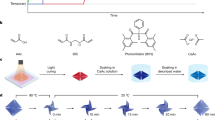
Abstract
Shape-changing smart substrates are revolutionizing implantable bioelectronics by offering minimal invasiveness and self-adaptive geometric conformity. However, precise manipulation of shapeshifting, which is critical for the successful implantation, are hindered by triggering accessibility and cumbersome customization. Here we introduce a digital 4D printing technique for fabricating customized shape memory hydrogel substrates enabling trigger-free shapeshifting with predefined onset periods. By controlling spatio-temporal light exposure, pixelated polymer sheets with heterogeneous networks are obtained, yielding intricate 3D original shapes after monomer removal. Meanwhile, spatially controlled onset time of the programmable shape recovery is introduced, attributed to local variation of polymeric network structure and the corresponding thermally-induced phase separation. This approach allows the complex 3D objects to execute autonomous multi-stage shape recovery through a single-step digital exposure process, which is not offered by existing techniques. In addition, the material exhibits negligible inflammation proven by in-vivo testing of male mice experiments. Demonstrated with self-winding bioelectronics, this method advances time-dependent adaptability of implantable devices and expands the functional scope of bioelectronics.
Similar content being viewed by others
Introduction
Soft implantable bioelectronics are emerging for various in vivo applications, exemplified by neural electrodes1,2,3,4, gastrointestinal robots5,6, and cardiac pacemakers7,8. Since they can interact directly with tissues and organs, these devices represent a transformative approach for advanced functionalities such as precise biosensing9,10,11, in-situ stimulation7,12,13, and long-term therapeutic interventions14,15. To minimize invasive implantation and maximize signaling precision, smart soft substrates which can execute shapeshifting on demand are innovated to support the electronics3,16,17,18,19,20,21. Specifically, shape memory polymers enable implantation in a temporarily tiny shape through micro incisions, and subsequently expand or morph in vivo upon stimuli to conform the tissues for improved detection.
Two aspects are critical for high-quality shape-memory substrates, including construction of the permanent shapes and control of the shapeshifting pathways. On one hand, permanent geometries are supposed to match the contour of the target tissue18,22. Given that bio interfaces are versatile and personal, 3D shaping and customization of the substrates turn out to be important. On the other hand, a precisely-controlled shapeshifting pathway can promise the substrate to work right in the target position without unexpected deployment3,20. Importantly, the two aspects are expected to be achieved simultaneously to accomplish the conforming task.
As for shape customization, 3D printing is a primary candidate. However, the layer-by-layer stacking mode is commonly time-consuming23. In addition, fabrication of 3D thin sheets, which are desired for the soft substrates, is usually challenging for most 3D printing techniques due to the resolution constraint and the supporting complexity. In contrast, a special 4D printing technique, which constructs monolayer 3D shapes via spatial stress release of 2D thin sheets with heterogenous network structure, has been born as a facile yet effective approach to realize fast shaping24,25,26,27,28,29,30,31,32. Meanwhile, this technique can be adopted to various responsive polymers those can perform shapeshifting upon specific stimuli such as temperature, light, electricity, magnet, and chemical compounds. However, to obtain an ideal 3D smart substrate is still challenging, since the accessibility to the stimuli remains problematic.
Recently, some hydrogels capable of reversible thermally-induced phase separation exhibit a unique shape memory effect33. They can be programmed and fixed in temporary shapes by hot water. Upon cooling to ambient temperature, the materials do not recover before certain onset periods controlled by the phase evolution kinetics. This unique behavior can apparently achieve stimuli-free and timed shapeshifting, since the recovery process begins autonomously in a constant environmental condition.
Herein, we report our progress of the 4D printing method for the shape memory hydrogels with the programmable recovery onset. A single planar digital exposure enabled simultaneously the effortless customization of complex geometries and the creation of multiple self-morphing pathways, which is a tough yet a desired task. The relationship between the photo curing kinetics, the macroscopic shape geometry, and the phase separation behavior was established. Implantable self-winding electrodes were then fabricated, further showing the merits of these smart 3D substrates. It advances the scientific understanding for responsive hydrogels with reversible phase separation as well as the practical toolkit for programmable and adaptive substrates.
Results
Figure 1a illustrates the 4D printing process. Precursor solution containing 78.7 wt% monomer (acrylate acid, AAc), 0.4 wt% crosslinker (N,N′-methylenebisacrylamide, BIS) aqueous solution with 1.0 wt% photo initiator and 0.1 wt% biocompatible dye was sandwiched between two slides and then digitally irradiated by a projector. Herein, 1.0 wt% concentration of photo initiator is chosen herein for its highest irradiation efficiency (Supplementary Fig. 1) and the biocompatible dye contributes to improving printing precision34. Notably, the most critical parameter to realize 4D printing is the irradiation time, which is denoted as tirr throughout the investigation to ensure clarity. The exposed side is defined as the front, and the other side is the back. A curing gradient was generated within the PAAc sheet due to light attenuation across its thickness (set as 0.3 mm unless specially noted), which is further enhanced by the biocompatible tartrazine dye. Unreacted monomers inside the sheet were removed when it was immersed in 1 M calcium acetate (CaAc) solution, driving the 2D sheet into a 3D shape due to the non-uniform volume change. At the same time, additional ionic crosslinking was formed along with the calcium coordinate complexation. As expected, the target PAAc-CaAc hydrogel not only owned complex shapes, but also exhibited shape memory behavior with unique recovery onset time (Fig. 1b). They can be deformed at 25 °C in a soft and homogeneous state, and the deformed shapes can be temporarily fixed at 90 °C due to the hardening effect raised by thermal-induced phase separation. After the heat treatment for a certain time, the temporary shapes do not recover at once upon stimulus (cooling to room temperature here) like common shape memory polymers. Instead, these temporary shapes maintain stably for certain onset periods (referred as tonset) before the hydrogels change to their original shapes, apparently providing a trigger-free autonomous manner. Mechanistically, this behavior is driven by internal mass diffusion that the phase-separated polymer chains are solvated and activated via water redistribution during the phase evolution. Further discussion conducted later.
a Scheme of the 4D printing for facile fabrication of 3D shapes via digital exposure and ionic crosslinking. b Trigger-free shape recovery process of the hydrogel at 25 °C after programming at 90 °C.
Geometrical control for fabricating the 3D original shapes was first investigated. A rectangular one-side exposure was chosen as the experimental model to clarify the influence of tirr on the outcome geometry (Fig. 2a). Specifically, conversion of the double bond representing the polymerization degree was measured using Fourier transform infrared (FTIR) spectra with the ratio of the vinyl peak at 980 cm−1 to the standard carbonyl peak at 1705 cm−1 (Supplementary Fig. 2)35. It shows that the conversion on both sides increases with tirr to construct a more complete network. Longer tirr resulted in higher conversion on both sides of the PAAc sheet (exceeding 85% after 300 s), which led to a denser network. This is consistent with the overall mass loss from the removal of AAc monomers (Supplementary Fig. 3). The conversion contrast between the two sides decreased over time, which exhibits a maximum value of 22% at 80 s and reduces to only 0.87% at 300 s. Unlike 4D printing of dry polymers, whose shapes are predominantly determined by monomer loss and associated volume shrinkage26,30,31, hydrogels undergo a more complex case due to the combined effects of water swelling and monomer loss. In particular, a looser network synthesized upon shorter exposure time not only leads to greater monomer loss but also results in increased water uptake. Both effects were analyzed here to identify the dominant factor. The volume ratio of the target PAAc-CaAc gel relative to the cured PAAc sheet changes from 1.1 at 80 s to 2.5 at 300 s, while the water content of the target gel shows negligible difference at around 45% (Supplementary Fig. 4). Therefore, monomer loss is considered as the dominating factor during the 2D-to-3D shape transformation.
a Double bond conversion curves of the front or back side and calculated conversion contrast (∆conversion). b Representative macroscopic images of hydrogel strips with rectangle irradiation patterns in size of 4.5 mm × 13.5 mm × 0.3 mm. This experiment was independently repeated three times with similar results. c Curvature alteration of the strips undergone various exposure time. d A hinge model to investigate the double-side irradiation, with b, t, θ corresponding to the width of middle rectangle, tirr and the bending angle of the hinge, respectively. The length of a was fixed at 4.5 mm. e Folding angles upon different tirr and irradiation width. f Folding angles of the samples with different thickness (b = 0.9 mm, t = 120 s). g Exposure patterns and photographs of the resulted 3D hydrogels. All scale bars are 5 mm.
Figure 2b shows that under one-sided irradiation of rectangular patterns, the curvature κ of the resulted PAAc-CaAc hydrogels decreases with time. tirr of 80 s and 100 s produces a significant gradient, forming spiral shapes along the film’s width instead of circular shapes. Extended irradiation reduced the κ from 7.2 cm−1 to 0.46 cm−1 at 300 s (Fig. 2c). Besides, irradiation for less than 80 s was insufficient to generate an intact film, because the practical energy delivered to the precursor did not reach the critical exposure threshold (Supplementary Fig. 5). While 300 s caused pattern divergence due to light scattering despite the addition of photo-absorber to minimize the scattering. On this basis, tirr between 80 s and 240 s were selected for further investigations. Additionally, increasing the film thickness from 0.1 mm to 0.5 mm enhanced the structure gradients, resulting in larger curvature (Supplementary Fig. 6).
To further enhance the shaping complexity, double-side irradiation setup was built. A hinge model was used to investigate the influencing factors. Two square regions were irradiated from both sides for 120 s, while the connecting middle region was exposed from one side for a variable time t (Fig. 2d). In this configuration, only the rectangular middle region develops a gradient, driving the sheet to form a hinge with a folding angle (θ). Unlike the curling process in Fig. 2a, the middle region would curl along the x direction, as the y direction was confined by the non-actuated areas (Supplementary Fig. 7 and Supplementary Fig. 8). Consequently, the middle width (b) primarily determines the driving proportion. A larger b and shorter t produced more pronounced bending. The resulting θ can range from 0° to 180° depending on the control of b, t, or material thickness (Fig. 2e, f).
On this basis, elegant exposure patterns were designed to induce spatial gradient heterogeneity, enabling the fabrication of complex 3D hydrogel structures (Fig. 2g). In these designs, the chessboard patterns represented the double-side exposure areas, while the grayscale patterns corresponded to varying one-sided tirr, not relative to other parameters like sample thicknesses. To ensure precise shaping at a small scale, the minimum pattern resolution was set to 200 μm (Supplementary Fig. 9). It is noted that, although the presented 4D printing method is not suitable to some types of 3D structures (e.g. lattice structures), it still provides two advantages in comparison with conventional 3D printing techniques. First, the preparation is fast since it only needs a single-layer light exposure. Second, complex 3D curved structures as exemplarily shown in Fig. 2g are exactly ideal to serve as substrates for flexible bioelectronics, enabling continuous curvatures that can achieve conformal match with tissue contours. They are in fact challenging to be fabricated via layer-by-layer 3D printing due to the poor self-supporting of the thin-walled structure.
It is known that polymer network structures, which can be regulated by crosslinker content or precursor concentration, significantly influence the phase separation behavior and the corresponding tonset before shapeshifting33. A looser network, which provides a larger characteristic size of phase separation upon heating, leads to a longer tonset. Extending to the current case, the tirr would influence the polymeric conversion degree, which not only determines the macroscopic geometries as analyzed before, but also simultaneously change the network chemical structures to induce the variation of the tonset (Fig. 3a). Mechanistically, increasing the tirr leads to higher conversion on both front and back sides, which constructs a denser network with less gradient (Fig. 3b). This weakens the phase separation and reduces the phase domain sizes attributed to the dense crosslinking structure. Consequently, the tonset decreases with the tirr due to the shortened internal water diffusion path. The shape recovery kinetics of the printed samples at 25 °C were measured to verify our hypothesis (Fig. 3c). As the irradiation extended from 80 s to 240 s, the tonset decreased from 7.4 min to 2.5 min (Fig. 3d). Microscopic phase separation states before (Fig. 3e) and after the heating programming (Fig. 3f) were characterized by SEM, which supports the results from a morphology perspective. Before the heating, the hydrogels undergone one-sided irradiation were loosely crosslinked at the back and densely at the front layer, reflected respectively by the large and small pores. The contrast diminishes until 240 s irradiation is applied. After the heating, the statistics pore size enlarges for the both sides. These results are generally consistent with the hypothesis in Fig. 3b. DSC curves show that the enthalpy decreases with the tirr which also implies the reduction of the overall phase separation degree (Supplementary Fig. 10). In addition, the tonset can be tuned in a large time window by varying the heating times from 10 s to 180 min (Supplementary Fig. 11). Specifically, the tonset ranged from 3 to 65 min for 80 s irradiation and 1 min to 21 min for 240 s irradiation, and all the shapes exhibited full recovery. Notably, long tonset is beneficial to complex surgical scenarios requiring operational precision, which ensures precise navigation and positioning processes without premature activation.
a The predictive relationship among the tonset, the conversion, and the tirr. b Illustration showing the phase separation behavior for the hydrogels irradiated by different time. c Shape recovery kinetics of the hydrogels printed by different tirr. Before recovery at 25 °C water, all the samples were folded at 90 °C water for 1 min. d The tonset of the hydrogels synthesized by different tirr. Data are presented as mean values +/- SD with n = 3 sample replicates. e SEM images of the samples before the heating programming. f SEM images of the samples quenched immediately after the heating programming in 90 °C water for 1 min. These images are representative of results from three tested samples. All the scale bars are 1 μm.
After separate investigation on the controllability of the 3D geometries and the tonset, the combined performance was studied. First, a Miura origami structure was fabricated using a simple digital exposure with all hinge areas irradiated for 120 s. After programming at 90 °C for 1 min, the structure was fixed in a compressed state with hinge closure. It presented a single-step shape-shifting at room temperature driven by the uniform tonset of the hinge (Fig. 4a and Supplementary Movie 1). More complex shape-shifting pathways can be achieved by setting multiple onsets in different regions. To illustrate this, pyramid (Fig. 4b (i)) and windmill (Fig. 4b (ii)) structures were chosen for the investigation. After the shape programming as shown in the upper part of Fig. 4b, designated areas irradiated for 120 s on both sides and 80 s on one side were temporarily fixed. Due to distinct tonset for the two regions, the samples began the first recovery stage after 2 min, as indicated by the blue arrows corresponding to the 120 s irradiation regions. After 10 min, the secondary recovery initiated at the 80 s irradiation regions, as indicated by the green arrows (Supplementary Movies 2 and 3). These results verified that by varying tirr regionally, 3D geometries with multiple autonomous shape-recovery stages could be achieved without additional triggers. Accordingly, small spiral connector enabling multi-shape recovery was fabricated, which is difficult for conventional molding or 3D printing (Fig. 4c). This connector was inserted as a flat strip between two separate rubber bands (mimicking soft tissues) during the tonset. After the first tonset, it curved to envelop the bands to achieve the linking. Sequentially, the two ends autonomously climbed over the bands, further solidifying the connection (Supplementary Movie 4). This demonstration shows the potential of the 4D printed parts for autonomous connecting of soft tissues or other bioelectronics in vivo.
a Single-step shape recovery of a Miura origami structure with a tonset of 2 min. The sample was programed at 90 °C for 1 min. b Dual-step recovery with two separate tonset. The chess board patterns relate to double-side exposure of 120 s. c A spiral connector with multiple tonset for bridging. All the scale bars are 5 mm.
With similar autonomous shapeshifting behavior under body temperature (Supplementary Fig. 12), low cell toxicity (Supplementary Fig. 13 and Fig. 14), and negligible inflammation (Supplementary Fig. 15 and Table 1), we hereafter demonstrate the application potential of the 3D materials as the substrates of bioelectronics. Self-winding electrodes for electromyography (EMG) of extraocular muscle were fabricated, which can provide valuable insights into the etiology and inform treatment strategies for ophthalmic diseases caused by disorders of extraocular muscles (e.g., myasthenia gravis and strabismus). Softness and conformality of EMG electrodes are critical for the signal quality, since they can reduce interfacial impedance to enable more accurate recordings. 4D-printed hydrogel electrodes were implanted on the surface of the extraocular muscle in a rabbit model, which provides the desired low modulus and spiral geometry for this application. As shown in Fig. 5a, the electrodes are inserted in a rigid state through the tiny gap under the muscle, and afterwards undergo an autonomous winding to wrap around the muscle (Fig. 5b). The necessity of the sequential shape transformation enabled by the patterning of multiple tonset was validated (Fig. 5c). Unlike a hydrogel electrode with uniform recovery onset that often fails in entire contact (Supplementary Movie 5), the sequential winding ensures smooth conforming to the target (Supplementary Movie 6). A liquid metal layer that consisted of EGaIn alloys and Ag particles was then deposited onto the 3D-curved hydrogel via post-printing to impart conductivity29 (Fig. 5d). With the thickness of conductive layer of 20 μm (Supplementary Fig. 16), the conductivity was stabilized at 1.25 × 106 S/m. Notably, the tonset ensured that the 3D shape can be fixed into a flatten state for the printing of the circuit, showing the merit of the shape memory effect. On this basis, two spiral hydrogel electrodes were successfully fabricated and wrapped around the obliquus inferior muscle (Fig. 5e). Action potentials were recorded in response to stimulation with square-wave currents ranging from 0.3 mA to 1 mA at a frequency of 15 Hz and a wave width of 1 ms. The amplitude of the recorded potentials increased with the applied current, indicating successful recording of the muscle electrophysiology (Fig. 5f). Such EMG measurements, enabled by soft and conformal 4D hydrogels, demonstrate excellent adaptability to various individuals (Supplementary Fig. 17) without noticeable tissue alternations (Supplementary Fig. 18). In addition, the electrodes can also function as stimulation ends to induce muscle contraction (Supplementary Movie 7). These results suggest that our approach could pave the way for the development of highly integrated 3D hydrogel devices. By combining high-fidelity monitoring and programmable stimulation in a single minimally invasive platform, the self-winding electrode is believed to be a potent tool for managing complex both ophthalmological and neurological diseases.
a Scheme of stimulation and potential recording of the extraocular muscle. b The sequential winding process of a hydrogel electrode. c Comparison between single-step and sequential recovery. d Circuit printing for fabricating a 3D electrode. e Photos showing the twining process of the spiral electrodes via the self-winding. f Action potential of the extraocular muscle under different stimuli currents, which is the representative result with n = 3 rabbit. All the scale bars are 5 mm.
A photopatterning-based 4D printing technique was developed in this work to fabricate 3D structures of trigger-free shapeshifting hydrogels. By controlling the tirr during the photocuring, the conversion gradient can be finely tuned, which determines the formation of the permanent 3D shapes after removal of the unreacted monomers. Simultaneously, the tonset of the shapeshifting can also be manipulated by the tirr, since the varied network structure alters the phase separation behavior of the hydrogel upon heating. Multi-stage autonomous shape recovery was further realized through simply adjusting the tirr for designated locations, which is a noteworthy progress for multi-shape morphing materials. The produced hydrogels are desired candidates as smart substrates for flexible bioelectronics due to the conformable 3D structure and the multi-shape morphing. Demonstrative devices such as autonomous spiral connectors and self-winding electrodes were fabricated, showing potential efficacy in complex scenarios. For perspective, how to realize decoupling of the tonset and the 3D shape formation requires exploration to further enhance the design flexibility.
Methods
Materials
Acrylic acid (AAc, purity>99%, lot#A800293), calcium acetate (CaAc, purity of 99%, lot# C850055), and bis(2,4,6-trimethylbenzoyl)-phenylphosphineoxide photo-initiator (Irgacure 819, purity of 98%, lot#P831909) were purchased from Macklin. N,N′-methylenebisacrylamide (BIS, purity of 99%, lot#146072) was obtained from Sigma-Aldrich. Tartrazine dye (purity of biological stain, lot#R094359) was provided by Rhawn. Semi-liquid metals (Semi-LM) were acquired from Shenyang Jiabei Trading Co., Ltd, by mixing and grinding EGaIn alloys with 33.3 wt% of Ag particles (average particle size of 2 μm, purity of 99.5%).
Preparation of the hydrogel precursor
14.42 g (0.2 mol) AAc monomer, 0.077 g (0.0005 mol) BIS crosslinker, and 0.20 g (0.0005 mol) photo-initiator Irgacure 819 were solved in 3.6 g DI water with magnetic stirring. 0.018 g tartrazine dye was added to the precursor for photo absorbing to create desirable polymerization gradient along the thickness direction and promise the planar resolution of the photopatterning. The precursor was stored in the dark at room temperature before use.
4D printing of the hydrogel
First, the hydrogel precursor including 78.7 wt% AAc, 0.4 wt% BIS, 1.0 wt% photo initiator and 0.1 wt% biocompatible dye was poured into a self-made mold consisting of two slides and a silicon rubber spacer. The width and length of the spacer were set as 1 cm and 2 cm, respectively. The thickness of spacer varied from 0.1 mm to 0.5 mm, in which 0.3 mm being used for most investigations unless otherwise noted. Afterwards, the precursor was irradiated for specific curing time (tirr) by a commercial projector (DELL1609WX with 10 mW/cm2 intensity) with one-side or double-side exposure patterns under ambient room environment. The obtained film was immersed in 1 M calcium acetate solution (pH of 7) for two days followed by three days of DI water for removal of the unreacted monomers and excess calcium acetate to get a 3D sample. Afterwards, the hydrogels can undergo shape memory process enabled by phase separation mechanism, whose characterization methods were explicated in the following section. The combination of 3D fabrication process with programmable shape memory behavior was identified as 4D printing. Notably, hydrogel samples with rectangular irradiation (size of 4.5 mm × 13.5 mm) were selected as standard models to investigate the geometrical controlling factors, whose curvatures were calculated as the reciprocal of the arc radius. The hydrogel electrode was fabricated by applying the semi-liquid metal (EGaIn alloys with 33.3 wt% of Ag particles) through a polyethylene mask onto the hydrogel surface at its flattened temporary shape.
Investigation of the shape memory behavior
Hydrogel samples made by different one-side exposure times (size of 4.5 mm × 13.5 mm × 0.3 mm) were folded into half by tweezers at 25 °C water and fixed at 90 °C water bath for a certain period. Afterwards, the fixed samples were transformed to 25 °C or 37 °C water to conduct shape recovery process. The whole shape recovery process was recorded by a digital video camera (Sony HDR-TD30E). The shape recovery ratio at a certain time was calculated by the ratio of the real-time angle extracted from the video and the overall deformation angle (180°). The onset period was defined as the time of reaching 5% shape recovery ratio.
Measurement of the double bond conversion
The polymerization conversion was characterized by double bond conversion measurement. The hydrogel precursor in a self-made mold with the size of 1 cm*1 cm*0.3 mm was irradiated from one side for different times to obtain cured hydrogel sheets with different degrees of conversion. The liquid precursor and all cured hydrogel samples were then analyzed using a Nicolet iS50 FTIR spectrometer in ATR mode. Notably, each cured hydrogel was measured on both the front and back surfaces to obtain the corresponding Fourier transform infrared (FTIR) spectra. The peak ratio between the vinyl group (980 cm–1) and the carbonyl group (1705 cm–1) was calculated as the double bond conversion αvinyl,t = 1 – (Avinyl/Acarbonyl)t/(Avinyl/Acarbonyl)t=0, where t is the light exposure time. The conversion contrast between the two sides was defined by Δα = (αfront-αback).
SEM morphology observation
The hydrogel samples before and after thermal programming were quenched by liquid nitrogen, followed by lyophilization to extract the water. The cross-section morphology of the samples was observed by scanning electron microscopy (SU-8010 SEM by Hitachi) at an acceleration voltage of 3 kV.
Measurement of the mass variation
Weight of the pristine cured PAAc sheet (size of 4.5 mm × 13.5 mm × 0.3 mm) was noted as mt, where t is the light exposure time. These PAAc sheets were immersed in water for 12 h until a fully swollen state to remove the unreacted monomers, followed by drying process in the oven of 70 °C for another 12 h to remove the water completely. Their weights in a fully swollen state and drying state were measured as mt(wet) and mt(dry), respectively. The mass loss was calculated as (mt- mt(dry))/mt × 100%, and water content was calculated as (mt(wet)-mt(dry))/mt(wet) × 100%. The volume expansion ratio of the PAAc-CaAc gels was estimated as mt(wet) /mt, assuming a constant density of 1 g/cm3 before and after treatment. Each data was obtained from five parallel samples.
DSC test
The hydrogel samples were cut into small pieces and sealed in DSC liquid sample pans for DSC measurements (DSC250 by TA), with a ramp rate of 3 °C/min over a temperature range of 20–80 °C.
Jacob’s curve test
The 0.2 mL precursor was poured into a self-made mold of 1 cm*1 cm*2 mm, and exposed to the light source with the intensity of 10 mW/cm2, for five irradiation times from 120 s to 420 s. The thickness of the irradiated five samples was measured by calipers.
Toxicity test
Cell viability was assessed using the Cell Counting Kit-8 (CCK-8) assay. The prepared hydrogel discs (with a diameter of 1 cm) were sterilized by ultraviolet (UV) irradiation for 30 minutes. The hydrogels were then soaked in DMEM/F-12 medium (0.1 g/mL) at 37 °C for 24 h to obtain the extract. Human corneal epithelial cells (Wuhan Procell Life Science & Technology Co., Ltd.) were cultured in DMEM/F12 (Gibco) at 37 °C with 5% CO2. Afterwards, cells were seeded in 96-well plates at densities of 1 × 10⁴ cells/well (1 day) and 5 × 10³ cells/well (3 day). For the culture for 7 days, cells were seeded in 24-well plates at 8 × 10³ cells/well in case of overwhelmed proliferation. Hydrogel extracts were added into the wells after overnight incubation for cell attachment, followed by incubation for 1, 3, 7 days, respectively. When specific periods of culture were finished, the supernatants in each well were removed and replaced by 100 µL and 500 µL medium (containing 10% CCK-8 reagent) for 96-well plates and 24-well plates, respectively. The cells were cultured for another 3 h before optical density (OD) testing. OD at 450 nm was measured using a plate reader (iMarkTM by Bio-Rad). Cell viability was calculated as (ODE−ODblank)/(ODNE−ODblank) × 100%, where ODE, ODNE, and ODblank are the mean OD values for the sample group, control group, and blank group, respectively.
For live/dead cell co-staining, the cells were co-cultured with hydrogels in 35 mm culture dishes. Specifically, the hydrogels were first uniformly coated on the bottom of the dishes, followed by seeding of cells at a density of 1 × 105 cells per dish onto the hydrogel surface. After 24 h of co-culture, the cell-hydrogel constructs were stained with Calcein-AM (1:1000) and PI (1:1000) to distinguish live (green fluorescence) and dead (red fluorescence) cells, respectively. The morphological characteristics and spatial distribution of cells on the hydrogel surface were then examined and imaged using the microscopic imaging system (DFC7000T by Leica).
Evaluation of in-vivo inflammatory
The animal experiments were conducted in accordance with guidelines for the care and use of laboratory animals and approved by the Ethics Committee of Zhejiang Cancer Hospital (Approval No. 2025-04-014). 6-week-old male BALB/c mice (body weight: ~20 g by Jiangsu GemPharmatech Co., Ltd.) were acclimatized for one week prior to experimentation. Notably, Sex was not a primary variable in the study design, as the endpoints assessed (subcutaneous hydrogel safety and biocompatibility) are not expected to differ fundamentally between sexes. Male mice were used to minimize variability associated with the estrous cycle in females, which is known to cause fluctuations in hormone levels that can significantly influence inflammation. Animals were housed under a 12 h light/dark cycle at 25 °C and 50% humidity, with ad libitum access to food and water.
Mice were randomly divided into three groups (n = 8 per group): sham, 4D printed hydrogel (named as 4D gel), and 4D printed hydrogel with liquid metal coating (named 4D-LM gel). Following anesthesia with 0.5% sodium pentobarbital (i.p.), a 10 mm dorsal incision was made to create a subcutaneous pocket. The 4D gel or 4D-LM gel was implanted into the pocket, and the incision was closed with surgical sutures. Sham-operated mice underwent identical procedures without hydrogel implantation. Postoperatively, mice were monitored daily for signs of distress or weight loss. At 7 days post-implantation, mice were euthanized. Blood samples were collected via retro-orbital puncture, allowed to clot at room temperature for 2 h, and centrifuged (3000 rpm, 15 min, 4 °C) to obtain serum. Serum levels of IL-1β, IL-6, TNF-α, IFN-γ, CXCL1, and CRP were quantified by Wuhan Servicebio Technology Co., Ltd. (China). Peri-implant tissues were harvested, fixed in 4% paraformaldehyde overnight, paraffin-embedded, and sectioned (3–5 μm thickness) for hematoxylin and eosin (H&E) staining. Data were presented as mean ± SD or median with interquartile range, as appropriate. Statistical comparisons among multiple groups were performed using one-way ANOVA or Kruskal–Wallis tests, followed by Tukey’s or Dunn’s post hoc tests where applicable. All analyses were conducted using GraphPad Prism 10, and statistical significance was defined as two-sided P < 0.05.
Numerical simulation of the deformation of the hydrogel
Finite element analysis (FEA) was performed using the commercial software ABAQUS to simulate the deformation behavior of the hydrogel via conventional static analysis. The hydrogel samples were modeled using eight-node 3D solid elements (C3D8R). To account for the contraction induced by incomplete conversion of the hydrogel, an equivalent thermal expansion approach was employed. Specifically, the mass loss-induced contraction ratio was converted into an equivalent shrinkage ratio by simulating a temperature reduction. The shrinkage ratios of the two sides of the sample were, respectively set as 20% and 30%. The hydrogel was assigned an elastic modulus of 0.1 MPa and a Poisson’s ratio of 0.48, while the constraining materials were treated as rigid materials.
In-vivo study of self-winding electrodes
This animal experiment was approved by the Ethics Committee of the Second Affiliated Hospital of Zhejiang University School of Medicine (SAHZU) (Approval No. 2024-176). Three 3-month-old New Zealand White rabbits (body weight: ~2.5 kg) were obtained from Hangzhou Medical College. The left eye was selected for the experiment. The rabbit was anesthetized with 3% pentobarbital sodium in the ear vein and placed on the experimental table. The operating area was disinfected with 75 vol% ethanol, and the hole towel was paved. Then the obliquus inferior muscle was separated by a surgical scissors and tweezers. For electromyographic (EMG) signal acquisition, a pair of spiral hydrogel electrodes was programmed into flatten states and fixed at 90 °C water for 30 s, followed by a quick cooling process at 25 °C for 10 s to avoid burning of the tissues. Two electrodes were then inserted to the obliquus inferior muscle and self-winded onto the surface acting respectively as the anode and the cathode for the EMG signal acquisition. Two hook platinum electrodes are used for the stimulation, by which the electrical stimulation of square-wave current with frequency of 15 Hz and wave width of 1 ms was applied to the muscle. The amplitude of the current is set as 0.3 mA, 0.5 mA, and 1 mA sequentially. A commercial patch electrode was placed at the front leg skin after shaving the rabbit hair working as the reference electrode. The EMG signal was collected by the biological data acquisition and stimulation system (BL-420F, Chengdu Techman Software Company, China), in which the gain is 100 mV. Besides, the EMG signal was processed by the low-pass filter with frequency of 10 kHz. For the electrical stimulation, a pair of hydrogel electrodes was self-winded to the muscle and acted as the input interface. The electrodes were connected to the biological data acquisition and stimulation system. Square-wave current with frequency of 15 Hz and wave width of 1 ms was applied to the muscle by the electrode, and the amplitude of the current was 0.3 mA.
Software and tools
The geometric simulations in Supplementary Figs. 7 and 8 were conducted by Abaqus 2021. Statistical analyses of Supplementary Fig. 15 were conducted using GraphPad Prism 10.0. All the other data analysis and graphing were performed using Origin 2021 unless noted.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data in this study are available in the main article, supplementary information, and source data file. All data underlying this study are available from the corresponding author upon request. Source data are provided with this paper.
References
Liu, Y. et al. Morphing electronics enable neuromodulation in growing tissue. Nat. Biotechnol. 38, 1031–1036 (2020).
Woodington, B. J. et al. Electronics with shape actuation for minimally invasive spinal cord stimulation. Sci. Adv. 7, eabg7833 (2021).
Bae, J.-Y. et al. A biodegradable and self-deployable electronic tent electrode for brain cortex interfacing. Nat. Electron. 7, 815–828 (2024).
Tringides, C. M. et al. Viscoelastic surface electrode arrays to interface with viscoelastic tissues. Nat. Nanotechnol. 16, 1019–1029 (2021).
Nan, K. et al. An ingestible, battery-free, tissue-adhering robotic interface for non-invasive and chronic electrostimulation of the gut. Nat. Commun. 15, 6749 (2024).
Li, S. et al. Bioresorbable, wireless, passive sensors for continuous pH measurements and early detection of gastric leakage. Sci. Adv. 10, eadj0268 (2024).
Ouyang, H. et al. Symbiotic cardiac pacemaker. Nat. Commun. 10, 1821 (2019).
Sunwoo, S.-H. et al. Soft bioelectronics for the management of cardiovascular diseases. Nat. Rev. Bioeng. 2, 8–24 (2024).
Madhvapathy, S. R. et al. Miniaturized implantable temperature sensors for the long-term monitoring of chronic intestinal inflammation. Nat. Biomed. Eng. 8, 1040–1052 (2024).
Deng, J. et al. Electrical bioadhesive interface for bioelectronics. Nat. Mater. 20, 229–236 (2021).
Wang, F. et al. 3D printed implantable hydrogel bioelectronics for electrophysiological monitoring and electrical modulation. Adv. Funct. Mater. 34, 2314471 (2024).
Huang, Y. et al. Bioelectronics for electrical stimulation: materials, devices and biomedical applications. Chem. Soc. Rev. 53, 8632–8712 (2024).
Liu, J., Ma, A. K. H., So, K. F., Lee, V. W. H. & Chiu, K. Mechanisms of electrical stimulation in eye diseases: a narrative review. Adv. Ophthalmol. Pract. Res. 2, 100060 (2022).
Song, Y., Min, J. & Gao, W. Wearable and implantable electronics: moving toward precision therapy. ACS Nano 13, 12280–12286 (2019).
Jonsson, A. et al. Therapy using implanted organic bioelectronics. Sci. Adv. 1, e1500039 (2015).
Zhao, Q., Li, C., Shum, H. C. & Du, X. Shape-adaptable biodevices for wearable and implantable applications. Lab Chip 20, 4321–4341 (2020).
Zhu, Z. et al. Tissue/Organ adaptable bioelectronic silk-based implants. Adv. Mater. 36, 2405892 (2024).
Zhang, Y. et al. Climbing-inspired twining electrodes using shape memory for peripheral nerve stimulation and recording. Sci. Adv. 5, eaaw1066 (2019).
Yi, J. et al. Water-responsive supercontractile polymer films for bioelectronic interfaces. Nature 624, 295–302 (2023).
Coles, L. et al. Origami-inspired soft fluidic actuation for minimally invasive large-area electrocorticography. Nat. Commun. 15, 6290 (2024).
Zhang, L. et al. Skin-inspired, sensory robots for electronic implants. Nat. Commun. 15, 4777 (2024).
Jang, T.-M. et al. Expandable and implantable bioelectronic complex for analyzing and regulating real-time activity of the urinary bladder. Sci. Adv. 6, eabc9675 (2020).
Shahrubudin, N., Lee, T. C. & Ramlan, R. An overview on 3D printing technology: Technological, materials, and applications. Procedia Manuf. 35, 1286–1296 (2019).
Huang, L. et al. Ultrafast digital printing toward 4D shape changing materials. Adv. Mater. 29, 1605390 (2017).
Nojoomi, A., Jeon, J. & Yum, K. 2D material programming for 3D shaping. Nat. Commun. 12, 603 (2021).
Fang, Z. et al. Modular 4D printing via interfacial welding of digital light-controllable dynamic covalent polymer networks. Matter 2, 1187–1197 (2020).
Sydney Gladman, A., Matsumoto, E. A., Nuzzo, R. G., Mahadevan, L. & Lewis, J. A. Biomimetic 4D printing. Nat. Mater. 15, 413–418 (2016).
Kim, J., Hanna, J. A., Byun, M., Santangelo, C. D. & Hayward, R. C. Designing responsive buckled surfaces by halftone gel lithography. Science 335, 1201–1205 (2012).
Hao, X. et al. Self-shaping soft electronics based on patterned hydrogel with stencil-printed liquid metal. Adv. Funct. Mater. 31, 2105481 (2021).
Zhang, Y. et al. 4D printing of a digital shape memory polymer with tunable high performance. ACS Appl. Mater. Interfaces 11, 32408–32413 (2019).
Zhao, Z. et al. Desolvation induced origami of photocurable polymers by digit light processing. Macromol. Rapid Commun. 38, 1600625 (2017).
Zang, T. et al. 4D printing of shape-morphing liquid crystal elastomers. Chem. Bio Eng. 1, 488–515 (2024).
Ni, C. et al. Shape memory polymer with programmable recovery onset. Nature 622, 748–753 (2023).
Grigoryan, B. et al. Multivascular networks and functional intravascular topologies within biocompatible hydrogels. Science 364, 458–464 (2019).
Feairheller, W. R. & Katon, J. E. The vibrational spectra of acrylic acid and sodium acrylate. Spectrochim. Acta Part A Mol. Spectrosc. 23, 2225–2232 (1967).
Acknowledgements
We thank National Natural Science Foundation of China (No. 52525312 to Q.Z., No. 52403170 to C.N. and U25A20321 to K.X,), National Key R&D Program of China (No. 2022YFB3805701 to Q.Z. and J.W.), Zhejiang Province “Van guard” and “Geese Leading” Research and Development Plan (No. 2022C03173 to X.J., No. 2024C03082 to Y.Z., and 2024C03001 to Q.Z.), and Central Guidance for Local Scientific and Technological Development Funding Projects (No. 2024ZY01057 to Q.F.). We thank Mrs. Na Zheng for the help of SEM characterizations at the State Key Laboratory of Chemical Engineering (Zhejiang University).
Author information
Authors and Affiliations
Contributions
C.N. and Q.Z. conceived the concept and wrote the manuscript. C.N. performed most of the experiments. Y.C., X.J., and Q.Z. supervised the project. C.Z., Y.Q. and Y.C. designed the EMG recording experiment. Z.Q. conducted the animal experiments with the assistance of X.Z. and Y.S. The circuit printing and characterization were realized by B.W. and K.X. The biocompatible evaluations were carried out by Q.K. and Y. Z. The underlying mechanism was analyzed by Q.F., Z.N., G.C., D.C., Z.F., J.W., and K.Y. All authors discussed the results.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Xabier Lopez de Pariza and the other anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ni, C., Zhang, C., Qin, Z. et al. 4D printing of trigger-free shape-memory hydrogels towards self-adaptive substrates for bioelectronics. Nat Commun 17, 677 (2026). https://doi.org/10.1038/s41467-025-67323-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-67323-1