Abstract
The rising demand for sustainable energy solutions has promoted significant interest in ionic thermoelectric materials, which convert low-grade waste heat into electrical energy through spatial temperature gradients. However, diurnal temperature variations, which offer potential for location-independent time-domain thermal energy, remain largely unexplored. To overcome the challenges of harvesting spatially limited thermal energy, this study presents an ionic thermoelectric cycle (t-ITC) designed for time-domain thermal energy harvesting, incorporating two gels with contrasting temperature coefficients. A temperature-adaptive self-regeneration (TASR) strategy is proposed to set the critical regeneration temperature (TCR) at the midpoint of temperature fluctuations, facilitating long-term device operation. The regeneration criteria are defined as neutralization of the electrochemical potential difference between separated cells, and a method based on shared counter-ion self-balancing is introduced. Employing a polyacrylamide-polyvinylpyrrolidone (PAM-PVP) matrix with KI3/KI and K3Fe(CN)6/K4Fe(CN)6 redox couples, both utilizing the same counter-ion K+ for regeneration, the t-ITC device attains a peak energy density of 3.28 kJ m–2 per cycle and a relative Carnot efficiency of 8.39% with 70% heat recuperation, under cycling conditions between 60 °C and 10 °C. This work highlights the potential of t-ITC devices for global-scale time-domain thermal energy on a global scale, across diverse environments, such as hot deserts and cold plateaus.
Similar content being viewed by others
Introduction
In the search for sustainable energy solutions, the capture of low-grade heat, typically defined as thermal energy at temperatures below 100 °C from various sources, has become a pivotal focus of research and development1. Traditional thermoelectric power generation techniques offer a means to directly harvest heat energy by establishing a spatial temperature difference between the heat source and the environment, drawing considerable interest2,3,4. However, low-grade heat, which is dispersed and has a minimal temperature difference compared to the surrounding environment, makes it difficult to generate large temperature gradients5. With the abundant thermal energy present in the environment, there is growing recognition of the potential to harvest time-domain temperature differences, such as diurnal fluctuations or periodic and intermittent variations in heat sources, which are not constrained by the spatial limitations typically associated with spatial temperature gradients6. These time-domain temperature differences can reach up to 50 °C in Earth’s deserts7, and 310 °C on Moon’s surface8 between day and night, offering substantial economic benefits9. Nevertheless, current methods for utilizing time-domain thermal energy often require thermal storage systems to convert these temperature variations into spatial temperature gradients, which are then coupled with traditional thermoelectric power generators10,11. Even when employing phase-change materials (PCMs) or radiative cooling materials to convert temporal temperature differences into spatial thermal gradients, the inherent loss of the time-domain temperature gradient persists. Consequently, conventional electronic thermoelectric (e-TE) materials exhibit low discharge capacity during nighttime when harvesting energy from time-domain temperature variations, resulting in a significant disparity between day and night discharge performance12,13,14,15,16 (Fig. S1).
Ionic thermoelectric (i-TE) energy conversion provides an alternative approach for capturing both spatial and time-domain temperature gradients. In spatial thermal energy, i-TE devices can operate in a generator mode, which includes voltage build-up, power output, and rest phases (Fig. 1a). The thermodiffusion-based i-TE, where charged ions move along the temperature gradient, and thermogalvanic-based i-TE, where ion pairs exhibit different reaction entropies at electrodes under varying temperatures, are both capable of generating thermal voltages17,18. Previous studies have explored the synergistic effects of thermodiffusion and thermogalvanic processes, resulting in a thermopower of 17 mV K–1 in Gelatin-KCl-K3Fe(CN)6/K4Fe(CN)6 i-TE gels19. However, in contrast to traditional electronic thermoelectric (e-TE) materials, which require a spatial temperature gradient for heat harvesting, the ionic thermoelectric (i-TE) materials offer greater versatility. Although the i-TE materials struggle to generate high discharge currents like e-TEs, they can operate in modes such as harvesting energy from time-domain temperature variations, utilizing ion-electrode interfaces rather than relying on direct electron flow through electrodes as in e-TE materials20,21. Consequently, a thermally regenerative electrochemical cycle was proposed using asymmetric copper hexacyanoferrate (CuHCF) and copper electrodes, separated by a cation-exchange membrane (CEM), to generate thermal potential via isothermal operations, involving two distinct redox reactions: KCuFeII/FeIII(CN)6 and Cu(NO3)2/Cu, building a thermodynamic cycle for converting heat to electricity22. This cycle consists of four stages: (1) heating at a high temperature TH in an isothermal state, (2) electrical charging to a voltage (VTH) with increased entropy, (3) cooling (TL) to reach an open-circuit voltage (VTL), and (4) discharge as entropy decreases through heat rejection into the environment23. However, this system requires external electrical charging or power supplies to facilitate the regeneration process, limiting its potential applications. Subsequently, a charging-free system was proposed using the K3Fe(CN)6/K4Fe(CN)6 and Prussian blue [KFeIIFeIII(CN)6, PB] without the need for electrical charging24. This approach requires that the two redox couples have opposite temperature coefficients, i.e., α = dEAg/Ag+/dT, where EAg/Ag+ is the electrical potential relative to the standard electrode of Ag/AgCl. This charging-free mode is referred to as the time-domain ionic thermoelectric cyclic mode (t-ITC) for time-domain thermal energy harvesting. The t-ITC also involves four steps: (1) heating, (2) forward discharging, (3) cooling, and (4) backward discharging (Fig. 1b). Forward discharging at TH requires a condition of \({E}_{Ag/A{g}^{+}}^{{\alpha }^{+}}\)>\({E}_{Ag/Ag+}^{{\alpha }^{-}}\), while backward discharging at TL needs a reverse condition, i.e., \({E}_{Ag/A{g}^{+}}^{{\alpha }^{+}}\)<\({E}_{Ag/Ag+}^{{\alpha }^{-}}\). Consequently, the electrode potentials (\({E}_{Ag/A{g}^{+}}^{{\alpha }^{+}}\) and \({E}_{Ag/Ag+}^{{\alpha }^{-}}\)) intersect at a temperature where the difference is zero, defined as the critical regeneration temperature (TCR). To ensure the t-ITC operates sustainably over extended periods, the TCR must meet the condition TL < TCR < TH. The t-ITC device using KFeIIFeIII(CN)6 and K3Fe(CN)6/K4Fe(CN)6 demonstrated a TCR of 37 °C and a device thermopower St-ITC of 1.47 mV K–1, obtained from \({S}_{t-ITC}={\alpha }_{+}-{\alpha }_{-}\), relative to the temperature coefficients of the redox couples with positive and negative temperature coefficients, which is always positive. However, identifying redox couples with opposite temperature coefficients, whose electrode potentials intersect within a suitable temperature range, and engineering the TCR to align with the uneven distribution of time-domain thermal energy on a global scale, or even in lunar environments, remains a major challenge.
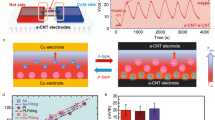
a Schematic illustration of spatial temperature gradient harvesting via the i-TE generator: (i) voltage build-up, (ii) power output, and (iii) rest phases; b Schematic illustration of time-domain temperature gradient harvesting via i-TE cycles: (i) voltage build-up, (ii) power output at high temperature, (iii) voltage build-up, and (iv) power output at low temperature; c Schematic illustration of the unequal electrode potentials of PAM/PVP-Fe(CN)63–/Fe(CN)64– and PAM-PVP-I3–/I– before the start of the regeneration treatment; d \({E}_{Ag/A{g}^{+}}^{{{I}_{3}}^{-}/{I}^{-}}\) for the PAM/PVP-I3–/I– and \({E}_{Ag/A{g}^{+}}^{Fe{({{{\rm{CN}}}})}_{6}^{3-/4-}}\) for PAM-PVP- Fe(CN)63–/Fe(CN)64– at the start of regeneration treatment (t0); e Schematic illustration of the electrode potential change for PAM/PVP-I3–/I– and PAM-PVP-Fe(CN)63–/Fe(CN)64– a t t = 6 h; f \({E}_{Ag/A{g}^{+}}^{{{I}_{3}}^{-}/{I}^{-}}\) for the PAM/PVP-I3–/I– and \({E}_{Ag/A{g}^{+}}^{Fe{({{{\rm{CN}}}})}_{6}^{3-/4-}}\) for PAM-PVP-Fe(CN)63–/Fe(CN)64– at t = 6 h; g Schematic illustration of the equal electrode potential for PAM-PVP-Fe(CN)63–/Fe(CN)64– and PAM/PVP-I3–/I– at the final state of the regeneration treatment (tend = 12 h); h \({E}_{Ag/A{g}^{+}}^{{{I}_{3}}^{-}/{I}^{-}}\) for the PAM/PVP-I3–/I– and \({E}_{Ag/A{g}^{+}}^{Fe{({{{\rm{CN}}}})}_{6}^{3-/4-}}\) for PAM-PVP-Fe(CN)63–/Fe(CN)64– at final stage of regeneration treatment tend; i Change in electrode potential and accumulated Qtransfer of electrons over treatment time.
This study introduces a quasi-solid-state t-ITC system for time-domain thermal energy harvesting using polyacrylamide-polyvinylpyrrolidone (PAM-PVP) hydrogel, achieving a device thermopower St-ITC of 2.4 mV K–1 through selective interactions between the polymer and redox couples. The iodine encapsulation approach with PVP also enhanced the device’s cycling stability, enabling over 300 cycles. A temperature-adaptive self-regeneration (TASR) strategy is proposed as a versatile solution for tuning the TCR. This strategy takes advantage of osmotic pressure and electrode potential gradients across the CEM during the TASR process, where the balance between ion diffusion and electrochemical potential supports the sustainability of time-domain thermal energy harvesting. With a TCR of 35 °C, the device demonstrated impressive discharge performance, reaching an energy density \({E}_{2h}^{{T}_{H}}\) of 1.7 kJ m–2 at TH = 60 °C and \({E}_{2h}^{{T}_{L}}\)1.58 kJ m–2 at TL = 10 °C under a 50 Ω and a 60 Ω external resistance, respectively. Additionally, the study showcases the modular design of the t-ITC device, which can be easily integrated into practical time-domain heat harvesting applications, such as in hot deserts and cold plateaus.
Results and discussion
Engineering T CR through temperature-adaptive self-regeneration strategy
In the t-ITC device described as below:
The initial concentrations of C(I3–/I–) = 0.4 M/0.4 M (on the left of the expression, with positive α) and C[Fe(CN)63–/4–] = 0.4 M/0.4 M (on the right of the expression, with negative α) were selected. C(I3–/I–) and C[Fe(CN)63–/4–] were denoted to the concentration ratios of I3– ions to I– ions (C[I3–]/ C[I3–]) and Fe(CN)63– ions to Fe(CN)64– ions (C[Fe(CN)63–/ C[Fe(CN)64–), respectively. The hydrogel of each side was prepared with a thickness of 1 mm. The initial open-circuit voltage Voc of the t-ITC device was near 70 mV, 120 mV, and 170 mV at temperatures of 10 °C, 25 °C, and 40 °C, respectively, based on the difference between \({E}_{Ag/A{g}^{+}}^{{\alpha }^{+}}\) and \({E}_{Ag/Ag+}^{{\alpha }^{-}}\). To ensure the proper cycling operation of the t-ITC device under both hot and cold conditions, the electrode potentials on both sides of the CEM must be equal at the designated TCR. Therefore, it is important to adjust TCR to fall within the time-domain temperature range (TL < TCR < TH). The temperature-dependent electrode potentials \({E}_{Ag/A{g}^{+}}^{{\alpha }^{+}}\) and \({E}_{Ag/Ag+}^{{\alpha }^{-}}\) can be expressed by the Nernst equation as follows25:
where E0, R, T, n, F, γ, and C are the standard potential, ideal gas constant, temperature, the number of moles of electrons transferred in the half-reaction, Faraday constant, activity coefficient, and concentration, respectively. The alteration in the ionic activity aO/R of the redox couples can affect the temperature-dependent electrode potential \({E}^{left/right}\). By applying the Nernst equation to compare \({E}_{Ag/A{g}^{+}}^{{\alpha }^{+}}\) and \({E}_{Ag/Ag+}^{{\alpha }^{-}}\) with different concentration ratios at the same temperature, it was observed that the variations in the activity coefficient ratios (γO/γR) of the redox couples do not follow a consistent pattern. Due to ionic interactions and hydration effects, accurately determining the ionic activity coefficient (γ) is challenging, and it is notably less than one under concentrated electrolyte conditions26. This variability makes it difficult to predict and regulate TCR by adjusting the concentration ratios of redox couples in different systems (Note S1). While a trial-and-error approach could theoretically be used to determine the corresponding concentration ratios, it remains inefficient and unclear for achieving the desired TCR. As a solution, a TASR strategy is proposed, which involves resetting the Voc to zero at the target TCR, generally set as the midpoint between high and low temperatures, i.e., TCR = (TH + TL)/2. Two conditions are necessary to implement TASR: i) the redox couple on the positive/negative side must share counter-ion, and ii) the t-ITC device electrodes must be connected to a matched external load and maintained at the target TCR. In this study, potassium ions and Nafion CEM were selected for the anionic redox couples of I3–/I– and Fe(CN)63–/Fe(CN)64–. Differences in ion migration rates and affinities between ions and the membrane may affect ion transport efficiency and system stability, influencing the overall performance. The shared counter-ion ensures that ion transport across the CEM is solely controlled by the concentration gradient on both sides, maintaining chemical equilibrium. A target TCR of 35 °C was selected to evaluate the feasibility of harvesting energy from time-domain temperature variations in hot climates, such as arid deserts. In these regions, daytime air temperatures can reach 40 °C, while solar radiation elevates device’s surface temperatures to higher than 60 °C. Nighttime temperatures then drop to ~10 °C, closely tracking the device’s surface temperature7. Detailed procedures are presented in Note S2 and Fig. S2.
At the initial state t0 (Fig. 1c), the as-fabricated t-ITC device was operated under isothermal conditions at 35 °C. A higher \({E}_{Ag/A{g}^{+}}^{{{I}_{3}}^{-}/{I}^{-}}\) enables the reduction reaction to be more likely to occur on the I3–/I– side of the t-ITC device, and the oxidation reaction occurs on the Fe(CN)63–/Fe(CN)64– side, with a device Voc of 161 mV (Fig. 1d). The t-ITC device was subsequently connected to a 50 Ω external load, which was matched with the effective internal resistance of the t-ITC device for steady self-discharging. The ΔV of \({E}_{Ag/A{g}^{+}}^{{{I}_{3}}^{-}/{I}^{-}}-{E}_{Ag/A{g}^{+}}^{Fe{({{{\rm{CN}}}})}_{6}^{3-/4-}}\) triggers electron flows from the Fe(CN)63–/Fe(CN)64– side to the I3–/I– side (right to the left) (Fig. 1e). As the treatment continued, I3– ions were reduced to I– ions, resulting in a decrease in the concentration ratio of I3–:I–, while Fe(CN)64– ions were oxidized to Fe(CN)63– ions, leading to an increase in the concentration ratio of Fe(CN)63–:Fe(CN)64–. This change in the valence state of the anionic redox couple causes an imbalance in anion concentrations on both sides of CEM, which drives the diffusion of potassium ions from the Fe(CN)63–/Fe(CN)64– sub-cell to the I3–/I– sub-cell through the CEM to maintain electric neutrality. The change in the ionic activity ratios of I3–/I– and Fe(CN)63–/Fe(CN)64– promoted a considerable decrease in the electrical potential \({E}_{Ag/A{g}^{+}}^{{{I}_{3}}^{-}/{I}^{-}}\) and an upwards shift in \({E}_{Ag/A{g}^{+}}^{Fe{({{{\rm{CN}}}})}_{6}^{3-/4-}}\) according to Eq. (1), suggesting a device Voc of 63 mV and a right shift in the TCR to 17 °C (Fig. 1f). Finally, the reaction is terminated after over 12 h of treatment (tend), with \({E}_{Ag/A{g}^{+}}^{{{I}_{3}}^{-}/{I}^{-}}={E}_{Ag/A{g}^{+}}^{Fe{({{{\rm{CN}}}})}_{6}^{3-/4-}}\), and the device Voc = 0 mV at 35 °C (Fig. 1g). The TCR shifted from 17 °C to 35 °C in the final state, as indicated by the EAg/Ag+-T curve (Fig. 1h). Figure 1i shows the Voc curve with the transfer charge quantity Q over regeneration treatment time with three 1-min disconnection operations every 4 h to stabilize the internal and external electric field balance. The QTransfer of 6.74 C indicated that 0.35 mol L–1 electrons are transferred from the Fe(CN)63–/Fe(CN)64– side to the I3–/I– side through an external circuit to balance the electrical potentials.
Figure 2 confirms the time-resolved variations in ion concentrations within the TASR process, as analyzed using UV–VIS spectroscopy. Figures 2a and S3a illustrate the evolution of concentrations of I3–/I– during the TASR treatment, indicated by UV-VIS absorption bands over time intervals of 2 h (from t0 to tend). The absorption observed prominently between 200 nm and 250 nm corresponds to the introduction of excess iodide ions and characteristic absorption peaks of the triiodide ion (I3–) appear at 288 nm and 351 nm. During the treatment process, a significant reduction reaction resulted in the partial reduction of I3– to I– ions. Concurrently, the intensity of the absorption band near 200 nm decreased, indicating the oxidation of Fe(CN)64– to Fe(CN)63– over the treatment duration (Fig. 2b and Fig. S3b). Variations in the tensities of characteristic UV–VIS absorption peaks provided insight into changes in the concentrations of corresponding active substances based on Lambert–Beers Law (Note S3)27. According to Lambert, absorption is directly proportional to the thickness of the irradiated cell, while according to Beer, absorption is directly proportional to concentration. By comparing the characteristic absorption peak intensity ratios of I3– (288 nm) to I– (225 nm), the normalized spectra in Fig. 2c demonstrated a decrease in the concentration ratio of C[I3–]/C[I–] from 1 to nearly 0.23 over the TASR treatment period28. Furthermore, Fig. 2d highlights enhanced absorption peak intensities near 300 nm and 420 nm, corresponding to the increased concentration ratio of C[Fe(CN)63–]/C[Fe(CN)64–] from 1 to nearly 7.38 following the TASR treatment29.
a UV-VIS spectra of the PAM-PVP-I3–/I– over TASR treatment time (t); b UV–VIS spectra of the PAM-PVP-Fe(CN)63–/Fe(CN)64– over TASR treatment time (t); c Normalized UV-VIS spectra of PAM-PVP-I3–/I–; d Normalized UV-VIS spectra of PAM-PVP-Fe(CN)63–/Fe(CN)64–; e Variations in concentration ratios and absorbance ratios of characteristic absorption bands in the UV–VIS spectra for PAM-PVP-I3–/I– and PAM-PVP-Fe(CN)63–/Fe(CN)64– during TASR treatment; f Electrical potentials (E) relative to Ag/AgCl standard electrode for the separated PAM-PVP-I3–/I– and PAM-PVP-Fe(CN)63–/Fe(CN)64–, along with the device voltage.
Figure 2e illustrates the variation in the concentration ratio of redox couples over the TASR treatment time at a TCR of 35 °C, derived from the absorption peak intensity ratios in the normalized UV-VIS spectra. The opposing trends in the concentration ratio changes of the redox couples confirmed the reverse direction of the redox reactions occurring on either side of the t-ITC device during the TASR treatment. The observed variation in the concentration ratios of I3–/I– and Fe(CN)63–/Fe(CN)64– during the treatment was about 0.3 mol L–1 (Fig. S4), aligning with the number of transferred electrons (QTransfer = 6.67 C, 0.35 mol L–1) shown in Fig. 1i. It is noteworthy that the 0.35 mol L–1 value of transferred electrons, as derived from QTransfer, was slightly higher than the QTransfer of about 0.3 mol L–1 determined from the UV-VIS spectra. This discrepancy is attributed to the increased activity coefficients of the ions involved. The shift in the concentration ratios of the redox couples contributed to the dynamic changes in the electrode potentials on both sides of the t-ITC system, which steadily decreased \({E}_{Ag/A{g}^{+}}^{{{I}_{3}}^{-}/{I}^{-}}\) and steadily increased \({E}_{Ag/A{g}^{+}}^{Fe{({{{\rm{CN}}}})}_{6}^{3-/4-}}\), thus achieving the necessary regeneration conditions for subsequent discharge under both hot and cold conditions (Fig. 2f).
Enhancing device thermopower and stability with hydrogel matrix
The device thermopower St-ITC and stability were further optimized by incorporating the PAM-PVP hydrogel matrix. Randomly arranged PVP chains were effectively linked to adjacent PAM chains through hydrogen bonding, creating a double-network crosslinked structure (Fig. 3a). The I3–/I– ions disrupted some of hydrogen bonding between PAM and PVP and interacted with two polymers (Fig. 3b). The interactions between I3–/I– and PAM-PVP can be separately investigated by Raman and UV-VIS spectroscopy. The interactions between I3–/I– and PAM can be observed by Raman spectra (top left of Fig. 3c), which identified peaks at 62 cm–1, 110 cm–1, 150 cm–1 and 226 cm–1. It was noted that the single Raman peak of I2 near 180 cm–1, shifted to about 150 cm-1, indicating coordination with donor atoms, such as I3–. The symmetrical I3– ion typically exhibits a single Raman-active ν1 symmetric stretch band at 110 cm–1, but additional vibrational modes, such as ν2 deformation vibration at 70 cm–1 and ν3 antisymmetric stretch within 130–140 cm–1, were activated due to interactions with amino and amide groups in PAM polymers, resulting in symmetry disruption30. Additionally, the interactions between I3– and PVP, which were always attributed to hydrogen bonding formation with two pyrrolidone residues in PVP, can be found in UV–VIS spectra31. As the PVP mass ratio increased from 1 to 10 wt.%, a broad UV–VIS absorption band (top right of Fig. 3c) appeared between 360 and 500 nm, reflecting complexation between PVP with I3– ions32. The intensities of these bands grew with a higher PVP ratio, indicating stronger PVP-I3– interactions, which enhanced iodide stability by the encapsulation function to reduce evaporation (bottom of Fig. 3c). The temperature coefficient (α) of the I3–/I– sub-cell is influenced by selective interactions between PAM-PVP polymer chains and I3– ions, while interactions with I– ions remain minimal. The absorption band of PVP-I3– also decreased with elevating temperature, because the binding constant between I3– and PVP decreased at higher temperature, further indicating their thermosensitive selective interactions (Fig. S5). This interaction affects the concentration ratio difference (ΔCr) of the redox couples and their entropy difference (ΔSr). A similar approach was employed by Zhou et al.33 to engineer the redox entropy of Fe(CN)63–/4– using the thermal sensitivity of guanidinium cations (Gdm+), which could also be applicable in this system. The α values of both I3–/I– and Fe(CN)63–/Fe(CN)64– redox couples in aqueous solutions and hydrogels at varying concentration ratios (with PAM-PVP representing 5% PVP). Incorporating the PAM-PVP polymer matrix significantly enhanced the α of I3–/I– from –0.6 mV K–1 to –1.13 mV K–1 with TCR = 35 °C. Conversely, the α of Fe(CN)63–/Fe(CN)64– remained stable at 1.3 mV K–1, indicating negligible interaction with the hydrogel chains. Thus, an optimized St-ITC of 2.4 mV K–1 was achieved under the condition of adjusting TCR to 35 °C (Fig. 3d). Based on Eq. (2), derived from the Nernst equation in Eq. (1) under the assumption that activity is proportional to species concentration.
where α0 R, n, and F are the temperature coefficient for CO/CR = 1, ideal gas constant, the number of moles of electrons transferred in the half-reaction, and Faraday constant, respectively. The temperature coefficient (α) of the PAM-PVP- I3–/I– and PAM-PVP- Fe(CN)63–/Fe(CN)64– sub-cells (Fig. S6-S8) exhibits concentration-dependent behavior. This was quantified via a three-electrode system across varied redox ion concentration ratios (C[I3–/I–] = 0.4 M/0.4 M, 0.21 M/0.59 M, 0.15 M/0.65 M; C[Fe(CN)63–/Fe(CN)64–] = 0.4 M/0.4 M, 0.63 M/0.17 M, 0.73 M/0.07 M). Critically, increasing the CO/CR ratio reduces positive α values, while decreasing CO/CR diminishes negative α values.
a Schematic structure of the PAM-PVP double network hydrogel matrix; b Schematic structure of the PAM-PVP-I3–/I– hydrogel; c Raman spectra of PAM-x I3– hydrogels (x represents concentrations of 0.01 M, 0.05 M, 0.1 M, 0.15 M, and 0.2 M) and UV-VIS spectra of PAM-PVP-I3–/I– hydrogel with different PVP ratios of 1% to 10%; d Comparison of temperature coefficients α of I3–/I– and Fe(CN)63–/Fe(CN)64– solutions (left column) and PAM-PVP-I3–/I– and PAM-PVP-Fe(CN)63–/Fe(CN)64– hydrogels (right column); e Stress–strain curves for PAM, PAM-5% PVP, PAM-5% PVP- Fe(CN)63–/Fe(CN)64–, and PAM-5% PVP- I3–/I– hydrogels, respectively; f Discharging voltage-time curve in one cycle of the t-ITC system with TCR of 35 °C under TH = 60 °C and TL = 10 °C conditions; g Two-hour output energy densities (E2h) during discharging at TH and TL; h Comparison of energy retention percentages for the t-ITC system with and without PVP addition during 300 cycles; i Relative Carnot efficiency (ηr) plotted against heat recuperation efficiency (ηHR) for the t-ITC system with varying active layer thicknesses.
Additionally, the PAM-PVP hydrogel exhibited a maximum tensile strain exceeding 1600%, about 1.5 times that of pristine PAM hydrogel, due to robust hydrogen bonding between PAM and PVP chains, enabling efficient energy dissipation (Fig. 3e)34. Although the electrolyte ions weaken hydrogen bond strength in the double-network polymer chain35, the maximum tensile strains for PAM-PVP-I3–/I– and PAM-PVP-Fe(CN)63–/Fe(CN)64– reached 1350% and 1270%, respectively at a concentration ratio of C(I3–/I–) = C[Fe(CN)63–/Fe(CN)64–] = 0.4 M/0.4 M, ensuring the t-ITC devices meet the flexibility standards. Additionally, it is noteworthy that the tensile stress-strain curves of both PAM-PVP-I3–/I– and PAM-PVP-Fe(CN)63–/Fe(CN)64– hydrogels are significantly influenced by variations in water content, exhibiting a clear trend where lower water content corresponds to decreased strain and increased stress (Fig. S9).
Figure 3f demonstrates the discharge performance of the t-ITC device in a consistent cycle between TH = 60 °C and TL = 10 °C, with a constant current of 1 mA cm–2. The electron transfer occurring in both forward and backward discharging processes resulted in fluctuations in the concentration ratio of redox couples, which in turn caused dynamic changes in the TCR and thermal voltage in the initial cycles. The TASR strategy can be applied repeatedly to restore the targeted TCR. Hydrophilic carbon paper (CP) with fibrous structures was employed (Fig. S10), offering abundant electrochemically active interfaces that facilitate electron exchange between redox couples and electrodes (Figs. S11, S12)36. As a result, the maximum power density (Pmax), reflecting peak instantaneous output, reached 1.6 W m–2 at TH = 60 °C (Fig. S13). The output voltage and energy densities under constant-resistance discharging conditions showed variation as a function of external loads, ranging from 10 Ω to 80 Ω, during a 2-h discharge at TH = 60 °C and TL = 10 °C, respectively (Fig. 3g, Fig. S14 and S15). The t-ITC device achieved a sustained energy output with high 2-h energy density \({E}_{2h}^{{T}_{H}}\) of 1.7 kJ m–2 at TH = 60 °C with a 50 Ω external resistance and \({E}_{2h}^{{T}_{L}}\) 1.58 kJ m–2 at TL = 10 °C with a 60 Ω external resistance, outperforming other quasi-solid-state i-TE devices for harvesting spatial temperature gradients (Fig. S16). Figure 3h presents a comparison of the energy retention in t-ITC devices during cycling tests, emphasizing the improved stability achieved by incorporating PVP. The cycling tests were carried out between 10 °C and 60 °C, with each charge lasting 10 min, followed by a 20-min discharge at a constant current of 1 mA cm–2. After 300 cycles, the PAM-only t-ITC device displayed a significant decline, retaining only 40% of its initial discharge energy (Fig. S17). This is likely due to iodine evaporation, which disrupted the electrolyte salinity balance during the heating-cooling cycles37. In contrast, the PAM-PVP-based t-ITC device exhibited further improvement in cycling stability, retaining more than 90% of its discharge energy after 300 cycles (Fig. S18). Encapsulation of I2 within PVP has been shown to enhance stability through hydrogen bonding interactions38, as demonstrated in antimicrobial applications. No I2 penetration or discoloration was observed on the electrode surface, and iodine penetration tests with PVP concentrations exceeding 5% further validated the improved stability (Fig. S19). When considering time-domain thermal energy harvesting, the cycling performance of the t-ITC device is notably better than that of other reported quasi-solid i-TE devices39,40. Beyond demonstrating stable cycling performance, the t-ITC device achieves symmetric diurnal discharge capacity under dynamically varying temperature differences through continuous TCR modulation. We performed five consecutive cycle tests where the maximum-minimum temperature difference varied continuously within a range of ±5 °C. Continuous TCR adjustment after each cycle ensured consistent and stable discharge capacity matching within each cycle. Furthermore, the TASR regeneration process was consistently completed within a short timeframe (Fig. S20). While the initial TASR process exhibits extended treatment times, subsequent cycles achieve rapid reconditioning (<30 min) for minor TCR adjustments (~5 °C), making it practical for diurnal applications.
The heat-to-electricity conversion efficiency (η) of the t-ITC system under isothermal conditions is calculated with in detail Note. S4 and Table. S3. The efficiency (η) is influenced by the device thickness, which affects the mass of active materials (mi) and total heat capacity (Cp). The t-ITC devices with different active layer thicknesses (2 mm, 1 mm, 500 μm, and 200 μm) were fabricated (Fig. S21). The Pmax of the t-ITC device was unaffected by thickness, as isothermal operation simplifies thermal management without relying on a temperature gradient. The energy density remained nearly constant across all thicknesses during the steady discharge cycle (Fig. S22). Theoretically, with a constant energy density, efficiency (η) can be improved by reducing the mass of active material (mi), even with a shorter discharge time. Reducing mi notably increases the relative charge transfer concentration (Ne-/Nredox couple), which shifts the concentration ratio of the redox couples, enhancing the dynamic thermal voltage during i-TE cycles, as described in Eq. (2). The relative Carnot efficiency (ηr) for the t-ITC device with an optimized 200 μm active layer reaches 3.93%, even without heat recuperation (ηHR = 0), marking the highest efficiency reported among quasi-solid-state i-TE systems in this temperature range (Fig. 3i). Additionally, the overall efficiency (η) is influenced by heat recuperation (ηHR), with the ηr of the t-ITC device reaching 6.36% and 8.39% for the 200 μm active layer when ηHR is 50% and 70%, respectively, showing obvious advantages compared with other hydrogel-based i-TE devices (Table S4).
Designing t-ITC modules for round-the-clock energy harvesting
Figure 4a, b depicts the method used to assemble the multi-cell module. Unlike traditional i-TE modules that use n-type or p-type legs, the t-ITC utilizes redox couples with opposing temperature coefficients, separated by the CEM. This design simplifies large-area manufacturing by eliminating the need for complex electrode connections (Fig. 4a). The interaction between these components creates ionically conductive pathways, including the PAM-PVP-I3–/I– hydrogel, CEM, PAM-PVP-Fe(CN)63–/Fe(CN)64– hydrogel, and electrode patterns on a PET substrate (Fig. 4b). The t-ITC devices were arranged in a 5 × 5 array, with Fig. 4c showing both partial and full views of the setup. Its flexibility allows it to conform to various surface shapes, making it adaptable for a wide range of practical applications. The TCR can be fine-tuned using the TASR strategy to match different time-domain heat sources. Additionally, by employing the deep eutectic solvent 30% formamide (FA)/H2O, the TCR of the t-ITC device can be further adjusted to function at temperatures as low as –20 °C by lowering the freezing point of the aqueous solvent (Note. S5, Fig. S23-26)41. The discharge energy density in a single cycle \({E}_{cycle}={E}_{2h}^{{T}_{H}}+{E}_{2h}^{{T}_{L}}\) varied with the adjusted TCR, under the same time-domain temperature range of TH – TL = 50 °C due to the changes in ionic conductivity (Fig. S27). A maximum energy density Ecycle of 3.28 kJ m–2 was achieved in the t-ITC device with a TCR of 35 °C. If 1% of the land area in Xinjiang, China (16,649 km2) were employed for harvesting time-domain temperature difference energy using the t-ITC system, it could generate 3.33 × 107 MJ of electricity each day, representing a considerable source of clean energy.
a Schematic illustration of the structure of integrated t-ITC module, featuring the electrode pattern on PET substrate, cation-exchange membrane (CEM), and hydrogels; b Overview of the connection mechanism within the t-ITC device; c Digital photographs of partial and full views of the t-ITC module; Maximum ambient air temperature distributions of Xinjiang, China in summer (averages in July and August from 2021 to 2023) (during d daytime; e, nighttime); f Discharge voltage-time curve of one complete cycle of the as-fabricated t-ITC module with TCR of 40 °C under a broad temperature range TH = 75 °C and TL = 5 °C; Maximum ambient air temperature distributions of western USA in summer (averages in July and August from 2021 to 2023) (during g daytime; h nighttime); i Discharge voltage-time curve of one complete cycle of the as-fabricated t-ITC module with TCR of 15 °C at TH = 40 °C and TL = –15 °C.
After successfully regenerating a single device, the TASR strategy was applied directly to the integrated module to demonstrate the need for large-area configurations. Two typical regions with significant time-domain thermal energy were chosen for this analysis: 1) the hot desert in Xinjiang, China, and 2) the cold Plateau in the western United States42,43. The diurnal temperature range (DTR) was denoted as ΔTd = TH(day)-TL(night), describing the variation between a high air temperature and a low temperature that occurs during the same day was applied to describe the time-domain temperature difference for different regions. The hot desert region is characterized by a distinctive mountain-basin structure and fragile ecosystems, with a ΔTd surpassing 50 °C44. Figure 4d, e present the monthly ΔTd of air temperature distribution for this desert region during the summer, based on CRU data45, reaching over 30 °C, with some extreme days exceeding 60 °C, described in Note S6. Given the intense solar radiation, the actual time-domain temperature difference on the device surface is expected to be even higher. To demonstrate the feasibility of harvesting energy from diurnal temperature variations, a field test of the t-ITC device was conducted in Shenzhen, a region characterized by relatively low natural diurnal temperature fluctuations, under cloudy conditions on June 24, 2025. Despite ambient air temperatures averaging 37 °C during the day and 26 °C at night, solar irradiation elevated the device’s surface temperature to 60 °C. This resulted in a substantial diurnal surface temperature variation of 34 °C, significantly exceeding the natural air temperature fluctuation (Figs. S28–S31). This outcome strongly supports the significant potential for energy harvesting utilizing diurnal thermal cycles.
To match those extreme climates with large ΔTd like Xinjiang, the TCR of the 5 × 5 t-ITC module was adjusted to 40 °C, allowing an efficient and large temperature range from 5 °C to 75 °C. In a time-domain cycle (Fig. 4f), the t-ITC module with a TCR of 40 °C was discharged at a constant current density of 1 mA cm–2, demonstrating continuous heat energy harvesting from 5 °C to 75 °C. The I–V curve of the t-ITC module showed a Voc of 2.1 V, Isc of 7.7 and Pmax of 5.4 mW at TH = 70 °C (Fig. S32). The cold plateau regions, with high latitudes and plateau terrain, also experience high average day-night temperature difference during the summer, with some areas reaching Tmin below 0 °C (Fig. 4g-h)43. To effectively capture the diurnal time-domain temperature differences in such regions, the TCR of the t-ITC module was set to 15 °C to match the local temperature range. Figure 4i shows a time-domain cycle of the t-ITC module with a TCR of 15 °C, operating between a hot condition of 40 °C and a cold condition of –10 °C for over 20 min at a constant current density of 0.5 mA cm–2. This demonstrated the effectiveness of the TASR strategy, even at lower temperatures, where ionic conductivity was reduced. The t-ITC module, with its high output power density, is capable of directly powering a light-emitting diode (LED) array and a commercial digital thermo-hygrometer (Fig. S33, Movie S1 and S2). Furthermore, Fig. S34 compares the voltage and power outputs of the integrated t-ITC module with other reported quasi-solid-state i-TE modules19,46,47,48,49,50,51, delivering ten times the power output of other quasi-solid-state devices, and underscoring the significance of the TASR strategy to tune TCR for global-scale time-domain heat sources. Moreover, the t-ITC device offers a more economically viable material solution compared to others (like e-TEs). Its substantially lower cost-performance metric enhances the feasibility of large-scale deployment for harvesting low-grade diurnal waste heat14,52 (Table S5).
In summary, this study presents a pioneering hydrogel-based t-ITC system designed to capture time-domain temperature thermal energy, such as diurnal temperature fluctuations, for efficient low-grade heat harvesting, providing a novel alternative to traditional spatial temperature gradient approaches. The temperature-adaptive mechanism of the t-ITC system precisely aligns electrode potentials, optimizing its performance under fluctuating thermal conditions and offering an adjustable critical regeneration temperature (TCR) to enhance its adaptability in dynamic environments. By incorporating PAM-PVP hydrogels and KI3/KI and K3Fe(CN)6/K4Fe(CN)6 redox couples, the system achieves an impressive thermopower of 2.4 mV K–1 and a peak power density of 1.6 W m–2 at 60 °C. Under isothermal conditions (10 °C and 60 °C), the integrated t-ITC modules efficiently power electronic devices, such as LED arrays and digital instruments. With a relative Carnot efficiency of 3.93% (and up to 8.39% with 70% heat recuperation) and excellent cycling stability, the t-ITC system holds substantial potential for real-world applications in low-grade heat conversion, including hot desert and cold plateau regions. This work establishes a strong foundation for further exploration of time-domain thermal energy, positioning the t-ITC system as a transformative solution for continuous and efficient energy harvesting from global-scale time-domain thermal energy in various environments.
Methods
Materials
K3Fe(CN)6 (MW = 329.25, ≥99.5%), K4Fe(CN)6 3H2O (MW = 422.39, 99.0%), KI (MW = 166, 99.8%), Polyvinyl pyrrolidone (PVP, MW = 40000, ≥95%) and formamide (FA) (MW = 45.04, 99.0%) were purchased from Aladdin Industrial Corporation (Shanghai, China). I2 (MW = 253.81, 99.0%) was provided by Macklin Biochemical Co., Ltd. (Shanghai, China). Acrylamide (MW = 71.08, ≥99%) monomer, N, N′-methylenebisacrylamide (BIS, MW = 154.17, 99%) cross-linker, ammonium persulfate (APS, MW = 228.2, ≥98%) initiator were purchased from Sigma-Aldrich. All chemical reagents were used as received without further purification. Graphite sheets and hydrophilic carbon paper were purchased from JingLong Special Carbon Company, Ltd. (China) and Toray Industries, Inc. (Japan), respectively. Cation-exchange membrane CEM (Nafion, N212, 50.8 μm) was provided by DuPont de Nemours, Inc. (USA).
Characterization
The time-domain thermopower measurement of the t-ITC was conducted under an isothermal condition. The temperatures of the two sides were controlled by a digital temperature control module (YEXIAN TCM-M207, China). The open-circuit voltage and temperature results were acquired using the LabVIEW program. The OCV was measured with a Keithley-2000 instrument and the current-voltage characterization of the device was performed with a Keithley 2400 instrument. There are approximately 50 points between 0 V to the corresponding open-circuit voltage at different temperatures. The voltage sweep rate is 0.1 s per point. The voltage and current were measured by a Keithley-2400 and Keithley-2450, respectively, during the voltage build-up and power output stages of continuous power output.
The ionic conductivities of the t-ITC devices were calculated from the first intercept of the high-frequency semicircle in the results of EIS measurements at different temperatures, where the response is fully resistive. EIS measurements were conducted between 10 kHz and 50 mHz with an AC amplitude of 10 mV. The structural and chemical bonding characteristics of the electrolyte were examined using FTIR-attenuated total reflectance techniques (Bruker Vertex 70 V, province, country). The electrochemical performances were measured on an electrochemical workstation (Zennium Pro, Germany) with a three-electrode configuration. A carbon paper (CP) piece was used as the working electrode, and platinum and Ag/AgCl electrodes were used as the counter and reference electrodes, respectively. The UV-VIS spectra were recorded on a Lamda 950 spectrophotometer. The samples for UV–VIS test were obtained from the t-ITC device with different TASR treatment time. FESEM image was recorded on HITACHI cold FE-SEM SU8230. Mechanical stress-strain curves were recorded on a universal mechanical test machine (Instron 3342) at room temperature.
Device fabrication
Different concentrations 1% to 10% of PVP was first dissolved in deionized water at 25 °C to make a homogeneous aqueous solution. To prepare PAM-PVP hydrogel, 10.0 g acrylamide was added in to make a 100 mL homogeneous aqueous solution. 2.19 × 10−5 mol APS initiator and 4.67 × 10−5 mol BIS cross-linker were added. The solution was degassed for 5 min in a vacuum chamber and then cured under UV-light for 60 min. The hydrogel was dried at 70 °C and immersed in different concentrations of K3Fe(CN)6/K4Fe(CN)6 and KI3/KI solutions until complete swelling. The t-ITC device was assembled with a laminar structure of two graphite current collectors, two CP electrodes, a CEM in the middle to separate the two cells, a cylinder spacer composed of Polydimethylsiloxane, and two different hydrogels inside the spacer. The as-fabricated t-ITC device and its internal structure were depicted in Fig. S35.
The t-ITC module containing 25 integrated units consisted of graphite-carbon paper electrode patterns, PET flexible substrates, hydrogels and CEM membranes. The module size was 100 mm × 100 mm × 0.5 mm (length × width × height) and contained 25 units. The carbon electrodes on both sides have been pasted on PET film in advance as flexible electrode patterns. The as-prepared hydrogels with K3Fe(CN)6/K4Fe(CN)6 and KI3/KI were connected to the electrodes and then compressed with the middle CEM membrane to simply fabricate the large-area t-ITC module.
Data availability
The relevant data generated in this study have been deposited in the Figshare database under accession code: https://doi.org/10.6084/m9.figshare.29909345.v2. Extra data are available from the corresponding author upon request.
References
Forman, C., Muritala, I. K., Pardemann, R. & Meyer, B. Estimating the global waste heat potential. Renew. Sustain. Energy Rev. 57, 1568–1579 (2016).
Crane, D. T. & Bell, L. E. Maximum temperature difference in a single-stage thermoelectric device through distributed transport properties. Int. J. Therm. Sci. 154, 106404 (2020).
Snyder, G. J. & Toberer, E. S. Complex thermoelectric materials. Nat. Mater. 7, 105–114 (2008).
He, J. & Tritt, T. M. Advances in thermoelectric materials research: looking back and moving forward. Science 357, eaak9997 (2017).
Yang, W., Bao, J., Liu, H., Zhang, J. & Guo, L. Low-grade heat to hydrogen: current technologies, challenges and prospective. Renew. Sustain. Energy Rev. 188, 113842 (2023).
Xiao, C., Naclerio, N. D., Hawkes, E. W. Energy harvesting across temporal temperature gradients using vaporization. In proc. IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS)) (2019).
Zhao, Y., Norouzi, H., Azarderakhsh, M. & AghaKouchak, A. Global patterns of hottest, coldest, and extreme diurnal variability on earth. Bull. Am. Meteorol. Soc. 102, E1672–E1681 (2021).
Williams, J. P., Paige, D. A., Greenhagen, B. T. & Sefton-Nash, E. The global surface temperatures of the Moon as measured by the Diviner Lunar Radiometer Experiment. Icarus 283, 300–325 (2017).
Zhong, Z. et al. Reversed asymmetric warming of sub-diurnal temperature over land during recent decades. Nat. Commun. 14, 7189 (2023).
Cui, T., Xuan, Y. & Li, Q. Design of a novel concentrating photovoltaic–thermoelectric system incorporated with phase change materials. Energy Convers. Manag. 112, 49–60 (2016).
Li, T. et al. Highly conductive phase change composites enabled by vertically-aligned reticulated graphite nanoplatelets for high-temperature solar photo/electro-thermal energy conversion, harvesting and storage. Nano Energy 89, 106338 (2021).
Liu, J. et al. All-day uninterrupted thermoelectric generator by simultaneous harvesting of solar heating and radiative cooling. Opt. Express 31, 14495–14508 (2023).
Liu, M. et al. Sustainable all-day thermoelectric power generation from the hot sun and cold universe. Small 20, 2403020 (2024).
Whalen, S. A. & Dykhuizen, R. C. Thermoelectric energy harvesting from diurnal heat flow in the upper soil layer. Energy Convers. Manag. 64, 397–402 (2012).
Omair, Z., Assawaworrarit, S., Fan, L., Jin, W., Fan, S. Radiative-cooling-based nighttime electricity generation with power density exceeding 100 mW/m2. iScience 25, (2022).
Wu, Z. et al. High-performance floating thermoelectric generator for all-day power supply. Nano Energy 133, 110443 (2025).
Wang, J. et al. “Thermal charging” phenomenon in electrical double layer capacitors. Nano Lett. 15, 5784–5790 (2015).
Quickenden, T. I. & Vernon, C. F. Thermogalvanic conversion of heat to electricity. Sol. Energy 36, 63–72 (1986).
Han, C.-G. et al. Giant thermopower of ionic gelatin near room temperature. Science 368, 1091–1098 (2020).
Bonetti, M. et al. Thermoelectric energy recovery at ionic-liquid/electrode interface. J. Chem. Phys. 142, 244708 (2015).
Zhao, D. et al. Ionic thermoelectric supercapacitors. Energy Environ. Sci. 9, 1450–1457 (2016).
Lee, S. W. et al. An electrochemical system for efficiently harvesting low-grade heat energy. Nat. Commun. 5, 3942 (2014).
Gao, C., Lee, S. W. & Yang, Y. Thermally regenerative electrochemical cycle for low-grade heat harvesting. ACS Energy Lett. 2, 2326–2334 (2017).
Yang, Y. et al. Charging-free electrochemical system for harvesting low-grade thermal energy. Proc. Natl Acad. Sci. USA 111, 17011–17016 (2014).
Bard, A.J., Faulkner, L. R., White, H. S. Electrochemical Methods: Fundamentals and Applications (John Wiley & Sons, 2022).
Stokes, R. H. & Robinson, R. A. Ionic hydration and activity in electrolyte solutions. J. Am. Chem. Soc. 70, 1870–1878 (1948).
Akash, M. S. H., Rehman, K. Ultraviolet-visible (UV-VIS) spectroscopy. In: Essentials of Pharmaceutical Analysis (eds Akash MSH, Rehman K) (Springer Nature, 2020).
Jung, S.-H., Yeon, J.-W., Kang, Y. & Song, K. Determination of triiodide ion concentration using UV-visible spectrophotometry. Asian J. Chem. 26, 4084–4086 (2014).
Veerakumar, P., Salamalai, K., Thanasekaran, P. & Lin, K.-C. Simple preparation of porous carbon-supported ruthenium: propitious catalytic activity in the reduction of ferrocyanate(III) and a cationic dye. ACS Omega 3, 12609–12621 (2018).
Gabes, W. & Gerding, H. Vibrational spectra and structures of the trihalide ions. J. Mol. Struct. 14, 267–279 (1972).
Zamora, J. L. Chemical and microbiologic characteristics and toxicity of povidone-iodine solutions. Am. J. Surg. 151, 400–406 (1986).
Oster, G. & Immergut, E. H. Ultraviolet and infrared spectral studies of polyvinylpyrrolidone1. J. Am. Chem. Soc. 76, 1393–1396 (1954).
Yu, B. et al. Thermosensitive crystallization–boosted liquid thermocells for low-grade heat harvesting. Science 370, 342–346 (2020).
Xu, Q.-J. et al. Tough Semi-interpenetrating Polyvinylpyrrolidone/polyacrylamide hydrogels enabled by bioinspired hydrogen-bonding induced phase separation. Chin. J. Polym. Sci. 42, 591–603 (2024).
Lewoczko, E. M., Kelly, M. T., Kent, E. W. & Zhao, B. Effects of temperature on chaotropic anion-induced shape transitions of star molecular bottlebrushes with heterografted poly(ethylene oxide) and poly(N,N-dialkylaminoethyl methacrylate) side chains in acidic water. Soft Matter 17, 6566–6579 (2021).
Wang, S. et al. High-performance cryo-temperature ionic thermoelectric liquid cell developed through a eutectic solvent strategy. Nat. Commun. 15, 1172 (2024).
Yeon, J.-W. & Jung, S.-H. Effects of temperature and solution composition on evaporation of iodine as a part of estimating volatility of iodine under gamma irradiation. Nucl. Eng. Technol. 49, 1689–1695 (2017).
Rackur, H. New aspects of mechanism of action of povidone-iodine. J. Hosp. Infect. 6, 13–23 (1985).
Chen, B. et al. Giant negative thermopower of ionic hydrogel by synergistic coordination and hydration interactions. Sci. Adv. 7, eabi7233 (2021).
Hu, J. et al. Double selective ionic gel with excellent thermopower and ultra-high energy density for low-quality thermal energy harvesting. Energy Environ. Sci. 17, 1664–1676 (2024).
Gao, Y., Qin, Z., Guan, L., Wang, X. & Chen, G. Z. Organoaqueous calcium chloride electrolytes for capacitive charge storage in carbon nanotubes at sub-zero temperatures. Chem. Commun. 51, 10819–10822 (2015).
Azarderakhsh, M., Prakash, S., Zhao, Y. & AghaKouchak, A. Satellite-based analysis of extreme land surface temperatures and diurnal variability across the hottest place on earth. IEEE Geosci. Remote S. Lett. 17, 2025–2029 (2020).
Whiteman, C. D., Haiden, T., Pospichal, B., Eisenbach, S. & Steinacker, R. Minimum Temperatures, diurnal temperature ranges, and temperature inversions in limestone sinkholes of different sizes and shapes. J. Appl. Meteorol. 43, 1224–1236 (2004).
Yao, J. et al. Recent climate and hydrological changes in a mountain–basin system in Xinjiang. China Earth-Sci. Rev. 226, 103957 (2022).
Harris, I., Osborn, T. J., Jones, P. & Lister, D. Version 4 of the CRU TS monthly high-resolution gridded multivariate climate dataset. Sci. Data 7, 109 (2020).
Yang, P. et al. Wearable thermocells based on gel electrolytes for the utilization of body heat. Angew. Chem. Int. Ed. 55, 12050–12053 (2016).
Duan, J. et al. P-N conversion in thermogalvanic cells induced by thermo-sensitive nanogels for body heat harvesting. Nano Energy 57, 473–479 (2019).
Zhang, L. et al. High power density electrochemical thermocells for inexpensively harvesting low-grade thermal energy. Adv. Mater. 29, 1605652 (2017).
Li, Y. et al. 3D hierarchical electrodes boosting ultrahigh power output for gelatin-KCl-FeCN4−/3− ionic thermoelectric cells. Adv. Energy Mater. 12, 2103666 (2022).
Li, Y. et al. Realizing record-high output power in flexible gelatin/GTA-KCl-FeCN4−/3− ionic thermoelectric cells enabled by extending the working temperature range. Energy Environ. Sci. 15, 5379–5390 (2022).
Li, Y. et al. Multilayer electrode strategy shorten thermal charging time and boost energy output in gelatin-based i-TE cells. Adv. Energy Mater. n/a, 2402621 (2024).
LeBlanc, S., Yee, S. K., Scullin, M. L., Dames, C. & Goodson, K. E. Material and manufacturing cost considerations for thermoelectrics. Renew. Sustain. Energy Rev. 32, 313–327 (2014).
Acknowledgements
S.-P.F. acknowledges the financial support from the General Research Fund (17203520, 17207422) and Collaborative Research Fund (C7082-21G, C6016-22G) from the Research Grants Council of Hong Kong Special Administrative Region and the Startup Grant of the City University of Hong Kong. W.S.L. acknowledges the support of the NSFC program for Distinguished Young Scholars (T2425012) and the Shenzhen Innovation Program for Distinguished Young Scholars (RCJC20210706091949018). Open Access made possible with partial support from the Open Access Publishing Fund of the City University of Hong Kong.
Author information
Authors and Affiliations
Contributions
Q.K.L., W.S.L. and S.P.F. conceived the idea. Q.K.L. and M.Y. designed experiments, analyzed data and wrote the paper. Q.K.L., M.Y., C.L.P. and X.Y.W. carried out experiments. Q.K.L. and S.H.W. conducted the characterizations. H.L. and Y.P.W. provided helpful suggestions and discussions. Q.K.L. and M.Y. All the authors revised the paper and made comments together. W.S.L. and S.P.F. directed the collaborative research, funding acquisition and supervision. All authors participated in the analysis and discussion.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Seok Woo Lee, who co-reviewed with Jia Li, Sheng Shen, and Peiyi Wu for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Li, Q., Yu, M., Pang, C. et al. Temperature adaptive self-regenerating ionic thermoelectric cycles for time domain thermal energy harvesting. Nat Commun 16, 8619 (2025). https://doi.org/10.1038/s41467-025-63645-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-63645-2