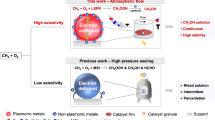
Abstract
Photocatalytic methanol dehydration to dimethyl ether (DME) offers a sustainable alternative to energy-intensive thermocatalysis, yet its practical application remains constrained by low efficiency. Herein, we designed Ni-doped plasmonic W18O49 nanowires that synergistically integrates low-coordinated W and Ni dual active sites with surface plasmon resonance for enhanced photocatalytic performance. The synergistic effect of W and Ni dual sites is amplified by plasmonic electron oscillations to facilitate the C-O bond cleavage and C-O-C coupling, driving efficient methanol-to-DME conversion. The optimized Ni0.66-W18O49 achieves a DME yield of 133.7 ± 3.3 mmol g-1 h-1 with 98.7% selectivity under 400 mW cm-2 illumination. The versatility of the catalyst is demonstrated through C2+ alcohol dehydration, achieving 40-80% rate enhancements and a recorded isobutylene yield of 3.7 mol g-1 h-1. This study highlights the huge potential of rationally engineered plasmonic semiconductors in solar-driven chemical synthesis, particularly for C-O bond activation and coupling reactions.
Similar content being viewed by others
Introduction
Dimethyl ether (DME) is extensively applied as a clean fuel, friendly aerosol, and green refrigerant1,2,3. According to the latest market analysis, the global DME market was valued at approximately USD 8.69 billion in 2023 and is expected to grow to USD 25.4 billion by 20364, reflecting a significant growth potential. Conventional industrial production relies on energy-intensive methanol dehydration over metal-supported aluminosilicate catalysts at elevated temperatures (230–400 °C) and pressures (0.5–1.5 MPa)5,6,7. This thermodynamic demand highlights the critical need for catalyst innovations enabling efficient methanol conversion under mild conditions. While solar-driven photocatalysis presents a sustainable alternative, current catalysts exhibit insufficient activity (DME generation rate of 4.3 mmol g−1 h−1) for industrial implementation8,9,10. Plasmonic photocatalysis offers a superior-active approach for chemical transformation by leveraging surface plasmon resonance (SPR) to concentrate optical energy at catalytically active interfaces11,12,13,14,15. Our group previously demonstrated the exceptional capability of plasmonic W18O49 nanowires in alcohol-to-alkene conversion, achieving an isobutylene production rate of 1.8 mol g−1 h−1 through plasmon-driven C-O bond activation16,17,18.
Unlike the intramolecular dehydration for alkene generation, DME synthesis necessitates a bifunctional mechanism involving sequential C-O bond cleavage and C-O-C coupling, and this process requires spatially distinct active sites19. While pristine W18O49 effectively facilitates C-O bond dissociation, its inability to mediate C-O-C coupling restricts DME productivity. Systematic density functional theory (DFT) simulations (Fig. 1) reveal atomic-level insights into methanol activation pathways. Low-coordinated W sites with coordination numbers (CN) of 4 and 5 were identified as primary methanol adsorption sites, with energies of −1.03 eV and −0.85 eV, respectively. These sites induce significant C-O bond elongation from 1.432 Å to 1.459 Å (CN = 5) and 1.464 Å (CN = 4), confirming their roles in bond activation. Upon introducing various metal atoms (Au, Ag, Cu, Pd, Pt, and Ni) into W18O49 for simulations (Supplementary Fig. 1), Ni-doping was found to disrupt the surface structure of W18O49, generating W = O dangling bonds and Ni site (Fig. 1). DFT simulations reveal that the Ni site and the adjacent W = O can synergistically adsorb methanol through Ni-O bonding and hydrogen bond (HB) interaction, with an energy of −1.20 eV. The O-H bond (0.977 Å) of methanol elongates to 1.015 Å, suggesting that the Ni site can activate the O-H bond for cleavage. This finding demonstrates the potential of doped Ni as active sites for the C-O-C coupling reaction. Therefore, constructing W and Ni dual active sites on plasmonic W18O49 without compromising its surface activity and SPR intensity is a promising strategy yet a significant challenge to enhance photocatalytic methanol dehydration for DME generation.
Density functional theory (DFT) calculations were used to simulate the adsorption energies of methanol on low-coordinated W sites (CN = 4 and 5) of W18O49, as well as on the Ni site of Ni-doped W18O49. HB hydrogen bond, CN coordination number.
Herein, we engineered W and Ni dual active sites on plasmonic W18O49 through one-step Ni doping, which significantly enhances photocatalytic methanol dehydration for DME production. The localized plasmon on the surface of the catalyst generates hot electrons, which amplify the synergistic effect of W and Ni sites to facilitate C-O bond cleavage and C-O-C coupling, collectively driving the methanol-to-DME conversion pathway. As a result, the reaction activation energy (Ea) over the optimized Ni0.66-W18O49 catalyst is reduced to 65.1 kJ mol−1 under 400 mW cm-2 irradiation, much lower than 142.4 kJ mol−1 of thermocatalysis. This significant reduction in Ea improves the DME generation rate to 133.7 ± 3.3 mmol g−1 h−1. The Ea can be further reduced to 23.4 kJ mol−1 as the irradiation intensity increases to 1000 mW cm-2, enhancing the DME generation rate to 784.7 mmol g−1 h−1. The dual-active-site architecture demonstrates broad applicability, enhancing intramolecular alcohol dehydration for alkene generation by 40–80% and achieving a recorded isobutylene production rate of 3.7 mol g−1 h−1. Our work demonstrates the superiority of plasmonic semiconductor systems in boosting chemical transformation.
Results and discussion
Photocatalytic methanol dehydration over plasmonic Nix-W18O49
To effectively incorporate Ni atoms into plasmonic W18O49, Ni-doped W18O49 was synthesized via a one-step solvothermal process, utilizing W(CO)6, NiCl2, and hydrochloric acid as precursors in ethanol solution20. Various amounts of Ni were incorporated into W18O49, designated as Nix-W18O49 (where x represents the actual weight percentage of doped Ni measured by inductively coupled plasma mass spectrometry (ICP-MS), Supplementary Table 1), to investigate the relationship between doped Ni and W atoms. For comparative studies, control samples including Co-, Pd-, Pt-, Au-, Ag-, and Cu-W18O49 with varying dopant concentrations were also synthesized.
The photocatalytic performance of Nix-W18O49 was evaluated by the methanol dehydration reaction (Fig. 2a), which is a primary catalytic process for DME generation. As depicted in Fig. 2b, the optimized Ni0.66-W18O49 photocatalyst exhibits a notably high production rate of DME (133.7 ± 3.3 mmol g−1 h−1) compared to the W18O49 (13.2 ± 1.2 mmol g−1 h−1), and the maximum methanol conversion rate reaches 41.5% per hour (Supplementary Fig. 2). DME is the main product with a selectivity of 97.6-98.8%, and small amounts of H2, CH4, and CO were detected by gas chromatography-mass spectrometry (GC-MS) during photocatalytic methanol dehydration over Nix-W18O49 (Supplementary Fig. 3). The generation of H2O during photocatalysis was confirmed by the 1H nuclear magnetic resonance spectroscopy (NMR, Supplementary Fig. 4). The molar ratio between consumed methanol and generated DME is 2.03 (Supplementary Fig. 5), proving the carbon balance during the photocatalytic methanol dehydration reaction. The photocatalytic methanol dehydration stability of Ni0.66-W18O49 was evaluated by 21 consecutive cycles (Fig. 2c), with results indicating negligible decay in DME yield. The comparisons of activity and selectivity for methanol dehydration over a range of metal-doped W18O49 catalysts are illustrated in Fig. 2d and Supplementary Fig. 6. The optimal DME production rates of Co-, Pd-, Pt-, Au-, Ag-, and Cu-W18O49 are 19.6, 30.6, 126.4, 95.7, 67.7, and 43.6 mmol g−1 h−1, respectively, which are all lower than that of Ni0.66-W18O49. The actual contents of doped metals in the optimal Co-, Pd-, Pt-, Au-, Ag-, and Cu-W18O49 samples were measured by ICP-MS (Supplementary Table 2), which are 0.61 wt%, 0.73 wt%, 1.04 wt%, 0.96 wt%, 0.75 wt%, and 0.69 wt%, respectively. The disparity underscores the distinctive catalytic properties of W18O49 conferred by Ni doping. Furthermore, the Ni0.66-W18O49 catalyst displays high activity in photocatalytic dehydration of C2+ alcohols to alkenes (Supplementary Fig. 7). As shown in Figs. 2e, f, compared to W18O49, the photocatalytic alkene production rates of Ni0.66-W18O49 are significantly enhanced, with the yields of ethylene from ethanol dehydration, propylene from propanol dehydration, propylene from isopropanol dehydration, butylene from 1-butanol dehydration, isobutylene from isobutanol dehydration, butylene and 2-butylene from 2-butanol dehydration, and isobutylene from tert-butanol dehydration increasing by 82% (32.4 mmol g−1 h−1), 54% (59.1 mmol g−1 h−1), 62% (497.6 mmol g−1 h−1), 40% (290.3 mmol g−1 h−1), 71% (675.4 mmol g−1 h−1), 56% (1607.5 mmol g−1 h−1), and 71% (3694.6 mmol g−1 h−1), respectively. Notably, a record isobutylene production rate of 3.7 mol g−1 h−1 was achieved in photocatalytic tert-butanol dehydration, highlighting the pivotal role of Ni-W18O49 in enhancing alcohol dehydration reactions.
a Schematic diagram of photocatalytic methanol dehydration. b DME generation rate and product selectivity over Nix-W18O49. Error bars correspond to the standard deviation determined from three independent measurements. c 21-times recycled photocatalytic methanol dehydration reaction over Ni0.66-W18O49. d DME generation rates over different element-doped W18O49 (Pd-W18O49, Pt-W18O49, Co-W18O49, Ni-W18O49, Au-W18O49, Ag-W18O49, and Cu-W18O49). e, f Photocatalytic dehydration reactions of other higher alcohols (C2, C3, and C4) over Ni0.66-W18O49 for alkene generation. Light source: 400 mW cm-2 supplied by a 300 W Xe lamp (Perfectlight, PLS-SXE300D) equipped with an AM 1.5 G filter. The measurements in Figs. 2d, e, f were only performed once.
The influence of light intensity on the photocatalytic methanol dehydration reaction has been meticulously investigated. As shown in Fig. 3a, a direct correlation between DME production rates and light intensity is observed, with a significant increase from 25.9 mmol g−1 h−1 to 784.7 mmol g−1 h−1 as the light intensity increased from 200 to 1000 mW cm−2. Concurrently, the methanol conversion improves dramatically from 10.4% to 94.9% per hour. Supplementary Fig. 8 displays that the photothermal effect of Ni0.66-W18O49 causes a substantial increase in the surface temperature from 92 to 175 °C. In contrast, the DME production rates of the thermocatalytic reaction are 0.027 to 1.87 mmol g−1 h−1 at the temperature from 92 to 175 °C, which are far lower than those of the photocatalytic reaction. Compared to the thermocatalytic performance of various reported catalysts shown in Supplementary Table 3, the photocatalytic performance of Ni0.66-W18O49 is highly competitive in DME generation.
a Photocatalytic DME generation rates and methanol conversion over Ni0.66-W18O49 under different light intensities, and thermocatalytic DME generation rates at the corresponding temperatures. b Surface temperatures of Ni0.66-W18O49 and its photocatalytic DME generation rates under different light irradiations (UV, Vis, UV-Vis, NIR, Vis-NIR, and UV-Vis-NIR, with a fixed intensity of 400 mW cm−2). Error bars correspond to the standard deviation determined from three independent measurements. c, d Arrhenius plots and the calculated Ea for methanol dehydration under different light irradiations (c) and AM 1.5 G light irradiation with different densities (200, 400, 600, 800, and 1000 mW cm−2) (d). e, f Continuous-flow reaction system (e) and DME production rates (f) over Ni0.66-W18O49 under concentrated natural solar light irradiation (the photocatalyst loading in the continuous-flow reaction system is 50 mg). Error bars correspond to the standard deviation determined from three independent measurements.
Investigations into the photothermal effects of Ni0.66-W18O49 induced by various light sources (with a fixed intensity of 400 mW cm−2) are displayed in Fig. 3b and Supplementary Fig. 9. The surface temperatures of Ni0.66-W18O49 under UV, visible (Vis), and combined UV-Vis irradiation were measured to be 51, 61, and 80 °C, respectively. Notably, the surface temperatures of Ni0.66-W18O49 rose to 106 °C under near-infrared (NIR), and 108 °C under Vis-NIR or full-spectrum (UV-Vis-NIR) irradiation. The significantly increased surface temperature demonstrates the predominant role of SPR in the NIR region in driving the photothermal effect. Moreover, the DME generation rates of Ni0.66-W18O49 under different light irradiations are measured. Figure 3b shows that Ni0.66-W18O49 exhibits a low DME generation rate under UV (1.8 ± 0.8 mmol g−1 h−1) or Vis (3.7 ± 1.1 mmol g−1 h−1) irradiation. However, the synergistic effect of UV and Vis light irradiation effectively enhances the DME generation rate to 33.8 ± 3.2 mmol g−1 h−1. The activity enhancement is further anabatic under NIR, Vis-NIR, and UV-Vis-NIR irradiation, with DME generation rates of 103.4 ± 1.8, 99.6 ± 3.4, and 133.7 ± 3.3 mmol g−1 h−1, respectively. These findings strongly suggest that the strong SPR of Ni0.66-W18O49 in the NIR region plays a vital role in photocatalytic methanol dehydration for DME generation. For reference, the thermocatalytic reaction at 108 °C only yields a mere DME (1.3 ± 0.8 mmol g−1 h−1), which shows a stark contrast to the photocatalytic performance. These results indicate the dominant contribution of plasmonic hot carriers in the superior photocatalytic methanol dehydration activity of Ni0.66-W18O49.
The Ea of methanol dehydration for DME generation over Ni0.66-W18O49 under various temperatures and different light irradiation conditions (with a fixed intensity of 400 mW cm−2) is further explored by the Arrhenius equation k = Ae-Ea/RT. Figure 3c illustrates the DME generation rates measured under different conditions, and the resulting Arrhenius plots reveal substantial differences in Ea. Under pure thermal condition, the Ea of methanol dehydration over Ni0.66-W18O49 is determined to be 142.4 kJ mol−1. Under UV, Vis, and UV-Vis light irradiation, the Ea is reduced to 127.9, 119, and 103 kJ mol−1, respectively. Notably, NIR and Vis-NIR light irradiations further lower the Ea to 71.8 and 73.5 kJ mol−1, respectively, indicating the significant role of plasmonic hot electrons in reducing the Ea of methanol dehydration. Full-spectrum light irradiation enhances hot electron generation, leading to a greatly reduced Ea of 66.4 kJ mol−1, which is 52.3% lower than that of thermocatalysis. Furthermore, the influence of light intensity on Ea was explored by measuring the methanol dehydration reaction kinetics. As shown in Fig. 3d and Supplementary Fig. 10, the calculated Ea decreases linearly with increasing AM 1.5 G light intensity. This trend suggests that higher light intensity generates more plasmonic hot electrons, which in turn significantly lower the Ea of methanol dehydration. The lowest Ea of 23.4 kJ mol−1 is achieved under 1000 mW cm−2 irradiation. As an exothermic reaction (ΔH, −23.9 kJ mol−1), the light energy is mainly used to reduce the Ea of methanol dehydration and promote the kinetics of DME generation. Photocatalytic and thermocatalytic dehydration Ea of different alcohols were measured, as shown in Supplementary Fig. 11, further confirming the reduced Ea under light irradiation. Consequently, these findings confirm the dominant contribution of plasmonic hot electrons to the photocatalytic methanol dehydration performance of Nix-W18O49.
We designed and constructed a continuous-flow reaction system to assess the photocatalytic methanol dehydration performance of Nix-W18O49 under solar light irradiation (Fig. 3e), and the experimental setup was given in Supplementary Fig. 12. The Ni0.66-W18O49 catalyst was integrated into a microfluidic reactor with a total volume of 2.5 mL. Methanol was injected into the reactor with nitrogen gas serving as the carrier, with an optimized flow rate of 22.4 mL min−1. Solar light, concentrated by a Fresnel lens, was directly irradiated on the reaction system to facilitate the methanol dehydration reaction, and the generated DME was collected for analysis. The performance of this continuous-flow system was evaluated at Jinan University Panyu Campus, Guangzhou, China, on December 06, 07, and 09, 2024. Figure 3f illustrates that the focused solar light intensity was 1.83 ± 0.03 W cm−2 at 8:30 AM, reaching a peak of 2.28 ± 0.03 W cm−2 at noon. In response, the DME production rate increased from 23.2 ± 3.8 mmol h−1 to a maximum of 34.9 ± 2.5 mmol h−1, demonstrating the practical applicability and high efficiency of Ni0.66-W18O49 under solar light irradiation.
Structural characterizations of catalysts Nix-W18O49
Pristine W18O49 displays a sea urchin-like morphology, with preferential growth along the <010> direction21, as shown in Fig. 4a and Supplementary Fig. 13a. High-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) images in Fig. 4b reveal a uniform atomic distribution throughout the W18O49 structure. In contrast, the Nix-W18O49 catalysts exhibit no significant morphological changes as the Ni content increases from 0.15 to 1.4 wt% (Supplementary Fig. 13b–f). However, a noticeable disruption in the surface structure is observed for Ni0.66-W18O49 (Fig. 4c), suggesting that Ni doping induces surface alterations. Despite this, the HAADF-STEM image in Fig. 4d does not clearly resolve the distribution of Ni atoms, likely due to the lower atomic number of Ni (28) than W (74). Energy-dispersive X-ray spectroscopy (EDS) elemental mapping, as shown in Fig. 4e–h, confirms the homogeneous distribution of Ni atoms in the W18O49 nanowires. The crystallinity of the Nix-W18O49 catalysts was investigated using X-ray diffraction (XRD), as shown in Fig. 4i, and a shift in the [010] peak to a lower angle indicates a structural change caused by Ni doping. Raman spectra, as shown in Fig. 4j and Supplementary Fig. 14, further demonstrate the structural change, with shifts of both ν(O-W-O) and δ(O-W-O) modes to lower wavenumbers in Ni0.66-W18O49. Notably, a new band at 963.8 cm−1 is observed in Nix-W18O49 catalysts, with the enhanced intensity as Ni doping increases, which is assigned to the surface terminal W = O stretching mode22. Low-valent Ni2+ substitutes for high-valent W6+, W5+, or W4+, and oxygen vacancies will spontaneously form to maintain charge balance in Nix-W18O49. This indicates that the surface-ordered crystal structure of W18O49 is disrupted by Ni doping, as shown in 4j, leading to the loss of some lattice oxygen atoms and the creation of coordinatively unsaturated W atoms on the surface23.
a–d High resolution transmission electron microscope (HRTEM) and HAAD-STEM images of W18O49 (a, b) and Ni0.66-W18O49 (c, d). e Dark-field STEM and f–h EDS element (W, O, and Ni) mapping images of Ni0.66-W18O49. i XRD pattern, j Raman spectra (insert figure is the surface structure model), and k UV-Vis-NIR DRS of W18O49 and Nix-W18O49. l EPR spectra of Ni0.66-W18O49 before and after light irradiation. m Ratio varies of low-valent W (W5+, W4+) in W18O49, Ni0.66-W18O49, and Ni1.4-W18O49 before and after light irradiation.
The intrinsic crystal structure of W18O49 has an abundance of low-valent W ions (W4+ and W5+), leading to its high electron density. This characteristic is evident in electron paramagnetic resonance (EPR) spectra, as shown in Supplementary Fig. 15. As the increase of Ni content in Nix-W18O49 from 0.15 to 1.4 wt%, there is a slight decrease in electron density. X-ray photoelectron spectroscopy (XPS) was employed to study the chemical states of W atoms in Nix-W18O49, and the contents of W5+ and W4+ in Nix-W18O49 are found to be similar to that in pristine W18O49, as detailed in Supplementary Figs. 16, 17. The doped Ni in Nix-W18O49 is detected as Ni2+ ion in their Ni 2p XPS spectra (Supplementary Fig. 18). Although the SPR intensity of Nix-W18O49 experiences a slight reduction in the UV-Vis-NIR diffuse reflectance spectroscopy (DRS), the overall SPR characteristics remain robust (Fig. 4k, Supplementary Fig. 19). The broad SPR band of W18O49 nanowires is due to the coupling of nanowires, demonstrated by finite-difference time-domain (FDTD) simulations (Supplementary Fig. 20). EPR spectra (Fig. 4l and Supplementary Fig. 21) show that Ni0.66-W18O49 has a significantly increased electron density after light irradiation, suggesting that an appropriate amount of Ni-doping facilitates photoelectron accumulation on Ni0.66-W18O4924. The influence of light irradiation on the chemical states of Ni and W elements in Nix-W18O49 was analyzed by XPS spectra, and the results in Supplementary Fig. 22 show that no significant change is found in the Ni 2p XPS spectra of Ni0.66-W18O49 after light irradiation, indicating that electron accumulation does not occur on Ni atoms. Notably, the variations in W5+ and W4+ contents are clearly depicted in Fig. 4m and Supplementary Fig. 23 (the W 4 f XPS spectra). The W5+ content in W18O49, Ni0.66-W18O49, and Ni1.4-W18O49 similarly increases from 8.0% to 15.1% after light irradiation, while Ni0.66-W18O49 exhibits the highest W4+ content of 14.1%. This result demonstrates that doping of 0.66 wt% Ni in W18O49 facilitates photoelectron trapping by high-valent W6+ or W5+ and generates more low-valent W4+ sites, thereby enhancing electron density for strong SPR. The SPR enhancement under light irradiation is more pronounced for Ni0.66-W18O49 compared to pristine W18O49, as evidenced by their enhanced UV-Vis-NIR DRS, as shown in Supplementary Fig. 24. The surface low-valent W atoms can strongly adsorb alcohols, which is crucial for photocatalytic alcohol dehydration25. Additionally, the stability of the structure and SPR of Ni0.66-W18O49 after photocatalysis was confirmed by XRD patterns (Supplementary Fig. 25) and UV-Vis-NIR DRS analysis (Supplementary Fig. 26).
To further elucidate the electronic state and the coordination environment of Ni and W atoms in Nix-W18O49, we have employed X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) spectroscopies. Figure 5a displays the normalized Ni K-edge XANES spectra for Ni0.66-W18O49 and Ni1.4-W18O49, with NiO and Ni foil as references. The energy absorption edges of doped Ni in Ni0.66-W18O49 and Ni1.4-W18O49 closely resemble that of NiO, indicating its oxidation state is close to Ni2+20,26. As depicted in Fig. 5b, the Fourier-transformed EXAFS (FT-EXAFS) spectra reveal distinct features for the references and the Ni- W18O49 samples. The Ni foil exhibits a predominant peak at 2.16 Å, corresponding to the Ni-Ni bond. The NiO reference displays two peaks, a strong peak at 2.54 Å is attributed to the Ni-Ni bond, and the weak peak at 1.64 Å is assigned to the Ni-O bond27,28. Comparatively, Ni0.66-W18O49 only shows a strong peak at 1.52 Å, attributed to the Ni-O bond, which suggests that Ni is atomically dispersed within Ni0.66-W18O49. However, Ni1.4-W18O49 exhibits two peaks of similar intensity at 1.52 and 2.62 Å, corresponding to the Ni-O bond of single Ni atom doping and the Ni-Ni bond characteristic of NiO28,29, respectively, which implies that Ni exists in two distinct coordination environments in Ni1.4-W18O49. Quantitative structural parameters derived from the Ni K-edge EXAFS fitting curves of Ni0.66-W18O49, Ni1.4-W18O49, NiO, and Ni foils are illustrated in Fig. 5c, d, and Supplementary Fig. 27, and the detailed results are tabulated in Supplementary Table 4. The CN of the Ni-O bond in Ni0.66-W18O49 is determined to be 6.37. For Ni1.4-W18O49, the CN of Ni-O and Ni-Ni bonds is 6.3 and 6.2, respectively. The reduced CN of the Ni-Ni bond in Ni1.4-W18O49, compared to the CN (12) of the Ni-Ni bond in NiO, suggests that Ni atoms in Ni1.4-W18O49 are closely spaced. Wavelet transforms (WT) of the EXAFS spectra, shown in Fig. 5e–h, were conducted to further elucidate the localized structure in both the K and R spaces. The WT intensity maximum for Ni0.66-W18O49 at 5.8 Å−1 in the K space and 1.52 Å in the R space corresponds to the Ni-O bond, which is distinct from the Ni-Ni bond observed in NiO and Ni foil references. For Ni1.4-W18O49, an additional WT intensity maximum is observed at 6.8 Å−1 in the K space and 2.62 Å in the R space, corresponding to the Ni-Ni bond.
a Normalized Ni K-edge XANES and b FT-EXAFS spectra of Ni0.66-W18O49, Ni1.4-W18O49, NiO, and Ni foil references. c, d The Ni K-edge EXAFS fitting curves for Ni0.66-W18O49 (c) and Ni1.4-W18O49 (d). e–h WT-EXAFS of Ni K-edge for Ni0.66-W18O49, Ni1.4-W18O49, NiO, and Ni foil references. i Normalized W L3-edge XANES and j FT-EXAFS spectra of W18O49, Ni0.66-W18O49, WO3, and Ni foil references. k The W L3-edge EXAFS fitting curves for Ni0.66-W18O49.
The normalized W L3-edge XANES spectra for W18O49 and Ni0.66-W18O49, with WO3 and W foil as references30, are shown in Fig. 5i. The white lines of W in W18O49 and Ni0.66-W18O49 nearly overlap, indicating that Ni doping does not alter the valence of W in W18O49. The FT-EXAFS spectra, as depicted in Fig. 5j, reveal a strong peak at 1.28 Å and a weak peak at 1.84 Å assigned to two different W-O bonds (W-O1 and W-O2) in the W18O49 crystal18,31. Quantitative structural parameters derived from the W L3-edge EXAFS fitting curves for W18O49 and Ni0.66-W18O49 are illustrated in Fig. 5k and Supplementary Fig. 28, and the detailed results are tabulated in Supplementary Table 5. The CN of W-O2 in Ni0.66-W18O49 is slightly lower than that of W-O2 in W18O49, and a lower CN of W is favorable to photocatalysis23,32.
Photocatalytic methanol dehydration mechanism study on Nix-W18O49
The photocatalytic mechanism of methanol dehydration on Nix-W18O49 has been elucidated by in-situ XPS measurement, as shown in Fig. 6a. After the addition of methanol, a distinct peak at 286.5 eV, corresponding to the C-O bond, emerges in the C 1 s XPS spectrum of Ni0.66-W18O4933. The C-O bond peak is more pronounced than that of pristine W18O49 (Supplementary Fig. 29), highlighting the enhanced methanol adsorption capacity of Ni0.66-W18O49. Upon light irradiation, the C-O bond peak diminishes, indicating the C-O bond cleavage of adsorbed methanol for the dehydration reaction, and this process takes place more slowly on W18O49 (Supplementary Fig. 29). Further insights come from the in-situ W 4 f XPS spectra, where the adsorbed methanol induces a shift in the W 4 f XPS peak to lower binding energy due to the electron-donating nature of methanol. The shift is more pronounced in Ni0.66-W18O49, confirming its superior methanol adsorption. Concurrently, the W5+ content decreases, suggesting that methanol adsorption occurs on surface low-coordinated W sites. Post-irradiation, the W5+ content returns to its initial level within 10 min, while the W4+ content increases from 9.1% to 14.7%, and further to 18.7% after 20 min of irradiation. The increase in W4+ content is attributed to photoelectron accumulation on Ni0.66-W18O49 in the methanol atmosphere. As light irradiation prolongs, the W 4 f XPS peak shifts back to high binding energy, revealing the consumption of surface-adsorbed methanol for DME generation (Fig. 6a and Supplementary Fig. 30). The adsorption and dehydration of methanol are also evident by the in-situ O 1 s XPS spectra (Supplementary Fig. 31).
a In-situ C 1 s and W 4 f XPS spectra of Ni0.66-W18O49 during photocatalytic methanol dehydration reaction. b, c In-situ FT-IR spectra of Ni0.66-W18O49 and W18O49 during photocatalytic methanol dehydration reaction under full-spectrum light irradiation. d Methanol-TPD pattern of the W18O49 and Ni0.66-W18O49 catalysts. e Photocatalytic mechanism of methanol dehydration reaction over Nix-W18O49.
The photocatalytic methanol dehydration processes over W18O49 and Ni0.66-W18O49 were further monitored by in-situ Fourier transform infrared (FT-IR) spectroscopy, as depicted in Fig. 6b. After the addition of methanol, new strong bands around 1010 ~ 1060, 1362 ~ 1462, and 2824 ~ 2984 cm−1, assigned to νC-O, δCH3, and νCH334,35, respectively, demonstrate the strong methanol adsorption on Ni0.66-W18O49. Under full-spectrum light irradiation, a new band around 1161.9 cm−1, corresponding to νC-O-C36,37, emerges and strengthens with prolonged light irradiation, verifying the methanol dehydration to the DME process. Furthermore, the νC-O band intensities for both Ni0.66-W18O49 and W18O49 increase with prolonged UV-Vis-NIR irradiation, which is attributed to the light-induced more low-valent W4+ and W5+ (Fig. 4m) for enhanced methanol adsorption. The signal of *OH intermediate was enhanced under full-spectrum light irradiation, and then became weak due to its removal from the catalyst surface as H2O. However, the νC-O-C band is weaker in the in-situ FT-IR spectra of W18O49 (Fig. 6c), confirming a higher activity of Ni0.66-W18O49 for photocatalytic methanol dehydration. When UV light is irradiated on Ni0.66-W18O49, no νC-O-C band is observed, but as the light switches to NIR, the νC-O-C band appears and strengthens with prolonged irradiation time (Supplementary Fig. 32), indicating that NIR-excited plasmonic hot electrons boost DME generation. These results demonstrate that the Ni doping in W18O49 enhances methanol adsorption, thereby boosting the plasmonic dehydration reaction for DME generation. The reaction pathway of C2+ alcohol (tert-butanol) dehydration over W18O49 and Ni0.66-W18O49 was monitored by the in-situ FT-IR spectroscopy, as shown in Supplementary Fig. 33. UV-irradiation can promote more tert-butanol adsorption on Ni0.66-W18O49, and NIR-irradiation promotes C-O bond cleavage for C = C bond generation. Temperature-programmed desorption (TPD) of methanol on W18O49 and Ni0.66-W18O49 are presented in Fig. 6d. The desorption peak observed at 112 °C and 188 °C corresponds to the physical adsorption and chemical adsorption of methanol, respectively. Notably, in comparison to W18O49, the desorption peak at 188 °C on the Ni0.66-W18O49 sample is significantly enhanced, indicating that the incorporation of Ni sites effectively improves the chemical adsorption capacity for methanol molecules38. While excessive Ni doping reduces the adsorption amount of methanol, restraining the dehydration reaction for DME generation (Supplementary Fig. 34).
Based on the results and discussions, a plausible mechanism for photocatalytic methanol dehydration over Ni-W18O49 is proposed, as illustrated in Fig. 6e. Ni doping in W18O49 induces surface reconstruction, creating W = O dangling bonds. DFT calculations (Fig. 1) have demonstrated that two distinct surface sites existed on Ni-W18O49 for methanol adsorption: low-coordinated W and Ni sites. The surface low-coordinated W atom can directly bond to the oxygen of methanol, forming *methanol(1), while the Ni and adjacent W = O have a synergy on adsorbing methanol by Ni-O bonding and a hydrogen bond interaction, resulting in *methanol(2). According to DFT simulations (Supplementary Figs. 35, 36), the O-H bond of *methanol(2) is activated for cleavage to form W-O-H and *CH3O as intermediates39. The excitations on Ni0.66-W18O49 under UV, Vis-NIR, and UV-Vis-NIR light irradiation were illustrated in Supplementary Fig. 37. Most photoelectrons excited by UV irradiation became trapped at low energy states within conduction band, thereby increasing the electron density. Upon Vis-NIR light irradiation, electrons on low-valent W atoms undergo strong oscillations due to SPR, creating an intense plasmonic field on the surface of W18O49 (Supplementary Fig. 38)18. SPR also excites low-energy electrons to high-energy hot electrons. Both *methanol(1) and *CH3O are located in the plasmonic field, and the plasmonic hot electrons can be transferred to *methanol(1) through the W-O bond, activating and cleaving the C-O bond in methanol for *CH3 generation and leaving OH adsorbed on W sites as W-O-H. However, *CH3O connected to Ni prevents hot electron transfer and protects its C-O bond. During transition state (TS), the cleavage of the C-O bond is the rate-determining step for the methanol dehydration reaction. The generated *CH3 from *methanol(1) attacks the oxygen atom of *CH3O to achieve C-O-C coupling for adsorbed DME (*DME) formation. Ultimately, a dehydration reaction between two W-O-H produces one H2O, and the catalyst returns to its initial state after releasing adsorbed H2O (*H2O). Meanwhile, a small part of doped Ni can produce a small amount of hydrogen, and the generated *CH3 can react with *H for CH4 byproduct generation as demonstrated in Fig. 2b. However, the thermal-activation for C-O cleavage and C-O-C coupling is hard due to the large activation energy (142.4 kJ mol−1) for thermocatalytic methanol dehydration. For W18O49, most methanol is adsorbed on surface low-coordinated W sites as *methanol(1). During TS, plasmonic hot electron transfer can break the C-O bond of *methanol(1), but the generated *CH3 is less likely to react with the oxygen atom of another *methanol(1), slowing the DME generation rate. Although DFT calculations still exhibit some discrepancies from actual experiences, they further demonstrate that Ni-doping in W18O49 reduces Ea of methanol dehydration as shown in Supplementary Figs. 35, boosting DME generation. Therefore, the synergy of W and Ni sites enhances the plasmonic photocatalytic methanol dehydration to DME. This photocatalytic mechanism underscores the synergistic interaction between distinct surface sites and the amplification effect of the semiconductor-plasmon on Nix-W18O49, thereby effectively facilitating the methanol dehydration process. While for C2+ alcohol dehydration, Ni0.66-W18O49 with more light-induced surface low-valent W4+ and W5+ can facilitate C2+ alcohol adsorption, meanwhile, its hot electrons promote C-O bond cleavage for alkene generation.
In summary, we present a one-step synthesis strategy for engineering dual active sites on plasmonic W18O49 through Ni doping, which remarkably enhances the photocatalytic methanol dehydration reaction for DME production. The distinct roles of W and Ni sites in the catalytic process have been elucidated: low-coordinated W sites facilitate C-O bond cleavage promoted by SPR-excited hot electrons and Ni sites enable C-O-C coupling for DME generation. The optimized Ni0.66-W18O49 catalyst dramatically reduces Ea to 65.1 kJ mol−1 under 400 mW cm−2 illumination, much lower than 142.4 kJ mol−1 required for thermocatalysis. This significant Ea reduction enables a good DME production rate of 133.7 ± 3.3 mmol g−1 h−1. Further increasing light intensity to 1000 mW cm−2 lowers Ea to 23.4 kJ mol−1 while boosting the DME generation rate to 784.7 mmol g−1 h−1. This Ni-doped W18O49 catalyst also demonstrates broad applicability in various alcohol dehydration for alkene generation, achieving a recorded photocatalytic isobutylene production rate of 3.7 mol g−1 h−1. This work showcases the exceptional potential of rationally designed plasmonic semiconductor systems in driving photochemical transformations, establishing a sustainable and energy-efficient paradigm for DME production.
Methods
Materials
Tungsten hexacarbonyl (W(CO)6, >97%), methanol (99.8%), anhydrous ethanol (99.8%), propanol (99.8%), isopropanol (99.8%), 1-butanol (99.8%), isobutanol (99.8%), 2-butanol (99.8%), tert-butanol (99.8%), NiCl2.6H2O (>98%), HAuCl4.3H2O (99.9%), H2PtCl6.6H2O (99.9%), CuCl2.2H2O (>98%), PdCl2 (99.9%), CoCl2.6H2O (99%), AgNO3 (99.9%), and hydrochloric acid (HCl, AR, 36% ~ 38%) were purchased from Aladdin and used without further purification. Deionized water (18.2 MΩ) was made by Arium Mini Plus ultrapure water system and used in all experiments.
Synthesis of catalysts
Nix-W18O49 samples were prepared by using a solvothermal method20. In a typical procedure, 150 mg of W(CO)6 was dissolved in 28.75 mL of ethanol, forming a bright yellow solution. Subsequently, different volumes (0.196, 0.392, 0.588, 0.784, and 0.980 mL) of a NiCl2·6H2O solution (10 mg mL−1) and 1.25 mL of concentrated HCl (36%-38%) were added into above solution under vigorous stirring. The resulting mixture was transferred to a 50 mL Teflon-lined stainless-steel autoclave and heated at 180 °C for 12 h. After cooling to room temperature, Nix-W18O49 catalysts were collected after centrifugation, washed thoroughly with ethanol, and dried in a vacuum oven. The other metal-doped W18O49 samples, including Au-W18O49, Ag-W18O49, Cu-W18O49, Pt-W18O49, Pd-W18O49, and Co-W18O49 were synthesized following above process using HAuCl4, AgNO3, CuCl2, H2PtCl6, PdCl2, and CoCl2 precursor solutions, respectively. The pristine W₁₈O₄₉ catalyst was prepared as a reference through the above synthetic process without adding the doped metal salts.
Photocatalytic methanol dehydration reaction tests
A 5 mg catalyst was plastered on a cover glass (9.6 cm2) and placed at the bottom of a 180 mL reaction chamber. The chamber was sealed with a thick quartz cover glass and purged with pure nitrogen for 20 min to remove oxygen. Illumination was supplied by a 300 W Xe lamp (Perfectlight, PLS-SXE300D) equipped with an AM 1.5 G filter, delivering a light intensity of 400 mW cm−2. The light intensity can be adjusted by modulating the working current of light source. 0.1 mL of methanol was injected into the chamber for reaction. Gaseous products were analyzed using gas chromatography-mass spectrometry (GC-MS, Panna Instruments A91Plus + AMD9P) equipped with a Plot-Q column. Photocatalytic reaction tests under different light irradiation were measured following the above process, employing different light-cutoff filters for NIR ( > 800 nm), Vis (420–780 nm), UV (200-400 nm), Vis-NIR ( > 420 nm), and UV-Vis (<800 nm) light irradiation. The reaction temperature was controlled by a thermal oil bath.
The flow reaction of photocatalytic methanol dehydration reaction was performed in heart-shaped micropipes engraved on a quartz plate, with a diameter of 1 mm and a total volume of 2.5 mL. 50 mg catalyst was coated on the inner walls of the micropipes, which were sealed with another quartz plate as a reaction chamber. Methanol was injected into the chamber with a flow rate of 50 μL min−1 through one inlet, while nitrogen was introduced through a second inlet at a flow rate of 22.4 mL min−1 to facilitate methanol flow along the micropipes. Sunlight was focused onto the quartz window of the chamber using a Fresnel lens, covering a total irradiation area of 16 cm2. The gas products were collected for detection by GC-MS.
The reaction activation energy calculation
The reaction activation energy of methanol dehydration is calculated by the Arrhenius equation as follows40:
where \({k}\) is the reaction rate constant, \({A}\) is the Arrhenius constant, \({E}_{a}\) is the activation energy, \({R}\) is the molar gas constant, and \(T\) is the surface temperature of the catalysts. The surface temperature of the catalysts was detected by a infrared thermal camera (Fluke Tis20 + ).
DFT calculations
All computational simulations were performed using the DS-PAW software based on density functional theory (DFT)41. The interaction between the valence electrons and the ionic cores was described using projector-augmented wave (PAW) pseudopotentials. During the structural optimization, the Perdew-Burke-Ernzerhof (PBE) functional, within the generalized gradient approximation (GGA), was applied. For the W18O49 (100) surface model (Supplementary Data 1), a 1 × 2 supercell containing 134 atoms was chosen. A vacuum space of approximately 15 Å was introduced above the slab to avoid interactions between periodic images. In simulations involving Ni doping, a single tungsten (W) atom on the surface was replaced by a nickel (Ni) atom within the same supercell (Supplementary Data 1). Only the surface atoms were allowed to relax during the optimization. The computational setup included a cutoff energy (Ecut) of 500 eV, a total energy convergence criterion of 1 × 10-4 eV, and a residual stress tolerance of 0.05 eV/Å. Additionally, a 2 × 1 × 1 Monkhorst-Pack k-point mesh was used.
The formation energy of CH3OH adsorption was calculated using the following expression:
where X represents either W18O49 (100) or Ni-W18O49 (100), and E(CH3OH-X), E(CH3OH), and E(X) denote the total energies of CH3OH adsorbed on X, CH3OH alone, and pristine X, respectively.
The Gibbs free energy (ΔG) was calculated using the thermodynamic expression:
where ΔEZPE denotes the zero-point energy correction and ΔS represents the entropy change of intermediate species.
Transition states were located using the climbing image nudged elastic band (CI-NEB) method42. The calculations employed 8 intermediate images between initial and final states, with a spring constant of 5.0 eV/Å2 and a force convergence threshold of 0.1 eV/Å.
In-situ FT×IR transmission measurement
In-situ FT-IR measurements were performed using a Thermo Scientific Nicolet iS50 FT-IR spectrometer equipped with a liquid-nitrogen-cooled HgCdTe detector. Spectra were collected with a resolution of 4 cm−1 by averaging 32 scans per measurement. The sample was placed in a custom-designed IR reaction chamber suitable for analyzing highly scattered powder samples in diffuse reflection mode. The chamber was sealed with ZnSe windows to ensure controlled experimental conditions. Before measurement, the sample was degassed at 150 °C for 1 h and subsequently purged with ultrapure nitrogen for 1 h. A mixed gas containing 0.1 mL of methanol was introduced into the chamber, allowing the sample to reach adsorption equilibrium over 30 min before irradiation. The light source used was provided by a xenon lamp coupled through an optical fiber and the light intensity reaching the sample surface was approximately 80 mW cm−2. The FT-IR spectra were recorded at 5-min intervals to monitor dynamic changes during the reaction43.
Catalysts characterizations
X-ray absorption spectroscopy (XAS) was carried out at the XAS Beamline located within the Australian Synchrotron (ANSTO) in Melbourne, Australia. A collection of Si (111) monochromator crystals, which were maintained at low temperatures using liquid nitrogen, was employed for these measurements. The energy of the electron beam utilized was 3.0 GeV. To reduce the harmonic components of the X-ray beam, optics featuring a silicon-coated collimating mirror and a rhodium-coated focusing mirror were integrated into the beamline. Data collection was performed in transmission mode with W foil used for energy calibration when measuring the W L3 edge, and in fluorescence mode with Ni foil used for energy calibration when measuring the Ni K edge. The size of the beam measured approximately 1 × 1 mm2. The XRD patterns were acquired using a Rigaku Rint-2500 diffractometer with Cu Kα radiation, scanning at a speed of 0.1° s−1. The morphologies of catalysts were examined using TEM (JEOL 2100) and HRTEM (JEM-3000F). HAADF-STEM images and elemental mapping were performed using a JEOL JEM-2100 F transmission electron microscope. The instrument was equipped with double spherical aberration (Cs) correctors for both the probe-forming and image-forming objective lenses, along with a Super-X EDS detector system. In-situ XPS spectra were obtained using an ESCALAB 250Xi+ analyzer, with Al Kα as the excitation source, and the sample was adsorbed with methanol, dried, and then placed into the reaction chamber, followed by vacuum pumping before testing. The sample was exposed to irradiation using a CEAULIGHT CEL-HXF300 light source, and XPS spectra were collected at 10-min intervals to monitor changes induced by light exposure. The constant analyzer energy mode was used for the acquisition of high-energy resolution spectra with a pass energy of 30 eV and an energy step size of 0.1 eV. UV-Vis-NIR DRS were collected by a UV-Vis-NIR spectrophotometer (JASO V-570). To study the influence of light irradiation on DRS, the powdered samples were firstly irradiated by xenon lamp for different times, and then were moved to UV-Vis-NIR spectrophotometer for DRS measurement. EPR spectra were detected by a Bruker A300 spectrometer. The specific doping metal content was determined by ICP-MS (Agilent 7850 ICP-MS). Raman spectra were collected by Raman microspectroscopy (HORIBA XploRA PLUS). The methanol-TPD patterns were obtained using a chemisorption analyzer (BelCata II, MicrotracBEL, Japan).
Data availability
Source data are provided in this paper.
References
Semelsberger, T. A. et al. Dimethyl ether (DME) as an alternative fuel. J. Power Sources 156, 497–511 (2006).
Park, S. H. et al. Applicability of dimethyl ether (DME) in a compression ignition engine as an alternative fuel. Energ. Convers. Manag. 86, 848–863 (2014).
Gogate, M. R. The direct dimethyl ether (DME) synthesis process from syngas I. Process feasibility and chemical synergy in one-step LPDMEtm process. Nat. Chem. Petrol. Sci. Technol. 36, 547–554 (2018).
Dimethyle Ether (DME) Market Size, Trends, Global Report-2037, Research Nester, Report ID: 3501, (2024).
Peinado, C. et al. Review and perspective: Next generation DME synthesis technologies for the energy transition. Chem. Eng. J. 479, 147494 (2024).
Chmielarz, L. Dehydration of methanol to dimethyl ether—current state and perspectives. Catalysts 14, 308 (2024).
Azizi, Z. et al. Dimethyl ether: a review of technologies and production challenges. Chem. Eng. Process. 82, 150–172 (2014).
Tang, Y. Q. et al. Enhanced photothermal dehydration of methanol over W18O49/Au/SAPO-34 catalysts with broadened light absorption. Rare Met. 43, 1139–1152 (2024).
Tao, L. et al. Efficient methanol-to-olefins conversion via photothermal effect over TiN/SAPO-34 catalyst. Catal. Lett. 152, 1651–1659 (2022).
Zhang, X. L. et al. Efficient photothermal alcohol dehydration over a plasmonic W18O49 nanostructure under visible-to-near-infrared irradiation. J. Photoch. Photobio. A 441, 114728 (2023).
Aslam, U. et al. Catalytic conversion of solar to chemical energy on plasmonic metal nanostructures. Nat. Catal. 1, 656–665 (2018).
Baffou, G. et al. Nanoplasmonics for chemistry. Chem. Soc. Rev. 43, 3898–3907 (2014).
Xin, Y. et al. Copper-based plasmonic catalysis: recent advances and future perspectives. Adv. Mater. 33, 2008145 (2021).
Zhou, L. N. et al. Quantifying hot carrier and thermal contributions in plasmonic photocatalysis. Science 362, 69–72 (2018).
Yuan, Y. G. et al. Steam methane reforming using a regenerable antenna–reactor plasmonic photocatalyst. Nat. Catal. 7, 1339–1349 (2024).
Li, J. et al. Solar-driven plasmonic tungsten oxides as catalyst enhancing ethanol dehydration for highly selective ethylene production. Appl. Catal. B-Environ. 264, 118517 (2020).
Lu, C. H. et al. Full-spectrum nonmetallic plasmonic carriers for efficient isopropanol dehydration. Nat. Commun. 13, 6984 (2022).
Tian, D. H. et al. Constructing high-active surface of plasmonic tungsten oxide for photocatalytic alcohol dehydration. Adv. Mater. 36, 2404738 (2024).
Yang, D. et al. Mechanism of methanol dehydration catalyzed by Al8O12 nodes assisted by linker amine groups of the metal-organic framework CAU-1. ACS Catal. 12, 12845–12859 (2022).
Mao, Y. Y. et al. Electronic structure manipulation via site-selective atomically dispersed Ni for efficient photocatalytic CO2 reduction. ACS Catal. 13, 8362–8371 (2023).
Lou, Z. Z. et al. Surfactant-free synthesis of plasmonic tungsten oxide nanowires with visible-light-enhanced hydrogen generation from ammonia borane. Chem. –Asian J. 10, 1291–1294 (2015).
Santato, C. et al. Crystallographically oriented mesoporous WO3 films: synthesis, characterization, and applications. J. Am. Chem. Soc. 123, 10639–10649 (2001).
Shi, Y. Z. et al. Photocatalytic toluene oxidation with nickel-mediated cascaded active units over Ni/Bi2WO6 monolayers. Nat. Commun. 15, 4641 (2024).
Wang, Y. L. et al. Photocatalytic ammonia synthesis from nitrate reduction on nickel single-atom decorated on defective tungsten oxide. Appl. Catal. B-Environ. Energy 341, 123266 (2024).
Duan, M. et al. Synergizing inter and intraband transitions in defective tungsten oxide for efficient photocatalytic alcohol dehydration to alkenes. JACS Au 2, 1160–1168 (2022).
Li, Y. G. et al. High-valence nickel single-atom catalysts coordinated to oxygen sites for extraordinarily activating oxygen evolution reaction. Adv. Sci. 7, 1903089 (2020).
Akri, M. et al. Atomically dispersed nickel as coke-resistant active sites for methane dry reforming. Nat. Commun. 10, 5181 (2019).
Rao, Z. Q. et al. Light-reinforced key intermediate for anticoking to boost highly durable methane dry reforming over single atom Ni active sites on CeO2. J. Am. Chem. Soc. 145, 24625–24635 (2023).
Yan, C. C. et al. Coordinatively unsaturated nickel–nitrogen sites towards selective and high-rate CO2 electroreduction. Energy Environ. Sci. 11, 1204–1210 (2018).
Gu, Y. et al. Boosting selective nitrogen reduction via geometric coordination engineering on single-tungsten-atom catalysts. Adv. Mater. 33, 2100429 (2021).
Zhang, K. X. et al. Regulation of the work function difference promotes in situ phase transition of WO3-x for efficient formate electrooxidation. ACS Appl. Mater. Interfaces 15, 15024–15035 (2023).
Zhou, J. Y. et al. Tailoring the coordination microenvironment of single-atom W for efficient photocatalytic CO2 reduction. Appl. Catal. B-Environ. Energy 350, 123911 (2024).
Zhang, G. et al. Selective CO2 electroreduction to methanol via enhanced oxygen bonding. Nat. Commun. 13, 7768 (2022).
Herrera, C. et al. Dimethyl ether synthesis via methanol dehydration over Ta-supported catalysts. Appl. Catal. A-Gen. 582, 117088 (2019).
Ozensoy, E. et al. NOx reduction on a transition metal-free γ-Al2O3 catalyst using dimethylether (DME). Catal. Today 136, 46–54 (2008).
Zhou, Z. Y. et al. Electrooxidation of dimethoxymethane on a platinum electrode in acidic solutions studied by in situ FTIR spectroscopy. J. Phys. Chem. C. 112, 19012–19017 (2008).
Mclnroy, A. R. et al. An infrared and inelastic neutron scattering spectroscopic investigation on the interaction of η-alumina and methanol. Phys. Chem. Chem. Phys. 7, 3093–3101 (2005).
Shi, Q. Q. et al. Experimental and mechanistic understanding of photo-oxidation of methanol catalyzed by CuO/TiO2-spindle nanocomposite: oxygen vacancy engineering. Nano Res 13, 939–946 (2020).
Xiong, F. et al. Methanol conversion into dimethyl ether on the anatase TiO2(001) Surface. Angew. Chem. Int. Ed. 55, 623–628 (2016).
Zhou, L. et al. Quantifying hot carrier and thermal contributions in plasmonic photocatalysis. Science 362, 69–72 (2018).
Blochl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Henkelman, G. A climbing image nudged elastic band method for finding saddle points and minimum energy paths. J. Chem. Phys. 22, 9901–9904 (2000).
Chung, S. H. et al. Origin of active sites on silica-magnesia catalysts and control of reactive environment in the one-step ethanol-to-butadiene process. Nat. Catal. 6, 363–376 (2023).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 22175076 for Z.L., 22222202 for Z. Z., U23A20136 for Z.W. and Z.L., 11904133 for J.L., 22102087 for X.L.), Science and Technology Plan Project of Guangzhou (No. 2023A03J0128), Guangdong S&T Program (2023B1212010008), the Fundamental Research Funds for the Central Universities and Outstanding Innovative Talents Cultivation Funded Programs for Doctoral Students of Jinan University.
Author information
Authors and Affiliations
Contributions
D.T. and Y.L. performed the experiments. X.L., Z.Z., and Z.L. supervised the project and wrote the manuscript. L.M., X.C., Y.C. and X.W. carried out the DFT calculations. J.L., Z.W. C.X., and B.L. analyzed experimental data. All authors discussed the results together and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests
Peer review
Peer review information
Nature Communications thanks Zhao-Qing Liu, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.”
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tian, D., Liang, Y., Zheng, Z. et al. Plasmonic Ni-doped W18O49 with dual active sites drives efficient methanol dehydration to dimethyl ether. Nat Commun 16, 10062 (2025). https://doi.org/10.1038/s41467-025-65040-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65040-3