Abstract
FGF receptors play pivotal roles in morphogenetic processes, including vertebrate neurulation. Planar cell polarity signaling coordinates cell orientation in the tissue plane and is essential for neural tube closure. Here, we demonstrate abnormal planar polarity in the Xenopus neuroectoderm depleted of FGFR1, suggesting a mechanistic connection between FGFR signaling and morphogenesis. FGFR1 associates with the core planar cell polarity protein Vangl2, leading to its phosphorylation at N-terminal tyrosines, a modification also induced by FGF8. Vangl2 phosphorylation requires FGFR1 activity in Xenopus embryos and mouse embryonic stem cells, extending our observations to mammals. A non-phosphorylatable Vangl2 construct exhibits increased binding to the receptor tyrosine kinase PTK7, suggesting a potential role of Vangl2 phosphorylation. By contrast, a phosphomimetic Vangl2 mutant shows reduced interactions with Prickle and PTK7, and disrupted planar polarity in the neuroectoderm. Together, these findings identify cross-talk between the FGFR1 and planar cell polarity pathways mediated by Vangl2 tyrosine phosphorylation.
Similar content being viewed by others
Introduction
The FGF receptor pathway is known to orchestrate cell specification and tissue patterning during embryogenesis. In addition to the regulation of cell fate, cell proliferation and differentiation, FGFR signaling has been implicated in the morphogenetic processes that involve all germ layers1,2,3,4,5,6. In vertebrates, the pathway consists of eighteen FGF ligands and four receptors from the receptor tyrosine kinase (RTK) superfamily. Upon stimulation, the FGFRs dimerize and the tyrosine kinase domains cross-phosphorylate each other, leading to the activation of downstream effectors such as ERK, PI3K, PLCγ, and STAT17,8,9. Although the ERK pathway is considered a principal transducer of FGFR activity in the embryo, FGFR-dependent developmental signaling has been proposed to depend on additional yet unidentified molecular targets. The knocked-in fgfr1 gene with mutated binding sites for the known mediators of FGF signaling fails to phenocopy the null mutant phenotype in mice, indicating that FGFR1 must possess still uncharacterized modes of signaling10,11.
The planar cell polarity (PCP) pathway coordinates cell alignment in the tissue plane12,13,14 and is required for many morphogenetic events in vertebrate embryos. Initially discovered in Drosophila, ‘core PCP’ components include the transmembrane proteins Van Gogh (Vang), Frizzled (Fz), and Flamingo/CELSR, as well as the cytoplasmic proteins Prickle (Pk) and Disheveled (Dvl). Due to feedback regulation, Vangl/Pk and Fzd/Dvl complexes segregate to opposite cell boundaries15,16,17,18,19,20,21. In vertebrates, Vang-like genes (Vangl) play important roles in neural tube morphogenesis22,23,24,25,26,27,28. Mouse studies have demonstrated genetic interactions of Vangl2 with other genes implicated in PCP, such as Wnt5, Ror2, Scribble, Celsr1 and the inactive receptor tyrosine kinase Ptk729,30,31.
In Xenopus early embryos, the enrichment of Vangl2 at the anterior surface of each cell is first evident in the posterior neural plate and subsequently extends anteriorly during neurulation, suggesting a posterior origin of the PCP-instructing signal32,33. Because of the posterior expression and activity of several FGF ligands during neurulation34,35,36,37, we asked whether the FGFR pathway plays a role in the establishment of PCP. In this study, we uncover a vertebrate-specific tyrosine phosphorylation of Vangl2 in response to FGFR1 and demonstrate a requirement of FGFR signaling for PCP in the Xenopus neural plate. Mechanistically, we find that the phosphorylation inhibits the association of Vangl2 with Pk3 and Ptk7, an RTK family pseudokinase that genetically interacts with Vangl2 in neural tube closure31,38,39.
Results
Requirement of FGFR1 for neural plate planar polarity
FGF signaling is required for anteroposterior patterning in the neural plate, neural induction34,35,40,41,42,43 and neural tube closure2. Whether these functions involve distinct pathway targets and whether a separate signaling branch is responsible for the control of collective cell behaviors remains to be determined.
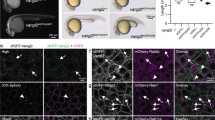
To investigate a potential link between FGFR1, a commonly expressed FGF receptor44,45, and the core PCP pathway, we examined the distribution of the Vangl2 protein in the plane of the Xenopus neural plate of embryos injected with a previously characterized FGFR1 morpholino oligonucleotide (MO)46. We found that injection of FGFR1 MO caused severe neural tube defects that were partially rescued by MO-resistant mouse FGFR1 construct (Supplementary Fig. 1). To avoid global effects on morphogenesis, we generated GFP-marked mosaic clones of FGFR1-morphant cells. Vangl2 was enriched at the anterior edges of the uninjected and control MO-injected neuroepithelial cells as reported previously32 (Fig. 1A, B). By contrast, the cells containing FGFR1 MO lacked the typical Vangl2 polarity that is characteristic of PCP (Fig. 1C, D; Supplementary Fig. 2). We did not observe significant effects of FGFR1 MO on the expression of the pan-neural marker Sox3, suggesting that cell specification was largely intact (Supplementary Fig. 3). Inhibition of the anterior enrichment of endogenous Vangl2 (Supplementary Fig. 4) and exogenous complexes of Vangl2 and Pk332,47,48 (Supplementary Fig. 5) has been confirmed using a dominant-negative FGFR1 construct (XFD)44. These observations suggest a role for FGFR1 signaling in establishing PCP in the neural plate.
A Experimental scheme. Adapted from Xenopus illustrations © Natalya Zahn (2022), via Xenbase (www.xenbase.org, RRID: SCR_003280). Thirty-two-cell Xenopus embryos were co-injected with 5 nl of control (Co) or FGFR1 morpholino (MO), 15 ng each, along with GFP RNA (90 pg) as a lineage tracer into one dorsal animal blastomere. Embryos were collected at stage 15 (st.15) and co-immunostained for Vangl2 (red) and GFP (green). The anteroposterior (AP) axis is indicated. Representative en face neural plate images are shown in (B) and (C), white box areas are magnified on the right to show merged (green+red) and single (red) channel images. B Control neuroectoderm with anteriorly polarized Vangl2 (arrows). C FGFR1MO-injected neuroectoderm lacking anterior Vangl2 accumulation (asterisks). D Quantification of GFP-positive cells with anteriorly enriched Vangl2 in CoMO- and FGFR1MO- injected neuroectoderm. Data are presented as a dot plot with horizontal lines indicating the mean. Each dot represents the percentage of polarized cells per embryo, with three embryos per group and 50–90 cells scored per embryo. Total numbers of cells (n) analyzed per group are indicated above each condition. Data are representative of four independent experiments, p = 0.00074, two-tailed unpaired Student’s t-test. Scale bar, 30 µm.
In addition to early neuroectoderm, we examined the distribution of Prickle2 (Pk2) that exhibits planar polarization at posterior edges of tailbud skin cells49. We find that FGFR signaling is required for Pk2 planar polarity in the skin (Supplementary Fig. 6), suggesting that this pathway may have a broad role in PCP establishment.
Tyrosine phosphorylation of Vangl2 in response to FGF receptor activation
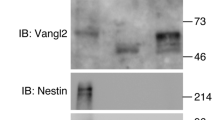
Although FGF ligand has previously been reported to modulate the polarization of Vangl2 in the developing mouse limb50, the mechanistic link between FGFR signaling and PCP remains unclear. We therefore asked whether endogenous FGFR1 might regulate Vangl2 tyrosine phosphorylation in vivo. Notably, the phosphorylation can be detected in untreated ectodermal explants of stage 12 embryos (Fig. 2A, B). The signal was reduced by the XFD construct (Fig. 2B, Supplementary Fig. 7), by FGFR1 MO (Fig. 2C) and after the treatment of the explants with the chemical inhibitor of FGFR activity SU540251 (Supplementary Fig. 8), indicating that this phosphorylation event requires endogenous FGFR1 function in Xenopus embryos. This conclusion was further supported by experiments using FGFR1/2 double knockout mouse embryonic stem (ES) cells52 (Fig. 2D–F). The latter result establishes FGFR1/2 as a significant contributors to Vangl2 phosphorylation in mouse ES cells, extending our findings to the mammalian model.
A–C FGFR signaling inhibition decreases Vangl2 tyrosine phosphorylation in Xenopus embryos. A experimental scheme for (B–C). Adapted from Xenopus illustrations © Natalya Zahn (2022), via Xenbase (www.xenbase.org, RRID: SCR_003280). Ectoderm explants were dissected from stage 9 embryos and used for pull-down with Vangl2 antibody. B Explants were dissected from embryos injected with HA-Vangl2 RNA (40 pg), with or without dominant-interfering FGFR1 (XFD) and cultured until stage 12. C Reduction of endogenous Vangl2 tyrosine phosphorylation in ectoderm explants (stage 16) depleted of FGFR1 with FGFR1 MO (20 ng). D–F Vangl2 tyrosine phosphorylation depends on FGFR signaling activity in mouse embryonic stem (ES) cells. D Experimental scheme. E, F Lysates from wild-type and FGFR1/2 double knockout mouse ES cells were precipitated with anti-Vangl2 (E) or anti-phosphotyrosine (pY) (F) antibodies and immunoblotted as indicated. Cyclin B1 is a negative control (F). Experiments were reproduced twice (B, E), three times (C).
To evaluate whether FGFR1 is directly connected to core PCP proteins, we tested whether FGFR1 physically associates with Vangl2. For both exogenous and endogenous proteins, we found that FGFR1 co-precipitates with Vangl2 (Fig. 3A, Supplementary Fig. 9), consistent with crosstalk between the two pathways. We next assessed a potential mechanism of PCP modulation by FGFR1. Vangl2 is known to be phosphorylated by Casein kinase I on serine/threonine residues in response to Wnt and Frizzled signaling, leading to reduced gel mobility and altered functional activity29,32,53,54,55,56. More recently, tyrosine phosphorylation of Vangl2 has also been reported57,58. We asked whether Vangl2 is phosphorylated at tyrosine residues in embryos expressing exogenous FGFRs. A strong signal was detected by anti-phosphotyrosine (pY) antibody in Vangl2 pulldowns from lysates of embryos co-expressing Vangl2 and FGFR1, FGFR2, FGFR4 and constitutively active FGFR1Y372 > C but not those co-expressing Vangl2 and Frizzled3 (Fig. 3B, Supplementary Fig. 10). These findings reveal a physical association between FGFR1 and Vangl2 and show that overexpressed FGFRs can stimulate Vangl2 phosphorylation.
Four- to eight-cell Xenopus embryos were injected with RNAs for GFP-Vangl2, PTK7-GFP and FGFR1-FLAG (100 pg each) (A) or RNAs for HA-Vangl2 (50 pg) and either FGFR1-FLAG (40 pg) or Frizzled3-FLAG (400 pg) (B), lysed at stage 12 and subjected to immunoprecipitation (IP) with anti-FLAG to assess FGFR1-Vangl2 binding (A) or anti-Vangl2 to assess Vangl2 tyrosine phosphorylation (B). PTK7-GFP is a negative control in (A). Frizzled3-dependent Vangl2 band shift in (B) reflects S/T phosphorylation54. An irrelevant portion of the membrane was removed. C FGF8 induces Vangl2 phosphorylation in Xenopus embryos. Embryos were injected with HA-Vangl2 with or without FGF8 plasmid DNAs (50 pg each), lysed at stage 14 and precipitated with HA-trap, followed by immunoblot with anti-pY antibodies. Experiments were reproduced four times (A), ten times (B) and three times (C).
Together, our findings point to Vangl2 as a molecular target of the FGFR pathway and demonstrate that FGFR is both necessary and sufficient for Vangl2 tyrosine phosphorylation in early embryos.
Lack of correlation between ERK activation and Vangl2 phosphorylation and a role for FGF ligands
An important question is whether FGFR signals to Vangl2 via a mechanism distinct from its signaling to the well-known mediator ERK7. Another critical issue is which FGF ligand activates Vangl2 phosphorylation in response to FGFRs. We compared the induction of Vangl2 tyrosine phosphorylation and phospho-ERK1/2 (active ERK) in ectodermal explants in response to FGFR and FGF2. FGFR1 triggered a dose-dependent increase in Vangl2 tyrosine phosphorylation, whereas only a weak synergy was observed between FGF2 and FGFR1, and FGF2 alone had no effect (Supplementary Fig. 8 and Supplementary Fig. 11). By contrast, FGF2 strongly synergized with FGFR1 in upregulating phospho-ERK1/2. Both phosphorylation events were sensitive to SU5402, indicating dependence on FGFR activity (Supplementary Fig. 11).
While searching for a potential inducer of Vangl2 tyrosine phosphorylation among the FGF ligands present during neurulation, we found that FGF8 can stimulate Vangl2 tyrosine phosphorylation (Fig. 3C). Since FGF8 is expressed in the posterior neural plate36,37, it is a good candidate for the ligand stimulating FGFR1 signaling to Vangl2.
We next tested FGFR1FCPG, a mutated FGFR1 construct lacking the binding sites for known signaling intermediates and deficient in the ability to activate most of the known FGFR1 signaling pathways10. FGFR1FCPG did not activate ERK, consistent with a previous study10, but induced Vangl2 tyrosine phosphorylation even more efficiently than wild-type FGFR1 (Fig. 4). This observation strengthens our hypothesis that Vangl2 is phosphorylated via a noncanonical pathway.
Four-to-eight cell embryos were injected into four animal blastomeres with GFP-Vangl2 RNA, FGFR1-FLAG or FGFR1FCPG-FLAG RNA (40 pg each), and were cultured until stage 12. GFP-Vangl2 was immunoprecipitated (IP) from embryo lysates using GFP-trap beads, followed by immunoblotting (IB) with anti-pY, anti-GFP, anti-FLAG, anti-pERK1/2 and anti-ERK1/2 antibodies. FGFR1FCPG containing mutations in the binding sites of known signaling mediators (see text) induced Vangl2 tyrosine phosphorylation but did not upregulate ERK1/2 activity. Experiment was reproduced three times.
Vangl2 N-terminal tyrosine phosphorylation inhibits planar polarity
To identify specific Vangl2 amino acid residues that become phosphorylated in response to FGFR1, we replaced tyrosine residues with phenylalanines. RNAs encoding FGFR1 and Vangl2 mutants with Y > F substitutions were co-injected into Xenopus embryos, and the phosphorylation was analyzed in HA-Vangl2 pulldowns using anti-pY antibody. The triple substitution of Y7, Y10, Y12 to phenylalanine strongly decreased FGFR1-induced Vangl2 tyrosine phosphorylation, while the Y341, Y342, Y343 substitutions had only a minor effect (Fig. 5A). Further mapping confirmed that Y10 and Y12 are essential and that the ‘kinase-dead’ form of FGFR1 (D623 > A) is completely inactive in this assay (Supplementary Fig. 12). Notably, the equivalent of the tyrosine phosphorylation site identified in Drosophila Van Gogh57 was not phosphorylated in this experiment (Y308, Supplementary Fig. 12). Our findings indicate that the N-terminal tyrosines Y10 and Y12 are the dominant sites in Vangl2 that are phosphorylated in response to FGFR1. The alignment of the N-terminal amino acid sequence shows that the identified sites are conserved among vertebrates (Fig. 5B).
A Mapping major phospho-tyrosine (pY) sites in Vangl2. Embryos were injected with 40 pg of RNA encoding FGFR1 and the indicated HA-Vangl2 constructs. Vangl2 phosphorylation was analyzed in anti-HA pull-downs from stage 13 embryo lysates. Immunoblotting (IB) was performed using anti-pY, anti-HA or anti-FLAG antibodies, as indicated. One representative set of duplicate samples is shown. The graph below shows average pY/HA intensity ratios for the duplicates. B Alignment of N-terminal amino acid sequences of Vangl2 from several chordate species and Drosophila Van Gogh. The N-terminal tyrosine cluster (in red) is conserved in vertebrates but not in Drosophila. C–E Planar polarization of Vangl2 tyrosine phosphosite mutants in the neuroectoderm. C Experimental scheme. Adapted from Xenopus illustrations © Natalya Zahn (2022), via Xenbase (www.xenbase.org, RRID: SCR_003280). Two dorsal blastomeres of 16-cell embryos were coinjected with myr-BFP RNA (80 pg) and RNAs encoding GFP-Pk3 (150 pg), HA-tagged Vangl2 (in D), Y7, 10, 12 > F (Y > F, in E) or Y7, 10, 12 > E (Y > E, in F) (20 pg each). Embryos were fixed at stages 14–15, and GFP and BFP fluorescence were imaged. Anterior enrichment of GFP-Pk3 is indicated by arrows (D–E’), while a non-polarized cell is marked by an asterisk (F–F’). The anteroposterior (AP) axis is indicated. Scale bar, 30 µm. (G) Quantification of GFP fluorescence for the GFP-Pk3-Vangl2 complexes in mosaically expressing cells is shown as a graph. Mean fluorescence is plotted along the cell circumference as a function of circular angle from 0 to 360 degrees relative to the AP axis, with fluorescence intensity shown in green (HA-Vangl2), pink (HA-Vangl2Y > F), and brown (HA-Vangl2Y > E) lines. The number of scored cells is indicated. The data are representative of four independent experiments.
We next asked whether the observed Vangl2 tyrosine phosphorylation may influence the phosphorylation at previously reported Cluster 1 and Cluster 2 serine/threonine sites29. Using antibodies specific for phospho-T78, S79, S82 and phospho-S14, S17 peptides, we observed reduced phospho-S14, S17-Vangl2 levels (Cluster 2) in the Y7,10,12 > F Vangl2 construct whereas the phosphorylation at the Cluster 1 was not affected (Supplementary Fig. 13). This finding indicates that tyrosine phosphorylation of Vangl2 promotes serine phosphorylation in the adjacent Cluster 2 sites.
Our subsequent experiments focused on the physiological significance of posttranslational modifications at these sites. To assess the ability of unphosphorylatable or phosphomimetic Vangl2 proteins to associate with other PCP proteins, we studied their interactions with Prickle, a known component of the anterior PCP complex32,47,48. RNAs encoding Pk3 and mutated Vangl2 constructs were co-injected into dorsal blastomeres at the 16-cell stage at doses that did not affect normal morphogenesis (Fig. 5C). Both wild-type and nonphosphorylatable Vangl2 constructs enabled anterior enrichment of Pk3 in neuroepithelial cells, while the phosphomimetic Vangl2 mutant did not. Thus, the N-terminal tyrosine phosphorylation of Vangl2 inhibits the anterior accumulation of Pk3 in the neuroectoderm (Fig. 5D–G).
We also tested whether Vangl2 tyrosine phosphorylation regulates the Vangl2-Pk3 association. This was carried out using a modified proximity biotinylation assay59. We observed reduced interaction of phosphomimetic Vangl2 with Pk3 as compared to the wild-type and nonphosphorylatable Vangl2 constructs (Fig. 6). These results suggest that Vangl2 tyrosine phosphorylation decreases the amount of the Vangl2-Pk3 complex, likely reducing the signaling activity of Vangl2.
A Schematic for the proximity biotinylation assay to assess the interaction between Vangl2 and Prickle3 (Pk3). Vangl2 is biotinylated (asterisks) when in proximity to the Pk3 fused to the large N-terminal fragment of a bacterial biotin ligase (BLN). B Animal blastomeres of four- to-eight-cell stage embryos were co-injected with 100 pg of FLAG-BLN-Pk3 RNA and 40 pg of HA-Vangl2, HA-Vangl2 Y7, 10, 12 > F (Y > F), or HA-Vangl2 Y7, 10, 12 > E (Y > E) RNAs. At stage 9, 20 nl of biotin (0.8 mM) was injected into the blastocoel. Embryos were collected at stage 13, and Vangl2 constructs were pulled down using anti-HA antibody. Biotinylation of Vangl2 and protein expression levels were assessed in independent duplicate samples using anti-biotin, anti-HA, and anti-FLAG antibodies. Data represent three independent experiments.
The significance of Vangl2 tyrosine phosphorylation for PCP has been expanded to the complex of Vangl2 and the receptor tyrosine kinase PTK7, a transmembrane protein that genetically interacts with Vangl231 and functions in PCP via an unknown mechanism31,38,39,60. We observed that wild-type Vangl2 co-precipitated with PTK7 in lysates of stage 12 Xenopus embryos (Fig. 7A, B). Notably, this complex formed more efficiently with nonphosphorylatable Vangl2, whereas it was strongly reduced with the phosphomimetic Vangl2 construct (Fig. 7A, B), arguing that Vangl2 phosphorylation may control the Vangl2-PTK7 association.
A, B Vangl2-PTK7 binding is enhanced by non-phosphorylated Vangl2 and reduced by phosphorylated Vangl2. Four-to-eight-cell Xenopus embryos were injected with RNAs encoding HA-Vangl2, HA-Vangl2 Y7Y10Y12 > F (Y > F) or HA-Vangl2 Y7Y10Y12 > E (Y > E) (40 pg each) as indicated, with or without PTK7-GFP RNA, 100 pg, into four animal blastomeres. Embryos were collected at stage 12 and PTK7-GFP (A) or HA-Vangl2 (B) were immunoprecipitated using GFP-trap or anti-HA antibody, respectively, to assess co-precipitated Vangl2 (A) or PTK7 (B). Protein levels in pull-downs and lysates were analyzed with anti-GFP and anti-HA antibodies. Irrelevant part of the membrane was removed (A). Data are representative of three experiments.
To address the significance of this observation, we asked whether PTK7 can functionally synergize with Vangl2 in the induction of neural tube defects. Notably, strong synergy was observed when PTK7 was coexpressed with nonphosphorylatable Vangl2 but not phosphomimetic Vangl2, pointing to the physiological role of Vangl2-PTK7 complex in the neural plate morphogenesis (Fig. 8A–D). Together, these experiments highlight a role of the Vangl2-PTK7 complex in PCP.
Neural folding defects were assessed in embryos injected with RNAs encoding PTK7 and different forms of Vangl2 (200 pg each) into two dorsal animal blastomeres (inset in A, red arrows). Dorsal views of representative embryos exhibit no A, mild B or severe C disruption of neural tube closure. Co-injection of PTK7 and Y > F Vangl2 RNAs causes stronger neural tube defects (NTDs) compared to the co-injection of the wild type or Y > E Vangl2 RNAs. The degree of neural tube closure was scored at stage 17 by the distance between the opposing neural folds near the brain-spinal cord border (arrowheads), anterior is to the top. Dashed line marks the midline. D Quantification showing frequencies of normal neural folds A, as compared to mild B and severe C defects. The number of scored embryos is shown in the graph. Data represent two independent experiments. E Model. Graded FGF/FGFR1 activity triggers Vangl2 tyrosine phosphorylation at posterior cell edges, inhibiting the formation of Vangl2-Pk3 and Vangl2-PTK7 complexes, leading to reduced posterior accumulation of Vangl2. PTK7 is proposed to act as a feedback regulator and FGFR1 antagonist, promoting Vangl2 dephosphorylation through phosphatase (PPase) recruitment.
Discussion
This work demonstrates that FGFR1 physically associates with Vangl2 and triggers its tyrosine phosphorylation. The lack of correlation between Vangl2 phosphorylation and ERK activation points to distinct signaling branches downstream of FGFR1. We propose that FGFR1 tyrosine kinase that is activated by an FGF ligand, such as posteriorly expressed FGF8, binds and directly phosphorylates Vangl2 in a spatially restricted manner (Fig. 8E). In the absence of additional evidence, we cannot formally exclude the involvement of other protein tyrosine kinases. It is also possible that the same Vangl2 tyrosine phosphorylation sites serve as substrates for different RTKs expressed in various embryonic tissues58. The requirement of FGFR1/2 signaling for Vangl2 phosphorylation in Xenopus embryos and mouse ES cells and the demonstration of its importance for PCP in both the neuroectoderm and the tadpole skin underscore the conservation of this signaling event in different embryonic tissues in vertebrates.
The anterior enrichment of endogenous Vangl2 protein is lost in the FGFR1-depleted neuroectoderm, suggesting an important role of FGFR1 in PCP. We found that the interaction of Pk3 with phosphomimetic Vangl2 assessed by proximity biotinylation was reduced, and the complex of Pk3 with phosphomimetic Vangl2 was not anteriorly polarized. These findings are consistent with N-terminal tyrosine phosphorylation preventing the formation of Vangl2-containing PCP complexes at the posterior edges of neuroepithelial cells (Fig. 8E). Our finding that phospho-S14, S17-Vangl2 levels are reduced in the phosphomutant Vangl2 construct indicates that the phosphorylation of Vangl2 at Cluster 2 residues mediates cross-talk between the FGF and PCP pathways during embryonic patterning and morphogenesis29,55,56. Future studies will define other biological processes affected by FGFR-dependent Vangl2 phosphorylation, including Vangl2 membrane trafficking55,61, lipidation62 or the regulation of Vangl2 degradation63,64.
Based on our analysis of Vangl2 phosphosite mutations, we expected to see enhanced planar polarity in FGFR1 knockdown experiments, but instead, we observed PCP loss. To explain this inconsistency, we propose that FGFR1 has another molecular target besides Vangl2. The observed loss of PCP after FGFR1 depletion could be due to its combined positive and negative effects on more than one target. Additionally, Vangl2 phosphorylation might affect PCP not only via the Prickle complex but through other PCP proteins, such as PTK7. Indeed, although the nonphosphorylatable Vangl2 behaved similarly to the wild-type when tested for the planar polarization of anterior Vangl2-Pk3 complexes, we discovered that it strongly bound PTK7, another planar polarity component. By contrast, the association of PTK7 with phosphomimetic Vangl2 was barely detectable, indicating that Vangl2 binds PTK7 in a phosphorylation-dependent manner.
Our findings extend previous reports of the genetic interactions between Vangl2 and PTK7 during mammalian neural tube closure31, and point to a mechanistic role of PTK7 in PCP regulation. Currently, it remains unknown how PTK7 and Vangl2 modulate each other. Importantly, the synergistic effect of Vangl2 and PTK7 on neural tube closure correlated with the phosphorylation status of N-terminal tyrosines in Vangl2, underscoring the physiological significance of this phosphorylation. Searching protein interaction databases such as BioGRID, we found multiple protein tyrosine phosphatases associated with PTK7. We propose that PTK7 functions in PCP as a feedback regulator, because it can facilitate Vangl2 phosphate removal by PTK7-associated phosphatases or compete with FGFR for Vangl2 binding. Additional loss-of-function experiments are needed to understand the function of PTK7 in PCP.
Methods
Plasmids and mRNA synthesis
Plasmids encoding Xenopus GFP- and HA-tagged Vangl248, mouse FGFR1-3xFLAG, FGFR1FCPG-3xFLAG10 and FGFR2-3xFLAG52, human FGFR1, FGFR1Y372 > C and FGFR465, dominant-interfering-FGFR1 (XFD)44, GFP32, mCherry59, Fz3-FLAG66, FLAG-BLN-tagged Pk359, pCS107-GFP-Pk249, pCS2-FGF837 have been described previously. pCS2-PTK7-GFP was a gift of Annette Borchers. The pCS2-myristoylated BFP-HA and the corrected GFP-Pk3 construct48, fitting the predicted protein sequence, were gifts of Miho Matsuda. HA-tagged Y > F and Y > E point mutants of Vangl2 and kinase-dead FGFR1D623 > A mutant67 were generated using gBlock fragments (Integrated DNA Technologies). The XFD-3xFLAG construct was generated by subcloning a PCR-amplified fragment encoding 3xFLAG and the SV40 polyadenylation sequence from the pCS107-FGFR1-3xFLAG plasmid10. The fragment was amplified using the forward primer 5′-CGGTCTAGAACCGGTGATTACAAGGATGACGACGAT-3′ and reverse primer 5′-TTTGAATTCAAAAGCTGGGTACCGGGCC-3′, and inserted at the C-terminal sequence of the dominant-negative FGFR1 (XFD) construct44. Capped mRNAs were synthesized using mMessage mMachine kit (Ambion, Austin, TX).
Xenopus embryo culture and microinjections
The study was conducted in strict accordance with the recommendations outlined in the Guide for the Care and Use of Laboratory Animals by the National Institutes of Health. The animal protocol received approval from the Institutional Animal Care and Use Committee (IACUC) at the Icahn School of Medicine at Mount Sinai. Xenopus laevis eggs were in vitro fertilized and cultured in 0.1x Marc’s Modified Ringer’s solution (MMR)54. For microinjections, 4–16 cell embryos were transferred to a 3% Ficoll 400 solution (GE Healthcare) in 0.6x MMR and were injected with 5–10 nl of a solution containing mRNAs or morpholino. To achieve mosaic expression of PCP complexes or FGFR1-depletion in the neural plate, the embryos were injected into two dorsal blastomeres of 16–32-cell embryos. The quantities of injected mRNAs were optimized in preliminary dose-response experiments, and the specific amounts are indicated in the figure legends.
Immunostaining, fluorescent protein detection, imaging and quantification
To identify PCP complexes in the neural plate, embryos were collected at stage 14, and the vitelline membrane was manually removed. RNAs encoding GFP-Pk3, HA-Vangl2 wild type and mutated Y7Y10Y12 > F or Y7Y10Y12 > E constructs and myr-BFP were co-injected at the established doses (as specified in the figure legends), which did not have an impact on normal development as was established in preliminary experiments. Embryos were fixed in MEMFA solution for 40 min. Neural plate explants were then dissected, mounted in the Vectashield mounting medium (Vector), and GFP-Pk3 and BFP fluorescence was detected and scored.
For detection of endogenous Vangl2, embryos were collected at stages 15-16, fixed in 2% TCA for 30 min, and stained with the rabbit polyclonal anti-Vangl2 antibody (1:100)32. Sox3 was detected with mouse monoclonal Sox3 antibody (1:100; DSHB, DA5H6). For tracing FGFR1-depleted cells, embryos were co-injected with 15 ng of FGFR1MO and 90 pg of GFP RNA. Mouse monoclonal (Santa Cruz, GFP-B2) or rabbit polyclonal (Invitrogen, A6544) anti-GFP antibodies were used for co-immunostaining at 1:100 and 1:500 dilutions, respectively.
Secondary antibodies were against mouse or rabbit IgG conjugated to Cy2 or Cy3 (1:500, Jackson ImmunoResearch). Standard specificity controls were conducted to verify the absence of cross-reactivity and to ensure that no staining occurred in the absence of primary antibodies.
Images of whole neural plate explants were acquired using tiling and subsequent stitching at the BC43 (Andor) confocal microscope. The quantification of planar polarity of the HA-Vangl2 constructs in complexes with GFP-Pk3 complexes (Vangl2, Vangl2 Y7Y10Y12 > F or >E constructs) was performed using the ImageJ software with the Azimuthal Average plugin available at https://imagej.nih.gov/ij/plugins/azimuthal-average.html, following the described steps54. myr-BFP fluorescence was used to identify cell boundaries in mosaic cells, which were selected using the circle tool in ImageJ. The fluorescence intensity of GFP-Pk3 was quantified along the radius, divided to 180 bins within cells expressing PCP complexes.
To determine the effect of FGFR1 MO (15 ng) on neural cell fate, Sox3 immunofluorescence was measured in 3 × 10⁴ µm² rectangular regions of interest on the FGFR1 MO-injected (GFP-marked) and uninjected sides of the neural plate in three independent embryos.
Preparation of ectodermal explants, proximity biotinylation
Four-to-eight-cell embryos were injected with RNAs or FGFR1 MO, as indicated in figure legends, vitelline membrane was removed at stage 9 and ectoderm explants were dissected and cultured with or without of 25–100 ng/ml of FGF2 with or without 100 µM SU5402 (Millipore Sigma) until siblings reached stage 12 or stage 16 when they were collected for immunoprecipitation and immunoblotting analysis.
For proximity biotinylation59,68, embryos were injected into the animal pole of four-to-eight-cell embryos with RNAs encoding FLAG-BLN-Pk3, 100 pg, and HA-Vangl2 constructs, 40 pg. The embryos were allowed to develop until stage 9, followed by injection of 20 nl of a 0.8 mM biotin (Millipore Sigma) solution into the blastocoel. Embryos were lysed, and the protein biotinylation was assessed in embryo lysates and pulldowns obtained with mouse anti-HA (12CA5) antibody. Stages are indicated in the figure legends. Immunoblotting was carried out with goat anti-biotin-HRP antibodies (Cell Signaling) or goat anti-biotin antibody (Pierce), anti-FLAG (M2, Millipore Sigma), rabbit anti-HA (Bethyl) antibodies, mouse anti-HA (12A5), rabbit anti-phospho-ERK (Cell Signaling), rabbit anti-ERK (Cell Signaling), mouse anti-Vangl2 (C8, Santa Cruz), rat monoclonal anti-Vangl2 (2G4, Millipore Sigma), and mouse anti-GFP (B2, Santa Cruz). Chemiluminescence was captured by ChemiDoc (BioRad), and the Biotin/HA chemiluminescence intensity ratios were quantified using the ChemiDoc software.
Immunoprecipitation, immunoblotting and Vangl2 tyrosine phosphorylation
To detect tyrosine phosphorylation of Vangl2, endogenous or exogenous HA- or GFP-tagged Vangl2 proteins were pulled down with rabbit polyclonal Vangl2 (Millipore Sigma, Abn2242), rabbit polyclonal Vangl2 H55 (Santa Cruz), rabbit HA (Bethyl), antibodies and Protein A Sepharose, or GFP- or HA-trap (Chromotek) beads. The analysis of tyrosine phosphorylation was performed using a mouse monoclonal anti-phospho-tyrosine antibody (Santa Cruz, pY20, sc508). For analysis of FGFR1-Vangl2 complexes, FGFR1-3xFLAG RNA was co-injected with GFP-Vangl2 (100 pg each) or PTK7-GFP RNA into four animal blastomeres at the four-to-eight cell stage. Embryos were lysed at stage 12, and FGFR1 was pulled down with FLAG beads (Millipore Sigma). Vangl2, PTK7 and FGFR1 were detected in pulldowns and lysates with anti-GFP (GFP-B2, Santa Cruz) or anti-FLAG (M2, Millipore Sigma) antibodies. Phosphorylation at specific serine/threonine sites of Vangl2 was analyzed by immunoblotting after pulldowns of HA-Vangl2 with anti-HA antibodies (12CA5) from stage 12 embryo lysates. Membranes were probed with rabbit monoclonal antibodies against phospho-T78/S79/S82 (Cluster 1; AP1206, AbClonal) and phospho-S15/S17 (Cluster 2; AP1204, AbClonal).
The association between endogenous Vangl2 and FGFR1 was analyzed in the neural progenitor cell line C17.269 and HEK293T cells. Confluent C17.2 or HEK293T cells, initially plated in 100 mm dishes at 10⁴ cells per cm² were lysed, and immunoprecipitation was performed using 1 µg of either control Protein A Sepharose-purified rabbit IgG or rabbit polyclonal Vangl2 antibody (Millipore Sigma, ABN2242). Pulldowns and lysates were probed with rabbit monoclonal FGFR1 (Cell Signaling, D8E4) and mouse monoclonal Vangl2 (Santa Cruz, C8) antibodies.
Vangl2 analysis in mouse embryonic stem (ES) cells
Mouse wild type and FGFR1/2 double knockout ES cells52 were cultured in DMEM supplemented with penicillin/streptomycin, 1 mM of sodium pyruvate, non-essential amino acids, 1 µM β-mercaptoethanol, 15% fetal bovine serum (FBS), 1000 U of mouse recombinant LIF (R&D systems), 1 µM of MEK1 inhibitor PD0325901 (Tocris), and 3 µM of GSK3 inhibitor CHIR99021 (Tocris) as described52. Before the lysis, cells were plated on gelatinized 60 mm culture dishes. On the next day cells were briefly washed with PBS and cultured for 4 h in medium without inhibitors, LIF and FBS, to alleviate potential effects on FGFR signaling. Immunoprecipitation was done with either 1 µg Vangl2 H55 (Santa Cruz) or 0.4 µg of anti-pY antibody (pY20, Santa Cruz) with the lysates from 5 × 106 cells. Vangl2 pulldowns were immunoblotted with mouse Vangl2-specific antibody (C8, Santa Cruz), pY pulldowns were immunoblotted with rat anti-Vangl2 monoclonal antibody (MABN750, Millipore Sigma) and rabbit Cyclin B1 antibody (H-433, Santa Cruz) that served as a negative control. Vangl2 and FGFR1 expression in lysates was analyzed with rat monoclonal Vangl2 (MABN750, Millipore Sigma) and rabbit anti-FGFR1 antibody (D8E4, Cell Signaling), respectively.
Statistics and presentation of data
Statistical analyses, histograms, and graphs were generated using Prism 10 and Microsoft Excel. A two-tailed Student’s t-test (Fig. 1, Fig. S3 and Fig. S4) was used to assess statistical significance. Drawings of embryonic stages presented in Fig. 1, Fig. 2, Fig. 5, Supplementary Fig. 4 and Supplementary Fig. 8 were taken from Xenbase (www.xenbase.orgRRID:SCR_003280)70.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data used in the analysis are contained within the main text or in the supplementary materials. Materials are available on request from the authors. Source data are provided with this paper.
References
Benazeraf, B. et al. A random cell motility gradient downstream of FGF controls elongation of an amniote embryo. Nature 466, 248–252 (2010).
Deng, C. et al. Fibroblast growth factor receptor-1 (FGFR-1) is essential for normal neural tube and limb development. Dev. Biol. 185, 42–54 (1997).
Nerurkar, N. L., Lee, C., Mahadevan, L. & Tabin, C. J. Molecular control of macroscopic forces drives formation of the vertebrate hindgut. Nature 565, 480–484 (2019).
Sun, X., Meyers, E. N., Lewandoski, M. & Martin, G. R. Targeted disruption of Fgf8 causes failure of cell migration in the gastrulating mouse embryo. Genes Dev. 13, 1834–1846 (1999).
Yang, X., Dormann, D., Munsterberg, A. E. & Weijer, C. J. Cell movement patterns during gastrulation in the chick are controlled by positive and negative chemotaxis mediated by FGF4 and FGF8. Dev. Cell 3, 425–437 (2002).
Ciruna, B. & Rossant, J. FGF signaling regulates mesoderm cell fate specification and morphogenetic movement at the primitive streak. Dev. Cell 1, 37–49 (2001).
Eswarakumar, V. P., Lax, I. & Schlessinger, J. Cellular signaling by fibroblast growth factor receptors. Cytokine Growth Factor Rev. 16, 139–149 (2005).
Ornitz, D. M. & Itoh, N. The Fibroblast Growth Factor signaling pathway. Wiley Interdiscip. Rev. Dev. Biol. 4, 215–266 (2015).
Dorey, K. & Amaya, E. FGF signalling: diverse roles during early vertebrate embryogenesis. Development 137, 3731–3742 (2010).
Brewer, J. R., Molotkov, A., Mazot, P., Hoch, R. V. & Soriano, P. Fgfr1 regulates development through the combinatorial use of signaling proteins. Genes Dev. 29, 1863–1874 (2015).
Ray, A. T. et al. FGF signaling regulates development by processes beyond canonical pathways. Genes Dev. 34, 1735–1752 (2020).
Butler, M. T. & Wallingford, J. B. Planar cell polarity in development and disease. Nat. Rev. Mol. Cell Biol. 18, 375–388 (2017).
Gray, R. S., Roszko, I. & Solnica-Krezel, L. Planar cell polarity: coordinating morphogenetic cell behaviors with embryonic polarity. Dev. Cell 21, 120–133 (2011).
Devenport, D. The cell biology of planar cell polarity. J. Cell Biol. 207, 171–179 (2014).
Strutt, H., Warrington, S., Madathil, A. C. K., Langenhan, T. & Strutt, D. Molecular symmetry breaking in the Frizzled-dependent planar polarity pathway. Curr. Biol. 33, 5340–5354 e5346 (2023).
Peng, Y. & Axelrod, J. D. Asymmetric protein localization in planar cell polarity: mechanisms, puzzles, and challenges. Curr. Top. Dev. Biol. 101, 33–53 (2012).
Tree, D. R. P. et al. Prickle mediates feedback amplification to generate asymmetric planar cell polarity signaling. Cell 109, 371–381 (2002).
Wang, Y. & Nathans, J. Tissue/planar cell polarity in vertebrates: new insights and new questions. Development 134, 647–658 (2007).
Basta, L. P., Joyce, B. W., Posfai, E. & Devenport, D. Epithelial polarization by the planar cell polarity complex is exclusively non-cell autonomous. Science 387, eads5704 (2025).
Song, Y., Jian, S., Teng, J., Zheng, P. & Zhang, Z. Structural basis of human VANGL-PRICKLE interaction. Nat. Commun. 16, 132 (2025).
Zhang, F., Li, S., Wu, H. & Chen, S. Cryo-EM structure and oligomerization of the human planar cell polarity core protein Vangl1. Nat. Commun. 16, 135 (2025).
Kibar, Z. et al. Ltap, a mammalian homolog of Drosophila Strabismus/Van Gogh, is altered in the mouse neural tube mutant Loop-tail. Nat. Genet 28, 251–255 (2001).
Murdoch, J. N., Doudney, K., Paternotte, C., Copp, A. J. & Stanier, P. Severe neural tube defects in the loop-tail mouse result from mutation of Lpp1, a novel gene involved in floor plate specification. Hum. Mol. Genet 10, 2593–2601 (2001).
Jessen, J. R. et al. Zebrafish trilobite identifies new roles for Strabismus in gastrulation and neuronal movements. Nat. Cell Biol. 4, 610–615 (2002).
Ciruna, B., Jenny, A., Lee, D., Mlodzik, M. & Schier, A. F. Planar cell polarity signalling couples cell division and morphogenesis during neurulation. Nature 439, 220–224 (2006).
Nikolopoulou, E., Galea, G. L., Rolo, A., Greene, N. D. E. & Copp, A. J. Neural tube closure: cellular, molecular and biomechanical mechanisms. Development 144, 552–566 (2017).
Torban, E., Wang, H. J., Groulx, N. & Gros, P. Independent mutations in mouse Vangl2 that cause neural tube defects in looptail mice impair interaction with members of the Dishevelled family. J. Biol. Chem. 279, 52703–52713 (2004).
Ybot-Gonzalez, P. et al. Convergent extension, planar-cell-polarity signalling and initiation of mouse neural tube closure. Development 134, 789–799 (2007).
Gao, B. et al. Wnt signaling gradients establish planar cell polarity by inducing Vangl2 phosphorylation through Ror2. Dev. Cell 20, 163–176 (2011).
Montcouquiol, M. et al. Identification of Vangl2 and Scrb1 as planar polarity genes in mammals. Nature 423, 173–177 (2003).
Lu, X. et al. PTK7/CCK-4 is a novel regulator of planar cell polarity in vertebrates. Nature 430, 93–98 (2004).
Ossipova, O., Kim, K. & Sokol, S. Y. Planar polarization of Vangl2 in the vertebrate neural plate is controlled by Wnt and Myosin II signaling. Biol. Open 4, 722–730 (2015).
Mancini, P., Ossipova, O. & Sokol, S. Y. The dorsal blastopore lip is a source of signals inducing planar cell polarity in the Xenopus neural plate. Biol. Open 10, https://doi.org/10.1242/bio.058761 (2021).
Cox, W. G. & Hemmati-Brivanlou, A. Caudalization of neural fate by tissue recombination and bFGF. Development 121, 4349–4358 (1995).
Lamb, T. M. & Harland, R. M. Fibroblast growth factor is a direct neural inducer, which combined with noggin generates anterior-posterior neural pattern. Development 121, 3627–3636 (1995).
Lea, R., Papalopulu, N., Amaya, E. & Dorey, K. Temporal and spatial expression of FGF ligands and receptors during Xenopus development. Dev. Dyn. 238, 1467–1479 (2009).
Fletcher, R. B., Baker, J. C. & Harland, R. M. FGF8 spliceforms mediate early mesoderm and posterior neural tissue formation in Xenopus. Development 133, 1703–1714 (2006).
Berger, H., Wodarz, A. & Borchers, A. PTK7 Faces the Wnt in Development and Disease. Front Cell Dev. Biol. 5, 31 (2017).
Hayes, M., Naito, M., Daulat, A., Angers, S. & Ciruna, B. Ptk7 promotes non-canonical Wnt/PCP-mediated morphogenesis and inhibits Wnt/beta-catenin-dependent cell fate decisions during vertebrate development. Development 140, 1807–1818 (2013).
Delaune, E., Lemaire, P. & Kodjabachian, L. Neural induction in Xenopus requires early FGF signalling in addition to BMP inhibition. Development 132, 299–310 (2005).
Kengaku, M. & Okamoto, H. bFGF as a possible morphogen for the anteroposterior axis of the central nervous system in Xenopus. Development 121, 3121–3130 (1995).
Streit, A., Berliner, A. J., Papanayotou, C., Sirulnik, A. & Stern, C. D. Initiation of neural induction by FGF signalling before gastrulation. Nature 406, 74–78 (2000).
Wilson, S. I., Graziano, E., Harland, R., Jessell, T. M. & Edlund, T. An early requirement for FGF signalling in the acquisition of neural cell fate in the chick embryo. Curr. Biol. 10, 421–429 (2000).
Amaya, E., Musci, T. J. & Kirschner, M. W. Expression of a dominant negative mutant of the FGF receptor disrupts mesoderm formation in Xenopus embryos. Cell 66, 257–270 (1991).
Kinoshita, N. et al. Mechanical stress regulates epithelial tissue integrity and stiffness through the FGFR/Erk2 signaling pathway during embryogenesis. Cell Rep. 30, 3875–3888 e3873 (2020).
Nichane, M., Ren, X. & Bellefroid, E. J. Self-regulation of Stat3 activity coordinates cell-cycle progression and neural crest specification. EMBO J. 29, 55–67 (2010).
Jenny, A., Darken, R. S., Wilson, P. A. & Mlodzik, M. Prickle and Strabismus form a functional complex to generate a correct axis during planar cell polarity signaling. EMBO J. 22, 4409–4420 (2003).
Chu, C. W. & Sokol, S. Y. Wnt proteins can direct planar cell polarity in vertebrate ectoderm. Elife 5, e16463 (2016).
Butler, M. T. & Wallingford, J. B. Control of vertebrate core planar cell polarity protein localization and dynamics by Prickle 2. Development 142, 3429–3439 (2015).
Gao, B. et al. Coordinated directional outgrowth and pattern formation by integration of Wnt5a and Fgf signaling in planar cell polarity. Development 145, dev163824 (2018).
Mohammadi, M. et al. Structures of the tyrosine kinase domain of fibroblast growth factor receptor in complex with inhibitors. Science 276, 955–960 (1997).
Molotkov, A., Mazot, P., Brewer, J. R., Cinalli, R. M. & Soriano, P. Distinct requirements for FGFR1 and FGFR2 in primitive endoderm development and exit from pluripotency. Dev. Cell 41, 511–526 e514 (2017).
Kelly, L. K., Wu, J., Yanfeng, W. A. & Mlodzik, M. Frizzled-induced van gogh phosphorylation by CK1ε promotes asymmetric localization of core PCP factors in Drosophila. Cell Rep. 16, 344–356 (2016).
Chuykin, I., Itoh, K., Kim, K. & Sokol, S. Y. Frizzled3 inhibits Vangl2-Prickle3 association to establish planar cell polarity in the vertebrate neural plate. J. Cell. Sci. 134, jcs258864 (2021).
Yang, W. et al. Wnt-induced Vangl2 phosphorylation is dose-dependently required for planar cell polarity in mammalian development. Cell Res 27, 1466–1484 (2017).
Strutt, H., Gamage, J. & Strutt, D. Reciprocal action of Casein Kinase Iepsilon on core planar polarity proteins regulates clustering and asymmetric localisation. Elife 8, https://doi.org/10.7554/eLife.45107 (2019).
Humphries, A. C. et al. A Van Gogh/Vangl tyrosine phosphorylation switch regulates its interaction with core Planar Cell Polarity factors Prickle and Dishevelled. PLoS Genet 19, e1010849 (2023).
Kim, Y. S. et al. An unanticipated discourse of HB-EGF with VANGL2 signaling during embryo implantation. Proc. Natl Acad. Sci. 120, e2302937120 (2023).
Chuykin, I., Ossipova, O. & Sokol, S. Y. Par3 interacts with Prickle3 to generate apical PCP complexes in the vertebrate neural plate. Elife 7, https://doi.org/10.7554/eLife.37881 (2018).
Lee, J. et al. PTK7 regulates myosin II activity to orient planar polarity in the mammalian auditory epithelium. Curr. Biol. 22, 956–966 (2012).
Lee, O. K. et al. Discs-Large and Strabismus are functionally linked to plasma membrane formation. Nat. Cell Biol. 5, 987–993 (2003).
Ying, J., Yang, Y., Zhang, X., Dong, Z. & Chen, B. Stearoylation cycle regulates the cell surface distribution of the PCP protein Vangl2. Proc. Natl Acad. Sci. USA 121, e2400569121 (2024).
Andre, P. et al. The Wnt coreceptor Ryk regulates Wnt/planar cell polarity by modulating the degradation of the core planar cell polarity component Vangl2. J. Biol. Chem. 287, 44518–44525 (2012).
Feng, D. et al. Regulation of Wnt/PCP signaling through p97/VCP-KBTBD7-mediated Vangl ubiquitination and endoplasmic reticulum-associated degradation. Sci Adv 7, https://doi.org/10.1126/sciadv.abg2099 (2021).
Gudernova, I. et al. Multikinase activity of fibroblast growth factor receptor (FGFR) inhibitors SU5402, PD173074, AZD1480, AZD4547 and BGJ398 compromises the use of small chemicals targeting FGFR catalytic activity for therapy of short-stature syndromes. Hum. Mol. Genet. 25, 9–23 (2016).
Shi, D. L., Goisset, C. & Boucaut, J. C. Expression of Xfz3, a Xenopus frizzled family member, is restricted to the early nervous system. Mech. Dev. 70, 35–47 (1998).
Lew, E. D., Furdui, C. M., Anderson, K. S. & Schlessinger, J. The precise sequence of FGF receptor autophosphorylation is kinetically driven and is disrupted by oncogenic mutations. Sci. Signal 2, ra6 (2009).
Chuykin, I. & Sokol, S. Y. Analysis of planar cell polarity complexes by proximity biotinylation in xenopus embryos. Methods Mol. Biol. 2438, 97–106 (2022).
Snyder, E. Y. et al. Multipotent neural cell lines can engraft and participate in development of mouse cerebellum. Cell 68, 33–51 (1992).
Zahn, N. et al. Normal Table of Xenopus development: a new graphical resource. Development 149, https://doi.org/10.1242/dev.200356 (2022).
Acknowledgments
We thank Phil Soriano, Annette Borchers, Miho Matsuda and Jean-Pierre Saint-Jeannet for plasmids, Elena Torban for the anti-Vangl2 antibodies. We are grateful to Phil Soriano for the FGFR1/2 −/− double knockout mouse ES cells, Evan Snyder for C17.2 cells, and Nikos Tzavaras and Shilpa Dilip Kumar from the ISMMS Microscopy Core Facility for cell polarity quantification advice. We also thank James Clark and Jean-Pierre Saint-Jeannet for comments on the manuscript and Sokol lab members for discussions. This research was supported by the NIH grant R35GM122492 to S.Y.S.
Author information
Authors and Affiliations
Contributions
I.C. and S.Y.S. initiated, designed the experiments and developed the project. I.C. performed all the experiments, analyzed the data and prepared the figures. I.C. and S.Y.S. wrote the manuscript. S.Y.S. acquired the funding. Both authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chuykin, I., Sokol, S.Y. FGF receptor modulates planar cell polarity in the neuroectoderm via Vangl2 tyrosine phosphorylation. Nat Commun 16, 7219 (2025). https://doi.org/10.1038/s41467-025-62400-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-62400-x