Abstract
Immune checkpoint inhibitor (ICI) resistance in hepatocellular carcinoma (HCC) poses a major therapeutic challenge. Here we present a Phase 2 trial evaluating stereotactic body radiotherapy (SBRT) combined with sintilimab and bevacizumab biosimilar (PD-1/VEGF blockade) to overcome resistance in ICI-refractory HCC. Twenty-one patients with progressive HCC after ICI therapy receive SBRT followed by sintilimab 200 mg and bevacizumab biosimilar 15 mg/kg every 3 weeks. The primary outcome, objective response rate in non-irradiated lesions is 33.3%, with a disease control rate of 66.7%. Median progression-free survival is 6.2 months, and estimated median overall survival is 24.4 months. SBRT achieves 100% local control, with 33.3% experiencing grade 3 or higher adverse events. Proteomic profiling reveals that responders exhibit lower baseline IFN-γ and elevated IL-6, while post-SBRT increases in IFN-γ, IL-2, and IL-6 correlate with improved outcomes. These results indicate that combination of SBRT in ICI-refractory HCC is effective, well-tolerated, and may be guided by cytokine assessment.
Similar content being viewed by others
Introduction
Hepatocellular carcinoma (HCC) accounts for 90% of all primary liver cancers; it ranks as the third leading cause of cancer-related death globally1, with an estimated 5-year survival rate of less than 4% for advanced-stage patients2. Immune checkpoint inhibitors (ICIs), particularly anti-PD-1/PD-L1 agents, have revolutionised systemic therapy, enabling durable responses in a subset of patients3. However, monotherapy yields limited objective response rates (ORRs) of 13–20% (e.g., durvalumab4 and tislelizumab5). Consequently, combination strategies integrating ICIs with antiangiogenic agents (anti-VEGF agents, e.g., bevacizumab), tyrosine kinase inhibitors (TKIs), or anti-CTLA-4 antibodies have emerged as first-line standards. Landmark phase 3 trials, including IMbrave150 (atezolizumab plus bevacizumab; ORR 27.3%6), ORIENT-32 (sintilimab plus bevacizumab biosimilar; ORR 21.0%7), CARES-310 (camrelizumab plus rivoceranib; ORR 26.8%8), HIMALAYA (tremelimumab plus durvalumab; ORR 20.1%4), and CheckMate-9DW (nivolumab plus ipilimumab; ORR 36%9), have demonstrated significantly improved outcomes over sorafenib or lenvatinib.
Despite these advances, at least 60% of patients receive no benefit from combination immunotherapy (IO), and the inevitable immunoresistance remains a formidable constraint to implementing ICIs4,6,8. These patients then become candidates for next-line treatment. Treatment options after progression on front-line ICI regimens remain poorly defined, as most approved second-line agents (e.g., regorafenib) have been validated in sorafenib-pretreated populations10. Currently, the choice for late-line therapy after front- or second-line ICI treatment is primarily empirical, with the shift between ICIs and antiangiogenic agents largely dependent on drug safety, patient comorbidities, physician and patient preferences, and local reimbursement policies.
Currently, data are available from several retrospective studies exploring the feasibility and efficacy of rechallenging with ICIs in the context of prior exposure11,12,13. However, the protocols have mainly focused on the use of different ICI combinations and the replacement of previously unused ICI-based regimens. The main rationale is that inhibiting non-cross-resistance might promote antitumour responses in patients who did not respond to previous ICI therapy14,15,16. These types of regimens are reported to have tolerable toxicity, with ORRs ranging from 16–30% and disease control rates (DCRs) ranging from 40%–55%11,12,13. The therapeutic effectiveness of a simply substituted immunotherapy category remains unsatisfactory, yet prospective data are scarce.
In addition to systemic therapy, locoregional therapies, such as radioembolization and radiotherapy, have been explored and are promising, with favourable results17,18. Radiotherapy has rapidly become a crucial component of multidisciplinary treatment for liver cancer due to advances in technology, resulting in the transition to stereotactic body radiotherapy (SBRT)19. SBRT allows high doses of radiation to be applied to the targeted tumour area in a limited number of fractions with less damage to the surrounding healthy tissues. Recent prospective and retrospective studies have reported that SBRT can achieve high rates of local tumour control at 2 years, ranging from 68–95%20,21, and is an effective local treatment for inoperable early-, intermediate-, and advanced-stage HCCs19. However, many patients experience out-of-field progression, highlighting the need for concurrent systemic disease control22.
Accumulating evidence has demonstrated that radiotherapy rewires the tumour microenvironment (TME) to enhance immunotherapeutic sensitivity through dual mechanisms: stimulating tumour cells to upregulate immune-recognition molecules that potentiate cancer cell detection and antitumour immune activation23,24,25,26,27 while concurrently inducing pro-inflammatory cytokines and chemokines that recruit effector immune cells into the TME24,28. Consequently, SBRT is being actively investigated in combination with systemic agents, particularly ICIs, for the treatment of metastatic lesions across multiple malignancies, including HCC29. Antiangiogenic therapies such as VEGF inhibitors further augment this approach by demonstrating radiosensitizing properties in tumours such as glioma30 and potentiating radiation-induced abscopal effects when combined with ICIs in HCC31. Nevertheless, whether SBRT-mediated tumoral or circulating immune microenvironment in reprogramming can resensitise patients with immunotherapy-refractory HCC to subsequent ICI regimens remains an unanswered clinical question.
Herein, we report the results of the ReUNION-1 study, a phase 2 trial of sintilimab (IBI308) and a bevacizumab biosimilar (IBI305) combined with SBRT after failure of anti-PD-1 treatment in patients with unresectable HCC. In light of the need to identify potential biomarkers and clarify the basis of the tumour response, we analysed serial blood samples obtained before treatment and at time points during treatment and identified many SBRT-induced and efficacy-correlated cytokines. This study will provide evidence for the efficacy of combining SBRT with ICI in patients with ICI-refractory HCC.
Results
Patient characteristics
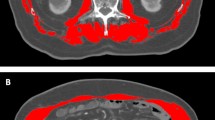
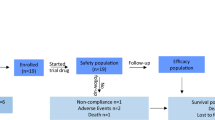
The trial schema is summarised in Fig. 1A and Supplementary Fig. S1A. Between March 2022 and April 2024, a total of 25 patients were screened for eligibility. Among them, one patient had no target lesion other than the proposed radiotherapy lesion; two patients underwent gastroscopy, suggesting untreated or incompletely treated oesophageal or gastric varices with bleeding or a high risk of bleeding; and one patient declined enrolment for personal reasons. Ultimately, 21 patients were enroled (Fig. 1B). The baseline demographics of the patients are summarised in Supplementary Dataset 1 and Supplementary Dataset 2. All 21 patients were included in the intention-to-treat (ITT) analysis, with a cut-off date of 31 August 2024 (Fig. 1B). The median duration of the latest line immunotherapy before enrolment was 7.4 months (interquartile range [IQR], 5.9–18.4 months). The median age was 55 years, and 81.0% of the patients were men. Nine patients had an Eastern Cooperative Oncology Group (ECOG) performance status (PS) of 0, and 12 had a PS of 1. All 21 patients had a Child‒Pugh grade of A at baseline. According to the Chinese Society of Clinical Oncology (CSCO) guidelines, the front-line TKI/PD-1 combination was selected based on availability and data from contemporary trials in China, despite differences in international guidelines. Nineteen patients were treated with at least one line of TKI plus PD-1. Specifically, 73.7% (14/19) of these patients received a combination of lenvatinib and camrelizumab or sintilimab, 21.1% (4/19) received apatinib and camrelizumab, and 5.3% (1/19) received both regimens. Additionally, one patient received anti-VEGF (bevacizumab) plus anti-PD-L1 (atezolizumab) treatment, and another received anti-CTLA-4 plus anti-PD-1 treatment. Details of the fractionation regimen for each patient receiving SBRT treatment (median dose 8 Gy/fraction, range 5–8 Gy in 5 fractions) and the target lesion dose-volume metrics used to assess efficacy, including the median volume receiving ≥5 Gy (V5, which was 0%), can be found in the Source data. Chronic HBV infection was the leading cause of liver disease (n = 18, 85.7%). Most patients were characterised by the following: prior one-line systemic therapy (n = 13, 61.9%), high baseline serum AFP levels (n = 12, 57.1%), multiple intrahepatic lesions (n = 18, 85.7%), and extrahepatic metastasis (n = 15, 71.5%).
A and profile (B). A total of 21 patients were enroled in the study and included in our analysis. Source data were provided as a Source Data file. ORR, objective response rate. DCR, disease control rate. PFS, progression-free survival. OS, overall survival. ECOG, Eastern Cooperative Oncology Group.
Treatment efficacy
With a median follow-up of 9.1 months (IQR, 7.6–16.1), 21 (100%) of the 21 patients were evaluable for the best overall response (BOR). A confirmed partial response (PR) was observed in 7 patients without complete response (CR), contributing to an ORR of 33.3%. Stable disease (SD) was observed in 7 patients, with a DCR of 66.7% (Fig. 2A and Table 1). Notably, reductions in the sizes of irradiated lesions after treatment compared with those at baseline were observed in 20 (95.2%) of the 21 enrolled patients (Fig. 2B). The local-regional control (LRC) rate was 100% (21/21, Fig. 2B), indicating excellent local control of SBRT treatment. Excitingly, lesions outside the irradiation field can also achieve very effective results (Supplementary Fig. S2A, S2B). Durable disease control, defined as continuous tumour control lasting ≥6 months, was observed in patients with both a PR and SD. One patient with a PR according to RECIST v1.1 maintained disease control for more than two years after IBI cessation. Three of the seven patients who experienced a PR remained alive, with no alternative systemic therapy required. Seven patients had progressive disease as their best response.
Maximal change in tumour size from baseline in target (A) non-irradiated lesion(s) and (B) irradiated lesion(s) (n = 21, patients with baseline and at least one post-treatment radiographic evaluation). Pentagrams denoted patients with PD based on new lesion(s) or progression of non-target lesion(s). Each bar represents an individual patient’s response, with colours indicating the response group. C Event-history plots of the treatment strategies of enrolled patients. Time zero is the start of the SBRT treatment. D Kaplan–Meier curves of (upper) progression-free survival (PFS) and (lower) overall survival (OS) in patients (n = 21). Change of individual tumour burden regarding the non-irradiated lesion(s) (E) and irradiated lesion(s) (F) over time from baseline. BOR, best overall response. PR, partial response. CR, complete response. SD, stable disease. PD, progressive disease. Source data were provided as a Source Data file.
By the data cut-off date for the final analysis, 18 patients experienced disease progression, the median progression-free survival (PFS) was 6.2 months (Fig. 2C, D, 95% CI: 3.7–15.1 months), and 2 patients were still under treatment (Fig. 2C). The 6-month and 12-month PFS rates were 57.1% (95% CI, 36.5%–75.5%) and 14.3% (95% CI, 5.0%–34.6%), respectively (Fig. 2D). Additionally, eight patients died, and the estimated median overall survival (OS) was 24.4 months (Fig. 2C, D, 95% CI: 9.2 months - NA). Kaplan-Meier estimates of the 6-month and 12-month OS rates were 95.2% (95% CI 77.3%–99.8%) and 33.3% (95% CI 17.2%–54.6%), respectively. The median duration of response (mDOR) was 5.70 months (95% CI, 2.0–12.4). Overall, local responses were strong and durable (Fig. 2E, F, Supplementary Fig. S2B). Moreover, neither the location of the metastases nor the response to prior ICI therapy nor PFS to prior ICI therapy was significantly associated with treatment response or clinical benefit (Supplementary Fig. S2C, S2D, Supplementary Table S1), indicating the generalisability of this regimen among HCC patients who were refractory to immunotherapy.
Safety
All patients included in the ITT analysis completed radiotherapy and at least one cycle of sequential treatment with sintilimab plus a bevacizumab biosimilar. Treatment-related adverse events (TRAEs) and treatment-emergent AEs (TEAEs) are listed in Table 2 and Supplementary Table S2, respectively. During the therapy period, TRAEs of any grade occurred in 17 of 21 patients (81.0%; see details in Table 2). No unexpected adverse events or treatment-related deaths occurred. Commonly observed TRAEs included decreased lymphocyte count (n = 8, 38.1%), decreased platelet count (n = 8, 38.1%), and increased alanine aminotransferase (n = 8, 38.1%) and aspartate aminotransferase (n = 8, 38.1%). Grade 3 or higher TRAEs (serious TRAEs) occurred in 7 (33.3%) patients, commonly including decreased platelet count (n = 2, 9.5%), decreased white blood cell count, neutropenia (n = 2, 9.5%), decreased platelet count (n = 2, 9.5%), and gastrointestinal haemorrhage (n = 2, 9.5%). Discontinuation of therapy occurred in 3 patients (14.3%) due to serious TRAEs. Two cases (9.5%) of grade 4 gastrointestinal haemorrhage were observed (Patient 4 after 12 cycles; Patient 5 after 10 cycles of sintilimab and bevacizumab biosimilar). Both patients stabilised after endoscopic haemostasis. One participant (4.8%) with grade 3 anaphylaxis was documented (Patient 11 during cycle 2 of sintilimab and bevacizumab biosimilar). The patient gradually recovered after treatment with corticosteroids, and sintilimab was permanently discontinued. One patient (4.8%) withdrew from the trial due to personal preference.
Biomarkers for treatment response and outcomes
Serial Olink proteomics assays for Immuno-Oncology in plasma (see details in Supplementary Dataset 3 and Supplementary Dataset 4), in parallel with flow cytometry analysis for immune lineages in PBMCs, were performed to compare responders and non-responders to identify possible biomarkers that could predict efficacy. A total of 52 samples were obtained and tested at the time points indicated in Supplementary Fig. S1. All samples passed quality control and were included in the analysis (Supplementary Fig. S3A).
At the T1 time point, responders (Rs, defined as patients with PR) exhibited significantly lower enrichment of IFN-γ (p = 0.025) and serum globulin (GLB, p = 0.047) and higher levels of CXCL1 (p = 0.036), TNF-α (p = 0.048), and IL-6 (p = 0.002) than non-responders (NRs, defined as patients with SD and PD; Fig. 3A, D, Supplementary Fig. S3B-C and S3G). Additionally, responders had a significantly higher percentage of CD4+ T cells (p = 0.019), white blood cell (WBC) count (p = 0.038), albumin-to-globulin ratios (A/G, p = 0.025), and neutrophil count (p = 0.046) than non-responders (Supplementary Fig. S3D and S3G). When the duration of response was considered, IFN-γ expression (p = 0.014) and AFP levels (p = 0.007) were lower in patients with clinical benefit response (CBR, patients with SD longer than 6 months and patients with PR) than in nonclinical benefit (NCB) patients (patients with SD less than 6 months and patients with progression disease [PD]), whereas VEGFA (p = 0.033), TWEAK (p = 0.026), CD4 (p = 0.049), IL-6 (p = 0.043), and TNF-α (p = 0.0031) levels were higher (Fig. 3B, D, Supplementary Fig. S3B and S3H). Additional biomarkers or characteristics, including age, sex, ECOG score, China liver cancer (CNLC) staging, enrichment of immune lineages, and serum AFP levels, were analysed for correlation with clinical efficacy (Supplementary Fig. S3I). Unsurprisingly, the levels of the classical tumour biomarker AFP were consistently lower in patients with CBR and long PFS at baseline.
Volcano plots illustrating the baseline expression of differentially expressed proteins from Olink analysis between the (A) Rs and NRs, (B) CBR and NCB groups, and (C) patients with long- and short-PFS. D The NPX values of IFN-γ for pre-treatment samples between the R (n = 5) and NR (n = 12) groups, CBR (n = 9) and NCB (n = 8) groups, and patients with long- (n = 7) and short- (n = 10) PFS. E The univariate logistic regression analysis of each baseline variable with PFS with a two-sided Wald test; hazard ratios (HR) and 95% confidence intervals (CI) were shown. F Kaplan–Meier curves for PFS and OS by baseline IFN-γ level (High, n = 9; Low, n = 8; log-rank test). G Correlation heatmap between protein markers and immune cell subsets. Distance measurement was performed using Euclidean distance. P values were computed using a two-sided t distribution approximation under the null hypothesis of no association. H Volcano plots illustrating the levels of differentially expressed proteins from Olink analysis and different clinical parameters tested by the central laboratory between different timepoints and baseline. I Longitudinal changes in circulating biomarkers from T1 to T3 for PDCD1 and VEGFA (n = 13), Th/Ts, CD8⁺ T-cell percentage, and TNF-α (n = 17 for T1, n = 15 for T3), and Terminal effector CD8⁺ T/Ts (n = 8). J Longitudinal changes from T1 to T4 for ARG1, PGF, TNFRSF4, CX3CL1 and VEGFA (n = 8), TNF-α (n = 17 for T1, n = 10 for T4), lymphocyte count and NLR (n = 21 for T1, n = 18 for T4). R, responders. ALB, albumin. AST, aspartate aminotransferase. A/G, ratio of serum albumin to globulin. Th/Ts, counts of CD4+ T cells/counts of CD8+ T cells. Terminal effector CD8+T/Ts, ratio of CD45RA+CCR7-CD8+T cells to total CD8⁺ T cells. Statistical significance was defined as p < 0.05. P values in volcano plots, box plots were derived from the two-sided Wilcoxon signed-rank test, uncorrected. Box plots indicated the interquartile range, with horizontal lines indicating the median and whiskers extending to the maxima and minima. Source data were provided as a Supplementary Datasets.
Intriguingly, levels of plasma IFN-γ were also decreased in patients with longer PFS than in those with shorter PFS (p = 0.0058, Fig. 3C, D). Survival analysis also showed that patients with a higher level of IFN-γ had poorer PFS and estimated OS (Fig. 3F). The median estimated OS was 24.4 months in the low-IFN-γ group and 9.3 months in the high-IFN-γ group (hazard ratio [HR], 4.12; 95% CI, 1.36–12.46; p = 0.009). The median PFS was 7.8 months in the low-IFN-γ group and 3.7 months in the high-IFN-γ group (HR, 3.19; 95% CI, 1.5–6.78; p = 0.025). Moreover, the existence of extrahepatic metastases (EHS) was correlated with shorter PFS (HR, 5.62; 95% CI: 1.23–25.62) but was not significantly correlated with a lower ORR (EHS vs. non-EHS: 26.7% vs. 50%, p = 0.354), and AFP < 400 μg/L was correlated with better PFS (HR, 0.09; 95% CI: 0.02–0.41) (Fig. 3E, F, Supplementary Fig. S3J and S3L).
We further investigated the dynamics of plasma proteins in patients at the time of BOR and disease progression. The results demonstrated a downward trend in the expression of PDCD1 (p = 0.02) and VEGFA (p = 0.0003), as well as in the ratio of CD4+ T cells to CD8+ T cells (Th/Ts, p = 0.002). In contrast, an increase in the abundance of CD8+ T cells (p = 0.006) and the ratio of terminal effector T cells (defined as CD45RA+CCR7-CD8+ T cells) to CD8+ T cells (p = 0.031) was observed when the patients achieved BOR (Fig. 3I), irrespective of response. Impressively, when the disease progressed, the expression levels of arginase 1 (ARG1) and placental growth factor (PGF) and the neutrophil-lymphocyte ratio (NLR) significantly increased compared with those at baseline, whereas the levels of the T-cell activation marker OX-40 and the T-cell chemotactic factor CX3CL1 significantly decreased (Fig. 3J).
Dynamics of cytokines and immune cells in the circulation during SBRT
Subsequently, we enumerated the differences in the levels of circulating immune cells and immuno-oncology-related proteins before and after SBRT (Fig. 4). A significant SBRT-induced increase in IL15, IL6, and CD70 and a decline of VEGFA, B cells, and total lymphocytes were detected. After SBRT, the median frequency of B cell and lymphocyte counts in unirradiated PBMCs decreased from 12.01% (IQR 8.92–15.72%) to 7.46% (IQR 4.35–11.11%) (p = 0.0017) and from 1.26 (IQR 0.96–1.75) G/L to 0.73 (IQR 0.46–1.14) G/L (p = 0.016), respectively, and the circulating abundance of VEGFA decreased from 9.68 (IQR 9.26–9.94) to 9.28 (IQR 9.04–9.59) (p = 0.0057). In contrast, the concentrations of circulating IL6 (3.756 versus 3.278, p = 0.0442), CD70 (4.090 versus 3.872, p = 0.0088), and IL15 (4.413 versus 4.030, p = 0.0091) and two other specific inflammatory markers, the NLR (3.644 versus 2.090, p = 0.0043) and the PLR (202.7 versus 111.2, p = 0.0058), were significantly elevated after SBRT (Fig. 4A, C, Supplementary Fig. S4A).
Level of (A) differentially expressed proteins from Olink analysis and (B) immune subsets and cytokines tested by the central laboratory before and after SBRT. C Levels of indicated biomarkers before and after SBRT: IL-15, MUC16, IL-6, VEGFA, CD27, and CD70 (n = 15 paired samples); lymphocyte count (n = 20); B-cell proportion in PBMC (n = 14). Volcano plots of SBRT-induced changes (Δ = pre–post) between (D) CBR and NCB groups, (E) long- and short-PFS, and (F) Rs and NRs groups; left: Olink proteins, right: immune subsets/cytokines. G For biomarkers showing consistent and significant divergence in volcano plots, violin plots were compared for their levels across patient subgroups before and after SBRT. Data were presented for IFN-γ, IL-2, and IL-6 (R group, n = 5 paired samples. NR group, n = 5. CBR group, n = 8. NCB group, n = 7. LPFS group, n = 6. SPFS group, n = 9), the IL-17 (R group, n = 3. NR group, n = 10. CBR group, n = 7. NCB group, n = 6. LPFS group, n = 6. SPFS group, n = 7). Statistical significance was defined as p < 0.05. P values in volcano plots, box plots, and violin plots were derived from the two-sided Wilcoxon signed-rank test, uncorrected. Box plots indicated the interquartile range, with horizontal lines indicating the median and whiskers extending to the maxima and minima. For violin plots, the white box represented the interquartile range, the horizontal line inside the box denoted the median, and the whiskers extended to the minimum and maximum values. Source data were provided as a Supplementary Datasets.
While our findings indicated that radiotherapy-induced perturbations in plasma cytokine profiles and immune cell subsets were associated with therapeutic response and PFS, only variations displaying consistent statistical significance across subgroup analyses were deemed to hold substantive relevance to clinical efficacy (Fig. 4A, C). Specifically, SBRT triggered a more prominent augmentation in plasma concentrations of IFN-γ, IL-6, and IL-17A in the CBR group and long-term survival patients (Fig. 4D, E, G) than in the NCB group and patients with short PFS. Additionally, a higher expression of IL-2 in post-SBRT plasma was associated with a better response to the ICI-combined treatment (Fig. 4D, F, G). Moreover, a more pronounced decrease in circulating MMP12 in pre-SBRT and post-SBRT blood samples was associated with improved PFS and clinical benefit (Fig. 4D, E, G). Taken together, inflammation signalling, such as via IFN-γ, IL-6, and IL-2, might be particularly essential in mediating SBRT-triggered immune responses.
Other biomarkers and subgroup analysis
Additional biomarkers or characteristics, including age, sex, ECOG score, prior lines of treatment, expression of PD-L1, infiltration by CD8+ T and CD4+ T cells at diagnosis, existence of liver metastasis, and serum PIVKA-II levels, were analysed for correlation with clinical efficacy (Figure S4B, S4C, Supplementary Table S1, and Supplementary Dataset 1), and no significant differences were observed.
Discussion
Hepatocellular carcinoma exhibits marked tumour heterogeneity and typically progresses after first-line therapies. Current guidelines recommend several second-line regimens based on phase 3 trials, most of which target resistance to sorafenib or lenvatinib monotherapy. Therefore, a critical evidence gap remains regarding the management of advanced HCC that is refractory to ICI-based combinations, representing a pivotal unmet need in contemporary HCC research.
Our literature review revealed that few studies have systematically evaluated radiotherapy-ICI combinations in ICI-refractory or recurrent HCC. Recent investigations focusing predominantly on alternative ICIs combined with TKIs (NCT04985136, rivoceranib + camrelizumab; NCT04696055, regorafenib + pembrolizumab), other targeted therapies (NCT05178043, anti-ALK-1 + nivolumab; NCT03695250, Indoleamine-2,3-dioxygenase (IDO) inhibitor + nivolumab), or TACE combined with regorafenib plus cadonilimab (ChiCTR2400085759) are still ongoing.
Beyond systemic therapies, locoregional interventions remain a cornerstone of HCC management. Globally, an estimated 50–60% of HCC patients receive such interventions during their disease course. For those who develop locoregional progression after systemic therapy, the integration of localised treatments (e.g., SBRT, TACE, or radiofrequency ablation) with ongoing or subsequent systemic regimens may be an alternative strategy to immediate transition to later-line agents32,33. Our phase 2 trial introduces the evidence for SBRT combined with anti-VEGF/PD-1 in anti-PD-1-resistant HCC, yielding three key findings. First, the triplet regimen demonstrated feasibility, with no grade 4–5 radiotherapy-related AEs and grade ≥3 TRAEs in 33.3% of patients—a safety profile comparable to or superior to that of PD-(L)1 inhibitor-SBRT combinations17,34,35. Second, despite limitations in cross-trial comparisons, this regimen demonstrated promising efficacy with an ORR of 33.3% and a DCR of 66.7%, exceeding the 5.9%–11.1% ORR observed for regorafenib plus pembrolizumab in ICI-resistant hepatocellular carcinoma within a prospective trial36. Median progression-free survival (6.2 months) and the estimated overall survival (24.4 months) further support its clinical efficacy. Moreover, the combination conferred consistent benefit in second- or later-line settings for patients with either primary or secondary ICI resistance. Third, SBRT-engaged systemic therapy elicited durable clinical benefit, with three patients sustaining disease control post-treatment—a phenomenon possibly linked to radiation plus ICI-induced immunogenic modulation. Mechanistically, SBRT may enhance tumour antigen exposure and reinvigorate exhausted T-cell populations, synergising with VEGF blockade to normalise immunosuppressive microenvironments. These observations position the SBRT-anti-VEGF/PD1 combination as a well-tolerated, biologically rational strategy for treating anti-PD-1-refractory HCC31.
Although the precise mechanisms underlying innate or acquired resistance to ICIs have not yet been fully elucidated, several hypotheses have emerged based on their known mechanisms of action and preclinical evidence37. These encompass deficient neoantigen processing and recognition38, suboptimal priming/activation of tumour-specific T cells39,40, impaired trafficking of effector T cells to tumour sites41, and the presence of T-cell inhibitory molecules or immunosuppressive cells within the TME42. Our findings align with preclinical evidence demonstrating radiotherapy’s ability to overcome immune exclusion through enhanced antigen presentation and systemic immunomodulation29,43. Radiation preserves native tumour antigen architecture while inducing polyclonal T-cell repertoire diversification and neoantigen release—a dual mechanism enabling abscopal responses29,44. The current study demonstrated that when combined with ICI-based systemic therapy, SBRT achieves a 100% local control rate for irradiated lesions. Notably, the ORR of non-irradiated lesions exceeded both the second-line ORR (7.4%) reported for regorafenib plus pembrolizumab in ICI-resistant populations36 and the historically observed probability of abscopal effects (< 30%)44, clinically supporting a synergistic interaction between radiotherapy and immunotherapy.
Most clinically validated biomarkers require analysis of tumour tissue samples (e.g., MSI-H colorectal cancer, which is a biomarker of immunotherapy responsiveness45). A major translational challenge in HCC biomarker development is the scarcity of tumour tissue, as most diagnoses rely on non-invasive criteria46, especially in patients after heavy treatment. To address this, we implemented longitudinal liquid biomarker profiling via Olink proteomics assay and flow cytometric immune monitoring. We identified remarkable circulating shifts in numerous inflammatory markers after SBRT, as well as markers related to treatment efficacy. Dynamic cytokine changes post-SBRT, particularly increased levels of IFN-γ, IL-2, and IL-6, correlated with prolonged PFS and better responses to combination therapy, indicating a pivotal role of radiotherapy in augmenting acute inflammation and T-cell-mediated immune responses and offering a practical alternative to tissue-based biomarkers.
As a central regulator of antitumour immunity, IFN-γ mediates T-cell infiltration, MHC/PD-L1 upregulation, and immunosuppressive microenvironment remodelling47. While preclinical models implicate radiotherapy-induced IFN-γ surges in systemic tumour control48,49, clinical validation in human cohorts remains limited. Our study bridges this gap by showing that baseline suppression of plasma IFN-γ was significantly associated with improved clinical outcomes in heavily pretreated HCC patients, including increased ORRs, improved disease control, and prolonged PFS. Paradoxically, elevated baseline TNF-α, a cytokine typically linked to tumour progression47, has emerged as a positive predictor of therapeutic response, with a post-treatment slump correlating with sustained clinical benefit. We postulated that the patients enrolled in this study had an immunosuppressive microenvironment resilient to ICI therapy, which may have evinced impairment or exhaustion of IFN-γ as well as the accumulation of TNF-α; thus, it is reasonable that the therapy abated TNF-α, and patients who had lower IFN-γ at baseline, as well as those whose IFNs can be provoked by SBRT, are more likely to experience abscopal effects and have a better prognosis after subsequent IBI treatment. These findings suggest that baseline IFN-γ exhaustion and TNF-α enrichment may characterise ICI-resistant HCC, where SBRT-mediated cytokine resetting might restore therapeutic susceptibility.
IL-2 dynamics further corroborated the immunostimulatory role of radiation, with post-SBRT elevation in responders and the CBR group aligning with its established function in T-cell activation47. Our data revealed that SBRT-induced IL-2 increases were more pronounced in treatment-responsive patients, reinforcing the premise that SBRT augments immunotherapeutic antitumour effects via T-cell activation. Interestingly, our findings concerning IL-6 conflict with those of previous reports, indicating that inhibiting IL-6 improves the prognosis of patients with HCC treated with radiotherapy and teslizumab plus anlotinib50. In our study, patients with elevated baseline IL-6 or post-radiation increases demonstrated superior survival, which is consistent with the null survival benefit of the TRIPLE-R trial from IL-6R blockade51 and the preclinical evidence of IL-6 dependency in radiotherapy-immunotherapy synergy in patients with cervical52 and pancreatic53 cancer. These divergent observations underscore the dual and heterogeneous nature of IL-6-dependent immunoregulation, necessitating disease-specific biomarker frameworks54. Taken together, our exploration of plasma biomarkers indicated a potential mechanism by which radiotherapy combined with immunotherapy could increase the abscopal response rate and confer a probable benefit.
While non-invasive biomarkers like the widely applicable CRAFTY score55 and inflammatory markers such as the neutrophil-to-lymphocyte ratio (NLR)56 and platelet-to-lymphocyte ratio (PLR)57,58 have been reported to predict immunotherapy responses in treatment-naïve HCC patients, our study failed to validate these findings in heavily pretreated patients. Intriguingly, the NLR and PLR spiked after SBRT, yet this pattern did not differ among the subgroups with distinct responses. These results indicated that predictive markers for efficacy are distinguishable between ICI-resistant and untreated populations, and the incorporation of SBRT may alter the predictive efficacy of the established biomarkers for immunotherapy.
Our findings should be interpreted in the context of several limitations. The single-arm phase 2 design with a cohort of 21 patients, while statistically predefined, limits subgroup analyses and generalizability. Critically, the absence of paired tumour biopsies prevents direct investigation of SBRT-induced immune remodelling within the tumour microenvironment. Furthermore, the regional preference for TKI/PD-1 combinations over atezolizumab/bevacizumab in the first-line setting may affect the applicability of our results to other healthcare systems. These gaps highlight the need for future randomized trials and translational studies with sequential tissue sampling to confirm and build upon our findings.
Nevertheless, the inclusion of high-risk patients—those with primary ICI resistance, multisite metastases, and extreme AFP elevation—enhances real-world applicability. These populations are generally more debilitated than those in most clinical studies, making the demonstration of safety and efficacy in this setting particularly meaningful. Anti-PD-1 refractory hepatocellular carcinoma still represents an area of unmet need, as treatment options are restricted to registered clinical trials or salvage options, such as anti-PD-1/PD-L1/CTLA4 antibodies, antiangiogenic drugs, or both combined. Therefore, the therapeutic options presented in this study retain considerable potential for populations that lack effective alternatives.
Although the primary endpoint (ORR ≥ 40%) was not achieved, the 33.3% response rate substantially exceeds historical controls for anti-PD-1-refractory HCC, signalling potential clinical utility that warrants validation in larger cohorts, especially given the limited options available in this setting. Together, these results substantiate the clinical efficacy of this combined treatment in HCC patients and link its effects to complex systemic immune responses, the latter requiring further confirmation. Key questions remain regarding optimal radiotherapy parameters (dose, target volume), selection and timing of immunotherapy, and biomarker-driven patient selection. Mechanistic studies dissecting radiation-induced inflammatory responses and antitumour immune reactions are warranted. Collaborative trials integrating multiomics biomarker platforms will be essential to validate these findings and refine combinatorial strategies.
Methods
Study design and participants
The open-label, single-arm, phase 2 study was conducted in accordance with the Declaration of Helsinki and all patients provided written informed consent before enrolment. The institutional review boards of Wuhan Union Hospital approved the study (Ethics number: [2022]0136-01). The trial was registered with chictr.org.cn (ChiCTR2200056068) on 31 January 2022, and the published study protocol59 was provided in the Supplementary Information. Analyses were conducted as planned in the preregistration. Patients did not receive financial compensation for participation in the study.
Patients aged 18 years or over with an ECOG performance status of ≤1 and clinically or pathologically diagnosed hepatocellular carcinoma (HCC) that had failed to respond to previous anti-PD-1 immunotherapy were eligible for enrolment. According to the Response Evaluation Criteria in Solid Tumour version 1.1 (RECIST v1.1), measurable disease was required if at least one tumour was amenable to SBRT treatment at a dose of 25–50 Gy in 5 fractions, and another measurable tumour lesion was present that did not undergo radiotherapy for efficacy assessment.
Other inclusion criteria included having an A Child-Pugh grade, a life expectancy of ≥3 months, and sufficient bone marrow and organ function. The exclusion criteria included a history of any other malignancy, metastases to the central nervous system, previous radiotherapy, autoimmune or interstitial lung disease, or serious comorbidities, such as congestive heart failure or uncontrolled diabetes.
Procedures
Patients underwent SBRT once daily for five consecutive days, targeting a single tumour lesion. Sintilimab (200 mg, Innovent Biologics, Suzhou, China) combined with bevacizumab biosimilar (15 mg/kg, Innovent Biologics, Suzhou, China) was administered intravenously every 3 weeks, starting 1–2 weeks after completion of SBRT. Treatment continued until disease progression, the occurrence of intolerable toxicity, patient withdrawal, death, or for a maximum duration of 24 months. All patients included in the ITT analysis had finished radiotherapy and at least one cycle of sequential treatment with sintilimab plus a bevacizumab biosimilar. Complete follow-up, including physical examination, information on adverse events, and radiographic evaluation, was performed every 9 weeks. Key laboratory tests and flow cytometry analysis of immune lineages in PBMCs were performed in the clinical laboratory of Wuhan Union Hospital.
Outcomes
The primary endpoint was the ORR, defined as the percentage of patients who had a RECIST v1.1-defined CR or PR; only non-irradiated (abscopal) lesions were RECIST eligible60. The secondary endpoints included DCR, PFS (time from the date of enrolment to the date of disease progression or death), OS (time from the date of enrolment to the date of death), and LRC rates. The PFS or OS times of patients who were still progression-free or alive at the time of the last follow-up were censored. Responders (Rs) were defined as patients with BOR of PR. Non-responders (NRs) were defined as patients with BOR of SD or PD. Clinical benefit responders (CBR) were defined as patients who achieved a BOR of PR or SD lasting longer than 6 months. Non-clinical benefit (NCB) was defined as patients with a BOR of PD or SD lasting less than 6 months. Long progression-free survival (LPFS) and short progression-free survival (SPFS) were defined as patients with PFS longer or shorter than the median PFS, respectively. LRC rates were defined as the proportion of patients without local progression of all irradiated target lesions from the baseline value according to the RECIST 1.1 assessment. Outfield lesions were defined as lesions outside the 500 cGy dosimetric line. The safety assessment spanned from informed consent through follow-up. Investigators evaluated adverse event (AE) incidence and severity per the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. AEs, including TEAEs, TRAEs and serious TRAEs, were monitored during treatment and up to 30 days after the last dose according to the study protocol. TRAEs were meticulously evaluated by investigators through a safety assessment protocol aligned with China NMPA’s Technical Guideline for Causality Assessment of Adverse Events in Drug Clinical Trials (2024 edition, https://www.cde.org.cn/main/news/viewInfoCommon/0a5ae4924881321c07cce100e99f2a5c). Three core criteria had to be fulfilled: temporal plausibility (onset/duration patterns matching known toxicity kinetics), biological gradient (dose/exposure-dependent changes confirmed via pharmacokinetic monitoring), and the exclusion of confounders (active ruling out disease progression, portal hypertension, and concurrent infections through serial imaging and laboratory profiling). The specific criteria are presented in Supplementary Dataset 5.
Plasma immune proteomics
Plasma samples were collected at the indicated time points according to the timeline described in Supplementary Fig. 1A, 1B. The main time points include baseline (T1)after SBRT and before IBIs treatment (T2), at best overall response (T3), and at disease progression (T4). All samples were tested in a single run in the same batch. Proteins in the plasma samples were measured using the Olink® Target Immuno-Oncology Panel (Olink Proteomics, Sweden) according to the manufacturer’s instructions, which includes the detection of 92 proteins by the proximity expansion assay (PEA) with 1 μL of each sample. In brief, pairs of oligonucleotide-labelled antibody probes bind to their target proteins. If the two probes come into proximity, their oligonucleotides hybridise in a pairwise manner.
The introduction of DNA polymerase initiates a proximity-dependent DNA polymerisation event, generating a unique PCR target sequence. This sequence is subsequently detected and quantified using a microfluidic real-time PCR instrument (Biomark HD, Fluidigm). The resulting Ct data undergo quality control and normalisation against a set of internal and external controls. The final assay output is reported as normalised protein expression (NPX) values, which are arbitrary units on a log2 scale, where higher values indicate greater protein expression. The four internal controls are designed to emulate and monitor the distinct stages of the PEA process. These include two incubation/immuno controls, an extension control, and a detection control. The internal controls are added to all samples as well as to the external controls, serving for quality assurance and data normalisation. The external controls comprise a negative control, which is utilised for determining the limit of detection (LOD), and triplicate interplate controls (IPCs), which are used for data normalisation.
Quality control of the data occurs in two stages. First, the run’s quality is assessed by calculating the standard deviation for both the detection control and the incubation/immuno controls. For a run to pass quality control, this standard deviation must be less than 0.2. Second, each sample undergoes quality control by comparing its detection control and one of the incubation control results against the run median. All assay validation data, including detection limits and intra- and inter-assay precision data, are available on the manufacturer’s website (www.olink.com). The complete dataset for the Olink analysis has been uploaded as Supplementary Dataset 4.
Immunohistochemical (IHC) analysis of the tumour immune microenvironment
We analysed 11 FFPE samples from pre-enrolled HCC primary or metastatic lesions: (a) 9 primary HCCs (8 surgical resections, 1 ultrasound-guided biopsy) and (b) 2 lung metastases (1 metastasectomy, 1 CT-guided biopsy). Tissues were collected at Union Hospital, Tongji Medical College, Huazhong University of Science and Technology (from 2019–2023; see details in Source data) with histopathological confirmation. IHC analysis was performed on 4-μm FFPE sections using antibodies against CD4 (Clone 4B12, Dako #M7310), CD8 (Clone C8/144B, Dako #M7103), and PD-L1 (Clone 22C3, Dako #SK006). Following EDTA-based antigen retrieval (pH 9.0, 95 °C, 20 min) and overnight primary antibody incubation at 4 °C, the sections were stained with EnVision FLEX/HRP (Dako) and DAB, and haematoxylin was used for counterstaining. The quantitative assessment included the following: 1) CD4⁺/CD8⁺ T-cell density (cells/mm²) counted in five random 400× HPFs spanning intratumoural and invasive margin regions and 2) PD-L1 combined positive score (CPS) = PD-L1⁺ cells (tumour/lymphocytes/macrophages)/viable tumour cells×100 (CPS ≥ 1 = positive, per the FDA 22C3 pharmDx criteria). Experienced pathologists performed all assessments.
Statistical analysis
This trial adopted Simon’s two-stage optimal design to evaluate the primary endpoint of ORR. Historical benchmarks defined the null (π₀ = 15%)7 and alternative (π₁ = 40%) response hypotheses. Early termination criteria stipulated trial discontinuation if ≤1 response occurred among the initial 9 evaluable participants (stage I). Progression to stage II disease required ≥2 responses, with final efficacy declared if >5/19 patients achieved responses (α = 0.05, power=80%). To account for 10% attrition, 21 patients were enrolled.
The efficacy-evaluable population comprised participants who received ≥1 treatment cycle with post-baseline imaging. The safety analysis included all patients who were administered ≥1 dose with follow-up toxicity assessments. Survival endpoints (PFS/OS) were analysed via Kaplan-Meier estimates with log-rank tests, whereas response rates (ORRs/DCRs) were calculated using Clopper-Pearson exact 95% CIs—Cox proportional hazard models generated HRs for survival comparisons.
For further analysis, subgroup comparisons between responders and non-responders were conducted using the exact Wilcoxon rank-sum test. The relationship between potential biomarkers and the radiographic response was assessed via Spearman’s rank correlation coefficient. Additionally, biomarker levels before and after treatment were compared using the paired Wilcoxon signed-rank test to evaluate treatment effects. The Wilcoxon rank-sum test was used for intergroup comparisons. Correlations between biomarkers were quantified via Spearman’s ρ, with longitudinal changes assessed using the paired Wilcoxon signed-rank test. All analyses were conducted bidirectionally (SPSS Statistics 26; R, version 4.3.1) at p < 0.05, with visualisation implemented in R and GraphPad Prism 9. The following R packages were employed: stats package for statistical testing; ggplot2 package for generating box plots, volcano plots and violin plots; swimplot package for patient-time trajectory plots; linkET package for correlation analysis; forestploter package for univariable forest plots; and the survminer and survival packages for Kaplan-Meier survival analysis and visualization. Data analysis of proteomics was conducted using the Olink NPX Signature Software (version 1.16.0) for quality control (QC) of Olink data.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Individual deidentified participant data (including data dictionaries) that underlie the results reported in this article are available from the corresponding author at junxue_xh@hust.edu.cn. A response to data access requests will be provided within 30 days from the date of the request. Restrictions on the use of controlled data, including data sharing and publication, will be governed by the terms of the data use agreements. The data supporting the findings of this study are available in the accompanying Supplementary Information. Raw data on plasma immune proteomics are provided in the PRoteomics IDEntifications (PRIDE) server under the accession code PAD000004. The study protocol has already been published59. Source data are provided with this paper.
References
Ezequiel, M. et al. Hepatocellular carcinoma: Epidemiology, diagnosis and treatment. JHEP Rep. 7, 101571 (2025).
Siegel, R. L., Giaquinto, A. N. & Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 74, 12–49 (2024).
Llovet, J. M. et al. Immunotherapies for hepatocellular carcinoma. Nat. Rev. Clin. Oncol. 19, 151–172 (2022).
Abou-Alfa, G. K. et al. Tremelimumab plus Durvalumab in unresectable hepatocellular carcinoma. NEJM Evid. 1, EVIDoa2100070 (2022).
Qin, S. et al. Tislelizumab vs Sorafenib as first-line treatment for unresectable hepatocellular carcinoma: a phase 3 randomized clinical trial. JAMA Oncol. 9, 1651–1659 (2023).
Finn, R. S. et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N. Engl. J. Med 382, 1894–1905 (2020).
Ren, Z. et al. Sintilimab plus a bevacizumab biosimilar (IBI305) versus sorafenib in unresectable hepatocellular carcinoma (ORIENT-32): a randomised, open-label, phase 2-3 study. Lancet Oncol. 22, 977–990 (2021).
Qin, S. et al. Camrelizumab plus rivoceranib versus sorafenib as first-line therapy for unresectable hepatocellular carcinoma (CARES-310): a randomised, open-label, international phase 3 study. Lancet 402, 1133–1146 (2023).
Yau, T. et al. Nivolumab plus ipilimumab versus lenvatinib or sorafenib as first-line treatment for unresectable hepatocellular carcinoma (CheckMate 9DW): an open-label, randomised, phase 3 trial. Lancet 405, 1851–1864 (2025).
Pinter, M., Scheiner, B. & Pinato, D. J. Immune checkpoint inhibitors in hepatocellular carcinoma: emerging challenges in clinical practice. Lancet Gastroenterol. Hepatol. 8, 760–770 (2023).
Scheiner, B. et al. Efficacy and safety of immune checkpoint inhibitor rechallenge in individuals with hepatocellular carcinoma. JHEP Rep. 5, 100620 (2023).
Wong, J. S. L. et al. Ipilimumab and nivolumab/pembrolizumab in advanced hepatocellular carcinoma refractory to prior immune checkpoint inhibitors. J. Immunother. Cancer 9, https://doi.org/10.1136/jitc-2020-001945 (2021).
Roessler, D. et al. Ipilimumab and nivolumab in advanced hepatocellular carcinoma after failure of prior immune checkpoint inhibitor-based combination therapies: a multicenter retrospective study. J. Cancer Res Clin. Oncol. 149, 3065–3073 (2023).
Fukumura, D., Kloepper, J., Amoozgar, Z., Duda, D. G. & Jain, R. K. Enhancing cancer immunotherapy using antiangiogenics: opportunities and challenges. Nat. Rev. Clin. Oncol. 15, 325–340 (2018).
Wahid, M. et al. Pembrolizumab’s non-cross resistance mechanism of action successfully overthrown ipilimumab. Crit. Rev. Oncol. Hematol. 111, 1–6 (2017).
Wei, S. C. et al. Distinct cellular mechanisms underlie anti-CTLA-4 and anti-PD-1 checkpoint blockade. Cell 170, 1120–1133.e1117 (2017).
Hu, Y. et al. Efficacy and safety of stereotactic body radiotherapy combined with Camrelizumab and Apatinib in patients with hepatocellular carcinoma with portal vein tumor thrombus. Clin. Cancer Res. 29, 4088–4097 (2023).
Lencioni, R. et al. EMERALD-1: A phase 3, randomized, placebo-controlled study of transarterial chemoembolization combined with durvalumab with or without bevacizumab in participants with unresectable hepatocellular carcinoma eligible for embolization. J. Clin. Oncol. 42, LBA432 (2024).
Hu, Y. et al. The role of stereotactic body radiotherapy in hepatocellular carcinoma: guidelines and evidences. J. Natl. Cancer Cent. 2, 171–182 (2022).
Kim, T. H. et al. Proton beam radiotherapy vs. radiofrequency ablation for recurrent hepatocellular carcinoma: A randomized phase III trial. J. Hepatol. 74, 603–612 (2021).
Scorsetti, M. et al. The challenge of inoperable hepatocellular carcinoma (HCC): results of a single-institutional experience on stereotactic body radiation therapy (SBRT). J. Cancer Res. Clin. Oncol. 141, 1301–1309 (2015).
Lee, Y. H., Tai, D., Yip, C., Choo, S. P. & Chew, V. Combinational immunotherapy for hepatocellular carcinoma: radiotherapy, immune checkpoint blockade and beyond. Front. Immunol. 11, 568759 (2020).
Liu, S. et al. Radiotherapy remodels the tumor microenvironment for enhancing immunotherapeutic sensitivity. Cell Death Dis. 14, 679 (2023).
Guo, S. et al. Radiation-induced tumor immune microenvironments and potential targets for combination therapy. Signal Transduct. Target. Ther. 8, 205 (2023).
Reits, E. A. et al. Radiation modulates the peptide repertoire, enhances MHC class I expression, and induces successful antitumor immunotherapy. J. Exp. Med. 203, 1259–1271 (2006).
Mollà, M. et al. Relative roles of ICAM-1 and VCAM-1 in the pathogenesis of experimental radiation-induced intestinal inflammation. Int. J. Radiat. Oncol. Biol. Phys. 57, 264–273 (2003).
Huang, J. et al. Combination of pembrolizumab and radiotherapy induces systemic antitumor immune responses in immunologically cold non-small cell lung cancer. Nat. Cancer. https://doi.org/10.1038/s43018-025-01018-w (2025).
Dovedi, S. J. et al. Fractionated radiation therapy stimulates antitumor immunity mediated by both resident and infiltrating polyclonal T-cell populations when combined with PD-1 blockade. Clin. Cancer Res. 23, 5514–5526 (2017).
Podesta, C., Kayani, M., Goody, R. & Samson, A. Combination treatment of HCC with SBRT and immune checkpoint inhibition. Crit. Rev. Oncol. Hematol. 192, 104191 (2023).
Kashiwagi, S. et al. Perivascular nitric oxide gradients normalize tumor vasculature. Nat. Med. 14, 255–257 (2008).
Sheng, H. et al. Antiangiogenic treatment facilitates the abscopal effect of radiation therapy combined with anti-PD-1 in hepatocellular carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 121, 534–546 (2025).
Llovet, J. M. et al. Locoregional therapies in the era of molecular and immune treatments for hepatocellular carcinoma. Nat. Rev. Gastroenterol. Hepatol. 18, 293–313 (2021).
Wang, H. et al. Efficacy and safety of radiotherapy to delay second-line systemic therapy in patients with oligoprogressive hepatocellular carcinoma: study protocol of a multicentre, single-arm, phase II trial. Ther. Adv. Med Oncol. 17, 17588359251334538 (2025).
Li, Z. et al. Neoadjuvant tislelizumab plus stereotactic body radiotherapy and adjuvant tislelizumab in early-stage resectable hepatocellular carcinoma: the Notable-HCC phase 1b trial. Nat. Commun. 15, 3260 (2024).
Chen, Y. et al. Envafolimab plus lenvatinib and transcatheter arterial chemoembolization for unresectable hepatocellular carcinoma: a prospective, single-arm, phase II study. Signal Transduct. Target Ther. 9, 280 (2024).
El-Khoueiry, A. B. et al. International, open-label phase 2 study of regorafenib plus pembrolizumab in patients with advanced hepatocellular carcinoma (HCC) previously treated with immune checkpoint inhibitors (ICI). J. Clin. Oncol. 42, 4007 (2024).
Desert, R. et al. Improving immunotherapy for the treatment of hepatocellular carcinoma: learning from patients and preclinical models. npj Gut Liver 2, 8 (2025).
McGranahan, N. et al. Clonal neoantigens elicit T cell immunoreactivity and sensitivity to immune checkpoint blockade. Science 351, 1463–1469 (2016).
Tay, C., Tanaka, A. & Sakaguchi, S. Tumor-infiltrating regulatory T cells as targets of cancer immunotherapy. Cancer Cell 41, 450–465 (2023).
Kamada, T. et al. PD-1(+) regulatory T cells amplified by PD-1 blockade promote hyperprogression of cancer. Proc. Natl. Acad. Sci. USA 116, 9999–10008 (2019).
Zhu, Y. et al. Disruption of tumour-associated macrophage trafficking by the osteopontin-induced colony-stimulating factor-1 signalling sensitises hepatocellular carcinoma to anti-PD-L1 blockade. Gut 68, 1653–1666 (2019).
Xie, Q., Zhang, P., Wang, Y., Mei, W. & Zeng, C. Overcoming resistance to immune checkpoint inhibitors in hepatocellular carcinoma: challenges and opportunities. Front. Oncol. 12, 958720 (2022).
Li, H. et al. External radiotherapy combined with sorafenib has better efficacy in unresectable hepatocellular carcinoma: a systematic review and meta-analysis. Clin. Exp. Med. 23, 1537–1549 (2023).
Ngwa, W. et al. Using immunotherapy to boost the abscopal effect. Nat. Rev. Cancer 18, 313–322 (2018).
Fan, A. et al. Immunotherapy in colorectal cancer: current achievements and future perspective. Int J. Biol. Sci. 17, 3837–3849 (2021).
Villanueva, A. Hepatocellular carcinoma. N. Engl. J. Med 380, 1450–1462 (2019).
Yi, M. et al. Targeting cytokine and chemokine signaling pathways for cancer therapy. Signal. Transduct. Target Ther. 9, 176 (2024).
Popp, I., Grosu, A. L., Niedermann, G. & Duda, D. G. Immune modulation by hypofractionated stereotactic radiation therapy: therapeutic implications. Radiother. Oncol. 120, 185–194 (2016).
Mills, B. N. et al. Stereotactic body radiation and interleukin-12 combination therapy eradicates pancreatic tumors by repolarizing the immune microenvironment. Cell Rep. 29, 406–421.e405 (2019).
Lin, Y. D. et al. Effect of cytokines on advanced hepatocellular carcinoma prognosis receiving radiotherapy and tislelizumab plus anlotinib: a single-center phase II clinical trial. Sci. Rep. 14, 11486 (2024).
Chen, I. M. et al. Phase 2 study of ipilimumab, nivolumab, and tocilizumab combined with stereotactic body radiotherapy in patients with refractory pancreatic cancer (TRIPLE-R). Eur. J. Cancer 180, 125–133 (2023).
Beyranvand Nejad, E. et al. IL-6 signaling in macrophages is required for immunotherapy-driven regression of tumors. J. Immunother. Cancer 9, e002460 (2021).
Perez-Penco, M. et al. The antitumor activity of TGFβ-specific T cells is dependent on IL-6 signaling. Cell. Mol. Immunol. 22, 111–126 (2025).
Fisher, D. T., Appenheimer, M. M. & Evans, S. S. The two faces of IL-6 in the tumor microenvironment. Semin Immunol. 26, 38–47 (2014).
Scheiner, B. et al. Prognosis of patients with hepatocellular carcinoma treated with immunotherapy - development and validation of the CRAFITY score. J. Hepatol. 76, 353–363 (2022).
Zhu, A. X. et al. Molecular correlates of clinical response and resistance to atezolizumab in combination with bevacizumab in advanced hepatocellular carcinoma. Nat. Med. https://doi.org/10.1038/s41591-022-01868-2 (2022).
Wang, J. H. et al. The prognostic value of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio in patients with hepatocellular carcinoma receiving atezolizumab plus bevacizumab. Cancers 14, https://doi.org/10.3390/cancers14020343 (2022).
Wu, Y. L. et al. Neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios as prognostic biomarkers in unresectable hepatocellular carcinoma treated with atezolizumab plus bevacizumab. Cancers 14, https://doi.org/10.3390/cancers14235834 (2022).
Zhang, J. et al. Efficacy and safety of SBRT combined with sintilimab and IBI305 in patients with advanced HCC and previously failed immunotherapy: study protocol of a phase 2 clinical trial. BMJ open 14, e077903 (2024).
McBride, S. et al. Randomized phase II trial of nivolumab with stereotactic body radiotherapy versus nivolumab alone in metastatic head and neck squamous cell carcinoma. J. Clin. Oncol. 39, 30–37 (2021).
Acknowledgements
The authors thank the patients, their families, the investigators, and all investigational site members involved in this study. The sintilimab and bevacizumab biosimilar used in this study were provided by Innovent Biologics (Suzhou) Co., Ltd. Support for plasma proteomic data processing was provided under the direction of the authors by Nanjing Simcere Medical Laboratory Science Co., Ltd. Responsibility for all opinions, conclusions, and data interpretation lies with the authors. This work was supported by the National Natural Science Foundation (82172977, 82573809, J.T.; 82573541, U21A20376, J.X.), Beijing Xisike Clinical Oncology Research Foundation (Y-HS202102-0152, J.X.), Project of Key Research and Development of Hubei Province (2022BCA021, J.X.), Science Fund for Distinguished Young Scholars of Hubei Province (2025AFA104, J.T.), Open Research Fund of Hubei Key Laboratory of Precision Radiation Oncology (jzfs002, J.T.), and the Open Research Fund of the Ministry of Education Key Laboratory on Signalling Regulation and Targeting Therapy of Liver Cancer (2023-MEKLLC-MS-002, J.T.).
Author information
Authors and Affiliations
Contributions
X.J., Z.T., W.G. and T.J. were the principal investigators and participated in the trial design, study management, data and toxicity review, review of the report, supervision of the study, and final approval of the report. T.J., X.J., Y.Y.Q. and W.B.C. contributed to the writing of the protocol, recruitment and treatment of the patients, data and trial management, data analysis and interpretation, and writing of the manuscript. T.J., X.J., Y.Y.Q., L.Z.Y. and W.B.C. participated in the recruitment and treatment of the patients, data and trial management, and report preparation. T.J., X.J., L.D.E., Z.J.F. and Z.D.J. were responsible for the statistical analysis and interpretation as well as data review. W.Z.L., L.Y.Q., G.M.H. and C.L. contributed to the analysis of proteomics and immune patterns and data visualisation. All authors reviewed and approved the final draft. All authors had full access to all the data in the study. The corresponding author holds final responsibility for the decision to submit the manuscript for publication.
Corresponding authors
Ethics declarations
Competing interests
L.Y.Q. and G.M.H. declare employment with Nanjing Simcere Medical Laboratory Science Co., Ltd. The remaining authors declare that they have no competing interests.
Peer review
Peer review information
Nature Communications thanks Yuhao Xie, Ignacio Melero, Antonio D’Alessio and the other anonymous reviewer(s) for their contribution to the peer review of this work. [A peer review file is available].
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tang, J., Yang, Y., Liu, D. et al. Stereotactic body radiotherapy with sintilimab and bevacizumab biosimilar in anti-PD-1 refractory hepatocellular carcinoma: the ReUNION-1 phase 2 trial. Nat Commun 17, 823 (2026). https://doi.org/10.1038/s41467-025-67528-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-67528-4