Abstract
Anticipated reactions to stressful situations are vital for the survival and well-being of organisms, and abnormal reactions results in stress-related disorders. The neuropeptide oxytocin is a key modulator ensuring well-adapted stress responses. Oxytocin acts on both neurons and astrocytes, but the molecular and cellular mechanisms mediating stress response remain poorly understood. Here, we focus on the amygdala, a crucial hub that integrates and processes sensory information through oxytocin-dependent mechanisms. Using an acute stress paradigm in mice, genetic and pharmacological manipulations combined with proteomic, morphological, electrophysiological and behavioral approaches, we reveal that oxytocinergic modulation of the freezing response to stress is mediated by transient Gαi-dependent retraction of astrocytic processes, followed by enhanced neuronal sensitivity to extracellular potassium in the amygdala. Our findings elucidate a pivotal role for astrocytes morphology-dependent modulation of brain circuits that is required for proper anticipated behavioral response to stressful situations.
Similar content being viewed by others
Introduction
Anticipated reactions to potentially dangerous situations are essential for animal survival, but their dysregulation can lead to pathological conditions such as post-traumatic stress disorders and chronic anxiety1,2. Rodents display similar responses to stress as stereotyped freezing behavior3. Understanding the cellular mechanisms mediating stressor detection and the resulting behavioral and physiological adaptations is crucial to design effective treatments for anxiety and other stress-related disorders. Despite notable progress in this field, the cellular and molecular mechanisms mediating adequate physiological and behavioral responses to stress remain elusive.
Importantly, stress response requires coordinated activity of various brain regions. Among these, the amygdala plays a central role, as lesions of this brain structure impair sensory information interpretation across mammalian species, including rodents, monkeys, and humans4. Projections from the amygdala basolateral complex (BLA) to the central nucleus (CeA) are particularly critical for stress-related behavior5. Sensory information is integrated in the BLA and transmitted to the CeA, which in turn modulates stress reactions as freezing behavior in rodents3,6. The activity of CeA neurons, especially in the laterocapsular part (CeL/C), is regulated by endogenous neuromodulators, notably oxytocin (OT)7,8.
OT is synthesized by neurons located in the hypothalamic accessory, supraoptic and paraventricular nuclei (PVN) that project to the pituitary and via axonal collaterals to various regions of the mammalian central nervous system9. OT modifies the balance between excitation and inhibition and regulates the salience of emotionally and socially relevant stimuli. In the context of the behavioral response to stress, this allows rapid switching between states of low and high arousal10,11,12. Within the CeL/C, OT modulates neuronal network activity and influences freezing. This highlights its role in regulating stress responses, and especially the acute stress-induced freezing in rodents6,7,11,12,13,14,15,16.
Remarkably, OT signaling involves not only neurons but also astrocytes as they express oxytocin receptors (OTR)17. Astrocytes are key regulators of neuronal function in the central nervous system18,19. They can detect and respond to neuronal activity by eliciting calcium transients; they release gliotransmitters, which in turn modulate synaptic transmission and they regulate neuronal excitability through neurotransmitter uptake and K+ buffering20. Consequently, astrocytes regulate the activity of neuronal circuits underlying behaviors in a brain-region specific manner17,20. Since OT is released in response to stressful conditions within the amygdala of rats21, this study aimed at determining whether and how astrocytes play a role in the oxytocinergic modulation of behavioral response to stress in vivo.
Here, we show that freezing response to acute stress is mediated by OTR- and Ca2+-dependent structural and functional changes in astrocytes, which in turn modulate neuronal activity in the CeL/C. Therefore, neuromodulatory signaling in astrocytes contributes to stress-induced neuronal hyperexcitability in the CeL/C.
Results
To investigate the role of astrocytes in oxytocinergic modulation of behavioral response to stress in vivo, we adapted a paradigm of acute stress-induced freezing in adult mice22. Animals were exposed to five 20-second-long sound stimuli followed by electrical foot shocks. These paired sound-shock sequences were randomly delivered over 10 minutes, and freezing was quantified during the 20 seconds following the electric foot shocks to assess the immediate stress response (Fig. 1a).
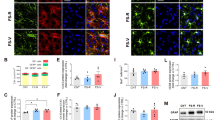
a Protocol for stress induction: Mice were submitted to five electrical foot shocks (1s, 0.6mA) at variable time intervals during a 10-min session. Naïve mice were placed in the same cage without foot shocks. GFAP OTR cKO model: rAAV-GFAP-Cre was injected bilaterally in OTR-lox mice to knock out the OTR in astrocytes 3 weeks before experiments. b Freezing behavior in WT and GFAP OTR cKO mice during the stress protocol. Left: Development of the freezing response during the habituation (Hab.) and after each electric shock exposure (Shock (n)). Right: quantification of the time spent freezing during the 20s following electric foot shock exposure in WT and GFAP OTR cKO mice. nWT=16 mice, nGFAP OTR cKO=13 mice. c Plasma corticosterone levels in control and GFAP OTR cKO mice in naïve and stress male mice. nWT=11 mice, nWT stress=10 mice, nGFAP OTR cKO=10 mice, nGFAP OTR cKO stress=9 mice. d Monitoring calcium activity in Paraventricular hypothalamic (PVN) OT neurons in freely moving animals. GCaMP6f was targeted to PVN OT neurons by crossing Ai148 mice with OT-Cre mice. GRIN lenses were implanted above the PVN, and GCaMP fluorescence was captured thanks to a miniscope. Scale bar = 100µm. e Proportion of neurons showing calcium transients in response to each electric foot shock, to some electric foot shocks, or showing no response. Peristimulus graph showing the mean variation of GCaMP6f fluorescence in PVN OT neurons before and after an electric foot shock. The purple square shows the tone duration, and the yellow bar indicates the moment of the foot shock. The pink trace is an overlay of neuronal response following the electric foot shock, while the grey one was obtained in mice exposed to a tone with no foot shock. f Heat map showing calcium responses to electric foot shocks of all PVN OT neurons tested. Calcium levels are encoded by color. n = 4 mice, 86 neurons. g Microdialysis probe implanted in the amygdala for repeated OT sampling in naïve conditions, during the acute stress protocol, and after CD1 mice returned to the home cage. h Quantification of the OT amount per microdialysate. n = 9 mice. i Monitoring calcium activity in CeA astrocytes of freely moving mice. GCaMP was targeted to CeA astrocytes or to OTR-expressing CeA astrocytes following injection of rAAV-GFAP-GCaMP6s in WT mice (n= 5) or rAAV-GFAP-DIO-GCaMP6s in OTR-Cre mice (n = 4), respectively. Scale bar = 100µm. j Peristimulus graph showing the mean variation of GCaMP fluorescence intensity in all CeA astrocytes (left panel) or in OTR-expressing astrocytes (right panel). The grey square highlights the tone duration, and the yellow bar indicates the application of the foot shock. The blue and purple traces are overlays of astrocyte response following the electric foot shock, while the grey one was obtained in mice exposed to a tone with no foot shock. k GCaMP was expressed in all CeA astrocytes by injecting rAAV-GFAP-GCaMP in WT mice. Mice received i.p. injections of saline just before the first stress induction (Trial 1, T1), and the OTR antagonist L368 or its vehicle (Saline) just before a second stress induction (Trial 2, T2). l Left: GCaMP6s fluorescence intensity traces illustrating an increase in Ca2+ signaling in CeA astrocyte following the foot shock during the first trial (grey traces), or the second one (blue or orange traces). Right: normalized integrals of the fluorescence signal. nsaline=5 mice, nL368=8 mice. Data are expressed as mean across animals ± SEM. Detailed statistics can be found in Source Data Table 1. *p < 0.05, **p < 0.01, ***p < 0,001, ****p < 0.0001. Drawings created in BioRender: Charlet, A. (2025) https://BioRender.com/bkv8qa0.
Astrocyte OTR in the CeL/C contributes to stress-induced freezing
We previously reported that roughly 20% of CeL/C astrocytes express OTR, and that those astrocytes are connected to neighbouring astrocytes through GAP junctions17. To directly assess whether astrocytic OT signaling influences the stress-induced freezing, we induced cell-specific loss of OTR by injecting an AAV encoding the Cre recombinase under the control of the glial fibrillary acidic protein (GFAP) promoter (rAAV-GFAP-Cre) to target astrocytes in the CeL/C of OTR fl/fl mice (GFAP OTR cKO, Fig. 1a and Supplementary Fig. 1a)17,23. This viral vector had a specificity between 95.8%-98,2% and an efficiency between 75,8% and 87,3% as indicated by astrocyte markers S100b, Aldh1l1 and GS (Supplementary Fig. 1a–d). In addition, the vector causes a significant decrease in the number of astrocytes expressing OTR without altering OTR expression in other cell types (p < 0.0001 and 0.7812, Supplementary Fig. 1e). We observed that GFAP OTR cKO mice failed to exhibit freezing behavior during the stress session as compared with WT control mice (p < 0.0001, Fig. 1b and Supplementary Fig. 1f). Intriguingly, this effect was not observed in female mice (p = 0.449, Supplementary Fig. 1g), indicating a putative sex-specific role of astrocytic OTR in the behavioral response to stress, which prompted us to focus on males. To test if the hormonal response to stress was similar between WT and GFAP OTR cKO mice, we quantified plasmatic corticosterone levels just after the stress protocol and the day before (to avoid any effect of the stress due to blood sampling). We observed a similar increase in plasmatic corticosterone levels in stressed versus naïve animals in both WT and GFAP OTR cKO mice (p = 0.677, Fig. 1c and Supplementary Fig. 1i), suggesting that the absence of freezing behavior was not due to an altered physiological response to stress. As observed by other studies24, females showed a higher corticosterone increase than males following the stress session (p = 0.023, Supplementary Fig. 1j)
Importantly, we tested whether the absence of freezing of GFAP OTR cKO mice could stem from changes in their nociceptive sensitivity. We performed a comprehensive analysis of nociception and pain sensitivity in these animals and did not observe any significant differences in thermal cold (p = 0.808), thermal heat (p = 0.297) and mechanical (p = 0.853) nociception, nor in spontaneous pain as tested with the conditioned place preference test compared to WT mice (p = 0.577, Supplementary Fig. 1k, l). We next examined whether the absence of freezing could be explained by GFAP OTR cKO mice displaying depressive-like symptoms. This appears unlikely as GFAP OTR cKO and WT mice performed similarly in the sucrose preference test and in the direct social interaction test (Supplementary Fig. m-n).
Acute stress induces PVN OT neurons activation, CeA OT release, and astrocyte recruitment
Based on the behavioral characterization, astrocytes are involved in oxytocinergic modulation of stress-induced freezing in vivo. To decipher the underlying cellular mechanisms, we tested whether the acute stress paradigm activated PVN OT neurons and CeA astrocytes in vivo. To this end, we first recorded the calcium activity of OT neurons in the PVN in freely moving mice during the acute stress paradigm (Fig. 1d). To monitor individual neuronal calcium responses, we genetically targeted the calcium sensor GCaMP6f to OT neurons by breeding floxed GCaMP6f transgenic mice (line Ai148D) with OT-Cre mice25. Gradient-Index (GRIN) lenses were chronically implanted above the PVN for imaging of calcium activity using a micro-endoscope in freely-moving mice (Fig. 1d, Supplementary Fig. 2a–c). Electric foot shocks induced calcium transients in 78% of the GCaMP6f-expressing OT neurons (n = 67/86; Fig. 1e, f). Interestingly, PVN OT neurons also responded to stress induced by tail suspension (p = 0.011) but not by cage shaking (p = 0.750, Supplementary Fig. 2d, e). These findings established that the acute stress protocol reliably activated OT neurons in the hypothalamus.
Next, we investigated whether acute stress triggered OT release in the CeA, which is a well-known target region of PVN OT neurons7. To this end, we used CD1 mice, which have larger brains, and implanted microdialysis probes within the CeA region to measure the release of endogenous OT in freely moving mice (Fig. 1g). We found that stress induced by five randomly presented auditory tone-shock pairings induces an increase in CeA OT concentration (p = 0.0162, Fig. 1h). This result showed that the foot shock-induced stress paradigm induces endogenous OT release in the CeA.
We then tested whether this acute stress paradigm induced a Ca2+ response in CeA astrocytes. To do so, we injected a rAAV carrying GCaMP6s under the control of the GFAP promoter (rAAV-GFAP-GCaMP6s) in the CeA and implanted optic fibers above the CeA to image Ca2+ activity in astrocyte population using fiber photometry in freely-moving mice (Fig. 1i). Similar to PVN OT neurons, electric foot shocks reliably increased Ca2+ transients in GCaMP6s-expressing astrocytes (p = 0.010, Fig. 1j and Supplementary Fig. 2h). To test if this response can be observed in OTR-expressing astrocytes that represents ~25% of CeL/C astrocytes (Supplementary Fig. 1e)17, we aimed to express GCaMP in OTR-expressing astrocytes. To validate our targeting, we first injected a rAAV-GFAP-DIO-mScarlet in OTR-Cre mice (Supplementary Fig 2f, g). Following this verification, we injected OTR-Cre mice26 with a Cre-dependent AAV targeting astrocytes and expressing GCaMP6s. Following the electrical foot shock, the fluorescence significantly increased (p = 0.0313, Fig. 1j and Supplementary Fig. 2h). Next, we aimed to test the involvement of OTR in this response. Given the non-ratiometric nature of the GCaMP6s, we opted for a longitudinal approach and achieved pharmacological inhibition of the OTR by injecting i.p. the antagonist L368 that cross the blood-brain barrier (Fig. 1k)27. We observed a significant reduction of the foot shock-induced Ca2+ response following L368, suggesting the involvement of the OTR in the stress-induced astrocyte Ca2+ signaling (Fig. 1l, Supplementary Fig. 2i, p = 0.451, ncontrol = 5, nL368 = 8). These results established that the acute stress paradigm activated OTR-expressing astrocytes and the general astrocyte population in the CeA.
Acute stress induces changes in the amygdala proteome
To gain insight into the molecular and cellular mechanisms induced by acute stress, we performed label-free quantitative mass spectrometry proteomics on amygdala samples snap frozen ~15 minutes following the acute stress protocol (Fig. 2a). Principal component analysis clearly separated naïve from stressed mice (Fig. 2b), indicating an extensive stress-induced remodeling of the amygdala proteome. Two large clusters of proteins were either up- or down-regulated upon stress (Fig. 2c and Supplementary Fig. 3). Further analysis revealed that up- and down-regulated proteins are involved in axonal activity and morphological remodeling of astrocytes, indicating that stress modifies protein levels in CeA astrocytes. Indeed, profound expression changes were observed among 59 astrocyte proteins (Supplementary Fig. 3), showing, among others, an increase in GFAP cytoskeleton protein. Analysis of gene ontology (GO) terms and protein-protein interactions revealed multiple signaling components, pathways, transmitter receptors and ion channels, most of them related to synaptic activity (Fig. 2d). Gene set enrichment analysis (GSEA) indicated that differentially expressed proteins are involved in multiple metabolic and signaling processes, as well as in structural cellular components (Supplementary Fig. 3b-c). Among them, we found a significant change in intermediate filament proteins (Fig. 2e, f). Collectively, these data revealed that acute stress induces changes in astrocyte proteome, notably upregulation of intermediate filaments.
a Experimental design for proteome analysis. b Principal component analysis (PCA) of the proteomic data. Dots represent proteomic data sets from individual control (sky blue) and stressed mice (dark blue). c Heat map and hierarchical clustering of differentially expressed proteins. d Network based on Protein-Protein Interaction (PPI) Map. Network of enriched terms obtained by analyzing top-300 genes by Metascape. Each node represents a term, and the node size is directly proportional to the number of input proteins grouped into each term. Node color denotes cluster identity. Gene ontology (GO) terms with a similarity score >0.5 are connected by an edge and the edge thickness represents the similarity score. e 1D Enrichment Analysis of gene set enrichment analysis (GSEA) data. Each point represents a gene set obtained after GSEA analysis of the proteomic data, displayed as the average fold change of the proteins within that gene set as a function of the adjusted p-value of the GSEA analysis. Significant GSEA terms (adjusted p-value < 0.05) are highlighted in red. f Bar plot of normalized enrichment scores (NES) obtained from GSEA analysis using the ranked fold changes and adjusted p-values for all the differentially expressed proteins. The bar chart highlights the cytoskeletal-related pathways, with pathways exhibiting both positive and negative enrichment under stress conditions. Detailed statistics can be found in Source Data Table 2.
OTR signaling mediates acute stress-induced changes in astrocyte morphology and K+-dependent neuronal depolarization
We next investigated whether the acute stress-induced changes in proteins entail changes in astrocyte morphology. To achieve this, we injected a rAAV-GFAP-GFP in the CeL/C of naïve and stressed mice. Using the IMARIS software, we performed three-dimensional reconstructions of GFP-expressing astrocytes from confocal images (Fig. 3a, b). We found that the acute stress protocol decreases the morphological complexity of astrocytes, which displayed reduced process ramification as measured by Sholl analysis (p < 0.0001, Fig. 3b, c and Supplementary Fig. 4b). Notably, this morphological response was absent from GFAP OTR cKO mice (p = 0.167, Fig. 3b, c and Supplementary Fig. 4b), suggesting an OTR-dependent mechanism. Stress-induced changes in morphology were accompanied by an OTR-dependent increase in GFAP expression in CeL/C astrocytes (p < 0.001, Fig. 3d and Supplementary Fig. 4b–d). Consistent with this result, the GFAP volume measured by immunofluorescence was inversely correlated to astrocyte process ramification (r² = 0.569, p < 0.0001, Fig. 3e and Supplementary Fig. 4b–d). Together, these results revealed that acute stress concomitantly induces OTR-dependent upregulation of GFAP in astrocytes and a reduction of their morphologic complexity.
a Imaging of CeL/C astrocytes in different mouse lines using targeted expression of eGFP using rAAV-GFAP-eGFP injection. Right: reconstruction of CeL/C astrocytes based on their eGFP expression. Scale bar = 20µm. b Representative images of reconstructed astrocytes from WT and GFAP OTR cKO mice under naïve and stress conditions. Scale = 20µm. Representative observation made in 8 different mice. c Estimation of astrocyte ramification using Sholl analysis. Left: Diagram depicting the Sholl method. Middle: Sholl plot showing the number of astrocyte processes crossing the concentric Sholl circles as a function of the distance from the soma. n = 8 mice per group, nnaïve=4032, nstress=3402, nnaïve GFAP OTR cKO=2843, nstress GFAP OTR cKO=2968 astrocytes. Right: Comparison of astrocyte ramification in different experimental groups based on mean maximum Sholl values. n = 8 mice. d Quantification of GFAP volume after immunohistochemical staining and 3D reconstruction. n = 8 mice. Scale bar = 400 and 25µm. e Correlation between astrocyte ramification revealed by GFP and their GFAP volume. nnaïve=333 astrocytes, nstress=489 astrocytes. Scale = 10µm. Scatter plot showing a negative correlation between GFAP volume and peak Sholl values in individual astrocytes. f Upper panel: cartoon illustrating whole-cell patch-clamp recordings from CeL/C neurons in acute slices. Lower panel: micrograph of a slice during a patch-clamp experiment. The puff pipette is in the left and the patch clamp pipette is on the right. Scale bar = 50µm. g Spontaneous spiking frequency of CeL/C neurons measured in loose-patch recordings. nnaïve=9, nstress=9. h Spider plot showing the log p-values given by the comparison of different electrophysiological features. Current steps (IStep) were used to measure the spike-firing threshold, the number of spikes at the second active step (nSpike), and the latency to the first spike. K+ puff (30mM in aCSF), was used to measure the difference in spike firing (δSpike), the amplitude of the K+-induced depolarization (δVm), and its rise/decay constants. i Amplitude of neuronal membrane depolarization during the K+ puff. nnaïve=20, nstress=20, nnaïve GFAP OTR cKO=17, nstress GFAP OTR cKO=21 neurons. Data are expressed as mean across animals ± SEM for bar graph c & d, across astrocytes for scholl analysis and correlation plot, and across neurons for (g)–(i). Detailed statistics can be found in Source Data Table 3. *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001. Drawings created in BioRender: Charlet, A. (2025) https://BioRender.com/bkv8qa0.
Given that astrocytic OTR are involved in acute stress-induced freezing (Fig. 1) and knowing that astrocytic regulation of neuronal excitability can depend on their morphology27,28, we first evaluated the spontaneous neuron activity by performing loose cell-attached recordings of CeL/C neurons (Fig. 3f). We observed an increased spontaneous firing frequency in neurons that come from stressed mice compared to their naïve counterpart (p = 0.028, Fig. 3g). To understand the origin of this increased activity of CeL/C neurons, we then characterized the excitability of CeL/C neurons in brain slices of naive and stressed mice using whole-cell patch-clamp recordings. We found that current injections induced similar voltage responses in neurons from WT naïve and stressed mice (prheobase=0.300, platency to spike = 0.372, pnumber of spikes = 0.521, Fig. 3h and Supplementary Fig. 5a). Neuronal excitability also depends on extracellular K+ concentration, which is ~3 mM in resting conditions and could increase to 10-12 mM during neuronal activity and to even higher values in pathological conditions19. To mimic these conditions, we applied a K+ puff (30 mM) to induce depolarization of neuronal membrane potential (Fig. 3h, i and Supplementary Fig. 5b), and after verification that such application does not lead to cell death (Supplementary Fig. 5f), we observed higher depolarization in stressed mice compared to naïve mice (p = 0.0002). The kinetics of this depolarization was modified at the level of decay (prise=0.086, pdecay < 0.0001, Fig. 3h and Supplementary Fig. 5b) and the K+ puff did not elicit higher spike frequency in neurons from stressed versus naive animals (pnumber of spikes = 0.094, Fig. 3h and Supplementary Fig. 5b). Importantly, we found that this stress-induced increase in K+-dependent neuronal depolarization was not significant in GFAP OTR cKO, as compared to naive mice (pdepolarization=0.062, prise = 0.088, pdecay = 0.025, pnumber of spikes = 0,676, Fig. 3h, i and Supplementary Fig. 5b). To investigate whether acute stress modify extracellular K+ sensitivity in a defined subset of CeL/C neurons, we analyzed their electrophysiological features with a principal component analysis. However, we failed to identify any characteristics associated with the increased in extracellular K+ sensitivity observed after stress (Supplementary Fig. 5c, d). Furthermore, we demonstrated that under stress conditions, the various effects described above did not alter hyperpolarizing potassium currents following an action potential (IAHP) (Supplementary Fig. 5e). We further analyzed the timing of those changes and found that the stress-induced depolarization observed initially was no longer detectable after three days (Supplementary Fig. 5g). Taken together, the results revealed that OTR-dependent signaling in astrocytes mediates acute stress-induced morphologic changes in astrocytes and an increase of K+-dependent depolarization of CeL/C neurons.
Stress-induced changes in astroglial coverage of synapses and regulation of their efficacy within the amygdala CeL/C
The acute stress-induced changes in astrocyte morphology raised the question of whether this also affected the astrocytic coverage of synapses. This is of physiologic importance as such changes can influence circuit function29. To address this, we injected rAAV-GFAP-GFP in the CeL/C to label astrocytes. We then stained for vGluT1, vGluT2, and Homer1 to visualize pre- and postsynaptic elements of excitatory glutamatergic synapses (Fig. 4a, b and Supplementary Fig. 6a, b). Confocal microscopy revealed a significantly reduced number of synapses contacted by astrocytes from mice subjected to acute stress compared to those from naïve mice (p = 0.001, Fig. 4c). These results suggest that acute stress prompted the retraction of astrocytic processes away from synapses. Notably, this difference was not observed in GFAP OTR cKO mice, indicating that this change in synaptic coverage was mediated by astrocytic OTR (p = 0.998, Fig. 4c). As a complementary method to visualize the coverage of synapses by astrocytic processes, we used transmission electron microscopy. In support of our observations by light microscopy, we observed a lower density of astrocytic processes surrounding synapses in CeL/C from stressed mice compared to naïve ones (p < 0.0001; Supplementary Fig. 6c). Together, these observations indicated that acute stress initiated an OTR-dependent retraction of astrocytic processes contacting synapses in the CeL/C.
a Imaging of CeL/C astrocytes using targeted expression of eGFP using rAAV-GFAP-eGFP. b Representative micrograph showing synapses and surrounding astrocytic processes following IMARIS-based 3D reconstruction. Arrowheads show a putative synapse contacted by an astrocyte. eGFP: astrocyte, vGlut1/2: post-synaptic, Homer1: pre-synaptic c Quantification of the contacts between synapses and astrocyte processes per reconstructed astrocyte. n = 5 animals in each group. d Analysis of BLA-to-CeL/C synaptic transmission using optogenetic stimulation of the BLA axons and recordings of the EPSCs in CeL/C neurons in naïve and stressed mice. Injection of AAV-CaMKII-ChR2 was performed in the BLA of WT mice, and a second pair of injection containing rAAV-GFAP-Cre was done in the CeL/C of OTR-lox mice. 3 weeks after, we measured the currents evoked by BLA neurons activation in CeL/C neurons of presynaptic BLA neurons. Lower right panel: representative recording of EPSCs in CeL/C neurons evoked during photostimulation. e, f Time course and quantification of photostimulation-evoked EPSCs amplitude. n (neurons, mice): WT naive=14, 5, WT stress=19, 5, GFAP OTR cKO naïve=17, 9, GFAP OTR cKO stress=13, 5. g Experimental approach to record K+ currents in CeL/C astrocytes following optogenetic stimulation of BLA neurons in naïve and stressed mice. AAV-CaMKII-ChR2 was injected in the BLA of mice, and 3 weeks after, the currents evoked by BLA neuron activation were measured in CeL/C astrocytes. Lower right panel: representative recording of photstimulation-evoked currents in control condition, after the application of Ba2+, and the subtraction of both (IK). h, i Time course and quantification of photostimulation-evoked astrocytic current induced by BLA neurons stimulation before (black) and after a 15-min incubation in Ba2+ (200µM, dark grey). The subtracted K+ current is shown in light grey. n (astrocyte, mice): WT naive=13,6, WT stress=11, 8, GFAP OTR cKO naïve=11, 6, GFAP OTR cKO stress=8, 6. j Quantification of Kir4.1 levels in naïve and stressed mice using 1mm punches of mice CeA was done on 400µm slices from naïve or stressed mice. k Image of representative Western blot reacted with antibody against Kir4.1 in six animals from the indicated experimental groups. l Quantification of Kir4.1 levels in amygdala punches. nnaïve=6, nstress=8 mice. Data are expressed as mean across animals ± SEM for (c) and (l), and as mean across cells ± SEM for (e), (f), (h), and (i). Detailed statistics can be found in Source Data Table 4. *p < 0.05, **p < 0.01. Drawings created in BioRender: Charlet, A. (2025) https://BioRender.com/bkv8qa0.
We next tested whether the reduced coverage of synapses by astrocytic processes induced by acute stress affected their transmission properties. Previous studies suggested a major role of basolateral amygdala (BLA) to CeL/C projections in the adaptation to stress-induced freezing29,30. To optogenetically activate BLA neurons, we injected rAAV-CaMKII-ChR2-eYFP in the BLA (Fig. 4d). After visual verification of the BLA location of eYFP expression, we stimulated ChR2-expressing BLA neurons by light (5 ms pulse, 20 Hz during 5 s) and recorded the synaptic response of CeL/C neurons using patch-clamp recordings in acute brain slices of both naïve and stressed animals (Fig. 4d and Supplementary Fig. 6d-g). Acute stress increased the overall amplitude of the evoked excitatory postsynaptic currents (eEPSCs) in CeL/C neurons as compared to naïve mice (pamplitude=0.037, Fig. 4e, f), similarly to31. To test the involvement of OTR signaling in astrocytes, we performed the same recordings in naïve and stressed GFAP OTR cKO mice. Under these conditions, the stress-induced increase in synaptic responses was abolished (p = 0.948, Fig. 4e, f). Interestingly, the amplitude of eEPSCs triggered by the two first photostimulations was not different between naïve and stressed mice (Fig. 4e, Supplementary Fig. 6f, p = 0.107). However, the BLA-evoked eEPSCs amplitudes in the middle of the stimulation train was increased in stressed compared to naïve mice (Fig. 4f, Supplementary Fig. 6f, p = 0.031), indicating an incremental mechanism. This could arise from an accumulation of extracellular K+ with repeated stimulations. Crucially, we quantified synaptic parameters but did not observe any difference between naïve and stressed animals of the same genotype (paired pulse ratio: p = 0.3590, failure rate: p = 0.4091, sEPSCs amplitude: p = 0.7292, sEPSCs frequency: p = 0.5835, eEPSCs rise time: p = 0.3824, eEPSCs decay time: p = 0.5280, Supplementary Fig. 6h). This indicates that direct synaptic plasticity is not responsible in the observed increased eEPSCs amplitude following stress exposure.
To test the implication of astrocytic K+ buffering in this phenomenon, we recorded K+ currents in CeL/C astrocytes following photostimulation of BLA neurons. We patch-clamped SR101-labelled astrocytes (Fig. 4g) and verified their astrocytic identity based on their electrophysiological response (IV curve, Supplementary Fig. 7a)32. To isolate the current due to K+ buffering, we subtracted from the current evoked by photostimulation of BLA neurons a second current recorded in the presence of Ba2+ (200 µM) to block K+ currents (Fig. 4g). This experiment revealed that the amplitudes of stimulation-induced K+ currents were significantly reduced in stressed mice compared with naïve mice (p = 0.030, Fig. 4h, i), indicating impaired K+ buffering in astrocytes. Since we cannot rule out a potential Ba2+ effect on neurons, we reproduced the experiment in GFAP OTR cKO mice. Crucially, the stimulation-induced K+ currents were not altered in GFAP OTR stressed mice compared with naïve mice (p = 0.159, Fig. 4h-i). A decrease in astrocyte K+ buffering capacity could arise from their process retraction but also from reduced expression of inwardly rectifying Kir4.1 channels19,33. Thus, we extracted membrane proteins of CeA samples from stressed and naïve mice and detected Kir4.1 protein levels by western blot analysis (Fig. 4j and Supplementary Fig. 7b-c). We found that the acute stress paradigm reduced Kir4.1 levels in CeA samples as compared with naïve controls (Fig. 4j-l). These results suggested that acute stress induces a retraction of astrocytic processes from synapses and diminishes the K+ buffering capacity at the BLA-to-CeL/C synapses, thereby modifying the strength of this connection.
These findings indicate that acute stress initiates an OTR-dependent retraction of perisynaptic astrocytic processes, leading to a diminished K+ buffering capacity at the BLA-to-CeL/C synapses, enabling adequate behavioral stress response.
Identification of G proteins mediating OTR-dependent effects in astrocytes
We next aimed to identify the signaling pathways downstream of OTR underlying changes in Ca2+ transients in astrocytes after acute stress.
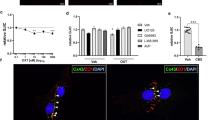
Previous proteomic analysis suggested that acute stress modifies GPCR and cAMP signaling pathways (Fig. 2d, Supplementary Fig. 3c and 8a). Thus, we studied the subtype of G-proteins activated by OTR in astrocytes using bioluminescence resonance energy transfer (BRET)-based biosensors34 (Fig. 5a). We found that upon OT binding, OTR couple to both Gαi and Gαq in cultured astrocytes (Fig. 5b and Supplementary Fig. 8b). We next identified the G proteins coupling OTR to Ca2+ transients in CeL/C astrocytes by combining OGB1-based calcium imaging and pharmacological manipulations (Fig. 5c). We applied biased OTR agonists that favor either Gαi (atosiban) or Gαq (carbetocin) pathways or a full agonist ([Thr4,Gly7]-oxytocin, TGOT) that activates both pathways34,35. We then measured calcium transients in SR101-positive astrocytes in brain slices in the presence of TTX to block action potential-dependent neuronal activity (Fig. 5c). All three agonists increased the frequency and AUC of Ca2+ transients compared to baseline activity (frequency: pTGOT = 0.024, patosiban = 0.001, pcarbetocin = 0.003; AUC: pTGOT = 0.002, patosiban = 0.0007, pcarbetocin = 0.0008, Fig. 5d and Supplementary Fig. 8c). Similar proportions of cells responded to each agonists (~50%, Supplementary Fig. 8c). The Gαq protein blocker YM254890 (500 nM, 30 min pre-incubation) prevented the carbetocin- and TGOT-induced calcium signaling (Carbetocin: Ca2+ transient frequency: p = 0.296, TGOT: Ca2+ transient frequency: p = 0.561, Supplementary Fig. 8d). The Gαi protein blocker pertussis toxin (PeTX, 5 µg/mL, 5 h pre-incubation) abolished calcium activity induced by atosiban (Ca2+ transient frequency: p = 0.316, Supplementary Fig. 8e), while the response to TGOT remained unaffected (Ca2+ transient frequency: p = 0.006, Supplementary Fig. 8e). In order to characterize the Ca2+ transients induced by OTR-Gαi and OTR-Gαq pathways, we compared the parameters of calcium event triggered by different G protein activation. We found that the rise constant and amplitude of Ca2+ transients induced by atosiban were lower as compared to those induced by carbetocin (prise=0.016, pamplitude = 0.013, Fig. 5e, f). Taken together, these pharmacological data indicated that activation of Gαi and Gαq proteins triggers distinct Ca2+ transient signatures in astrocytes.
a Verification of the biased OTR activation triggered by atosiban and carbetocin. BRET donors and acceptors were expressed in primary astrocyte cultures from mice brains temporal lobe. b Induced changes in BRET ratio due to OTR activation by OT in live astrocytes. nPBS=3 and nOT=6 in each condition. c Upper left panel: cartoon showing the recording of Ca2+ activity of CeL/C astrocytes. Lower left panel: micrograph showing SR101-identified astrocytes. Right: pseudo-colorized picture of the OGB1 Ca2+ indicator fluorescence before and after bath application of OTR agonists. Scale = 100µm. d Upper panel: mean trace of the normalized to the baseline OGB1 fluorescence recorded in CeL/C astrocytes in response to TGOT (500nM, blue), Carbetocin (500nM, light green), or Atosiban (500nM, blue). The vertical line indicated the start of agonist application. Lower panel: frequency of Ca2+ transients in CeL/C astrocytes. nTGOT=57 astrocytes over 13 slices, natosiban=49 astrocytes over 9 slices, ncarbetocin=54 astrocytes over 13 slices. e Comparison of calcium activity triggered by different OTR agonists. Spider plots showing the p-value of the comparison between the basal state and after the application of different agonists for different calcium signaling parameters. f Right: spider plot showing the p-value of a one-way ANOVA to compare calcium events triggered by the three different agonists. Left: Bar plots showing mean rise time and height of calcium transients triggered by indicated OTR agonists normalized to baseline values. Data are expressed as mean across cell ± SEM. Detailed statistics can be found in Source Data Table 5. *p < 0.05, **p < 0.01, ***p < 0.001. Drawings created in BioRender: Charlet, A. (2025) https://BioRender.com/bkv8qa0.
OTR-Gαi signaling mediates CeL/C astrocytes morphological reorganization and controls neuron sensitivity to extracellular K+
Given the involvement of OTR-Gαi in CeL/C astrocytes Ca2+ activity, we sought to elucidate the potential role of OTR-Gαi-mediated Ca2+ transients in CeL/C astrocytes. To do so, we incubated brain slices with different OTR agonists for 1 h and assessed astrocyte GFAP levels by histology (Fig. 6a). While the full (TGOT) and Gαi- biased (atosiban) agonists increased CeL/C astrocytes GFAP volume (pTGOT=0.007, patosiban < 0.001), the Gαq-biased agonist (carbetocin) did not (pcarbetocin=0.404) (Fig. 6b). This suggested an involvement of Gαi, but not Gαq, in the modification of the GFAP cytoskeleton. Similarly, TGOT increased the K+-induced depolarization of CeL/C neurons (p = 0.003), an effect prevented by the co-incubation of TGOT with the OTR antagonist dOVT (p > 0.999) or by genetic deletion of OTR (GFAP OTR cKO, p > 0.999) (Fig. 6c-d and Supplementary Fig. 9a–c). These results indicate that astrocytic OTR-Gαi mediates the OT-induced hypersensitivity of CeL/C neurons to extracellular K+. Similarly, atosiban but not carbetocin increased the K+-evoked depolarization of CeL/C neurons (patosiban=0.022, pcarbetocin > 0.999; Fig. 6d and Supplementary Fig. 9b, c).
a Analysis of OTR-Gαi activation on CeL/C astro-neuronal network. Brain slices were incubated for 1h in the different OTR agonists before fixation and GFAP staining. b Left: Representative micrograph showing GFAP staining of CeL/C astrocytes in presence of the indicated agonists. Scale = 400 and 25µm. Right: Mean values of the GFAP volume in CeL/C astrocytes under indicated conditions. Data are mean across slices: nControl=11, nTGOT=13, natosiban=13, ncarbetocin=14 slices. All groups had n=4 animals. c Electrophysiological recordings from CeL/C neurons following OTR agonist application. Micrograph of slice during patch-clamp experiment showing the puff pipette on the left and the patch clamp recording pipette on the right. Scale = 20µm. d Left: Spider showing the p-values given by the comparison of different electrophysiological features. Current steps were used to measure the spike-firing threshold, the number of spikes at the second active step (nSpike), and the latency to the first spike. K+ puffs (30mM in aCSF), were used to measure the difference in spike firing (δSpike), the amplitude of the K+-induced depolarization (δVm), and its rise/decay constants. Right: Amplitude of membrane depolarization during the K+ puff. nControl=20, nTGOT=20, natosiban=20, ncarbetocin=21, nTGOT+dOVT=21, nGFAP OTR cKO+TGOT=18 neurons. e Model summarizing roles of astrocytes in OTR-mediated stress behavior. Under naive conditions (left) CeL/C astrocytes express few GFAP filaments and buffer potassium at the BLA-to-CeL/C synapse. After acute stress induction (right), astrocytes retract their processes away from synapses and decrease K+ buffering leading to an increased strength of BLA-to-CeL/C synapses. Data are expressed as mean across cell ± SEM. Detailed statistics can be found in Source Data Table 6 and other electrophysiological parameters of CeL/C neurons can be found in Supplementary Fig. 6. *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001. Drawings created in BioRender: Charlet, A. (2025) https://BioRender.com/bkv8qa0.
Collectively, these findings suggest that the OTR-Gαi pathway in CeL/C astrocytes recapitulate features of acute stress-induced morphological alterations in astrocytes and adjacent neurons sensitivity to extracellular K+ (Fig. 6e).
Discussion
We here provide evidence that acute stress induces a series of OTR- and Ca2+ dependent changes in astrocytes of the amygdala, which ultimately impact synaptic transmission in this brain area. Proteomic analysis hinted us towards stress-induced structural remodeling of astrocytes, as verified by three-dimensional reconstruction of astrocytes morphology. Interestingly, OTR-dependent astrocyte plasticity was associated with an increased sensitivity of CeL/C neurons to extracellular K+, a decreased in astrocyte synaptic coverage, and an increased strength of BLA-to-CeL/C synapses. Crucially, this morpho-functional plasticity depends upon the recruitment of OTR-Gαi pathway in astrocytes. We propose that this mechanism is key to achieve anticipated behavioral response to a potential danger, contributing to animal survival.
Previous studies have shown that activation of OTR in the CeA decreases freezing behavior induced by Pavlovian fear conditioning in rodents during fear expression and extinction, but not during fear acquisition8,27,36. In contrast, in the context of acute stress, GFAP OTR cKO mice do not exhibit freezing behavior, indicating that astrocytic OTR-mediated signaling is necessary for this response. The acute stress paradigm used in our study is similar to a fear acquisition protocol, where a conditional stimulus (sound) precedes an unconditional stimulus (shock). Given that removal of astrocytic OTR in the CeA prevent stress induced freezing in these conditions, we suggest that OTR astrocytes are not only involved in immediate stress expression but also in fear memory acquisition37. Therefore, the OTR’s roles in freezing behavior may stem from the varying effects of OT depending on the physiological state of the animal38. We cannot exclude a potential effect of astrocytic OTRs on fear memory extinction; nevertheless, our experimental design does not allow us to decipher such a function since mice lacking CeL/C astrocytic OTRs do not freeze, even during the conditioning session. In summary, OT enables freezing in response to a potential danger during acute stress, while it may trigger a fight-or-flight response when facing a known danger during fear. This bidirectional regulation of freezing behavior is surprising, although a similar effect has been recently observed in the basolateral amygdala. In this region, optogenetic stimulation of dopaminergic neurons targeting fear-responding Rspo2+ neurons impairs fear extinction. On the other hand, stimulating reward-activated Ppp1r1b+ neurons accelerated fear extinction39. Similarly to this projection-dependent dual role of dopamine, OT may also regulate freezing behavior differently depending on its neuronal and glial targets. Therefore, the physiological relevance of OT, and its action on astrocytes, is then of critical importance: it enables the animal to execute the appropriate behavior in response to a potential threat.
The fact that stress response relies on the initial activation of ~20% of the CeL/C astrocyte population expressing the OTR is surprising but can be explained in part by a communication between OTR-expressing astrocytes and their neighbors via gap junctions, thereby relaying the oxytocinergic message to a larger astrocyte population17,40. A cardinal feature of the cellular response to acute stress was the retraction of astrocyte processes leading to reduced coverage of CeL/C synapses. This type of structural plasticity has also been observed in the lateral amygdala, where fear conditioning induce the retraction of astrocytic processes from synapses29. Interestingly, similar OT-dependent structural changes were also observed in the hypothalamus after prolonged stimulations such as lactation29,37,38,40. These results suggest that altering their morphology is a general mechanism by which astrocytes respond to disruption of brain homeostasis. Of importance, OTR removal from CeA astrocytes prevented stress-induced freezing in male, but not in female mice, suggesting potential sex-specific mechanisms. Sex differences in the OT/OTR system have been reported41,42, with female mice having more OTR in the CeA as compared to males43. Knowing this general difference of OTR expression, one could wonder whether the same proportion of CeL/C astrocyte express the OTR, and whether their OT sensibility are similar between males and females.
The described inverse correlation between GFAP levels and branching of astrocytes resembles the reactive astrocytes state, where essential physiological functions such as K+ buffering are altered44. Modifications of astrocyte physiology have been found in several pathological models but a proper correlation between those parameters have never been established within the same model45,46,47. Here we propose that loss of cellular homeostasis are detected by astrocytes that restructure their cytoskeleton by increasing the amount/length of GFAP intermediate filament in their proximal processes while retracting their thin processes, probably by depolymerizing subcortical actin that is their main structural component48. This reorganization occurs in parallel with an upregulation of the transcription factor STAT3, of the intermediate filaments vimentin, and of the K+ channel Kir4.1 (Supplementary Fig. 8a). While our results are based on ex vivo experiments and may not fully reflect in vivo conditions, the data indicate that the variation of neuronal excitability might be due to a defect in K+ clearance. However, expression of Kir4.1 has recently been shown to modulate synapse strength by acting at the presynaptic level49, and we therefore cannot exclude a similar effect under our conditions. Astrocytes can rapidly modify their morphology upon strong neuronal activity in calcium-dependent manner48,50. In accordance with our findings, it was reported that Gαi pathways activate the transcription factor STAT51, which is a key mediator of astrocyte reactivity in many pathological contexts49. Moreover, two recent studies linked Gαi-GPCR activation and astrocyte morphology52,53, indicating this pathway to be a potentially common mechanism for the regulation of astrocyte morphology. Especially, Soto and colleagues showed a decreased astrocyte morphology and homeostatic functions in mice from a model of compulsive and anxiety-related behaviors. Chemogenetic stimulation of the Gαi pathway in astrocytes reverse these phenotypes53. Thus, it is tempting to speculate that astrocytic Gαi proteins evoke intracellular Ca2+ transients that subsequently modify astrocyte morphology during demanding physiological and behavioral challenges, whose dysregulation might be linked to pathological conditions.
Remarkably, both OTR-Gαi and OTR-Gαq pathway activation led to Ca2+ transients in astrocytes, which induced distinct cellular responses. How do astrocytes differentiate between these two signaling pathways to trigger an appropriate reaction? It seems plausible that concomitant activation of Gαq and Gαi signaling pathways may occur upon OTR activation. To support this hypothesis, we observed that albeit Gαi blocking by pertussis toxin prevent atosiban but not TGOT-induced Ca2+ signaling, Gαq blocking prevents astrocytes response to both carbetocin and TGOT, indicating that TGOT-induced Ca2+ response does not rely on Gαi activation only. This can be explained by the property of Gβγ subunits of the Gαi protein to enhance subthreshold Gαq signaling54, suggesting that a concomitant activation of these proteins is needed to generate Ca2+ transients. Such signal may encode a specific message, as suggested by the distinct features in the Ca2+ events triggered by astrocytic Gαi- or Gαq-OTR pathways. This discovery suggests that these pathways likely exert differential effects on cellular functions such as gliotransmission or morpho-functional changes. In line with this, our results indicate that an OT-induced astrocyte morphological change only occurs upon activation of OTR-Gαi, but not Gαq, pathway. This finding is supported by recent studies, which elegantly show that whilst Gαi-GPCR signaling is linked with changes in astrocyte shape52, Gαq-GPCR activation does not regulate GFAP expression20. While this suggests that Gαi pathway is a potentially common signaling involved in the regulation of astrocyte morphology, we cannot exclude the possibility of a complementary calcium-independent mechanism involved in the stress-induced astro-neuronal network plasticity.
A striking feature of our observations is the short amount of time needed to establish cellular and molecular changes following stress exposure. Astrocytes’ shape functions modifications are frequently described in pathological contexts44, however, such changes in physiological contexts remains unexplored. In the present study, we used a 10 minutes stress session followed by 5-10 minutes to anesthetize the mice and perform the slices or fixate the tissues. The morphological and homeostatic changes that occur within 15-20 minutes can seems surprisingly fast but is supported by live imaging of astrocyte processes where a significant retraction can be observed after 10-20 minutes following LTP induction48. Finally, proteomic analysis of amygdala samples after acute stress showed profound changes of the protein networks with an unexpected range and magnitude, given the short duration of stress exposure. Although this data set do not allow to identify in which cell type these proteomic modification occurs, it revealed a strong signature for altered astrocyte functionality across various classes of proteins, including channels, transporters, receptors, intermediate filaments and Gαi pathway. This broad coverage was afforded by the deep proteome profiling spanning >8000 proteins, which, to the best of our knowledge, represents the largest amygdala proteome dataset to date. While we focused on astrocyte-specific stress response proteins, these data will provide a rich resource for further mining by the community.
In conclusion, our study suggests that acute stress induces OTR-dependent astrocyte morpho-functional plasticity associated with a decreased synaptic coverage and an increased strength of BLA-to-CeL/C synapse. Together, this novel mechanism may represent the basis of proper anticipated behavioral response to stressful situations, contributing to mammalian survival.
Methods
Animals
All experiments were conducted in accordance with European Union rules and approbation from the French Ministry of Research (01597.05). For ex vivo and in vivo experiments, male and female C57BL/6j mice were used except CD-1 mice (Charles Rivers, 022) for microdialysis. For ex vivo experiments animals were between 1 and 3 months old. To target GCaMP6f to OT neurons in the PVN, Ai148 mice (Jackson Laboratory, 030328) were crossbred with OXT-Cre (Jackson Laboratory, 024234). Animals were housed under standard conditions with food and water available ad libitum and maintained on a 12 h light/dark cycle.
Viral transduction following stereotaxic injections
Animals were deeply anesthetized with 4% isoflurane, and their heads were fixed in a KOPF (model 955) stereotaxic frame. Metacam (Meloxicam, 20 mg/kg) was injected subcutaneously to limit inflammation and local analgesics were injected locally at the incision site (Bupivacaïne, 5 mg/kg, and Lurocaïne, 2 mg/kg). The skull was then exposed, and two holes were drilled according to coordinates adapted from Paxinos and Watson brain Atlas. For injections in the CeL/C, we injected 200nL of the appropriate rAAV at the following coordinates: rostrocaudal = −1.25 mm, mediolateral = ±3.1 mm, dorsoventral = −5.0 mm. To stimulate BLA neurons, we injected 100nL of AAV9-CaMKIIa-hChR2(H134)-EYFP at the following coordinates: rostrocaudal = −1.8 mm, mediolateral = ±3.4 mm, dorsoventral = −5.0 mm. The wound was then sutured and the weight of the animals was followed for five days to verify the post-operatory recuperation. After ~3 weeks, mice were used for experiments.
Behavior analysis
Stress protocol
The day before the stress protocol, animals were introduced in the stress box with a smooth white plastic floor and walls for ten minutes for habituation and internal control. Mice were exposed five times to a 20s-long CS tone (2 kHz, 60 dB) with a variable inter-tone interval (20 to 180 seconds). The next day, stripe patterns were placed on the walls of the box and the smooth white plastic floor was removed, revealing the grid floor of the box. Animals were re-introduced in the stress box for 10 min and exposed to five 20-second long sound stimuli with different inter-tone time intervals. The last second of these tones coincided with a 1s-long foot shock of 0.6 mA. Freezing behavior, speed and distance traveled was quantified using the ANY-maze software. Freezing was defined as periods longer than 2 seconds during which mice did not move in a time window of 20 s following the foot shock. For subsequent ex vivo experiments, mice were injected with anesthetics directly after the end of the stress protocol and slices were performed in the following 10 to 20 minutes.
Sucrose preference test
Cages were equipped with two feeding bottles, one with sucrose 1% and the other drinking water. At 24 h after the beginning of the experiment, we reversed the position of the bottles to avoid a potential place preference. Bottles were weighted before the experiment, 24 h and 48 h after. We then calculated the sucrose preference according to the following equation:
Conditioned place preference
In this test, animals develop a preference to a clonidine-paired chamber due to both pain relief in this environment and avoidance for the saline-paired chamber associated with ongoing pain. The apparatus (42553 Conditioned Place Preference for Mice, Ugo Basile, Gemonio, Italy) consists of 2 Plexiglas chambers separated by manually operated doors. Two chambers (size 16 ×25 x 25 cm) distinguished by the texture of the floor and by the wall patterns. On the first, second and third days (pre-conditioning), animals were free to explore the apparatus during 30 min, and the time spent in each chamber was recorded using ANY-maze™ software. Animals spending more than 75% or less than 25% of the total time in one chamber were excluded from the study. On the 4th day (conditioning) animals were intrathecally injected with either saline solution (10 µL, morning) or clonidine solution (10 µg in 10 µL in the afternoon) and restricted in one chamber for 15 min, switching chamber between the morning and the afternoon. On the 5th day the animals were free to explore the 3 chambers and the time spent in each chamber was recorded for 30.
Von Frey test
The mechanical threshold of hind paw withdrawal was evaluated using von Frey hairs (Aniphy Vivo-Tech, Salon-de-Provence, France). Mice were placed in clear Plexiglas® boxes on an elevated mesh screen. After a habituation time of around 10 min, the filaments were pressed on the plantar surface of each hind paw in a series of ascending forces (0.6 to 8 grams). Each filament was applied five times per paw, until it just bent, and the threshold was defined as three or more withdrawals observed out of the 5 trials. The mean of sensitivity of the two hind paws was calculated.
Hargreaves test
The latency for hind paw withdrawal in response to thermal stimulation was determined using the Hargreaves method. Mice were placed in clear Plexiglas® boxes and testing began after exploration and grooming behaviors ended (15 min). The infrared beam of the radiant heat source (37570 Plantar Test, Ugo Basile, Gemonio, Italy) was applied to the plantar surface of each hind paw. The experimental cut-off to prevent damage to the skin was set at 15 s. Three measures of the paw withdrawal latency were obtained per animal and averaged for each hind paw.
Dry ice test
Hind paw withdrawal latency in response to cold thermal stimulation was determined using the dry ice method. Mice were placed in clear Plexiglas® boxes. After a 15 min habituation period, a cut 2 mL syringe filled with dry ice powder was placed against the glass surface under the animal’s paws. This source of cold under the animal’s plantar surface caused the animal’s paw to retract. Latency times were recorded manually using a stopwatch. Three measurements of paw withdrawal latency (spaced at least 15 min apart) are then taken and averaged for each hind paw.
Social interaction test
Social interactions were assessed in the cage of the same size of the home cage, with a bit of dust at the bottom. For this test, interactions between a test subject and a stimulus mouse were systematically scored and videotaped for 5 min. Light intensity was 150lux. All cages were bring in the test room for acclimatization of 30 min. Then, the stimulus and test mice were introduced in the same time in a clean cage for 5 min. The stimulus mice were from the same background C57BL6, same sex, same age and used maximum twice a day. Their weight was on average 7% less compared to test mice. Order of testing was counterbalanced according to genotype and stimulus mice. The times the test subject spent performing social behaviors (i.e. facial sniffing (oral-to oral contact), anogenital sniffing (oral-to-base sniffing), direct touching with a paw and pursuit of the stimulus mouse) were determined in real time with an ethological keyboard (ANY-maze software). After testing, each mouse was returned to its home cage and a new clean cage was used for the next mice. The percent of time spent engaged in social behavior compared to total time (5 min) was calculated for the measure of social interactions:
Corticosterone measurements in blood
Sampling
Blood sample (40 μl) were collected from tail tips following a scissor incision using a calibrated sodium-heparinized capillary (40 µl, Hirschmann™ Disposable Capillary Tubes, Fisher Scientific, Illkirch, France). The blood was then supplemented with 10 µl of heparin (843 µM; Sigma-Aldrich, St. Quentin Fallavier, France). Subsequently, 10 µl of D4-CORT (5 µM, Sigma-Aldrich) in 99.1% H2O/0.1% formic acid (v/v; Fisher Scientific) was added to the mixture. Proteins were precipitated acetonitrile (ACN 100%, Fisher Scientific) and samples were centrifuged at 20,000 g (4 °C, 30 min). The supernatant was collected and dried under vacuum (SpeedVac, Thermo Fisher). The dried samples were then resuspended in 20 μl of 20% ACN/0.1% formic acid (v/v). The supernatants was collected and stored at −80 °C.
Dosage
Corticosterone measurements were conducted using a Dionex Ultimate 3000 HPLC system (Thermo Electron, Villebon-sur-Yvette, France) coupled to a triple quadrupole Endura mass spectrometer (Thermo Electron). The samples were loaded onto a heated Hypersil aQ column (100 x 1 mm, 1.9μm, with a flow rate of 90 μl/min; Thermo Electron) at 40 °C. Qualification and quantification were performed using multiple reaction monitoring (MRM) based on the isotopic dilution method. Compound identification was achieved by comparing the precursor ions, selective fragment ions, and retention times with those of the isotopically labeled standards (IS). The selection of monitored transitions, along with the optimization of collision energy and RF lens parameters, was automated (Supplementary Table “HPLC and MSMS details”). System control was driven by Xcalibur v4.0 software (Thermo Electron).
Microdialysis
Male CD1 mice (25-30 g; 7-8 weeks old; Charles River, Germany) were used due to their larger size. Animals were stereotaxically implanted with a U-shaped microdialysis probe (molecular cut-off: 18 kDa; length of membrane 1.5 mm) under isoflurane anaesthesia (Isoflurane Baxter, Baxter Deutschland GmbH, Germany) targeting the right CeA (AP: −1.0; lateral +2.4; DV: −5.0 mm from bregma; Paxinos and Watson, 1998). After a 48 h post-surgery recovery, the microdialysis probe was first perfused without sampling for 90 min (3.3 µl/min, Ringer’s solution, adjusted to pH 7.4), before 3 consecutive 30 min microdialysates from the CeA were collected (1) under basal conditions, (2) during exposure to 5 random foot-shocks (0.6 mA, 1 s) over a 10 min period in a conditioning chamber (45 ×22 x 40 cm, transparent Perspex box with a steel grid floor), and (3) again in the home cage. Samples were kept at −20 °C until radioimmunological analysis of OXT (detection limit: 0.5 pg/sample, intra-assay variability of <8% cross-reactivity with other neuropeptides including arginine vasopressin <0.7%). Brains were frozen and the perfusion sites were histologically verified on 40 µm cryo-cut stained brain slices.
In vivo imaging
In vivo micro-endoscope calcium imaging
To visualize calcium level in PVN OT neurons, Ai148:OXT-Cre mice were stereotactically implanted with a chronic GRIN lens (0.5 mm in diameter; 7.4 mm in length, Inscopix) ± 200 µm above the PVN at the following coordinates: rostrocaudal = −0.85 mm, mediolateral = +1.1 mm, dorsoventral = −4.9 mm with a 10-degree angle (right hemisphere). The lens was securely fixed to the skull with a three-component dental cement (C&B-Metabond, Parkell Inc.). After a recovery period of at least two weeks, animals were implanted with a baseplate to attach the micro-endoscope (nVista 3, Inscopix). A tethered dummy micro-endoscope was connected to the baseplate for habituation to the recording procedures. Following an additional week for recovery and habituation, the real miniature microscope was attached to the baseplate one day before the experiment. Imaging data were collected at 10 Hz, with excitation light power 0.8-1.2 mW/mm² (40-60% of maximal power, nVista 3, Inscopix). Stacks of calcium images were first preprocessed to correct time-invariant pixels or reconstruct frames lost during the acquisition, and were then adjusted for motion artifacts in the x-y plans using Inscopix Data Processing software 1.9.1 (Inscopix). Subsequently, image stacks were exported to ImageJ, where the recording background was subtracted. Regions of interest (ROI) corresponding to individual neurons were manually defined over z-stacked images, and the raw average fluorescence from each ROI was quantified and exported using Fiji software. Raw traces are further processed and normalize using custom-made scripts (Matlab, MathWorks). Due to the potential bleaching of the GCaMP6f fluorescence within the session, an exponential curve was fitted and subtracted from the raw signal of each cell during each session to ensuring consistency in the fluorescence measurements. Subsequently, F0 was determined using Gaussian mixture modeling, where the observed intensity values from each cell in each session were approximated as a mixture of Gaussian distributions via an expectation-maximization procedure. The mean of the distribution containing the lowest values was identified as the F0 for each neuron and session. ΔF/F0 are calculated base on bleaching-corrected calcium traces and the estimated F0, and then are normalized using envelope normalization. For control experiments, each mouse was subjected to a randomized protocol that included multiple instances (2–6 each) of tail suspension (lifting by the experimenter) and cage shaking, with intervals of 10–60 seconds. Baseline and post-event quantification windows were manually defined for each animal.
Fiber photometry
3-4 weeks after viral injection, fiber-optic cannula (fiber core, 400μm; 0.37 NA; 5 or 5.5 mm length; Doric Lenses, Quebec City, Canada) was implanted approximately 0.1 mm above the injection site and fixed to the skull with dental cement and Super-Bond C&B. After 5-7 days recovery, mice were handled by the experimenter for 3-5 days and acclimated to the fiber-optic patch cord connection for 3-5 additional days. Fluorescence intensity was measured with a dual-color fiber photometry setup equipped with two LED light sources, 465 and 560 nm (Doric Lenses). The light intensity at the end of fiber-optic patch cord was adjusted to between 50 and 80 µW. Data were acquired by lock-in mode in Doric Neuroscience Studio. Acquired data were processed by custom-made python codes. After data denoising with a Savitzky-Golay filter, photobleaching was corrected for by a computed control channel generated by fitting with an exponential curve with the corresponding signal. Normalized ΔF (ΔF/F%) was calculated by the equation:
where Fraw is the Ca2+-dependent fluorescence changes and Fcontrol is the value in control channel computed for minimizing the photobleaching effect. For experiments using OTR antagonist, L368-899 (10 mg/kg) was injected 30 minutes before the stress session. Animals with inconsistent basal fluorescence between days were removed from the analysis.
Proteomic analysis
Tissue sample preparation
Mice brains were sliced as described for ex vivo electrophysiology. Using a 1mm-diameter punch, the CeA was dissected bilaterally on two 400 µm slices per hemisphere (4 punches per mouse), snap-frozen and stored at −80 °C. Anesthesia, slicing and punch sampling procedure took 10 to 20 minutes following the stress protocol. Isolated amygdala tissues from naïve and stressed mice (n = 5 per group) were disrupted using a lysis solution containing 1% Sodium-dodecylsulfate, 50 mM Tris (pH 8.0), and a protease inhibitor cocktail (Merck) without EDTA. The tissues were subjected to sonication in AFA-tube TPX strips (Covaris) using a Covaris LE220R-Plus instrument (300 s, average power of 162.5, peak power of 325, 50% duty factor, 200 cycles per burst). Tolerance of the dithering settings were set at ± 5 mm in the x and z axis, and 4.5 mm in the y axis, while operating at a velocity of 20 mm/s. Protein concentrations of the sonicated lysates were measured using a BCA assay (Pierce). For mass spectrometer analysis, protein digestion and peptide generation were performed using 1 μg of protein per condition. Due to the limited amount of initial material, we utilized the automated version of the SP3 protocol for protein digestion and peptide purification. The Bravo system is designed to handle 96 samples at once, performing all necessary steps such as protein reduction and alkylation, magnetic bead aliquoting, protein clean-up using SP3, protein digestion, and peptide recovery.
LC-MS/MS
The quantification of tryptic peptides from the whole proteome was performed using an EASY-nLC 1200 system (Thermo Fischer Scientific) connected to a Fusion mass spectrometer (Thermo Fischer Scientific). Peptides were separated by reverse-phase liquid chromatography employing 0.1% formic acid (solvent A) and 80% acetonitrile (solvent B) as the mobile phases. The separation of peptides was performed using an Acclaim PepMap trap column (Thermo Fisher Scientific, C18, 20 mm × 100μm, 5μm C18 particles, 100 Å pore size) and a nanoEase M/Z peptide BEH C18 analytical column (Waters, 250 mm × 75μm 1/PK, 130 Å, 1.7μm). The gradient started with a 3% concentration of solvent B for the first 4 min, thereafter increasing to 8% and further rising to 10% after 6 min. The concentration of solvent B increased to 32% after 68 min and to 50% after 86 min. Between the 87th and 94th minute of the gradient, the concentration of solvent B reached 100%. The system was re-equilibrated after 95 min using 3% of solvent B for 10 min. The eluted peptides were subjected to ionization and introduced into the mass spectrometer using the Nanospray flex ion source (Thermo Fischer Scientific) and a Sharp Singularity nESI emitter (ID = 20μm, OD = 365μm, L = 7 cm, α = 7.5°) (FOSSILIONTECH), which was connected to a SIMPLE LINK UNO-32 (FOSSILIONTECH). A constant spray voltage of 2.5 kV was applied to the emitter, and the capillary temperature of the ion transfer tube was set to 275 °C. The Fusion mass spectrometer was used in data-independent (DIA) mode with a scan range of 400-1000 m/z, an Orbitrap resolution of 60000 FWHM, an AGC target of 3e6, and a maximum injection duration of 20 ms. The acquisition of data-independent MS/MS spectra was performed at a resolution of 30000 FWHM. The maximum injection duration was set to 50 ms and the AGC target was set to 1e6. The acquisition strategy included employing 26 isolation windows, each having a width of 23.3 m/z and overlapping by 1Th. The collision energy used for fragmenting the precursor ions was set to 27, and a fixed initial mass of 200 m/z was used to get the MSMS spectra.
Data analysis
Raw data obtained from DIA measurements were analyzed using DIA-NN version 1.8.0. A spectral library was developed based on the predictions made by the fasta file. In addition, a fasta file containing commonly seen protein contaminant was included for the purpose of predicting the spectral library. The spectral library prediction used the default values. The default parameters of DIA-NN were used for the processing of the raw files. The precursor matrix output tables were filtered to include only values with a false discovery rate <0.01. Additionally, values were filtered based on a channel q-610 value < 0.01 and a translated q-value < 0.01. MBR feature was activated to enhance data integrity. Output tables generated by DIA-NN were then analyzed with R-Studio, using the dplyr package for data wrangling, and ggplot2 for visualisation. Differential expression and statistical analyses were carried out using the limma package. Gene Set Enrichment Analysis (GSEA) was conducted with the fgsea package, heat maps were created using the pheatmap package and Perseus software as explained thereafter.
Statistical analysis
GSEA was performed using the R package fgsea, which implements a fast preranked GSEA algorithm. Proteins were ranked according to the (log₂ fold change) from the differential abundance analysis. For each protein set, fgsea computes an enrichment score using a Kolmogorov-Smirnov-like running-sum statistic on the ranked list. To assess significance, fgsea generates a null distribution of enrichment scores by repeated permutation of the protein-level statistics and recalculation of the enrichment score for each protein set. The nominal p-value is defined empirically as the proportion of null enrichment scores that are at least as extreme as the observed enrichment score for that protein set. Multiple testing correction across all tested protein sets was performed using the Benjamini-Hochberg procedure. The resulting FDR-adjusted p-values are reported in the Fig. 2e, f and Source Data Table 2.
Immunofluorecence staining and image analysis
Tissue fixation
Mice were anesthetized using ketamine (Ketamin 1000, 400 mg/kg) and xylazine (Paxman, 80 mg/kg) administered intraperitoneally. 10 to 20 minutes following stress protocol, mice were then perfused transcardially with PBS, followed by 4% PFA. Brains were dissected and post-fixed overnight in 4% PFA at 4 °C.
Astrocyte morphology assessment
50μm vibratome sections were cut, collected, and stored in PBS 1X overnight at 4 °C. Slices were then incubated in a PBS1X-Gelatin 0.02%-Triton 0.25% solution for permeabilization and saturation of non-specific sites during 1 h at RT. Incubation of the first antibodies took place overnight at 4 °C and slices were then washed and incubated with secondary antibodies overnight at 4 °C. Finally, slices were mounted in an Abberior antifade medium and left for one day at 4 °C in the dark for polymerization. All antibodies used and their concentrations are given in the reagent table (Supplementary Table “List of reagents”). Z-stack images of the CeL were captured (6-8 images per animal) with a depth of 50μm and 1μm intervals, employing a 40x magnification on the Nikon AX confocal system (Nikon Imaging Center, Heidelberg). All images used for analysis were taken with the same confocal settings (pinhole, laser intensity, digital gain, and digital offset). Imaris software (version 10.0, Oxford Instruments) was used for reconstruction and morphological analysis of astrocytes. CeL/C segmentation was performed by superimposing the putative CeL/C delimitation onto the corresponding CeL/C-containing brain images (Paxinos Mouse Brain Atlas) to achieve the best fitting overlay. Raw confocal files were imported into Imaris and subjected to surface reconstruction, involving background subtraction and setting surface detail to 0.5 µm with the largest diameter at 0.5 µm. Immunofluorescent signals from GFAP staining or virally-expressed eGFP were utilized as templates for surface and filament reconstructions. Astrocytes falling below a volume of <250 µm³ or exceeding >5500 µm³ were excluded from data analysis. A separate masked channel was generated for filament analysis, employing automated threshold detection with maximum seed point placements. Manual intervention ensured each astrocyte possessed precisely one starting point at the soma center. The efficiency of viruses were calculated as the number of cells co-expressing virus-dependent of GFP (or mScarlet) and astrocyte markers over the number of cells expressing astrocyte markers. The specificity were calculated as the number of cells co-expressing virus-dependent of GFP (or mScarlet) and astrocyte markers over the number of cells expressing GFP (or mScarlet). Morphological analysis involved calculating average values for each animal (surface area, volume, etc.). Sholl analysis was conducted using Imaris in the filament reconstruction mode. Each data point on the respective Sholl plots corresponds to the mean Sholl intersections, accounting for the average of the respective cohort with error bars indicating ±SEM, calculated across all astrocytes per animal. Identification of synapses in contact with astrocytes was performed after immunohistochemical amplification of virally expressed GFP in astrocytes, and staining of pre- and postsynaptic markers vGlut1/2 and Homer1. High magnification confocal images were acquired, which were then processed into our Imaris-assisted pipeline. The background was subtracted (surface detail at 0.2 µm, largest diameter at 0.2 µm), and debris measuring <0.1 µm³ was disregarded as nonspecific and filtered out in a subsequent step. vGlut1/2 and Homer1 puncta were reconstructed from high magnification confocal images. These putative synapses were kept for analysis if they followed the following criteria: 1) close apposition of pre- and postsynaptic components based on vGlut1/2 and Homer 1 colocalizsation, 2) volume of putative synapses must be at least 0.1 µm3, and 3) must be in a 50 nm-range of the astrocyte surface.
Ex vivo recordings
Slice preparations
Mice were anesthetized using ketamine (Ketamine 1000, 400 mg/kg) and xylazine (Paxman, 80 mg/kg) administered intraperitoneally. Intracardiac perfusion was then performed using an ice-cold NMDG-based aCSF containing (in mM): NMDG (93), KCl (2.5), NaH2PO4 (1.25), NaHCO3 (30), MgSO4 (10), CaCl2 (0.5), HEPES (20), D-glucose (25), L-ascorbic acid (5), thiourea (2), sodium pyruvate (3), N-acetyl-L-cysteine (10) and kynurenic acid (2). This solution was bubbled in 95% O2/5% CO2 gas all the time of the experiment and its pH was adjusted to 7.3-7.4 using HCl. After decapitation, the brain was swiftly removed in the same ice-cold dissection aCSFs and hemisectionned. 350μm-thick horizontal slices containing the CeA were obtained using a Leica VT1000S vibratome. Slices were then stored in 37 °C NMDG aCSF for 10 min before placing them in the holding chamber at room temperature containing normal aCSFs for 1 h minimum before any experiments were conducted. Normal aCSF used for all ex vivo experiments was composed of (in mM): NaCl (124), KCl (2.5), NaH2PO4 (1.25), NaHCO3 (26), MgSO4 (2), CaCl2 (2), D-glucose (15), adjusted for pH values of 7.3-7.4 with HCl or NaOH and continuously bubbled in 95% O2/5% CO2 gas. Osmolality was set to 300 ± 10 mOsm/L. For pharmacologic experiments, slices were incubated in aCSF supplemented with 500 nM of agonist and/or antagonist for 1 h. Slices were then transferred to an immersion-recording chamber superfused at 2 ml/min with aCSF. The anesthesia and slicing procedure took 10 to 20 minutes.
Electrophysiology
Whole-cell patch-clamp recordings of CeL/C neurons were done using infrared oblique light visualization. For astrocyte recordings, slices were incubated in 1 µM SR101 at 37 °C for 20 min and washed for 1 h. Subsequent patch-clamp recordings from astrocytes were guided by SR101 fluorescence. Borosilicate glass electrodes (OD 1.5 mm, ID 0.86 mm; Sutter Instrument) were pulled using a horizontal flaming/brown micropipette puller (P97; Sutter Instrument) to obtain 3.5-7 MΩ recording pipettes. These pipettes were filled with an intracellular solution containing (in mM): K-Glu (125), HEPES (20), NaCl (10), and ATPNa2 (3). The pH was adjusted to 7.3-7.4 with KOH, and osmolality was adjusted at 300 ± 10 mOsm/L with water. Recordings were acquired with an Axon MultiClamp 700B Amplifier coupled to a Digidata 1440 A Digitizer (Molecular Devices) at 20 kHz using either the pClamp 10 software suite for puff experiments or using WinWCP 4.2.2 freeware (John Dempster, SIPBS, University of Strathclyde, UK) for photostimulation experiments. Series resistance was monitored and manually compensated (~50% typically). Spontaneous action potential firing was measured over 10-minute-long recordings. At the beginning of each recording, current pulses ranging from −10pA to 100pA with 10pA steps were applied for 300 ms per sweep to neurons or voltage pulses ranging from −100mV to 100 mV with 20 mV steps lasting 100 ms for astrocytes. For neurons, the membrane potential drift was estimated with the Savitsky-Golay polynomial local filter from the scipy library and subtracted from the membrane potential. Spikes were then detected with the find_peaks function of the Scipy library for each sweep as peaks with a height and a prominence higher than 10 mV. The latency of spikes was quantified as the time between the beginning of the first step that triggers a spike and the first spike. The number of spikes was measured in the second step, which triggers action potentials. Spike hyperpolarizations were measured on the first step that evoked an action potential. This protocol was repeated three times, and the median results were used for further analysis. For astrocytes, voltage-current curves were constructed by measuring the current needed to reach the command voltage during the last 25 milliseconds of the voltage step. For K+ puff application, the injected current was adapted to hold the neuronal membrane at −60mV. Basal activity of neurons was recorded for 20 s, and then a 20s-long puff of a 30 mM KCl-containing aCSF was done at ~100 µm of the recorded neuron. The membrane potential drift was estimated after a stringent Gaussian filter from the pyABF library, thereby avoiding overestimation of this value due to action potentials. This drift was then subtracted from the original traces, and spikes were detected with the find_peaks function of the Scipy library as peaks with a height and a prominence higher than 20 mV. Spikes and membrane potential were measured in the 20 s baseline, the 20 s K+ puff application, and the 20 s washout. The sum of spikes and the median of membrane potential were computed over these three periods. For depolarization kinetics, linear functions were fitted to the first five seconds of the rising/decay phase of the K+-induced depolarization on the Gaussian-filtered trace. This protocol was repeated three times, and the median values of measures were used for statistical analysis. For IAHP recordings, CeL/C neurons membrane potential were clamped at −20mV, depolarized at +20 mV for 100 milliseconds, and clamped again at −20mV for 50 seconds. We reproduced this protocol 5 times for each neuron. The AUC of each IAHP were measured as the integral from its maximum peak after the end of stimulation until its membrane potential return to baseline. The decay was measured as the time necessary for the current to decrease by 63%. For optostimulation of BLA neurons, the neuronal membrane potential was held in voltage clamp at −60mV. Basal activity was recorded for 20 s of basal activity before stimulating glutamatergic BLA neurons with 5 ms blue light pulses at 20 Hz for 10 s. For each recording, EPSC were detected with the find_peaks function of the Scipy library as peaks higher than three times the standard deviation estimated during the 20 s baseline. Frequency and amplitude of these EPSC were measured in 5 seconds-long quantification windows taken just before the photostimulation, during the five first seconds of the photostimulation, and at the end of the recording. After each light pulse, we estimated a failure as the absence of evoked EPSC, or we estimated the rise and decay of the evoked EPSC. Rise and decay constants were measured on exponential and linear fits as the time needed to go from 10 to 90% or from 90 to 10% of the EPSC amplitude, respectively. This recording was repeated three times and the medial value of the three recordings was used for statistical analysis. The paired pulse ratio were calculated as the ratio between the second over the first EPSC evoked by the photostimulation of BLA axon terminals. For K+ currents recordings, astrocytes were patch-clamped at −80mV and basal membrane current was recorded during 20 s before stimulating glutamatergic BLA neurons with 5 ms blue light pulses at 20 Hz for 10 s. Subsequently, recordings were performed in the presence of Ba2+ to block K currents and BLA neurons were stimulated again after 15 min. Evoked currents were estimated as the minimum value during the photostimulation subtracted from the minimum value during baseline. The K+ current was calculated by subtraction of currents evoked in the presence of Ba2+
Quantification of cell mortality
A 20s-long puff of aCSF or a 30 mM KCl-containing aCSF was done at ~100 µm of the recorded neuron in brain slices from naive and stress mice. Slices were then washed 5 min with PBS then incubated for 10 min with blue trypan (4%), washed again in PBS for 5 min 3 times, subjected to chemical fixation overnight in 4% PFA at 4 °C and observed on by confocal microscopy.
Ex vivo calcium imaging
After SR101 incubation, the synthetic calcium indicator OGB1-AM was bulk loaded as previously described55, reaching final concentrations of 0.0025% (20 μM) for OGB1-AM, 0.002% for Cremophor EL, 0.01% for Pluronic F-127 and 0.5% for DMSO in aCSF and incubated for 1 h at 37 °C Slices were then washed in aCSF for at least 1 h before imaging. Only astrocytes co-labeled for SR101 and OGB1 were used. The spinning disk confocal microscope for calcium imaging was composed of a Zeiss Axio examiner microscope with a ×40 water immersion objective (numerical aperture of 1.0), mounted with an X-Light Confocal Unit–CRESTOPT spinning disk. Images were acquired at 2 Hz with an optiMOS sCMOS camera (Qimaging). Cells within a confocal plane were illuminated for 20 ms at 575 nm for SR101 and 80 ms at 475 nm for OGB1 using a Spectra 7 LUMENCOR. The different hardware elements were synchronized through the MetaFluor 7.8.8.0 software (Molecular Devices). Because astrocytes are mechanosensitive, OTR agonists (500 nM) were bath applied for 20 s and not puff-applied to avoid mechanical stimulation. All calcium imaging experiments were conducted at room temperature and cells with an unstable baseline were discarded.
All analyses were conducted as described55. Astrocytic calcium levels were measured in manually outlined regions of interest (ROI) comprising the cell body using ImageJ software. Subsequent offline data analysis was performed using a custom-written Python-based script. To take into account slice micro-movements, the SR101 fluorescence values were subtracted from the OGB1 ones. Then, a linear regression was applied to each trace to correct for photobleaching. Calcium transients were detected using the find_peaks function of the SciPy library as fluorescence variation exceeding 8 time the standard deviation and a prominence exceeding it 5 times (for more details, see55). The number of peaks and the area under the curve was quantified before and after the drug application. All data were normalized according to the duration of the recording, and astrocytes were labeled as ‘responsive’ when their AUC or their calcium transient frequency at least doubled after drug application. Because the time after stimulation is longer than the baseline (10 min vs. 5 min), the probability of observing a spontaneous calcium peak is stronger after stimulation. To avoid this bias, astrocytes with only one calcium peak during the whole recording were not considered responsive. Each calcium transients were then isolated, their area were estimated with the trapezoid method and their duration were measured as their half-maximum full width (HMFW). The rise and decay phases of Ca2+ transients were fitted with linear functions and the coefficient of these slopes was used as rise and decay constants. To compare effects of different agonists (Figure S6), evoked Ca2+ activity was normalized to the baseline values as ratio (baseline/drug effect).
BRET experiments in astrocytes
Purifed astrocytes were isolated from P2 C57BL6 mice whole brains by magnetic-activated cell sorting (MACS) (Miltenyi Biotec, Bergisch Gladbach, Germany) with anti-GLAST (ACSA 1; (Jungblut et al., 2012)) MicroBeads according to the manufacturer’s instructions. Cells were plated on poly-L-lysine-coated (Sigma Aldrich, St. Louis, MO, USA) T75 flasks in complete medium, composed by minimal essential medium (MEM, Invitrogen, Life Technologies, Carlsbad, CA, USA) supplemented with 20% fetal bovine serum (FBS) (Gibco, Life Technologies, Carlsbad, CA, USA) and glucose (5.5 g/L, Sigma Aldrich, St. Louis, MO, USA). Cells were grown in a humidified incubator at 37 °C and 5% CO2. Two days before the experiment, astrocytes were harvested with trypsin 0,25% (Gibco, Life Technologies, Carlsbad, CA, USA) and plated in 12-well plates in complete medium at a density to ensure ~70% confluent cultures at 24 h after seeding. Typically, 80,000 cells/cm² surface area of the culture dish were seeded in cell-specific medium. Astrocytes were transfected using Lipofectamine 3000 (Invitrogen, Life Technologies, Carlsbad, CA, USA) according to the manufacturer’s protocol with a DNA to Lipofectamine ratio of 1:3 w/v. A transfection enhancer, the 3000 enhancer reagent (1:2, DNA: Reagent, w/v), was used along with the Lipofectamine 3000 transfection reagent. 1.25 µg of plasmids DNA were transferred to each well of the 12-well plates. The cDNA amounts used per well were as following: Gαx Rluc8 (300 ng), GFP10-Gγ2 (250 ng), Gβ1 (300 ng) and OTR (400 ng).
BRET experiments were performed 72 h after transfection. Astrocytes were detached with trypsin 0.25% and resuspended in Krebs-Ringer’s-HEPES (KRH) buffer containing (in mM) NaCl (125), KCl (5), MgSO4 (1.2), KH2PO4 (1.2), CaCl2 (2), glucose (6) and HEPES-NaOH (25), pH 7.4.
For specific validation experiments, BRET biosensors were expressed in HEK293 cells. To this end, cells were co-transfected using plasmids encoding Gαq-97-Rluc8 or Gαi2-91-Rluc8 and GFP10-Gγ2, Gβ1, and the OTR. Two days after transfection, cells were washed twice, detached, and resuspended with PBS with 0.5 mM MgCl2 at room temperature. Cells were distributed on 96-well microplates (100 µg of proteins/well) (Optiplate, PerkinElmer Life Sciences) and incubated at room temperature for 5 min in the presence or absence of YM (60 nM) and two minutes with PBS or OT (10 µM).
BRET measurements were performed as previously described using the Infinite F500 multimode microplate reader (Tecan)34. The signals emitted by the Rluc8 donor that allows the sequential integration of light signals was detected with two filter settings (Rluc8 filter, 370-450 nm; GFP10 filter, 510-540 nm). The data were recorded by Infinite F500 after the addition of 5 μM Deep Blue C coelenterazine and the BRET signal was calculated as the ratio between GFP10 emission and the light emitted by Rluc8 after 2 min stimulations with maximal dose of OT (1 µM) or vehicle (PBS). All measurements were performed at 37 °C. The BRET signal was calculated as the ratio between GFP10 emission and the light emitted by Rluc8. The changes in BRET induced by OT were expressed on graphs as “OT-induced changes in BRET ratio” using the following formula:
Tissue preparation, protein extraction and western blotting
Ten to twenty minutes following the end of the acute stress protocol, brains from stressed or naïve (home cage controls) mice were sliced as described for ex vivo electrophysiology. Using a 1 mm-diameter punch, the CeA was dissected bilaterally on two 400 µm slices per hemisphere (4 punches per mouse, tissue weight <4 mg), snap-frozen and stored at −80 °C until processing. To enrich membrane protein fraction protein extraction was performed using the Mem-PERTM Plus Membrane Protein Extraction Kit (Thermoscientific) according to the manufacturer’s instructions. Briefly, after rinsing, CeA punches were transferred into grinders (Wheaton; Dounce tissue grinder 1 mL, #357538) with 500 µL of permeabilization buffer containing protease (1:100, Protease inhibitor cocktail, Sigma-Aldrich, #P8340) and phosphatase (1:100, Phosphatase inhibitor cocktail 2, Sigma-Aldrich, #P5726) inhibitors and grinded until even suspension. Homogenates were transferred in 2 mL tubes, incubated for 10 min at 4 °C under constant rotation followed by centrifugation at 16,000 g for 15 min at 4 °C. After removal of the supernatant containing cytosolic proteins, the pellet was resuspended in 500 µL of solubilization buffer with protease and phosphatase inhibitors. Samples were incubated for 30 min at 4 °C under constant rotation, centrifuged at 16,000 g for 15 min at 4 °C and the supernatant containing membrane proteins was collected and stored at −80 °C. Because of very low amounts of input tissue, we used a protein concentration method. Membrane protein samples were incubated with 77% trichloroacetic acid on ice for 30 min and centrifuged at 16,000 g for 15 min at 4 °C. After supernatant removal, pellets were rinsed with ice cold acetone and centrifuged at 16,000 g for 5 min at 4 °C. After supernatant removal, pellets were resuspended in loading buffer (NuPAGE® LDS sample buffer and sample reducing agent, Invitrogen) and denaturated 10 min at 70 °C. Samples were loaded on 4-20% Criterion™ TGX Stain-Free Precast Gels (Bio-rad) and migration was performed 1 h at 170 V in Tris-glycine buffer (Bio-rad) followed by Stain-Free gel activation using a ChemiDoc MP Imaging System (Bio-rad). Proteins were transferred on a nitrocellulose membrane with the Trans-Blot Turbo™ Transfer System (Bio-Rad). Total protein load per well was evaluated after imaging the membrane under UV light. After 3x10min rinses in 9 mM NaCl Tris-HCl Buffer + 0.1% Tween 20 (TBST), membranes were blocked in TBST + 5% milk for 1 h at room temperature (RT) and incubated overnight at 4 °C sequentially with the following primary antibodies: anti-Kir4.1 (1:1000, Rabbit; Alomone, #APC-035), anti-actin or anti-Tom20 in TBST + 5% milk. After 3 x 10 min washed in TBST, membranes were incubated for 1 h at RT with Horse Radish Peroxidase (HRP)-conjugated secondary antibodies (1:5000, Vector laboratories) diluted in TBST + 5% milk. Membranes were incubated with Clarity Western ECL substrate (Bio-rad) and the signal was detected with ChemiDoc MP Imaging System. Band intensity was quantified with Image Lab Version 5.2.1 and normalized to total loaded protein load. In-between signal detections, peroxidase activity was inactivated using a 20 min incubation of membranes in 1% Na-Azide in TBST at RT followed by 3 x 10 min wash in TBST. When necessary, antibodies were stripped using a 20 min incubation in stripping solution (ReBlot Plus Mild Antibody Stripping Solution, Millipore). Western blotting was performed with Biorad Criterion Stain Free gels, which allow to detect all proteins on the membrane via a reaction of the trial compound with tryptophan residues and thus do not require to normalize the protein concentration per well prior to protein migration.
CeL/C ultrastructural analysis
Mice were anesthetized with intraperitoneal injection of 100 mg/kg ketamine chlorhydrate and 5 mg/kg xylazine and transcardially perfused with glutaraldehyde (2.5% in 0.1 M PBS at pH 7.4). 1 mm amygdala punches were immersed in the same fixative overnight, rinsed several times in phosphate buffer, and postfixed in 1% osmium for 1 hour. Finally, samples were dehydrated in graded ethanol series and embedded in Embed 812 (EMS). Ultrathin sections were cut with an ultramicrotome (Leica), stained with uranyl acetate (1% (w/v) in 50% ethanol), and examined by transmission electron microscopy (TEM; Hitachi H7500 equipped wuth an AMT Hamamatsu digital camera). The areas for image acquisition by TEM were chosen randomly (50 images per side per animal) and the density of synapses was identical in both naïve and acute stress conditions (3.47 ± 0.08 vs 3.62 ± 0.08, respectively).
Statistical analysis
Except for the principal component analysis that was performed using the scikit python library, all statistical tests were performed using GraphPad Prism version 8.0.0 (GraphPad Software). Parametrical tests were performed if data showed normal distribution and equality of group variances. Otherwise, non-parametric tests were used. All values, group compositions, and statistical tests for each experiment are detailed in Source Data file.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The raw data generated in this study have been deposited in the Zenodo repository accessible via https://doi.org/10.5281/zenodo.13912907. The raw proteomic data generated in this study have been deposited in the Pride repository accessible via https://www.ebi.ac.uk/pride, project number PXD053856. In addition, all data that support the findings of this study are available from the corresponding authors upon request. Statistical data are provided in the Source Data Tables 1-6. Source data are provided with this paper.
Code availability
All the MatLab and Python codes for the experiments and analysis presented in this manuscript can be found in the github repository https://github.com/Team-Charlet/Baudon_et_al and accessible via https://doi.org/10.5281/zenodo.17870954.
References
Craske, M. G. & Stein, M. B. Anxiety. Lancet 388, 3048–3059 (2016).
McDonald, A. J. & Mott, D. D. Functional neuroanatomy of amygdalohippocampal interconnections and their role in learning and memory. J. Neurosci. Res. 95, 797 (2017).
Duvarci, S. & Pare, D. Amygdala microcircuits controlling learned fear. Neuron 82, 966–980 (2014).
LeDoux, J. E. Emotion circuits in the brain. Annu Rev. Neurosci. 23, 155–184 (2000).
Zimmerman, J. M., Rabinak, C. A., McLachlan, I. G. & Maren, S. The central nucleus of the amygdala is essential for acquiring and expressing conditional fear after overtraining. Learn Mem. 14, 634–644 (2007).
Terburg, D. et al. The basolateral amygdala is essential for rapid escape: a human and rodent study. Cell 175, 723–735.e16 (2018).
Knobloch, H. S. et al. Evoked axonal oxytocin release in the central amygdala attenuates fear response. Neuron 73, 553–566 (2012).
Viviani, D. et al. Oxytocin selectively gates fear responses through distinct outputs from the central amygdala. Science 333, 104–107 (2011).
Jurek, B. & Neumann, I. D. The oxytocin receptor: from intracellular signaling to behavior. Physiol. Rev. 98, 1805–1908 (2018).
Lefevre, A. et al. Oxytocinergic feedback circuitries: an anatomical basis for neuromodulation of social behaviors. Front Neural Circuits 15, 688234 (2021).
Maldonado, P. P. et al. Oxytocin shapes spontaneous activity patterns in the developing visual cortex by activating somatostatin interneurons. Curr. Biol. 31, 322–333.e5 (2021).
Takayanagi, Y. & Onaka, T. Roles of oxytocin in stress responses, allostasis and resilience. Int J. Mol. Sci. 23, 150 (2021).
Baldi, E. et al. Oxytocin and fear memory extinction: possible implications for the therapy of fear disorders?. Int J. Mol. Sci. 22, 10000 (2021).
Moscarello, J. M. & Penzo, M. A. The central nucleus of the amygdala and the construction of defensive modes across the threat-imminence continuum. Nat. Neurosci. 25, 999–1008 (2022).
Olivera-Pasilio, V. & Dabrowska, J. Oxytocin promotes accurate fear discrimination and adaptive defensive behaviors. Front Neurosci. 14, 583878 (2020).
van den Burg, E. H. & Hegoburu, C. Modulation of expression of fear by oxytocin signaling in the central amygdala: From reduction of fear to regulation of defensive behavior style. Neuropharmacology 173, 108130 (2020).
Wahis, J. et al. Astrocytes mediate the effect of oxytocin in the central amygdala on neuronal activity and affective states in rodents. Nat. Neurosci. 24, 529–541 (2021).
Baudon, A., Clauss Creusot, E., Althammer, F., Schaaf, C. P. & Charlet, A. Emerging role of astrocytes in oxytocin-mediated control of neural circuits and brain functions. Prog. Neurobiol. 217, 102328 (2022).
Kofuji, P. & Newman, E. A. Potassium buffering in the central nervous system. Neuroscience 129, 1045–1056 (2004).
Nagai, J. et al. Behaviorally consequential astrocytic regulation of neural circuits. Neuron 109, 576–596 (2021).
Ebner, K., Bosch, O. J., Krömer, S. A., Singewald, N. & Neumann, I. D. Release of oxytocin in the rat central amygdala modulates stress-coping behavior and the release of excitatory amino acids. Neuropsychopharmacology 30, 223–230 (2005).
Broussot, L. et al. A non-canonical GABAergic pathway to the VTA promotes unconditioned freezing. Mol. Psychiatry 27, 4905–4917 (2022).
Lee, H.-J., Macbeth, A. H., Pagani, J. H. & Young, W. S. Oxytocin: the great facilitator of life. Prog. Neurobiol. 88, 127–151 (2009).
Tao, Y. et al. Sex differences in a corticosterone-induced depression model in mice: Behavioral, neurochemical, and molecular insights. Brain Res. 1823, 148678 (2024).
Daigle, T. L. et al. A suite of transgenic driver and reporter mouse lines with enhanced brain-cell-type targeting and functionality. Cell 174, 465–480.e22 (2018).
Ryan, P. J., Ross, S. I., Campos, C. A., Derkach, V. A. & Palmiter, R. D. Oxytocin-receptor-expressing neurons in the parabrachial nucleus regulate fluid intake. Nat. Neurosci. 20, 1722–1733 (2017).
Pannasch, U. et al. Connexin 30 sets synaptic strength by controlling astroglial synapse invasion. Nat. Neurosci. 17, 549–558 (2014).
Zhou, B., Zuo, Y. & Jiang, R. Astrocyte morphology: Diversity, plasticity, and role in neurological diseases. CNS Neurosci. Ther. 25, 665–673 (2019).
Ostroff, L. E., Manzur, M. K., Cain, C. K. & Ledoux, J. E. Synapses lacking astrocyte appear in the amygdala during consolidation of Pavlovian threat conditioning. J. Comp. Neurol. 522, 2152–2163 (2014).
Zhang, W.-H., Zhang, J.-Y., Holmes, A. & Pan, B.-X. Amygdala Circuit Substrates for Stress Adaptation and Adversity. Biol. Psychiatry 89, 847–856 (2021).
Li, H. et al. Experience-dependent modification of a central amygdala fear circuit. Nat. Neurosci. 16, 332–339 (2013).
Nishiyama, A., Yang, Z. & Butt, A. Astrocytes and NG2-glia: what’s in a name?. J. Anat. 207, 687–693 (2005).
Pannasch, U. & Rouach, N. Emerging role for astroglial networks in information processing: from synapse to behavior. Trends Neurosci. 36, 405–417 (2013).
Busnelli, M. et al. Functional selective oxytocin-derived agonists discriminate between individual G protein family subtypes. J. Biol. Chem. 287, 3617–3629 (2012).
Passoni, I., Leonzino, M., Gigliucci, V., Chini, B. & Busnelli, M. Carbetocin is a Functional Selective Gq Agonist That Does Not Promote Oxytocin Receptor Recycling After Inducing β-Arrestin-Independent Internalisation. J. Neuroendocrinol. 28, n/a (2016).
Campbell-Smith, E. J., Holmes, N. M., Lingawi, N. W., Panayi, M. C. & Westbrook, R. F. Oxytocin signaling in basolateral and central amygdala nuclei differentially regulates the acquisition, expression, and extinction of context-conditioned fear in rats. Learning & Memory 22, 247.
Oliet, S. H. R. et al. Neuron-glia interactions in the rat supraoptic nucleus. Prog. Brain Res 170, 109–117 (2008).
Theodosis, D. T., Poulain, D. A. & Oliet, S. H. R. Activity-dependent structural and functional plasticity of astrocyte-neuron interactions. Physiol. Rev. 88, 983–1008 (2008).
Zhang, X., Flick, K., Rizzo, M., Pignatelli, M. & Tonegawa, S. Dopamine induces fear extinction by activating the reward-responding amygdala neurons. Proc. Natl. Acad. Sci. USA 122, e2501331122 (2025).
Theodosis, D. T., Montagnese, C., Rodriguez, F., Vincent, J. D. & Poulain, D. A. Oxytocin induces morphological plasticity in the adult hypothalamo-neurohypophysial system. Nature 322, 738–740 (1986).
Carter, C. S. et al. Is Oxytocin ‘Nature’s Medicine’?. Pharm. Rev. 72, 829–861 (2020).
de Vries, G. J. Sex differences in vasopressin and oxytocin innervation of the brain. Prog. Brain Res 170, 17–27 (2008).
Gigliucci, V. et al. Oxytocin receptors in the Magel2 mouse model of autism: Specific region, age, sex and oxytocin treatment effects. Front Neurosci. 17, 1026939 (2023).
Escartin, C., Guillemaud, O. & Carrillo-de Sauvage, M.-A. Questions and (some) answers on reactive astrocytes. Glia 67, 2221–2247 (2019).
Khakh, B. S. & Sofroniew, M. V. Diversity of astrocyte functions and phenotypes in neural circuits. Nat. Neurosci. 18, 942–952 (2015).
Lawal, O., Ulloa Severino, F. P. & Eroglu, C. The role of astrocyte structural plasticity in regulating neural circuit function and behavior. Glia 70, 1467–1483 (2022).
Verhoog, Q. P., Holtman, L., Aronica, E. & van Vliet, E. A. Astrocytes as guardians of neuronal excitability: mechanisms underlying epileptogenesis. Front Neurol. 11, 591690 (2020).
Henneberger, C. et al. LTP induction boosts glutamate spillover by driving withdrawal of perisynaptic astroglia. Neuron 108, 919–936.e11 (2020).
Ceyzériat, K., Abjean, L., Carrillo-de Sauvage, M.-A., Ben Haim, L. & Escartin, C. The complex STATes of astrocyte reactivity: How are they controlled by the JAK-STAT3 pathway?. Neuroscience 330, 205–218 (2016).
Wang, Y.-F. & Hatton, G. I. Astrocytic plasticity and patterned oxytocin neuronal activity: dynamic interactions. J. Neurosci. 29, 1743–1754 (2009).
Jiang, M. & Bajpayee, N. S. Molecular mechanisms of go signaling. Neurosignals 17, 23–41 (2009).
Cheng, Y.-T. et al. Inhibitory input directs astrocyte morphogenesis through glial GABABR. Nature 617, 369–376 (2023).
Soto, J. S. et al. Astrocyte Gi-GPCR signaling corrects compulsive-like grooming and anxiety-related behaviors in Sapap3 knockout mice. Neuron S0896-6273(24)00541–5 https://doi.org/10.1016/j.neuron.2024.07.019 (2024).
Pfeil, E. M. et al. Heterotrimeric G Protein Subunit Gαq Is a Master Switch for Gβγ-Mediated Calcium Mobilization by Gi-Coupled GPCRs. Mol. Cell 80, 940–954.e6 (2020).
Baudon, A. et al. Calcium imaging and BAPTA loading of amygdala astrocytes in mouse brain slices. STAR Protoc. 3, 101159 (2022).
Acknowledgements
This work was supported by the Centre National de la Recherche Scientifique contract UPR3212, the Université de Strasbourg contract UPR3212; the NeuroStra Interdisciplinary Thematic Institute of the ITI 2021-2028 programme; the Agence Nationale de la Recherche (ANR, French Research Foundation) grants n° 23-CE37-0015-01, 19-CE16-0011-0, 19-CE37-0019 and 20-CE18-0031, the FRM Equipe grant EQU202403018071, the Region Grand Est 19_GE9_043 grant (to AC); the Graduate School of Pain EURIDOL, ANR-17-EURE-0022 (to AC, ECC and VGre); the CNRS International Research Project grant ICOT2023 (to AC and VGri); the FRM Fellowships FDT202204015114 (to AB) and FDT202504020277 (to VGre); The Taiwan National Science and Technology Council fellowship 113-2917-I-564-020 (to K-YW); the Graduate School GRK2174 of the German Research Foundation (to BDB), the EMBO Short Term Fellowship and the Procope Mobility Fellowship (to EV); the graduate school Signaling and Integrated Networks in Biology (to TL); a Fondation Vaincre Alzheimer pilot grant (to LBH); the Deutsche Krebshilfe project 70114190 (to JK); the DFG grants 465/34-1 and 465/36-1 (to IDN), AL 2466/2-1 (to FA) and SFB1158-3 (to VGri). We thank Noémie Willem and the ComptOpt plateform (Comportement et Optogénétique, CNRS and CoRTecS Université de Strasbourg) for nociception experiments, and the SMPMS plateform for corticostesrone quantification (CNRS and CoRTecS Université de Strasbourg). We thank Carole Escartin for feedback on Western blot experiments, Yuval Podpecan and Moritz Wimmer for assistance with confocal microscopy and image analysis, and all our colleagues for the enriching scientific discussions led during the various scientific meetings we attempted.
Author information
Authors and Affiliations
Contributions
Project conception, AC; Methodology, AA, AB, AC, FA, FWP, IDN, JK, K-YW, LB, MB, NR, PD, QK, VGre, VGri; Proteomic analysis, ASA, JK; 3D reconstructions and analysis: ACK, JAM, BD-B, EME, FA, FF, FS, JH, NR, QK, RB, TS, VGri; Electron microscopy, FWP, VD; In vitro BRET, FP, MB; Ex vivo patch-clamp electrophysiology, AB, ECC, PRP, VGre; Ex vivo calcium imaging, AB, CD, FP; In vivo calcium imaging, AA, K-YW, YY; Western blot: LB, M-AC-dS, TL; Behavior analysis: ACe, MK, VGre; Corticosterone dosage, YG, VA, VH, VGre; Oxytocin dosage, FCD, IDN; Writing, AB, AC, CPS, FA, FWP, K-YW, LB, VGre; Ilustrations, AB, AC, VGre; Project administration and supervision, AC.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Marta Navarrete and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Baudon, A., Grelot, V., Wang, KY. et al. Stress induces oxytocin-Gαi-dependent remodeling of astrocytes to shape neuronal response in the amygdala. Nat Commun 17, 1364 (2026). https://doi.org/10.1038/s41467-025-68114-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-68114-4