Abstract
Parathyroid hormone (PTH) analogs with improved actions in vivo could lead to optimized treatments for bone and mineral ion diseases. Rapid clearance from the circulation and short dwell times on the PTH receptor limit the efficacies of conventional PTH peptides currently in medical use. Here, we seek to enhance PTH peptide efficacy using two distinct peptide lipidation strategies. First, we append a lipid chain to the peptide’s C-terminus in a fashion to promote binding to serum albumin and hence prolong the peptide’s circulation half-life in vivo. Second, we append a lipid chain to a lysine side chain in a fashion designed to anchor the peptide to the cell membrane as the ligand is bound to the receptor and hence increase its dwell time on the receptor. We find that both strategies of lipidation can profoundly enhance the efficacy of PTH peptides in vitro and in mice. Our results could lead to the development of modified PTH analogs with optimized therapeutic utility.
Similar content being viewed by others
Introduction
The parathyroid hormone (PTH) receptor (PTH1R) plays essential roles in calcium and phosphate homeostasis via interaction with PTH, as well as in the development of bone and other tissues via interaction with PTH-related peptide (PTHrP)1. Recombinant PTH(1-34) (teriparatide) and a PTHrP(1-34) analog (abaloparatide) are FDA-approved for the treatment of osteoporosis2, and other longer-acting peptides, including a PTH(1-14)/PTHrP(15-36) hybrid peptide, called LA-PTH (eneboparatide)3, and a self-cleaving PTH(1-34)-based pegylated compound called TransCon PTH(1-34)(palopegteriparatide)4, are in clinical trials or approved for the treatment of hypoparathyroidism5. For most PTH peptide-based therapies developed so far, efficacy in vivo is limited by a relatively short half-life in the body, which typically is within minutes to an hour6, resulting in a need for frequent delivery, i.e. at least once-daily injection for hypoparathyroidism7. A further potentially limiting factor affecting efficacy is the duration of the desired biological effect achieved by a ligand on its target receptor, which in turn is determined at least in part by how long the ligand remains bound to the receptor. Receptor internalization and desensitization responses activated in target cells of bone and kidney also likely limit the efficacy that an injected PTH ligand achieves in vivo8,9.
A variety of strategies have been explored to overcome the factors that limit the biological effects achieved by a PTH1R ligand in vivo. Unmodified PTH agonist peptides such as PTH(1-34) bind to the PTH1R receptor with affinities in the nanomolar range, and produce signaling responses that are limited in duration, in part due to dissociation of the ligand-receptor complex10,11. Amino acid modifications have thus been introduced in the PTH peptide scaffold to increase binding affinity and hence prolong the dwell time of the ligand on the receptor and the resulting signaling responses in cells, which is the strategy employed in the development of LA-PTH (eneboparatide)3,12,13, currently in phase 3 clinical trials as a potential treatment for hypoparathyroidism14. The protracted pharmacodynamic actions of LA-PTH are not explained by pharmacokinetics, as peptides of this class clear from the circulation at rates comparable to that of PTH(1-34)15.
Another approach at extending PTH action in vivo involves the introduction of backbone modifications that confer resistance to degradation by proteases that are likely encountered by the ligand at the injection site and in blood. PTH- and PTHrP-based peptide analogs containing periodic replacements of native α-amino acids with homologous β-amino acids, which interfere with protease recognition and/or action, have thus been shown to exhibit full potency on the PTH1R and marked resistance to proteases in vitro, as well prolonged calcemic effects upon injection in mice16,17,18,19. Clearance via renal filtration is yet another mechanism that can limit peptide efficacy in vivo. Strategies to overcome this problem include pegylation, to push ligand size near or above the glomerular filtration limit. This approach has been shown to be effective for PTH(1-34) in mice20 and contributes to the extended therapeutic effects achieved by TransCon PTH in patients4,21.
Peptide lipidation can be a very effective approach for improving the functional properties that an exogenous peptide ligand exhibits in vivo. Lipid modification has been used most notably in the development of peptides in the glucagon, glucagon-like peptide 1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) family of hormones as treatments for diabetes and obesity22. A key feature of semaglutide, the long-acting GLP-1 analog in use as a once-weekly injection therapy for obesity, is thus an 18-carbon (C18) fatty diacid chain appended to the side chain of a lysine at position 20 in the 31-amino acid peptide chain23,24. Such lipid modifications alter the biophysical properties of the peptide ligand and can thus have pleiotropic effects on function via a variety of mechanisms. These include the stabilization of the ligand’s structure, increasing its resistance to protease attack, promoting its binding to serum carrier proteins to prolong circulation half-life, modulating its interaction with the cell membrane to increase its local concentration around the receptor, or directly altering the ligand’s action at the receptor, including the processes of binding, receptor activation, and the internalization, trafficking and desensitization responses23,25,26,27. Recently, Zorzi et al. reported a generalizable strategy for promoting ‘piggy-back’ binding of a target peptide to serum albumin, which has a serum half-life of approximately three weeks, to thereby profoundly extend the peptide’s circulation half-life and its pharmacodynamic actions28. The strategy involves linking a target peptide to a C-terminal hepta-residue tag sequence, EYEK(palm)EYE, comprised of an amino acid sequence that is optimized for high affinity yet reversible binding to albumin and is linked at its central lysine to a C16 palmitoyl chain, which further promotes albumin binding. Conjugating this acylated motif to the C-terminus of a Factor-XII-derived peptide thus extended the half-life of that peptide from ~13 minutes to ~7 h and similarly prolonged its anti-blood coagulating effects in rodents.
A requirement of any such peptide modification strategy is the preservation of affinity for the target receptor. PTH binds to the PTH1R, which is a prototypical class B GPCR, and thus utilizes a mechanism that involves docking of the peptide’s 15–34 region to the receptor’s N-terminal extracellular domain (ECD), and the insertion of the peptides N-terminal portion into the orthosteric pocket of the receptor’s hepta-helical transmembrane domain bundle (TMD). Two previous studies have explored lipidation for PTH peptides. Liu et al. thus shows that attachment of a dipalmitoly (2xC16) lipid group to the C-terminus of PTH(1-14) promotes binding to lipid micelles and improves cAMP signaling potency of the peptide in PTH1R-expressing HEK293 cells by ~100-fold relative to unmodified PTH(1-14)29. A more recent study by Ruan et al. shows that attaching a C18 diacid to Lys27 of PTH(1-34) promotes binding of the peptide to serum albumin in vitro and, upon single injection into mice, results in a 5-fold longer half-life in the blood and more prolonged increases in blood calcium levels, as compared to PTH(1-84)30. These results encourage the view that lipid modification can be an effective strategy for optimizing PTH ligands for actions in cells and in vivo.
In the current study, we explored the effects of lipid modifications on the actions in vitro and in vivo of a variety of PTH ligand analogs. We thus synthesized analogs of PTH(1-34), a commonly used and fully active PTH peptide, as well as minimized M-PTH(1-14)-derived peptides containing the previously reported C-terminal tag motif optimized for albumin-binding28, as well as other M-PTH(1-14) and M-PTH(1-11) peptides containing a lipid chain appended more directly to the side chain of a lysine at position 11 or 13 in a configuration designed for anchoring the ligand to the plasma membrane while the ligand is bound to the receptor’s orthosteric pocket. Our results demonstrate that such lipid appendages can indeed have profound impacts on the pharmacological actions that a PTH ligand exhibits in cells and in mice and can thus lead to robust changes in bone and calcium metabolism, even for peptides as small as the PTH(1-11) fragment that would otherwise be inactive in vivo.
Results
Palm-PTH(1-34): effects of the C-terminal albumin-binding motif on actions in cells and in vivo
We first assessed the effects of linking the palmitoylated (C16) heptapeptide tag sequence [EYEK(palm)EYE] reported by Zorzi et al. to be optimized for serum albumin binding28 to the C-terminus of PTH(1-34). A polyethylene glycol (PEG)2-like (20 atom) spacer was placed between the N-terminal Glu of the hexapeptide tag and Phe34 of PTH. The palmitoyl group was added post-synthetically to the central lysine of the tag (see Supplemental Methods for specifics on peptide chemistry). The resulting Palm-PTH(1-34) (Fig. 1A) was assessed for cAMP signaling potency in SGS-72 cells, which are human osteoblastic cells that endogenously express the PTH1R and stably express the glosensor cyclic adenosine monophosphate (cAMP) reporter. Palm-PTH(1-34) stimulated cAMP formation in these cells with a potency that was not significantly different from that of unmodified PTH(1-34) (pEC50s ~ 9.65 ± 0.1 and 9.60 ± 0.01, respectively, p (t-test) = 0.7, n = 10) and with a comparable maximum efficacy (Fig. 1B).
A Schematic of Palm-PTH(1-34). PTH(1-34) is C-terminally fused to a hepta-residue tag EYEK(palm)EYE optimized for albumin binding and containing a palmitoyl group (palm) linked to the central lysine side chain amine; a (PEG)2 (20 atom) spacer joins Phe34 of PTH to Glu1 of the tag. B cAMP responses in SGS-72 human osteoblast-derived cells expressing the glosensor cAMP reporter; data are calculated as the area-under-the-curve (AUC) of plots of luminescence vs. time (60’) normalized to the PTH(1-34) maximum and are means ± SEM of 10 independent experiments. C–F Pharmacodynamic and pharmacokinetic measurements of PTH(1-34) and Palm-PTH(1-34) in 10-week-old female CD1 mice; peptides injected at a dose of 10 nmol/kg (C–E) or 50 nmol/kg (F). Blood concentrations of ionized calcium (Ca++, C) and inorganic phosphorus (Pi, D) as a function of time after subcutaneously (SC) injection of peptide or vehicle; p < 0.05 (2-way ANOVA, followed by LSD test): *, peptide vs. vehicle; +, PTH(1-34) vs. Palm-PTH(1-34). E Peptide concentrations in blood plasma as a function of time after SC injection. The inset shows the data plotted with the y-axis in log10 scale. Data for x = 1–24 h were fit to a monophasic decay curve (dashed lines); the corresponding values of T1/2, AUC, Cmax (peak y), and Tmax (peak x) are shown. F Concentrations of tetramethylrhodamine (tmr)-labeled ligands in urine as a function time after SC injection of PTH(1-34)tmr or Palm-PTH(1-34)tmr; tmr fluorescence (lex = 531 ± 25 nm, lem = 595 ± 60 nm) at each time point was corrected for background fluorescence (t = 0) and net fluorescence was converted to peptide concentration using a standard curve formed with the corresponding tmr-peptide; p < 0.05 (two-sided t-test): *, PTH(1-34) vs. Palm-PTH(1-34). The inset shows the data plotted with the x-axis extended to 8 h; corresponding values of AUC (0-24 h) and Tmax (peak x) are also shown. In C-F, t = 0 represents pre-dose. Data are means ± SEM of values from six (C, D), three (E) or five (F) mice per group. Source data with exact p values are provided in the Source Data File. Corresponding changes in blood Ca++ and plasma peptide concentrations after IV injection are shown in Supplementary Fig. S-2.
Injection of PTH(1–34) into animals and humans is well documented to in an acute rise in blood ionized calcium (Ca++) levels and a concomitant decrease in blood inorganic phosphate (Pi)[5][6][15]. PTH increases directly or indirectly blood calcium by acting on target cells in bone and kidney while it decreases blood phosphate by reducing urinary Pi reabsorption. We hence assessed the pharmacodynamic (PD) actions of Palm-PTH(1-34) in vivo by subcutaneously (SC) injecting 10-week-old mice (strain CD-1) with either Palm-PTH(1-34) or PTH(1-34), each at a dose of 10 nmol/kg-body weight, or with vehicle and measuring effects on blood Ca++ and plasma Pi at times after injection. Both injected PTH peptides caused blood Ca++ levels to rise significantly above the pre-injection level by 1 h after injection, but while the response to PTH(1-34) was transient, that to Palm-PTH(1-34) was markedly more robust and prolonged. The response to PTH(1-34) thus peaked at 2 h and returned to baseline by 6 hours after injection, while that to Palm-PTH(1-34) attained a peak at 6 h that was significantly higher than that of PTH(1-34) and remained slightly higher than vehicle control levels even at the 24-h time point (Fig. 1C). Similarly, Palm-PTH(1-34) induced a more pronounced decrease in plasma Pi levels than did PTH(1-34) and the effect persisted for at least 6 h after injection (Fig. 1D).
A separate experiment was performed in intact mice to further assess the effects of a single injection of Palm-PTH(1-34) vs PTH(1-34) or vehicle, on markers of mineral ion homeostasis and bone metabolism. The peptides were injected SC, at a dose of 50 nmol/kg and at 2, 7 and 24 h after injection blood plasma as well as urine was collected for analysis. Consistent with the changes in blood ionized calcium and Pi observed in the experiment of Fig. 1 C and D, injection of Palm-PTH(1-34) resulted in significantly greater increases in plasma total calcium and significantly greater decreases in plasma Pi levels than did injection of PTH(1-34), as measured at 7 h post-injection (Supplemental Fig. S-1A,B). At 2 h post injection, plasma total calcium tended to be increased to similar levels with the two peptides, as compared to the levels in mice injected with vehicle, but the differences were not significant, and variations between replicate measurements were wide, as compared to those in the measurements of ionized calcium in whole blood shown in Fig. 1C, which presumably reflects technical differences in the assay formats for whole blood ionized calcium vs. plasma total calcium (see Methods). Plasma Pi levels were similarly and significantly reduced at 2 hours post injection for both peptides, and no change in either plasma Pi or total calcium was detected at 24 h-post injection (Supplemental Fig. S-1A, B).
Injection of Palm-PTH(1-34) also resulted in a significant increase in urine calcium levels at 24 h-post injection and relative to the levels in the vehicle-injected mice, while PTH(1-34) injection tended to increase urine calcium only at 7 h and the change was not significant; urine Pi was elevated in both peptide-injected groups at 2 h post-injection but the change was significant only for PTH(1-34) (Supplemental Fig. S-1C, D). Neither ligand resulted in a change in CTX1, but injection of Palm-PTH(1-34) caused blood levels of 1,25-dihydroxyvitamin D to increase to approximately twice the levels observed in mice injected with PTH(1-34) at 7 h post-injection, and the levels remained elevated relative to those in the vehicle-injected mice at 24-h post-injection (P < 0.001), by which time the levels in mice injected with PTH(1-34) had returned to baseline (Supplemental Fig. S-1E, F). These results on serum 1,25-dihydroxyvitamin D, together with the sustained effects on blood phosphorus levels establish that Palm-PTH(1-34) induces responses in kidney that are more sustained than those induced by conventional PTH(1-34).
To evaluate the pharmacokinetic (PK) properties of Palm-PTH(1-34), we measured the concentrations of the peptide, or that of PTH(1-34), in blood at times after SC injection. The levels of the two peptides were determined using a commercial ELISA developed for human PTH(1-34) and calibrating the assay with standard curves generated with either Palm-PTH(1-34) or PTH(1-34). By this approach, we found that while both peptides could be detected in the blood soon after injection, Palm-PTH(1-34) attained a maximum concentration (Cmax) that was at least 30-times that of PTH(1-34) (Cmax = 28.1 nM vs. 0.65 nM), and the time to attain the maximum was about 6-times slower for the lipidated analog than for the non-lipidated peptide (Tmax = 60 mins. vs. 10 mins, Fig. 1E). In addition, Palm-PTH(1-34) could be detected at the last 24 h post-injection time point, whereas PTH(1-34) was not detected past six h post-injection (Fig. 1E). Fitting the data obtained at the 1–24 h time points (i.e, from the peak onwards and thus excluding the initial absorption phase) to a monophasic decay function yielded an estimated half-life (T1/2) of 1.9 h for the lipidated peptide vs. 0.3 h for PTH(1-34). Similarly marked differences in the pharmacokinetic, as well as pharmacodynamic (blood Ca++) properties of the two peptides were obtained following intravenous (IV) injection of the peptides in mice (Suppl. Fig. S-2).
We also evaluated the renal filtration properties of PTH(1-34) and Palm-PTH(1-34) by injecting fluorescent tmr-labeled versions of the peptides into mice and monitoring the appearance of tmr fluorescence in urine at times after injection. Each analog contained the tmr moiety appended to the side chain of Lys13, a modification known to have little if any impact on the functional properties of PTH ligands20. Spot urine was collected at times after injection and measured for tmr fluorescence in a plate reader, which was converted to peptide concentration by extrapolating from standard curves generated with each control peptide. Following injection of PTH(1–34)tmr, tmr fluorescence was observed as a robust peak at the 1 h time point and then then diminished to near base-line levels by 24 h. In contrast, after injection of Palm-PTH(1-34)tmr, the tmr fluorescence reached a much lower peak that occurred at 2 h post-injection before diminishing to near base-line levels by 24 h, such that the area-under-the-curve (AUC) values obtained for the data with Palm-PTH(1-34)tmr was approximately 1/2 that obtained with PTH(1-34)tmr (Fig. 1F). These data are consistent with a slower rate of excretion of Palm-PTH(1-34)tmr into the urine, relative to PTH(1-34)tmr, and thus a more prolonged persistence of the lipidated peptide in the circulation. Although we did not evaluate the possibility that a fraction of the tmr fluorescence measured in these experiments derives from excreted free tmr that became detached from the peptide by some unknown mechanism operating in vivo after injection, we note that such a scenario would likely impact the two peptides similarly, given that the tmr is similarly linked to a lysine at position-13 in each ligand, and hence would not be expected to contribute to the differences in the quantities in urine observed for the two peptides.
The pharmacokinetic data together indicate that the lipid modification in Palm-PTH(1-34) prolongs the peptide’s bioavailability in the circulation and reduces its rate of renal elimination into the urine and are thus consistent with the hypothesis that the lipidated C-terminal tag promotes binding to serum albumin. The data do not exclude, however, the possible involvement of other contributing mechanisms, such as a reduction in the rate of absorption from the subcutaneous injection site compartment, an increase in distribution of the peptide to and subsequent release from soft tissues, and enhanced resistance to proteases.
To directly evaluate peptide binding to albumin, we conducted in vitro biolayer interferometry (BLI) experiments in which we analyzed the binding of Palm-PTH(1-34) as well as Palm-M-PTH(1-14) to bovine serum albumin that had been pre-adsorbed to the optical probes of the detector. We found that each lipidated peptide exhibited a dose-dependent increase in binding to albumin that was characterized by a maximum (Bmax) value at least three-fold higher, and an estimated KD value at least three-fold lower than that obtained for the corresponding non-lipid counterpart peptide (Suppl. Fig. S-3). These data thus support the notion that the C-terminal EYEK(palm)EYE tag attached to each of these two lipidated PTH peptides indeed promotes direct binding to serum albumin.
Palm-PTH(1-34) normalizes blood Ca++ in parathyroidectomized mice
We then assessed the translational potential of Palm-PTH(1-34) by using parathyroidectomized (PTX) mice as a model of surgical hypoparathyroidism31. The parathyroid glands were surgically removed and four days later the mice were used for the experiment, by which time the mice had developed frank hypocalcemia (Fig. 2A), which characterizes hypoparathyroidism. Upon SC injection of PTH(1-34) control peptide at a dose of 50 nmol/kg, blood Ca++ levels in the PTX mice rose briefly but did not attain the Ca++ levels of sham-surgery control mice, and the levels returned to baseline by 6 h post-injection. In contrast, upon injection of Palm-PTH(1-34), at a dose of 50 or 10 nmol/kg, blood Ca++ levels rose to near or within the normal range and persisted at those levels for at least 24 h (Fig. 2B). Even at a dose of 2.0 nmol/kg, Palm-PTH(1-34) increased blood Ca++ to levels similar to those attained with PTH(1-34) injected at a 25-fold higher dose.
GFP-guided parathyroidectomy or sham surgery was performed on two-month old PTH-Cre;Rosa-mT/mG mice four days prior to the day of the experiment. A Baseline blood Ca++ concentrations in the sham-operated and parathyroidectomized (PTX) mice assessed 0.5 to 1 h prior to injection. Boxplot shows Median, 25th and 75th percentile as well as minima and maxima of n = 5 (Sham) or 30 (PTX) mice per group, p-value determined by two-sided t-test. B Blood Ca++ concentrations as a function of time after SC injection of PTH(1-34) at a peptide dose of 50 nmol/kg-body weight, or Palm-PTH(1-34) at a peptide dose of 2, 10 or 50 nmol/kg-body weight or vehicle. Data are means ± SEM of n = 6 mice per group; p < 0.05 (2-way ANOVA followed by LSD test): *, peptide vs. vehicle; +, PTH(1-34) vs. Palm-PTH(1-34), #, PTX-vehicle vs. Sham-vehicle. Values at t = 0 represent those shown in (A) distributed by injection group. Source data with exact p values and deaggregation by sex (19 females, 17 males) are provided in the Source Data File. Concentrations of bone and mineral metabolism markers in blood plasma collected 24 h post-injection are shown in Supplemental Fig. S-4.
In parallel to the robust increases in blood Ca++, serum levels of 1,25-dihydroxyvitamin D3 at 24 h post-injection were elevated approximately 4-fold in the PTX mice injected with the 50 nmol/kg dose of Palm-PTH(1-34), as compared to the levels in PTX mice injected with vehicle (P < 0.0001), but were unchanged in the PTX mice injected with PTH(1-34) at the same dose as compared to the vehicle controls (Suppl. Fig. S-4).
The C-terminal albumin-binding motif prolongs the actions of M-PTH(1-14) in cells and in mice
The pronounced improvements in PK/PD properties that the EYEK(palm)EYE tag conferred to PTH(1-34) led us to ask if the same tag could provide similar improvements to shorter length agonist PTH peptides, such as M-PTH(1-14). M-PTH(1-14) is modified (as indicated by “M”) with six amino acid replacements that cumulatively improve agonist potency by at least 1000-fold, relative to the nearly inert native PTH(1-14), as assessed in cell-based signaling assays32. Despite the improvements in vitro, M-PTH(1-14) has been found to be inactive when tested in vivo33, which likely is due to the peptide’s small size (~1700 Daltons) leading to a rapid rate of renal clearance as well as a rapid rate of dissociation from the receptor34. We thus synthesized Palm-M-PTH(1-14) containing the EYEK(palm)EYE tag linked to the peptide’s C-terminus via a PEG2-like (20 atom) spacer. The resulting Palm-M-PTH(1-14) analog exhibited a potency for stimulating cAMP formation in SGS-72 cells that was greater than that of non-lipidated M-PTH(1-14), as assessed both during the Ligand-on phase and after washout of the unbound ligand (Fig. 3A, B). Interestingly, the improvements in signaling potency of up to 10-fold observed for Palm-M-PTH(1-14) vs. M-PTH(1-14) in SGS-72 cells were not observed in the studies with Palm-PTH(1-34) vs. PTH(1-34) (Fig. 1B), suggesting that the lipid moiety as attached to the C-terminus of the PTH(1-14) scaffold, but not as attached to the C-terminus of the longer PTH(1-34), might provide favorable interactions with the cellular plasma membrane, a mechanism explored in the section below on membrane anchoring.
A Amino acid sequences of PTH(1-34), M-PTH(1-14) and Palm-M-PTH(1-14) and cAMP signaling responses in SGS-72 cells assessed in the presence of varying concentrations of ligand (left Ligand on), and after rinsing the cells to remove unbound ligand (right, washout). Responses were assessed as the area under the curve (AUC) of the cAMP-dependent luminescence signal vs. time plots and are plotted as a function of ligand concentration. Curves were fit to the data using a non-linear regression equation. B Curve fit parameters for the data shown in (A). Data are means ± SEM of 4 independent experiments. p-value determined by two-sided t-test (C) Blood Ca++ concentrations as a function of time after IV injection of peptide (10 nmol/kg-body weight) or vehicle in 10-week-old intact female CD1 mice. Data are means ± SEM values from six mice per group; p < 0.05 (2-way ANOVA followed by LSD test): *, vs. M-PTH(1-14); +, Palm-M-PTH(1-14) vs. PTH(1-34). Values at t = 0 represent samples collected 0.5 to 1 hr prior to injection. Source data with exact p values are provided in the Source Data File.
The favorable cAMP responses observed in vitro for Palm-M-PTH(1-14) prompted us to assess the effects of the peptide in vivo. We thus compared Palm-M-PTH(1-14) vs control peptides for effects on blood Ca++ levels in intact adult CD-1 mice after a single-dose intravenous (IV) injection. Palm-M-PTH(1-14) (10 nmol/kg) caused blood Ca++ levels to increase for at least 7 h after injection, whereas an equal molar dose of PTH(1-34) caused blood Ca++ to increase for no longer than 5 h, and, as expected, injection of non-lipidated M-PTH(1-14) resulted in no change in blood Ca++ (Fig. 3C).
Lipid-mediated membrane anchoring promotes actions in cells and in vivo by M-PTH(1-14) and M-PTH(1-11)
The observation that C-terminal lipidation on M-PTH(1-14) increased the efficacy of the peptide in the washout phase of the SGS-72 cell cAMP assay led us to explore other lipidation strategies for the M-PTH(1-14) scaffold. We first synthesized several peptides with a C16 palmitoyl or palmityl chain linked directly, i.e. without a spacer, to the peptide scaffold at either the N-terminus or the C-terminal residue and assessed the signaling actions of the peptides in GP-2.3 cells, which are HEK293-derived cells that are stably transfected to express the glosensor reporter as well as the human PTH1R. These cells thus provide PTH1R signaling functionality similar to that of SGS-72 cells, albeit at higher surface receptor at levels. None of the modifications improved potency of cAMP generation, relative to the non-lipidated M-PTH(1-14) parent peptide or the prior Palm-M-PTH(1-14) analog containing the EYEK(palm)EYE tag (Suppl. Fig. S-5).
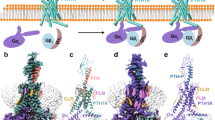
We then explored other modes of lipid attachment to specifically assess the possibility that a lipid chain appended to the M-PTH(1-14) scaffold in an appropriate manner might act to promote signaling by anchoring the ligand to the plasma membrane as the ligand is situated in the receptor’s orthosteric pocket. We were inspired to pursue such a mechanism based on the recent X-ray crystal and Cryo-EM structures obtained for the PTH1R in complex with various PTH ligands35,36,37,38. These structures thus reveal that the 1–14 portion of the ligand is situated in the receptor’s orthosteric pocket as an α-helix with the N-terminus positioned deep within the pocket and the 11–14 region located closer to the extracellular surface, such that the side chain at position 11 in the ligand projects towards a crevice that occurs between the extracellular ends of TM helices 1 and 2 while the side chain of residue 13 projects towards a crevice at the opposite end of the pocket between the extracellular ends TM helices 5 and 6. We further noted that certain designed peptide analogs of glucagon and/or GLP-1 contained a lipid moiety appended to the side chain of position 1025,39, which, upon amino acid sequence comparison40, aligns with residue 11 of PTH40. We thus hypothesized that a lipid chain appended via an appropriate spacer to the side chain of either residue 11 or 13 in the peptide would project through the respective crevice and insert into the lipid bilayer of the surrounding plasma membrane to thereby anchor the ligand in place as it is bound to the receptor (Fig. 4A). This model further predicts that a lipid appended at position 12 in the peptide, rotated by ~100° relative to residue 11 or 13, would project into the inner wall of the binding pocket, and thus not provide such an anchoring effect on the ligand-receptor complex (Fig. 4A).
A Left, schematics of N-terminal PTH peptides containing a C16 palmitoyl (palm) group appended to the side chain amine of a C-terminal lysine via an Arg (R) dArg(r), Glu(E) or dGlu(e) spacer; the structure of K11(R)palm-PTH(1-11), and peptide sequences. Right; Models of the receptor TMD (gray, with helix H1 in turquoise) bound to a PTH fragment peptide (red) containing a hypothetical lipid chain (orange scribble line) appended to the side chain of a C-terminal Lys at position 11, 12 or 13. Views are from the extracellular surface and side. Images were generated using Pymol and PDB.6nBF (LA-PTH-PTH1R-Gs cryo-EM structure). Lipid chains at positions 13 and 11 were drawn manually to depict their postulated extension through the H5-H6 and H1-H2 clefts, respectively. The lipid chain at position 12 depicts a clash with the inner wall of the binding pocket. B cAMP responses in GP-2.3 cells to M-PTH(1-14), K13(R)palm-PTH(1-13), K12(R)palm-PTH(1-12) and K11(R)palm-PTH(1-11); responses were calculated as the AUCs of the cAMP-dependent luminescence vs. time plots observed after ligand addition (Ligand-on, 20’) and in the same cells after rinsing to remove unbound ligand (washout, 120’), normalized to the maximum response observed for M-PTH(1-14) and are plotted as a function of peptide concentration. p-values were determined by two-sided t-test. C Schematic of N-terminal PTH peptides containing octadecanedioic acid (dioic) group appended to the side chain amine of a lysine via an Arg (R), spacer. D cAMP responses in GP-2.3 cells induced by PTH(1-34) or a PTH(1-11) or PTH(1-13) peptide containing either a palmitoyl or dioic group appended to a lysine side chain. Responses calculated as in (B) but normalized to the maximum response of PTH(1-34). Data are means ± SEM of four independent experiments. Curves were fit to the data using a non-linear regression equation; corresponding potency (pEC50) values are shown in the inset. Source data with exact p values are provided in the Source Data File. Data for related peptides are shown in Supplemental Figure S-6 and Supplemental Tables S-1 and S-3.
To test the above hypothesis, we synthesized N-terminal PTH fragment peptides containing a C16 palmitoyl (palm) chain appended to the side chain amine of a C-terminal lysine located at either position 11, 12 or 13, via a spacer amino acid that was either L-Glu (E), dGlu (e), L-Arg (R) or dArg (r) (Fig. 4A). The cAMP signaling responses induced by the peptides were assessed in GP-2.3 cells both in the presence of the ligand (Ligand-on phase) and after rinsing the cells to remove unbound ligand (washout phase). Figure 4B shows dose-response potency curves generated from the AUCs of the time vs. cAMP-dependent luminescence responses obtained in these assays for the three peptides containing an L-Arg spacer. Both K13(R)palm-PTH(1-13) and K11(R)palm-PTH(1-11) exhibited a potency in the Ligand-on phase that was comparable to that of M-PTH(1-14) and the potency of each ligand was largely maintained during the washout phase, whereas the potency of M-PTH(1-14) was diminished by ~90-fold following washout. K12(R)palm-PTH(1-12) exhibited a potency that was ~14-fold weaker than that of M-PTH(1-14) in the Ligand-on phase, and its potency decreased ~2-fold further after the washout. These results are consistent with the hypothesis that the lipid chains extending from positions 11 or 13 in the ligand provide a stabilizing effect on the ligand-receptor complex to thereby enable prolonged signaling, whereas the lipid chain extending from position 12 provides less of a stabilizing effect and hence leads to a diminished and more transient signaling response, as compared to the responses seen with the peptides lipidated at position 11 or 13.
Similar findings were obtained with the three peptides in this series containing a dArg spacer amino acid, whereas the peptides having L-Glu or dGlu as the spacer exhibited potencies during both the Ligand-on and washout phases that were weaker than those of the corresponding L-Arg and dArg counterpart peptides, as especially apparent for the PTH(1-11) analogs (Suppl. Fig. S-6 and Suppl. Table 1). These data suggest that the presence of a spacer between the peptide backbone and the lipid appendage is important for determining peptide bioactivity. Moreover, the superior performance of the Arg/dArg spacer relative to the Glu/dGlu spacers suggests a beneficial interaction between the positively charged guanidinium group of Arg and dArg in the spacer and the negatively charged headgroups of lipids in the cell membrane41.
We ruled out the possibility that the effects of the lipid appendages on the cAMP signaling actions of the N-terminal PTH peptides involved interactions with the receptor’s N-terminal extracellular domain (ECD) as we found that the lipids provided nearly the same enhancing effects on cAMP signaling in GD-5y cells, which stably express a PTH1R construct, PTH1R-delNTyfp, that lacks the receptor’s ECD, as those observed in GP-2.3 cells expressing the intact PTH1R (Suppl. Fig. S-7, Suppl. Table S-2). Indeed, the side chain lipids at positions 11 and 13 improved the signaling potency of the N-terminal fragment peptides after washout in GD-5y cells by as much as 200-fold, relative to the potency of the non-lipidated M-PTH(1-14) peptide.
As a further test of the hypothesis that the lipid chain extensions at position 11 or 13 promote signaling by inserting into the lipid compartment of the plasma membrane, we synthesized peptides incorporating a C18 diacid chain (dioic) at those positions to thereby introduce a polar carboxyl group at the distal end of the fatty acid chain (Fig. 4C). The rationale was that this distal polar carboxylate would be less effective at inserting into the hydrophobic component of the lipid bilayer of the plasma membrane, as compared to the fully apolar C16 chain of the palmitoyl group, and thus not provide the same stabilizing effect on the complex as achieved with the palmitoyl group. Consistent with this model, K13(R)dioic-PTH(1-13) and K11(R)dioic-PTH(1-11) each showed a potency for cAMP generation in GP-2.3 cells that while comparable to that of the corresponding palmitoyl peptide during the Ligand-on phase, was markedly weaker than that of the palmitoyl peptide as measured during the washout phase (Fig. 4D; Suppl. Table S-3). The results thus support a model by which the apolar palmitoyl chains of K13(R)palm-PTH(1-13) and K11(R)palm-PTH(1-11) promote complex stability and hence signaling via a mechanism involving in situ anchoring of the receptor-bound ligand to the surrounding cellular plasma membrane.
Lipidation at the C-terminus of Exendin4 has been shown to slow the rate at which this GLP-1R peptide agonist induces GLP1R internalization and to impair recruitment of βarrestin226. We assessed our lipidated PTH analogs for inducing receptor internalization using HEK293 cells stably expressing a PTH1R derivative, PHL2-PTH1R, containing a pH-sensitive GFP inserted into the ECD (GPG-10 cells), which permit the monitoring of ligand-induced PTH1R trafficking into acidic endosomes as a time-dependent increase in GFP fluorescence42. Both K13(R)palm-PTH(1-13) and Palm-M-PTH(1-14) dose-dependently induced internalization of the PHL2-PTH1R into such acidic compartments, and these responses were inhibited by the dynamin GTPase inhibitor, Dyngo4A, consistent with a dynamin/clathrin-coated pit-mediated process of receptor internalization (Suppl. Fig. S-8A). As expected, a PTHrP(7-36) antagonist peptide did not induce internalization. The internalization responses induced by K13(R)palm-PTH(1-13) at sub-micromolar concentrations were significantly weaker than those induced by Palm-M-PTH(1-14), suggesting a possible inhibitory effect of the lipid at position 13 on the internalization process.
Fluorescent microscopy imaging analysis performed in HEK293 cells stably expressing βarrestin2yfp and transiently transfected with the PTH1R revealed that by 30 minutes after addition, K13(R)palm-PTH(1-14)tmr, formed clusters that colocalized with βarrestin2yfp and were presumably located in endosomes with the PTH1R, as also seen with non-lipidated PTH(1-34)tmr and M-PTH(1-14)tmr (Suppl. Fig. S-8B). The lipidated PTH(1-14) analog thus retains the capacity to recruit βarrestin2 to the receptor.
Effects of lipidated PTH peptides on bone metabolism in humanized PTH1R mice
The positive results obtained with the lipidated N-terminal PTH peptides in cells encouraged us to evaluate these peptides for effects on calcium and bone physiology using mice expressing the human PTH1R in place of the mouse PTH1R (humanized PTH1R mice)43. We first assessed the capacity of K11(r)palm-PTH(1-11) to increase blood ionized calcium levels after a single acute injection in five-month-old male humanized PTH1R mice. The results shown in Suppl. Fig. S-9A demonstrate that whereas injection of non-lipidated M-PTH(1-11) had no effect on blood calcium, injection of K11(r)palm-PTH(1-11) resulted in an increase in serum calcium that was at least as robust and prolonged as that induced by PTH(1-34) injected at the same peptide dose of 50 nmol/kg.
We then assessed the lipidated PTH peptides for longer-term effects in vivo by performing repeated daily injection studies. Repeated daily injections of PTH(1-34) in humans and animals is well known to result in pronounced changes in bone metabolism and bone structure through direct effects on osteoblasts coupled with indirect effects on osteoclasts44, but shorter-length N-terminal peptides have so far been found to be inactive in vivo33. For the current studies, we injected five-month-old male humanized PTH1R mice43 with three lipidated PTH peptides that are predicted to function via three distinct modes of action: 1) K11(r)palm-PTH(1-11) as a lipidated N-terminal peptide that mediates potent and prolonged signaling actions via in-situ membrane anchoring; 2) K11(R)dioic-PTH(1-11) as a peptide of similar structure and acute cAMP signaling potency, but deficient in prolonged signaling due to a polar lipid substituent, and 3) Palm-PTH(1-34) as a full-length peptide with prolonged actions in vivo due to extended pharmacokinetics via albumin binding. Control mice were injected with unmodified PTH(1-34) and vehicle. Peptides were injected at a dose of 50 nmol/kg/day, or, for K11(R)dioic-PTH(1-11), at a dose of 500 nmol/kg/day. We note that in a prior pilot experiment, we injected mice with K11(r)palm-PTH(1-11) at dose of 500 nmol/kg/day and observed excessive hypercalcemia after 3–4 days of injection, and so a 50 nmol/kg/day dose was selected for that peptide in the current experiment, as also guided by the positive calcemic response induced by the peptide at a dose of 50 nmol/kg in the single injection experiment presented in Suppl. Fig. S-9A. We also note that the polar end group of the diacid C18 chain in K11(R)dioic-PTH(1-11) is not expected to result in a substantial difference in peptide pharmacokinetics as compared to that of K11(r)palm-PTH(1-11) containing the fully apolar palmitoyl C16 chain, given the similar sizes of the two acyl chains and their likely similar affinities for binding albumin23,45. The mice were injected daily for three weeks and 24 h after the last injection were humanely euthanized, and bone and blood samples were collected for analysis. Compared to vehicle-injection controls, body weights remained unchanged after peptide analog injection (Fig. S-9B).
Microcomputed tomography (μCT) analysis of the isolated femurs revealed that injection of either K11(r)palm-PTH(1-11) or Palm-PTH(1-34) resulted in profound increases in trabecular bone mass in the distal metaphyses, indicated by quantitatively significant increases in trabecular bone volume fraction (Tb.BV/TV), bone mineral density (Tb.BMD), and the number of bone trabeculae (Tb.N.) that were at least as substantial as those observed with PTH(1-34), whereas injection of K11(R)dioic-PTH(1-11) resulted in no significant increase in these parameters despite being injected at a dose 10-fold higher than that used for the other peptides (Fig. 5A and B, Suppl. Fig. S-9C, Suppl. Table 4).
Five-month-old male humanized PTH1R mice were injected daily for three weeks with either vehicle, PTH(1-34) Palm-PTH(1-34), K11(r)palm-PTH(1-11), each at a dose of 50 nmol/kg/day, or with K11(r)dioic-PTH(1-11) at a dose of 500 nmol/kg/day and femurs were isolated at the end of the experiment for analysis. A Representative µCT radiographs of the isolated femurs; views are in the sagittal plane at the distal metaphysis (top) and in the transverse plane at the midshaft (bottom). Mouse identification numbers are shown adjacent to each image. B Quantitative µCT parameters of trabecular bone volume fraction (BV/TV), trabecular bone mineral density (BMD, as mg of hydroxyapatite per cm3) and trabecular number (TB.N) measured in the distal metaphysis. C Mechanical strength, as work to fracture, determined by a three-point bending test of the femurs. Data are means ± SD of measurements from five mice indicated by points with adjacent mouse identification numbers. Statistical differences vs. vehicle were analyzed by one-way ANOVA with Dunnetts multiple comparison test: *, p < 0.05; **, p < 0.01; ***, p < 0.001; ****, p < 0.0001; ns, not significant. Source data with exact p values are provided in the Source Data File. Additional data are shown in Supplemental Material for µCT (Fig. S-9A and Table S-4), and mechanical testing (Fig. S-9B).
Histological sections of the distal femur stained with trichrome to show mineralized tissue (blue-green) revealed marked increase in trabecular bone in the bones of mice injected with Palm-PTH(1-34) or K11(r)palm-PTH(1-11), as compared to those injected with vehicle, PTH(1-34) or K11(R)dioic-PTH(1-11) (Suppl. Fig. S-10).
In the area of the femur mid-shaft, which is normally comprised mostly of high-density cortical bone, the μCT radiographs revealed that injections of either K11(r)palm-PTH(1-11) or Palm-PTH(1-34) resulted in marked accumulations of what appeared to be low-density woven bone on the inner as well as outer surfaces of the mid-shaft wall (Fig. 5A). The markedly irregular contours of the mineralized bone in this region led us to limit our quantitative measurement at the mid-shaft to only the higher-density cortical bone areas. These measurements indicated significant increases in cortical bone porosity with injection of PTH(1-34), K11(r)palm-PTH(1-11) or especially Palm-PTH(1-34), relative to vehicle-injected controls, and no change with K11(R)dioic-PTH(1-11), as well as moderate decreases in cortical thickness with injection of Palm-PTH(1-34) or K11(r)palm-PTH(1-11) although the effect on thickness was significant with only the latter peptide (Suppl. Fig. S-9C, Suppl. Table 4).
Despite the apparent irregularities in certain bone structural properties, injection of Palm-PTH(1-34) or K11(r)palm-PTH(1-11) resulted in significant increases in the mechanical strength of the femurs, as assessed by the work-to-fracture as well as the toughness-to-fracture under a three-point bending load (Fig. 5C, Suppl. Fig. S-9D). These increases in the bone-strength parameters observed for Palm-PTH(1-34) and K11(r)palm-PTH(1-11) were comparable to those observed for PTH(1-34) (P > 0.05), although only PTH(1-34) significantly increased bending rigidity (Suppl. Fig. S-9D). Injection of K11(R)dioic-PTH(1-11) had little or no effect on any bone structural parameter. The effects of Palm-PTH(1-34) and K11(r)palm-PTH(1-11) on bone toughness and resistance to fracture seem to parallel the increases in the trabecular bone structural parameters at the distal femur as assessed by μCT (Fig. 5A and B, S-9 C and D) and by histology (Suppl. Fig S-10). The difference in the effects of Palm-PTH(1-34) and K11(r)palm-PTH(1-11) vs. PTH(1-34) on mechanical bending rigidity, on the other hand, might be related to the differences in the effects of the peptides on cortical bone structural properties at the mid-shaft, as the quantitative μCT data indicated that the two lipidated peptides induced significantly greater increases in cortical porosity at this site as compared to PTH(1-34) (Suppl. Fig. S-9C and Table S-4).
Measurements of serum collected 24 h after the last injection showed that relative to the levels in vehicle-injected mice, total serum calcium was modestly yet significantly increased with injection of Palm-PTH(1-34) but not with the other peptides, while serum phosphorus levels were not changed in any peptide-injected group (Suppl. Fig. S-11). In parallel with the marked changes in bone structure, serum levels of CTX1 and the bone formation marker, P1NP, were significantly elevated in mice injected with either K11(r)palm-PTH(1-11) or Palm-PTH(1-34), while PTH(1-34) injection significantly increased P1NP but not CTX1, and no change in these markers occurred in mice injected with K11(R)dioic-PTH(1-11) (Suppl. Fig. S-11).
We performed a similar 3-week injection experiment in five-month-old male humanized PTH1R mice to assess K13(r)palm-PTH(1-13) and Palm-M-PTH(1-14) along with M-PTH(1-14), PTH(1-34) and vehicle injection as controls. In this experiment, Palm-M-PTH(1-14) and K13(r)palm-PTH(1-13) were each injected at a dose of 500 nmol/kg/day, while PTH(1-34) was injected at a dose of 50 nmol/kg/day. Bone and blood samples were analyzed at the study end point, as described in the experiment above. MicroCT analysis of the isolated femurs revealed that injection of K13(r)palm-PTH(1-13) or Palm-M-PTH(1-14) resulted in marked increases in trabecular bone mass in the distal metaphyses, as well as dramatic accumulations of low-density woven bone on the endocortical surfaces at the mid-shaft while M-PTH(1-14) resulted in little or no change in these parameters (Suppl. Fig. S-12 A, B). Palm-M-PTH(1-14) also induced marked accumulations of woven bone mass on the periosteal bone surfaces.
Histological analysis of the proximal tibiae confirmed that injection of either K13(r)palm-PTH(1-13) or Palm-M-PTH(1-14) led to dramatic increases in bone mass as well as in fibrotic tissue surrounding the bone that together ultimately filled much of the marrow cavity area under the growth plates (Suppl. Fig. S-12 C, D). These changes in bone structure seen in the mice injected with K13(r)palm-PTH(1-13) and Palm-M-PTH(1-14) were paralleled by significant increases in serum levels of P1NP, which were unchanged in mice injected with non-lipidated M-PTH(1-14) (Suppl. Fig. S-12E). Mechanical testing analysis revealed a moderate increase in the work to ultimate moment parameter with injection of K13(r)palm-PTH(1-13). These also revealed a decrease in bending rigidity and an increase in the toughness with injection of Palm-M-PTH(1-14), which paralleled the decrease in cortical bone thickness, the increase in cortical bone porosity, and the increase in woven bone that were apparent in the μCT data and images obtained for the bones of mice treated with this Palm-M-PTH(1-14) analog.
Discussion
Peptide lipidation has gained footage as an easily adoptable design strategy to improve the efficacy in vivo of a bioactive peptide. Use of the approach is most notably demonstrated in the development of peptides of the incretin family that target the class B GPCRs for GLP-1, glucose-dependent insulinotropic polypeptide (GIP) and/or glucagon for the treatment of diabetes and obesity22,46. The GLP-1 analog, semaglutide, thus has a C18 diacid chain coupled to a lysine at position 20 in the 31-amino acid peptide chain23,24. The main beneficial effect of the lipid in semaglutide appears to be enhancement of PK properties via binding to serum albumin although other effects may be involved as discussed in the introduction23,24. Our current studies demonstrate that lipidation can also be an effective means to alter the actions of PTH peptide ligands in vivo.
From a therapeutic standpoint, our studies have potential relevance to diseases such as osteoporosis and hypoparathyroidism, for which other non-lipidated PTH analogs are already in use. For both diseases, the therapeutic efficacy attained by a given PTH peptide is dependent, at least in part, on the duration of the biological effect induced by the ligand with each cycle of administration44,47. For osteoporosis, where the goal is to increase bone mass and strength, the optimal profile seems be a pulsatile, peak-to-trough mode-of-action, as is achieved by daily subcutaneous injection of non-modified PTH(1-34) (teriparatide), or a substituted PTHrP(1-34) analog (abaloparatide)44,47. For hypoparathyroidism, where the goal is to maintain a constant 24-h control of serum calcium levels, a more sustained mode of action is desirable, and hence underlies the development of PTH analogs designed to have prolonged action in vivo5. Such approaches explored so far include modifications that delay the rate of renal clearance of the administered PTH, as is obtained by increasing the effective bulk size of the ligand, for example through pegylation4,20 or via conjugating the peptide to vitamin D to promote binding to vitamin D-binding serum proteins48, or through peptide side chain or back bone modifications that promote sustained binding to and signaling action on the target PTH1R, as used in LA-PTH3,15 to improve stability and or resistance to protease degradation in vivo18,49.
Our current results indicate that lipidation is also an effective strategy for prolonging and enhancing the actions of short N-terminal PTH(1-14) or even PTH(1-11) analogs in vivo and provide evidence that the enhancing effects might occur through two distinct mechanisms -- effects on PK via binding to serum albumin and effects on receptor engagement via lipid interactions with the plasma membrane -- depending on the nature and position of the lipid appendage employed.
To specifically extend the PK properties of PTH analogs we employed the C-terminal lipidated hepta-residue tag motif, EYEK(palm)EYE, developed by Zorzi et al. for serum albumin binding28. We established that this C-terminal addition on PTH(1-34) largely preserves signaling potency in cells and sustains the calcemic and hypophosphatemic responses in normal mice as well as provides for sustained normalization of blood calcium in a parathyroidectomized mouse model of surgical hypoparathyroidism. In support of an effect on pharmacokinetics, we showed that the lipidated Palm-PTH(1-34) analog persists in the blood after SC injection with a half-life at least six-times longer than that of PTH(1-34). We also showed that fluorescent Palm-PTH(1-34)tmr analog appeared in the urine at a much slower rate and in lower total abundance after SC injection than did PTH(1-34)tmr, confirming a delayed rate of renal clearance for the lipid-modified peptide.
We further confirmed using BLI spectroscopy that the EYEK(palm)EYE motif in Palm-PTH(1-34) as well as in Palm-M-PTH(1-14) promoted binding to serum albumin, as compared to the corresponding non-lipidated peptide which exhibited only weak if any binding. Together, our in vitro and in vivo data strengthen the notion that C-terminal lipidation of PTH(1-34) and C-terminally shortened peptide analogs can be an effective strategy for prolonging PTH1R agonist peptide action in vivo and potentially for developing new ligands as therapeutics for hypoparathyroidism. Our studies extend those of Ruan et al., which show that a PTH(1-34) peptide modified at Lys27 with a C18 diacid exhibits enhanced binding to serum albumin and thus induces sustained calcemic actions in rodents, as compared to PTH(1-34)30.
We extended our observations to the shorter-length N-terminal PTH fragment peptides, M-PTH(1-14) and M-PTH(1-11), which represent minimized agonist probes that bind only to the receptor’s TMD region, unlike PTH(1-34) which binds to both the ECD and TMD regions, as typical for each endogenous peptide hormone that binds to a class B GPCR. M-PTH(1-14) and M-PTH(1-11) analogs contain up to six affinity- and/or cAMP signaling-enhancing amino acid substitutions that cumulatively result in cAMP-signaling potencies in cells comparable to that of PTH(1-34). The lack of activity detected for such smaller PTH peptides in vivo33 can likely be explained in part by their relatively rapid rates of renal clearance combined with their faster rates of dissociation from the target receptor34. We showed that linking the EYEK(palm)EYE motif to the C-terminus of M-PTH(1-14) can confer activity in vivo, as Palm-M-PTH(1-14) upon IV injection in mice induced elevations in serum calcium that were even more robust and more prolonged than those induced by an equivalent dose of PTH(1-34), whereas M-PTH(1-14) was, as predicted, without effect.
We also showed that this C-terminal modification did not interfere with the cAMP signaling potency of M-PTH(1-14) as assessed in the Ligand-on format. Intriguingly, however, we observed that the signaling response was considerably prolonged after ligand washout, as compared to that of M-PTH(1-14), which suggested the possibility that the lipid appendage on Palm-M-PTH(1-14) could act to anchor the ligand to the cell membrane and thereby prevent its removal during rinsing and facilitate its diffusion to the receptor. In support of such a possibility, Liu et al. showed that a PTH(1-14) peptide containing a C-terminal dipalmitoyl appendage exhibited a four-fold slower rate of diffusion in micelle-like nanostructures, as analyzed by fluorescence correlation spectroscopy, and an approximate 100-fold improvement in cAMP signaling potency in HEK293/PTH1R cells, as compared to the unmodified PTH(1-14) control peptide29.
We then considered the possibility that a lipid moiety appended to a PTH(1-14) peptide at a certain position might provide a membrane-anchoring effect to stabilize the ligand-receptor complex in a productive geometry that leads to prolonged signaling. We were motivated to pursue this possibility by two sets of prior observations. First, in previous structure-activity relationship studies we identified the residue at position 11 in PTH(1-14) as a particularly important determinant of receptor-binding affinity and signaling potency, and that extensions in the length of the position 11 side chain from that of the native leucine, to that of arginine, and finally to that of homoarginine led to progressive enhancements in receptor binding and signaling potency50. The accompanying receptor mutational data in these studies further led to the prediction that the position-11 side chain projects between the extracellular ends of TMD helices 1 and 2 and towards the surrounding lipid bilayer, which is now supported by the recent high-resolution structures obtained for the PTH1R in complex with various peptide ligands provide35,36,38,51,52. Second, we noted that peptides in a series of GLP1/glucagon chimeras designed for dual agonism at the GLP1R and GCGR25, as well as a GLP1/glucagon/exendin4 chimera designed for tri-agonism at the GLP1R, GIPR and GCGR39, contained a lipid chain appended to a lysine at position 10, which aligns with residue position 11 in PTH40.
We tested our membrane-anchoring hypothesis by synthesizing a series of N-terminal PTH peptides containing a lipid moiety appended to a C-terminal lysine located at position 11, 12 or 13 at the end of the peptide chain. By having the lipid attached to a sidechain at one of these three positions we sought to probe different projection angles for the lipid according to a 100° rotation per residue along the alpha-helix axis such that the lipids at position 11 and 13 would project towards gaps located at opposing ends of the upper portion of the receptor’s TMD pocket, whereas the lipid at position 12 would clash with the inner wall of the pocket (Fig. 4 A). The functional data we obtained with these peptides in cells are fully consistent with these predictions. Thus, K11(R)palm-PTH(1-11) and K13(R)palm-PTH(1-13) each exhibited a potency for stimulating cAMP formation in SGS-72 cells that was about 10-fold greater than that of K12(R)palm-PTH(1-12), as measured during the Ligand-on phase, and the difference was about 20-fold when measured during the washout phase. The signaling potencies of K11(R)palm-PTH(1-11) and K13(R)palm-PTH(1-13) were thus largely maintained after the washout step, whereas that of the non-lipidated M-PTH(1-14) parent peptide was reduced by ~200-fold as compared to the potencies measured in the Ligand-on phase.
We further showed that K11(R)dioic-PTH(1-11) and K13(R)dioic-PTH(1-13) containing a lipid chain with a terminal polar group were at least 20-fold less effective at maintaining signaling after washout than their respective counterpart peptides containing a fully apolar palmitoyl lipid chain. The overall results thus support our hypothesis that a lipid appended to the side chain at position 11 or 13 in an N-terminal PTH peptide can prolong signaling e.g. by anchoring the peptide to the plasma membrane as it is bound to the receptor’s orthosteric pocket, although more direct methods of analysis, such as by Cryo-EM are needed to fully evaluate this proposed model. In further support of such a model, however, we note that recent Cryo-EM structures of the GLP1R, GCGR and GIPR in complex with either a GLP1R/GCGR/GIPR triagonist (peptide 20), or a GLP1R/GCGR dual agonist (MEDIO382), each of which contains a palmitoyl group linked to a lysine at position-10, show that the lipid chain in each peptide indeed extends through a crevice located between the extracellular ends of TM H1 and H2 of the receptor and hence project downwards along the outer face of receptor and interface with the surrounding lipid of the membrane53,54. Moreover, the potency of each of these peptides for stimulating cAMP signaling in cells expressing the GLP1R or GCGR was at least 1000-fold greater than that of the corresponding non-lipidated peptide, supporting the model by which the lipids help stabilize a productive interaction with the target receptor.
In Cryo-EM structures of class BGPCRs in complex with other lipidated peptides of this class, the lipid chains were not resolved due to high thermal motion. In these cases, the lipids were appended to a lysine located in the C-terminal portion of the peptide that contacts the receptor’s ECD, and thus may not be close enough to the cell surface to promote stability through a strong membrane anchoring effect. Examples of this include the C18 diacid linked to lysine at position 20 in semaglutide in complex with the GLP1R55, and the C18 diacid linked to lysine at position 20 in tirzepatide, a GIPR/GLP1R dual agonist, in complex with either the GLP1R or GIPR56.
A potentially important contributing factor to the potency of a lipidated ligand is the nature of the spacer used to connect the lipid to the peptide, especially if the spacer makes contact with functional determinants on the receptor23,53,54. For our PTH peptides, we showed that an Arg or dArg is more compatible with function than is a Glu or dGlu as a spacer for linking the fatty acid to the peptide via the side chain amine of a lysine at position 11 or 13. These findings are generally consistent with our previous structure-activity relationship studies of PTH(1-14) peptides in which we found beneficial effects on potency upon replacement of the normal leucine at position 11 with arginine and especially homoarginine50.
An additional factor we did not explore is cross-reactivity with other GPCRs and potential toxicity in vivo; although we note that animal body weights were maintained over the course of 3-weeks of daily injection, except with Palm-M-PTH(1-14) at dose of 500 nmol/kg, for which weights decreased by week 3 (Suppl. Fig. S-11F). This reduction in body weight could be due to transient hypercalcemia that occurs from excess PTH1R signaling in bone and or kidney cells after each injection, although blood total calcium levels in this group were normal by 24 h after the last injection (Suppl. Fig. S-11E).
Further questions raised by our findings include the extent that lipidation may alter the functional properties of a PTH ligand in different PTH1R-expressing target cell types, namely, those of bone, kidney and growth plate chondrocytes, the impact on signaling through various second-messenger pathways, including the Gαs/cAMP/PKA, Gq/PLC/PKC and βarrestin/ERK1/2 pathways, the trafficking of ligand-receptor complexes to different subcellular compartments, such as endosomes vs the plasma membrane, as well as the dosing regimens that can be used for such lipidated PTH peptides to achieve optimal effects on bone and mineral ion metabolism. A challenge in the development of any new PTH-based ligand as a therapy for either hypoparathyroidism or osteoporosis is to identify a dose regimen that leads to favorable effects on the balance of bone turnover and calcium mobilization, as excess PTH1R signaling in bone and kidney target cells can result in hypercalcemia and/or excess bone turn-over5,47,57. The presumably unfavorable increases in cortical porosity that we observed with daily injection of the Palm-M-PTH(1-14) analog in the study represented in the data of Supp. Fig S-12 are thus likely attributable to the high dose (500 nmol/kg/day) of peptide utilized in that experiment. While further work is needed to more fully evaluate the therapeutic potential of the new peptides we report herein, our study nevertheless provides initial proof-of-concept support for the notion that lipidation can be an effective strategy for enhancing the efficacy in vivo of even minimized PTH agonist peptides.
In summary, we showed that signaling and biological actions of PTH(1-34) and shorter-length peptides, including M-PTH(1-11) in cells and in mice can be profoundly enhanced by peptide side chain lipidation. While an appended lipid modification could impact peptide hormone function by several different routes, we presented evidence in support of two distinct modes of action, as determined by the configuration of the lipid appendage. We thus showed that attachment of a previously reported EYEK(palm)EYE motif to the C-terminus of PTH(1-34) as well as to that M-PTH(1-14) prolongs the calcemic response induced upon single injection of the ligand into mice, likely via a mechanism involving a reduced rate of renal clearance due to enhanced association with serum albumin. We also showed that lipid modification at position 11 or 13 in M-PTH(1-14) and even a PTH(1-11) peptide, prolongs the cAMP signaling responses induced by those ligands in cells, and greatly augments the impact that the peptides have on bone metabolism upon repeated daily injection into mice, via a mechanism proposed to involve in situ anchoring of the peptide to the plasma membrane as the ligand is bound to the receptor. While additional work is needed to determine, for example, the high-resolution molecular structure of a lipidated PTH ligand-PTH1R complex and to thus help evaluate the proposed modes of action on the receptor, as well to help further define the effects that these ligands have on bone and calcium metabolism in vivo and hence their potential therapeutic utility towards diseases such as hypoparathyroidism and osteoporosis, our current studies should help prompt such further efforts aimed in these new directions.
While finalizing our work for publication, a study was published on a new lipidated PTH peptide called MBX 2109 that is in development by MBX Biosciences Inc. as a potential treatment option for hypoparathyroidism58. This peptide is designed as a prodrug and is comprised of a PTH(1-32) core sequence with a C18 fatty diacid attached at the C-terminus via an introduced lysine (Lys33) and a second C18 diacid attached at the N-terminus via a di-amino acid motif (Sar0-dLys-1), which undergoes autocyclization and cleavage at physiological pH and temperature to release the C-terminally acylated PTH(1-32) peptide as the active agent. The phase 1 clinical data reported in this study on the safety, pharmacokinetics, and calcium mobilizing properties of the peptide support the intended goal of using the peptide as a once-weekly subcutaneous injection-based treatment option for hypoparathyroidism. The results also further highlight the utility of using lipidation as a strategy for extending and enhancing the functional properties of PTH peptides in vivo.
Methods
Peptides and reagents
Peptides were synthesized at the Massachusetts General Hospital Biopolymer Core facility, except with M-PTH(1-14) analogs with direct lipid attachments (Suppl. Fig. S-5) which were synthesized at the University of Wisconsin. Solid-phase peptide synthesis was performed in a microwave synthesizer or manually using Fmoc/tBu chemistry with H-Rink amide ChemMatrix resin and Oxyma/DIC-mediated coupling. Site-specific addition of either a palmitoyl (palm, C16H31O) or octadecanedioic acid (dioic, C18H33O3) group to the free amine of either a lysine side chain or the alpha amino of an N-terminal amino acid was performed on the peptide resin after removal of the orthogonal protection group. Fluorescent ligand analogs were derived by covalently linking tetramethyl rhodamine (tmr) to the free amine of a lysine side chain. Peptide sequences are shown in the corresponding figures. Specific information on chemical synthesis is provided in Supplemental Methods.
Plasmids and cell lines
Functional assays were performed in stably transfected cell lines derived from HEK-293 cells (ATCC Cat. # CRL-1573) and expressing the glosensor cAMP reporter (plasmid p22f, Promega) along with either the human PTH1R (GP-2.3 cells), pHL2-hPTH1R, in which pHluorin2, a pH-sensitive GFP variant, is inserted into the E2 region of the hPTH1R’s ECD (GPG-10 cells), PTH1R-delNtyfp, in which the receptor’s N-terminal extracellular domain (ECD) is replaced by yellow fluorescent protein (GD-5y cells), or β-Arrestin2yfp (GBR-24 cells). Assays were also performed in SGS-72 cells, which are derived from the human osteoblastic cell line Saos-2 (ATCC Cat. # HTB-85) and stably express glosensor.
Glosensor cAMP assays
cAMP assays were performed in cell lines stably expressing the glosensor cAMP reporter9. Confluent cell monolayers in 96-well white plates were rinsed with CO2-independent medium (ThermoFisher Cat # 18045088) containing BSA (sigma A8412) at 0.1% w/v (CIDB), and then loaded with luciferin (Biotium) at 0.5 mM in CIDB (CIDB-Luc) for 30 minutes then treated with test ligands in CIDB and luminescence signals were recorded in an Envision plate reader (PerkinElmer, Waltham, MA, USA) at 60-second repeat cycles for 30 minutes (Ligand-on phase), then rinsed twice with CIDB, and after adding fresh CIDB-Luc, luminescence was again recorded for 90 minutes (washout phase). Peak luminescence signals, which typically occurred at 10-15 minutes after ligand addition, or the AUCs of time vs. luminescence plots were plotted vs. ligand concentration to derive potency curves.
Receptor internalization in GPG-10 cells
Ligand-induced PTH1R internalization responses were assessed in GPG-10 cells (HEK293 cells stably expressing pHL2-PTH1R containing a pH-sensitive GFP in the ECD)42. These cells permit detection of receptor internalization to acidic endosomes as an increase in GFP fluorescence at 535 nm upon excitation at 485 nm relative to 535 nm fluorescence upon excitation at 405 nm. Confluent cell monolayers in black 96-well plates were rinsed with HANKS balanced salts solution containing bovine serum albumin (0.1% w/v) and 10 mM HEPES buffer at pH 7.4 (HBSS), then incubated in HBSS for 10 minutes during which fluorescence at 535 nm with alternating excitation at 485 and 405 nm was recorded in the Envision plate reader at 1 minute repeat cycles to establish base line; test peptides were then added and fluorescence was again monitored for 90 minutes. Data were analyzed as a ratio of fluorescence intensities upon excitation at 485/405 nm over the course of 90 minutes. AUCs were calculated for the corresponding fluorescence ratio vs. time plots obtained at each ligand concentration. The role of dynamin in the PTH1R internalization response was assessed by pre-treating the cells with Dyngo4A (SelleckChem Cat# S716) at a concentration of (3 × 10−5M) or with DMSO (0.1%) as control in HBSS for 30 minutes prior to adding ligand9.
Fluorescence microscopy analysis of PTHtmr analog internalization and β-arrestin2yfp recruitment
Ligand-induced recruitment of β-arrestin2yfp was assessed in GBR-24 cells, which stably express β-arrestin2yfp 9. The cells in six-well plates were transiently transfected with plasmid DNA encoding PTH1R-WT and 24 h later seeded onto glass coverslips in 6-well plates. At 48 h post-transfection, the cells were rinsed with HBSS and treated with K13(tmr),Nle8,21-rPTH(1-34) or K13(R)palm,K14(tmr)-M-PTH(1-14), each at 100 nM, for 30 minutes, then rinsed, fixed with 3.7% paraformaldehyde (Boston BioProducts Cat. No. BM-158) and mounted on a glass microscope slide with EverBrite mounting media (Biotium Cat. No 23002) containing 4′,6-diamidino-2-phenylindole (DAPI). Slides were imaged by fluorescence microscopy using a Nikon Eclipse fluorescence microscope equipped with a CCD camera configured with SPOT imaging software. Regions of interest were digitally expanded 3-fold to improve views.
Biolayer Interferometry (BLI) Analysis of binding to bovine serum albumin (BSA)
Binding to and dissociation from BSA was assessed for PTH(1-34), Palm-PTH(1-34), M-PTH(1-14) and Palm-M-PTH(1-14) by Biolayer Interferometry using a Gator plus 8-channel analyzer (Gator Bio Inc.). The optical biosensor tips (Gator Probe Streptavidin; item No. 160002) were applied and hydrated by immersion for 30 minutes in PBS/0.05% tween20 (PBST); the tips were then immersed in biotinylated BSA (Pierce ThermoFisher-Cat# 29130) at a concentration of 10 ug/cc (0.14 uM) for 200 s, rinsed in PBST for 30 s, followed by immersion in PBST containing various concentrations (156-5000 nM) of test peptide for 300 secs (association phase) followed by immersion in PBST for 300 seconds (dissociation phase), during which times the optical signals were recorded and analyzed by the integrated analysis software. The net shifts in wavelength (nm, subtracted for background defined as the endpoint measurement) were plotted as a function of time using Graph Pad Prism. Curves were fit to the data using the Association-Dissociation non-linear regression model in Graph Pad Prism with the following constraints: HotnM = 5000, Kon and Koff = shared, and Time0 = 300.
Pharmacology studies in mice
All mice were housed in a specific pathogen-free (SPF) facility under controlled environmental conditions (temperature: 20–24 °C, humidity: 40–60%) with a 12 h light/dark cycle. Animals were provided with ad libitum access to autoclaved standard rodent chow and water. Mice were housed in groups of 3-5 five animals in individually ventilated cages (IVCs) containing autoclaved bedding and nesting material. Environmental enrichment, such as nesting material and shelters, was provided in all cages. Mice were treated in accordance with the ethical guidelines adopted by Massachusetts General Hospital, following a protocol approved by the Institutional Animal Care and Use Committee (IACUC) at MGH. Animals were housed in a temperature- and humidity-controlled vivarium with 12 h day-night light cycles. Intact CD-1 (Charles River Laboratories), PTH-Cre;Rosa-mT/mG mice31 or humanized PTH1R mice (CD-1/C57BL/6)43 were used for PTH analog injection studies. Mice were injected either subcutaneously in the interscapular region or intravenously in the tail, with either vehicle (10 mM citric acid/150 mM NaCl/0.05%Tween-80, pH 5.0) or vehicle containing a test PTH peptide formulated to provide a peptide dose ranging from 2 to 500 nmol/kg-body weight; doses are indicated in the figure legends, and were selected based on results of in vitro cAMP signaling assays presented in the figures and tables, as well as the Results of pilot and prior published studies15,31,43.
Acute calcemic responses in intact CD-1 mice
For short-term responses to a single injection, tail vein blood (40-100 ul) was collected 0.5 to 1 h prior to injection (t = 0) and at times from 0.5 to 24 h after injection into heparinized capillary tubes (MultiCap-S, Siemens Medical Solutions, USA) and ionic calcium was measured directly in whole blood upon collection using a RAPIDLabs model 348 Ca++/pH analyzer (Siemens Medical Solutions, USA).
Calcemic responses in Parathyroidectomized (PTX) mice
GFP-guided parathyroidectomy or sham surgery was performed on two-month-old PTH-Cre;Rosa-mT/mG mice31. Mice were used for PTH injection studies four days after surgery, by which time the mice exhibited significant hypocalcemia. Blood Ca++ concentrations were assessed as described above for intact mice. Mice were euthanized at 24 h after the last injection under isoflurane anesthesia, during which blood was collected from the abdominal aorta, transferred to a MiniCollect Blood Collection Tube (Gold Cap 0.8 mL, Greiner Bio-One, Cat# 450472), centrifuged at 8000xg for 15 minutes at 4°C and the serum supernatant was collected for analysis. Spot urine was collected into microfuge tubes, centrifuged at 8000xg for 15 minutes at 4°C, and the supernatant was collected.
Pharmacokinetic studies-
Blood PTH analog concentrations
10-week old female CD-1 mice were injected SC or IV with PTH(1-34) or Palm-PTH(1-34), each at a dose of 10 nmol/kg-body weight, and at serial time points ranging from 2 minutes to 24 h post-injection, blood (~15 ul) was collected from the tail vein into glass capillary tubes, transferred to a tube containing protease inhibitor cocktail (Sigma, Cat. No. P8340), centrifuged at 1000xg for 15 minutes at 4°C, and the serum supernatant was analyzed using an ELISA kit specific for human PTH(1-34) (QuidelOrtho Corp. San Diego, CA USA, Catalog No. 60-3900) calibrated with standard curves generated with dilutions of each corresponding test peptide, PTH(1-34) or Palm-PTH(1-34). Serum peptide concentrations were analyzed graphically as a function of time to derive values of AUC, Cmax (peak y) and Tmax (peak x) and fit to a monophasic decay equation to derive T1/2 values.
Urinary clearance of PTH analogs
10-week-old female CD-1 mice were injected SC injected tetramethylrhodamine (tmr)-labeled PTH(1-34)tmr and Palm-PTH(1-34)tmr was assessed by collecting spot urine at serial time points (2–24 h) after SC injection of each peptide at a dose of 50 nmol/kg/bodyweight, diluting the urine 1:7 in HB, and measuring 100 ul of each sample in a black 96-well plate in an Envision plate reader for tmr fluorescence (λex = 531 ± 25 nm, λem = 595 ± 60 nm). The fluorescence in urine collected from the mice 0.5 to one hour prior to injection (t = 0) was subtracted and the resulting net fluorescence was converted to tmr-peptide concentration using a standard curve formed with the corresponding tmr-labeled peptide.
Peptide-injection studies in humanized hPTH1R mice
Peptide were assessed in five-month-old male humanized PTH1R mice43 after a single or repeated daily injection of test peptides. Our previous study in which we generate and characterize these humanized PTH1R mice shows the basal skeletal phenotype and mineral ion physiology of these mice is not different from that of control wild-type mice43. For short-term responses to a single injection, mice were injected subcutaneously with vehicle or vehicle containing either PTH(1-34), K11(r)palm-PTH(1-11) or M-PTH(1-11), each at a peptide dose of 50 nmol/kg body weight and tail vein blood (40-100 ul) was collected 0.5–1 h prior to injection (t = 0) and at 1, 2, 4 and 6 h after injection into heparinized capillary tubes (MultiCap-S, Siemens Medical Solutions, USA) and ionic calcium was measured directly in whole blood upon collection using a RAPIDLabs model 348 Ca++/pH analyzer (Siemens Medical Solutions, USA). For repeat injection studies, humanized PTH1R mice were subcutaneously injected daily with vehicle or vehicle containing either PTH(1-34), Palm-PTH(1-34) or K11(r)palm-PTH(1-11), each at a peptide dose of 50 nmol/kg/injection, or containing K11(R)dioic-PTH(1-11) at a dose of 500 nmol/kg/injection, for three weeks. At 24 h after the last injection, mice were euthanized under isofluorane anesthesia, during which bladder urine and cardiac blood was collected. The blood was collected using a 21 G BD Vacutainer (Becton, Dickinson and Co., Cat No. 368652) into serum blood collection tubes (BD Vacutainer SST, Becton, Dickinson and Co., Cat No. 367986, centrifuged at 8000 × g for 12 minutes at 4°C and the serum supernatant was collected for analysis. Femurs were dissected at necroscopy.
Assays of biochemical markers in blood and urine
Plasma, serum and urine samples were measured for total calcium using a Calcium (CPC) LiquiColor Test kit (Stanbio Laboratory, Catalog No. 0150-250) and for inorganic phosphorus (Pi) using a Colorimetric Phosphate Assay Kit (Abcam Waltham, MA 02453, USA; Catalog No. ab65622). Serum or plasma levels of C-terminal telopeptide type I collagen (CTX-1), N-terminal propeptide of type I procollagen (P1NP) and 1,25-dihydroxyvitamin D3 were measured using EIA kits (Immunodiagnostic Systems Inc., Fountain Hills, AZ, USA; Catalog Nos. AC-06F1, AC-33F1 and AC-62F1, respectively). Endogenous PTH in serum was measured using an ELISA kit specific for mouse PTH(1-84) (QuidelOrtho Corp. San Diego, CA USA, Catalog No. No. 60-2305). Cross-reactivity of the ELISA towards human PTH(1-34) and Palm-PTH(1-34) was <1/100,000 the reactivity towards mPTH(1-84).
Microcomputed tomography (μCT) and mechanical testing of femurs
Right femurs were isolated from 5-month-old male humanized PTH1R mice after 3-weeks of daily injection and stored wrapped in PBS-saturated gauze at -20°C until analysis. Bones were analyzed using a benchtop micro-tomographic imaging system (µCT 40, Scanco Medical AG, Brüttisellen, Switzerland). Samples were scanned with a 10-µm isotropic voxel size, 70 kV peak potential (kVp), 114 µA X-ray tube intensity, and 300 ms integration time. Intramedullary trabecular bone and total volume were assessed in the distal femoral metaphysis in a region beginning at the peak of the growth plate and extending proximally 1.0 mm (100 transverse slices). Parameters at the femoral mid-shaft were assessed in a region spanning 0.5 mm (50 transverse slices) and are reported as the means of the measurements across all slices in the region. Low-density woven bone present at the mid-shaft, which was present in femurs isolated from mice treated with lipidated PTH analogs, was excluded in the measurements of cortical bone thickness and cortical bone thickness.
After analysis by μCT, the femurs were tested in three-point bending to assess their mechanical properties. Testing was performed using an electrical force materials testing machine (Electroforce 3230, Bose Corporation, Eden Prairie, MN). The bending fixture had a bottom span length of 8 mm. The test was performed with the load point in displacement control moving at a rate of 0.1 mm/sec with force and displacement data collected at 50 Hz. All bones were positioned in the same orientation during testing with the cranial surface resting on the supports and being loaded in tension. Bending rigidity (EI, N-mm2), apparent modulus of elasticity (Eapp, GPa), ultimate moment (Mult, N-mm), apparent ultimate stress (σapp, MPa) work to fracture (Wfrac, mJ), and apparent toughness to fracture (Uapp, mJ/mm3) were calculated based on the force and displacement data from the tests and, for apparent material properties, the mid-shaft geometry, as defined by the minimum moment of inertia (Imin) determined by µCT (see equations in Supplemental Fig S-9D). Work to fracture, the energy required to cause the femur to fracture, was calculated by finding the area under the force-displacement curve using the Riemann Sum method. Bending rigidity was calculated using the linear portion of the force-displacement curve.
Statistical analysis
Statistical analysis was performed by using GraphPad Prism version 8.4.3 for Mac (GraphPad Software, San Diego, CA, USA). Data are presented as the means ± standard error (SE) or standard deviation (SD) of the mean, as indicated in the figure legends. For comparison of two groups, data were analyzed by students two-sided t-test. For comparison of more than two groups, data were analyzed by one-way ANOVA followed by Dunnett’s or Bonferroni’s multiple comparison test, or by two-way ANOVA followed by Fisher’s LSD test, as indicated in the figure legends. P values < 0.05 were considered statistically significant. Box and whisker plots show individual mouse measurements as points, with whiskers indicating the range, boxes the 25th to 75th percentile, a line indicating the median and a plus indicating the mean.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Data supporting the findings of this study are available in the article and its Supplementary information. Source data are provided as Source Data file and may be obtained from the corresponding authors upon request. Source data are provided with this paper.
References
Cheloha, R. W., Gellman, S. H., Vilardaga, J. P. & Gardella, T. J. PTH receptor-1 signalling-mechanistic insights and therapeutic prospects. Nat. Rev. Endocrinol. 11, 712–724 (2015).
Kostenuik, P. J., Binkley, N. & Anderson, P. A. Advances in osteoporosis therapy: focus on osteoanabolic agents, secondary fracture prevention, and perioperative bone health. Curr. Osteoporos. Rep. 21, 386–400 (2023).
Shimizu, M. et al. Pharmacodynamic actions of a long-acting PTH Analog (LA-PTH) in thyroparathyroidectomized (TPTX) rats and normal monkeys. J. Bone Miner. Res. 7, 1405–1412 (2016).
Khan, A. A. et al. Efficacy and safety of parathyroid hormone replacement with transcon PTH in hypoparathyroidism: 26-week results from the phase 3 PaTHway trial. J. Bone Miner. Res. 38, 14–25 (2023).
Bilezikian, J. P. et al. Hypoparathyroidism. J. Clinical Endocrinol. Metab. 105, 1722–1736 (2020).
Satterwhite, J. et al. Pharmacokinetics of teriparatide (rhPTH[1-34]) and calcium pharmacodynamics in postmenopausal women with osteoporosis. Calcif. tissue Int. 87, 485–492 (2010).
Winer, K. K. Advances in the treatment of hypoparathyroidism with PTH 1-34. Bone 120, 535–541 (2019).
White, A. D. et al. Spatial bias in cAMP generation determines biological responses to PTH type 1 receptor activation. Sci. Signal. 14, eabc5944 (2021).
Portales-Castillo, I. et al. Altered signaling and desensitization responses in PTH1R mutants associated with Eiken syndrome. Commun. Biol. 6, 599 (2023).
Hoare, S. R., Gardella, T. J. & Usdin, T. B. Evaluating the signal transduction mechanism of the parathyroid hormone 1 receptor. effect of receptor-G-protein interaction on the ligand binding mechanism and receptor conformation. J. Biol. Chem. 276, 7741–7753 (2001).
Dean, T., Vilardaga, J. P., Potts, J. T. Jr & Gardella, T. J. Altered selectivity of parathyroid hormone (PTH) and PTH-related protein (PTHrp) for distinct conformations of the PTH/PTHrp receptor. Mol. Endocrinol. 22, 156–166 (2008).
Maeda, A. et al. Critical role of parathyroid hormone (PTH) receptor-1 phosphorylation in regulating acute responses to PTH. Proc. Natl Acad. Sci. USA 110, 5864–5869 (2013).
Hattersley, G., Dean, T., Corbin, B. A., Bahar, H. & Gardella, T. J. Binding selectivity of abaloparatide for PTH-Type-1-receptor conformations and effects on downstream signaling. Endocrinology 157, 141–149 (2016).
Takacs, I. et al. An open-label phase 2 study of eneboparatide, a novel PTH receptor 1 agonist, in hypoparathyroidism. J. Clin. Endocrinol. Metab. 109, 2199–2209 (2024).
Noda, H. et al. Optimization of PTH/PTHrP hybrid peptides to derive a long-acting PTH analog (LA-PTH). JBMR Plus 4, e10367 (2020).
Liu, S. et al. Altered signaling at the PTH receptor via modified agonist contacts with the extracellular domain provides a path to prolonged agonism in vivo. Proc. Natl Acad. Sci. 119, e2212736119 (2022).
Liu, S. et al. Use of backbone modification to enlarge the spatiotemporal diversity of parathyroid hormone receptor-1 signaling via biased agonism. J. Am. Chem. Soc. 141, 14486–14490 (2019).
Cheloha, R. W. et al. Development of potent, protease-resistant agonists of the parathyroid hormone receptor with broad beta residue distribution. J. Med Chem. 60, 8816–8833 (2017).
Cheloha, R. W., Maeda, A., Dean, T., Gardella, T. J. & Gellman, S. H. Backbone modification of a polypeptide drug alters duration of action in vivo. Nat. Biotechnol. 32, 653–655 (2014).
Guo, J. et al. Prolonged pharmacokinetic and pharmacodynamic actions of a pegylated parathyroid hormone (1-34) peptide fragment. J. Bone Miner. Res. 32, 86–98 (2017).
Holten-Andersen, L. et al. Design and preclinical development of transcon PTH, an investigational sustained-release PTH replacement therapy for hypoparathyroidism. J. Bone Miner. Res. 34, 2075–2086 (2019).
Gutgesell, R. M., Nogueiras, R., Tschop, M. H. & Muller, T. D. Dual and triple incretin-based co-agonists: novel therapeutics for obesity and diabetes. Diab. Ther. 15, 1069–1084 (2024).
Knudsen, L. B. & Lau, J. The discovery and development of liraglutide and semaglutide. Front Endocrinol. 10, 155 (2019).
Lau, J. et al. Discovery of the once-weekly glucagon-like peptide-1 (GLP-1) analogue semaglutide. J. Med Chem. 58, 7370–7380 (2015).
Ward, B. P. et al. Peptide lipidation stabilizes structure to enhance biological function. Mol. Metab. 2, 468–479 (2013).
Lucey, M. et al. Acylation of the incretin peptide exendin-4 directly impacts glucagon-like peptide-1 receptor signaling and trafficking. Mol. Pharm. 100, 319–334 (2021).
Willard, F. S. et al. Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight. https://doi.org/10.1172/jci.insight.140532. (2020).
Zorzi, A., Middendorp, S. J., Wilbs, J., Deyle, K. & Heinis, C. Acylated heptapeptide binds albumin with high affinity and application as tag furnishes long-acting peptides. Nat. Commun. 8, 16092 (2017).
Liu, Y. et al. Triblock peptide-linker-lipid molecular design improves potency of peptide ligands targeting family B G protein-coupled receptors. Chem. Commun. 51, 6157–6160 (2015).
Ruan, S., Yang, G., Dong, Y., Shangguan, W. & Lu, W. Discovery of a Long-Acting Parathyroid Hormone 1-34 Analogue to Treat Hypoparathyroidism. Mol. Pharm. 18, 3260–3271 (2021).
Bi, R. et al. Diphtheria toxin- and GFP-based mouse models of acquired hypoparathyroidism and treatment with a long-acting parathyroid hormone analog. J. Bone Miner. Res. 31, 975–984 (2016).
Shimizu, N., Guo, J. & Gardella, T. Parathyroid hormone (1-14) and (1-11) analogs conformationally constrained by {alpha} aminoisobutyric acid mediate full agonist responses via the Juxtamembrane region of the PTH 1 receptor. J. Biol. Chem. 276, 49003–49012 (2001).
Murrills, R. J. et al. In vitro and in vivo activities of C-terminally truncated PTH peptides reveal a disconnect between cAMP signaling and functional activity. Bone 35, 1263–1272 (2004).
Dean, T. et al. Mechanisms of ligand binding to the parathyroid hormone (PTH)/PTH-related protein receptor: selectivity of a modified PTH(1-15) radioligand for GalphaS-coupled receptor conformations. Mol. Endocrinol. 20, 931–943 (2006).
Ehrenmann, J. et al. High-resolution crystal structure of parathyroid hormone 1 receptor in complex with a peptide agonist. Nat. Struct. Mol. Biol. 25, 1086–1092 (2018).
Zhao, L.-H. et al. Structure and dynamics of the active human parathyroid hormone receptor-1. Science 364, 148–153 (2019).
Kobayashi, K. et al. Endogenous ligand recognition and structural transition of a human PTH receptor. Mol. Cell 82, 3468–3483.e3465 (2022).
Cary, B. P. et al. Molecular insights into peptide agonist engagement with the PTH receptor. Structure. https://doi.org/10.1016/j.str.2023.04.002. (2023).
Finan, B. et al. A rationally designed monomeric peptide triagonist corrects obesity and diabetes in rodents. Nat. Med. 21, 27–36 (2015).
Wang, X. et al. Molecular insights into differentiated ligand recognition of the human parathyroid hormone receptor 2. Proc. Natl. Acad. Sci. 118, https://doi.org/10.1073/pnas.2101279118 (2021).
Bouchet, A., Lairion, F. & Disalvo, E. A. Role of guanidinium group in the insertion of l-arginine in DMPE and DMPC lipid interphases. Biochimica et. Biophysica Acta (BBA) - Biomembranes 1798, 616–623 (2010).
Sato, T. et al. Comparable initial engagement of intracellular signaling pathways by parathyroid hormone receptor ligands teriparatide, abaloparatide, and long-acting PTH. JBMR Plus 5, e10441 (2021).
Daley, E. J. et al. Actions of parathyroid hormone ligand analogues in humanized PTH1R knockin mice. Endocrinology 163, https://doi.org/10.1210/endocr/bqac054 (2022).
Martin, T. J., Sims, N. A. & Seeman, E. Physiological and pharmacological roles of PTH and PTHrP in bone using their shared receptor, PTH1R. Endocr. Rev. 42, 383–406 (2021).
Knudsen, L. B. et al. Potent derivatives of glucagon-like peptide-1 with pharmacokinetic properties suitable for once daily administration. J. Med Chem. 43, 1664–1669 (2000).
Campbell, J. E. et al. GIPR/GLP-1R dual agonist therapies for diabetes and weight loss—chemistry, physiology, and clinical applications. Cell Metab. 35, 1519–1529 (2023).
Tay, D., Cremers, S. & Bilezikian, J. P. Optimal dosing and delivery of parathyroid hormone and its analogues for osteoporosis and hypoparathyroidism - translating the pharmacology. Br. J. Clin. Pharm. 84, 252–267 (2018).
Hall, D. B., Kostyla, C. H., Hales, L. M. & Soliman, T. M. Preclinical development of EXT608, an investigational parathyroid hormone derivative with extended half-life for the treatment of hypoparathyroidism. JBMR Plus 8, ziae045 (2024).
Levine, P. M. et al. O-GlcNAc engineering of GPCR peptide-agonists improves their stability and in vivo activity. J. Am. Chem. Soc. 141, 14210–14219 (2019).
Shimizu, M., Carter, P. H., Khatri, A., Potts, J. T. Jr & Gardella, T. J. Enhanced activity in parathyroid hormone-(1-14) and -(1-11): novel peptides for probing ligand-receptor interactions. Endocrinology 142, 3068–3074 (2001).
Zhai, X. et al. Molecular insights into the distinct signaling duration for the peptide-induced PTH1R activation. Nat. Commun. 13, 6276 (2022).
Zhao, L.-H. et al. Molecular recognition of two endogenous hormones by the human parathyroid hormone receptor-1. Acta pharmacologica Sinica. https://doi.org/10.1038/s41401-022-01032-z (2022).
Zhao, F. et al. Structural insights into multiplexed pharmacological actions of tirzepatide and peptide 20 at the GIP, GLP-1 or glucagon receptors. Nat. Commun. 13, 1057 (2022).
Li, Y. et al. Structural analysis of the dual agonism at GLP-1R and GCGR. Proc. Natl Acad. Sci. 120, e2303696120 (2023).
Zhang, X. et al. Structure and dynamics of semaglutide- and taspoglutide-bound GLP-1R-Gs complexes. Cell Rep. 36, 109374 (2021).
Sun, B. et al. Structural determinants of dual incretin receptor agonism by tirzepatide. Proc. Natl Acad. Sci. 119, e2116506119 (2022).
Pasieka, J. L. et al. Etiology and pathophysiology of hypoparathyroidism: a narrative review. J. Bone Miner. Res. 37, 2586–2601 (2022).
Carney, P., Cutler, G. B. Jr., Schneider, K., Zhang, F. & DiMarchi, R. MBX 2109, a Once-weekly parathyroid hormone replacement therapy prodrug: phase 1, first-in-human, randomized trial. J. Clin. Endocrinol. Metab. 110, 940–950 (2024).
Acknowledgements
The authors thank Yingshe Zhao and Marie Demay of the MGH Center for Skeletal Research Imaging and Biomechanical Testing Core (NIH P30 AR070542) for assistance with histological measurements; Ruiye Bi, Monica Reyes, Aaria Prakash, Johanna Schmitt and Sung-Hee Yoon of the MGH Endocrine Unit for assistance with mouse experiments, and Greg Gipson of the MGH Cardiovascular Research Center for assistance with BLI experiments. Work at MGH was supported by NIH grants P01 DK011794, R01 DK113039, to TJG and HJ, and P30 AR075042 to TJG and MLB. JH was supported by a scholarship from the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG). Work at UW-Madison was supported by NIH grant R01 GM056414 and its successor, R35 GM151985 to SHG. RWC was supported in part by Biotechnology Training Grant T32 GM008349.
Author information
Authors and Affiliations
Contributions
T.J.G., H.J., A.K., R.W.C. and S.H.G. conceived and planned experiments. J.H., H.N., A.K., A.K.A., R.W.C., T.D., M.B. and D.J.B. carried out experiments. M.M. and M.L.B. contributed to interpretation of results. J.H. and T.J.G. wrote the initial manuscript. All authors provided critical feedback on data analysis and manuscript preparation and helped shape the research.
Corresponding author
Ethics declarations
Competing interests
HN, AK and TJG are inventors on a patent relating to lipid-modified PTH analogs (US patent # 11975050). The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks David Goltzman, Elena Tsourdi, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Höppner, J., Noda, H., Anitha, A.K. et al. Prolonging parathyroid hormone analog action in vitro and in vivo through peptide lipidation. Nat Commun 16, 4487 (2025). https://doi.org/10.1038/s41467-025-59665-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-59665-7