Abstract
Cervical cancer remains a significant health burden, and effective screening is essential, yet the age-specific performance of HPV primary screening is rarely studied. This multicenter study evaluates age-specific performance of primary human papillomavirus (HPV) testing as cross-sectional and longitudinal screening for cervical cancer among 28,501 Chinese women. At baseline, HPV screening with cytology triage demonstrates higher sensitivity (96.9% vs. 79.7%) but slightly lower specificity (88.8% vs. 92.7%) than cytology with HPV triage for cervical intraepithelial neoplasia grade 2 or worse (CIN2+). HPV sensitivities remain high across age groups (≤35: 100.0%; 36–45: 96.4%; >45: 96.8%), consistently exceeding the corresponding cytology sensitivities (66.7%, 75.7%, and 84.9%). Over a three-year follow-up, HPV screening maintains superior sensitivity (93.1% vs. 58.1%) and slightly lower specificity (89.6% vs. 92.4%) compared with cytology. Age-stratified analyses in the longitudinal setting reveal trends similar to those observed in the cross-sectional analyses. Women positive for HPV16/18 have a 43.0% three-year risk of CIN2+, compared with 0.2% for HPV-negative women. These findings support the durable protection of HPV-based screening and the feasibility of extending screening intervals.
Similar content being viewed by others
Introduction
Cervical cancer is the fourth leading cause of cancer-related mortality and morbidity among women worldwide1. High-income countries with organized screening programs have seen a decline in cervical cancer incidence, whereas developing countries, including China, continue to face rising cases2,3. Cervical cancer screening remains one of the most effective strategies for reducing the disease burden4,5. Despite the National Cervical Cancer Screening Program being in place for over 15 years, significant challenges persist in its implementation across China.
Currently, Pap smear tests and visual inspection with acetic acid or Lugol’s iodine are the most commonly used screening methods in China6. Although cytology-based screening has contributed to reducing cervical cancer incidence and mortality for decades, its relatively low sensitivity—approximately 60% for conventional cytology and 70% for liquid-based cytology (LBC)—necessitates repeated screening7,8. Additionally, cytology screening relies heavily on medical personnel and diagnostic services, raising concerns about its long-term sustainability within China’s national screening program9.
Strong evidence has indicated that cervical cancer screening with high-risk oncogenic human papillomavirus (HR-HPV) testing is more effective for the prevention of precancer and cancer than cervical cytology10,11,12,13,14. Moreover, an extension of the screening interval is permitted for primary HPV screening14,15. Thus, the World Health Organization (WHO) has recommended HPV nucleic acid testing as the preferred primary screening method for cervical cancer. Additionally, WHO has recommended that 70% of women undergo a high-performance screening before the age of 35 and again by the age of 45, by 203016. However, only clinically validated HPV tests could be used for cervical cancer screening17. China has issued guidelines on HPV assays for cervical screening in the Chinese population18. Nevertheless, there is a lack of longitudinal studies evaluating the age-specific clinical performance of high-performance HPV testing in cervical cancer screening in China.
In this work, following Chinese guidelines, we first evaluate the combined clinical performance of three high-performance HPV tests—HBRT-14 (DNA-based, qPCR), DH3 (DNA-based, hybrid capture), and Aptima (mRNA-based)—for cervical cancer screening in the general Chinese population, both at baseline and after a 3-year follow-up. We further assess the age-specific clinical performance of HPV testing across different age groups (≤35, 36–45, and >45 years) as recommended by the WHO. We find that primary HPV screening with cytology triage demonstrates higher sensitivity but slightly lower specificity than cytology-based screening for detecting cervical intraepithelial neoplasia (CIN) grade 2 or worse (CIN2+). The sensitivity of HPV screening with LBC triage remains consistently high across all age groups. The adoption of high-performance HPV testing could enhance the quality of population-level cervical cancer screening, contributing to the WHO’s target of screening 70% of women.
Results
Distribution of HPV and cytological diagnoses at baseline
Among 28,501 screened participants (Table 1), 3497 (12.3%) were 14 HR-HPV positive (814, HPV16/18 positive; 2683, other HR-HPV positive), while 2327 (8.2%) had abnormal cytology results, including 884 cases of low-grade squamous intraepithelial lesions (LSIL) or worse (LSIL+) and 1443 cases of atypical squamous cervical cells of undetermined significance (ASC-US). As expected, the abnormal rate of cytology increased with age, from 5.2% (223/4300) in women aged ≤35 years to 13.1% (459/3514) in women aged >55 years. HPV16/18 positive rate also showed the same trend with age, rising from 2.1% (89/4300) to 3.9% (128/3514). In addition, the prevalence of HR-HPV slightly declined from 11.0% (472/4300) in women aged ≤35 years to 10.9% (1053/9677) in women aged 36–45, before rising to 15.3% (537/3514) in women aged >55 years.
Clinical performance of screening in the cross-sectional setting
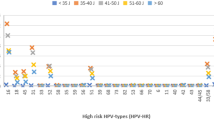
Results of sensitivity and specificity of the three HPV tests (HBRT-H14, DH3, and Aptima) with reflex LBC are shown in Fig. 1. Overall, HBRT-H14, DH3, and Aptima had comparable cross-sectional and longitudinal primary sensitivity for CIN2+ and CIN grade 3 or worse (CIN3+). To increase statistical power, we therefore conducted a pooled analysis based on the test results from three assays.
Individual HPV assays (HBRT-H14: rhombus; DH3: circle; Aptima: triangle) and the combined three HPV assays (square) with LBC triage are shown for detecting CIN2+ (A) and CIN3+ (B) in the cross-sectional setting, and CIN2+ (C) and CIN3+ (D) in the longitudinal setting. Each symbol represents the sensitivity or specificity of the corresponding assay or the combined three assays, with point estimates shown and 95% CIs indicated by error bars. Sensitivity comparisons between assays were performed using two-sided Fisher’s exact tests: cross-sectional CIN2+ (A, Exact P = 0.731), cross-sectional CIN3+ (B, Exact P = 0.575), longitudinal CIN2+ (C, Exact P = 0.669), and longitudinal CIN3+ (D, Exact P = 0.187). HPV human papillomavirus, LBC liquid-based cytology, CIN cervical intraepithelial neoplasia, CIN2+ CIN grade 2 or worse, CIN3+ CIN grade 3 or worse, CI confidence intervals.
At the baseline screening, 359 of the 28,501 (1.7%) participants were diagnosed with CIN2+, and 219 (0.8%) were CIN3+ (Table 2). Among those with CIN2+, 96.9% (348/359) were HPV positive and only 3.1% (11/359) were HPV negative; 79.7% (286/359) were diagnosed with ASC-US or worse (ASC-US+) and 20.3% (73/359) with negative for intraepithelial lesion or malignancy (NILM). When identifying CIN2+ as an endpoint (Table 2), the clinical sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of HPV16/18 genotyping were 66.3%, 98.0%, 29.2%, and 99.6%, respectively. When women were grouped by age (≤35, 36–45, and >45 years), the sensitivity (all >61%), specificity (all >97%), and PPV (all >25%) of HPV16/18 genotyping decreased with age. In women aged ≤35 years, the sensitivity and PPV of HPV16/18 were greater than 80% and 30%, respectively, whereas the sensitivity and PPV of LSIL+ were below 34% and 16%, respectively. Furthermore, HPV screening with LBC triage had higher sensitivity (96.9% vs. 79.7%) and NPV (100.0% vs. 99.7%) but had lower specificity (88.8% vs. 92.7%) and PPV (10.0% vs. 12.3%) than LBC screening with HPV triage. In terms of age groups, the sensitivities of HPV screening with LBC triage were similar across age groups, all exceeding 96% (≤35: 100.0%, 36–45: 96.4%, and >45: 96.8%; P = 0.854), though specificity was significantly higher in the younger groups compared to the older group (89.7% and 90.4% vs. 87.5%; P < 0.001). By contrast, the sensitivity of LBC screening with HPV triage increased with age (P = 0.010), with women aged ≤35 years having a sensitivity of only 66.7%, whereas the specificity decreased with age (95.3% and 94.8% vs. 90.6%; P < 0.001). When identifying CIN3+ as an endpoint, similar results were observed in Table 2.
Moreover, the relative sensitivity and specificity of HPV testing to LBC were 1.22 (95% confidence interval [CI], 1.15–1.29; PMcNemar’s (McN) < 0.001) and 0.96 (95% CI, 0.95–0.96; PMcN < 0.001) for CIN2+, and 1.13 (95% CI, 1.06–1.19; PMcN < 0.001) and 0.96 (95% CI, 0.95–0.96; PMcN < 0.001) for CIN3+ in the entire study population (Table 3). After grouping by age, the relative sensitivity for CIN2+ declined from 1.50 for ≤35 years, 1.33 for 36–45 years, to 1.14 for >45 years. The relative specificity slightly rose from 0.94 for ≤35 years, 0.95 for 36–45 years, to 0.97 for >45 years. We also found similar relative sensitivity and specificity for CIN3+.
Clinical performance of screening in the longitudinal setting
Finally, 23,071 (80.9% follow-up rate) women were followed up for 3 years (Table 4). Among them, a total of 611 (2.6%) participants were cumulatively diagnosed with CIN2+, and 333 (1.5%) were diagnosed with CIN3+.
The sensitivity of HPV16/18 genotyping was greater than 50%, the specificity was higher than 98%, and the PPV was up to 43%. Additionally, the sensitivity of HPV screening with LBC triage for CIN2+ was greater than 90% (569/611, 93.1% [95% CI, 90.8–94.9%]), while the sensitivity of LBC screening with HPV triage was less than 60% (355/611, 58.1% [95% CI, 54.2–62.0%]). The PPV (19.7% vs. 17.2%) and NPV (99.8% vs. 98.8%) of HPV screening with LBC triage were also higher than those of LBC with reflex HR-HPV, while the specificity of HPV screening with LBC triage was slightly lower than (89.6% vs. 92.4%) that of LBC with reflex HR-HPV.
By age group, the sensitivities of HPV16/18 genotyping were greater than 46%, the specificities were higher than 97%, and the PPVs exceeded 39%. Additionally, the sensitivity of HPV screening with LBC triage grouped by age was much higher than that of LBC screening with HPV triage across all age groups (≤35 years: 89.4% vs. 51.5%; 36–45 years: 91.7% vs. 54.1%; >45 years: 94.9% vs. 62.3%). The specificity of HPV screening with LBC triage, however, was slightly lower than that of LBC screening with HPV triage in each age group: ≤35 years (90.8% vs. 95.1%), 36–45 years (91.3% vs. 94.5%), and >45 years (88.2% vs. 90.3%). The PPVs and NPVs of the two algorithms were similar across age groups. Similar results were found when CIN3+ was used as the endpoint, as shown in Table 4.
Moreover, in the entire study population under the longitudinal screening setting, the relative sensitivity of HPV testing to LBC was 1.60 (95% CI, 1.49–1.72; PMcN < 0.001) for CIN2+, and 1.49 (95% CI, 1.37–1.62; PMcN < 0.001) for CIN3+; the relative specificity was 0.97 for both CIN2+ and CIN3+. Restricting the analysis to women by age group, a decline in relative sensitivity was observed with increasing age (Table 3). Specifically, the relative sensitivity was highest in women ≤35 years, with values of 1.74 for CIN2+ and 1.82 for CIN3+. In women aged 36–45 years, the relative sensitivity was 1.69 for CIN2+ and 1.62 for CIN3+. The lowest relative sensitivity was found in women aged >45 years, with 1.52 for CIN2+ and 1.38 for CIN3+.
Discussion
To the best of our knowledge, this is the first study to prospectively evaluate the age-specific clinical performance of multiple HPV tests as primary screening for cervical cancer in accordance with Chinese guidelines. Our findings provide strong evidence supporting the effectiveness of HPV testing as a primary screening tool in both cross-sectional and longitudinal evaluations for detecting CIN2+ and CIN3+ lesions, with sensitivities exceeding 93% and specificities greater than 88%.
The age-specific pattern of HPV positivity observed in our study, with higher rates among older women, differs from that typically reported in Western countries and may be population-specific19,20,21,22. This pattern may result from low HPV vaccination coverage23, age- and region-related differences in screening participation24, and cohort or biological factors such as reduced immune clearance, postmenopausal changes, and lower condom use, which may increase susceptibility to new or reactivated HPV infections25.
In our study, primary HPV screening with cytology triage demonstrated significantly higher sensitivity but slightly lower specificity than primary cytology screening with HPV triage across the overall population and in almost every age group. Similar findings have been reported in previous studies. A meta-analysis of 40 cross-sectional studies evaluating HPV testing for CIN2+ detection found that validated HPV testing had a pooled sensitivity of 89.9% compared to 72.9% for cytology, while the pooled specificity estimates were 89.9% for HPV testing and 90.3% for cytology8. Additionally, a study of 4075 women screened using both cytology and HPV testing reported sensitivity and specificity for detecting CIN3+ of 88.2% and 78.8% for PCR-based HPV testing, and 90.8% and 72.6% for signal amplification-based HPV testing, respectively. In comparison, cytology alone had a much lower sensitivity of 61.3% and a specificity of 82.4%26. Several HPV testing methods that separate HPV16/18 genotyping from other HR-HPV types were used for cervical cancer screening. A HERMES (HEllenic Real life Multicentric cErvical Screening) study recruited 4009 women 25–55 years old from Greece and reported HR-HPV testing with HPV16/18 genotyping and reflex LBC for CIN2+ presented the optimal combination of cross-sectional sensitivity (82.9%) and specificity relative to cytology alone (0.99)27. A real-world cross-sectional study from Mexico found HR-HPV (HPV16/18 genotyping) with reflex LBC has a sensitivity of 86.6% (95%CI: 79.4–92.0%) for CIN2+, but a lower specificity with only 34.0%28. Another study involving 34,254 women aged 30 years or older from the American ATHENA (Addressing the Need for Advanced HPV Diagnostics) study, which included a 3-year follow-up, compared ten cervical cancer screening strategies. The study found that HPV16/18 genotyping with reflex cytology for other HR-HPV types had a sensitivity of 72.1% and a specificity of 85.2% for detecting CIN2+29. Although results varied across different populations, HPV testing with LBC triage has been shown in multiple studies to provide a good balance between sensitivity and specificity for cervical cancer screening27,29. Furthermore, validated HPV assays have demonstrated comparable longitudinal safety and better protection against cervical cancer than cytology alone13,30.
Our study found that the relative sensitivity of HPV testing in primary screening compared to LBC is higher in younger women. Similarly, previous studies have reported that the relative sensitivity of primary HPV testing compared to cervical cytology is greater in women under 30 or 35 years than in older women13,26,31,32. Furthermore, the relative sensitivity of primary HPV screening with LBC triage compared to LBC screening with HPV triage remained higher in older women at both our cross-sectional and longitudinal analyses. In addition, the sensitivity of HPV screening with LBC triage was comparable across all age groups, whereas its specificity was higher among women aged ≤45 years than those aged >45 years. These findings support HPV testing as a more appropriate primary screening strategy for older women, in line with earlier evidence33. However, we also found that LBC with reflex HPV testing demonstrated higher sensitivity in older women than in younger women. A possible explanation is that the prevalence of LSIL+ lesions increases with age, making cytological abnormalities more apparent and thus easier to detect. Consistent with this, in our study, the prevalence of LSIL+ lesions rose from 1.6% among women aged ≤35 years to 3.8% among those aged 46–55 years (Table 1). Nevertheless, with further increases in age, particularly after menopause, the diagnostic accuracy of cytology may decline again34. This reduction is partly attributable to age-related anatomical changes, such as regression of the squamocolumnar junction into the endocervical canal, which limits access to the transformation zone and may lead to inadequate sampling. Similarly, in our study, among women older than 55 years, the proportion of LSIL+ lesions slightly decreased to 3.4%, whereas the proportion of ASC-US increased to 9.7%, suggesting that cytological interpretation becomes increasingly challenging in this age group.
HPV16/18 genotyping, which is most strongly associated with cervical cancer, along with the detection of other high-risk HPV types, helps facilitate the further triage of primary HPV-positive women. Our data showed that the cross-sectional clinical sensitivity of HPV16/18 for detecting CIN2+ was over 60%, with a 3-year longitudinal sensitivity of approximately 50%. In comparison, other HR-HPV types combined with LBC triage demonstrated a cross-sectional sensitivity of around 30% and a longitudinal sensitivity exceeding 40%. These findings highlight that, besides HPV16/18, other HR-HPV types also play a significant role in cervical cancer screening, particularly in the context of long-term follow-up. Similar observations have been reported in other studies35,36. A U.S.-based prospective cohort study found that HPV16/18 had a cross-sectional sensitivity of 62.0%, while an extended genotyping model including HPV16/18/31/33/35/39/45/51/52/56/58/59/68 achieved a higher sensitivity of 94.7%. In terms of longitudinal performance, HPV16/18 alone showed a sensitivity of 58.2%, compared to 94.6% with the extended panel35. Additionally, a study conducted among cytology-negative women in China reported that HPV types 52, 58, and 33 demonstrated comparatively high sensitivity in detecting CIN2+36. Furthermore, in our study, the absolute cross-sectional risk and 3-year longitudinal risk (PPV) of CIN2+ in HPV16/18-positive individuals were as high as 29% and 43%, respectively, and the corresponding risk was higher in women under 45 years compared to older women. The cross-sectional risk was 9.95% in the HR-HPV-positive and ASC-US+ cases, and the longitudinal risk was 19.66%, respectively. Consequently, HPV16/18-positive individuals have the highest immediate and progressive risks37,38. These women should be promptly referred to colposcopy, especially younger women. In contrast, other HR-HPV-positive individuals after cytological triage could be recommended for colposcopy referral or 12-month follow-up with repeat testing. This strategy could substantially reduce the burden on colposcopy services while being cost-effective12,28. Some European and American countries support the strategy39,40. Moreover, since cytological tests are more prone to false negatives than HPV tests—potentially delaying appropriate treatment—a negative HPV test result offers greater long-term reassurance against cervical cancer than a negative cytological diagnosis13,30.
Results from our prospective cross-sectional and longitudinal study supported the relative clinical significance of these tests in cervical cancer screening. Specifically, the immediate and 3-year absolute risks (PPV) of developing CIN2+ in the HPV-negative population were just 0.04% and 0.21%, respectively (for CIN3+, these risks were only 0.02% and 0.06%)-much lower than counterpart risks (0.28% and 1.22%) in the NILM population diagnosed by LBC (CIN3+: 0.11% and 0.56%). These findings indicated that HPV-negative women had significantly lower immediate and longitudinal risks of developing cervical cancer-related abnormalities, and suggested that HPV testing could allow more tailored and less frequent screening intervals, reducing unnecessary procedures and anxiety for patients, while also optimizing the use of healthcare resources15,28,41.
The study has several strengths. Its greatest strength is that it is a multicenter, large-scale, multi-method, and prospective longitudinal study that utilizes multiple HPV testing methods. Second, the strict quality control processes in cytological and pathological diagnostics ensured the accuracy of the results. Third, HPV testing and clinical diagnosis were performed in blind settings to minimize potential biases. However, there are some limitations. First, 81% of participants in the screening cohorts completed follow-up, a level of adherence that is common in real-world studies. For example, other studies evaluating the efficacy of HPV-based screening for cervical cancer prevention, including the NTCC, ART, and POBASCAM trials, reported follow-up completion rates of less than 75%13. Second, HPV-negative and NILM individuals were not referred for colposcopy, which may result in a potential missed diagnosis. However, double-negative individuals are less likely to develop CIN within 3 years42, and this strategy could greatly reduce the burden of colposcopy and be cost-effective. Moreover, double-negative individuals were tested for HPV and LBC testing in the fourth year.
In conclusion, our study demonstrates that HPV testing as a primary screening method provides reliable clinical performance for both cross-sectional and longitudinal cervical screening in accordance with Chinese guidelines. Our findings emphasize the importance of integrating HPV testing into routine screening programs, either as a supplement to or replacement for cytology, particularly for younger women. The use of validated HPV tests could significantly enhance the quality of population-level cervical cancer screening, supporting the WHO’s goal of screening 70% of women.
Methods
Ethical approval
The study was approved by the ethics committees of Cancer Hospital of Chinese Academy of Medical Sciences (CHCAMS) (No. 16-111/1190 and No. 20180920001), and Henan Cancer Hospital (No. 201655), and informed consent was obtained from all participants.
Study population and design
A total of 29,166 women, screened with one of three different HPV tests (HBRT-H14: 9980, DH3: 9698, and Aptima: 9488), aged 21–64 years, from nine centers in China (three centers per HPV test method) consented to participate in HPV-based cervical cancer screening. The inclusion criteria were as follows: (1) Women aged 21–64 years with an intact uterus; (2) No history of cervical cancer or precancerous lesions; (3) Willingness to undergo colposcopy and biopsy; (4) Not currently pregnant; women were eligible if at least 8 weeks postpartum; Exclusion criteria: Women with a history of hysterectomy or uterine ablation/resection. After excluding 665 (2.3%) ineligible participants, a total of 28,501 women (HBRT-H14: 9829, DH3: 9379, and Aptima: 9293) without gynecological issues were included at baseline. The sample size was estimated based on the cumulative 3-year incidence of CIN2+, which has been reported to range from 1.13 to 2.1% among women with normal baseline cytology.
A flowchart of the screening cohort enrollment is presented in Fig. 2. At baseline, all participants underwent both HPV testing and LBC. Women with positive baseline results were followed for 3 years, and in the final year, all participants were retested with both HPV and LBC. Briefly, screening recommendations at baseline and during follow-up were based on HPV testing and cytology results. Women who tested positive for HPV16/18 (or HPV16/18/45 for Aptima) were immediately referred to colposcopy, regardless of the cytological results. Similarly, women diagnosed with LSIL+ were immediately referred to colposcopy, irrespective of the HPV test results. Women who were HR-HPV positive with ASC-US were also referred for colposcopy. For follow-up, women with a positive HR-HPV test and normal cytology, ASC-US with a negative HR-HPV test at baseline, or a negative cervical biopsy (Normal or CIN grade 1 (CIN1)) were recommended for repeat cytology at 12 and 24 months. If follow-up cytology showed ASC-US+, a new colposcopy was performed. Women with negative HPV tests and normal cytology at baseline, as well as those diagnosed with CIN1 or less within the first 2 years, were recalled for screening in the fourth year and underwent LBC and HPV tests. The primary study endpoint was histologically confirmed CIN2+.
HPV human papillomavirus, LBC liquid-based cytology, HR-HPV High-risk HPV, LSIL+ low-grade squamous intraepithelial lesion or worse, ASC-US atypical squamous cells of undetermined significance, NILM negative for intraepithelial lesion or malignancy, CIN cervical intraepithelial neoplasia, CIN2+ CIN grade 2 or worse.
Sample collection
All participants first underwent gynecological examinations. Two cervical specimens were collected from each subject during the same visit, one sample was deposited in PreservCyt solution (Hologic, Inc., Bedford, USA) for cytological examination, and the other was placed in a tube containing cell preservation solution for HPV test (for HBRT-14 testing, Female Sample Collection Kit, Hybribio Biochemistry Ltd., Chaozhou, China; for DH3 testing, Specimen Preservation Solution, DALTONbio, Hangzhou, China; for Aptima testing, 1 mL specimen in PreservCyt solution was transferred to Aptima Specimen Transfer Kit, Hologic, Inc., Bedford, USA).
Liquid-based cytologic diagnosis
All of the diagnostic procedures were strictly double-blind. Based on the Bethesda 2014 classification, two cytologists reviewed and reported cytology slides; if the diagnoses were inconclusive, a third cytologist was consulted. LBC examination that identified ASC-US+.
HPV testing
All the samples were tested with HPV following the manufacturer’s instructions. HBRT-H14 is a TaqMan-based real-time PCR assay that targets the E6 and E7 regions of 14 HR-HPV types, allowing simultaneous separate genotyping of HPV16 and HPV18 from the 12 other HR-HPV types (HPV31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68) and the human β-globin gene43. If the cycle threshold (Ct) is ≤40 for both the human β-globin gene channel and at least one HPV type channel, the sample is considered HPV-positive.
The DH3 HPV assay is a liquid-based RNA–DNA hybrid capture technique that detects HPV16/18 and 12 additional HR-HPV types without nucleic acid amplification44. The 1.0 pg/mL cutoff value was used as the threshold for HPV positivity. The Aptima mRNA tests include two assays, Aptima mRNA assay and Aptima HPV 16 18/45 Genotype Assay. Aptima mRNA assay qualitatively detects the expression of HPV E6/E7 mRNA from all 14 HR-HPV types of HPV through real-time amplification. The Aptima HPV16 18/45 genotype assay detects HPV16 and 18/45 in cervical specimens from women with Aptima HPV assay-positive results. A signal/cutoff (S/CO) ratio of 1.0 was the recommended cutoff value. The FDA approves the Aptima HPV assays for screening women for HPV mRNA. HBRT-14 and DH3 are the two HPV DNA tests to receive National Medical Products Administration (NMPA) certification, according to China’s guidelines for HPV assay for cervical screening. In addition, the clinical performance of HBRT-H1443, DH345, and Aptima46 has been validated to be similar to that of HC2.
Pathological diagnosis
Biopsy or cervical scraping specimens obtained under colposcopy were sent for histological diagnosis to the Department of Pathology of Henan Cancer Hospital, CHCAMS, Guangdong Provincial Women and Child Health Hospital, Women’s Hospital at Zhejiang University School of Medicine, and Qilu Hospital at Cheeloo College of Medicine, Shandong University. The CIN grading reporting system was used for classification. Each diagnosis was reviewed by an experienced pathologist. In cases of disagreement between the initial diagnosis and review, the specimen was sent to CHCAMS for a final interpretation.
Statistics and reproducibility
In this study, all participants were not randomized and underwent both HPV testing and LBC. All medical personnel involved in the study, including gynecologists, cytotechnicians, and pathologists, were blinded to the HPV testing results to ensure unbiased evaluation. The sample size was estimated based on the cumulative 3-year incidence of CIN2+, previously reported to range from 1.13 to 2.1% among women with normal baseline cytology. According to the Guidelines for the Technical Review of HPV Nucleic Acid Detection and Genotyping Reagents in China, at least 60 CIN2+ cases are required by the end of follow-up. Using the lowest estimated incidence (1.13%), approximately 5310 women with NILM cytology (60/1.13%) would need to be enrolled. Assuming that approximately 83% of participants would have NILM cytology results and allowing for a 30% loss to follow-up, the initial recruitment target was set at around 9140 women. Considering additional practical factors, the final estimated sample size was approximately 9300 to ensure adequate power to detect the required number of CIN2+ cases. A total of 29,166 women consented to participate in HPV-based cervical cancer screening and were screened using one of three HPV assays (HBRT-H14: 9980; DH3: 9698; and Aptima: 9488). After applying the inclusion and exclusion criteria, 665 participants (2.3%) were excluded, leaving 28,501 women for baseline analyses. The study design is described in detail in the “Study population and design” section.
Histologically confirmed CIN2+ and CIN3+ were used as study endpoints. The total number of CIN2+ and CIN3+ cases was calculated for both cross-sectional settings (baseline screening) and longitudinal settings (both baseline and follow-up screenings). CIN2+ or CIN3+ cases diagnosed in the first year were classified as cross-sectional cases. CIN2+ or CIN3+ cases diagnosed between the first and the fourth year were classified as longitudinal cases. In the longitudinal analysis, since CIN2 is typically treated, only participants without prior CIN2 were included in the CIN3+ group. We calculated sensitivity, specificity, PPV (absolute risk), and NPV for LBC and HPV testing (HBRT-H14, DH3, and Aptima) in detecting CIN2+ and CIN3+ lesions. The 95% CIs for sensitivity, specificity, PPV, and NPV were calculated using the Wilson score method. Analyses were conducted for the overall population and stratified by age groups (≤35, 36–45, and >45 years). Considering the balance of sample sizes, the Chi-square test was used to compare the differences in clinical performance between the two groups (≤45 vs. >45 years). Since HPV45 prevalence in the Chinese population was only 0.7%47, we analyzed the HPV16/18 genotyping, including HPV16/18 detected by three assays and HPV45 detected by Aptima. Primary HPV screening with LBC triage referred to HPV16/18 positive, and other HR-HPV positive triaged by LBC diagnosis to find ASC-US+, while LBC screening with HPV triage was defined as LSIL+ diagnosed by LBC, and ASC-US triaged by HPV testing that found HR-HPV positive. We also estimated the relative sensitivity and specificity of HPV testing to LBC. The 95% CIs for these ratios were calculated using the log-transformed (Wald) method. A two-tailed McNemar’s (McN) test was conducted to compare differences between HPV and LBC. The level of statistical significance was set at 0.05. All statistical analyses were performed using SPSS (version 18.0, IBM Corp). As this was a population-based screening study, the detections were not independently replicated or repeated. However, the results are expected to be reproducible under similar study conditions, and the direction and magnitude of the observed associations are unlikely to change.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Source data for Fig. 1 are provided with this paper. Public sharing of the complete dataset is restricted by national regulations governing medical data. The data can be made available from the authors upon reasonable request, subject to compliance with applicable laws and institutional ethical approvals. Source data are provided with this paper.
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263 (2024).
Gu, X. et al. Incidence and mortality of cervical cancer in China in 2015. J. Natl. Cancer Cent. 2, 70–77 (2022).
Zhang, S. et al. Cancer incidence and mortality in China, 2015. J. Natl. Cancer Cent. 1, 2–11 (2021).
Small, W. Jr. et al. Cervical cancer: a global health crisis. Cancer 123, 2404–2412 (2017).
Zhao, F. & Qiao, Y. Cervical cancer prevention in China: a key to cancer control. Lancet 393, 969–970 (2019).
Zhao, Y. X. et al. [Analysis of the reported data of National Cervical Cancer Screening Program in Rural Areas in China from 2009 to 2018]. Zhonghua Yi Xue Za Zhi 101, 1863–1868 (2021).
Cuzick, J. et al. Overview of the European and North American studies on HPV testing in primary cervical cancer screening. Int. J. Cancer 119, 1095–1101 (2006).
Koliopoulos, G. et al. Cytology versus HPV testing for cervical cancer screening in the general population. Cochrane Database Syst. Rev. 8, CD008587 (2017).
Zhao, F., Lang, J., Qiao, Y. & Zhu, L. How can China achieve WHO’s 2030 targets for eliminating cervical cancer? BMJ 386, e078641 (2024).
Arbyn, M. et al. Evidence regarding human papillomavirus testing in secondary prevention of cervical cancer. Vaccine 30, F88–F99 (2012).
Arbyn, M. et al. Accuracy and effectiveness of HPV mRNA testing in cervical cancer screening: a systematic review and meta-analysis. Lancet Oncol. 23, 950–960 (2022).
Bhatla, N. & Singhal, S. Primary HPV screening for cervical cancer. Best Pract. Res. Clin. Obstet. Gynaecol. 65, 98–108 (2020).
Ronco, G. et al. Efficacy of HPV-based screening for prevention of invasive cervical cancer: follow-up of four European randomised controlled trials. Lancet 383, 524–532 (2014).
Wright, T. C. et al. Primary cervical cancer screening with human papillomavirus: end of study results from the ATHENA study using HPV as the first-line screening test. Gynecol. Oncol. 136, 189–197 (2015).
Bulkmans, N. W. et al. Human papillomavirus DNA testing for the detection of cervical intraepithelial neoplasia grade 3 and cancer: 5-year follow-up of a randomised controlled implementation trial. Lancet 370, 1764–1772 (2007).
World Health Organization. Reaching 2030 cervical cancer elimination targets—new WHO recommendations for screening and treatment of cervical pre-cancer. Available from: https://www.who.int/news-room/events/detail/2021/07/06/default-calendar/reaching-2030-cervical-cancer-elimination-targets. Accessed 6 July 2021.
Arbyn, M. et al. 2020 list of human papillomavirus assays suitable for primary cervical cancer screening. Clin. Microbiol. Infect. 27, 1083–1095 (2021).
Center for Medical Device Evaluation. NMPA. Guidelines for the technical review of human papillomavirus (HPV) nucleic acid detection and genotyping reagents. Available from: https://www.cmde.org.cn//flfg/zdyz/zdyzwbk/20170405151300482.html. Accessed 5 April 2015.
Kang, L. N. et al. A prospective study of age trends of high-risk human papillomavirus infection in rural China. BMC Infect. Dis. 14, 96 (2014).
Yin, X. et al. HPV prevalence and distribution characteristics in postmenopausal women from Nanjing, China. BMC Women’s Health 24, 68 (2024).
Han, S. et al. Prevalence, trends, and geographic distribution of human papillomavirus infection in Chinese women: a summative analysis of 2,728,321 cases. BMC Med. 23, 158 (2025).
Yao, B. et al. Real-world effectiveness of cytology and HPV-based screening strategy in cervical cancer screening: a cross-sectional population-based study in Chengdu, China. PLoS ONE 19, e0299651 (2024).
Chen, J. et al. Estimated human papillomavirus vaccine coverage among females 9-45 years of age—China, 2017-2022. China CDC Wkly 6, 413–417 (2024).
Zhang, M. et al. Cervical Cancer Screening Coverage - China, 2018-2019. China CDC Wkly. 4, 1077–1082 (2022).
Gonzalez, P. et al. Behavioral/lifestyle and immunologic factors associated with HPV infection among women older than 45 years. Cancer Epidemiol. Biomark. Prev. 19, 3044–3054 (2010).
Kulasingam, S. L. et al. Evaluation of human papillomavirus testing in primary screening for cervical abnormalities: comparison of sensitivity, specificity, and frequency of referral. JAMA 288, 1749–1757 (2002).
Chatzistamatiou, K., Moysiadis, T., Moschaki, V., Panteleris, N. & Agorastos, T. Comparison of cytology, HPV DNA testing and HPV 16/18 genotyping alone or combined targeting to the more balanced methodology for cervical cancer screening. Gynecol. Oncol. 142, 120–127 (2016).
Torres-Ibarra, L. et al. Comparison of HPV-16 and HPV-18 genotyping and cytological testing as triage testing within human papillomavirus-based screening in Mexico. JAMA Netw. Open 2, e1915781 (2019).
Cox, J. T. et al. Comparison of cervical cancer screening strategies incorporating different combinations of cytology, HPV testing, and genotyping for HPV 16/18: results from the ATHENA HPV study. Am. J. Obstet. Gynecol. 208, 184 e181–184.e111 (2013).
Ostrbenk Valencak, A. et al. Clinically validated HPV assays offer comparable long-term safety in primary cervical cancer screening: a 9-year follow-up of a population-based screening cohort. Int. J. Cancer 156, 788–801 (2025).
Dillner, J. et al. Long term predictive values of cytology and human papillomavirus testing in cervical cancer screening: joint European cohort study. BMJ 337, a1754 (2008).
Ronco, G. et al. Results at recruitment from a randomized controlled trial comparing human papillomavirus testing alone with conventional cytology as the primary cervical cancer screening test. J. Natl. Cancer Inst. 100, 492–501 (2008).
Safaeian, M., Solomon, D. & Castle, P. E. Cervical cancer prevention—cervical screening: science in evolution. Obstet. Gynecol. Clin. North Am. 34, 739–760, ix (2007).
Zhou, Q. et al. The clinical performance of different cervical cancer screening strategies in premenopausal and postmenopausal women. Menopause 32, 640–647 (2025).
Del Mistro, A. et al. Human papilloma virus genotyping for the cross-sectional and longitudinal probability of developing cervical intraepithelial neoplasia grade 2 or more. Int. J. Cancer 143, 333–342 (2018).
Bai, A., Xue, P., Li, Q., Jiang, Y. & Qiao, Y. Diagnostic value of high-risk HPV other than type 16/18 in high-grade cervical neoplasia among cytology-negative women: a multicenter retrospective study. Cancer Med. 12, 14794–14805 (2023).
Schiffman, M. et al. A cohort study of cervical screening using partial HPV typing and cytology triage. Int. J. Cancer 139, 2606–2615 (2016).
Agorastos, T. et al. Primary screening for cervical cancer based on high-risk human papillomavirus (HPV) detection and HPV 16 and HPV 18 genotyping, in comparison to cytology. PLoS ONE 10, e0119755 (2015).
Cuschieri, K. et al. Eurogin roadmap 2017: triage strategies for the management of HPV-positive women in cervical screening programs. Int. J. Cancer 143, 735–745 (2018).
Workowski, K. A. et al. Sexually Transmitted Infections Treatment Guidelines, 2021. MMWR Recomm. Rep. 70, 1–187 (2021).
Lobin, C. et al. Cost-effectiveness analysis of alternative screening strategies for the detection of cervical cancer among women in rural areas of Western Kenya. Int. J. Cancer 155, 1257–1267 (2024).
Chao, A. et al. Incidence and outcome of acquisition of human papillomavirus infection in women with normal cytology—a population-based cohort study from Taiwan. Int. J. Cancer 126, 191–198 (2010).
Xu, L., Ostrbenk Valencak, A., Poljak, M. & Arbyn, M. Evaluation and optimization of the clinical accuracy of Hybribio’s 14 high-risk HPV with 16/18 genotyping assay within the VALGENT-3 Framework. J. Clin. Microbiol. 58, e00234-20 (2020).
Zhao, X. et al. The performance of human papillomavirus DNA detection with type 16/18 genotyping by hybrid capture in primary test of cervical cancer screening: a cross-sectional study in 10,669 Chinese women. Clin. Microbiol. Infect. 24, 1322–1327 (2018).
Fu, Y. et al. Head-to-head comparison of DH3 HPV test and HC2 Assay for detection of high-risk HPV infection in residual cytology samples from cervical cancer screening setting: baseline and 3-year longitudinal data. Microbiol. Spectr. 10, e0157021 (2022).
Cook, D. A. et al. Aptima HPV Assay versus Hybrid Capture((R)) 2 HPV test for primary cervical cancer screening in the HPV FOCAL trial. J. Clin. Virol. 87, 23–29 (2017).
Zhong, F. et al. HPV genotyping of cervical histologic specimens of 61,422 patients from the largest women hospital in China. Front. Oncol. 13, 1161631 (2023).
Acknowledgements
This study was supported by the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (CIFMS 2021-I2M-1-004), with grant ownership attributed to Y.L.Q., W.C., and F.H.Z. The authors are grateful for the support and cooperation of all the study participants.
Author information
Authors and Affiliations
Contributions
Study concepts and study design: J.Y., Y.L.Q., and W.C; clinical management and data collection: S.K.Z., Z.F.L., W.C., X.P.L., X.D.C., B.H.K., X.B.S., S.M.W., and J.Y.; quality control data and algorithms: W.C., X.Z., Q.J.P., J.Y., F.H.Z., and Y.L.Q.; data analysis, interpretation, statistical analysis, and manuscript preparation: J.Y.; manuscript review: Y.L.Q.; all authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Sveinung Wergeland Sørbye and the other anonymous reviewer(s) for their contribution to the peer review of this work. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yin, J., Zhang, S., Wang, S. et al. Age-specific clinical performance of HPV-based vs. cytology-based cervical cancer screening in China. Nat Commun 17, 521 (2026). https://doi.org/10.1038/s41467-025-67215-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-67215-4