Abstract
Cycloaddition reaction offers an ideal and powerful method for the construction of cyclic structures with molecular complexity and diversity. The exploration of versatile synthons, especially in situ formed species, for the assembly of poly-functionalized heterocycles in one-pot is an intriguing and challenging area in synthetic chemistry from the perspective of atom economy and bond formation efficiency. Herein, we report an asymmetric formal [1 + 2 + 2]-cycloaddition of readily accessible diazoamides with enamines and carbonyl compounds. This cascade reaction involves a 1,3-dipolar cycloaddition of ketones/aldehydes with the key zwitterionic intermediate that generated in situ through an intermolecular metal carbene transfer to nucleophilic enamines, which offers a direct access to spiro-tetrahydrofurans containing four consecutive stereocenters in high yields and excellent enantioselectivity through chiral copper complex or chiral dirhodium carboxylates catalysis.
Similar content being viewed by others
Introduction
Cycloaddition reactions represent ideal protocols for the rapid assembly of cyclic and polycyclic architectures with molecular complexity and structural diversity1,2,3. In this vein, the asymmetric [3 + 2]-cycloaddition holds significant interest for the formation of five-membered carbocyclic and heterocyclic ring systems, which are of indispensable value in the synthetic and medical chemistry communities4,5,6,7. In particular, these reactions have evolved as a viable protocol for assembling spiro skeletons containing all carbon quaternary stereocenters8,9,10,11,12, but they pose significant challenges in synthetic chemistry and asymmetric catalysis. Efficient methods and versatile catalytic protocols for these structural motifs are therefore highly desirable13,14,15,16.
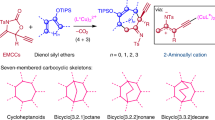
In established strategies for the [3 + 2]-cycloadditions, using pre-synthesized and bench stable 1,3-dipoles is generally inevitable, which has become a major drawback that significantly restrict the utility for the diversity-oriented-synthesis (Fig. 1a, path a)17,18. However, the application of in situ formed 1,3-dipoles in enantioselective cycloaddition19,20,21,22,23,24,25,26,27, especially those formed through an intermolecular addition process28,29,30,31, has received limited attention (Fig. 1a, path b), probably due to their relatively low stability and high reactivity, which are the main obstacles for the asymmetric induction. Whereas, this formal [1 + 2 + 2]-cycloaddition reaction features obvious advantages in comparison to the conventional [3 + 2] versions, such as high bond formation efficiency (enabling the formation of at least three σ-bonds in one reaction) and exponential enhancement of structural diversity and flexibility with readily available reagents. For example, convergent synthesis of poly-substituted tetrahydrofurans is possible by a 1,3-dipolar cycloaddition of alkenes with carbonyl ylides, which are generated in situ by carbene transfer to a carbonyl compound (Fig. 1b)32,33.
a General protocols for the catalytic [3 + 2]- and [1 + 2 + 2]-cycloadditions. b Catalytic [3 + 2]-cycloaddition of in situ formed carbonyl ylide with alkene. c Interception transformations of in situ formed zwitterionic intermediate & our hypothesis. d This work: formal [1 + 2 + 2]-cycloaddition via interception of zwitterionic intermediate with carbonyl compound.
Unlike structurally analogous carbonyl ylide species, the utilization of an in situ formed zwitterionic intermediate as a 1,3-dipole in cycloaddition is rare34,35,36,37,38. During the course of our studies in catalytic asymmetric metal carbene gem-difunctionalization39,40,41,42, we disclosed a carbene gem-dialkylation reaction through strategic interception of a zwitterionic intermediate with electrophiles for the expeditous construction of all-carbon quaternary stereocenters (Fig. 1c, path c)43,44. The competitive formal Csp2-H insertion reaction45,46, cyclopropanation47,48, or nucleophilic addition reactions (Fig. 1c, path d)49 could be suppressed when methylene iminium ion, which derived in situ from 1,3,5-triazinane in the presence of chiral phosphoric acid (CPA), is employed as the trapping agent. The versatile application of this interception protocol set the stage for the invention of multi-component reactions, and promoted us to explore these transient zwitterionic intermediates in [3 + 2]-cycloaddition with dipolarophiles (Fig. 1c, path e).
Described herein is an asymmetric formal [1 + 2 + 2]-cycloaddition of diazoamides with enamines and carbonyl compounds, which involves an untapped synthetic use of the key in situ formed zwitterionic intermediate as a C3 synthon in 1,3-dipolar cycloaddition with carbonyl compounds (Fig. 1d). In this reaction, the selective activation of carbonyl compounds through the use of an appropriate Lewis acid is pivotal for achieving the desired cycloaddition, which renders the carbonyl compounds as highly effective dipolarophiles, facilitating their interception of the reactive zwitterionic intermediate while simultaneously suppressing unwanted side reactions. In the case with isatins as dipolarophiles, Cu(CH3CN)4PF6 complexed with a commercially available chiral Box ligand was found to be the optimal catalyst, which not only promoted the formation of zwitterionic intermediates via copper carbene transfer reaction, but also activated carbonyl compounds in the following stereoselective cycloaddition50, resulting in the synthesis of spiro-tetrahydrofurans containing four consecutive stereocenters in high yields and excellent enantioselectivity. Whereas, when non-cyclic ketones or aldehydes were employed as dipolarophiles, inexpensive Zn(OTf)2 was found to be the potent Lewis acid for the activation of corresponding carbonyl compounds, forming the spiro-tetrahydrofurans with chiral zwitterionic intermediates derived from chiral dirhodium complex catalyzed carbene transfer reaction. Significantly, this cycloaddition reaction offers a complementary protocol for the stereoselective construction of chiral tetrahydrofurans, which are regioisomers of those formed by traditional [3 + 2]-cycloadditions, such as [3 + 2]-cycloaddition of carbonyl ylide with alkenes (Fig. 1b)32,33, or [3 + 2]-annulation using donor-acceptor cyclopropanes as C3 building blocks (Fig. 1d, path f)34,35.
Results
Optimizations for catalytic reactions
We initiated our investigation by exploring the three-component cycloaddition reaction using enamine 1a, 3-diazoindolin-2-one 2a, and N-methylisatin 3a as model substrates (Table 1 and see Supplementary Table 1 in SI for detail). When Cu(CH3CN)4PF6 was employed as the catalyst (5 mol%), the designed formal [1 + 2 + 2]-cycloaddition reaction occurred smoothly in DCM to give 2,3-dihydrofuran 5a in 75% yield with 3:1 dr (entry1). Subsequently, a variety of chiral BOX ligands were examined for the exploration of the asymmetric catalytic version of this cycloaddition reaction. Although benzyl-BOX L1 and Py-BOX L2 failed to deliver the desired products with the reactants retained (entries 2 and 3), the desired product 5a was obtained in 28% yield with 31% ee by using tert-butyl-BOX L3 as the chiral ligand (entry 4). Using diphenyl-BOX ligand L4 and cyclopentane-derived L5 dramatically improved the enantioselectivity to 82% and 85% ee, respectively (entries 5 and 6). Further screening of chiral ligands with different substituents on the aryl motif of the Ph-BOX ligands (L6-L10, entries 7-11) revealed that L6 showed better results in terms of yields and selectivity (entry 7, 82% yield, 92% ee). Then, different solvents were studied using Cu(CH3CN)4PF6 combined with ligand L6 as the catalyst (entries 12-23), and the results revealed that DCM gave superior results in terms of reactivity and selectivity. In addition, screening of various additives demonstrated that addition a catalytic amount of sodium tetrakis(3,5-trifluoromethyl)-phenylborate (NaBARF, 6.0 mol%) had a negligible influence on enantioselectivity (91% ee) but had a negative effect on the yield (entry 24). To our delight, when an organic base, N,N-diisopropylethylamine (DIPEA, 10 mol%), and activated 4 Å molecular sieves were used, the yield reached 92% (87% isolated yield) with excellent enantioselectivity maintained (entry 25, 92% ee). In this scenario, DIPEA may serve as a ligand to regulate the catalytic activity of the copper complex. This is evidenced by the fact that when 10 mol% of DIPEA was used as additive, the reaction rate for the formation of insertion product 4a slowed down drastically (see Supplementary Fig. S7 and S8 in SI for detail). Whereas, when Lewis acid, such as Zn(OTf)2, was employed as additive for the activation of carbonyl compounds, the isolated yield drops obviously. This decline is mainly attributed to the slowly decomposition of the cycloadduct in the presence of the Lewis acid catalyst (see Supplementary Fig. S9 in SI for detail).
Substrates scope
With the optimized conditions in hand, we set out to explore the substrate generality of this three-component reaction. As shown in Fig. 2-I, a variety of enamines 1 proceeded smoothly with 3-diazoindolin-2-one 2a, and N-methylisatin 3a to afford the annulation adducts 5, which bear four adjacent quaternary and tertiary carbon stereocenters, in generally good to high yields and excellent enantioselectivities. Various N-alkyl-substituted (E)-acrylates that bear benzyl, p-methoxybenzyl, methyl, dimethyl substitution, or allyl groups performed well to generate the products 5a-5e in good yields with 90–92% ee. Furthermore, cyclic amino motifs were well-tolerated. The enamines derived from tetrahydroisoquinoline, morpholine, or piperidine gave the corresponding products 5f-5i in >85% yields with 92–98% ee. Moreover, both the L-proline and paroxetine derived enamines also worked well under these conditions, stereoselectively producing 5j and 5k in 90% and 70% yields, respectively. In addition, propargyl ester derived enamine performed smoothly, affording product 5 l in 75% yield with 90% ee and 11:1 dr.
Reaction conditions: 1 (0.10 mmol), 2 (0.15 mmol), 3 (0.10 mmol), Cu(CH3CN)4PF6 (1.8 mg, 5.0 mol%), L6 (2.9 mg, 6.0 mol%), DIPEA (1.8 μL, 10 mol%), and 4 Å MS (50 mg) in DCM (2.0 mL) at 30 °C. Yields are given in isolated yields. aThe reaction was conducted at 25 °C.
Subsequently, the substrate scope with respect to the diazo compounds was further elaborated (Fig. 2-II). A series of diazooxindoles bearing different substituents on the phenyl ring reacted with (E)-3-(dibenzylamino)acrylate 1a and isatin 3a under the standard conditions. These results revealed that the electronic effects and the position of the substituents on the diazooxindoles had negligible influence on the enantioselectivities. The corresponding three-component products 6a-6e were obtained in moderate to good yields with 88–95% ee. Then, the influence of the substituents on the aromatic ring of isatin has been explored. A wide range of substituents on the aryl motif having different electronic properties at C5, C6, or C7 positions of isatins did not have significant impact on the reactivity, smoothly furnishing spirotetrahydrofuran oxindoles 6f-6j in high yields with 90–91% ee and >5:1 dr. However, no reaction occurred with isatin having substitution at the C4 position, which might be due to the steric effects. Notably, the benzo[b]thiophene-2,3-dione and benzofuran-2,3-dione also worked very well under current conditions, forming polycyclic compounds 6k and 6 l in 75% and 76% yield with 90% and 93% ee, respectively. Using isatin derivatives with different N-protection groups, including benzyl, isopropyl, propargyl, tert-butyldimethylsilyl (TBS), ethyl, and allyl, all performed well in this reaction, providing the corresponding adducts 6m-6u in high yields with up to >20:1 dr and excellent ee. However, other type of diazo compounds, such as ethyl diazoacetate or phenyl diazoacetate, are not work under current conditions. The absolute configuration of products 6k and 6 u was determined by single-crystal X-ray diffraction analysis, and the other products were tentatively assigned by analogy.
Encouraged by these promising results, we then considered the possibility of extending this three-component reaction to linear ketones, and even to aldehydes. However, only insertion product 4b was obtained in 72% yield when amide 7a containing a phenylethynyl group was employed as dipolarophile for the reaction with methyl (E)-3-(dibenzylamino)acrylate 1b and N-methyl 3-diazoindolin-2-one 2b under above optimal conditions using chiral copper complex as catalyst (see Supplementary Table 2, entry 2 in SI for detail). These results implied that the key zwitterionic intermediate was formed smoothly under these conditions, since the insertion product 4b was formed in high yield. Thus, exploration of appreciate and compatible Lewis acid for the selective activation of this type of carbonyl compounds was conducted. After optimization using 1b, 2b, and 7a as model substrates (see Supplementary Tables 2–4 in the Supplementary Information for details), the best conditions were identified by using a combination of Rh2(R-TBPTTL)4 (1.0 mol%) with Zn(OTf)2 (10 mol%) as the effective catalytic system, DCM as the solvent at 0 °C for 12 h, affording the desired product 8a in 65% yield as a single diastereomer with 92% ee (see Supplementary Table 4, entry 5 in the Supplementary Information for detail). With these optimal reaction conditions in hand, the substrate generality of this Rh-catalyzed asymmetric formal [1 + 2 + 2]-cycloaddition was investigated (Fig. 3). Notably, both the alkynyl ketones and aldehydes were compatible in this reaction, delivering the chiral furan derivatives 8a-8e in 65-75% yields with up to 98% ee. Moreover, the cinnamaldehyde derivative and a variety of substituted benzaldehydes were also suitable substrates, and allowed the generation of the desired products 8f-8i in good to high yields with 91-94% ee. It should be mentioned that one equivalent Zn(OTf)2 was used in the case with bromo-substituted benzaldehyde to ensure the synthetically useful yield. And benzaldehydes with electron-donating substitutions did not gave any desired product under current conditions. Then, a variety of N-substituted diazo compounds 2 and different esters of enamines 1 were subjected to the optimal conditions, such as allyl, propinyl, and benzyl groups, providing the cycloaddition products 8k–8n in good yields with 90–99% ee. Furthermore, aldehyde containing a conjugated eneyne motif underwent the reaction smoothly, delivering the poly-functionalized product 8o in 54% yield with 93% ee. The remaining eneyne unit provides a practical synthetic handle for further structural diversification. However, alkyl substituted enolizable ketone does not work under current conditions, only providing the insertion product 4a with majority of the electrophile remaining intact. The absolute configuration of product 8a was determined to be (2 R,3 R,4 R,5 R) via single-crystal X-ray diffraction analysis, and the other products were tentatively assigned by analogy.
Reaction conditions: enamine 1 (0.10 mmol), carbonyl compound 7 (0.10 mmol, 1.0 eq.), Zn(OTf)2 (3.6 mg, 10 mol%), Rh2(R-TBPTTL)4 (2.5 mg, 1.0 mol%), and diazo compound 2 (0.15 mmol) in DCM (2.0 mL). Yields are given in isolated yields and the products are obtained as single diastereomers. aThe reaction was conducted at –20 °C. bThe p-bromobenzaldehyde (0.20 mmol, 2.0 eq.) in combination with 1.0 eq. of Zn(OTf)2 were used.
Mechanistic studies and proposed mechanism
To obtain some insight into the mechanistic details, a series of control experiments were conducted. Initially, the reaction of the formal Csp2 − H insertion product 4a with 3a was performed under standard conditions, and both materials remained intact (Fig. 4a, and see Supplementary Fig. S1 in SI for details). This observation suggested that compound 4a is not a possible intermediate in this reaction, and a stepwise reaction pathway is unlikely. Moreover, no reaction occurred by the combination of 1a and 3a under the optimal conditions (Fig. 4b, and see Supplementary Fig. S2 in SI for details). In addition, treatment of 2a with 3a under the same conditions did not lead to any identifiable product, and the diazo compound 2a decomposed into a complex mixture with most of 3a recovered (Fig. 4c, and see Supplementary Fig. S3 in SI for details). These results implied that the current reaction could neither be initiated via addition of 1a with 3a, nor undergo carbonyl ylide formation pathway through carbene intermediate interaction with the carbonyl substrate 2a32,33. The reaction with deuterated water was carried out under the standard conditions, and product 5a was not deuterated (Fig. 4d, and see Supplementary Fig. S4 in SI for details), which confirmed that an external H-exchange process between iminium and enamine species did not occur prior to the cycloaddition with 3a. Moreover, no reaction happened when control experiment of prepared donor-acceptor cyclopropane with N-methylisatin 3a was conducted under optimal copper-catalyzed conditions (see Supplementary Fig. S11 in SI for details). These results implied that the cyclopropane species might not be the possible intermediate in this reaction. In addition, the proton NMR monitoring of the crude reaction did not observe any potential signal of cyclopropane formation (see Supplementary Fig. S8 and S10 in SI for details).
a Control experiment with C-H insertion product 4a and 3a under standard conditions. b Control experiment of 1a with 3a under standard conditions. c Control experiment of 2a with 3a under standard conditions. d Control experiment of model reaction under standard conditions in the presence of D2O. e Nonlinear effect study results of Cu-catalyst catalysis. f Linear effect study results of Rh-catalyst catalysis.
In the Cu-catalyzed reaction, we have conducted a set of experiments with ee-varied chiral ligand L6, enamine 1a, 3-diazoindolin-2-one 2a, and N-methylisatin 3a under the otherwise same optimal conditions. These results clearly disclosed a positive nonlinear relationship between the enantiomeric excess of L6 and the ee value of the product 5a, indicating a high-order catalyst aggregate could be the origin of this phenomenon in the copper-catalyzed reaction (Fig. 4e, and see Supplementary Fig. S5 in SI for detail), but other explanations are also possible51,52,53,54,55. The study of Cu/L6 ratios revealed that around 1:1.2 is optimal, further increasing the amount of chiral ligand L6 had no influence on the ee values, but the yield dropped obviously (see Supplementary Table 7 in SI for details). These observations implied the formation of either less reactive or nonreactive dimer Cu(L6)2 complexes in this system, which is consistent with the observed positive nonlinear effect (NLE). Furthermore, detailed kinetic studies were performed through monitoring the initial reaction rates (see Supplementary Table 8 in SI for details). These observations revealed second-order dependence of the reaction rates on the concentration of the copper catalyst, which also aligned with the dual activation model. In addition, when chiral dirhodium complex Rh2(R-TBPTTL)4 was used as the catalyst instead of copper complex in the reaction with aldehyde 7c as dipolarophile, a linear relationship was observed between the enantiomeric excesses of the obtained product 8c and the ee values of corresponding used chiral Rh2(R-TBPTTL)4, which implied the involvement of one molecule of chiral dirhodium catalyst in the enantio-determining transition state (Fig. 4f, and see Supplementary Fig. S6 in SI for detail).
Based on these results and previously reported literature41,42,43,44,45, a plausible reaction pathway has been proposed (Fig. 5). In the Cu-catalyzed reaction (Fig. 5a), the copper carbene intermediate A-1 is generated from diazo compound 2a in the presence of the copper catalyst, followed by nucleophilic addition with enamine 1a to produce the iminium ion B-1, and the copper catalyst might migrate instantaneously to the oxygen atom to give a relative more stable enolate form species B’-143. Direct H-shift would occur with B-1 or B’-1 to produce the formal insertion product 4a in the absence of trapping reagent45. In the following 1,3-dipolar cycloaddition reaction, the chiral copper catalyst demonstrated a dual interaction model according to the observed nonlinear effect (Fig. 4e), which not only promoted the formation of the cooper associated enolate species B’-1 that serves as the C3 synthon, but also ensued the excellent stereoselectivity in the following cycloaddition reaction through activation of isatin 3 via coordination with the two carbonyl groups56, delivering the chiral spirotetrahydrofuran oxindoles 5 or 6 with excellent enantioselectivity via proposed transition state C-1 and regenerating the copper catalyst. Whereas, in the Rh-catalyzed version, similar rhodium carbene generation, followed by addition with electronic rich enamine to form the rhodium associated enolate species B’-2 has been proposed (Fig. 5b)57. On the other hand, the dirhodium complex could not effectively activate the non-cyclic ketones or aldehydes due to it lower Lewis acidity under current conditions, and no cycloaddition adduct was formed in the absence of external Lewis acid (see Supplementary Table 4 in SI for detail). Thus, additional Lewis acid, Zn(OTf)2, was introduced in this case, which could interact with the non-cyclic carbon compounds 7 effectively and selectively, producing the chiral cyclic adducts 8 via C-2 according to the observed stereochemistry of these formed chiral cycloadducts.
a Proposed catalytic cycle under chiral copper complex catalysis. b Proposed catalytic cycle under chiral dirhodium complex and Lewis acid cooperative catalysis.
Gram-scale reaction and applications
To demonstrate the synthetic utility of this asymmetric [3 + 2]-cycloaddition reaction, a four-component gram-scale reaction was conducted under the optimal conditions, in which mono-Boc-protected piperazine and terminal alkyne were used instead of presynthesized enamine, providing 2.68 g of pure product 5i in 79% yield with 93% ee as single diastereoisomer (Fig. 6a). Further transformations of obtained enantioenriched spiro-tetrahydrofurans 5 and 8 were performed to illustrate the synthetic potential of this method (Figs. 6b and 6c). Oxidative debenzylation of 5a in the presence of ceric ammonium nitrate (CAN) gave amine product 9 in 88% yield with 90% ee. Treatment of the propargyl alcohol derived product 5 l with tosyl azide in the presence of copper(I) thiophene-2-carboxylate (CuTc) afforded triazole 10 in 90% yield with 90% ee via a click-reaction. The elimination product 11 could be obtained in 95% yield from 5i by using m-CPBA as the oxidant in the presence of K2CO3 in N, N-dimethylformamide (DMF). Methylation of 5i yielded 12 in high yield. The click-reaction product 13 could also be formed from 8 m with tosyl azide in high yield. Intramolecular alkyne hydroarylation occurred smoothly with 8o in the presence of gold catalyst, leading to the tetracyclic product 14 in 73% yield. In all these cases, the enantiomeric excesses were well conserved. In addition, these obtained products on cell viability was evaluated via CCK8 assay in different human cancer cell lines with camptothecin (CPT) as control (see Supplementary Table 9 in SI for details). According to these results, compound 5e exhibits significant anticancer potency than other tested molecules with 5-fluorouracil (5-Fu) as comparison towards HCT116 cancer cells (see Supplementary Fig. S15 in SI for details: 5e, IC50 = 3.1 0.5 μM).
a Gram-scale reaction for the synthesis of 5i. b Synthetic transformations of products 5. c Synthetic transformations of products 8.
Discussion
In conclusion, we have developed an enantioselective formal [1 + 2 + 2]-cycloaddition reaction of diazoamides with enamines and carbonyl compounds, which provides a practical and atom-economic approach to dual spiro-tetrahydrofurans containing four consecutive stereocenters in high yields and excellent enantioselectivity. In this reaction, selective activation of the carbonyl compounds through the use of an appropriate Lewis acid is pivotal for achieving the desired 1,3-dipolar cycloaddition with in situ formed zwitterionic intermediates. The Cu(CH3CN)4PF6 complexed with a commercially available chiral Box ligand demonstrated robust catalytic ability and selectivity control in the case with isatins as dipolarophiles, which not only promoted the formation of zwitterionic intermediates, but also activated carbonyl compounds in the following stereoselective cycloaddition. When non-cyclic ketones or aldehydes were employed as dipolarophiles, inexpensive Zn(OTf)2 was found to be the potent Lewis acid for the activation of linear carbonyl compounds, forming the corresponding spiro-tetrahydrofurans with chiral zwitterionic intermediates derived from chiral dirhodium complex catalyzed carbene transfer reaction. This cycloaddition reaction features mild conditions, broad substrates scope, and high bond formation efficiency, which offers an essential complementary protocol for the stereoselective construction of chiral tetrahydrofurans that are regioisomers of those formed by other reported cycloadditions. However, alkyl substituted enolizable ketone/aldehyde does not work under current conditions. Preliminary anti-tumor activity study of these obtained chiral molecules indicates that some compounds exhibit high anticancer potency against colon cancer cells (HCT-116 cells, IC50 = 3.1 ± 0.5 μM). The broad utility demonstrated herein would set the stage for the invention of multi-component reactions through the strategic design of interception protocols targeting these proposed intermediates. Further synthetic applications based on this chemistry are underway in our laboratory, and will be reported in due course.
Methods
General procedure for the chiral copper complex catalyzed asymmetric formal [1 + 2 + 2]-cycloaddition
To an oven-dried 10 mL Schlenk tube equipped with a magnetic stirring bar was added Cu(MeCN)4PF6 (1.8 mg, 0.005 mmol, 5.0 mol%), ligand L6 (2.9 mg, 0.006 mmol, 6.0 mol%), and dissolved in dried dichloromethane (DCM, 1.0 mL) under argon atmosphere. After the resulting mixture was stirred at 30 °C (or 25 °C for the indicated cases) for 0.5 h. Then, a mixture of enamine 1 (0.10 mmol, 1.0 equiv.), diazo compound 2 (0.15 mmol, 1.5 equiv.), isatin 3 (0.10 mmol, 1.0 equiv.), and iPr2NEt (1.3 mg, 0.01 mmol, 10 mol%) in DCM (1.0 mL), and 4 Å molecular sieve (50 mg) were added to the above reaction mixture. After addition, the reaction mixture was stirred for additional 12 ~ 36 h under these conditions until consumption of the materials (monitored by TLC). Then the reaction mixture was purified by column chromatography on silica gel without any additional treatment (hexanes: EtOAc = 20:1 to 3:1) to give the pure products 5 or 6 in good to high yields with excellent enantioselectivity.
General procedure for the chiral dirhodium complex and Zn(OTf)2 cooperative catalyzed asymmetric formal [1 + 2 + 2]-cycloaddition
To a 10-mL oven-dried vial containing a magnetic stirring bar, Rh2(R-TBPTTL)4 (2.5 mg, 1.0 mol%), Zn(OTf)2 (3.6 mg, 10 mol%), carbonyl compound 7 (0.10 mmol, 1.0 equiv.), and enamine 1 (0.10 mmol, 1.0 equiv.) in dry DCM (1.0 mL), was added a solution of diazo compound 2 (0.15 mmol, 1.5 equiv.) in dry DCM via a syringe pump over 1 h under an argon atmosphere at 0 °C. After addition, the reaction mixture was stirred overnight under these conditions until consumption of the material (monitored by TLC) was complete. Then the reaction mixture was purified by column chromatography on silica gel without any additional treatment (hexanes: EtOAc = 20:1 to 10:1) to give 8 in good to high yields with excellent enantioselectivity.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data supporting the findings of this study are available within the paper and its Supplementary Information. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Center accession code numbers CCDC 2489845 (6k), 2289772 (6 u), 2414774 (8a). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. All data are available from the corresponding author upon request.
References
Kacprzak, K., Skiera, I., Piasecka, M. & Paryzek, Z. Alkaloids and isoprenoids modification by copper(I)-catalyzed Huisgen 1,3-dipolar cycloaddition (click chemistry): Toward new functions and molecular architectures. Chem. Rev. 116, 5689–5743 (2016).
Bilodeau, D. A., Margison, K. D., Serhan, M. & Pezacki, J. P. Bioorthogonal reactions utilizing nitrones as versatile dipoles in cycloaddition reactions. Chem. Rev. 121, 6699–6717 (2021).
Roglans, A., Pla-Quintana, A. & Solà, M. Mechanistic studies of transition-metal-catalyzed [2 + 2 + 2] cycloaddition reactions. Chem. Rev. 121, 1894–1979 (2021).
Hashimoto, T. & Maruoka, K. Recent advances of catalytic asymmetric 1,3-dipolar cycloadditions. Chem. Rev. 115, 5366–5412 (2015).
Breugst, M. & Reissig, H.-U. The Huisgen reaction: milestones of the 1,3-dipolar cycloaddition. Angew. Chem. Int. Ed. 59, 12293–12307 (2020).
Tang, S., Zhang, X., Sun, J., Niu, D. & Chruma, J. J. 2-Azaallyl anions, 2-azaallyl cations, 2-azaallyl radicals, and azomethine ylides. Chem. Rev. 118, 10393–10457 (2018).
Akter, M., Rupa, K. & Anbarasan, P. 1,2,3-Triazole and its analogues: new surrogates for diazo compounds. Chem. Rev. 122, 13108–13205 (2022).
Wu, M.-Y., He, W.-W., Liu, X.-Y. & Tan, B. Asymmetric construction of spirooxindoles by organocatalytic multicomponent reactions using diazooxindoles. Angew. Chem. Int. Ed. 54, 9409–9413 (2015).
Jia, Z.-J., Shan, G., Daniliuc, C. G., Antonchick, A. P. & Waldmann, H. Enantioselective synthesis of the spirotropanyl oxindole scaffold through bimetallic relay catalysis. Angew. Chem. Int. Ed. 57, 14493–14497 (2018).
Day, J., McKeever-Abbas, B. & Dowden, J. Stereoselective synthesis of tetrahydroindolizines through the catalytic formation of pyridinium ylides from diazo compounds. Angew. Chem. Int. Ed. 55, 5809–5813 (2016).
Zhang, D., Lin, L., Yang, J., Liu, X. & Feng, X. Asymmetric synthesis of tetrahydroindolizines by bimetallic relay catalyzed cycloaddition of pyridinium ylides. Angew. Chem. Int. Ed. 57, 12323–12327 (2018).
Zhou, Z. et al. Switchable regioselectivity in amine-catalysed asymmetric cycloadditions. Nat. Chem. 9, 590–594 (2017).
Doerksen, R. S. et al. Ruthenium-catalyzed cycloadditions to form five-, six-, and seven-membered rings. Chem. Rev. 121, 4045–4083 (2021).
Stanley, L. M. & Sibi, M. P. Enantioselective copper-catalyzed 1,3-dipolar cycloadditions. Chem. Rev. 108, 2887–2902 (2008).
Garayalde, D. & Nevado, C. Gold-containing and gold-generated 1,n-dipoles as useful platforms toward cycloadditions and cyclizations. ACS Catal. 2, 1462–1479 (2012).
Xu, P.-W. et al. Catalytic enantioselective construction of spiro quaternary carbon stereocenters. ACS Catal. 9, 1820–1882 (2019).
Burke, M. D. & Schreiber, S. L. A planning strategy for diversity-oriented synthesis. Angew. Chem. Int. Ed. 43, 46–58 (2004).
Kato, N. et al. Diversity-oriented synthesis yields novel multistage antimalarial inhibitors. Nature 538, 344–349 (2016).
Cohen, Y., Cohen, A. & Marek, I. Creating stereocenters within acyclic systems by C-C bond cleavage of cyclopropanes. Chem. Rev. 121, 140–161 (2021).
Pirenne, V., Muriel, B. & Waser, J. Catalytic enantioselective ring-opening reactions of cyclopropanes. Chem. Rev. 121, 227–263 (2021).
Sokolova, O. O. & Bower, J. F. Selective carbon-carbon bond cleavage of cyclopropylamine derivatives. Chem. Rev. 121, 80–109 (2021).
Wei, L., Chang, X. & Wang, C.-J. Catalytic asymmetric reactions with N-metallated azomethine ylides. Acc. Chem. Res. 53, 1084–1100 (2020).
Wang, J., Blaszczyk, S. A., Li, X. & Tang, W. Transition metal-catalyzed selective carbon-carbon bond cleavage of vinylcyclopropanes in cycloaddition reactions. Chem. Rev. 121, 110–139 (2021).
Cheng, Q.-Q., Deng, Y., Lankelma, M. & Doyle, M. P. Cycloaddition reactions of enoldiazo compounds. Chem. Soc. Rev. 46, 5425–5443 (2017).
Kelleghan, A. V., Meza, A. T. & Garg, N. K. Generation and reactivity of unsymmetrical strained heterocyclic allenes. Nat. Synth. 3, 329–336 (2024).
Li, Y.-L., Zhang, P.-C., Wu, H.-H. & Zhang, J. Palladium-catalyzed asymmetric tandem denitrogenative Heck/Tsuji–Trost of benzotriazoles with 1,3-dienes. J. Am. Chem. Soc. 143, 13010–13015 (2021).
Zhou, X.-S. et al. Asymmetric [3 + 2] photocycloaddition of β-keto esters and vinyl azides by dual photoredox/nickel catalysis. J. Am. Chem. Soc. 145, 12233–12243 (2023).
Chen, X.-H., Wei, Q., Luo, S.-W., Xiao, H. & Gong, L.-Z. Organocatalytic synthesis of spiro[pyrrolidin-3,3′-oxindoles] with high enantiopurity and structural diversity. J. Am. Chem. Soc. 131, 13819–13825 (2009).
Lin, S., Deiana, L., Zhao, G.-L., Sun, J. & Córdova, A. Dynamic one-pot three-component catalytic asymmetric transformation by combination of hydrogen-bond-donating and amine catalysts. Angew. Chem. Int. Ed. 50, 7624–7630 (2011).
Aggarwal, S., Vu, A., Eremin, D. B., Persaud, R. & Fokin, V. V. Arenes participate in 1,3-dipolar cycloaddition with in situ-generated diazoalkenes. Nat. Chem. 15, 764–772 (2023).
Hayashi, H. et al. In silico reaction screening with difluorocarbene for N-difluoroalkylative dearomatization of pyridines. Nat. Synth. 1, 804–814 (2022).
Padwa, A. Domino reactions of rhodium(II) carbenoids for alkaloid synthesis. Chem. Soc. Rev. 38, 3072–3081 (2009).
Hou, B. L. et al. Facile generation of bridged medium-sized polycyclic systems by rhodium-catalysed intramolecular (3 + 2) dipolar cycloadditions. Nat. Commun. 12, 5239 (2021).
Parsons, A. T. & Johnson, J. S. Catalytic enantioselective synthesis of tetrahydrofurans: a dynamic kinetic asymmetric [3 + 2] cycloaddition of racemic cyclopropanes and aldehydes. J. Am. Chem. Soc. 131, 3122–3123 (2009).
de Nanteuil, F., Serrano, E., Perrotta, D. & Waser, J. Dynamic kinetic asymmetric [3 + 2] annulation reactions of aminocyclopropanes. J. Am. Chem. Soc. 136, 6239–6242 (2014).
Liu, H.-K. et al. Selectivity switch in a rhodium(II) carbene triggered cyclopentannulation: divergent access to three polycyclic indolines. Angew. Chem. Int. Ed. 58, 4345–4349 (2019).
Zhang, M.-C. et al. Cu-catalyzed asymmetric dearomative [3 + 2] cycloaddition reaction of benzazoles with aminocyclopropanes. Chem 5, 156–167 (2019).
Wei, W. et al. Catalytic asymmetric redox-neutral [3 + 2] photocycloadditions of cyclopropyl ketones with vinylazaarenes enabled by consecutive photoinduced electron transfer. Angew. Chem. Int. Ed. 63, e202406845 (2024).
Bao, M., Zhou, S., Hu, W. & Xu, X. Recent advances in gold-complex and chiral organocatalyst cooperative catalysis for asymmetric alkyne functionalization. Chin. Chem. Lett. 33, 4969–4979 (2022).
Xie, X., Dong, S., Hong, K., Huang, J. & Xu, X. Catalytic asymmetric difluoroalkylation using in situ generated difluoroenol species as the privileged synthon. Adv. Sci. 11, 2307520 (2024).
Zhou, S. et al. Enantioselective oxidative multi-functionalization of terminal alkynes with nitrones and alcohols for expeditious assembly of chiral α-alkoxy-β-amino-ketones. J. Am. Chem. Soc. 143, 14703–14711 (2021).
Hong, K. et al. Asymmetric three-component reaction of enynal with alcohol and imine as an expeditious track to afford chiral α-furyl-β-amino carboxylate derivatives. ACS Catal. 12, 14185–14193 (2022).
Dong, S. et al. Construction of chiral quaternary carbon centers via asymmetric metal carbene gem-dialkylation. Angew. Chem. Int. Ed. 62, e202302371 (2023).
Yao, M., Dong, S. & Xu, X. Asymmetric carbene transformations for the construction of all-carbon quaternary centers. Chem. Eur. J. 30, e202304299 (2024).
Yun, S. H., Xia, L., Kim, S. H. & Lee, Y. R. Rh(II)-catalyzed chemoselective synthesis of 3-substituted oxindoles by C(sp2)-H and C(sp2)-N functionalization of β-enaminoesters. Asian J. Org. Chem. 5, 1142–1147 (2016).
Dong, K. et al. Catalyst-directed divergent catalytic approaches to expand structural and functional scaffold diversity via metallo-enolcarbene intermediates. ACS Catal. 11, 4712–4721 (2021).
Pellissier, H. Recent developments in asymmetric cyclopropanation. Tetrahedron 64, 7041–7095 (2008).
Bloomer, B. J. et al. Mechanistic and structural characterization of an iridium-containing cytochrome reveals kinetically relevant cofactor dynamics. Nat. Catal. 6, 39–51 (2023).
Zhang, C. et al. Gold(I)-catalyzed intramolecular cyclization/intermolecular cycloaddition cascade as a fast track to polycarbocycles and mechanistic insights. Nat. Commun. 12, 1182 (2021).
Liao, S., Sun, X.-L. & Tang, Y. Side arm strategy for catalyst design: modifying bisoxazolines for remote control of enantioselection and related. Acc. Chem. Res. 47, 2260–2272 (2014).
Satyanarayana, T., Abraham, S. & Kagan, H. B. Nonlinear effects in asymmetric catalysis. Angew. Chem. Int. Ed. 48, 456–494 (2009).
Chen, X.-H., Zhang, W.-Q. & Gong, L.-Z. Asymmetric organocatalytic three-component 1,3-dipolar cycloaddition: control of stereochemistry via a chiral Brønsted acid activated dipole. J. Am. Chem. Soc. 130, 5652–5653 (2008).
Liu, S., Chan, K. L., Lin, Z. & Sun, J. Asymmetric synthesis of remotely chiral naphthols and naphthylamines via naphthoquinone methides. J. Am. Chem. Soc. 145, 12802–12811 (2023).
Momiyama, N., Konno, T., Furiya, Y., Iwamoto, T. & Terada, M. Design of chiral bis-phosphoric acid catalyst derived from (R)−3,3’-di(2-hydroxy−3-arylphenyl)binaphthol: catalytic enantioselective diels-alder reaction of α,β-unsaturated aldehydes with amidodienes. J. Am. Chem. Soc. 133, 19294–19297 (2011).
Li, X.-Z., He, Y.-P. & Wu, H. Multicomponent cyclizative 1,2-rearrangement enabled enantioselective construction of 2,2-disubstituted pyrrolinones. Angew. Chem. Int. Ed. 63, e202317182 (2024).
Moskowitz, M. & Wolf, C. Catalytic enantioselective ynamide additions to isatins: concise access to oxindole alkaloids. Angew. Chem. Int. Ed. 58, 3402–3406 (2019).
Kisan, H. K. & Sunoj, R. B. Asymmetric cooperative catalysis in a three-component reaction: mechanism and origin of enantio- and diastereoselectivities. Org. Lett. 18, 3746–3749 (2016).
Acknowledgements
Support for this research from the National Natural Science Foundation of China (22371309), and the Guangdong Basic and Applied Basic Research Foundation (2024B1515040025) is greatly acknowledged.
Author information
Authors and Affiliations
Contributions
X.F.X. supervised the project and wrote the manuscript. S.D. and M.Y. designed the experiments. S.D., M.Y., Y.W., and X.D.X. performed the experiments and analyzed the data. X.J. carried out the anticancer activity evaluation. All authors contributed to discussions.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests
Peer review
Peer review information
Nature Communications thanks Hai-Ming Guo and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dong, S., Yao, M., Wang, Y. et al. Asymmetric formal [1 + 2 + 2]-cycloaddition of diazoamides with enamines and carbonyl compounds. Nat Commun 16, 10624 (2025). https://doi.org/10.1038/s41467-025-65612-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-65612-3

![Fig. 1: Catalytic [3 + 2]- and formal [1 + 2 + 2]-cycloadditions.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-65612-3/MediaObjects/41467_2025_65612_Fig1_HTML.png)
![Fig. 2: Substrate scope for the Cu-catalyzed asymmetric formal [1 + 2 + 2]-cycloaddition.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-65612-3/MediaObjects/41467_2025_65612_Fig2_HTML.png)
![Fig. 3: Substrate scope for the Rh-catalyzed asymmetric formal [1 + 2 + 2]-cycloaddition.](http://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fs41467-025-65612-3/MediaObjects/41467_2025_65612_Fig3_HTML.png)


