Abstract
Nirmatrelvir-ritonavir is generally recommended to be initiated within five days of COVID-19 symptom onset. This study examined the association between the timing of nirmatrelvir-ritonavir initiation and post-acute outcomes more precisely using territory-wide data in Hong Kong. We included patients aged ≥18 years who tested positive for SARS-CoV-2 between March 16, 2022, and November 9, 2023, and were hospitalized with COVID-19. Treatment groups were formed based on the time from the positive RT-PCR date to nirmatrelvir-ritonavir initiation. Among 15,978 patients who received nirmatrelvir-ritonavir, 10,028 (62.8%) patients were included in Day 0 group, 4973 (31.1%) in Day 1 group, and 977 (6.1%) in Day 2 or later group. The control group comprised 22,312 patients who did not receive nirmatrelvir-ritonavir. Compared with the control group, the risks of post-acute mortality were significantly lower in Day 0 group (hazard ratio [HR] 0.51, 95% CI 0.46–0.56; p < 0.0001) and Day 1 group (HR 0.66, CI 0.59–0.74; p < 0.0001), but not in Day 2 or later group. Meta-regression results showed that more immediate initiation was associated with lower risks of death and all-cause hospitalization. Our findings suggested that the antiviral should be prescribed immediately after COVID-19 diagnosis for achieving its greatest benefit on improving post-COVID outcomes.
Similar content being viewed by others
Introduction
COVID-19 is increasingly recognized as a disease with potential long-term health consequences extending beyond the acute phase of infection. Patients may experience a wide range of post-acute complications after recovering from the acute phase of COVID-19, affecting pulmonary, cardiovascular, renal, and other organ systems1. These complications may lead to severe outcomes. A systematic review has highlighted that patients who survive an index hospital admission with COVID-19 remain at significant risk for long-term hospital readmission and death following discharge2.
Nirmatrelvir-ritonavir is an authorized oral antiviral for treating symptomatic COVID-19 in non-hospitalized individuals who are at high risk of developing severe COVID-193. Literature has well demonstrated its short-term benefits in outpatient populations4,5. Apart from that, recent studies showed that prescribing nirmatrelvir-ritonavir during the acute infection phase may lower the risk of post-COVID conditions6. Similar long-term benefits on preventing post-acute sequelae were also observed among inpatients and immunocompromised patients with COVID-197,8.
In many clinical guidelines for managing patients with COVID-19, nirmatrelvir-ritonavir is generally recommended to be prescribed within 5 days of diagnosis or symptom onset; however, the duration of acute symptoms is typically short, especially for Omicron infections, which have a median duration of five days9, raising questions about the timing and necessity of antiviral treatment in some cases. A previous study indicated that initiation of nirmatrelvir-ritonavir treatment 0 to 1 days after symptom onset or diagnosis demonstrated a lower risk of acute conditions, compared to starting the treatment 2 or more days after onset10. Given the connection between acute disease severity and long-term adverse health outcomes11, we hypothesize that immediate initiation of nirmatrelvir-ritonavir following diagnosis may provide greater benefits in reducing the risk of post-COVID mortality, hospitalization, and sequelae. In this study, we examined the associations between the timing of nirmatrelvir-ritonavir initiation and post-acute outcomes among hospitalized patients with COVID-19 in Hong Kong.
Results
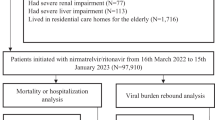
Among 83,204 patients with positive RT-PCR test results between March 16, 2022, and November 9, 2023, 38,290 patients met the eligibility criteria and were included in the study cohort (Fig. 1), of whom 18,977 (49.6%) were female, and 19,313 (50.4%) were male. The median age of the study cohort was 75.0 years (IQR, 63.0–86.0). The median follow-up time was 365.0 days (IQR, 335.0–365.0). The treatment groups comprised 15,978 patients who received nirmatrelvir-ritonavir, of whom 10,028 (62.8%) were in the Day 0 group, 4973 (31.1%) in the Day 1 group, and 977 (6.1%) in the Day 2 or later group. The control group consisted of 22,312 patients who did not receive nirmatrelvir-ritonavir. The median age was 75.0 years (IQR, 59.0–86.0) for the control group, 76.0 years (68.0–85.0) for the Day 0 group, 75.0 years (65.0–85.0) for the Day 1 group, and 73.0 years (62.0–83.0) for the Day 2 or later group (Table 1). The control group had a smaller proportion of patients who received three or more doses of vaccine (42.9% [9570/22312]) compared with Day 0 group (67.6% [6781/10028]), Day 1 group (62.6% [3111/4973]), and Day 2 or later group (56.9% [556/977]). The control group had a larger proportion of patients who received dexamethasone (32.8% [7309/22312]) compared with Day 0 group (10.6% [1062/10028]), the Day 1 group (10.6% [527/4973]), and the Day 2 or later group (12.6% [123/977]). The control group had a larger proportion of patients who received remdesivir (25.8% [5765/22312]) compared with the Day 0 group (7.4% [740/10028]), Day 1 group (6.7% [335/4973]), and the Day 2 or later group (9.1% [89/977]).
Flowchart of patient inclusion and exclusion.
Study covariates were well balanced after standardized mortality ratio weighting was applied, except for the week of the index date in the comparison between Day 0 group and the control group (Supplementary Figs. 1–3), which was included in the cause-specific Cox models for doubly robust adjustment. The scaled Schoenfeld residual plots did not show clear evidence suggesting violation of the proportional hazards assumption (Supplementary Figs. 4–6).
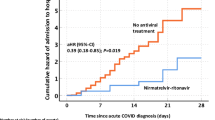
For the outcome of post-acute death, compared to the control group, the risks were lower in both Day 0 group (hazard ratio [HR] 0.51, 95% CI 0.46–0.56; p < 0.0001) and Day 1 group (HR 0.66, CI 0.59–0.74; p < 0.0001) (Fig. 2 and Fig. 3A). For the outcome of post-acute all-cause hospitalization, in comparison with the control group, the risks were lower in both Day 0 group (HR 0.73, CI 0.70–0.77; p < 0.0001) and Day 1 group (HR 0.82, CI 0.78–0.87; p < 0.0001). There is no significant difference between the Day 2 or later group and the control group in the risks of post-acute death and all-cause hospitalization. Unadjusted and adjusted risk differences and hazard ratios are provided in Supplementary Tables 5, 6. Meta-regression results showed declining effect of nirmatrelvir-ritonavir along with the initiation time in respect to post-acute death (regression coefficient 0.22, CI 0.13–0.30; p < 0.0001) (Fig. 3B) and post-acute all-cause hospitalization (regression coefficient 0.11, CI 0.07–0.14; p < 0.0001) (Fig. 3C). Details on the interpretation of the regression coefficients are provided in Supplementary Tables 7, 8.
Cumulative incidence curves of the treatment groups and control group for the outcomes of A death and B all-cause hospitalization. The shaded areas represent the 95% confidence bands.
A Forest plot stratified by different times to treatment initiation. B Bubble plot of cause-specific hazard ratio versus time to nirmatrelvir-ritonavir initiation for the outcome of death. C Bubble plot of cause-specific hazard ratio versus time to nirmatrelvir-ritonavir initiation for the outcome of all-cause hospitalization. For A, in the control group, there were 22,312 patients. The measure of centre for the error bars is hazard ratio, and the error bars represent the 95% confidence intervals from cause-specific Cox proportional hazards models. The statistical test used was a two-sided Wald test. For the outcome of post-acute inpatient death, the number of events was 2817, and the cumulative incidence (95% CI) on Day 365 was 14.18% (13.69, 14.67). For the outcome of post-acute all-cause hospitalization, the number of events was 10,372, and the cumulative incidence (95% CI) on Day 365 was 53.90% (53.18, 54.60). The axis is in log scale. For B and C, the measure of centre for the error bars is the hazard ratio, and the error bars represent the 95% confidence intervals from cause-specific Cox proportional hazards models. The shaded areas represent the 95% confidence bands from meta-regression models. Sizes of the circles are proportional to the weights. Sample sizes of each group: Day 0, n = 10028; Day 1, n = 4973; Day 2, n = 597; Day 3, n = 211; Day 4, n = 82.
In terms of post-acute sequelae, for the outcomes of atrial fibrillation and acute respiratory distress syndrome, Day 0 group had the lowest risk, followed by Day 1 group, and then Day 2 or later group, compared to the control group (Fig. 4). A similar pattern also occurred for the outcome of incident single organ impairment.
Covariates were adjusted for using standardized mortality ratio weighting. The measure of centre for the error bars is the hazard ratio, and the error bars represent the 95% confidence intervals from cause-specific Cox proportional hazards models. The statistical test used was a two-sided Wald test. The axis is in log scale.
Results from the subgroup analyses showed notable differences in the effect sizes among different initiation times on post-acute death and all-cause hospitalization between patients aged <65 years and ≥65 years (Supplementary Fig. 7), in which declining patterns were relatively weaker in the younger individuals. The declining patterns of effect sizes were generally similar between males and females, and between those receiving less than 3 doses of vaccine and those receiving 3 or more doses of vaccine (Supplementary Figs. 8, 9).
Sensitivity analyses showed results that were robust and consistent with those from the main analysis, indicating that the effectiveness of nirmatrelvir-ritonavir was higher when initiated immediately after diagnosis (Supplementary Figs. 10–14). In the comparisons between treatment groups, for the outcome of post-acute death, Day 0 group had significantly lower risk compared to Day 1 group (HR 0.80, 95% CI 0.70–0.91; p = 0.00063) and Day 2 or later group (HR 0.58, 95% CI 0.46–0.74; p < 0.0001), and Day 1 group had significantly lower risk compared to Day 2 or later group (HR 0.75, 95% CI 0.59–0.94; p = 0.013) (Supplementary Figs. 15–17). For the outcome of post-acute all-cause hospitalization, the risk was significantly lower in Day 0 group than in Day 1 group (HR 0.90, 95% CI 0.85–0.95; p < 0.0001) and in Day 2 or later group (HR 0.77, 95% CI 0.69–0.85; p < 0.0001), and the risk was significantly lower in Day 1 group than in Day 2 or later group (HR 0.86, 95% CI 0.77–0.95; p = 0.0033).
Discussion
Various investigations have shown the benefits of nirmatrelvir-ritonavir in reducing the risk of post-acute death, hospitalization, and sequelae6,7,8. However, the five-day post-diagnosis period of treatment initiation is somewhat vague, particularly given that the acute infection period for circulating SARS-CoV-2 variants is relatively short. In this study, we examined the associations of prescribing nirmatrelvir-ritonavir at different time post-diagnosis with post-acute outcomes in Hong Kong, where Omicron variants accounted for nearly all SARS-CoV-2 infections. Our study primarily demonstrated that immediate initiation of nirmatrelvir-ritonavir was associated with greater reductions in risks of post-acute mortality and all-cause hospitalization. Alongside evidence of early initiation providing acute benefits10, our findings demonstrate additional long-term advantages related to early nirmatrelvir-ritonavir prescriptions. This supports the recommendation for immediate treatment, especially since most clinical guidelines only suggest a five-day window. However, it is important to note that false-negative results from rapid antigen tests may delay antiviral treatment12, presenting a practical challenge.
The observed relationship between immediate nirmatrelvir-ritonavir prescription and long-term outcomes in this study is likely supported by the timely antiviral effect on alleviating or even halting the progression of acute severity, which has commonly been shown to be a risk factor for post-COVID outcomes13,14,15. According to Wong et al.10, prescribing nirmatrelvir-ritonavir treatment 0-1 days after symptom onset or diagnosis was significantly associated with lower risk of all-cause mortality and hospitalization 28-day post-infection, compared to later prescriptions. The potential underlying mechanism is that immediate nirmatrelvir-ritonavir inhibits early viral replication, thereby preventing or slowing down progression to severe disease11,16. In addition, nirmatrelvir-ritonavir may help diminish the immune dysregulation, which can lead to inflammation and damage to tissues and organs. By alleviating the immune response through early viral suppression, nirmatrelvir-ritonavir treatment may lower the risk of subsequent immune-related complications that contribute to post-acute conditions6,15. Timely antiviral treatment may facilitate a more robust recovery during the acute phase, helping the body to achieve homoeostasis effectively.
According to our subgroup analysis, the association of immediate initiation with higher effectiveness was more pronounced among older patients (aged above 65 years) than among younger patients (aged 65 years or below). It has been shown that older patients are generally at a higher risk of prolonged symptoms associated with COVID-19. The infection may trigger or worsen age-related chronic conditions, including cardiovascular and respiratory diseases17. Aging causes significant alterations in immune function, including immunosenescence and inflamm-aging, which weaken the body's ability to effectively clear infections and reduce vaccine responsiveness18. Therefore, immediate nirmatrelvir-ritonavir initiation may be particularly crucial for older patients by facilitating early suppression of viral replication, improving long-term outcomes.
The notable strength of this cohort study is that it used real-world inpatient data from all public hospitals. In Hong Kong, nearly 90% of hospital admissions occur in public settings. Given the close clinical monitoring during inpatient stays, the validity of study data, especially the antiviral prescription records, was ensured. Nevertheless, there were several major limitations in this study. Firstly, our study did not employ virologic rebound as a marker of viral persistence for the study outcome10, given that the accumulating evidence suggests no relationship between virologic rebound and nirmatrelvir-ritonavir treatment, as well as acute COVID-19 conditions19,20,21. Secondly, as the study period was dominated by a surge in Omicron infections22; caution should be exercised when generalizing the results to other variants. Nevertheless, considering the constant or decreasing trend of acute severity as well as the duration of acute phase across variants and sub-lineages23,24,25, our suggestion of immediate nirmatrelvir-ritonavir prescription is likely to remain robust for newly emerging variants, provided that no antiviral resistance develops26. Thirdly, our study did not investigate other antivirals, such as remdesivir, owing to the relatively small proportion of recipients. Fourthly, this observational study was likely subject to residual confounding, such as health-seeking behaviour. Lastly, due to missingness and potential recall bias, we did not use optional self-report symptom onset dates to define the index date in the main analysis. Instead, we used positive PCR test dates, which were objectively recorded and consistently available across patients. We believe they provide a meaningful basis for hospital or public health system management.
In conclusion, our study primarily demonstrated that immediate initiation of nirmatrelvir-ritonavir following diagnosis was associated with greater reductions in the risks of post-acute mortality and hospitalization, potentially due to early inhibition of viral replication and early alleviation of acute COVID-19 progression from the antiviral effect. In addition to the general recommendation for a five-day prescription period, our findings suggest that nirmatrelvir-ritonavir should be prescribed immediately after a COVID-19 diagnosis to maximize the benefits of the antiviral treatment not only in mitigating acute severity but also in reducing the risk of adverse long-term outcomes.
Methods
Settings, study design and data sources
This retrospective cohort study utilized real-world, territory-wide data from the Hospital Authority and the Department of Health in Hong Kong. The Hong Kong Hospital Authority is the statutory body managing services provided by public hospitals, which are accessible to all Hong Kong residents. The services cover all patients infected with COVID-19 in Hong Kong27. The Hospital Authority’s electronic health record system contains extensive patient data, encompassing demographics, dates of registered death, hospital admissions, emergency department visits, diagnoses, prescriptions, procedures, and laboratory results. The data from this system were linked to the COVID-19 vaccination records from the Department of Health. These population-based databases have been widely utilized in prior research5,7. To protect patient confidentiality, the data were anonymized with pseudo numbers. The study was reported according to STROBE guidelines.
Participants
This study included COVID-19 patients who had a positive RT-PCR test result between March 16, 2022, and November 9, 2023. The start date corresponded to when nirmatrelvir-ritonavir became available in Hong Kong, and the end date allowed for at least 21 days of follow-up prior to the study’s data cutoff on November 30, 2023 (this 21-day time frame is frequently adopted in post-COVID-19 studies)27. This period was dominated by the Omicron variant.
Another inclusion criterion was that patients were admitted to the hospital within 3 days before or after the index date (the date of the positive RT-PCR test result), which is a widely accepted definition of patients hospitalized with COVID-19 to account for potential time lags in confirming COVID-19 diagnoses7, especially during surges in SARS-CoV-2 infection. Eligible patients were also required to be aged 18 years or older.
Patients with contraindications to nirmatrelvir-ritonavir were excluded. Contraindications included the use of specific drugs within the 90 days prior to the index date (i.e., amiodarone, lumacaftor-ivacaftor, rifampicin, apalutamide, phenobarbital, rifapentine, carbamazepine, phenytoin, St John’s Wort [hypericum perforatum], ivosidenib, and primidone), severe renal impairment (i.e., an estimated glomerular filtration rate of less than 30 mL/min per 1.73 m², dialysis, or renal transplantation), and severe liver impairment (i.e., cirrhosis, hepatocellular carcinoma, or liver transplantation)5,28. In addition, patients prescribed with molnupiravir within 21 days of the index date were excluded, and patients prescribed with nirmatrelvir-ritonavir before the index date were excluded.
Ethics approval was obtained from the Joint Clinical Research Ethics Committee of The Chinese University of Hong Kong and New Territories East Cluster (CUHK-NTEC) (Ref No. 2023.006). Since this was a retrospective study using secondary data with no personal identifying information involved, the requirement for informed consent was waived by the ethics committee.
Procedures
Eligible patients were categorized into treatment and control groups. The treatment group consisted of patients who were prescribed nirmatrelvir-ritonavir, but not molnupiravir, within 21 days of the index date. The control group comprised patients who were not prescribed nirmatrelvir-ritonavir or molnupiravir within 21 days of the index date. Based on time to nirmatrelvir-ritonavir initiation (defined as the time difference in days between the index date and the nirmatrelvir-ritonavir initiation date), the treatment group was further divided into 3 groups: Day 0 group, Day 1 group, and Day 2 or later group, which comprised patients who initiated nirmatrelvir-ritonavir 0 day, 1 day, and 2 or more days after the index date, respectively10.
Covariates were selected based on literature and previous knowledge5,7. The covariates included in the analysis were age; sex; previous SARS-CoV-2 infection; the Charlson Comorbidity Index (CCI) calculated according to diagnoses before the index date (relevant ICD-9-CM codes are provided in Supplementary Table 1); concomitant pharmacological treatments initiated within 3 days before or after the index date (i.e., dexamethasone, prednisolone, remdesivir, and others [interferon-beta-1b, baricitinib, or tocilizumab]); admission to the intensive care unit (ICU) or use of ventilatory support within 3 days before or after the index date (relevant ICD-9-CM codes are provided in Supplementary Table 2); vaccination status (unvaccinated, 1–2 doses, or ≥3 doses; doses received less than 14 days before the index date were considered incomplete and did not count towards the total number of vaccine doses); and the week of the index date. Treatment choice was determined by physicians’ judgement, with reference to the Health Authority’s COVID-19 clinical guidelines. For example, according to the guidelines, remdesivir may be considered when patients could not tolerate oral antivirals or have moderate-to-severe symptoms. We controlled for other COVID-19 treatments mentioned above as covariates to further adjust for the baseline characteristics associated with treatment choice. Linear terms of the quantitative covariates were used in the models. A list of the covariates is provided in Supplementary Table 3.
Outcomes
The primary outcome of this study was post-acute inpatient death. In Hong Kong, more than 90% of deaths occur in public hospitals29, and thus the death records in our data were highly representative. The secondary outcomes were all-cause hospitalization and cause-specific hospitalization due to each of the 13 post-acute sequelae, which were selected based on evidence indicating a higher risk of these conditions among COVID-19 patients in Hong Kong27. These sequelae included congestive heart failure, atrial fibrillation, coronary artery disease, deep vein thrombosis, chronic pulmonary disease, acute respiratory distress syndrome, interstitial lung disease, seizure, anxiety, post-traumatic stress disorder, end-stage renal disease, acute kidney injury, and pancreatitis. In assessing each of the sequelae, individuals with a history of the disease of interest prior to the index date were excluded. We also classified the sequelae into outcomes related to organ impairment, including impaired lung function, impaired heart function, impaired kidney function, incident single organ impairment, and incident multi-organ impairment30. ICD-9-CM codes and the classification scheme are provided in Supplementary Table 4.
All of the outcomes were ascertained starting from 21 days after the index date. Patients were followed up from the index date until the earliest of the following: (1) inpatient death, (2) the outcome of interest in each specific analysis, (3) 365 days after the index date, or (4) the end of data availability (November 30, 2023).
Statistical analysis
Baseline characteristics of eligible patients were outlined in a table with appropriate summary statistics. For the outcomes of post-acute death and all-cause hospitalization, cumulative incidence curves were plotted with risk tables.
To control for the covariates, standardized mortality ratio weighting was applied31. Propensity scores were estimated by logistic regression models and were used to calculate weights. Covariate balance was assessed using the standardized mean difference (SMD), where a covariate with an SMD larger than 0.1 was considered imbalanced and included in the cause-specific Cox models (more details below) for doubly robust adjustment32.
To evaluate the effectiveness of nirmatrelvir-ritonavir with different times to initiation, cause-specific Cox proportional hazards models were employed to compare each of the three treatment groups (Day 0 group, Day 1 group; and Day 2 or later group) with the control group. Death prior to 21 days was treated as a competing event. The proportional hazards assumption was examined using plots of scaled Schoenfeld residuals.
To further investigate the association between the effectiveness of nirmatrelvir-ritonavir and time to initiation, hazard ratios were obtained from separately comparing patients who initiated nirmatrelvir-ritonavir on Day 0, Day 1, Day 2, Day 3, and Day 4 with the control group in terms of post-acute inpatient death and all-cause hospitalization. These hazard ratios were log-transformed and meta-regressed on time to initiation, assuming inverse-variance weighted linear mixed-effects models. The Wald test was used to assess the statistical significance of the association. We plotted hazard ratios versus time to initiation along with the fitted model to visualize the potential association.
Subgroup analyses were conducted to assess the effectiveness of nirmatrelvir-ritonavir in preventing post-acute inpatient death and all-cause hospitalization with different times to initiation stratified by age (≤65 years or >65 years), by sex (female or male), and by vaccination status (<3 doses or ≥3 doses). Sensitivity analyses included (1) using a 30-day time frame instead of the 21-day time frame in the main analysis; and (2) excluding patients who initiated nirmatrelvir-ritonavir on Day 5 or later; (3) including district as a covariate indicating which one of the 18 administrative districts patients were from, for those with data available; (4) incorporating the optionally self-reported symptom onset dates into the definition of the index date, that is, the index date was defined as the symptom onset date when available, and as the positive PCR test date when the symptom onset date was missing; (5) using Fine-Gray subdistribution hazards models that treat death prior to day 21 as a competing event. Additionally, we made pairwise comparisons for the outcomes of post-acute inpatient death and all-cause hospitalization among the three treatment groups using cause-specific Cox proportional hazards models, with subgroup analyses stratified by age.
All statistical analyses were conducted using R statistical software (version 4.4.2).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The Hong Kong Hospital Authority and Department of Health, the Government of the Hong Kong Special Administrative Region, are the data custodians, and data requests to these parties can be made via email (hacpaaedr@ha.org.hk) and website (https://www.dh.gov.hk/english/aboutus/aboutus_pps/aboutus_pps.html), respectively. In this study, the participants' surveillance data and medication records were provided by the Hong Kong Hospital Authority. The vaccination history was extracted from the COVID-19 surveillance database provided by the Department of Health in Hong Kong. Restrictions apply to the availability of these data, which were used under an agreement for the purposes of scientific research. The authors do not have the right to transfer or release the data, in whole or in part, and in whatever form or media, or to any other parties or place outside of Hong Kong, and must fully comply with the duties under the law relating to the protection of personal data, including those under the Personal Data (Privacy) Ordinance and its principles in all aspects.
References
Desai, A. D., Lavelle, M., Boursiquot, B. C. & Wan, E. Y. Long-term complications of COVID-19. Am. J. Physiol. -Cell Physiol. 322, C1–C11 (2022).
Newman, J. et al. Longer term post-hospital morbidity and mortality following admission with COVID-19: a systematic review and meta-analysis. ERJ Open Res. 01281–2024. https://doi.org/10.1183/23120541.01281-2024 (2025).
Fact sheet for healthcare providers: emergency use authorization for Paxlovid™. https://www.fda.gov/media/155050/download (accessed April 14, 2025).
Hammond, J. et al. Oral nirmatrelvir for high-risk, nonhospitalized adults with COVID-19. N. Engl. J. Med. 386, 1397–1408 (2022).
Wong, C. K. H. et al. Real-world effectiveness of early molnupiravir or nirmatrelvir-ritonavir in hospitalised patients with COVID-19 without supplemental oxygen requirement on admission during Hong Kong’s omicron BA.2 wave: a retrospective cohort study. Lancet Infect. Dis. 22, 1681–1693 (2022).
Xie, Y., Choi, T. & Al-Aly, Z. Association of treatment with nirmatrelvir and the risk of post-COVID-19 condition. JAMA Intern. Med. 183, 554–564 (2023).
Wang, H. et al. Association of nirmatrelvir-ritonavir with post-acute sequelae and mortality in patients admitted to hospital with COVID-19: a retrospective cohort study. Lancet Infect. Dis. 24, 1130–1140 (2024).
Lin, G. et al. Association of nirmatrelvir-ritonavir with post-acute sequelae and mortality among patients who are immunocompromised with COVID-19 in Hong Kong: a retrospective cohort study. Lancet Rheumatol. 7, e108–e117 (2025).
Menni, C. et al. Symptom prevalence, duration, and risk of hospital admission in individuals infected with SARS-CoV-2 during periods of omicron and delta variant dominance: a prospective observational study from the ZOE COVID Study. Lancet 399, 1618–1624 (2022).
Wong, C. K. H. et al. Optimal timing of nirmatrelvir/ritonavir treatment after COVID-19 symptom onset or diagnosis: target trial emulation. Nat. Commun. 14, 8377 (2023).
Cai, M., Xie, Y., Topol, E. J. & Al-Aly, Z. Three-year outcomes of post-acute sequelae of COVID-19. Nat. Med. 30, 1564–1573 (2024).
Rubin, R. Paxlovid is effective but underused-here’s what the latest research says about rebound and more. JAMA 331, 548–551 (2024).
Mateu, L. et al. Determinants of the onset and prognosis of the post-COVID-19 condition: a 2-year prospective observational cohort study. Lancet Reg. Health Eur. 33, 100724 (2023).
Tsampasian, V. et al. Risk factors associated with post−COVID-19 condition: a systematic review and meta-analysis. JAMA Intern. Med 183, 566 (2023).
Greenhalgh, T., Sivan, M., Perlowski, A. & Nikolich, J. Ž Long COVID: a clinical update. Lancet 404, 707–724 (2024).
Pujadas, E. et al. SARS-CoV-2 viral load predicts COVID-19 mortality. Lancet Respir. Med. 8, e70 (2020).
Mansell, V., Hall Dykgraaf, S., Kidd, M. & Goodyear-Smith, F. Long COVID and older people. Lancet Healthy Longev. 3, e849–e854 (2022).
Bajaj, V. et al. Immunity, and COVID-19: How Age Influences the Host Immune Response to Coronavirus Infections?. Front Physiol. 11, 571416 (2021).
Wong, G. L. et al. Incidence of viral rebound after treatment with nirmatrelvir-ritonavir and molnupiravir. JAMA Netw. Open 5, e2245086 (2022).
Wong, C. K. H. et al. Viral burden rebound in hospitalised patients with COVID-19 receiving oral antivirals in Hong Kong: a population-wide retrospective cohort study. Lancet Infect. Dis. 23, 683–695 (2023).
Harrington, P. R. et al. Evaluation of SARS-CoV-2 RNA rebound after nirmatrelvir/ritonavir treatment in randomized, double-blind, placebo-controlled trials - United States and international sites, 2021-2022. MMWR Morb. Mortal. Wkly Rep. 72, 1365–1370 (2023).
Chong, K. C. et al. Changes in all-cause and cause-specific excess mortality before and after the Omicron outbreak of COVID-19 in Hong Kong. J. Glob. Health 13, 06017 (2023).
Guo, Z. et al. Comparing the incubation period, serial interval, and infectiousness profile between SARS-CoV-2 Omicron and Delta variants. J. Med. Virol. 95, e28648 (2023).
Levy, M. E. et al. Reduced likelihood of hospitalization with the JN. 1 or HV. 1 Severe Acute Respiratory Syndrome Coronavirus 2 variants compared with the EG. 5 variant. J. Infect. Dis. 230, 1197–1201 (2024).
Padilla, S. et al. Long COVID across SARS-CoV-2 variants, lineages, and sublineages. iScience 27, 109536 (2024).
Service, R. F. Bad news for Paxlovid? Resistance may be coming. Science 377, 138–139 (2022).
Lam, I. C. H. et al. Long-term post-acute sequelae of COVID-19 infection: a retrospective, multi-database cohort study in Hong Kong and the UK. EClinicalMedicine 60, 102000 (2023).
Wan, E. Y. F. et al. Effectiveness of molnupiravir and nirmatrelvir–ritonavir in hospitalized patients with COVID-19: a target trial emulation study. Ann. Intern Med 176, 505–514 (2023).
Chung, R. Y.-N. et al. Healthcare inequalities in emergency visits and hospitalisation at the end of life: a study of 395 019 public hospital records. BMJ Support Palliat. Care https://doi.org/10.1136/bmjspcare-2020-002800 (2021).
Dennis, A. et al. Multi-organ impairment and long COVID: a 1-year prospective, longitudinal cohort study. J. R. Soc. Med. 116, 97–112 (2023).
Brookhart, M. A., Wyss, R., Layton, J. B. & Stürmer, T. Propensity score methods for confounding control in nonexperimental research. Circ. Cardiovasc. Qual. Outcomes 6, 604–611 (2013).
Funk, M. J. et al. Doubly robust estimation of causal effects. Am. J. Epidemiol. 173, 761–767 (2011).
Acknowledgements
We thank the Hospital Authority and the Department of Health, Hong Kong Government, for providing the data for this study. The Centre for Health Systems and Policy Research, funded by the Tung Foundation, is acknowledged for the support throughout the conduct of this study. This research was funded by Health and Medical Research Fund [EKY: grant numbers COVID190105, COVID19F03, INF-CUHK-1, COVID1903003], RGC Collaborative Research Fund (CKPM: C6036-21GF), CUHK Direct Grant (KCC: 2025.059), National Natural Science Foundation of China (KCC: 72574190), and RGC theme-based research schemes (CKPM: T11-705/21-N). The funders of the study had no role in study design, data collection, data analysis, data interpretation, writing of the manuscript, or the decision to submit for publication. This research was funded by National Natural Science Foundation of China (SZ: 12401648), the Noncommunicable Chronic Diseases - National Science and Technology Major Project of China (SZ: 2023ZD0519300), the Young Elite Scientists Sponsorship (YESS) Program by CAST (SZ: 24JCQNJC00610), the Natural Science Foundation of Tianjin Municipal Science and Technology Commission (SZ: 24JCQNJC00610), and Tianjin Medical University start-up funding (SZ).
Author information
Authors and Affiliations
Contributions
Study design and conceptualisation: K.C.C., Y.W., H.W., G.L. Data collection and pre-processing: G.L., Z.G., H.W., C.H.K.Y., T.Y.C., Y.W., E.K.Y., K.C.C. Data analysis and interpretation: G.L., Y.W., H.W., Z.G., S.Z., K.C.C. Writing - Original Draft: K.C.C., G.L., X.X., C.T.H., C.L., S.Z. Writing - Review and Editing: C.K.H.W., Z.G., K.L., A.Y., C.K.P.M., D.S.C.H., E.K.Y. E.K.Y. and K.C.C. have accessed and verified all the data. All authors critically reviewed the manuscript and gave final approval for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chong, K.C., Wei, Y., Wong, C.K.H. et al. Greater benefits of immediate nirmatrelvir-ritonavir initiation for post-COVID outcomes: a population-based retrospective cohort study. Nat Commun 16, 9872 (2025). https://doi.org/10.1038/s41467-025-64851-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-64851-8