPS for RuO2 catalyst after OER suggested the formation of the Ru>4+ species and severe leaching of Ru species from the surface. We utilized ICP-OES to monitor the cation dissolution into the electrolyte during electrolysis37. As shown in Fig. 4e and Supplementary Fig. 31, only slight dissolution of Ru and Pt ions was observed during the electrolysis for Pt-RuO2 catalysts. In contrast, Ru ions leached from the RuO2 catalyst rapidly at the beginning of electrolysis, resulting in a mass loss of exceeding 50 μg of Ru ions in the electrolyte after 24 h. These results evidenced that the Pt incorporation can effectively stabilize the overall structure of RuO2 electrocatalysts during OER progress, as supported by previous DFT calculations demonstrating enhanced stability of lattice oxygen due to the expanded crystal lattice.
Origin of the enhanced stability and activity
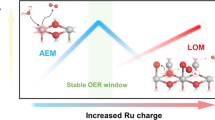
Currently, the prevailing OER mechanisms under acidic OER conditions primarily involve the adsorbate evolution mechanism (AEM) and the lattice oxygen-mediated mechanism (LOM)4 (Supplementary Fig. 32). To experimentally verify the OER pathway of Pt-RuO2, we conducted operando differential electrochemical mass spectroscopy (DEMS) measurements assisted by 18O isotope labeling to directly distinguish AEM and LOM routes (Fig. 5a). The catalyst surface was labeled with 18O isotopes, and we detected the evolved O2 in H216O/ 0.5 M H2SO4 electrolyte during OER. The evolved 32O2 reflected the AEM pathway, while the 34O2 signal reflected the LOM pathway15. As shown in Fig. 5b, the ratio of 34O2 / 32O2 detected on Pt-RuO2 is much lower than that on RuO2, indicating the LOM pathway is suppressed by approximately 50% on Pt-RuO2. The DEMS results of Pt-RuO2 in H218O/ 0.5 M H2SO4 electrolyte also verified that predominant occurrence of AEM mechanism over Pt-RuO2 electrocatalysts (Supplementary Fig. 33). Additionally, the ratio of consumed lattice oxygen measured by XPS spectra of fresh and post-reaction catalysts was reduced in Pt-RuO2 compared to RuO2, reflecting that less lattice oxygen was involved during OER for Pt-RuO2 (Fig. 5c, Supplementary Fig. 34 and Table 9)38.
a Schematic illustration of the operando DEMS. PTFE, polytetrafluoroethylene. b DEMS signals of 36O2 (18O18O), 34O2 (16O in the electrolyte + 18O in the lattice) and 32O2 (16O in the electrolyte + 16O in the electrolyte) from the reaction products for 18O-labelled Pt-RuO2 and RuO2 catalysts in H216O/0.5 M H2SO4 electrolyte. c Lattice O contents calculated from O 1 s XPS spectra of fresh and post-reaction of Pt-RuO2 and RuO2. d, e In-situ ATR-SEIRAS spectra of Pt-RuO2 (d) and RuO2 (e). f Potential dependence of peak intensity of characteristic vibration adsorption of surface-adsorbed *OOH. g Computed free energy profiles of OER via AEM pathway on surfaces of RuO2 (110) and Pt-RuO2 (110) under an electrode potential of 0 and 1.23 V, based on implicit solvation model. h, i Density of states of O 2p (h) and Ru 4 d (i) of RuO2 (110) and Pt-RuO2 (110). j The calculated demetallation energies of surface Ru of RuO2 (110) and Pt-RuO2 (110). k Schematic diagram of enhancing the OER activity and stability over the doped region of Pt-RuO2. RDS, rate-determining step.
We conducted in situ attenuated total reflectance surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS) to detect the intermediates during OER. For Pt-RuO2, a distinct peak located at ~1231 cm−1 can be attributed to the O–O stretching of surface *OOH species, a typical intermediate for AEM pathway16 (Fig. 5d). The peak intensity increased with the applied potential, indicating a AEM pathway for Pt-RuO2 catalyst. Additionally, the formation of the *OOH intermediate is regarded as the rate-determining step (RDS), and the higher intensity of *OOH species for Pt-RuO2 indicated the higher OER activity. In contrast, RuO2 exhibited a weak peak at the location of *OOH species ( ~ 1229 cm−1) (Fig. 5e). Besides, the peak intensity remains nearly unincreased from 1.2 V to 1.65 V, which demonstrated the sluggish formation of *OOH species for RuO2 catalyst (Fig. 5f).
The model was constructed that can well simulate the active area with local compressive strain in Pt-RuO2 catalyst (Supplementary Figs. 35–37, Supplementary Data 1, 2), to deepen insight of the enhanced OER performance on Pt-RuO2 utilizing DFT. The OER barriers were calculated for RuO2 and Pt-RuO2, with particular emphasis on two Ru sites: one located near the Pt atom (designated as Ru1) and another situated farther from Pt (designated as Ru2) (Supplementary Fig. 38a). The optimized structures revealed that Ru1 is under compressive strain, while Ru2 is under tensile strain. DFT results showed that the energy barrier at Ru1 is lower than that at Ru2 (Supplementary Fig. 38b) and Ru site in RuO2 (Fig. 5g), indicating that the Ru site with compressive strain near Pt dopant serves as the primary active site for the reaction. The OER pathway was calculated using an implicit solvation model to account for solvent effects and simulate a more realistic reaction environment (Supplementary Note 5)39,40,41,42. It was observed that an –OO specie on the Ru coordinatively unsaturated site (CUS) is stabilized by a neighboring –OH group on the Ru fully coordinated bridge site (BRI), forming an -OO---H intermediate. This indicates that the -OO---H intermediate is thermodynamically more favorable than the conventional −OOH intermediate (Supplementary Fig. 39). Theoretical calculations were conducted to evaluate the possibility of Pt sites acting as active centers by binding with additional oxygen atoms under oxidative reaction conditions. The results revealed a significantly higher energy barrier for the RDS in the OER for this configuration (Supplementary Fig. 41), rendering it thermodynamically unfavorable.
Electronic structure analysis was performed to pinpoint the physical origin of the correlation between strain and OER performance of catalysts. As shown in Fig. 5h, the O 2p band (εp) in Pt-RuO2 downshifted slightly, moving further away from the Fermi level. This shift resulted in a reduction in the redox activity of lattice oxygen during the electrochemical process, thus suppressing the LOM pathway, consistent with DEMS results38. The d-band center of Ru 4 d (εd) was also downshifted from −1.579 eV in RuO2 to −1.651 eV in Pt-RuO2 (Fig. 5i), mitigating the strong interaction between the Ru active site and oxo-intermediate, accelerating the RDS of the *O to *OOH conversion43,44. We evaluated the enthalpy changes for Ru atom demetallation as a descriptor for electrocatalyst stability. The energy cost for Ru demetallation increased (Fig. 5j), indicating more stable Ru sites in Pt-RuO2 than in RuO2. Consequently, the enhanced OER stability and activity for the active area can be attributed to the compressive Pt-O-Ru local structure. The combination of compressive strain and ligand structure resulted in a suppressed LOM pathway, appropriate adsorption strength of oxo-intermediate, and stable Ru sites (Fig. 5k).
To further demonstrate the impact of strain heterogeneity on OER performance, we systematically investigated the correlation between theoretical quantified strain and experimentally determined catalytic activity and stability. The OER performance of Pt-RuO2 catalysts with varying Pt contents (e.g., 0.48, 0.94 and 3.38 at%, designated as Pt1-RuO2, Pt2-RuO2, Pt3-RuO2) and Re-, Cu-doped RuO2 catalysts were systematically evaluated (Supplementary Figs. 42, 43). As shown in Supplementary Figs. 44, 45, the catalytic activity and stability are intimately linked to the degree of strain heterogeneity in RuO2, regardless of the varying Pt concentration or doping element. This intrinsic correlation arises from the modulation of the electronic structure induced by strain. The εd and εp values of surface Ru and O atoms exhibit a consistent trend corresponding to the level of compressive stress caused by varying single-atom Pt concentrations or other larger dopants (Supplementary Figs. 46, 47). The introduction of larger atoms generates localized compressive strain, which significantly influences the electronic structure and thereby enhances the catalytic performance of RuO2. Notably, Pt3-RuO2 exhibits the highest εp, indicating more lattice oxygen participation and instability. However, Pt3-RuO2 shows superior stability compared to Pt1-RuO2 and undoped RuO2 (Supplementary Fig. 42). This enhanced stability can be attributed to increased bulk tensile strain, which contributes to stabilizing the catalyst structure, as evidenced by significantly reduced Ru leaching during OER (Supplementary Figs. 48). Overall, these findings highlight the dual role of strain heterogeneity in concurrently enhancing catalytic activity and improving stability.
Demonstration in acidic overall water splitting
To evaluate the feasibility of Pt-RuO2 for larger-scale hydrogen production in industrial operating systems, we assembled a PEMWE device employing Pt-RuO2 (2 mg/cm−2) as anode and 40% Pt/C (1 mg/cm−2) as cathode catalysts, sprayed on the both sides of a Nafion 212 membrane, as schematically illustrated in Fig. 6a. At 80 °C, the Pt-RuO2 PEM electrolyzer achieved an industrially required hydrogen production current density of 1, 2 and 3 A cm−2 at a cell voltage of 1.567, 1.673 and 1.791 V, respectively, that surpassing the relevant DOE performance 2025 target (3 A cm−2@1.9 V) (Fig. 6b). Upon applying a constant current density of 500 mA cm−2, there was no discernible increase in cell voltage even after 500 h of electrolysis (Fig. 6c), which is in stark contrast to the commercial RuO2 that deactivated within a mere 70 h at a lower current density of 200 mA cm–2 (Supplementary Fig. 49) and recently reported Ru-based electrocatalysts (Supplementary Table 11), indicating the great practical applicability potential of Pt-RuO2.
a Schematic diagram of the PEMWE device. b Polarization curve and c chronopotentiometry curve of the PEMWE device using Pt-RuO2 catalyst as anodic catalyst and commercial Pt/C as cathodic catalyst, obtained at 80 °C with Nafion 212 membrane, no cell voltages were iR compensated. Inset of (c) is the photograph of the PEMWE device.
Discussion
In summary, we investigated a concept of strain heterogeneity on the activity and stability of RuO2 as efficient OER catalysts. DFT simulations and characterization measurements evidenced that Pt with larger radii was atomically doped into the surface layers of RuO2 to replace partial Ru sites, resulting in the expansion of the RuO2 lattice, but the local compressive strain was observed in the Pt-doped region. The compressively strained region is recognized as active area, where the combined effect of compressive strain and Pt ligand result in an appropriate adsorption strength of oxo-intermediate, effectively suppressed LOM pathway and stabilized Ru sites, thus improving the activity and stability of Pt-RuO2 catalyst. Additionally, the incorporation of Pt induces a bulk tensile strain structure that enhances the structural stability, avoiding the structural collapse of catalysts. Moreover, this work demonstrates a tenfold increase in the long-term durability of RuO2 catalysts, highlighting the significant contribution of strain engineering to their enhanced performance. This work enriched the strain engineering applied in the design of electrocatalysts with high performance and demonstrated the practical implementation of Ir-free Ru-based catalysts in PEMWE devices for efficient and clean H2 generation.
Methods
Chemicals and materials
Ruthenium (III) chloride hydrate (RuCl3·xH2O, 35.0 ~ 42.0% Ru basis) was purchased from Heowns Biochem Technologies, LLC, Tianjin. Chloroplatinic acid hexahydrate (H2PtCl6, Pt≥ 37.5%), H218O (99%) was purchased from Macklin. Commercial ruthenium (IV) oxide (RuO2, 99.9% metals basis) were purchased from Sigma-Aldrich. Commercial Pt/C (40 wt% Pt) catalyst was purchased from Johnson Matthey Company. The TGP-H-060 carbon paper (CP) was purchased from Toray. 5 wt% Nafion ionomer and Nafion 212 membrane was purchased from DuPont Co.
Materials synthesis
RuO2: For the preparation of RuO2, 51.9 mg of RuCl3 was dissolved into 20 mL of deionized (DI) water with ultrasonic treatment for 10 min. Then, the RuCl3 solution was transferred into a 50 mL Teflon-lined autoclave and heated at 120 °C for 10 h to generate Ru-Cl hydrate. After natural cooling to room temperature, the product was washed three times alternately with water and ethanol, followed by collection via centrifugation. After vacuum drying for 10 h at 60 °C, the dried Ru-Cl hydrate was annealed in an air atmosphere under 400°C for 2 h to generate RuO2.
Pt-RuO2: Ru-Cl hydrate was obtained through hydrothermal methods and then immersed in a 10 mL aqueous solution of H2PtCl6 for 2 h at 60 °C to facilitate an ion exchange reaction. After natural cooling to room temperature, the resulting product was washed three times with water and ethanol alternately, and collected by centrifugation. It was subsequently dried under vacuum at 60 °C for 10 h. The dried Pt-doped Ru-Cl hydrate was annealed in an air atmosphere under 400 °C to generate Pt-RuO2. Catalysts with varying Pt ratios were synthesized using the same procedure, with adjustments to the H2PtCl6 solution concentrations: 0.7 mg mL−1 for Pt1-RuO2 (0.48 at% Pt-RuO2), 1.3 mg mL−1 for Pt2-RuO2 (0.94 at% Pt-RuO2), and 6.5 mg mL−1 for Pt3-RuO2 (3.38 at% Pt-RuO2). The Pt content in the final products was determined using ICP-OES.
The preparation processes of M-RuO2 (M= Au, Ag, Cu, Re) catalysts are similar to that of Pt2-RuO2, without the variation of metals agent, those are HAuCl4, AgNO3, CuCl2, and ReCl3.
Characterization
The crystal structures of the as-synthesized samples were characterized using a powder X-ray diffraction (PXRD, Rigaku D/Max-2500) with a Cu Kα radiation (λ = 1.5408 Å) source in the 2θ range of 5-85°. The morphology and crystalline-amorphous phase were investigated by SEM on JEOL JSM-7500F and TEM by Talos F200X G2 AEMC instruments. HAADF-STEM imaging was executed utilizing a JEM-ARM200F AEMC instrument operated at an accelerating voltage of 200 kV. The ICP-OES was carried out on Agilent 7800. The element valence was studied by XPS on Thermo Scientific K-Alpha+PHI5000. EPR was measured on CIQTEK EPR200-Plus. The X-ray absorption find structure spectra (Pt L3-edge and Ru K-edge) were collected at 1W1B station in Beijing Synchrotron Radiation Facility (BSRF).
Electrochemical measurements under a three-electrode system
All the electrochemical measurements were carried out on Chenhua CHI-660E electrochemical workstation. A mercurous sulfate electrode (Hg/Hg2SO4) was used as reference electrodes in 0.5 M H2SO4 for both OER and HER tests, with a calibration value of 0.694 V (Supplementary Fig. 23), and graphite rod was used as counter electrode. The electrolyte (0.5 M H2SO4) was freshly prepared and promptly utilized (pH is 0.3 ± 0.1), which was prepared by adding 28.05 mL of sulfuric acid (95%) into a container with 500 mL of deionized water, followed by adjusting the volume to 1000 mL in a volumetric flask. To prepare the working electrode, 5 mg of the catalysts was dispersed in 260 μL of a solution containing 150 μL of ethanol, 100 μL of deionized water, and 10 μL of 5% Nafion solution, followed by ultrasonication for 30 min. Then 9 μL catalyst ink was dropped on a GCE (Φ = 5 mm) and dried in air. The catalyst loading (mloading) on the working electrode was 0.88 mg cm−2, that was calculated according to the following equation:
where mcatalyst is the mass of catalyst dropped on the electrode, which is equal to 0.173 mg (9 μL × 5 mg / 260 μL). Selectrode is the geometric area of GCE, which is equal to 0.196 cm2 (0.252 cm2 × 3.14).
For electrochemical measurements on CP, the catalysts were sprayed onto CP using an airbrush. First, 15 mg of catalyst was dispersed in 2 mL of isopropanol, and then 40 μL of 5% Nafion solution was added. After 30 min of sonication, the homogeneous ink was sprayed onto 2 cm × 2 cm CP heated to 90 °C. The CPs were weighed before and after spraying to ensure a total catalyst loading of approximately 2.5 mg cm−2.
All the potentials were calibrated with respect to the reversible hydrogen electrode (RHE) potentials without iR-compensated according to the Nernst equation:
When testing LSV polarization curves, three independent curves were measured with a scan rate of 5 mV s−1 to ensure the reproducibility of electrochemical performance. The LSV polarization curves were obtained without iR-correction.
For Cdl measurements, CV cycling was performed at various scan rates (ν: 20, 40, 60, 80, 100, 120, and 150 mV s−1) within the non-faradaic region (0.25–0.35 V vs. Hg/Hg2SO4). The Cdl was determined by the following equation:
where Δj represents the difference in anodic and cathodic current densities at the midpoint potential (Δj = janodic - jcathodic). The slope of Δj-ν curve is regarded as Cdl. The ECSA of the catalyst was determined by the equation of ECSA = Cdl/Cs, where Cs is 0.035 mF cm−2. EIS measurements were collected with frequencies ranging from 100 KHz to 0.1 Hz. EIS tests were performed at 1.45 V (versus RHE) from 0.1 Hz to 100 kHz, and the results are presented in the form of a Nyquist plot. The OER stability was evaluated on CP by chronopotentiometry at 10 mA cm−2 and on GCE by CV accelerated aging tests between 1.35 and 1.55 V vs. RHE.
DEMS tests
In situ DEMS experiments were performed to verify the OER pathway using a QAS 100 device (Linglu Instruments, Shanghai). A flexible carbon cloth (CC) was used as the current collector for the classical electrolysis cell required for DEMS tests. The 1 cm × 1 cm CC was ultrasonically cleaned in dilute hydrochloric acid and acetone, thoroughly rinsed with ultrapure water, and air-dried. Next, 8 mg of catalysts were first dispersed in 1 mL of isopropanol, and then 20 μL of 5% Nafion solution was added. After 30 min of sonication, the homogeneous ink was sprayed onto the prepared CC, and heated to 90 °C. The classical DEMS electrochemical cell setup is illustrated in Fig. 5a. The CC electrode loaded with catalyst was placed directly on a PTFE waterproof breathable membrane and connected to the electrochemical workstation using conductive copper tape. A plastic spacer with a hole was positioned on the CC, and both sides of the spacer were coated with vacuum grease to prevent gas leakage. 2 mL of 0.5 M H2SO4 was prepared using H218O as the solvent electrolyte, and the counter electrode (Pt foil) and reference electrode (Hg/Hg2SO4) were inserted. The oxygen generated from the OER was directly pumped into the mass spectrometer chamber through the PTFE membrane under a high vacuum for detection. This setup ensures high collection efficiency, rapid response time, and effective real-time monitoring of the mass spectrometry signals of evolved oxygen.
First, the catalysts were performed four LSV cycles (1.2 – 1.7 V vs. RHE at 5 mV s−1) in 18O-labelled 0.5 M H2SO4, and the mass signals of the gaseous products 32O2, 34O2 and 36O2 were recorded (Supplementary Fig. 33). The CV was performed in the range of 1.2–1.6 V at 50 mV s−1 to further labelled the catalyst surface with 18O isotopes. The catalysts were washed with DI water to remove H218O physically adsorbed on the surface. Then, the 18O-labelled catalysts were operated in H216O/ 0.5 M H2SO4 electrolyte. Again, the gaseous products of different molecular weights were recorded. The experimental set-up of DEMS is shown in Supplementary Fig. 50.
In situ Raman spectroscopy
The in-situ Raman tests were performed to clarify the dynamic variation in RuO2 structure during OER. Raman spectra were acquired using a Renishaw inVia Raman spectrometer equipped with a 50× objective and a 50 mW 532 nm laser. The spectra were collected with an exposure time of 10 s and averaged over 10 exposures. A homemade electrochemical cell with a reference electrode (AgCl/Ag) and a counter electrode (Pt wire) was used in the in-situ measurements. The working electrodes were prepared by spraying the catalyst ink onto CP, conducted in 0.5 M H2SO4. The applied potential was increased from OCP to 1.65 V vs. RHE. The experimental set-up of in situ Raman is shown in Supplementary Fig. 51.
In situ ATR-SEIRAS measurement
In-situ ATR-SEIRAS tests were performed to detect the reaction intermediates during OER to verify the reaction mechanism. A Bruker INVENIO S FT-IR spectrometer, equipped with an MCT detector cooled with liquid nitrogen, was used for the measurements. The homemade three-electrode electrochemical cell was employed, with a saturated AgCl/Ag as the reference electrode and a Pt wire as the counter electrode. Catalysts sprayed on the CP served as working electrode, directly placed on a fixed-angle Au-coated Si prism (60°). The Au thin layer was deposited through electrochemical plating. ATR-SEIRAS measurements consisted of 48 scans, each with a spectral resolution of 16 cm–1. The background spectrum of the working electrode was obtained under open-circuit conditions. The spectra were processed using OPUS software. The experimental set-up of in situ ATR-SEIRAS is shown in Supplementary Fig. 52.
ICP-OES analysis of the ion dissolution
Ion dissolution was measured by analysis of the concentration of ions dissolved in electrolyte via ICP-OES, to investigate the composition stability of catalysts. The test measurements were the same as the OER stability tests on CP by chronopotentiometry at 10 mA cm−2, only the volume of electrolyte is reduced to 10 mL, low volume of electrolyte is beneficial to fulfill the ICP-OES sensitivities. 4 mL of electrolyte was sampled after operating for 0, 0.5, 2, 6, 12, and 24 h, followed by replenishment to 10 mL with pristine electrolyte for the subsequent sampling period.
PEMWE tests
The membrane electrode assembly (MEA) was prepared via a catalyst-coated membrane (CCM) method. Pt-RuO2 and commercial 40% Pt/C were used as cathode and anode catalysts, respectively. The anode ink consisted of 16 mg Pt-RuO2, 1 mL isopropanol, 1 mL DI water, and 148 μL of 5% Nafion. The cathode ink comprised 8 mg of 40% Pt/C, 0.5 mL isopropanol, 0.5 mL DI water, and 74 μL of 5% Nafion. After at least 1 h of ultrasonication, the inks were air-sprayed onto the two sides of the 2 cm × 2 cm Nafion 212 membrane (without treatment). The MEA was weighed before and after airbrushing to control the total catalyst loading to approximately 2 mg cm−2 for the anode and 1 mg cm−2 for the cathode. The prepared MEA was then immersed into DI water for several hours for further measurements.
The PEM electrolyzer was integrated by pressing the cathodic diffusion layer, MEA, porous transport layers (PTL), and two Ti end-plate with flow field together. A 0.3 mm thick titanium felt with 0.3 μm Pt-coating was used as the PTL in anode, and carbon paper was used as the diffusion layer for cathode. The active area was about 4 cm2. The PEM cell operated at 80 °C, with DI water supplied as the reactant through peristaltic pump circulation. A battery test system (CT-4008-5V100A-NTFA, NEWARE) was used to evaluate the performance of Pt-RuO2. The polarization curves were tested within a range (0–3500 mA cm−2), and the stability was evaluated by chronopotentiometry tests at the current densities of 500 mA cm−2. The polarization curves and stability tests in PEMWE were obtained without iR-correction.
Computational details
DFT calculations were conducted using the Vienna Ab-initio Simulation Package (VASP)45. The exchange-correlation function was described with the Perdew-Burke-Ernzerhof (PBE) functional under the generalized gradient approximation (GGA) framework, utilizing the projector augmented wave (PAW) method46. A 5 × 2 × 1 k-point grid and an energy cut-off of 520 eV were utilized in all computations. To prevent interactions between periodic images, a vacuum layer of at least 20 Å was introduced in the z-direction. The convergence criteria for the total energy was established at 1 × 10−5 eV per atom, with the residual Hellmann-Feynman force being 0.02 eV per Å. Spin-polarized calculations were employed for all systems. Solvation effects were accounted for using the VASPsol code with a dielectric constant of 78.4 to simulate the H2O solvent environment (PBE + SOL). In our study, we modeled the OER intermediates at a coverage of 1/8 monolayer on the RuO2(110) surface. Free energy corrections for intermediate species were carried out with VASPKIT. Gas-phase H2 and H2O molecules were optimized within a box having dimensions of 10 × 10 × 10 Å, employing Gamma point sampling for the Brillouin zone. The free energy of gas-phase O2 (G[O2]) was derived from G[O2] = 4.92 (eV) + 2 G[H2O] – 2 G[H2], by using the experimental Gibbs free energy of the reaction 2H2O(l) → O2(g) + 2H2(g) in standard conditions.
Data availability
The data that support the findings of this study are available within the article and its Supplementary Information files. All other relevant data supporting the findings of this study are available from the corresponding authors upon request. Source data are provided with this paper.
References
Kibsgaard, J. & Chorkendorff, I. Considerations for the scaling-up of water splitting catalysts. Nat. Energy 4, 430–433 (2019).
Chu, S. & Majumdar, A. Opportunities and challenges for a sustainable energy future. Nature 488, 294–303 (2012).
Miao, L., Jia, W., Cao, X. & Jiao, L. Computational chemistry for water-splitting electrocatalysis. Chem. Soc. Rev. 53, 2771–2807 (2024).
An, L. et al. Recent development of oxygen evolution electrocatalysts in acidic environment. Adv. Mater. 33, 2006328 (2021).
Wang, T., Cao, X. & Jiao, L. PEM water electrolysis for hydrogen production: Fundamentals, advances, and prospects. Carb. Neutrality 1, 21 (2022).
Liu, R.-T. et al. Recent advances in proton exchange membrane water electrolysis. Chem. Soc. Rev. 52, 5652–5683 (2023).
Gao, J., Tao, H. & Liu, B. Progress of nonprecious-metal-based electrocatalysts for oxygen evolution in acidic media. Adv. Mater. 33, 2003786 (2021).
Lin, Y., Dong, Y., Wang, X. & Chen, L. Electrocatalysts for oxygen evolution reaction in acidic media. Adv. Mater. 35, 2210565 (2023).
Chen, Z. et al. Advances in oxygen evolution electrocatalysts for proton exchange membrane water electrolyzers. Adv. Energy Mater. 12, 2103670 (2022).
Tobias, R., Hong Nhan, N., Detre, T., Robert, S. & Peter, S. Electrocatalytic oxygen evolution reaction in acidic environments – reaction mechanisms and catalysts. Adv. Energy Mater. 7, 1601275 (2017).
Geiger, S. et al. The stability number as a metric for electrocatalyst stability benchmarking. Nat. Catal. 1, 508–515 (2018).
Hainan, S. & WooChul, J. Recent advances in doped ruthenium oxides as high-efficiency electrocatalysts for the oxygen evolution reaction. J. Mater. Chem. A 9, 15506 (2021).
Wu, Z.-Y. et al. Non-iridium-based electrocatalyst for durable acidic oxygen evolution reaction in proton exchange membrane water electrolysis. Nat. Mater. 22, 100–108 (2023).
Lin, C. et al. In-situ reconstructed Ru atom array on α-MnO2 with enhanced performance for acidic water oxidation. Nat. Catal. 4, 1012–1023 (2021).
Yunzhou, W. et al. Stabilizing highly active Ru sites by suppressing lattice oxygen participation in acidic water oxidation. J. Am. Chem. Soc. 143, 6482–6490 (2021).
Wang, Y. et al. Unraveling oxygen vacancy site mechanism of Rh-doped RuO2 catalyst for long-lasting acidic water oxidation. Nat. Commun. 14, 1412 (2023).
Xu, Y. et al. Strain‐modulated Ru‐O covalency in Ru‐Sn oxide enabling efficient and stable water oxidation in acidic solution. Angew. Chem. Int. Ed. 63, e2023160 (2024).
Wang, J. et al. Exceptionally active and stable RuO2 with interstitial carbon for water oxidation in acid. Chem 8, 1673–1687 (2022).
Qin, Y. et al. RuO2 electronic structure and lattice strain dual engineering for enhanced acidic oxygen evolution reaction performance. Nat. Commun. 13, 3784 (2022).
Chaudhary, P., Zagalskaya, A., Over, H. & Alexandrov, V. Strain‐dependent activity‐stability relations in RuO2 and IrO2 oxygen evolution catalysts. ChemElectroChem 11, e202300659 (2023).
Thao, N. T. T. et al. Colossal dielectric perovskites of calcium copper titanate (CaCu3Ti4O12) with low-iridium dopants enables ultrahigh mass activity for the acidic oxygen evolution reaction. Adv. Sci. 10, 2207695 (2023).
Zhang, D. et al. Construction of Zn-doped RuO2 nanowires for efficient and stable water oxidation in acidic media. Nat. Commun. 14, 2517 (2023).
Man, I. C. et al. Universality in oxygen evolution electrocatalysis on oxide surfaces. ChemCatChem 3, 1159–1165 (2011).
Mavrikakis, M., Hammer, B. & Nørskov, J. K. Effect of strain on the reactivity of metal surfaces. Phys. Rev. Lett. 81, 2819–2822 (1998).
Hammer, B. & Nørskov, J. K. Electronic factors determining the reactivity of metal surfaces. Surf. Sci. 343, 211–220 (1995).
Orr, K. W. P. et al. Strain heterogeneity and extended defects in halide perovskite devices. ACS Energy Lett. 9, 3001–3011 (2024).
Qi, Y., Ban, C. & Harris, S. J. A new general paradigm for understanding and preventing Li metal penetration through solid electrolytes. Joule 4, 2599–2608 (2020).
Tang, T. et al. Unconventional bilateral compressive strained Ni–Ir interface synergistically accelerates alkaline hydrogen oxidation. J. Am. Chem. Soc. 145, 13805–13815 (2023).
Rossmeisl, J., Qu, Z. W., Zhu, H., Kroes, G. J. & Nørskov, J. K. Electrolysis of water on oxide surfaces. J. Electroanal. Chem. 607, 83–89 (2007).
Zhu, W. et al. Direct dioxygen radical coupling driven by octahedral ruthenium–oxygen–cobalt collaborative coordination for acidic oxygen evolution reaction. J. Am. Chem. Soc. 145, 17995–18006 (2023).
Lang, R. et al. Single-atom catalysts based on the metal–oxide interaction. Chem. Rev. 120, 11986–12043 (2020).
Zhu, Y. et al. Iridium single atoms incorporated in Co3O4 efficiently catalyze the oxygen evolution in acidic conditions. Nat. Commun. 13, 7754 (2022).
Wu, Z. et al. Recent progress of vacancy engineering for electrochemical energy conversion related applications. Adv. Funct. Mater. 31, 2009070 (2021).
Hao, S. et al. Dopants fixation of ruthenium for boosting acidic oxygen evolution stability and activity. Nat. Commun. 11, 5368 (2020).
Wang, Q. et al. Long-term stability challenges and opportunities in acidic oxygen evolution electrocatalysis. Angew. Chem. Int. Ed. 62, e202216645 (2023).
Fang, W.-C., Huang, J.-H., Chen, L.-C., Su, Y.-L. O. & Chen, K.-H. Effect of temperature annealing on capacitive and structural properties of hydrous ruthenium oxides. J. Power Sources 160, 1506–1510 (2006).
Hodnik, N. et al. New insights into corrosion of ruthenium and ruthenium oxide nanoparticles in acidic media. J. Phys. Chem. C. 119, 10140–10147 (2015).
Shi, Z. et al. Customized reaction route for ruthenium oxide towards stabilized water oxidation in high-performance PEM electrolyzers. Nat. Commun. 14, 843 (2023).
Rao, R. R. et al. Towards identifying the active sites on RuO2(110) in catalyzing oxygen evolution. Energy Environ. Sci. 10, 2626–2637 (2017).
Rao, R. R. et al. Operando identification of site-dependent water oxidation activity on ruthenium dioxide single-crystal surfaces. Nat. Catal. 3, 516–525 (2020).
Inico, E., Di Liberto, G. & Giordano, L. Stability and solvation of key intermediates of oxygen evolution on TiO2, RuO2, IrO2 (110) surfaces: A comparative DFT study. Chem. Cat. Chem. 16, e202400813 (2024).
Di Liberto, G., Pacchioni, G., Shao-Horn, Y. & Giordano, L. Role of water solvation on the key intermediates catalyzing oxygen evolution on RuO2. J. Phys. Chem. C. 127, 10127–10133 (2023).
Liu, H. et al. Eliminating over-oxidation of ruthenium oxides by niobium for highly stable electrocatalytic oxygen evolution in acidic media. Joule 7, 558–573 (2023).
Bai, X. et al. Low ruthenium content confined on boron carbon nitride as an efficient and stable electrocatalyst for acidic oxygen evolution reaction. Angew. Chem. Int. Ed. 62, e202308704 (2023).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Acknowledgements
This work was financially supported by the National Key R&D Program of China (No. 2021YFB4000200 (L.J.)), the National Natural Science Foundation of China (Nos. 52025013 (L.J.) and 22121005 (L.J.)), and the Fundamental Research Funds for the Central Universities. High-energy X-ray absorption experiment was conducted on the 1W1B beamline at the Beijing Synchrotron Radiation Facility, and we acknowledge L. Zheng for discussion and data analysis.
Author information
Authors and Affiliations
Contributions
X.C. and L.M. contributed equally to the paper. X.C., L.M., and L.J. conceived and designed this work. X.C. performed the experiments. L.M. carried out the calculations with assistance from H.Q. and G.L. R.M. carried out the PEMWE tests. X.C. and L.M. co-wrote the manuscript. W.J., T.J., and L.J. discussed the results and assisted with manuscript preparation. The project was supervised by L.J.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Michal Bajdich, Porun Liu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Cao, X., Miao, L., Jia, W. et al. Strain heterogeneity in RuO2 for efficient acidic oxygen evolution reaction in proton exchange membrane water electrolysis. Nat Commun 16, 6217 (2025). https://doi.org/10.1038/s41467-025-58570-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-58570-3
This article is cited by
-
Novel ternary metal layered double hydroxide NiFeWMo-supported carbon spheres as a bifunctional electrocatalyst for oxygen evolution reaction and methanol oxidation
Journal of Materials Science (2026)
-
Accelerating Electron Transfer via Ternary Heterostructures for Enhanced Acidic Oxygen Evolution Performance
Chemical Research in Chinese Universities (2026)