Abstract
Achieving uniform and stable hole transport layers (HTL) is crucial for large-area perovskite solar cells (PSCs). However, current self-assembled monolayer (SAM)-based HTL suffer from weak interfacial adhesion, poor film uniformity, and limited stability, constraining their scalability. Our study proposes an integrated HTL strategy with in situ SAM anchoring during NiOx synthesis, forming a type of scalable, high-performance, and durable HTL. This strategy significantly enhances molecular ordering, energy level alignment, and charge extraction, leading to a more pronounced performance improvement in large-area slot-die coating modules. PSCs with this HTL achieved a champion PCE of 26.02% (with an active area of 0.0655 cm2), while large-area modules reached 22.80% at 23.25 cm2, 21.45% at 87.45 cm2, and 20.21% at 749.276 cm2 (certified at 19.50%), showing superior scalability. Moreover, the irradiation and thermal stability, key concerns for commercial applications, were significantly improved. The encapsulated industrial-scale modules successfully passed the three module quality tests (MQTs) in the IEC 61215-2-2021 standard: outdoor exposure (MQT 08), UV pre-conditioning (MQT 10), and wet-high temperature operating life (MQT 13) tests.
Similar content being viewed by others
Introduction
P-i-n structured perovskite solar cells (PSCs) have demonstrated great potential in photovoltaics, particularly for commercial-scale large-area module applications, due to their high efficiency, low-temperature fabrication, and superior charge transport properties1,2,3,4. In recent years, with in-depth research into device structures and materials, significant progress has been made in the development of hole transport layers (HTL) based on self-assembled monolayers (SAM)5,6. SAM materials offer adjustable molecular structures7,8,9,10, excellent interface engineering capabilities11,12, and a simple solution deposition process13,14, which can enhance carrier transport efficiency by tuning the alignment of interface energy levels. By utilizing SAM as the HTL, the power conversion efficiencies (PCEs) of p-i-n type PSCs have surpassed 26.95%15, demonstrating their enormous potential in the development of high-efficiency photovoltaic devices.
However, for commercial-scale large-area modules, SAM-based HTL still suffers from inadequate interfacial adhesion16,17, poor film uniformity18, and inconsistencies in large-area deposition19,20, limiting device reproducibility and long-term operational stability21,22. Conventional SAM modification methods typically employ a segmented strategy. When SAM is directly deposited onto the surface of conductive metal oxides (ITO, FTO, and NiOx) via solution methods, the lack of sufficient surface functional groups such as hydroxyl (–OH) groups and charge anchoring sites on the substrate, results in low chemical activity, which makes it difficult for SAM molecules to form strong bonding during deposition23,24. As a result, weak interface adhesion leads to the gradual desorption of SAM due to environmental factors25,26. Additionally, the roughness on the surface of conductive metal oxides causes localized charge concentration and inadequate physical coverage, further diminishing the uniformity of the SAM and the overall interface bonding strength27,28. This non-uniformity not only affects the efficiency of charge injection at the interface, but also significantly increases charge recombination, thereby limiting further enhancement of the device performance29.
Recent studies have attempted to enhance the anchoring effect of SAM by introducing –OH groups or other active functional groups onto the surface of the underlying metal oxides. Han et al. used atomic-layer deposition to create an ITO substrate with a fully covalent –OH covered surface for SAM anchoring30. You et al. optimized the dispersion and surface –OH groups of NiOx nanoparticles (NPs) by H2O2 treatment, and improved the uniformity of self-assembly of [4-(3,6-dimethyl-9H-carbazol-9-yl) butyl] phosphonic acid (Me-4PACz) onto the surface of NiOx31. However, such segmented HTL strategies present inherent limitations: On one hand, it often requires multi-step pretreatment, which increases the complexity and cost of the process. On the other hand, the low-temperature solution deposition process lacks sufficient chemical energy to establish strong bonding between the SAM and the substrate32. While these strategies improve small-area device performance, they remain inadequate for ensuring the fabrication consistency and long-term stability required for large-area commercial modules.
To address these challenges, our study proposes an innovative integrated HTL strategy based on in situ SAM-coordinated NiOx NPs. wherein SAM molecules are incorporated during the synthesis of NiOx NPs, enabling direct chemical bonding between NiOx and SAM, thus addressing the deficiencies of conventional segmented HTL approaches in interfacial adhesion, film uniformity, and large-area deposition consistency. Specifically, we first synthesized high-quality surface hydroxylated NiOx (H–NiOx) with the assistance of Na2O2, and then achieved the in situ anchoring reaction of [2-(9H-carbazol-9-yl)ethyl] phosphonic acid (2PACz) with the –OH groups of H–NiOx surface by the hot injection method (integrated 2PACz–NiOx). Compared to conventional segmented HTL approaches, where SAM is deposited onto a preformed NiOx layer (post-functionalization strategy), the in situ-coordinated SAM in the integrated HTL exhibits a more uniform molecular arrangement, improved charge transport properties, and enhanced conductivity. This strategy enables one-step HTL deposition, significantly reducing non-uniformities in large-area film formation, making it ideal for industrial-scale PSC module fabrication.
Devices incorporating the integrated HTL achieved a champion PCE of 26.02% (0.0655 cm2). Furthermore, large-area perovskite solar modules fabricated via slot-die coating demonstrated PCEs of 22.80%, 21.45%, and 20.21% for active areas of 23.25 cm2, 87.45 cm2, and 749.276 cm2, respectively, confirming the superior scalability of this approach. More importantly, the strong anchoring effect of the SAM in integrated HTL further improves the operational stability of the devices. According to ISOS protocols, the integrated HTL-based PSCs retained 91% of their initial PCE after 1000 h under ISOS-L-3 conditions and 88% after 500 h under ISOS-D-3 conditions. Additionally, encapsulated modules successfully passed key IEC 61215-2-2021 reliability module quality tests (MQT 08, MQT 10, MQT 13), demonstrating the commercial viability of this strategy for high-performance large-area perovskite photovoltaic applications.
Results
Na2O2 pretreatment induces surface hydroxylation of NiOx NPs
In the integrated HTL strategy proposed in this study (Fig. 1a), the surface hydroxylation of NiOx serves as a crucial chemical anchoring site, which is related to the bridging uniformity and stability of the SAM shell. Density functional theory (DFT) calculations (Supplementary Fig. 1) indicate that the introduction of –OH groups provides adsorption sites for 2PACz, leading to a more stable adsorption configuration through hydrogen bonding. To achieve this, control NiOx (C–NiOx) was first synthesized via a conventional two-step method33,34, followed by Na2O2 treatment to induce surface hydroxylation, yielding hydroxyl-enriched NiOx (H–NiOx), and the reaction mechanism is shown in Supplementary Fig. 2. Transmission electron microscopy (TEM) images (Fig. 1b, c) show that the C–NiOx exhibits severe aggregation, forming irregular nanoparticles with an average size of ~9 nm. Following Na₂O₂ treatment, agglomeration was suppressed, and the particle size was decreased to ~5 nm. The particle size distribution statistics (Fig. 1d) further confirmed that H–NiOx exhibits a reduced average particle size (from 9 nm in C–NiOx to 6 nm) with a narrower size distribution. The root mean square (RMS) roughness of H–NiOx film is reduced from 15.50 nm to 8.06 nm after adding 3 mol% Na2O2 (Supplementary Fig. 3). The improvement of the NPs dispersion is beneficial to the anchoring of in-situ 2PACz and the reduction of the leakage current and recombination when applied as HTL in perovskite devices.
a Synthesis of H–NiOx and integrated 2PACz–NiOx NPs. b, c TEM images of C–NiOx and H–NiOx NPs. d Size distribution of C–NiOx and H–NiOx NPs. e Adsorption energy of –NO3 and –OH on NiOx NPs. f, g XPS spectra of Ni 2p core levels for C–NiOx and H–NiOx films.
Supplementary Fig. 4 shows the Fourier transform infrared (FTIR) spectra of C–NiOx and H–NiOx NPs. An obvious vibration peak of NO3− was observed at 1353 cm−1 in the C–NiOx, originating from the synthesis reaction35. Compared with the C–NiOx, the peak area ratio (A-OH/ANO3−) of H–NiOx was significantly enhanced, indicating that –OH effectively replaces surface-adsorbed NO3− ions. DFT calculations (Fig. 1e) reveal that the adsorption energy (Eads) of –OH (−6.78 eV) is lower than that of NO3− (−4.60 eV), further supporting the preferential substitution of NO3⁻ by –OH, leading to hydroxyl-enriched H–NiOx. Supplementary Fig. 5 shows the X-ray diffraction (XRD) patterns of the C–NiOx and H–NiOx NPs; similar peak position and higher peak intensity are observed from H–NiOx than those in the C–NiOx. Figure 1f, g shows the high-resolution of Ni 2p XPS spectra, the three characteristic peaks at 857.2, 855.6, and 853.9 eV represent the Ni4+, Ni3+ and Ni2+, respectively. It can be seen that compared with the C–NiOx, the peak intensity ratio (INi3+/INi2+) of H–NiOx significantly increased from 2.08 to 2.55, indicating that the oxidation of Na2O2 increased the content of Ni3+, which is conducive to inducing the formation of additional holes and improving the conductivity of the HTL36.
These characterizations confirmed that the Na2O2 treatment resulted in H–NiOx with uniform small particle size and purified surface hydroxyl groups, which provided a highly surface-active NiOx substrate for the composite HTL strategy, and the increased INi3+/INi2+ ratio also made it possible to prepare HTL with excellent performance.
In situ coordination of SAM on H–NiOx enables integrated HTL
To address the inhomogeneity and desorption of non-anchored molecules in the traditional segmented 2PACz–NiOx HTL, we used a hot injection method to achieve the in situ anchoring reaction of 2PACz onto –OH groups on the surface of H–NiOx NPs, forming the integrated 2PACz–NiOx HTL. Detailed synthesis process details can be found in the Supplementary Information. The heat generated during the hot injection provides sufficient energy to facilitate 2PACz anchoring onto hydroxyl-enriched H–NiOx. Centrifugation, washing, and redispersion steps effectively remove the unanchored molecules, yielding integrated 2PACz–NiOx with a uniform and well-ordered molecular arrangement. Different from the traditional two-step film formation method to prepare HTL, the integrated 2PACz–NiOx enables one-step film formation, simplifying the fabrication process (Fig. 2a).
a Schematic diagram of segmented 2PACz–NiOx and integrated 2PACz–NiOx films. b–d KPFM images of surface potential of H–NiOx, segmented 2PACz–NiOx and integrated 2PACz–NiOx films, respectively. e J–V characteristics of the ITO/H–NiOx, segmented 2PACz–NiOx and integrated 2PACz–NiOx/Ag devices in the dark. f Schematic illustration of energy level diagram for the PSCs devices with H–NiOx, segmented 2PACz–NiOx and integrated 2PACz–NiOx.
Supplementary Fig. 6 shows the FTIR spectra of H–NiOx, 2PACz and integrated 2PACz–NiOx, respectively. In integrated 2PACz–NiOx HTL, besides the Ni–O absorption peak at 668 cm−1, the appearance of characteristic (C = N at 1593 cm−1) and peak shifts confirm the successful anchoring of 2PACz. The TEM image of integrated 2PACz–NiOx is shown in Supplementary Fig. 7, due to the formation of a shell layer on the H–NiOx surface by 2PACz, the steric hindrance effect between particles makes the integrated 2PACz–NiOx show highly uniform dispersion. The time-of-flight secondary ion mass spectrometry (ToF-SIMS) with 3D images of segmented 2PACz–NiOx and integrated 2PACz–NiOx are shown in Supplementary Fig. 8, the results show that 2PACz in integrated 2PACz–NiOx was uniformly anchored on the H–NiOx surface in a bulk manner, which further confirmed the successful progress of the above reaction.
To verify the uniformity of the composite HTL layer, kevin probe microscopy (KPFM) was used to measure the surface potential distribution of H–NiOx, segmented 2PACz–NiOx and integrated 2PACz–NiOx, respectively (Fig. 2b–d). We found that the segmented introduced 2PACz had uneven coverage on the H–NiOx layer, while the integrated introduced 2PACz almost completely and evenly covered the surface of H–NiOx as expected. EDS mapping results of P element from segmented 2PACz–NiOx and integrated 2PACz–NiOx further confirms the difference in uniformity between the two introduction methods of 2PACz (Supplementary Fig. 9).
Uniform film formation is beneficial to the further preparation of HTL with excellent electrical properties. Figure 2e shows the conductivity of H–NiOx, segmented 2PACz–NiOx and integrated 2PACz–NiOx on ITO substrates. The conductivity of integrated 2PACz–NiOx is significantly improved due to the uniform and well-ordered molecular anchoring on the surface. The ultraviolet photoelectron spectrometer (UPS) spectra of H–NiOx, segmented 2PACz–NiOx, integrated 2PACz–NiOx and perovskite films are shown in Supplementary Figs. 10, 11, respectively. We calculated the Fermi level (EF) and valence band maximum (VBM) of the binding energy from the UPS measurement, the conduction band minimum (CBM) was obtained from the VBM and Eg values measured by the UV-visible absorption spectrum (Supplementary Figs. 10, 11 and Supplementary Table 1)37,38, and the energy level diagram was drawn as shown in Fig. 2f. Compared with H–NiOx, the VBM of segmented 2PACz–NiOx and integrated 2PACz–NiOx are both shifted downward, among which the VBM of integrated 2PACz–NiOx is further shifted to −5.45 eV, which enhances the energy level matching between the HTL and the perovskite layer, providing more efficient hole extraction. Supplementary Fig. 12 shows the contact angle of the perovskite solution on three different films. The segmented 2PACz–NiOx HTL exhibits a larger contact angle than H–NiOx. Due to the more uniform and orderly of 2PACz in the integrated HTL, the contact angle is further increased, enhancing the Gibbs free energy for heterogeneous nucleation39.
Bottom interface crystallization kinetics
Epoxy resin-assisted exfoliation was employed to peel the perovskite films from ITO/H–NiOx, ITO/segmented 2PACz–NiOx and ITO/integrated 2PACz–NiOx, enabling the investigation of perovskite crystallization on different HTLs40,41. The preparation and exfoliation processes of perovskite films are shown in Supplementary Fig. 13 and Fig. 3a. Supplementary Fig. 14 shows an image of the exfoliated perovskite film from ITO/integrated 2PACz–NiOx.
a Exfoliation method of the bottom perovskite films. b–d SEM images for the bottom of the perovskite films on H–NiOx, segmented 2PACz–NiOx and integrated 2PACz–NiOx. e–g GIWAXS 2D patterns for the bottom of the perovskite films from H–NiOx, segmented 2PACz–NiOx and integrated 2PACz–NiOx. h–j PL maps of the exfoliated perovskite films from H–NiOx, segmented 2PACz–NiOx and integrated 2PACz–NiOx.
Bottom-view scanning electron microscope (SEM) images of exfoliated perovskite films from three different HTLs are displayed in Fig. 3b–d, with corresponding cross-sectional SEM images shown in Supplementary Fig. 15. Perovskite films grown on H–NiOx exhibit a small, inhomogeneous grain size with noticeable PbI2 precipitation and pronounced stratification during the longitudinal growth. Comparatively, the perovskite film on segmented 2PACz–NiOx exhibits a more uniform morphology with larger grains, and the stratification effect is significantly suppressed. Furthermore, perovskite films on integrated 2PACz–NiOx exhibit a flatter and denser morphology with large grain size and no observable PbI2 precipitation. Cross-sectional SEM reveals vertically aligned columnar grain growth, which facilitates efficient carrier transport in the full device stack.
The XRD patterns of the bottom perovskite were shown in Supplementary Fig. 16. The precipitation of PbI2 is promoted by the electron-proton transfer reaction between the overcoordinated Ni3+ sites and the perovskite precursor35,42,43, which is observed in the H–NiOx sample but is suppressed after the introduction of 2PACz. Grazing incidence wide-angle X-ray scattering (GIWAXS) was used to further investigate the perovskite crystallinity at the buried interface, as displayed in Fig. 3e–g. The diffraction rings located at the q values of 10.16 nm−1, 14.38 nm−1, 17.57 nm−1, and 20.28 nm−1 correspond to the (100), (111), (120), and (200) crystallographic planes, respectively44. In addition, a prominent diffraction ring corresponding to PbI2 was observed at q of 9.27 nm−1 in the H–NiOx sample. After the introduction of 2PACz, the diffraction ring of PbI2 on segmented 2PACz–NiOx is significantly weakened, while the bottom of the perovskite on integrated 2PACz–NiOx further exhibits standard α-phase perovskite with no diffraction ring of PbI2. This is consistent with the conclusion of the XRD study. The 1D integral analysis for GIWAXS patterns was conducted in Supplementary Fig. 17, which further illustrates the suppression of PbI2 peak and enhanced crystallinity at the perovskite-HTL interface in the integrated 2PACz–NiOx HTL.
In addition, according to the photoluminescence (PL) mapping of the peeled perovskite films (Fig. 3h–j), it can be seen that after introducing 2PACz in two different methods, both the PL intensities of the perovskite films are significantly higher than that of the H–NiOx sample. Notably, compared with segmented 2PACz–NiOx HTL, PL Mapping of integrated 2PACz–NiOx HTL exhibits greater uniformity, indicating reduced non-radiative recombination and the formation of high-quality perovskite films.
Photovoltaic performance and stability
To systematically evaluate the optoelectronic performance and operational stability of devices, we prepared p-i-n PSCs incorporating three different HTLs. The device architecture of p-i-n PSCs (ITO/integrated 2PACz–NiOx/perovskite/C60/BCP/Ag) was revealed in Fig. 4a. Photoluminescence (PL) and time-resolved PL (TRPL) spectra of different perovskite films were measured first, as shown in Fig. 4b, c. The PL intensity of perovskite films on integrated PACz-NiOx at 798 nm is lower than that on H–NiOx and segmented PACz-NiOx, indicating enhanced hole transport efficiency. Supplementary Table 2 shows the average carrier lifetime (τave) of perovskite films, the sample of ITO/segmented 2PACz–NiOx/perovskite exhibited a τave of 276.70 ns, which is shorter than that of the ITO/H–NiOx/perovskite film (464.51 ns). With integrated 2PACz–NiOx HTL, τave further decreased to 212.41 ns, confirming enhanced hole extraction and collection in the perovskite layer.
a Structural diagram of the device. b, c Steady-state PL and TRPL spectra of perovskite films on ITO/H–NiOx, ITO/segmented 2PACz–NiOx and ITO/integrated 2PACz–NiOx. d, e The J–V curves of devices with the active area of 0.0655 cm2 and 1 cm2. f EQE and integrated current density spectra. g Steady-state power outputs of the different devices. h, i ISOS-L-3 and ISOS-D-3 stability tests for the different devices based on H–NiOx, segmented 2PACz–NiOx and integrated 2PACz–NiOx.
Furthermore, the segmented 2PACz–NiOx HTL-based PSCs achieved a champion PCE of 25.12% with an active area of 0.0655 cm2 (Fig. 4d), with an open-circuit voltage (Voc) of 1.181 V, a short-circuit current density (Jsc) of 26.01 mA/cm2 and a fill factor (FF) of 81.78%. With integrated 2PACz–NiOx HTL, device performance is further improved compared with the H–NiOx device (Voc of 1.098 V, Jsc of 25.26 mA/cm2, FF of 79.03%, and PCE of 21.92%), achieving a champion PCE of 26.02% (Voc of 1.189 V, Jsc of 26.06 mA/cm2, and FF of 83.98%), and the forward and reverse scan curves of three different devices are shown in Supplementary Fig. 18. In addtition, the devices with an active area of 1 cm2 were also prepared to initially explore the advantages of integrated 2PACz–NiOx film uniformity in large-area PSCs (Fig. 4e). Compared with the PSCs based on H–NiOx and segmented 2PACz–NiOx, the integrated 2PACz–NiOx exhibits an enhanced Voc of 1.184 V, FF of 82.83%, and PCE of 24.89%. Figure 4f shows the external quantum efficiency (EQE) spectrum. The EQE of segmented 2PACz–NiOx and integrated 2PACz–NiOx devices mainly increase in the range of 330–800 nm than that of H–NiOx, resulting in the increased Jsc (24.51-25.45-25.59 mA/cm2). These results are consistent with those of the J–V curves.
To further characterize the charge transport and recombination behavior, a series of measurements were performaed. The dark J–V curves of the three devices are shown in Supplementary Fig. 19. The segmented 2PACz–NiOx device showed a J0 of 1.12 × 10−5 mA/cm2, which is lower than the J0 of H–NiOx sample (2.85 × 10−5 mA/cm2). When integrated 2PACz–NiOx is used as the HTL, the J0 of the device is further reduced (5.43 × 10−6 mA/cm2), which confirms that the non-radiative coincidence of charge carriers in the integrated 2PACz–NiOx device is more effectively suppressed. Supplementary Fig. 20 shows the Nyquist plots of the electrochemical impedance spectroscopy (EIS) measurement of H–NiOx, segmented 2PACz–NiOx and integrated 2PACz–NiOx devices in the low and high frequency ranges. The fitting circuit of the EIS measurement is shown in Supplementary Fig. 21, and the fitting parameters are summarized in Supplementary Table 3. The device based on integrated HTL exhibited a lower Rct of 78.19 Ω and higher Rrec of 9101 Ω, indicating faster carrier transport and suppressed nonraditative recombination.
The transient photovoltage (TPV) curve in Supplementary Fig. 22 shows that the integrated 2PACz–NiOx HTL-based device exhibits a longer decay time to 50% photovoltage (344 μs) compared to that of the H–NiOx (221 μs) and segmented 2PACz–NiOx (262 μs), indicating enhanced suppression of non-radiative recombination. Supplementary Fig. 23 presents the relationship between Voc versus and the logarithm of light intensity. Through linear fitting, the ideality factor of the integrated 2PACz–NiOx-based device (1.25) is significantly lower than that of the H–NiOx (1.98) and segmented 2PACz–NiOx (1.48), reflecting the lower defect state density. The defect density was futher evaluated using the space-charge-limited current (SCLC) technique with the device structure of ITO/HTL/perovskite/Spiro-OMeTAD/Au (Supplementary Fig. 24)45. The relative trap state density (Nt) and trap-filled limit voltage (VTFL) were calculated for different films. Compared to H–NiOx, the Nt of hole-only device based on segmented 2PACz–NiOx decreased from 2.41 × 1015 cm−3 to 1.83 × 1015 cm−3, and the Nt of the integrated device further decreased to 1.73 × 1015 cm−3. Thermal admittance spectroscopy (TAS) analysis was used to further quantify the energy distribution of the trap density of states (tDOS) of segmented 2PACz–NiOx and integrated 2PACz–NiOx, as shown in Supplementary Fig. 25. In the shallow energy level region, the defect density of integrated 2PACz–NiOx is lower than that of segmented 2PACz–NiOx, which is attributed to the reduction of interface trap states and the formation of ordered dipole layers in integrated 2PACz–NiOx, thereby improving the interface contact between perovskite and HTL and promoting efficient hole extraction.
In addition to the high efficiency, the long-term operation performance is a critical parameter for PSCs. The photocurrent steady-state output (SPO) was measured for 300 s at the maximum power point (MPP) to evaluate device stability under operational conditions (Fig. 4g). The integrated 2PACz–NiOx device shows a stable PCE of 26.02% (biased 1.04 V) and Jsc 25.01 mA/cm2, demonstrating the best operational stability among the three devices. Advanced ISOS stability tests (ISOS-L-3, light soaking; ISOS-D-3, shelf life) were conducted to assess long-term device reliability (Fig. 4h, i)46. Compared to H–NiOx and segmented 2PACz–NiOx, the integrated 2PACz–NiOx device demonstrates significantly improved long-term stability, which should be attributed to more ordered and stable molecular anchoring, resulting in a more uniform and denser buried interface30. While forming a perovskite film with lower defect state density, the migration of halogen ions during long-term operation is suppressed. As a result, the integrated 2PACz–NiOx device retained 88% of its initial PCE after 1000 h (ISOS-L-3) and 91% after 500 h (ISOS-D-3), further validating its reliability for prolonged operation.
Scalability and operational stability for commercial applications
Since the integrated 2PACz–NiOx HTL mitigates SAM inhomogeneity in large-area film formation, it exhibits greater potential for perovskite module fabrication. Perovskite film was deposited by a slot-die coating process under atmospheric conditions (Supplementary Fig. 26). By optimizing the coating parameters for different areas separately and the final parameters were determined (Supplementary Table 4). A 12-point thickness check confirmed that the film had good thickness uniformity (Supplementary Fig. 27). The module structure is FTO/ integrated 2PACz–NiOx/perovskite/C60/BCP/Cu, and the cross-sectional SEM image is shown in Supplementary Fig. 28. PSC modules were fabricated with active areas of 23.25 cm2, 87.45 cm2 and 749.276 cm2, respectively.
As a result, PSC modules based on integrated 2PACz–NiOx achieved PCEs of 22.80% (Voc of 9.106 V, Jsc of 3.144 mA/cm2, and FF of 79.64%), 21.45% (Voc of 16.92 V, Jsc of 1.657 mA/cm2, and FF of 76.51%) and 20.21% (Voc of 54.15 V, Jsc of 0.490 mA/cm2, and FF of 76.17%; certified at 19.50%) for active areas of 23.25 cm2, 87.45 cm2 and 749.276 cm2, respectively (Fig. 5a–c and Supplementary Fig. 29). Detailed module performance statistics are summarized in Supplementary Tables 5–7. Notably, the performance enhancement of the integrated 2PACz–NiOx HTL strategy becomes more pronounced in larger-area modules.
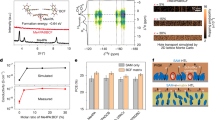
a–c The J–V curves of solar modules with the active area of 23.25 cm2, 87.45 cm2 and 749.276 cm2, respectively. d Statistic of PCE, \(\Delta\)PCE and S2 of PSCs with different active areas based on 15 devices. e Schematic diagram of SAM distribution with different active areas. f–h IEC 61215-MQT 08, IEC 61215-MQT 10 and IEC 61215-MQT 13 of the encapsulated solar modules with the active area of 749.276 cm2 based on segmented 2PACz–NiOx and integrated 2PACz–NiOx.
To validate this observation, 15 devices with active areas of 0.0655 cm2, 1 cm2, 23.25 cm2, 87.45 cm2 and 749.276 cm2 were prepared, the repeatability parameter statistics are shown in Supplementary Tables 8–22, and the PCE distribution statistics are summarized in Fig. 5d. We also calculated the relative PCE increment (\(\Delta\)PCE) of segmented 2PACz–NiOx and integrated 2PACz–NiOx compared with H–NiOx. In addition, the variance (S2) statistics of the PCE distribution of 15 devices under different active areas are analyzed to assess the reproducibility of different HTL preparation methods. Due to the limitations of the slot-die coating process, the non-uniform distribution and inadequate anchoring of SAM in segmented 2PACz–NiOx lead to suboptimal efficiency improvement in large-area modules (\(\Delta\)PCE of 7.92% for the modules with the active area of 749.276 cm2), and significant efficiency variation among samples of the same area (S2 of 0.663). On the contrary, the integrated 2PACz–NiOx, employing in-situ SAM introduction and one-step film formation, enables uniform SAM anchoring, effectively enhancing PCE in large-area modules (\(\Delta\)PCE of 23.16% for 749.276 cm2) while maintaining minimal efficiency variation (S2 of 0.331).
Supplementary Fig. 30 shows the PL maps of large-area modules for devices based on segmented 2PACz–NiOx and integrated 2PACz–NiOx. The non-uniform SAM coverage in the segmented 2PACz–NiOx leads to poor quality of perovskite films, whereas the one-step film formation of integrated 2PACz–NiOx provides a high-quality substrate for the uniform growth of perovskite. To intuitively compare the difference in SAM distribution uniformity under two HTL fabrication methods, the contact angles of the perovskite solution on the two HTLs were tested at different positions on a 30 cm × 40 cm substrate. The point positions and measurement results are shown in Supplementary Figs. 31, 32, respectively. Based on integrated 2PACz–NiOx, the contact angles of the perovskite solution at different positions exhibit smaller differences, which is due to the uniform and orderly anchoring of 2PACz, confirming the results of large-area PL maps. Figure 5e shows the schematic diagram of the SAM coverage variations during the large-area scaling. This demonstrates that the integrated 2PACz–NiOx strategy effectively mitigates SAM-related defects in large-area preparation, providing a viable approach for PCE enhancement in PSC modules. As a complementary comparison, we summarized the reported values of PSCs with similar active areas (Supplementary Fig. 33, Supplementary Table 23). Excitingly, the PCEs of devices with different areas baesd on integrated 2PACz–NiOx are all at a high level, which further proves its good reference value for the application of scalable moudles.
For industrial applicability, PSC modules with the active area of 749.276 cm2 based on segmented 2PACz–NiOx and integrated 2PACz–NiOx were encapsulated through the multilayer thin-film encapsulation (MFE) technique (Supplementary Fig. 34)47. The ISOS stability test (ISOS-L-2, light soaking) was used to preliminarily evaluate the operational stability under elevated temperatures (Supplementary Fig. 35), Notably, the PCE of the encapsulated modules based on integrated 2PACz–NiOx retained more than 95% of the initial PCE after 2000 h. Three key sequence tests (MQT 08, MQT 10 and MQT 13) in IEC 61215-2-2021 were further adopted as the guideline to evaluate the outdoor exposure durability, UV pretreatment effects, and wet high-temperature operating life (WHTOL, double 85 condition) for device stability (Fig. 5f–h)22,48. The detailed protocols of the three sequence tests are summarized in Supplementary Table 24, and images of industrial testing equipments are shown in Supplementary Figs. 36–38. According to the I 3 d XPS spectra (Supplementary Fig. 39) and SEM images (Supplementary Fig. 40) of the bottom perovskite before and after aging, compared with the obvious halogen ion migration and interface degradation in the segmented 2PACz–NiOx, the integrated 2PACz–NiOx alleviates the degradation of the buried interface due to its uniform and dense molecular anchoring, reducing the attenuation of device performance. Following outdoor exposure (cumulative irradiance: 60 KWh/m2), total UV irradiation (15 KWh/m2), and WHTOL testing (1000 h), the modules based on integrated 2PACz–NiOx retained over 95% of the initial efficiency (Initial/final PCEs are summarized in Supplementary Table 25), meeting module testing standards and demonstrating strong excellect industrial application potential.
Discussion
This study presents an integrated HTL strategy based on in situ SAM-coordinated NiOx, overcoming the limitations of conventional segmented HTL, where SAMs are deposited onto pre-formed NiOx layers. By achieving one-step in situ anchoring during NiOx synthesis, this approach significantly enhances interfacial adhesion, film uniformity, and charge transport properties, making it highly suitable for large-area PSC fabrication. PSC devices with this integrated HTL achieved a champion PCE of 26.02% (0.0655 cm2), while slot-die coating large-area modules reached 22.80% (23.25 cm2), 21.45% (87.45 cm2), and 20.21% (749.276 cm2). The strategy also demonstrated excellent stability, retaining 91% of the initial PCE after 1000 h (ISOS-L-3) and 88% after 500 h (ISOS-D-3). Furthermore, 749.276 cm2 encapsulated modules passed IEC 61215-2-2021 reliability tests (MQT 08, MQT 10, MQT 13), confirming its commercial viability. This integrated HTL strategy effectively addresses the scalability and stability challenges of SAM-based HTLs, providing a robust and industrially viable pathway for high-performance, large-area PSCs.
Method
Materials
N, N′-dimethylformamide (DMF), dimethyl sulfoxide (DMSO), isopropyl alcohol (IPA), chlorobenzene (CB) and ethanol (EtOH) were purchased from Sigma-Aldrich. Lead iodide (PbI2), lead bromide (PbBr2), cesium iodide (CsI), methylamine hydrochloride (MACl), methylamine hydrobromide (MABr), formamidine hydroiodide (FAI), phenethyl ammonium iodide (PEAI), ethylene diammonium diiodide (EDADI) and 2PACz were purchased from Xi’an Yuri Solar Co., Ltd. Sodium hydroxide (NaOH), sodium peroxide (Na2O2) and nickel nitrate hexahydrate (Ni(NO3)2·6H2O) were supplied by Shanghai Aladdin Biochemical Technology Co., Ltd. 2,9-dimethyl-4,7-diphenyl-1,10-phenanthroline (BCP) and C60 were from Tokyo Chemical Industry. Epoxy resin (JIKOOBOND-8103) was from Dongguan Ji. Koobond Industry Investment Co., Ltd.
Synthesis of C–NiOx and H–NiOx
NiOx NPs were synthesized by chemical precipitation method33,34,35. Ni(NO3)2·6H2O (0.5 mol) was dissolved in deionized water (100 mL) and stirred at room temperature to obtain a green solution. Then, adjusting the solution pH to 10 by dropping NaOH solution (10 mol/L). After stirring for 10 min, the resulting colloidal precipitate was washed three cycles of centrifugation with deionized water and absolute ethanol. The prepared product was dried in vacuum at 65 °C for 12 h and then calcined at 270 °C for 2 h to obtain dark-black control NiOx NPs (C–NiOx). The H–NiOx solution was obtained by adding Na2O2 (3 mol%) to the C–NiOx aqueous dispersion (20 mg/mL), and after stirring at room temperature for 20 min, H–NiOx NPs were further obtained by centrifugation and drying.
Synthesis of integrated 2PACz–NiOx
The collected H–NiOx NPs were redispersed in IPA (30 mL) and heated to 65 °C. 2PACz (0.25 mg/mL) was added dropwise to the IPA dispersion and stirred for 5 h, and then cooled naturally to room temperature. The solution was washed three times of centrifugation with EtOH to remove unanchored SAM molecules, and the integrated 2PACz–NiOx NPs were collected after drying and redispersed in DMF (20 mg/mL) for later use.
Exfoliation processes
JIKOOBOND-8103 UV resin (20 μL) was dripped onto the surface of the prepared perovskite film, and a transparent glass was used to cover the resin. The sample was fixed with clips around the edges and then transferred to the UV lamp (398 nm, 20 W) for 90 s. After UV curing, the perovskite was peeled off from the HTL side with a knife to complete the exfoliation processes.
Small-size solar cells fabrication
ITO/glass substrates were cleaned sequentially with detergent, deionized water, acetone/ethanol (1:1, v/v) mixture, and deionized water. After drying, the substrates were treated with UV ozone for 5 min. H–NiOx and integrated 2PACz–NiOx layers were deposited onto the ITO substrate by spin-coating at 2000 r.p.m. for 30 s, and annealed at 110 °C for 10 min in ambient atmosphere. Segmented 2PACz–NiOx layer is prepared in two steps. First, the NiOx film was prepared by the same deposition process as H–NiOx, then 2PACz (0.5 mg/mL in EtOH) was further spin-coating at 3000 r.p.m. for 30 s on the NiOx film, and annealed at 100 °C for 10 min to obtain the segmented 2PACz–NiOx layer. PbI2 (2% excess), PbBr2, CsI, MABr and FAI were mixed with the chemical formula of Cs0.05(FA0.95MA0.05)0.95Pb(I0.95Br0.05)3 with 12% MACl addition in DMF/DMSO (4:1, v/v) and stirred for 3 h to prepare perovskite precursor solution (1.4 M). The perovskite films were deposited by spin-coating (1000 r.p.m., 10 s/5000 r.p.m., 30 s), 200 μL of CB was used as an antisolvent 10 s before the end of the spinning, and then the film was annealed at 100 °C for 60 min. PEAI (0.2 mg/mL) and EDADI (0.1 mg/mL) were dispersed together in IPA solution and spin-coated on the perovskite surface at 5000 r.p.m. for 30 s as a passivation layer. C60 (40 nm) and BCP (6 nm) were deposited by thermal evaporation sequentially. Finally, the Ag electrode was deposited by thermal evaporation with a thickness of 120 nm and an area of 0.0655 cm2 and 1 cm2.
Solar modules Fabrication
PSC modules of 23.25 cm2, 87.45 cm2 and 749.276 cm2 were prepared on FTO glass substrates with a size of 7 cm × 7 cm, 12 cm × 12 cm and 30 cm × 40 cm. The serial interconnection of the module was realized by P1, P2, and P3 lines, which were patterned using a laser scribing system with 1064 nm (P1) and 532 nm (P2 and P3), respectively. The effect of P1-P2-P3 cutting is imaged using a 3D confocal microscope (VK-X3050, KEYENCE, America) and showed in Supplementary Fig. 41. The HTLs and perovskite layers were prepared by a slot-die-coating process. PSC modules of 23.25 cm2, 87.45 cm2 were prepared by precision medium-sized slot-die coating device (ST-200A, Suzhou Honest Intelligent Science and Technology Co., Ltd., China) and small vacuum dryer (KT-CM150, Suzhou Kejingte Intelligent Technology Co., Ltd., China). The precision large-sized slot-die coating device (SC300, Huzhou Yihuo Optoelectronics Co., Ltd.) and vacuum drying apparatus (CH-SZ300A, Huzhou Yihuo Optoelectronics Co., Ltd.) are used to prepare the large modules of 749.276 cm2. Taking a 23.25 cm2 module as an example, fill the H–NiOx and integrated 2PACz–NiOx solution into the slot-die mold, the height between the substrate and the die was kept at 110 μm, with a coating speed of 15 mm/s and a dispense rate of 1.25 μL/s to prepare the H–NiOx and integrated 2PACz–NiOx layers, respectively. 2PACz solution was further coated (120 μm, 10 mm/s and 0.55 μL/s) on the H–NiOx film to prepare the segmented 2PACz–NiOx layer. The annealing process of the above HTLs is consistent with the small-size solar cells. The perovskite precursor solution (1.0 M) of Cs0.05(FA0.95MA0.05)0.95Pb(I0.95Br0.05)3 with 12% MACl addition was mixed in DMF/DMSO (9:1, v/v) and stirred for 3 hours. The height between the substrate and the die was kept at 100 μm, with a coating speed of 10 mm/s and a dispense rate of 1.01 μL/s. The freshly coated precursor film was light yellow in color, which was then subjected to a vacuum extraction step. Maintain the vacuum pressure of 10 Pa for 60 s and then return to atmospheric pressure. After the vacuum extraction step, the perovskite film was annealed at 150 °C for 20 mins. The IPA solution of PEAI (0.2 mg/mL) was coated as a passivation layer. C60, BCP deposition process is consistent with small-size solar cells. Finally, 80 nm Cu electrode was deposited by thermal evaporation.
Encapsulation progress
A multilayer thin-film encapsulation technique (MFE) has been applied to encapsulate the devices45. Si3N4 of about 2 μm was prepared by plasma enhanced chemical vapor deposition (PECVD) first, and then acrylate of about 10 μm was prepared by ink-jet printing, finally Si3N4 of about 2 μm was prepared by PECVD to complete the encapsulation. The cover glass is 30 cm × 40 cm × 0.11 cm in size, with 29 cm × 39 cm of polyolefin encapsulant (Mitsui, Solar ASCE TR02BA, 500 μm) between the solar module and the cover glass. A butyl rubber edge seal (Quanex SET LP03, 3948) was applied to the glass as a frame, 0.5 cm from its edge. The sample is laminated in a vacuum laminator (KT-DX4040, Suzhou Kejingte Intelligent Technology Co., Ltd., China) at 120 °C. The vacuum in the chamber is reduced to a minimum within 300 s. Then, the pressure applied to the sample is gradually increased to 180 kPa within 120 s and maintained at 180 kPa for 450 s, after which the pressure is released and the chamber is returned to atmospheric pressure. Finally, the sample is placed outside the laminator for air cooling.
Characterization
Crystalline structure was obtained by XRD (Bruker, D8 ADVANCE, Germany), and the shape and particle size of NiOx NPs were measured by TEM (JEOL JEM-2100). XPS (Thermo Fisher, ESCALAB Xi + , America) was employed to study the chemical bonding state. Fourier transform infrared spectroscopy (Nicolet iS50) confirmed chemical reactions. The surface wettability of the film was measured by optical contact angle measurement (DS 100S), and the surface uniformity of different films was characterized by AFM (SPM-9700HT). SEM (FEI, Quanta250, America) were used to investigate the morphology of perovskite films. GIWAXS measurements were performed at beamline BL14B1 of the Shanghai Synchrotron Radiation Facility. PL and TRPL spectra were carried out by Spectrofluorometer (Edinburgh instruments, FS5, UK). PL maps of small-area perovskite films were carried out by a laser Raman spectrometer (Renishaw inVia Qontor Extect electronics co., Hong Kong, China). The absorption/transmission properties of NiOx and perovskite films were determined using a UV-vis spectrometer (HITACHI, U-3010, Japan). The J–V measurements of the modules with an active area of 749.276 cm2 was performed by a steady-state solar module simulator (GSIV-22A0606P, Gsolar Power Co., Ltd.). The light source adopts LED combined light source, and the intensity of the light was 100 mW/cm calibrated by a KG-5 Si diode. The J–V measurements of other active area devices (0.0655 cm2, 1 cm2, 23.25 cm2 and 87.45 cm2) were performed by a chemical workstation (CHI660E, Chenhua Instrument Corp, China). The light source was a solar simulator (Newport, 94023 A) to match AM 1.5 G. The intensity of the light was 100 mW/cm2, calibrated by a KG-5 Si diode. The operational stability test was measured at the MPP for the unencapsulated devices (SCS600, Zolix Instruments Co., Ltd.). The EIS was performed on an Electrochemical Workstation (CHI600E, Chenhua Instrument Crop, China), and the data were analyzed by the Z-View program. The EQE measurements using a solar cell Spectral Response/QE/IPCE Measurement System (Solar Cell Scan 100, ZOLIX INSTRUMENTS CO., LTD, China) were calibrated with a standard single-crystal Si photovoltaic cell. ISOS-L-3 tests were carried out under MPP tracking by Solar Cell Burn-in System (SCS600 series, ZOLIX INSTRUMENTS CO., LTD, China). The PL maps of the module perovskite films were obtained using the perovskite battery EL/PL test machine (GPEL-P0303, Gsolar Power Co., Ltd.). IEC 61215-MQT 08, MQT-10, and MQT-13 using solar cell light decay test box (GCD-24A1206, Gsolar Power Co., Ltd.), single lamp UV test box (Shanghai Hou Yao Test Equipment Co., Ltd.) and wet high temperature environment box (Zhenghang Instrument Equipment Co., Ltd.), respectively.
DFT Calculations
All DFT calculations were performed with VASP49,50,51 using the Perdew-Burke-Ernzerhof generalized gradient approximation (GGA) for solids (PBEsol)52 as the exchange-correlation functional in combination with the projector-augmented wave method53 to describe the interactions between the core ions and valence electrons. The plane-wave cutoff was set to 520 eV. GGA + U approximation54 was adopted to account for the on-site Coulomb interaction between the localized 3 d electrons of Ni atoms, with the effective Hubbard U parameter set to 7.0 eV, which results in a 3.8 eV band gap of bulk NiO in good agreement with experiments. The non-polar (001) surface was selected in all calculations, which was represented by a slab model constructed based on the optimized cubic (Fm-3m) NiO bulk structure. The slab model contains eight layers with each layer consisting of 8 Ni2+ and 8 O2– ions, accordingly, the parameters of the two-dimensional square lattice of the surface model is a = b = 8.24 Å. This is the smallest lattice that can represent the standard cubic structure and host the antiferromagnetism of NiO. The PBEsol+U optimized lattice parameters correspond to 4.09 Å of the standard Fm-3m, slightly smaller than the experimental value ~ 4.17 Å measured at above 400 °C55, confirming that the choice of DFT calculation parameters is adequate. Bared as well as OH− and NO3− adsorbed NiO surfaces were calculated using this model with using a 6 × 6 × 1 Γ-centered k-point mesh for the Brillouin zone integration. The Na+ cations were placed to be bonded to a surface O2– cation to ensure the charge neutrality of the computational models for both OH− and NO3− adsorption. 2PACz takes a bidentate motif when adsorbed at the NiO (001) surface. The introduction of OH− stabilizes the 2PACz and NiO structures through hydrogen bonding, reducing the Eads from −3.29 eV to −4.38 eV.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Source data are provided with this paper.
References
Sangale, S. S. et al. Colloidal ink engineering for slot-die processes to realize highly efficient and robust perovskite solar modules. Adv. Mater. 37, 2420093 (2025).
Du, J. et al. Face-on oriented self-assembled molecules with enhanced π–π stacking for highly efficient inverted perovskite solar cells on rough FTO substrates. Energy Environ. Sci. 18, 3196–3210 (2025).
Shin Thant, K.K. et al. Comprehensive review on slot-die-based perovskite photovoltaics: mechanisms, materials, methods, and marketability. Adv. Energy Mater. 15, 2403088 (2025).
Yang, Z. et al. Slot-die coating large-area formamidinium-cesium perovskite film for efficient and stable parallel solar module. Sci. Adv. 7, eabg3749 (2021).
Al-Ashouri, A. et al. Conformal monolayer contacts with lossless interfaces for perovskite single junction and monolithic tandem solar cells. Energy Environ. Sci. 12, 3356–3369 (2019).
Isikgor, F. H. et al. Molecular engineering of contact interfaces for high-performance perovskite solar cells. Nat. Rev. Mater. 8, 89–108 (2023).
Li, Z. et al. Stabilized hole-selective layer for high-performance inverted p-i-n perovskite solar cells. Science 382, 284–289 (2023).
Wu, H. et al. Tailored lattice-matched carbazole self-assembled molecule for efficient and stable perovskite solar cells. J. Am. Chem. Soc. 147, 8004–8011 (2025).
Zhang, X. et al. A spiro-type self-assembled hole transporting monolayer for highly efficient and stable inverted perovskite solar cells and modules. Energy Environ. Sci. 18, 468–477 (2025).
Li, M., Liu, M., Qi, F., Lin, F. R. & Jen, A. K. Y. Self-assembled monolayers for interfacial engineering in solution-processed thin-film electronic devices: design, fabrication, and applications. Chem. Rev. 124, 2138–2204 (2024).
He, X. et al. Enhancing hole transport uniformity for efficient inverted perovskite solar cells through optimizing buried interface contacts and suppressing interface recombination. Angew. Chem. Int Ed. 63, e202412601 (2024).
Qu, S. et al. Redox mediator-modified self-assembled monolayer stabilizes a buried interface in efficient inverted perovskite solar cells. Energy Environ. Sci. 18, 3186–3195 (2025).
Pininti, A. R. et al. Resolving scaling issues in self-assembled monolayer-based perovskite solar modules via additive engineering. Adv. Energy Mater. 15, 2403530 (2025).
Ren, Z. et al. Poly(carbazole phosphonic acid) as a versatile hole-transporting material for p-i-n perovskite solar cells and modules. Joule 7, 2894–2904 (2023).
National Renewable Energy Laboratory, best research-cell efficiency chart. https://www.nrel.gov/pv/cell-efficiency (2025).
Li, C. et al. Pros and cons of hole-selective self-assembled monolayers in inverted PSCs and TSCs: extensive case studies and data analysis. Energy Environ. Sci. 17, 6157–6203 (2024).
Dai, Z. et al. Connecting interfacial mechanical adhesion, efficiency, and operational stability in high-performance inverted perovskite solar cells. ACS Energy Lett. 9, 1880–1887 (2024).
Zhou, Y. et al. Interfacial modification of NiOx for highly efficient and stable inverted perovskite solar cells. Adv. Energy Mater. 14, 2400616 (2024).
Liu, S. et al. Buried interface molecular hybrid for inverted perovskite solar cells. Nature 632, 536–542 (2024).
Guo, R. et al. Refining the substrate surface morphology for achieving efficient inverted perovskite solar cells. Adv. Energy Mater. 13, 2302280 (2023).
Howard, I. A. et al. Coated and printed perovskites for photovoltaic applications. Adv. Mater. 31, 1806702 (2019).
Dong, B. et al. Self-assembled bilayer for perovskite solar cells with improved tolerance against thermal stresses. Nat. Energy 10, 342–353 (2025).
Wang, D. et al. Rigid molecules anchoring on NiOx enable >26% efficiency perovskite solar cells. Joule 9, 101815 (2025).
He, J. et al. Improved anchoring of self-assembled monolayer on hydroxylated NiOx film surface for efficient and stable inverted perovskite solar cells. Adv. Funct. Mater. 35, 2413104 (2025).
Li, T. et al. In situ dehydration condensation of self-assembled molecules enables stabilization of CsPbI3 perovskites for efficient photovoltaics. Adv. Funct. Mater. 34, 2409621 (2024).
Chen, C.-H. et al. Methylthio substituent in SAM constructing regulatory bridge with photovoltaic perovskites. Angew. Chem. Int Ed. 64, e202419375 (2025).
Kralj, S. et al. Impact of the TCO microstructure on the electronic properties of carbazole-based self-assembled monolayers. ACS Mater. Lett. 6, 366–374 (2024).
Park, S. M. et al. Low-loss contacts on textured substrates for inverted perovskite solar cells. Nature 624, 289–294 (2023).
Zhan, L. et al. Anchorable polymers enabling ultra-thin and robust hole-transporting layers for high-efficiency inverted perovskite solar cells. Angew. Chem. Int Ed. 64, e202422571 (2025).
Tang, H. et al. Reinforcing self-assembly of hole transport molecules for stable inverted perovskite solar cells. Science 383, 1236–1240 (2024).
Yu, S. et al. Homogenized NiOx nanoparticles for improved hole transport in inverted perovskite solar cells. Science 382, 1399–1404 (2023).
Er-Raji, O. et al. Tuning self-assembly of hole-selective monolayers for reproducible perovskite/silicon tandem solar cells. Small Methods 9, 2401758 (2025).
Zhang, H., Zhao, C., Yao, J. & Choy, W. C. H. Dopant-free NiO nanocrystals: a low-cost and stable hole transport material for commercializing perovskite optoelectronics. Angew. Chem. Int Ed. 62, e202219307 (2023).
Jeon, Y.-S. & Park, N.-G. Nickel oxide thin films formed from the ph-adjusted solutions for inverted perovskite solar cells. ACS Energy Lett. 10, 1266–1274 (2025).
Wang, S. et al. Critical role of removing impurities in nickel oxide on high-efficiency and long-term stability of inverted perovskite solar cells. Angew. Chem. Int Ed. 61, e202116534 (2022).
Feng, Z. et al. Doped-NiOx seed layer on textured substrates for low-loss contacts in perovskite solar cells. Adv. Energy Mater. 15, 2405016 (2024).
Li, C. et al. Efficient inverted perovskite solar cells with a fill factor over 86% via surface modification of the nickel oxide hole contact. Adv. Funct. Mater. 33, 2214774 (2023).
Wang, H. et al. Modulating buried interface with multi-fluorine-containing organic molecule toward efficient NiOx-based inverted perovskite solar cell. Nano Energy 111, 108363 (2023).
Su, H. et al. Surface energy engineering of buried interface for highly stable perovskite solar cells with efficiency over 25%. Adv. Mater. 36, 2306724 (2024).
Xu, R. et al. Optimizing the buried interface in flexible perovskite solar cells to achieve over 24% efficiency and long-term stability. Adv. Mater. 36, 2308039 (2024).
Zhu, X. et al. Photoinduced cross-linkable polymerization of flexible perovskite solar cells and modules by incorporating benzyl acrylate. Adv. Funct. Mater. 32, 2202408 (2022).
Boyd, C. C. et al. Overcoming redox reactions at perovskite-nickel oxide interfaces to boost voltages in perovskite solar cells. Joule 4, 1759–1775 (2020).
Zhang, J. et al. Obstructing interfacial reaction between NiOx and perovskite to enable efficient and stable inverted perovskite solar cells. Chem. Eng. J. 426, 131357 (2021).
Qin, M., Chan, P. F. & Lu, X. A systematic review of metal halide perovskite crystallization and film formation mechanism unveiled by in situ GIWAXS. Adv. Mater. 33, 2105290 (2021).
Isikgor, F. H. et al. Self-assembled monolayer dyes for contact-passivated and stable perovskite solar cells. Adv. Energy Mater. 15, 2402630 (2025).
Khenkin, M. V. et al. Consensus statement for stability assessment and reporting for perovskite photovoltaics based on ISOS procedures. Nat. Energy 5, 35–49 (2020).
Xu, R. et al. Buried interface regulation for efficient and stable perovskite minimodules. Nano Energy 133, 110406 (2025).
International Electrotechnical Commission (IEC), Terrestrial Photovoltaic (PV) Modules design qualification and type approval-part 2: test procedures (IEC 61215-2:2021). https://webstore.iec.ch/en/publication/61350 (2021).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal–amorphous-semiconductor transition in germanium. Phys. Rev. B 49, 14251–14269 (1994).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B Condens Matter 54, 11169–11186 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B Condens Matter 50, 17953–17979 (1994).
Dudarev, S. L., Botton, G. A., Savrasov, S. Y., Humphreys, C. J. & Sutton, A. P. Electron-energy-loss spectra and the structural stability of nickel oxide: an LSDA+U study. Phys. Rev. B 57, 1505–1509 (1998).
Kakherskyi, S. et al. Structural, microstructural, chemical, and optical properties of NiO nanocrystals and films obtained by 3D printing. Appl Phys. A 127, 715 (2021).
Acknowledgments
This work is supported by the National Key Research and Development Program of China (2022YFB3803304), National Natural Science Foundation of China (62275213, 22473088, 62305261), Natural Science Basic Research Program of Shaanxi (Program No.2023-JC-QN-0693, 2025JC-JCQN-076), and the Key Research and Development Program of Shaanxi Province (2023-YBGY-301, 2023-YBGY-447). We acknowledge the computing resources provided by Xi’an Jiaotong University’s HPC platform and the Computing Center in Xi’an.
Author information
Authors and Affiliations
Contributions
H.D. designed the study and supervised the work. Y.S. conducted the most experimental. R.X. and H.T. designed and carried out the PL mapping procedure and data analysis. J.W. and W.C. helped prepare the devices and characterized the thin film. J.L. performed the DFT calculations. P.L. and B.J. assisted in the stability testing of module cells. Y.S. and R.X. prepared the first draft of the manuscript. J.X., J.D., F.Y., and Z.W. assisted in revising the final version of the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Meng Li, Luigi Castriotta, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sun, Y., Xu, R., Dai, J. et al. In situ coordinated HTL strategy for high-performance and scalable perovskite solar cells. Nat Commun 16, 9110 (2025). https://doi.org/10.1038/s41467-025-64111-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64111-9
This article is cited by
-
Self-Assembled Monolayers in Inverted Perovskite Solar Cells: A Rising Star with Challenges
Nano-Micro Letters (2026)
-
Solution-processed halide perovskite solar cells: from coating to modules
Journal of Materials Science: Materials in Energy (2026)