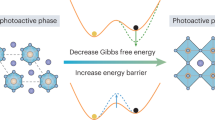
Abstract
The phase instability of perovskite materials remains a significant obstacle to their practical application in photovoltaics. Herein, we present a high-configurational-entropy strategy based on formamidinium ion (FA+) to fabricate the photoactive phase-stable halide perovskites through incorporating 2-amino-1,3,4-thiadiazole (2NTD). 2NTD optimally balances interactions with the [PbI6]4– octahedral frameworks while enhancing the rotational freedom of FA+. This synergistic effect amplifies FA+ anisotropy and elevates configurational entropy. Moreover, 2NTD effectively inhibits the formation of I2/I3– species and passivates the associated trap-state, thereby reducing the self-degradation behavior within perovskite films caused by undesirable iodine species. This improvement significantly enhances the crystallization and phase-stability of the perovskites under I2-rich conditions. Consequently, efficiencies of 26.63% (certified 26.40%) for a 0.09-cm2 inverted PSCs, 25.34% for a 1-cm2 device, and 23.08% for a 12.96-cm2 mini-module were obtained. Moreover, the target device exhibits a minimized non-radiative voltage loss of 69 mV and an improved long-term operational stability.
Similar content being viewed by others
Introduction
Perovskite solar cells (PSCs) have emerged as a transformative technology in photovoltaics (PV), achieving promising power conversion efficiencies (PCEs) exceeding 26% within just a decade of intensive research1. Their solution-based fabrication processes2, tunable bandgaps3, and exceptional optoelectronic properties make them a promising candidate for next-generation renewable energy solutions4. However, the intrinsic instability of perovskites, attributed to their soft ionic lattice nature, renders them vulnerable to phase transition from the photoactive α-phase to non-photoactive δ-phase under environmental stressors5. These uncontrolled phase transitions undermine device longevity and reliability, posing significant challenges for fabricating PSCs6. Addressing this phase instability issues is crucial for unlocking the full potential of PSCs in practical applications.
The crystal structural integrity of the photoactive phase fundamentally governs the operational stability of PSCs7. Extensive studies have been conducted to explore the degradation mechanisms and phase instability of perovskite films under environmental factors (oxygen, light, heat) and external electric fields8,9,10,11,12. Traditionally, oxygen-induced degradation predominantly occurs at the interface, where oxygen reacts with electrons to form superoxide ions (O2–), thus triggering MAPbI3 decomposition into CH3NH2, PbI2, and HI13. Subsequently, I2 and H2O formed by oxidation of HI14. Compared to oxygen, light and heat stress are considered as the key factors that influence the device’s operational stability due to that rational encapsulation can effectively prevent the erosion of oxygen. Light and/or thermal aging exacerbates degradation through ion migration, halide phase segregation, and PbI2 photolysis, which also releases I215. The accumulation of iodine species (I2/I3–) in the interface and bulk accelerates self-degradation, structural deterioration, micro-shorts and electrode delamination11. Moreover, I2 volatilization disrupts the perovskite lattice from its photoactive to non-photoactive phases6. The degradation mechanism of perovskites induced by iodine vapor involves a coupled process of oxidative corrosion and phase transition. Under photothermal, the cleavage of Pb-I bonds produces iodine radicals (I‧) and iodine vacancies (VI). These radicals recombine with free I⁻ ions to form I2, which then undergoes photodissociation into reactive I‧ species, triggering an autocatalytic degradation cycle16,17. I2 also destabilizes the [PbI6]4– octahedral framework via redox reactions, inducing lattice distortion and vacancy defects (Pb0, I– vacancies)18,19. This structural degradation deteriorates charge carrier dynamics by increasing non-radiative recombination and reducing ion migration activation energy. Therefore, suppressing the generation and accumulation of iodine species is critical for enhancing the operational reliability of PSCs.
It is well-established that formamidinium (FA+) based perovskites have consistently demonstrated record-breaking PCEs in PSCs, but remain challenged by stability concerns20. The orientation of FA+ within perovskite materials plays a critical role in the structural stability21,22. In the photoactive cubic phase, FA+ exhibit disordered anisotropy with high configurational entropy, whereas the non-photoactive hexagonal phase features strongly preferential FA+ orientations with reduced entropy22,23. Thermodynamics factors govern this phase stability, with entropy-driven disorder-to-order transitions contributing to the inherent instability of the cubic phase. Compositional engineering, such as incorporating mixed A-site cations, increases the configurational entropy of FA+-based perovskite, effectively suppressing transition to non-photoactive phases24,25. The high-entropy configuration of FA+ not only stabilizes the phase but also enhances defect tolerance and improves charge carrier dynamics19. Moreover, the inherent disorder in high-entropy FA+ configurations disrupts the formation of deep-level traps and reduces ion migration, which is beneficial for achieving superior optoelectronic performance and extended device lifetimes26. The high-entropy FA+ configuration establishes an averaged lattice, where local lattice strain is neutralized by randomized cation distribution, effectively suppressing δ-phase formation24. This prevented the degradation of photoactive perovskite phase under operational stresses. However, iodine-mediated phase transitions pose a significant threat to perovskite structural integrity. Therefore, it’s desirable to suppress the iodine-induced phase transition of perovskite for durable PSCs via enhancing the rotational freedom of FA+.
Here, we have fabricated photoactive phase-stable perovskites for efficient solar cells via strategically introducing thiadiazole isomers into the precursors. While 2-amino-1,3,4-thiadiazole (2NTD) is unlikely to substitute into the perovskite lattice, its presence at grain boundaries and interfaces can alter local environments that couple to FA+ dynamics, which induces a high-entropy configuration of FA+, significantly stabilizing the photoactive phase. 2NTD exhibits high adsorption energy toward defects, such as VI, PbI, and VPb, enabling optimal interactions with [PbI6]4– octahedra frameworks. The enhanced FA+ dynamic flexibility reduces charged traps and non-radiative voltage losses. Moreover, 2NTD mitigates self-degradation caused by accumulated I2/I3–, alleviating lattice distortions and improving structural stability under operational stress. The synergistic effects yielded p-i-n PSCs (0.09-cm2) with a high PCE of 26.63% (certified 26.40%), scalable to 25.34% for a 1-cm2 cell and 23.08% for a 12.96-cm2 mini-module. Additionally, the target devices retain 70% of their initial efficiencies after 1248 h of thermal aging at 85 °C and 86% of its original PCE after 1040 h of maximum power point (MPP) tracking under 1-sun illumination (white light LED array), demonstrating great thermo-optical stability. The proposed strategy raises the energy barrier against undesirable phase transformations and suppresses lattice distortions, which is critical for achieving stable perovskite materials and devices.
Results
Phase transition in I2-rich environment
To investigate the iodine-mediated degradation mechanisms of perovskite films, accelerated aging tests were conducted by exposing the films to I2 vapor environment6. Grazing incidence wide-angle x-ray scattering (GIWAXS) was employed to analyze the structural and crystal phase changes of perovskite after varying durations of I2 vapor exposure (t = 0 h, 1 h, and 3 h, as shown in Fig. 1a–d). The GIWAXS results revealed that, in the control perovskite films, the non-photoactive phase, specifically the hexagonal δ-phase 2H (qxy = 0.84 Å–1) and the hexagonal polytypes 4H (qxy = 0.83 Å–1)27, emerged after 3 h of I2 vapor exposure, indicating that the I2 vapor induced instability of photoactive phase and accelerated its transition. Furthermore, UV-vis absorption spectroscopy measurements were performed on aged perovskite films. The light absorption intensity of the control films decreased significantly with increasing the exposure duration, and only after 45 min, light absorption almost completely disappeared (Supplementary Fig. 1). Moreover, prolonged exposure to I2 vapor (over 4 h) in the control film resulted in an abnormal phenomenon—a redshifted absorption band edge, highlighting the instability of photoactive perovskite in I2-rich environment. This instability arises from the large size and high polarizability of I2, which interacts effectively with mobile I– in the perovskite lattice, forming I3– or other intermediate species6,28. These reactions trigger halogen-ion redistribution and defect generation within the perovskite structure29.
GIWAXS patterns of the control perovskite films aged under I2 vapor for a 0 h, b 1 h, and c 3 h. d The corresponding integrated intensity profiles of GIWAXS patterns. e The molecular structures, electrostatic potential (ESP) maps, and dipole moments of the three aminothiadiazole isomers. f Enlarged 1H-NMR spectra of FAI, 2NTD, and FAI blended with 2NTD (The gray shaded regions indicate the chemical shift of the amino hydrogen in 2NTD relative to the amino group hydrogen in FA+). g Adsorption energies of three aminothiadiazole isomers on VI, PbI, and VPb models of perovskite. Cross-sectional differential charge density maps of 2NTD adsorbed on h VI and i PbI models of perovskite.
To mitigate these undesirable phase transitions, we strategically selected three aminothiadiazole isomers as functional molecules. These isomers—2-amino-1,3,4-thiadiazole (2NTD), 5-amino-1,2,4-thiadiazole (5NT4D), and 5-amino-1,2,3-thiadiazole (5NT3D)—were chosen based on their structural similarities and potential to interact with the perovskite lattice. Density functional theory (DFT) calculations were conducted to determine the electrostatic potential (ESP) distributions and molecular dipole moments of these molecules. As shown in Fig. 1e, the thiadiazole N in all molecules shows negative ESP, while the –NH2 group within the molecules possesses the positive ESP. These characteristics make the aminothiadizole molecules promising functional modulators for the crystal growth of perovskite through molecular interactions. Moreover, the selected three isomers exhibits different dipole moments (3.52 D for 5NT4D, 3.56 D for 2NTD, and 5.27 D for 5NT3D), offering variable interaction energy with the precursors. It was reported that a larger dipole moment enhanced the intermolecular interactions30, leading to stronger interactions with FA+ and potentially promoting more ordered arrangements of FA+. Conversely, a lower dipole moment weakened the interactions, thereby mitigating the defect passivation effect in perovskites31. Therefore, we hypothesize that the 2NTD molecule with a moderate dipole moment possessed the ability to both passivate defects and modulate FA+ dynamics. This defect modulation, together with strain buffering, played a positive role in suppressing phase transition behavior under I2-rich conditions and improving device stability.
Fourier transform infrared spectroscopy (FT-IR) spectroscopy was conducted to investigate the interactions between FA+ and aminothiadiazole isomers (Supplementary Fig. 2). The results revealed that after modification with aminothiadiazole, the amino and FA+ skeletal vibrations shifted to higher wavenumbers, confirming the formation of hydrogen bond between FA+ and the functional molecules32. Nuclear magnetic resonance spectroscopy of hydrogen (1H-NMR) analyses further elucidated these interactions, as shown in Fig. 1f, the hydrogen atoms of the amino group in FA+ showed a downfield shift, while the amino hydrogen in 2NTD exhibited an upfield shift. Considering that there were almost no changes in the signals of protons for the 2NTD + CsI system (Supplementary Fig. 3), the above mentioned changes in chemical shift could be attributed to the interactions between FA+ and 2NTD through hydrogen bonding33. In contrast, for 5NT4D, the interaction primarily involved the hydrogen atoms on the thiadiazole ring, as evidenced by only a slight shift in the amino group’s hydrogen of FA+, indicating relatively weak interaction between 5NT4D and FA+. Meanwhile, the interaction between 5NT3D and FA+ split two hydrogen signals (around 8.75 ppm) of FA+ upon mixing, revealing that the strong interaction disrupts the symmetry of FA+ (Supplementary Fig. 4)34. This disruption suppressed intermolecular rotation, weakening the dynamic exchange of FA+ and reducing rapid conformational changes in solution. These findings align with the previously discussed influence of dipole moments on FA+ entropy change. 1H-NMR spectra of thiadiazole-PbI2 were also studied (Supplementary Fig. 5). Among the studied thiadiazole derivatives, 2NTD exhibited the most pronounced chemical shift in the proton, indicative of the coordination interaction with [PbI6]4⁻ octahedral. The strong interaction between 2NTD and [PbI6]4⁻ octahedral framework would enhance the rotational freedom of FA+ cations35.
DFT calculations were further conducted to explore the charge density distributions and adsorption energy of the three aminothiadiazole isomers on the defects, including iodine vacancies (VI), anti-site defect (Pb occupying I sites, PbI), and Pb vacancies (VPb). The adsorption energy of the aminothiadiazole isomers for these defects are summarized in Fig. 1g. Among them, 2NTD exhibited the strongest adsorption, followed by 5NT4D, while 5NT3D shows the weakest adsorption. Cross-sectional charge density distributions of the 2NTD-modified system revealed significant charge density variation at iodine vacancies (Fig. 1h). This charge redistribution predominantly arose from charge transfer involving the sulfur atom in the thiadiazole ring and the exocyclic amino group36. Similar trends were observed for 5NT4D and 5NT3D (Supplementary Fig. 6). For anti-site defects (Pb occupying I sites), the charge density variation was largely localized on the nitrogen atoms of the thiazole ring (Fig. 1i and Supplementary Fig. 7). For Pb vacancies, the three isomers exhibited distinct adsorption behaviors: both 2NTD and 5NT4D adsorb at the N atom adjacent to the carbon connected to the amino group, whereas 5NT3D interacted with a meta-positioned N atom (Supplementary Fig. 8). Despite sharing identical functional groups, the molecular configuration of the isomers led to significant differences in adsorption behavior. In the case of 2NTD, the close proximity of the amino group to the nitrogen atom on the thiadiazole ring promotes the formation of intramolecular hydrogen bonding, which increased molecular rigidity and reinforcing the perovskite skeleton37. In contrast, the amino groups in 5NT4D and 5NT3D are spatially farther from the thiadiazole nitrogen atoms, which increases molecular flexibility and results in lower adsorption energies38. Therefore, 2NTD, with its optimal dipole moment and enhanced molecular rigidity, established strong hydrogen bonds with FA+, effectively increasing the FA+ dynamics. This property would reinforce the perovskite structure and contribute to the improved phase stability.
Phase transition suppression in I2-rich environment
To assess the ability of aminothiadiazole isomers to suppress the phase transition in perovskite films under I2 vapor exposure, accelerated aging tests were conducted under I2-rich condition. GIWAXS results (Supplementary Figs. 9 and 10) reveal that 5NT4D and 5NT3D modified samples exhibit non-photoactive signals at 0.83 Å–1 and 0.84 Å–1, respectively, after 3 h of I2 vapor treatment. In contrast, 2NTD-modified perovskite films displayed high-resolution (100) diffraction rings without any δ-phase signals after 3 h of I2 vapor treatment. Although the (100) peak intensity of the α-phase for the aged film somewhat decreased, which should be attributed to a partial loss of crystallinity or increased lattice disorder under harsh conditions. This result indicated that 2NTD effectively mitigated phase transition of perovskite under I2-rich conditions (Fig. 2a–d). Similar results could be found from the X-ray diffraction (XRD) patterns (Supplementary Fig. 11). The control film exhibited serious phase transition from α to δ-phase after only 1 h of I2 vapor aging, as evidenced by the significant increased peak intensity of 11.7° (δ-phase)39. After 3 h of aging, most of the control perovskite was transformed to δ-phase. Conversely, aminothiadiazole-modified films, especially for 2NTD, showed suppressed δ-phase even after 8 h of I2 vapor aging, confirming the role of 2NTD in stabilizing photoactive phase perovskite, which were fully consistent with the surface-sensitive GIWAXS data. Additionally, UV-vis absorption spectroscopy was performed on the aged perovskite films (Supplementary Fig. 12). The incorporation of 2NTD significantly improved film stability by preventing I2-induced degradation, thereby stabilizing their photoactive phases. This observation aligns well with the GIWAXS and XRD findings, reinforcing the conclusion that 2NTD effectively suppresses I2-mediated phase transition and degradation.
GIWAXS patterns of 2NTD-modified perovskite films aged under iodine vapor for a 0 h, b 1 h, and c 3 h. d The corresponding integrated intensity profiles of GIWAXS patterns. e–g Polarized Raman spectra of the control and 2NTD-modified perovskite single crystals at 0°, 30°, 60°, and 90° polarization angles under 300 K. h Simulated vibrational entropy of the FA+ in the control and 2NTD-modified FAPbI3 (inset: the models of the control and 2NTD-treated α-phase FAPbI3). sin2ψ-2θ fitting curves for the control and aminothiadiazole isomers-modified perovskite films i before and j after iodine vapor treatment.
Temperature-dependent polarized Raman spectra (Supplementary Fig. 13) were acquired at 250 K, 300 K, 350 K and 400 K with different polarization angles (0°, 30°, 60° and 90°) to investigate FA+ cation orientational configurations and vibrational entropy (Fig. 2e–g and Supplementary Fig. 14). Vibrational signals below 150 cm–1 were assigned to [PbI6]4– octahedral vibrations (Fig. 2e), while higher-frequency modes corresponded to FA+ motions, including torsional (τ), bending (δ), NH2 rocking (ρ), NH2 stretching (ν) and CH wagging (ω) (Fig. 2f, g)21,40. At low temperature (250 K), both samples exhibited negligible vibrational signals of FA+, which gradually intensified with increasing temperature. Notably, the control FAPbI3 exhibited pronounced polarization angle dependence, with almost undetectable FA+ signals at polarization angles of 0° and 30°, indicating orientation-sensitive [PbI6]4– octahedral and FA+ vibrational coupling requiring specific alignment with the incident light polarization. In contrast, the angular dependence was greatly suppressed for all FA+ vibrational signals in the 2NTD-modified sample, indicating the absence of orientation-preference and greater dynamic disorder, i.e., higher orientational entropy for FA+27. The vibrational entropy (Svib) of FA+ was then extracted from these Raman spectra via employing the Boltzmann entropy expression within the harmonic approximation for both samples (Supplementary Note 1 and Supplementary Table 1)41. At 250K, where thermal energy is insufficient to activate vibrational modes, 2NTD modification induced only marginal Svib enhancement in FAPbI3. When the temperature was higher than 300 K, an increase in Svib could be obtained for the 2NTD modified samples at different temperatures, suggesting the increased vibrational freedom of FA+ in the target sample22,42. We also calculated the Svib for both samples at different temperatures using DFT calculations. 2NTD-modified FAPbI3 exhibited a higher Svib than that of the control one at all temperatures (Fig. 2h), confirming the reduction of vibrational barrier for FA+. Additionally, 15 out of 24 kinds of vibrational modes for FA+ exhibited lower vibrational frequencies upon 2NTD modification, further indicating a softening of the local environment and enhanced molecular freedom of FA+ (Supplementary Fig. 15 and Supplementary Video 1, Supplementary Video 2). These results collectively indicated that the vibrational modes of FA+ in 2NTD-modified crystals exhibited greater randomness and lacked a preferred orientation when compared to the control sample.
Molecular dynamics simulations were performed to investigate FA+ dynamics under different conditions: (i) 25 °C, (ii) 25 °C with iodide vacancies (VI), (iii) 85 °C, and (iv) 85 °C with VI. Conformational snapshots at 0, 1, 10, 50, and 100 ps were extracted for analysis. The angular displacement between the FA+ central carbon atom and the X-axis-aligned basal plane was quantitatively measured. 2NTD modification significantly enhanced the angular displacement of FA+ at both 25 °C and 85 °C compared to the control sample (Supplementary Figs. 16–19 and Supplementary Tables 2–9). This trend persisted under VI conditions at these temperatures. Crucially, VI-containing systems with 2NTD exhibited greater rotational freedom, which is beneficial for suppressing phase transitions. Additionally, the calculated Gibbs free energy difference for α-to-δ phase transition (ΔGα→δ) increased from 2.00 eV (control) to 4.05 eV (2NTD) at 300 K (Supplementary Fig. 20), suggesting that the introduction of 2NTD substantially increased the energy barrier for α-to-δ phase transition, thereby stabilizing the α-phase perovskite. These results highlight the critical role of 2NTD in stabilizing perovskite structures via interacting with Pb-I octahedral and FA+.
Time-of-flight secondary ion mass spectrometry (ToF-SIMS) was used to examine the distribution of 2NTD within the perovskite film. 2NTD molecules distributed primarily on the surface of perovskite film, with a gradually reduced concentration from the surface to the bulk (Supplementary Fig. 21). X-ray photoelectron spectroscopy (XPS) depth profiling on perovskite films was also collected to confirm the distribution of 2NTD molecules. S 2p signal was used as the indicator for 2NTD molecules. As shown in Supplementary Fig. 22, the 2NTD-treated film exhibited clearly S 2p signal even under the depth of 150 nm, whereas no S 2p signal was detected in the control sample. These findings demonstrated that 2NTD not only existed on the surface but also penetrated the shallow layer (about 150 nm) of perovskite film, in good agreement with the ToF-SIMS results, supporting the surface/interfacial interactions and passivations. As the 2NTD was added into the perovskite precursor, it engaged into the whole nucleation and crystal process of perovskite, offering positive effect on the crystallization. After annealing, 2NTD molecules were extruded to the surface and shallow layer of perovskite, which continued to influence the phase stability via coordination and hydrogen bonding with the Pb-I octahedra and FA+. Grazing-incidence X-ray diffraction (GIXRD) was performed to analyze the influence of aminothiadiazole isomers on the (012) diffraction plane of the perovskite lattice (Fig. 2i, j, Supplementary Figs. 23, 24, Supplementary Table 10, and Supplementary Note 2). Before I2 vapor treatment, the introduction of aminothiadiazole isomers reduced lattice strain in perovskite crystals, with measured stress values of 4.94 MPa (Control), 2.67 MPa (5NT4D), 2.09 MPa (2NTD), and 2.90 MPa (5NT3D), respectively. This alleviated lattice strain was attributed to the formation of a stress-buffering layer within the upper-middle region of the perovskite film, as supported by ToF-SIMS and XPS depth profiling, which confirmed that 2NTD molecules were predominantly located in the surface and shallow layer (about 150 nm) of the perovskite. Notably, after I2 vapor treatment, the lattice strain in control films increased significantly to 11.11 MPa, while films treated with aminothiadiazole isomers almost retained their original lattice size, especially in the case of the 2NTD-based sample. These findings suggest that aminothiadizole not only effectively released the lattice strain, but also provided a protective effect against I2 vapor erosion43. Mechanistically, these functional molecules interacted with the surface [PbI6]4– octahedra, causing subtle changes in bond lengths and angles at the surface and adjacent layers44,45. This interaction generated a strain gradient that propagates into the bulk, modifying the overall lattice structure and stress distribution46. Consequently, the [PbI6]4– octahedra in the bulk become slightly distorted, leading to alterations in the size and shape of the A-site cavities, which further adjusted the rotational energy barrier for FA+ 35. Therefore, 2NTD-modification reduced lattice strain and induced changes in FA+ vibrational characteristics, suggesting a surface–interface–bulk coupling that contributed to the improved phase stability.
XPS further revealed coordination interactions between aminothiadiazole isomers and the [PbI6]4– octahedral framework. As shown in Supplementary Fig. 25, the Pb 4 f core levels shifted to high binding energy upon isomer treatment, especially for the 2NTD-based sample. A similar phenomenon can be observed for I 3 d orbitals. These binding energy shifts were attributed to the strengthened Pb–I bonds within the [PbI6]4– framework47. Additionally, the passivation of under-coordinated Pb2+ by N atoms in the thiadiazole isomers promotes Pb–N charge delocalization, leading to a high-binding-energy shift, consistent with DFT calculations. These results indicate that 2NTD treatment optimized the interactions with [PbI6]4– octahedral framework and increased the dynamic freedom of FA+48. UV-vis absorption spectra and bandgap calculations of aminothiadiazole isomers-modified perovskite films reveal that lattice variations do not alter the bandgap (1.53 eV) of perovskite (Supplementary Fig. 26), which was related to both the alleviation of lattice strain and the enhanced freedom of FA+. Reduced residual stress led to more uniform lattice parameters, thus stabilizing the electronic band structure and preventing bandgap fluctuations. Meanwhile, sufficient spatial and rotational freedom of FA+ enabled dynamic disorder, which acted to buffer local stress and maintain average crystal symmetry, further preserving the bandgap stability. Ultraviolet photoelectron spectroscopy (UPS) further demonstrates enhanced n-type characteristics in modified perovskites, facilitating electron transport (Supplementary Figs. 27, 28). In short, the incorporation of 2NTD significantly reduces lattice strain of perovskite even in I2-rich environment. Moreover, 2NTD provided sufficient spatial freedom for FA+, contributing to enhanced lattice uniformity and defect passivation, maintaining high lattice symmetry and buffered structural stress, all of which were beneficial for improving the optoelectronic properties (such as photoluminescence (PL) lifetime, carrier mobility, trap state density, carrier recombination dynamics, and stability) of the perovskites.
Iodide oxidation suppression and crystal growth regulation
The detrimental effects of triiodide (I3–) on perovskite materials, such as ion migration-induced defect formation and structural degradation, highlight the necessity of fundamentally suppressing I3– generation. To study how 2NTD influences the precursor solution stability, UV-vis absorption spectroscopy was employed. As shown in Fig. 3a, b, distinct I3– absorption peaks at ~360 nm appeared in pristine FAI solution within 3 h (aged at 50 oC, in air), with intensity increasing over time, which was attributed to I– oxidation49. In contrast, FAI solutions containing 2NTD exhibited no detectable I3– absorption peaks even after 48 h accelerated aging, indicating superior oxidation resistance and effective suppression of I3– formation. Due to the spectral overlap between 5NT4D/5NT3D and I3–, the oxidation process of I– cannot be clearly decoupled (Supplementary Fig. 29). However, the continuous increase in the absorption intensity at 360 nm suggests that 5NT4D and 5NT3D exhibit limited efficacy in suppressing iodide oxidation. Notably, the addition of 2NTD to the aged FAI solution demonstrated strong reducing capability, effectively converting I3– back to I– within 10 s (Fig. 3c). This observation suggests that 2NTD possessed detrimental iodine-species scavenging capability, which could play a crucial role in enhancing the phase stability of perovskite materials50. Moreover, fresh 2NTD and oxidized 2NTD (aged FAI mixed with 2NTD) solutions were characterized by 1H-NMR and High-Resolution Mass Spectroscopy (HRMS). The detrimental I3– was reduced to I⁻, while the sulfur in 2NTD was oxidized to sulfoxide (Supplementary Fig. 30). To directly illustrate the passivation of I-related defects, the depth-resolved I⁻ and I3⁻ signal profiles were collected by measuring ToF-SIMS. As shown in Supplementary Fig. 31, the control film exhibited a clearly I3⁻ signal within the surface and bulk region, whereas the 2NTD-treated sample maintained negligible I3⁻ intensity throughout the probed depth, indicating effective suppression of polyiodide species. These results demonstrated that 2NTD effectively reduced I-related trap states and self-degradation reactions, thereby enhancing the film stability.
UV-vis absorption spectra of FAI/DMF solutions a without and b with 2NTD addition aged for different durations. c UV-vis absorption spectra and photographs of the aged FAI/DMF solution before and after 2NTD addition (inset: photographs to demonstrate the color change of the aged FAI solution before and after 2NTD addition). Time-dependent in situ-GIWAXS patterns of d the control and e 2NTD-modified perovskite films during spin-coating and annealing processes. Time-resolved integrated peak area intensity for f δ-phase and g α-phase of the control and 2NTD-modified samples. Surface SEM images of h the control and i 2NTD-modified perovskite films.
To gain insight into the effect of 2NTD on the crystallization process of perovskite, in situ GIWAXS measurements were conducted throughout the entire spin-coating and annealing process (Fig. 3d, e). Stage I corresponds to the period before antisolvent dripping, while Stage II represents the phase transformation from the δ-to-α phase. In both control and 2NTD-treated perovskite films, the hexagonal 2H phase (0.84 Å–1) was the only crystalline phase observed during stage II. For the control perovskite, stage II lasted approximately 29 s (from 28 s to 57 s). However, with the introduction of 2NTD into the perovskite, stage II extended to 33 s (from 28 s to 61 s), enabling complete phase conversion. The prolonged phase transition time is likely attributed to the reduced ion diffusion rate induced by 2NTD, which promotes the formation of larger and higher-quality grains. As shown in Fig. 3f, g, intensity-time profiles at q = 0.84 and 1.00 Å–1 for both perovskite films were extracted. The results demonstrate that 2NTD delays the emergence and retards the transition of δ-phase, facilitating stabilized and orderly arrangement of intermediate phases. Additionally, the α-phase extracted at q = 1.00 Å–1 exhibited higher and more uniform intensity, indicating the formation of high-crystallinity perovskite films with reduced defects. XRD was also performed on aminothiadiazole isomers-modified perovskite films (Supplementary Fig. 32). The full width at half maximum (FWHM) of the (100) diffraction plane was calculated to be 0.178° (control), 0.143° (5NT4D), 0.132° (2NTD), and 0.176° (5NT3D), verifying the enhanced crystallinity upon 2NTD treatment.
Scanning electron microscopy (SEM) was used to study the morphologies of perovskite films. Compared to the control films (Fig. 3h), the incorporation of 2NTD resulted in a more uniform grain size distribution (Fig. 3i). Statistical analysis of grain sizes (Supplementary Fig. 33) revealed that the 2NTD-modified sample exhibited the largest average grain size of 440 nm, compared to 390 nm, 350 nm, and 340 nm for 5NT4D, 5NT3D, and the control sample, respectively. Cross-sectional SEM images revealed that the 2NTD-modified PSCs exhibited reduced grain boundaries along the vertical direction compared to the control one (Supplementary Fig. 34). Moreover, the isomers treated samples demonstrated a notable decrease in the root-mean-square roughness as evidenced by atomic force microscopy (AFM) images (Supplementary Fig. 35). The reduction in surface roughness of the perovskite films contributes to a decrease in interfacial carrier recombination and energy loss, ultimately improving charge collection efficiency51. Trap-state densities in perovskite films were quantitatively analyzed using space charge limited current (SCLC) measurements (Supplementary Figs. 36, 37 and Supplementary Note 3). The hole-trap state densities were calculated to be 1.62 × 1015 cm–3, 1.18 × 1015 cm–3, 1.31 × 1015 cm–3, and 1.42 × 1015 cm–3 for the control, 2NTD, 5NT4D, and 5NT3D-based films, respectively. Similarly, the electron-trap state density for the control one was determined to be 1.93 × 1016 cm–3, which decreased to 1.64 × 1016 cm–3, 1.78 × 1016 cm–3, and 1.85 × 1016 cm–3 for 2NTD, 5NT4D, and 5NT3D-based films, respectively. These results confirm that isomer treatment, particularly with 2NTD, improves film quality and reduces trap-state density, which is beneficial for achieving high-quality perovskite films and high-performance devices.
Photovoltaic performance and stability assessment
To evaluate the impact of 2NTD on the PV performance of PSCs, inverted device with architecture of FTO/NiOx/MeO−2PACz/perovskite/C60/BCP/Ag was fabricated (Fig. 4a). After systematical optimization, the control device with aperture area of 0.09 cm2 achieved a PCE of 25.14%, with a VOC of 1.155 V, JSC of 26.01 mA cm–2, and FF of 83.70% (Fig. 4b). Introducing 5NT3D improved the PCE to 25.40%, while 5NT4D-treatment delivered a higher PCE of 26.10% (Supplementary Fig. 38). Notably, an impressive PCE of 26.63%, along with a VOC of 1.180 V, JSC of 26.35 mA cm–2, and FF of 85.66% was obtained for the champion device treated with 2NTD (Fig. 4c). The significantly enhanced device performance relied primarily on the improved VOC and FF, which should be originated from the optimized perovskite crystallinity, reduced grain boundary, passivated defects, and improved carrier lifetime. External quantum efficiency (EQE) spectra verified the accuracy of our sunlight simulator (Fig. 4d and Supplementary Fig. 39). MPP tracking further supported the improved device performance. The 2NTD-treated device delivered a high steady-state output efficiency of 26.19% with no degradation within 500 s (Fig. 4e), significantly outperforming that of the control device (Supplementary Fig. 40). The enhancement in the operational stability was attributed to the moderate coordination between 2NTD and the perovskite octahedral structure, which facilitated the formation of FA+ in a high-entropy state coupled with lattice strain relief, defect passivation, and suppression of I2/I3–. One of the best devices was sent to an independently third party for certification (National Photovoltaic Product Quality Inspection and Testing Center, China), a PCE of 26.40% with small hysteresis effect was achieved (Supplementary Fig. 41), validating our results. Statistics for 60 individual PSCs further indicate that the aminothiadiazole modified devices exhibit better reproducibility with smaller standard deviation compared to the control (Fig. 4f, Supplementary Fig. 42, and Supplementary Tables 11–14).
a Schematic diagram of the fabricated device structure. J–V curves for b the control and c 2NTD-modified PSCs (aperture area: 0.09-cm2) from the reverse and forward scans. d EQE curves and integral current curves of the control and 2NTD-modified PSCs. e Steady-state power output of the 2NTD-modified PSCs under 1-sun illumination. f Statistics of PCEs for 60 individual devices. g J–V curves for the 2NTD-modified PSCs with aperture area of 1-cm2 from the reverse and forward scans. h Photograph of the fabricated mini-module. i J–V curves for the 2NTD-modified mini-module with active areas of 12.96-cm2 from the reverse and forward scans.
To study the potential of the strategy in upscaling fabrication of PSCs, large area device with aperture area of 1-cm2 was assembled (Fig. 4g). The 2NTD modulated devices achieved efficiencies of 25.34%, which is higher than that of the corresponding control device (Supplementary Fig. 43), and consistent with the trend observed in small area devices. This scalability with maintained efficiency emphasizes the robustness of the 2NTD-modified strategy. Given the prominent efficiency for large area devices, we further fabricate a minimodule on a 5 × 5 cm2 substrate with an active area of 12.96 cm2 (consisted of six sub-cells connected in series) (Fig. 4h). The control minimodule exhibited a PCE of 20.13% (VOC = 6.80 V, JSC = 4.19 mA cm–2, FF = 70.64%) from the reverse scan and a PCE of 19.31% from the forward scan (Supplementary Fig. 44). When 2NTD was introduced into the device, the VOC was significantly improved to 7.00 V and the FF was enhanced to 77.94%, resulting in a PCE of 23.08% with negligible hysteresis effects (Fig. 4i). This improvement highlights the scalability and robustness of the proposed strategy.
To investigate carrier recombination mechanisms in devices, light-intensity-dependent JSC and VOC analyses were conducted (Fig. 5a and Supplementary Fig. 45). All devices exhibited an exponential factor of α = 0.94, derived from JSC-light intensity dependence plots, suggesting the negligible bimolecular recombination43. The ideality factor (n) was further extracted from VOC vs light intensity plots. Compared to the control one (n = 1.82), aminothiadiazole-treatment reduced the value of n, with 2NTD-based device reaching a minimal n = 1.15. This result indicates that 2NTD treatment effectively suppressed the trap-assisted recombination52. Non-radiative voltage loss (\({V}_{{loss}}^{{non}-{rad}}\)), predominantly governed by trap states, emerges as the critical bottleneck for VOC in perovskite photovoltaics. Quantitative EQEEL analysis reveals that aminothiadiazole isomers achieves \({V}_{{loss}}^{{non}-{rad}}\) suppression from 99 mV for the control to 69 mV for the 2NTD-modified devices (Fig. 5b and Supplementary Note 4). This reduction correlates directly with defect passivation efficacy and enhanced VOC. The performance ranking based on molecular structure (2NTD < 5NT4D < 5NT3D) highlights the role of steric configuration in mitigating Shockley-Read-Hall recombination53. To characterize the density of localized states near the absorption edge, Urbach energy (EU) was systematically investigated (Fig. 5c and Supplementary Fig. 46). Five individual devices were measured for each condition, and the average values were calculated: 2NTDavg (14.91 meV) <5NT4Davg (16.48 meV) <5NT3Davg (18.50 meV) <Controlavg (21.01 meV). These results indicate that control films exhibited broad Urbach tails, reflecting substantial carrier trapping-recombination losses. In contrast, 2NTD modification effectively reduced defect density at the absorption edge, improving the utilization efficiency of photogenerated carriers54. This reduction in localized states enhances the optoelectronic properties and overall device performance. A similar conclusion could be reached from the time-resolved PL spectroscopy (Supplementary Note 5, Supplementary Fig. 47 and Supplementary Table 15). The average PL lifetimes were calculated to be 1786 ns (control), 2313 ns (5NT4D), 2320 ns (2NTD), and 2233 ns (5NT3D), indicative of the enhanced carrier lifetimes in the modified samples when compared to the control one. This result suggested effective suppression of non-radiative recombination in perovskite films upon aminothiadiazole treatment, which minimized the non-radiative voltage loss.
a VOC vs. light intensity for the control and aminothiadiazole isomers-treated devices. b EQEEL as a function of injection current density for the control and aminothiadiazole isomers-treated devices (inset: photograph displays the electroluminescence image of a 2NTD-modified device). c Urbach energy (EU) of the control and aminothiadiazole isomers treated perovskite films (The error bars represent the standard deviation from five devices). d Charge collection efficiency (ηcc) profiles of the control and aminothiadiazole isomers-modified PSCs. e TPV and f TPC decay curves of the control and aminothiadiazole isomers-treated devices. g Stability of the unencapsulated control and aminothiadiazole isomers-modified devices under 85 oC heating in a N2-filled glovebox (The error bars represent the standard deviation from three devices). h Stability of the unencapsulated PSCs with MPP tracking at room temperature under one sun equivalent white LED illumination.
The charge collection efficiency (ηcc) of the devices was then quantitatively evaluated through intensity-modulated photocurrent spectroscopy (IMPS) and intensity-modulated photovoltage spectroscopy (IMVS) measurements (Fig. 5d, Supplementary Fig. 48, Supplementary Table 16, and Supplementary Note 6). The aminothiadiazole isomers treated devices show higher carrier recombination times under different light intensities compared to the control one, indicating reduced recombination. Notably, the 2NTD modified device demonstrates a highest ηcc of 93.59% under 10% light intensity, outperforming all other devices. The higher ηcc values correlate well with the improved JSC and FF in 2NTD-treated devices55. Transient photovoltage (TPV) and transient photocurrent (TPC) measurements further verified these findings (Fig. 5e,f). The 2NTD-treated device exhibited a faster charge extraction time (τtr = 0.82 μs) and an extended carrier recombination lifetime (τrec = 32.45 μs) compared to that of the control devices (τtr = 1.19 μs, τrec = 3.00 μs). The significantly reduced τtr and extended τrec collectively confirm that 2NTD treatment optimized the charge transport and improved device performance56. Electrochemical impedance spectroscopy (EIS) measurements confirm the increased interfacial recombination resistance (Rrec) in the aminothiadiazole isomers treated device compared to the control one (Supplementary Fig. 49 and Supplementary Table 17)57. The built-in potential (Vbi) fitted based on the Mott-Schottky plots was then extracted to b 0.895 and 0.819 V for the 2NTD treated and control devices (Supplementary Fig. 50), respectively. The enhanced Vbi is favorable to charge separation and extraction, as confirmed by the TPC decay. These findings collectively demonstrate that 2NTD treatment significantly enhances charge transport, suppresses recombination, and improves charge separation, ultimately contributing to the superior performance of the 2NTD-modified PSCs.
In addition to improved PV performance, the 2NTD-induced high entropy perovskite film demonstrates significantly enhanced stability in PSCs. Shelf-life measurements were first conducted by storing the unencapsulated devices in the dark at 25 °C in an N2-filled glovebox (Supplementary Fig. 51 and Supplementary Table 18). The control device retained 70% of its initial efficiency after 2016 h of storage, whereas the 2NTD-modified device exhibited minimal efficiency loss over the same period (93.2% remained). Besides, the operational stability of the device under stresses of thermal and/or light was more critical for their practical application, because these stresses often resulted in the formation and accumulation of detrimental iodine species (I2/I3⁻), which accelerated perovskite decomposition and phase instability. We therefore systematically investigated the thermal stability of different devices under 85 °C hotplate in a N2-filled atmosphere (Fig. 5g and Supplementary Tables 19−22). The 2NTD-treated device retains 70.00% of its initial PCE after 1248 h of thermal aging, outperforming all control groups. This enhanced thermal stability indicates that 2NTD modification effectively suppresses iodide oxidation and self-degradation of perovskite, providing functional resilience under elevated temperatures, which was also partly related to the intrinsic thermal stability of the thiadiazole isomers with high thermal decomposition temperatures (Supplementary Fig. 52). Additionally, 2NTD modification suppressed δ-phase formation and reduced grain boundaries in thermally aged perovskite films, as evidenced by SEM and XRD results (Supplementary Fig. 53), confirming the inhibition of thermally activated decomposition and phase transition. Operational stability under continuous white-light LED illumination (1 sun) with maximum power point tracking (MPPT) further highlights the benefits of 2NTD treatment (Fig. 5h and Supplementary Table 23). After 1040 h of MPPT, the 2NTD-modified device retained 86.0% of its PCE, far surpassing the control device, which maintained only 56.8% of its initial PCE after 870 h. The 2NTD-modified perovskite film exhibited fewer pinholes and suppressed δ-phase formation against light soaking (Supplementary Fig. 54). The aging tests under ambient conditions (55% relative humidity) were also conducted (Supplementary Fig. 55 and Supplementary Table 24). The target devices retained over 70% of their initial PCE after 96 h of continuous testing, while the control one maintained 68% of the original PCE. In short, 2NTD treatment was correlated with surface/near-surface stabilization and altered FA+ dynamics, thus contributing to the improved phase stability and device performance.
Discussion
In summary, we thoroughly investigated the effect of aminothiadiazole isomers, particularly 2NTD, on FA+ configurational entropy and perovskite phase stability using a combination of theoretical and experimental characterization. 2NTD showed optimal interactions with [PbI6]4– octahedra, effectively regulating the rotational freedom and anisotropy of FA+. This resulted in a disordered, high-entropy alignment for FA+, which in turn improved photoactive phase stability. Additionally, 2NTD exhibited distinct capabilities in suppressing and passivating detrimental I2/I3– species, mitigating perovskite self-degradation, and enhancing operational stability. These effects collectively enabled the fabrication of high-quality perovskites. Device (aperture area: 0.09 cm2) with 2NTD treatment achieved a high efficiency of 26.63% (certified 26.40%) along with a minimal non-radiative voltage loss of 69 mV and improved operational stability. Importantly, this approach maintained great scalability, delivering efficiencies of 25.34% for a 1-cm2 device and 23.08% for 12.96-cm2 mini-module. These advancements underscore the transformative role of the 2NTD-induced FA+ high-entropy state, which enhances phase stability, passivates defects, suppresses iodide oxidation, and ensures long-term durability of halide perovskites. This work paves the way for addressing critical issues related to efficiency and reliability in inverted PSCs.
Methods
Materials
All the chemicals were used directly without further purification. Nickel oxide nanoparticle (99.999%), lead iodide ( ≥ 99.999%), formamidinium iodide (FAI, 99.9%), and C60 (99.5%) were purchased from Advanced Election Technology Co. Ltd. [2-(3,6-Dimethoxy-9H-carbazol-9-yl)ethyl]phosphonic acid (MeO−2PACz, >99%) was purchased from TCI. CsCl (99.999%) was purchased from Alfa. 5-amino-1,2,4-thiadiazole (5NT4D), 2-amino-1,3,4-thiadiazole (2NTD), and 5-amino-1,2,3-thiadiazole (5NT3D) were purchased from J&K Scientific Ltd. Methylammonium iodide (MAI, 99.9%) and 2,9-dimethyl-4,7-diphenyl-1,10-phenanthroline (BCP, 99%) was purchased from Xi’an Yuri Solar Co. Ltd. Dimethylformamide (DMF, 99.8%), dimethyl sulfoxide (DMSO, 99.9%), isopropyl alcohol (IPA, 99.5%) and Ethyl alcohol (EtOH, 99.5%) were purchased from Sigma-Aldrich. 1,3-Diaminopropane Dihydroiodide (PDAI2, ≥99.5%) was purchased from Xi’an e-Light New Material Co, Ltd. Hydrogen peroxide (H2O2, ≥30%) was purchased from RESO.
Device fabrication
For the small-sized solar cell, a patterned FTO glass substrate was cleaned by sequentially washing with detergent, deionized water, acetone, and isopropanol (IPA). Before use, the FTO was treated with ultraviolet ozone for 15 min. Then, the substrate was spin-coated with a thin layer of NiOx nanoparticle (10 mg/mL aqueous solution containing 40 mol.% H2O2) at 2000 rpm for 40 s, and annealed in ambient air at 110 °C for 20 min, then cooled down naturally and transferred to the glove box. 0.5 mg/mL MeO−2PACz was deposited on the NiOx film at 3000 rpm for 25 s and annealed at 110 °C for 10 min.
The RbCsFAMA-based perovskite solution was prepared by mixing 691.52 mg of PbI2, 232.16 mg of FAI, 11.92 mg of MAI, 12.63 mg of CsCl, and 9.56 mg of RbI in 1 mL of a mixed solvent (DMF:DMSO = 4:1, v/v), which was stirred for 12 h before use. Aminothiadiazole isomers (5NT4D, 2NTD, 5NT3D) were dissolved in perovskite precursor solutions at 1 mg/mL. The perovskite solution was filtered with 0.22 μm PTFE filter. After that, the perovskite ink was deposited on the FTO/NiOx/MeO−2PACz/passivator substrate by spin coating at 1000 rpm for 8 s with a ramp of 200, and 5000 rpm for 25 s (2000 rpm ramp). 20 seconds into the second step, 120 μL of anisole was deposited onto the substrate. The wet film was then annealed at 110 °C for 20 min. After that, 1 mg/mL PDAI2 was coated on the perovskite surface at 4500 rpm for 25 s and annealed at 110 °C for 5 min. Finally, 20 nm C60, 6 nm BCP and 120 nm Ag were deposited by thermal evaporation.
Minimodule fabrication: a vacuum-assisted deposition technology was adopted. The perovskite ink was first spin-coated on the substrate by a two-step process (1000 rpm for 8 s and 5000 rpm for 10 s) to obtain a wet film. The wet film was then placed into the sample chamber connected to the vacuum pump. After starting the vacuum pump, with the decrease of pressure, the solvent components in the wet film were quickly volatilized and removed. After being held for 10 s at a pressure of 10 Pa, the perovskite film was removed and annealed at 110 °C for 20 min. After that, 1 mg/mL PDAI2 was coated on the perovskite surface at 4500 rpm for 25 s, and then annealed at 110 °C for 5 min. Finally, 20 nm C60, 6 nm BCP and 120 nm Ag were deposited by thermal evaporation. The minimodule consisted of six perovskite sub-cells in series fabricating on a 5 × 5 cm2 FTO glass substrate. Modules were interconnected through P1, P2, and P3 lines. The lines were drawn on the substrate by a laser etching system with a wavelength of 1064 nm, a frequency of 20 KHz, and a scan speed of 2000. The P1 line with a width of 150 μm was obtained by 30 etchings at a power of 16 W, the P2 line with a width of 500 μm was obtained by a single etching at a power of 6 W, and the P3 line with a width of 100 μm was obtained by a single etching at a power of 10 W. Finally, a minimodule with a width of 1100 μm for a single dead area, and with 12.96 cm2 active area was fabricated.
Synthesis of FAPbI3 single crystals: To grow FAPbI3 single crystals, FAI and PbI2 were dissolved in GBL with a ratio of 1:1 (molar) at room temperature. After vigorous stirring for 24 h, clear FAPbI3 precursor solution was obtained, and the concentration of the solution was controlled at 0.8 M. Before using the solution, 1.25 mg of 2-amino-1,3,4-thiadiazole (2NTD) was added into the solution with stirring for 10 min to disperse it completely. Then, the prepared solution was further filtered through 0.8 μm–pore size polytetrafluoroethylene (PTFE) filters. To grow large FAPbI3 single crystals, small crystals (1 to 2 mm) were grown by slow heating process from 80 °C to 120 °C, and maintained at 120 °C for 12 h. Finally, the small crystals with a regular shape were picked out as seeds. Second, to obtain large crystals, three to five seeds were transferred to fresh solution, and its temperature was raised to 120 °C and then maintained for 2 days. Then, each pristine seed was grown to a larger one ( ~ 4-5 mm).
Characterization
The UV-Vis absorption spectroscopy was conducted by the PerkinElmer Lambda 950 ultraviolet-visible spectrophotometer. XRD was measured by Bruker D8 advance using Cu Kα radiation (40 kV, 40 mA). The selected scan rate is 5°/min and the step size is 0.02 s. AFM was measured by Bruker Innova Atomic force microscopy. XPS and UPS were measured by the Thermo-fisher ESCALAB Xi+ system. The radiation at the time of measurement was produced by a monochromatic excitation of a 75 W Al Kα source centered on 1486.7 eV for XPS measurement. As for UPS, He I ultraviolet radiation source with an energy of 21.22 eV was used. FTIR was measured by Thermo-Fisher Nicolet Is5. SEM images were obtained by Hitachi S4800 field emission scanning electron microscope (Hitachi High Technologies Corporation). (In situ) Grazing-incidence wide-angle x-ray scattering (GIWAXS) was obtained at the BL14B1 beamline of Shanghai Synchrotron Radiation Facility (SSRF). The grazing incidence angle was 0.4o. The GIWAXS data was collected every 2 seconds synchronously from 0 s to 400 s. The liquid state 1H NMR measurements were recorded on JNM-ECZ400S/L1 spectrometer (TMS as an internal standard (δ = 0)). TRPL spectra were measured by the FLS980 series fluorescence spectrometer with an excitation wavelength of 500 nm. Urbach energy (EU) was detected by HS-EQE (ENLITECH PECT-600) and obtained by fitting the tail of the band. The depth profile of the perovskite film deposited on the ITO substrate was recorded by using ToF-SIMS (model ION ToF-SIMS 5). Cationic mode: the pulsed primary Bi3+ ion source was operated at 30 keV and 0.5 pA on a 100*100 μm2 area to bombard the sample surface to produce secondary ions. The sputtering was performed with a O2 beam operated at 1 keV and 150 nA on a 300*300 μm2 area. Anionic mode: the pulsed primary Bi3+ ion source was operated at 30 keV and 0.5 pA on a 100*100 μm2 area to bombard the sample surface to produce secondary ions. The sputtering was performed with a Cs+ beam operated at 1 keV and 30 nA on a 300*300 μm2 area. Temperature dependent polarized Raman spectroscopy was performed using a Thermo Fisher Scientific DXR2Xi Raman microscopy equipped with a 633 nm diode-pumped solid-state laser. Raman spectra of the perovskite single crystal were measured at different temperatures (250 K, 300 K, 350 K and 400 K) under various polarization angles (0°, 30°, 60° and 90°), respectively. Electrochemical impedance spectroscopy (EIS) was conducted using a potentiometer (CHI604E, CH instrument) under dark within the frequency range of 1 MHz to 10 mHz, with an AC amplitude of 5 mV. Mott-Schottky analysis was performed using a potentiometer (CHI604E, CH instrument) at a frequency of 1000 Hz and an applied voltage range from 0 V to 1.5 V, with an AC amplitude of 5 mV. Transient photovoltage (TPV) and transient photocurrent (TPC) signals were detected by Piaos 4.0 system (FLUXiM) with a 60 mW LED light source. For Transient Photo-Current (TPC) measurement, the offset voltage was set to 1.1 V for the devices. Besides, the light-pulse length, the setting time and the follow-up time were all 100 ms. For Transient Photo-Voltage (TPV) measurement, we applied the Open Circuit Voltage Decay (OCVD), which was also called the larger signal TPV. It measured the devices which were in the open-circuit condition. First, the carriers were generated by the pulsed light and then the light was turned off to simultaneously measure the attenuation of a voltage signal. The light-pulse length, the setting time and the follow-up time were all 100 ms. IMPS and IMVS measurements (Paios 4.0 system, FLUXiM) were performed at log arithmic light intensities ranging from 10% to 80% of 1-sun white LED illumination (10% intervals), with a frequency range of 100 Hz to 1 MHz. Electroluminescence (EL) spectra were measured under dark conditions using the ENLITECH REPS-VOC system to characterize the performance of PSCs. In the EQEEL measurement, the J-V curve was recorded with a starting voltage of -0.1 V, a terminating voltage of 1.5 V, and a step size of 0.02 V. Current-voltage characteristics were measured by a Keithley 2400 source under standard AM 1.5 G (100 mW/cm2, SAN EI: Japan). The light intensity is calibrated by standard silicon solar cells calibrated by Newport. Current density-voltage (J-V) curves are collected from forward scan and reverse scan (-0.1 V to 1.5 V). The voltage step is 20 mV and the delay time at each point is 100 ms. The area of the small area PSCs is 0.12 cm2, and the J-V curve is obtained by using a metal mask with an area of 0.09 cm2 to cover the cell. The area of the large area PSCs is 1.10 cm2, and the J-V curve is obtained by using a metal mask with an area of 1 cm2 to cover the cell. Monochromatic EQE spectra were measured as functions of wavelength under alternating current mode, utilizing monochromatic incident light intensity equivalent to 1 × 1016 photons cm⁻2 with bias voltage maintained at 0 V (QE-R3011). Light intensity emitted by the solar simulator was calibrated based on a standard silicon solar cell provided by PV Measurements. The thermal stability of devices was measured regularly after heating on 85 °C. The maximum power point (MPP) tracking was conducted within a custom-built N2-filled box under 1 sun illumination (white light LED array) at room temperature (Multi-Channels Solar Cells Stability Test System, Wuhan 91PVKSolar Technology Co. Ltd, China).
Computational method
Adsorption energy for functional molecules adsorbed on perovskite with iodine vacancies (VI), lead occupied the interstitial defects of iodine (PbI), and lead vacancies (VPb) were employed in the Density functional theory (DFT) calculations, which were performed to investigate atomic-level electronic and structural properties using the DMOL3 and CASTEP packages. The Perdew-Burke-Ernzerhof generalized gradient approximation (PBE-GGA) with norm-conserving pseudopotentials was employed for exchange-correlation interactions, while core-electron effects were treated via the frozen-core projector-augmented wave method. A plane-wave cutoff energy of 500 eV was applied, with self-consistent field convergence thresholds set at 10⁻5 eV for energy and 0.02 eV/Å for atomic forces.
Vibration entropy of FA+ was calculated by DFT calculation with the Vienna ab initio simulation package (VASP). The exchange-correlation energy is described by the PBE-GGA exchange-correlation energy functional. The structure optimizations of perovskite were carried out by allowing all atomic positions to vary and fixing lattice parameters until the energy difference of successive atom configurations was less than 10-6 eV. The force on each atom in the relaxed structures was less than 0.015 eV/Å. The cutoff energy for the plane-wave was set to 400 eV. The k-point spacing was set to be smaller than 0.03 Å-1 over Brillouin zone (BZ).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All the main data are available in the main text, the Supplementary Information, and the Source Data file. All other data of this study are available from the corresponding authors on request. Source data are provided with this paper.
References
Chen, H. et al. Improved charge extraction in inverted perovskite solar cells with dual-site-binding ligands. Science 384, 189–193 (2024).
Xu, T. et al. Surface chemistry-induced reconstruction of inorganic perovskites for efficient and stable inverted solar cells. Joule 9, 101826 (2025).
Luo, M. et al. Hybrid perovskites unlocking the development of light-emitting solar cells. Nat. Rev. Mater. 9, 295–297 (2024).
Tian, R. et al. Material and device design of flexible perovskite solar cells for next-generation power supplies. Adv. Mater. 36, 2311473 (2024).
Zhang, Y. et al. Improved fatigue behaviour of perovskite solar cells with an interfacial starch–polyiodide buffer layer. Nat. Photonics 17, 1066–1073 (2023).
Ren, X. et al. Mobile iodides capture for highly photolysis- and reverse-bias-stable perovskite solar cells. Nat. Mater. 23, 810–817 (2024).
Zhu, H. et al. Long-term operating stability in perovskite photovoltaics. Nat. Rev. Mater. 8, 569–586 (2023).
Ma, S. et al. Development of encapsulation strategies towards the commercialization of perovskite solar cells. Energy Environ. Sci. 15, 13–55 (2022).
Li, N. et al. Barrier reinforcement for enhanced perovskite solar cell stability under reverse bias. Nat. Energy 9, 1264–1274 (2024).
Hui, W. et al. Stabilizing black-phase formamidinium perovskite formation at room temperature and high humidity. Science 371, 1359–1364 (2021).
Wang, S. et al. Accelerated degradation of methylammonium lead iodide perovskites induced by exposure to iodine vapour. Nat. Energy 2, 16195 (2016).
Frolova, L. A. et al. The chemical origin of the p-type and n-type doping effects in the hybrid methylammonium–lead iodide (MAPbI3) perovskite solar cells. Chem. Commun. 51, 14917–14920 (2015).
Chi, W. & Banerjee, S. K. Achieving resistance against moisture and oxygen for perovskite solar cells with high efficiency and stability. Chem. Mater. 33, 4269–4303 (2021).
Niu, G. et al. Review of recent progress in chemical stability of perovskite solar cells. J. Mater. Chem. A 3, 8970–8980 (2015).
Akbulatov, A. F. et al. Probing the intrinsic thermal and photochemical stability of hybrid and inorganic lead halide perovskites. J. Phys. Chem. Lett. 8, 1211–1218 (2017).
Tuo, B. et al. A novel radical-reaction interruption strategy for enhancing the light stability of perovskite solar cells. Energy Environ. Sci. 17, 2945–2955 (2024).
Eames, C. et al. Ionic transport in hybrid lead iodide perovskite solar cells. Nat. Commun. 6, 7497 (2015).
Chen, T. et al. Inhibition of defect-induced α-to-δ phase transition for efficient and stable formamidinium perovskite solar cells. Nat. Commun. 14, 6125 (2023).
Liu, X. et al. Stabilization of photoactive phases for perovskite photovoltaics. Nat. Rev. Chem. 7, 462–479 (2023).
Li, W. et al. Chemically diverse and multifunctional hybrid organic–inorganic perovskites. Nat. Rev. Mater. 2, 16099 (2017).
Naqvi, F. H. & Ko, J.-H. Lattice dynamics and phase transitions in FAPbBr3 single crystals: Temperature- and pressure-dependent Raman spectroscopy. J. Raman Spectrosc. 54, 1138–1149 (2023).
Chen, T. et al. Entropy-driven structural transition and kinetic trapping in formamidinium lead iodide perovskite. Sci. Adv. 2, e1601650 (2016).
Mishra, A. et al. A complete picture of cation dynamics in hybrid perovskite materials from solid-state NMR spectroscopy. J. Am. Chem. Soc. 145, 978–990 (2023).
Tian, Y. et al. High-entropy hybrid perovskites with disordered organic moieties for perovskite solar cells. Nat. Photonics 18, 960–966 (2024).
Yi, C. et al. Entropic stabilization of mixed A-cation ABX3 metal halide perovskites for high performance perovskite solar cells. Energy Environ. Sci. 9, 656–662 (2016).
Wang, Y. et al. High-entropy perovskites for energy conversion and storage: Design, synthesis, and potential applications. Small Methods 7, 2201138 (2023).
Zhou, J. et al. Molecular contacts with an orthogonal π-skeleton induce amorphization to enhance perovskite solar cell performance. Nat. Chem. 17, 564–570 (2025).
Svensson, P. H. & Kloo, L. Synthesis, structure, and bonding in polyiodide and metal iodide−iodine systems. Chem. Rev. 103, 1649–1684 (2003).
Ni, Z. et al. Evolution of defects during the degradation of metal halide perovskite solar cells under reverse bias and illumination. Nat. Energy 7, 65–73 (2021).
Zhang, H. et al. Modulating the dipole moment of secondary ammonium spacers for efficient 2D Ruddlesden-Popper perovskite solar cells. Angew. Chem. Int. Ed. 63, 202318206 (2024).
Wang, H. et al. Interface dipole management of D–A-type molecules for efficient perovskite solar cells. Angew. Chem. Int. Ed. 63, 202404289 (2024).
Huang, Z. et al. Anion-pi interactions suppress phase impurities in FAPbI3 solar cells. Nature 623, 531–537 (2023).
Cao, Q. et al. Efficient and stable inverted perovskite solar cells with very high fill factors via incorporation of star-shaped polymer. Sci. Adv. 7, eabg0633 (2021).
Jiang, X. et al. Understanding the role of fluorine groups in passivating defects for perovskite solar cells. Angew. Chem. Int. Ed. 62, 202313133 (2023).
Ou, Z. et al. Role of A-site cation hydrogen bonds in hybrid organic–inorganic perovskites: A theoretical insight. J. Phys. Chem. Lett. 16, 802–810 (2025).
Ren, P. et al. Surface terminated 3D/2D heterojunction enables efficient and stable perovskite solar cells. Adv. Funct. Mater. 35, 2416250 (2024).
Wang, W. et al. Pressure-induced ultrawidely tunable and ultrapure photoluminescence in 2D perovskites via fluorination of organic cations. Chem. Mater. 36, 9594–9602 (2024).
Liu, S. et al. Stable surface contact with tailored alkylamine pyridine derivatives for high-performance inverted perovskite solar cells. Adv. Mater. 37, 2415100 (2024).
Jeon, N. J. et al. Compositional engineering of perovskite materials for high-performance solar cells. Nature 517, 476–480 (2015).
Jeong, H. J. & Shin, H. How to apply Raman spectroscopy for single crystals and thin films of organic–inorganic lead halide perovskite. Korean J. Chem. Eng. 41, 3693–3701 (2024).
Brehm, G. et al. Estimation of the vibrational contribution to the entropy change associated with the low- to high-spin transition in Fe(phen)2(NCS)2 complexes: Results obtained by IR and Raman spectroscopy and DFT calculations. J. Phys. Chem. A 106, 12024–12034 (2002).
Liu, H. et al. Reversible phase transition for durable formamidinium-dominated perovskite photovoltaics. Adv. Mater. 34, 2204458 (2022).
Yang, J. et al. Crystal phase and band edge modulation of MA- and Br-free CsFA-based perovskites for efficient inverted solar cells and minimodules. Energy Environ. Sci. 18, 1732–1744 (2025).
Ghosh, D. et al. Lattice expansion in hybrid perovskites: Effect on optoelectronic properties and charge carrier dynamics. J. Phys. Chem. Lett. 10, 5000–5007 (2019).
Fisicaro, G. et al. Local order and rotational dynamics in mixed A-cation lead iodide perovskites. J. Phys. Chem. Lett. 11, 1068–1074 (2020).
Cui, C.-Y. et al. Rational design on chemical regulation of interfacial microstress engineering by matching Young’s modulus in a CsPbBr3 perovskite film with mechanical compatibility toward enhanced photoelectric conversion efficiency. ACS Appl. Mater. Interfaces 14, 20257–20267 (2022).
Li, W. et al. Light-activated interlayer contraction in two-dimensional perovskites for high-efficiency solar cells. Nat. Nanotechnol. 17, 45–52 (2022).
Liu, Z. et al. Efficient and stable FA-rich perovskite photovoltaics: From material properties to device optimization. Adv. Energy Mater. 12, 2200111 (2022).
Sun, D. et al. Chemical reduction of iodine impurities and defects with potassium formate for efficient and stable perovskite solar cells. Adv. Funct. Mater. 33, 2303225 (2023).
Bitton, S. & Tessler, N. Perovskite ionics – elucidating degradation mechanisms in perovskite solar cells via device modelling and iodine chemistry. Energy Environ. Sci. 16, 2621–2628 (2023).
Chen, Y. et al. Synergistic transition metal ion co-doping and multiple functional additive passivation for realizing 25.30% efficiency perovskite solar cells. Energy Environ. Sci. 16, 5243–5254 (2023).
Wang, Q. et al. Tailored succinic acid-derived molecular structures toward 25.41%-efficiency and stable perovskite solar cells. Adv. Mater. 36, 2307709 (2024).
Aalbers, G. J. W. et al. Effect of sub-bandgap defects on radiative and non-radiative open-circuit voltage losses in perovskite solar cells. Nat. Commun. 15, 1276 (2024).
He, A. et al. Unraveling the impact of bromine incorporation on the hybrid perovskite film phase evolution mechanism by in situ characterization. Adv. Energy Mater. 13, 2300957 (2023).
Lv, Y. et al. High-efficiency perovskite solar cells enabled by anatase TiO2 nanopyramid arrays with an oriented electric field. Angew. Chem. Int. Ed. 59, 11969–11976 (2020).
Chen, X. et al. Minimizing the buried interfacial energy loss using a fluorine-substituted small molecule for 25.92%-efficiency and stable inverted perovskite solar cells. Energy Environ. Sci. 17, 7342–7354 (2024).
Thiesbrummel, J. et al. Ion-induced field screening as a dominant factor in perovskite solar cell operational stability. Nat. Energy 9, 664–676 (2024).
Acknowledgements
The authors thank the financial support from the National Key Research and Development Program of China (Grant No. 2022YFB4200500 (Q.P.)), the National Natural Science Foundation of China (Grant Nos. 22375133 (Y.W.), 22379101 (Q.P.) and 22075190 (Q.P.)), the Natural Science Foundation of Sichuan Province (Grant No. 2024NSFSC0001 (Q.P.)), the Fundamental Research Funds for the Central Universities (Grant No. YJ2021129 (Y.W.)), and the Creative Project of Engineering Research Center of Alternative Energy Materials & Devices, Ministry of Education, Sichuan University (Grant No. AEMD-C202202 (Y.W.)). The authors thank beamline BL14B1 and BL03HB at Shanghai Synchrotron Radiation Facility (SSRF) for providing the beam time. The authors also thank Yanping Huang from Center of Engineering Experimental Teaching, School of Chemical Engineering, Sichuan University for the help of NMR measurements.
Author information
Authors and Affiliations
Contributions
Y.W. conceived the idea and designed the experiments. Y.W. and Q.P. supervised the project. X.C. fabricated solar cells and characterized the devices. Q.W. carried out the TRPL. L.S., W.H., and Q.W. contributed to the GIWAXS test and analysis. B.F. performed the DFT calculations. Q.W., P.X., and Z.X. assisted with device fabrication and characterizations. X.C. and Y.W. analyzed the data and wrote the original draft. Y.W., X.X., and Q.P. reviewed and edited the draft. All authors discussed, commented on, and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Weiguang Chi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, X., Hui, W., Wang, Q. et al. Entropy-driven strategy stabilizes photoactive halide perovskites for inverted solar cells. Nat Commun 16, 9717 (2025). https://doi.org/10.1038/s41467-025-64728-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64728-w