Abstract
The Onchocerciasis Control Programme in West Africa implemented vector control (VC) and ivermectin mass drug administration (MDA) to eliminate blindness, intensifying efforts in Special Intervention Zones (SIZ). Togo aims to eliminate onchocerciasis transmission (EOT) by 2030. We use the EPIONCHO-IBM model to project microfilarial prevalence trends across Togo’s five regions by SIZ status, MDA coverage (65%-80%) and VC efficacy (60%-100%). We compare projections with prevalence surveys (400 villages, 1970–2017) stratified by hypoendemic, mesoendemic, hyperendemic, and holoendemic baseline endemicity, and calculate EOT probabilities for 2024, 2027, and 2030. Combined VC and MDA reduced prevalence nationwide. After cessation of VC, prevalence continued to decline in hypo-to-mesoendemic areas under annual MDA, while hyperendemic areas required biannual MDA. In holoendemic areas, prevalence rebounded even with biannual MDA, indicating that alternative strategies are needed. EPIONCHO-IBM reproduces Togo’s onchocerciasis trends throughout five decades of intervention and provides a transferable framework to guide policy towards 2030 goals.
Similar content being viewed by others
Introduction
Onchocerciasis, caused by the filarial nematode Onchocerca volvulus and transmitted by Simulium blackflies, remains a public health concern, particularly in sub-Saharan Africa where mean microfilarial prevalence in 2018 exceeded 25% in some countries1. The Global Burden of Disease 2021 Study estimated 1.3 (0.8–1.9) million disability-adjusted life-years, and 20 (18–22) million people infected2. Ocular, cutaneous and neuro-hormonal sequelae cause substantial morbidity3,4 and excess mortality5. Interventions include vector control (VC) and mass drug administration (MDA) of ivermectin for prolonged periods, owing to the 10-year (average) adult worm lifespan6.
The Onchocerciasis Control Programme in West Africa (OCP, 1974–2002) aimed to eliminate onchocercal blindness through weekly aerial larviciding of Simulium damnosum sensu lato (s.l.) riverine breeding sites for ≥14 years7. In the late 1980s, ivermectin MDA was introduced alongside VC or, in some Western Extension foci, as the sole intervention8. In 2012, the goal shifted from morbidity control to elimination of transmission (EOT)9.
Achieving EOT with MDA largely depends on pre-control endemicity (baseline microfilarial prevalence), determined by vector biting rates10. The impact of MDA on transmission is influenced by therapeutic coverage (proportion of population receiving treatment) and adherence (proportion of eligibles consistently taking treatment). It has been proposed that a minimal therapeutic coverage of 65% of total population (80% of eligibles) should be reached and sustained for at least 15–17 years to achieve elimination goals11,12.
The World Health Organization’s 2021–2030 Roadmap on neglected tropical diseases (NTDs) aims at verifying onchocerciasis EOT in 12 (31%) endemic countries by 203013. Verification of EOT requires stop-MDA surveys and, if successful, post-treatment surveillance (PTS) for 3–5 years following MDA cessation11. Togo, having eliminated four other NTDs14, aims to achieve onchocerciasis EOT by 203015. The epidemiology of onchocerciasis is heterogeneous across its five regions, from north to south: Savanes, Kara, Centrale, Plateaux and Maritime (Fig. 1, and Supplementary Information: Text 1.1, Table 1 and Fig. 1), each further sub-divided into prefectures. The blackfly-prolific Oti River Basin and its tributaries (Kara, Kéran and Mô) (Fig. 1, and Supplementary Information: Text 1.2), as well as at-risk hard-to-reach villages pose particular challenges to EOT15,16.
a Location of Togo in West Africa. b Five regions of Togo and its main river basins. Togo regions are indicated by grey borders and names; main rivers are indicated by light blue and names. River basins with superscript 1 are named but not depicted. Mô is a tributary of the Oti River. Ogou and Amou are tributaries of the Mono River. Yoto and Haho flow towards the delta of the Maritime Region. Maps were drawn by the authors in R. Data sources: country outlines and coastlines in a from: Natural Earth (public domain, www.naturalearthdata.com); administrative boundaries in b from: GADM v4.0 accessed via the geodata R package (v0.6-2; CC BY 4.0, https://github.com/rspatial/geodata); river networks/basins in b from: HydroSHEDS (HydroRIVERS/HydroBASINS; CC BY 4.0, www.d-maps.com).
Onchocerciasis control in Togo began with the OCP in the northwest, gradually expanding southward (Supplementary Fig. 2a and Supplementary Table 2). Savanes, Kara and a portion of Centrale were included in the OCP early VC Phases II and III East (starting in 1976–77). The remaining areas of Centrale, Plateaux and Maritime, were covered by the Southern Extension (starting in 1988–89), and annual ivermectin MDA was introduced. By the end of the OCP in 2002, Kara and parts of Savanes and Centrale had not achieved satisfactory entomo-epidemiological results17 and were included in the OCP Special Intervention Zones (SIZ, Supplementary Fig. 2b), receiving aerial larviciding until 2007 and intensified, biannual MDA until 201218. After the OCP/SIZ, Togo continued onchocerciasis control through annual or biannual MDA (Supplementary Information: Text 1.2).
Modelling studies and recent reviews suggest that EOT may be attainable in areas with low to moderate baseline endemicity through sustained, high-coverage annual MDA19,20. However, highly-endemic foci will likely require alternative treatment strategies (ATS), such as moxidectin MDA19,21, potentially reinforced by community-directed “slash-and-clear” VC22.
Following 2015–2017 stop-MDA surveys in Maritime, similar surveys commenced in Savanes in 202323. To support evidence-based decision-making by the National Onchocerciasis Control Programme (NOCP), we modelled, using the individual-based, stochastic EPIONCHO-IBM transmission model10, the temporal trends of O. volvulus microfilarial prevalence in Togo’s regions, comparing modelled projections with data from 400 endemic villages surveyed over time. We calculated the likelihood of EOT for each prefecture to determine which areas can begin stop-MDA surveys or may require ATS.
Results
Prevalence trends by region
Microfilarial prevalence trends were modelled for a total of 400 villages. Figures 2–6 present modelling results for the 140 OCP villages with recorded baseline microfilarial prevalence (BMP) estimates. Supplementary Figs. 8–14 present results for 252 villages without BMP estimates. (An additional 8, non-OCP villages also had BMP estimates.) Villages with BMP estimates were categorised into four endemicity levels: hypoendemic ( > 0% but <40%), mesoendemic ( ≥ 40% but <60%), hyperendemic ( ≥ 60% but <80%) and holoendemic ( ≥ 80%) microfilarial prevalence20. EPIONCHO-IBM10 was implemented across four BMP values: 30%, 50%, 70% and 90% to capture these endemicity levels within Togo’s five regions, SIZ status and intervention history (Table 1). Three (“minimal”, “reference” and “enhanced”) intervention scenarios were modelled (Table 2).
For Special Intervention Zone (SIZ): a hypoendemic village (n = 1) with simulated 30% BMP, 100% VC efficacy and biannual MDA. b mesoendemic village (n = 1) with simulated 50% BMP and biannual MDA. c mesoendemic villages (n = 2) with simulated 50% BMP, 100% VC efficacy and biannual MDA. For Non-Special Intervention Zone (non-SIZ): d mesoendemic village (n = 1) with simulated 50% BMP. e hyperendemic villages (n = 2) and 70% BMP with 100% VC efficacy. Yellow circles represent microfilarial prevalence of hypoendemic villages, orange circles indicate microfilarial prevalence of mesoendemic villages, and brown circles denote microfilarial prevalence of hyperendemic villages. Error bars are 95% (Wilson score) confidence intervals (95% CIs). For each BMP setting and intervention scenario, the average of 100 model repeats was used to calculate the mean microfilarial prevalence dynamics (blue lines). Dark blue lines represent the reference scenario; light blue lines above and below dark blue lines indicate the minimal and enhanced scenarios, respectively. Vertical coloured lines indicate: start of VC (light green); start of annual MDA (red); end of VC (dark green); start of biannual MDA (orange). Further information about the individual villages plotted here can be found in Supplementary Table 10. Source data and code are available47.
For Special Intervention Zone (SIZ, all villages): a hypoendemic villages (n = 2) with simulated 30% BMP and biannual MDA. b mesoendemic villages (n = 2) with simulated 50% BMP and biannual MDA. c hyperendemic villages (n = 6) with simulated 70% BMP and biannual MDA. d holoendemic villages (n = 2) with simulated 90% BMP and biannual MDA. Yellow circles represent microfilarial prevalence of hypoendemic villages, orange circles indicate microfilarial prevalence of mesoendemic villages, and brown circles denote microfilarial prevalence of hyper- and holoendemic villages. Error bars are 95% (Wilson score) confidence intervals (95% CIs). For each BMP setting and intervention scenario, the average of 100 model repeats was used to calculate the mean microfilarial prevalence dynamics (blue lines). Dark blue lines represent the reference scenario; light blue lines above and below dark blue lines indicate the minimal and enhanced scenarios, respectively. Vertical coloured lines indicate: start of VC (light green); start of annual MDA (red); end of VC (dark green); start of biannual MDA (orange). Further information about the individual villages plotted here can be found in Supplementary Table 10. Source data and code are available47.
For Special Intervention Zone (SIZ): a hyperendemic villages (n = 2) with 70% BMP and biannual MDA. For Non-Special Intervention Zone (non-SIZ): b hypoendemic villages (n = 13) with 30% BMP. c mesoendemic villages (n = 26) with 50% BMP. d hyperendemic villages (n = 5) with 70% BMP. Yellow circles represent microfilarial prevalence of hypoendemic villages, orange circles indicate microfilarial prevalence of mesoendemic villages, and brown circles denote microfilarial prevalence of hyperendemic villages. Error bars are 95% (Wilson score) confidence intervals (95%CIs). For each BMP setting and intervention scenario, the average of 100 model repeats was used to calculate the mean microfilarial prevalence dynamics (blue lines). Dark blue lines represent the reference scenario; light blue lines above and below dark blue lines indicate the minimal and enhanced scenarios, respectively. Vertical coloured lines indicate: start of VC (light green); start of annual MDA (red); end of VC (dark green); start of biannual MDA (orange). Further information about the individual villages plotted here can be found in Supplementary Table 10. Source data and code are available47.
a hypoendemic villages (n = 12) with simulated 30% BMP and annual MDA. b hypoendemic villages (n = 4) with 30% BMP and biannual MDA. c mesoendemic villages (n = 15) with 50% BMP and annual MDA. d mesoendemic villages (n = 12) with 50% BMP and biannual MDA. e hyperendemic villages (n = 12) with 70% BMP and annual MDA. f hyperendemic villages (n = 13) with 70% BMP and biannual MDA. Yellow circles represent microfilarial prevalence of hypoendemic villages, orange circles indicate microfilarial prevalence of mesoendemic villages, and brown circles denote microfilarial prevalence of hyperendemic villages. Error bars are 95% (Wilson score) confidence intervals (95%CIs). For each BMP setting and intervention scenario, the average of 100 model repeats was used to calculate the mean microfilarial prevalence dynamics (blue lines). Dark blue lines represent the reference scenario; light blue lines above and below dark blue lines indicate the minimal and enhanced scenarios, respectively. Vertical coloured lines indicate: start of VC (light green); start of annual MDA (red); end of VC (dark green); start of biannual MDA (orange). Further information about individual villages plotted here can be found in Supplementary Table 10. Source data and code are available47.
a hypoendemic villages (n = 6) with simulated 30% BMP. b hyperendemic village (n = 1) with 70% BMP. Yellow circles represent microfilarial prevalence of hypoendemic villages, and brown circles denote microfilarial prevalence of a hyperendemic village. Error bars are 95% (Wilson score) confidence intervals (95%CIs). For each BMP setting and intervention scenario, the average of 100 model repeats was used to calculate the mean microfilarial prevalence dynamics (blue lines). Dark blue lines represent the reference scenario; light blue lines above and below dark blue lines indicate the minimal and enhanced scenarios, respectively. Vertical coloured lines indicate: start of VC (light green); start of annual MDA (red); end of VC (dark green). Further information about the individual villagesplotted here can be found in Supplementary Table 10. Source data and code are available47.
Savanes
Villages with BMP estimates in Savanes prefectures were hypo- to mesoendemic in SIZ and meso- to hyperendemic in non-SIZ areas (Fig. 2). Prevalence decline was primarily driven by VC with enhanced (90%) or 100% efficacy (Fig. 2a–e), eventually leading to ≥90% EOT probability following initiation of MDA in hypo- to mesoendemic areas (Supplementary Table 12). Some SIZ villages lacking recorded BMP (Oti River Basin) had high prevalence, following hyperendemic simulation trends (Supplementary Fig. 8c). Non-SIZ villages lacking BMP estimates followed hypoendemic trends (Supplementary Fig. 9).
Kara
Villages in Kara (all prefectures in SIZ) with recorded BMP estimates encompassed all endemicity levels (Fig. 3). The intervention scenarios best capturing infection trends were: minimal for hypoendemicity (Fig. 3a), reference for mesoendemicity (Fig. 3b), and minimal (Mô River Basin), reference (Kara River Basin) or enhanced (Kara River Basin) for hyperendemicity (Fig. 3c). Holoendemic villages aligned with the enhanced (Kéran River Basin) intervention scenario (Fig. 3d), with the model capturing the observed prevalence rebound after VC cessation. Two-fifths (30/74) of villages without BMP estimates followed hyper- to holoendemicity trends (Supplementary Information: Table 11, Fig. 10c–d).
Centrale
Villages with recorded BMP in Centrale were hyperendemic in SIZ and ranged from hypo- to hyperendemic in non-SIZ areas. The SIZ hyperendemic villages (Mô River Basin) followed minimal, reference or enhanced intervention scenarios (Fig. 4a). Model projections for non-SIZ hypo- and mesoendemic villages (Fig. 4b, c) suggested a similar impact across intervention scenarios, likely because VC and MDA started roughly at the same time, reducing variability. Non-SIZ hyperendemic villages (Mono River Basin) followed the enhanced intervention scenario (Fig. 4d). Villages without recorded BMP ranged from hypo- to holoendemic (Supplementary Figs. 11–12). Nearly all SIZ villages (13/14) lacking BMP followed hyper- to holoendemic trends (Mô River Basin) (Supplementary Information: Table 11, Fig. 11).
Plateaux
Villages in Plateux (all non-SIZ) with recorded BMP were evenly distributed among hypo-, meso- and hyperendemicity (Fig. 5). Model outputs were similar for hypo- and mesoendemicity, as in Centrale (Fig. 5a–d). Prevalence trends in hyperendemic villages were mostly captured by the enhanced intervention scenario, with some following the minimal and reference scenarios (Fig. 5e, f, Mono River Basin). Several villages without BMP estimates followed hyperendemic trends (Supplementary Information: Table 11, Fig. 13). Prefectures continued with annual MDA or switched to biannual MDA in 2014 (Supplementary Table 2).
Maritime
Villages in Maritime with recorded BMP were predominantly hypoendemic, and their modelled prevalence trends followed the reference and enhanced intervention scenarios (Fig. 6a). The hyperendemic village depicted in Fig. 6b was consistent with the enhanced scenario. Villages without BMP estimates generally exhibited low endemicity, except in Yoto and possibly Avé prefectures, where some trends suggested hyperendemicity following the enhanced intervention scenario (Supplementary Information: Table 11, Fig. 14).
Elimination probabilities
Supplementary Tables 12–17 present EOT probabilities per region, baseline endemicity, SIZ status and intervention scenario. In Savanes, all SIZ hypo- to (100% VC efficacy) hyperendemic villages and nearly all (86%) non-SIZ villages are projected to have reached ≥90% EOT probability by 2024. However, in the northwestern part of Savanes non-SIZ, surveys conducted in early to mid-1970s (not in the OCP database), indicated baseline hyperendemicity (Supplementary Fig. 1). As non-SIZ areas of Savanes did not receive biannual MDA and VC ceased in 1993 (Table 1), the projected EOT probabilities are <5%. SIZ villages without BMP estimates following hyperendemic trends are projected to have <90% probability of reaching EOT by 2024. Extending biannual MDA to 2027 or 2030 in putative hyperendemic villages does not improve their EOT probabilities, remaining at <5%, 20–59%, and 60–89%, under minimal, reference, and enhanced interventions, respectively (Supplementary Table 12).
In Kara and Centrale, most hypo- and mesoendemic villages (regardless of SIZ status and intervention scenario) are projected to have reached ≥90% EOT probability by 2024. Some non-SIZ mesoendemic villages of Centrale following the minimal scenario and annual MDA would only reach 60–89% EOT probability, even with MDA extended to 2030. Hyperendemic villages following the enhanced scenario are projected to have reached ≥90% EOT probability by 2024. By contrast, those following the minimal or reference scenarios would have 5–19% or 60–89% EOT probability by 2024, respectively. If treatment continues until 2030, the former’s EOT probability would increase to 20–59%. As in Savanes, holoendemic villages have <5% EOT probability (Supplementary Tables 13–14).
In Plateaux, owing to its lower BMP compared to Kara and Centrale, VC started later and not all prefectures adopted biannual MDA. Hypoendemic villages (irrespective of treatment frequency) and mesoendemic villages following reference and enhanced scenarios with annual or biannual MDA, are projected to have reached ≥90% EOT probability by 2024. The same applies to mesoendemic villages following the minimal scenario under biannual MDA. Mesoendemic villages following the minimal scenario and hyperendemic villages following the enhanced scenario under annual MDA, would have had 60–89% EOT probability by 2024. Hyperendemic villages following minimal and reference scenarios under annual MDA would only reach <5% EOT probability by 2024 or 2030. Those following minimal and reference scenarios under biannual MDA would have reached, respectively, <5% and 5–19% EOT by 2024, with the latter increasing to 20–59% if biannual MDA were extended until 2030. By contrast, hyperendemic villages following the enhanced scenario and already under biannual MDA could reach ≥90% EOT if biannual treatment continues until 2030 (Supplementary Table 15).
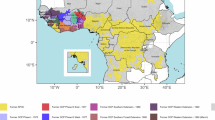
In Maritime, only annual MDA had been implemented by 2018. Being the least endemic region, all hypoendemic villages, and those mesoendemic villages following the reference and enhanced scenarios, are projected to have reached ≥90% EOT probability regardless of whether treatment ceased in 2014 or 2020. However, in one confirmed (with) and several putative (without BMP) hyperendemic villages, the EOT probability is 20–59% in 2020. Extending treatment in such villages to 2024 or 2030 would increase it to 60–89% (Supplementary Tables 16–17). Supplementary Tables 18–29 list villages (with and without BMP estimates) by region, SIZ status, and river basin for which EPIONCHO-IBM projects EOT probabilities <90% if MDA stops in 2027. Supplementary Tables 32–34 present prefecture-level likelihood of reaching EOT when simulating that ivermectin MDA stops in 2024, 2027 or 2030. Fig. 7 illustrates the (categorical) likelihood of reaching EOT if ivermectin MDA stops in 2027 across Togo. Supplementary Table 35 summarises current control strategies and recommendations by prefecture.
Details of the calculation of the prefecture-wide (joint) EOT probabilities are given in Supplementary Text 8. Categories are defined as: Very likely ( ≥ 90%); Likely (50–89%); Possibly (5–49%); Unlikely (0.01- < 5%), and Very unlikely ( < 0.01%). The pie-charts indicate the proportion of the total number of villages surveyed in each prefecture that are projected to reach <5%, 5–19%, 20–59%, 60–89% or ≥90% EOT probability, with the size of the pie-charts reflecting the number of villages (exact numbers are given in Supplementary Table 33; detailed information on those villages with projected EOT probability <90% is presented in Supplementary Tables 18–29). Black borders indicate regions (see Fig. 1); white borders correspond to prefectures. The map for Togo (with regions and prefectures) was drawn by the authors in R using the geodata package (version 0.6-2; CC BY 4.0, https://github.com/rspatial/geodata). Source data and code are available47.
Discussion
Modelling analyses of detailed spatiotemporal O. volvulus prevalence data from the OCP and other sources provided a unique opportunity to quantify the combined impact of VC and ivermectin MDA in a former OCP country. The epidemiology of onchocerciasis in Togo has changed profoundly over nearly 50 years of intervention, with some prefectures on the verge of reaching EOT and others projected not to reach EOT with current strategies. Our results illustrate the power of mathematical modelling in evaluating past, current and future epidemiology of onchocerciasis, to assist programmes in decision-making and resource allocation to maximise their chances of reaching the 2030 elimination goals.
We identified regional epidemiological patterns, long-term prevalence declines, and subsequent increases in some cases. In Savanes non-SIZ, (hypo- to hyperendemic) villages with 90-100% VC efficacy had reached a very low microfilarial prevalence by the time MDA started. This contrasts with other regions where VC was likely less impactful, underscoring the need for combining VC and MDA. After VC cessation, hypo- to mesoendemic villages performed well under MDA. Annual MDA sustained previous gains in hyperendemic villages, but only biannual MDA led to further prevalence declines. Holoendemic villages experienced prevalence increases after VC cessation, in both data and model outputs, even with biannual MDA.
According to EPIONCHO-IBM, achieving ≥90% EOT probability by 2024 in hypo- and mesoendemic areas nationwide seems feasible, and stop-MDA surveys could start if not already underway. Conversely, modelled prevalence in areas with hyper- and holoendemic villages, such as the Oti River Basin (Savanes), and the Kéran and Mô River Basins (Kara and Centrale), declines to a pseudo-equilibrium (a seemingly steady state of infection maintained by the opposing effects of treatment and microfilarial prevalence recovery under conditions of intense inter-treatment transmission) since 2007, and are unlikely to achieve EOT under the current (biannual) MDA strategy, even if continued until 2030. Infection prevalence of 0.1–1.0% in blackflies was found for these river basins (2015), and of 0.5–0.8% in Mô (2018–2019), indicating active transmission24,25. In non-SIZ (northwestern) Savanes, where VC started and ended early15, surveyed villages (with or without BMP estimates) are scarce in our database. However, based on surveys indicating hyperendemicity in the 1970s, EOT likelihood in Tandjouaré and Tône prefectures is <90%. Stop-MDA surveys in 2022 indicated some villages of potential concern in these prefectures26.
Whilst most of Maritime would likely have reached ≥90% EOT probability by 2020, some villages without BMP estimates, mostly in Yoto Prefecture, followed hyperendemic trends with moderate (60–89%) EOT probability. Active transmission was confirmed in Yoto during the 2020–2023 stop-MDA survey, prompting focal biannual MDA27.
Limitations. Although baseline annual biting rates (ABRs) were reduced during VC by its assumed efficacy, they were modelled as bouncing back to pre-control levels one year after VC cessation28. This conservative assumption is supported by several entomological studies conducted in Togo and other OCP countries. In the Mô River Basin, ABRs in 2015–2019 (12,000–16,000 bites/person/year)24,25 were comparable to pre-VC values (16,000–47,000). In areas of Burkina Faso bordering with Togo, ABRs returned to or surpassed baseline levels within two years of stopping VC (Loaba: 8617 compared to 6090; Ziou Zabré: 30,739 compared to 11,879)7. In Niger, the Goulbi River Basin experienced a bounce-back to 14,000 bites/person/year within one year after ceasing vector control, compared to baseline values of 14,35029. Even in areas experiencing deforestation as in the Wawa River in Ghana (a water course shared with Plateaux), biting rates rebounded to pre-control levels after VC ended, reaching 2400–4000 compared to 2800–4050 bites/person/month30. Notwithstanding, deforestation, particularly in western Plateaux and southern Centrale31, may have led to secular changes in vector density32 not considered in the model. Future modelling studies should account for secular trends in ABRs when substantial changes in transmission conditions have been documented33. Although we modelled information from 400 villages, there may be many others, certainly those with >2000 people, for which we have no epidemiological data as they were only incorporated into the NOCP in 201823. Because we may not have fully captured within-region heterogeneity, our prefecture-wide EOT likelihood must be interpreted with caution. Also, EPIONCHO-IBM models closed populations, not accounting for movement of people and/or flies between villages or cross-border migration that could jeopardise EOT by re-introduction of infection from less-well controlled areas26. Recently, EPIONCHO-IBM has been used to model the effects, on introduction or re-introduction of infection, of an influx of immigrants with varying worm burdens arriving from an area with ongoing transmission into an onchocerciasis-free setting with local blackfly populations34.
In conclusion, Togo has made considerable progress towards onchocerciasis EOT owing to VC and ivermectin MDA, switching to biannual frequency where necessary. However, areas with confirmed or putative high baseline endemicity pose challenges to achieving nation-wide EOT. EPIONCHO-IBM has proven its usefulness in interpreting epidemiological data, supporting decisions on stop-MDA surveys (e.g., in hypo- to mesoendemic areas with ≥15 years of high-coverage MDA and/or biannual treatment). In hyper- and holendemic areas with low EOT probabilities (e.g., in Kara and Centrale), the model suggests that ATS21 should be considered. In particular, biannual moxidectin MDA supplemented, if feasible, by “slash-and-clear” VC would be beneficial22,35. EPIONCHO-IBM could be used in other former OCP countries to inform policy decisions towards the 2030 elimination goals.
Methods
Ethics statement
This study analysed de-identified, aggregate village-level microfilarial prevalence data collected in Togo between 1970 and 2017 by the OCP, and national and partner programmes, which obtained ethical approval from their respective boards at the time of the surveys (e.g., from the Togo Bioethics Committee for Research in Health (Comité de Bioéthique pour la Recherche en Santé), with authorization and approval granted by the Togo Ministry of Health)24,25. In the OCP, a memorandum of agreement was signed covering all operational issues as well as clearance for epidemiological surveys. A committee consisting of OCP Chief of Units ensured that work plans and methodology were correctly followed by field technicians. Communities were free to participate in the taking of skin snip samples5. For this study, no human participants were recruited and no individual-level identifiable data were used; therefore, no institutional review board approval and informed consent were required for the present (modelling) analysis.
Prevalence data
Cross-sectional surveys (1970–2017) provided microfilarial prevalence data for 400 endemic villages (Supplementary Information: Text 2, Table 1, Figs. 3–5). Villages were considered to have recorded baseline endemicity estimates if surveyed for O. volvulus microfilarial prevalence before the start of ivermectin MDA or <3 years after starting VC7,36 (n = 148 with BMP estimates; 140 in the OCP database). Other villages lacked baseline assessments (n = 252) but had surveys conducted after the start of interventions (Supplementary Table 1). We used crude rather than age- and sex-standardised microfilarial prevalence because the latter was missing in 12% of the surveys, and there was a 0.99 Pearson’s correlation coefficient between crude and standardised prevalence (Supplementary Fig. 6)36. Wilson-score 95% confidence intervals (95% CIs)37 were calculated for each prevalence estimate.
Four endemicity levels (hypo-, meso-, hyper- and holoendemic)20 were used to categorise villages with BMP estimates, for which 30%, 50%, 70% or 90% BMP values were modelled to capture their endemicity levels. For villages without recorded BMP, all four endemicity levels were simulated to identify their most likely initial endemicity category according to modelled microfilarial prevalence trajectories (Supplementary Table 1). Baseline annual biting rates (ABRs, bites/person/year) were estimated by interpolating the relationship between microfilarial prevalence and ABR using the EPIONCHO-IBM transmission model10, generating ABR = 290 (for 30%), 615 (50%), 2,200 (70%), and 60,000 (90%) (Supplementary Information: Text 3, Tables 3–4).
EPIONCHO-IBM
EPIONCHO-IBM is a stochastic, individual-based model simulating O. volvulus infection dynamics in a closed population (village)10 (500 individuals for this work). It tracks the number of (male and female) adult worms and microfilarial load in each human host over time, and the mean number of infective, L3 larvae per blackfly vector. Adult worm and microfilarial mortality rates are parasite-age dependent, and adult female worm fecundity decreases with worm age10. Human exposure is age- and sex-dependent38, and overdispersed among individuals following a gamma distribution with shape and rate parameter kE (=0.3 for this work)10. Parasite population abundance is regulated by density-dependent processes within humans and vectors39, which contribute to endemic stability and intervention resilience10,40. A description of the model is provided in Hamley et al.10 (code available at: https://github.com/mrc-ide/EPIONCHO.IBM)41.
Modelling interventions and scenarios
EPIONCHO-IBM was implemented across the four endemicity levels aforementioned within Togo’s five regions, considering their SIZ status and intervention history (Table 1). Supplementary Table 2 provides intervention details at prefecture level. Modelled interventions comprised VC and ivermectin MDA. Ivermectin MDA was modelled by incorporating microfilaricidal and embryostatic effects42, and a permanent sterilising effect on adult female worms43. Therapeutic coverage (proportion of individuals receiving ivermectin at each round in the total population) was simulated as the mean treatment probability in any treatment round. A fixed proportion of systematic non-adherence (SNA) was used to represent eligible individuals never receiving treatment10. VC was simulated by reducing ABR based on assumed efficacy for the entire larviciding duration. ABR values were assumed to return to initial levels one year after VC cessation28. Supplementary Tables 5–9 provide reported coverage data by region and year.
In addition to the three intervention scenarios described in Table 2 (and Supplementary Text 4), a 100% VC efficacy44 was simulated in Savanes, varying therapeutic coverage and SNA as per the three main scenarios. Biannual MDA started in 2003 or 2014 in some regions (Table 1). In 2020, MDA was modelled annually nationally due to the COVID-19 pandemic45. Microfilarial prevalence trends were simulated until 2030, with the last (annual or biannual) treatment in 2029.
A total of 100 model repeats were run for each of the three intervention scenarios, four endemicity levels, five regions and SIZ status. The mean of the 100 runs yielded mean microfilarial prevalence dynamics over time. Prevalence trends were visualized using the “minimal” and “enhanced” scenarios as the upper and lower uncertainty bounds, with “reference” scenario representing the average. Simulated trends are presented alongside survey prevalence estimates with 95% CIs per region and SIZ status to illustrate infection trends and compare model outputs with observations (village surveys). The proportion of the population examined for O. volvulus skin microfilariae decreased from >80% at the beginning of the OCP to <70% between 2006 and 2015 (Supplementary Information: Text 5, Fig. 7). Supplementary Table 10 lists the villages with recorded BMP.
To identify the intervention scenarios followed by villages lacking BMP estimates, we used the (median) Mean Squared Error (MSE) between observed and simulated microfilarial prevalence trends across model repeats, with the smallest MSE values indicating the best-fit scenarios (Supplementary Information: Text 6, Table 11).
Elimination probabilities
For simulation of EOT probabilities, MDA frequencies from 2018 (annual or biannual, depending on prefecture) were used. The probability of reaching EOT for each scenario, region and SIZ status was calculated as the percentage of 100 model runs yielding zero microfilarial prevalence 50 years after stopping MDA35 in 2024, 2027 or 2030. For Maritime, where MDA ended in some prefectures in 2014 and in all by 2020, simulations ceasing MDA in 2014 or 2020 were performed (Table 1 and Supplementary Table 2). Five EOT probability categories were defined: <5%, 6–19%, 20–59%, 60–89% and ≥90%. Supplementary Information: Text 8, Tables 30–31 present the rationale and calculations of prefecture-wide likelihoods (joint probabilities) of achieving EOT.
We followed the five principles of the NTD Modelling Consortium regarding Policy-Relevant Items for Reporting Models in Epidemiology of NTDs (PRIME-NTD)46 (Supplementary Information: Text 8, Table 36).
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All the information used for the analyses presented here is contained in the figures, tables and Supplementary Information of this paper, and in the (publicly available) epidemiological database (https://doi.org/10.1371/journal.pntd.0012312.s001; https://doi.org/10.1371/journal.pntd.0012312.s003) found in Vinkeles Melchers et al15.
Code availability
The EPIONCHO-IBM model code is available at: https://github.com/mrc-ide/EPIONCHO.IBM41. The following link provides the branch for the modelling analysis presented in the paper: https://github.com/mrc-ide/EPIONCHO.IBM/tree/togo-elimination-analysis. The R code to reproduce the modelling analysis and figures (https://doi.org/10.5281/zenodo.17351356) can be found in Amaral & Basáñez47.
References
Schmidt, C. A. et al. The prevalence of onchocerciasis in Africa and Yemen, 2000-2018: a geospatial analysis. BMC Med. 20, 293 (2022).
GBD 2021 Diseases and Injuries Collaborators Global incidence, prevalence, years lived with disability (YLDs), disability-adjusted life-years (DALYs), and healthy life expectancy (HALE) for 371 diseases and injuries in 204 countries and territories and 811 subnational locations, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 403, 2133–2161 (2024).
Burnham, G. Onchocerciasis. Lancet 351, 1341–1346 (1998).
Colebunders, R. et al. Reducing onchocerciasis-associated morbidity in onchocerciasis-endemic foci with high ongoing transmission: a focus on the children. Int. J. Infect. Dis. 116, 302–305 (2022).
Little, M. P., Breitling, L. P., Basáñez, M. G., Alley, E. S. & Boatin, B. A. Association between microfilarial load and excess mortality in onchocerciasis: an epidemiological study. Lancet 363, 1514–1521 (2004).
Plaisier, A. P., Alley, E. S., van Oortmarssen, G. J., Boatin, B. A. & Habbema, J. D. F. Required duration of combined annual ivermectin treatment and vector control in the Onchocerciasis Control Programme in West Africa. Bull. World Health Organ. 75, 237–245 (1997).
Hougard, J. M., Alley, E. S., Yaméogo, L., Dadzie, K. Y. & Boatin, B. A. Eliminating onchocerciasis after 14 years of vector control: a proved strategy. J. Infect. Dis. 184, 497–503 (2001).
Boatin, B. The onchocerciasis control programme in West Africa (OCP). Ann. Trop. Med. Parasitol. 102, S13–S17 (2008).
World Health Organization. Accelerating work to overcome the global impact of neglected tropical diseases—A roadmap for implementation. https://www.who.int/publications/i/item/WHO-HTM-NTD-2012.1 (2012).
Hamley, J. I. D., Milton, P., Walker, M. & Basáñez, M.-G. Modelling exposure heterogeneity and density dependence in onchocerciasis using a novel individual-based transmission model, EPIONCHO-IBM: implications for elimination and data needs. PLoS Negl. Trop. Dis. 13, e0007557 (2019).
World Health Organization. Guidelines for stopping mass drug administration and verifying elimination of human onchocerciasis. Criteria and procedures. https://www.who.int/publications/i/item/9789241510011 (2016).
Makenga Bof, J. C., Ntumba Tshitoka, F., Muteba, D., Mansiangi, P. & Coppieters, Y. Review of the national program for onchocerciasis control in the Democratic Republic of the Congo. Trop. Med. Infect. Dis. 4, 92 (2019).
World Health Organization. Ending the neglect to attain the Sustainable Development Goals: a road map for neglected tropical diseases 2021–2030. https://www.who.int/publications/i/item/9789240010352 (2021).
World Health Organization African Region. WHO Director-General congratulates Togo on becoming first country to eliminate four neglected tropical diseases. https://www.who.int/news/item/22-08-2022-who-director-general-congratulates-togo-on-becoming-first-country-to-eliminate-four-neglected-tropical-diseases (2022).
Vinkeles Melchers, N. V. S. et al. Impact of ivermectin and vector control on onchocerciasis transmission in Togo: assessing the empirical evidence on trends in infection and entomological indicators. PLoS Negl. Trop. Dis. 18, e0012312 (2024).
World Health Organization & African Programme for Onchocerciasis Control. Report of the fifth activity review and planning meeting of the Special Intervention Zones (SIZ): Ouagadougou 8-10 November 2006. https://apps.who.int/iris/handle/10665/276197?show=full (2006).
World Health Organization & Onchocerciasis Control Programme in West Africa. Onchocerciasis control in Special Intervention Zones including Sierra Leone in the OCP area. Plan of action and budget: Ouagadougou 4-6 December 2002. https://apps.who.int/iris/handle/10665/342151 (2002).
Yaméogo, L. Special intervention zones. Ann. Trop. Med. Parasitol. 102, S23–S24 (2008).
NTD Modelling Consortium Onchocerciasis Group The World Health Organization 2030 goals for onchocerciasis: insights and perspectives from mathematical modelling. Gates Open Res. 3, 1545 (2019).
Mutono, N. et al. Elimination of transmission of onchocerciasis (river blindness) with long-term ivermectin mass drug administration with or without vector control in sub-Saharan Africa: a systematic review and meta-analysis. Lancet Glob. Health 12, e771–e782 (2024).
Boussinesq, M., Fobi, G. & Kuesel, A. C. Alternative treatment strategies to accelerate the elimination of onchocerciasis. Int. Health 10, i40–i48 (2018).
Jacob, B. G. et al. Community-directed vector control to supplement mass drug distribution for onchocerciasis elimination in the Madi mid-North focus of Northern Uganda. PLoS Negl. Trop. Dis. 12, e0006702 (2018).
End Neglected Tropical Diseases in Africa. Control of Neglected Tropical Diseases: Annual Work Plan (1 October, 2017-30 August, 2017) Togo, FY2018. http://web.archive.org/web/20230614035201/https://endinafrica.org/wp-content/uploads/2018/04/Togo-Work-Plan-FY2018-Final.pdf (2017).
Komlan, K. et al. Onchocerca volvulus infection and serological prevalence, ocular onchocerciasis and parasite transmission in northern and central Togo after decades of Simulium damnosum s.l. vector control and mass drug administration of ivermectin. PLoS Negl. Trop. Dis. 12, e0006312 (2018).
Johanns, S. I. et al. Onchocerca volvulus-specific antibody and cellular responses in onchocerciasis patients treated annually with ivermectin for 30 years and exposed to parasite transmission in central Togo. PLoS Negl. Trop. Dis. 16, e0010340 (2022).
World Health Organization & Global Onchocerciasis Network for Elimination. Summary of presentations and discussions of GONE webinar: Togo on its path towards the elimination of onchocerciasis (11 June 2024). https://cdn.who.int/media/docs/default-source/ntds/onchocerciasis/global-onchocerciasis-network-for-elimination-(gone)/webinar-reports/gone-webinar-report-togo-jun-2024-eng-fr.pdf?sfvrsn=5e782f71_3&download=true (2024).
Act to End Neglected Tropical Diseases | West - FY 2023 Work plan-Togo (October 1, 2022-September 30, 2023). https://web.archive.org/web/20240326094740/https://www.actntdswest.org/sites/default/files/inline-files/Act%20West%20FY23%20Workplan-Togo.pdf (2023).
Routledge, I. et al. Modelling the impact of larviciding on the population dynamics and biting rates of Simulium damnosum (s.l.): implications for vector control as a complementary strategy for onchocerciasis elimination in Africa. Parasit. Vectors 11, 316 (2018).
World Health Organization & Global Onchocerciasis Network for Elimination. Niger progrès vers l'élimination de l’onchocercose. GONE webinar (15 December 2022). https://www.who.int/multi-media/details/global-onchocerciasis-network-for-elimination---niger-15-december-2022 (2022).
Lamberton, P. H. L. et al. Onchocerciasis transmission in Ghana: biting and parous rates of host-seeking sibling species of the Simulium damnosum complex. Parasit. Vectors 7, 511 (2014).
Global Forest Watch. Location of tree cover loss in Togo. https://www.globalforestwatch.org/dashboards/country/TGO/?category=forest-change&map=eyJjYW5Cb3VuZCI6dHJ1ZX0%3D (2024).
Wilson, M. D. et al. Deforestation and the spatio-temporal distribution of savannah and forest members of the Simulium damnosum complex in southern Ghana and south-western Togo. Trans. R. Soc. Trop. Med. Hyg. 96, 632–639 (2002).
Ngave, N. F. et al. Reduction in Onchocerca volvulusinfection prevalence and intensity in Logo Health Zone in Ituri, Democratic Republic of the Congo, in the absence of interventions: results of screening for clinical trials of moxidectin vs. ivermectin in 2010 and 2021–2023. https://doi.org/10.21203/rs.3.rs-7151190/v1 (2025).
Stapley, J. N., Basáñez, M.-G., Ramani, A., Walker, M. & Hamley, J. I. D. Modelling the effects of immigration on the re-introduction of onchocerciasis. https://doi.org/10.21203/rs.3.rs-7122282/v1 (2025).
Kura, K. et al. Can mass drug administration of moxidectin accelerate onchocerciasis elimination in Africa?. Philos. Trans. R. Soc. Lond. B Biol. Sci. 378, 20220277 (2023).
O’Hanlon, S. J. et al. Model-based geostatistical mapping of the prevalence of Onchocerca volvulus in West Africa. PLoS Negl. Trop. Dis. 10, e0004328 (2016).
Brown, L. D., Cai, T. T. & DasGupta, A. Interval estimation for a binomial proportion. Stat. Sci. 16, 101–133 (2001).
Filipe, J. A. N. et al. Human infection patterns and heterogeneous exposure in river blindness. Proc. Natl. Acad. Sci. USA 102, 15265–15270 (2005).
Basáñez, M.-G. et al. River blindness: mathematical models for control and elimination. Adv. Parasitol. 94, 247–341 (2016).
Hamley, J. I. D. et al. Structural uncertainty in onchocerciasis transmission models influences the estimation of elimination thresholds and selection of age groups for seromonitoring. J. Infect. Dis. 221, S510–S518 (2020).
EPIONCHO-IBM. An individual-based, stochastic model of the Onchocerca volvulustransmission system. https://github.com/mrc-ide/EPIONCHO.IBM (2025).
Basáñez, M.-G. et al. Effect of single-dose ivermectin on Onchocerca volvulus: a systematic review and meta-analysis. Lancet Infect. Dis. 8, 310–322 (2008).
Plaisier, A. P. et al. Irreversible effects of ivermectin on adult parasites in onchocerciasis patients in the Onchocerciasis Control Programme in West Africa. J. Infect. Dis. 172, 204–210 (1995).
Boatin, B. et al. Patterns of epidemiology and control of onchocerciasis in West Africa. J. Helminthol. 71, 91–101 (1997).
Act to End Neglected Tropical Diseases. West program. Semi-annual report (April 1–September 30, 2021), FY2021. https://www.actntdswest.org/sites/default/files/inline-files/Act%20West%20FY%2021%20Semi%20Annual%20Report%20II.docx.pdf (2021).
Behrend, M. R. et al. Modelling for policy: the five principles of the Neglected Tropical Diseases Modelling Consortium. PLoS Negl. Trop. Dis. 14, e0008033 (2020).
Amaral, L.-J. & Basáñez, M.-G. GitHub repository: mrc-ide/EPIONCHO.IBM. https://doi.org/10.5281/zenodo.17351356 (2025).
World Health Organization & Onchocerciasis Control Programme in West Africa. Expert Advisory Committee, 23rd session: Ouagadougou 23-27 September 2002. Country-specific onchocerciasis control issues: Benin, Burkina Faso, Côte d’Ivoire, Ghana, Guinea, Guinea-Bissau, Mali, Niger, Sénégal, Sierra Leone, Togo. https://iris.who.int/handle/10665/311573 (2002).
World Health Organization & Onchocerciasis Control Programme in West Africa. Report of the evaluation of the ivermectin distribution in four countries: Côte d’Ivoire, Benin, Togo and Ghana. Ouagadougou 4 May 1996 https://iris.who.int/handle/10665/339196 (1996).
End Neglected Tropical Diseases in Africa. Control of Neglected Tropical Diseases: Annual Work Plan (1 October 2015-2030 September 2016) Togo, FY2016. https://web.archive.org/web/20230729131709/https://endinafrica.org/wp-content/uploads/2018/04/Togo-Work-Plan-FY2016.pdf (2015).
Acknowledgements
L.-J.A. was funded by La Caixa Foundation (grant B005782). M.W. and M.-G.B. acknowledge funding by the Bill & Melinda Gates Foundation through the NTD Modelling Consortium (grants OPP1184344 and INV-030046). M.-G.B. acknowledges funding from the MRC Centre for Global Infectious Disease Analysis (grant MR/X020258/1), funded by the UK Medical Research Council (MRC). This UK-funded award is carried out in the frame of the Global Health EDCTP3 Joint Undertaking. Special thanks go to Dr Paul Cantey for his introduction to the Togo team, and to Prof. Robert Colebunders for his support. Dr Natalie Vinkeles Melchers provided useful advice on the assumptions used for the control interventions implemented in Togo that informed Table 1. We also acknowledge Dr Philip Milton for his guidance during the early stages of the work, and Mr Aditya Ramani for calculating the range of annual biting rates for the holoendemic settings presented in Supplementary Information and advice on the Mean Square Error calculations and the preparation of the GitHub link to the code contained in the Zenodo link. This paper is dedicated to the memory of Prof. Yao Kassankogno whose contribution to data availability was invaluable. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The corresponding authors had final responsibility for the decision to submit for publication.
Author information
Authors and Affiliations
Contributions
Conceptualization: L.-J.A., M.-G.B. Data exploration: L.-J.A., J.I.D.H. Data Curation: J.-L.A. Formal analysis: L.-J.A. Investigation and methodology: L.-J.A., J.I.D.H., M.W., M.-G.B. Software: J.I.D.H. Resources: L.-J.A., R.N.B., A.S., M.-G.B. Visualization: L.-J.A., M.W., M.-G.B. Supervision: M.W., M.-G.B. Funding acquisition: L.-J.A., M.-G.B. Project administration: R.N.B., M.-G.B. Writing—original draft: L.-J.A., M.-G.B. Writing—review and editing: L.-J.A., R.N.B., A.S., M.-D.M., K.P., I.G.T., S.A., M.D., P.G., J.I.D.H., M.W., M.-G.B.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Shakir Bilal and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Amaral, LJ., Bronzan, R.N., Seim, A. et al. Reaching elimination of onchocerciasis transmission with long-term vector control and ivermectin treatment in Togo. Nat Commun 17, 779 (2026). https://doi.org/10.1038/s41467-025-67451-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-67451-8