Abstract
Magnetic refrigeration, utilizing the magnetocaloric effect in solid-state magnets, has emerged as a promising cooling technology. The development of practical ultra-low-temperature magnetic refrigeration applications has been limited by the absence of high-performance magnetic refrigerants. Herein, we provide a frustrated Gadolinium-dominated magnet, Gd2B2MoO9 oxide, which exhibits remarkable ultra-low temperature magnetocaloric performances. The maximum magnetic entropy changes reach 13.3 (76.6), 33.7 (194.0) and 45.1 (259.8) J/kgK (mJ/cm3K) under relatively low magnetic field changes of 0-1, 0-2 and 0-3 T, respectively. A minimum temperature of 0.16 K has been achieved by a custom-built quasi-adiabatic demagnetization apparatus. These magnetocaloric performances surpass those of the commercial ultra-low temperature magnetic refrigerant Gd3Ga5O12 and outperform most of recently reported materials. Combined with its relatively high density and environmental stability, the geometrically frustrated Gd2B2MoO9 oxide is established as an attractive ultra-low temperature magnetic refrigerant.
Similar content being viewed by others
Introduction
Ultra-low temperature conditions are crucial for various cutting-edge technological and scientific fields, such as space exploration, superconductor science, quantum physics and technology1,2,3,4,5,6,7. However, current refrigeration technologies rely heavily on liquid He, which is a depleting resource and increasingly difficult to obtain8. Thus, there is an urgent need to explore alternative cooling methods that can operate without liquid He8,9,10,11. Magnetic refrigeration (MR)11,12,13, which utilizes the magnetocaloric (MC) effect in solid-state magnets, offers an excellent alternative. The MC effect, an inherent magneto-thermal property of magnets11,12,13, can be primarily quantified by the magnetic entropy change (−ΔSM) under certain magnetic field variations (Δμ0H). Ultra-low temperature magnetic refrigerants are generally effective only close to and slightly above their magnetic ordering temperatures (TM). Historically, the low-temperature MR concept was first demonstrated experimentally by adiabatic demagnetization refrigeration (ADR) in the early 1930s14,15,16. Early ultra-low temperature ADR mainly utilized water-containing salts, such as Ce2Mg3(NO3)12·24H2O, Gd2(SO4)3·8H2O, CrK(SO4)2·12H2O, and Fe(SO4)2(NH4)·12H2O15,16,17,18, in which the magnetic ions are separated by water molecules, resulting in ultra-low TM. However, these materials exhibit poor environmental stability and low density of magnetic ions, which limits their widespread applications.
Over the past thirty years, systematic MC characterization has been undertaken in some rare-earth (RE)-dominated magnetic solids19,20,21,22,23,24,25,26,27,28, and some trivalent Gadolinium (Gd)- and divalent Europium (Eu)-dominated magnets with low TM are found to demonstrate notable low-temperature MC performances21,22,23,24,25,26. This is related to the high-spin-only ground state of Gd3+ and Eu2+ ions (J = S = 7/2), which results in low magnetic anisotropy and large magnetic entropy. However, high-performing magnetic refrigerants that are effective at ultra-low temperature below 1 K are still rare, since a higher density of magnetic ions usually results in stronger exchange interactions and thus higher TM19,20,21,22,23,24. Currently, the commercialized ultra-low temperature magnetic refrigerant is gadolinium gallium garnet (Gd3Ga5O12), which exhibits prominent MC performances with −ΔSMmax of 38.4 J/kgK (Δμ0H = 0–7 T) as well as high density (7.09 g/cm3)27,28.
We herein shifted our attention to geometrically frustrated (GF) magnets, which present a diverse class of magnets with peculiar cryogenic physical phenomenon such as large ground state degeneracy, unconventional phase transitions, and unusual quantum spin states29,30,31,32,33,34,35. The magnetic ions in GF systems are generally arranged in nearly equilateral triangle lattices, which prevents the nearest neighbor exchange interactions from being satisfied simultaneously29,30,31. It has also been theoretically proposed around three decades ago that magnetic frustration can enhance low-temperature MC response by suppressing TM with high magnetic ion density and thus maintaining large magnetic entropy at low-temperatures33. As stated above, the Gd3Ga5O12 oxide27,28, the only commercialized ultra-low temperature magnetic refrigerant up to present, shows a typical three corner-sharing garnet type GF structure. Koskelo et al. have provided a comparative study of the MC effects in several Gd-dominated magnets with different GF lattices, and they found that the lattice geometry and degree of magnetic frustration play vital roles in their low-temperature MC performances34. We have recently unveiled the MC properties of pyrochlore-type GF structured Gd2Ti2O7 oxide with strong magnetic frustrations, and a moderate MC performance with −ΔSMmax of 15.43 J/kgK under Δμ0H of 0–5 T was realized35. Nonetheless, the reported GF magnets with prominent ultra-low temperature MC performances remain quite limited due to the lack of material systems with a high density of magnetic ions and a suitable degree of magnetic frustration.
In this work, we present a Gd-dominated GF magnet with moderate magnetic frustration, Gd2B2MoO9 oxide, possessing excellent ultra-low temperature MC performances. This material adopts an antiferromagnetic semiconductor ground state and exhibits remarkable maximum magnetic entropy changes of 13.3 (76.6) and 33.7 (194.0) J/kgK (mJ/cm3K) under low magnetic field changes of 0–1 and 0–2 T, respectively. Utilizing a custom-built quasi-adiabatic demagnetization apparatus, we successfully achieved a minimum temperature of 0.16 K, which is low enough for solid-state quantum computers36,37. These MC performances are significantly better than those of the commercial ultra-low temperature magnetic refrigerant Gd3Ga5O12 and surpass most of the recently reported materials, highlighting Gd2B2MoO9 as a promising candidate for next-generation cryogenic cooling applications.
Results
Structural features and multi-technique characterization of Gd2B2MoO9 oxide
We fabricated the polycrystalline sample of Gd2B2MoO9 oxide using a conventional solid-state reaction route. Its structure and phase purity were examined by powder X-ray diffraction (XRD) at room temperature (RT) and Rietveld refinement using Fullprof38. The resulting XRD profiles (Fig. 1a) show a reliable fit to a triclinic structure (space group P1). The refinement quality factors of RB, Rwp, Rexp and χ2 are all with reasonably low values of 3.85%, 5.04%, 3.07% and 2.69, respectively. The derived lattice parameters of a, b, c and α, β, γ are 5.201(1), 6.948(8), 10.321(2) Å and 73.987(2), 76.146(5), 72.626(4)o, respectively. More detailed, refined structural and refinement parameters are listed in Table S1. The low refinement parameter values confirm the single-phase nature of Gd2B2MoO9 oxide.
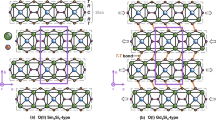
a Experimental and Rietveld-refined powder X-ray diffraction (XRD) profiles indicate the high-quality of the studied sample. b Crystal structure diagram shows the formation of a geometrically frustrated (GF) diamond lattice by the nearest Gd-Gd atoms. c1–c5 Local nearest oxygen coordination environments of Gd1, Gd2, B1, B2, and Mo atoms, respectively.
Figure 1b, c illustrates the schematic structure and nearest-neighbor oxygen environments of Gd, B, and Mo. Two inequivalent Gd sites (Gd1 and Gd2) are each coordinated by eight O atoms, forming distorted GdO8 hexagonal bipyramids (Fig. 1c1, 1c2). The Gd1–O and Gd2–O bond lengths are 2.159(5)−2.781(9) Å and 2.237(6)−2.675(8) Å, respectively. The Gd2–O–Gd2 bond angles range from 100.716(1)° to 115.010(1)°, while the Gd1–O–Gd1 angle is 100.851(0)°. The nearest Gd-Gd atoms form an isolated, slightly distorted, GF diamond lattice (Fig. 1b).
There are two inequivalent B sites (B1 and B2), each bonded in a trigonal planar geometry to three O atoms (Fig. 1c3, 1c4), with B1–O and B2–O bond lengths of 1.397(2)−1.593(1) Å and 1.353(2)−1.672(3) Å, respectively. The Mo atom is coordinated by four O atoms, forming MoO4 tetrahedra (Fig. 1c5) with bond lengths of 1.559(3)−1.996(3) Å. Each GdO8 bipyramid shares a corner with one MoO4 tetrahedra and edges with three other GdO8 bipyramids.
To comprehensively verify the chemical composition and structural features of Gd2B2MoO9 oxide, we employed a combination of spectroscopic and microscopic techniques. The chemical valence states of the constitute elements were determined by RT X-ray photoelectron spectroscopy (XPS). High-resolution core-level XPS spectra for Gd, B, and Mo, along with peak-fitting analyses (Fig. 2a–c), show that the experimental data agree well with the fitted results, confirming the presence of Gd3+, B3+, and Mo6+ in the oxide. Similar to those in recently reported Gd-containing oxides23,35, the profile of 4 d5/2 peak can be fitted by two components, demonstrates the existence of spin-orbit coupling interaction of 4d and 4 f electrons.
a–c Core-level XPS spectra and peak-fitting results for Gd, B, and Mo elements confirm the presence of Gd3+, B3+, and Mo6+ valence states. d High angle angular dark-field scanning transmission electron microscopy (HADDF-STEM) image proves the formation of geometrically frustrated (GF) diamond lattice by Gd-Gd atoms. e Schematic of the nearest Gd-Gd diamond lattice. Gd atoms are red, and Mo atoms are green. f–i Scanning transmission electron microscopy-based elemental energy-dispersive X-ray spectroscopy mapping.
Morphology, chemical, and structural characteristics of Gd2B2MoO9 oxide were further evaluated using scanning electron microscopy (SEM), high-resolution transmission electron microscopy (TEM), and scanning transmission electron microscopy (STEM) coupled with elemental energy-dispersive X-ray spectroscopy (EDS). The constituent elements of Gd, B, Mo and O elements are all uniformly distributed from the microscale to nanoscale (Figs. S1, S2) with the average atomic molar ratios of 15.3(3)%, 13.6(8)%, 7.2(2)% and 63.9(5)%, well-aligned with the nominal composition. The average particle size of Gd2B2MoO9 oxide was determined to be 2.26(8) μm (Fig. S3). These SEM and TEM images, together with elemental EDS mappings, demonstrate the single-phase nature and good homogeneity of the present Gd2B2MoO9 oxide.
An atom-resolved HAADF-STEM image and the corresponding EDS maps, acquired along the [111] zone axis (Figs. 2d, 2f-i), clearly resolve the Gd and Mo atomic columns. Four of the nearest Gd–Gd atoms, forming an isolated diamond lattice, are visible; the enlarged structural diagram (Fig. 2e) further confirms the Gd-dominated diamond-type GF structure of Gd2B2MoO9 oxide.
First-principle insights into the ground state of Gd2B2MoO9 oxide
To complement the experimental characterization, density functional theory (DFT) calculations were conducted to explore the magnetic and electronic properties of Gd2B2MoO9 oxide in its ground state. Spin-polarized generalized gradient approximation (GGA) plus Hubbard U (GGA + U) approach with Ueff values up to 8 eV was performed using VASP software39,40,41. The magnetic ground state was firstly evaluated by computing the total energy (Etot) for various constructed potential spin configurations, including non-magnetic (NM), ferromagnetic (FM), as well as three distinct antiferromagnetic (AFM) states, as illustrated in Fig. 3a–d. The derived values of Etot for various potential magnetic structures with different Ueff values are all summarized in Table S2, in which the AFM configuration exhibits the lowest Etot value, indicating that Gd2B2MoO9 oxide favors an AFM ground state.
a–d Schematic representations of possible magnetic structures, including parallel (ferromagnetic, FM) and three typical anti-parallel (antiferromagnetic, AFM) states. Gd atoms are dark red, B atoms are blue, Mo atoms are green, and O atoms are gray. e Spin-polarized partial and total density of states (DOS) indicate a typical AFM semiconductor character and large spontaneous polarization of Gd3+ ions at the ground state. f–h Charge-density difference visualizations with 2D projections and aerial view show the localized charges around Gd3+ atoms.
Further insights into the electronic structure were obtained by calculating the spin-polarized total density of states (DOS) and the atom-projected DOS for Gd (4 f), B (1 s), Mo (5 s), and O (2p) orbitals of Gd2B2MoO9 oxide without and with different Ueff values up to 8 eV are illustrated in Figs. 3, S4, respectively. Both spin-majority and spin-minority channels in all the DOS are nearly identical, and a distinct band gap (BG) is observed at the Fermi level regardless of the Ueff values, confirming the AFM semiconductor nature of Gd2B2MoO9 oxide. The total DOS is dominated by the Gd(4 f) partial DOS, with significant splitting observed, reflecting strong spontaneous polarization of the Gd3+ ions. The deduced BG value is 2.27(5) eV without Ueff, and it gradually rises to 2.43(8) eV with increasing Ueff up to 8 eV, as illustrated in Fig. S5. Moreover, the calculated average magnetic moments Mcal values range from 6.89(4) to 7.07(3) μB for Gd1, and from 6.91(3) to 7.06(5) μB for Gd2 atoms, respectively, closely matching the theoretical value for a free Gd3+ ion (7.0 μB).
Charge distribution and transfer characteristics were also analyzed using 2D maps and aerial view (Fig. 3f–h). O2− ions share electrons with Gd3+, B3+, and Mo6+ ions, indicating a covalent bonding nature in Gd2B2MoO9 oxide. Greater electronic overlap is observed between Mo6+ and O2− atoms compared to Gd3+ and O2− atoms. The aerial view reveals a positive peak for O2− and a negative peak for B3+ atoms, while charges around Gd3+ atoms are well-localized, resulting in the large magnetic moments observed.
Experimental determination of low-temperature magnetic and thermal properties of Gd2B2MoO9
To comprehensively characterize the magnetic phase transition (MPT) and magnetic frustration properties of Gd2B2MoO9 oxide, a series of magnetic and thermodynamic measurements was performed. The magnetic field (μ0H) and temperature (T) dependent magnetization (M) were measured up to 5 T and down to 0.4 K. The T-dependent inverse magnetic susceptibility, 1/χ (χ = M/μ0H), under μ0H = 1 T, exhibits high-temperature linearity consistent with the Curie-Weiss (C–W) law (Fig. 4a), \(\chi (T)={{{\rm{C}}}}/(T-{\theta }_{{{{\rm{P}}}}})\), wherein C and θ are the C-W constant and paramagnetic Curie temperature, respectively. Additionally, \(C=N{({\mu }_{B}{\mu }_{eff})}^{2}/3{k}_{B}\), wherein N is Avogadro’s number, kB is the Boltzmann constant, and μeff is the effective magnetic moment. Linear fitting above 6 K yields a negative θP of −3.11(5) K, indicative of AFM interactions at the ground state. This is further supported by the downturn in the χΜΤ products at low temperatures (Fig. S6). The effective magnetic moment, μeff, is deduced to be 7.97(3) μB/Gd, closely matching the theoretical value of the free Gd3+ ion (7.94 μB).
a Temperature dependence of inverse magnetic susceptibility 1/χ (T) curves and Curie-Wiess fitting line under a magnetic field of 1 T; inset: Temperature dependence of magnetic susceptibility χ(T) curves under μ0H of 0.05, 0.2 and 0.5 T by field-cooled (FC) and zero-field-cooled (ZFC) protocols demonstrating a typical antiferromagnetic ground state and a field-induced metamagnetic transition from antiferromagnetic to ferromagnetic-like state. b Temperature dependence of heat capacity C(T) curves under different magnetic fields. c Magnetic field dependence of magnetization M(μ0H) curves at several selected temperatures.
Further insights were obtained from χ(T) measurements under field-cooled (FC) and zero-field-cooled (ZFC) protocols from 0.4 to 1.8 K with μ0H values of 0.05, 0.2, and 0.5 T using a He-3 refrigerator (inset of Fig. 4a). Evidently, χ increases monotonously with decreasing T, peaking near its Néel temperature (TN) of 0.79(5) K, before decreasing as T increases. These observations confirm the AFM ground state of Gd2B2MoO9 oxide under low magnetic fields and align with DFT calculations. Whereas, under higher μ0H of 0.5 T, χ increases monotonously with decreasing T and shows a saturation tendency at low temperatures, illustrating the occurrence of a field-induced metamagnetic transition from AFM ground state to ferromagnetic-like state. Additionally, the T-dependent χZFC and χFC curves demonstrate high reversibility, which is advantageous for MR applications.
The T-dependent heat capacity, C(T), was measured from 4 K down to 0.1 K under various magnetic fields (Fig. 4b). The existence of a pronounced C(T) peak under zero-field and low magnetic fields demonstrates the long-range magnetic ordering in Gd2B2MoO9 oxide, well-aligned with those by magnetic measurements and DFT calculations. The MPT temperature (TN) was deduced to be 0.51(3) K from the inflection point in the temperature-dependent zero-field d(C/T)/dT curve (Fig. S7). The frustration parameter, f = |θP | /TN30,33,34,35, was considered as a measurement of magnetic frustration level. Generally, no magnetic frustration when f value is lower than 1, whereas, being strong magnetic frustration if f was higher than 1030,42. For the present Gd2B2MoO9 oxide, a f value of 6.1(4) has been obtained, which suggests a moderate magnetic frustration, favorable for ultra-low temperature MR33,34,35. At higher magnetic fields, only a broad peak is observed in C(T), with the peak temperature increasing as μ0H increases, consistent with magnetization results. The M(μ0H) curve (Fig. 4c) at low temperatures shows that M increases rapidly at low μ0H and saturates under high μ0H above ~2.0 T. A nearly linear behavior in M(μ0H) curves at high temperatures can be noted, consistent with the PM state. These findings corroborate DFT results, further confirming the dominate role of gadolinium in the magnetism of Gd2B2MoO9 oxide.
Discussion
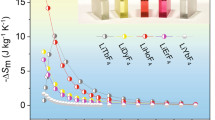
The MC performance of Gd2B2MoO9 oxide was firstly assessed using −ΔSM(T, Δμ0H) (Fig. 5a, b) calculated from M(μ0H) (Fig. S8) and C(T, μ0H) (Fig. 4b) data with the temperatures ranging from 0.5 to 10 K and 0.1 to 4 K, respectively. Subsequently, we estimated the relative cooling power (RCP) (Fig. S9) from the resulting −ΔSM(T, Δμ0H) (Fig. 5a) data. The detailed calculation methods43 are provided in the supplementary file. Although some differences in values exist, which are well within the intrinsic uncertainties documented by Pecharsky & Gschneidner43, the −ΔSM(T, Δμ0H) results from both approaches are consistent. The −ΔSM value increases with decreasing temperature under each Δμ0H, reaches a maximum (−ΔSMmax), and then decreases. Under low Δμ0H (0–0.5, 0–1, and 0–1.5 T), as temperature falls below ~0.5 K (Fig. 5b), the values of −ΔSM change from positive to negative, indicating a transition from conventional to inverse MC effect, consistent with the AFM ground state inferred from magnetization and DFT results. In the section below, we use the ΔSM results calculated from M(μ0H) data for discussion and comparison since most of the reported results are also estimated by this method. Moreover, the −ΔSMmax and RCP values all increase significantly with increasing Δμ0H (Fig. S9), reaching 13.3, 33.7, and 55.0 J/kgK and 35.0, 113.9, and 378.5 J/kg under Δμ0H = 0–1, 0–2, and 0–5 T, respectively. Despite the fact that these −ΔSMmax and RCP values are still lower than several updated Gd-based metal-organic framework materials and divalent Eu-based oxides24,25,26,27,28,44,45,46, the present Gd2B2MoO9 oxide evidently has its own benefits of high density (5.76(5) g/cm3), easy to be fabricated by a simple solid-state route, as well as good environmental stability.
Temperature dependence of magnetic entropy change −ΔSM(T) derived from (a) M(H) data and (b) C(T) results. c Maximum volumetric entropy change −ΔSVmax (light green column, left scale) and maximum mass entropy change −ΔSMmax (light purple column, right scale) under Δμ0H = 0–2 T, which are much larger than commercial ultra-low temperature magnetic refrigerant Gd3Ga5O12 and surpass most of the benchmark ultra-low temperature magnetic refrigerants.
For practical MR applications, minimizing system size is essential, making the volumes of MC materials and magnets critical factors. As such, evaluating the magnetic entropy change in volumetric terms (−ΔSV), expressed in the unit of mJ/cm3K, under relatively low Δμ0H (0–2 T) achievable by permanent magnets, is more suitable for assessing the MC performance of magnetic refrigerants. We therefore summarize the −ΔSmax values under Δμ0H = 0–2 T, both per unit mass and per unit volume, for Gd2B2MoO9 oxide, in comparison with commercial Gd3Ga5O12 oxide and other recently reported ultra-low temperature MC materials44,45,46,47,48,49,50,51,52,53,54 (Fig. 5c). While the mass-based −ΔSmax value of Gd2B2MoO9 oxide under Δμ0H = 0–2 T is lower than that of some reported materials24,25,26, its volumetric −ΔSVmax is approximately 1.88 times higher than commercial Gd3Ga5O12 and exceeds most recently realized ultra-low temperature MC materials27,28,46,47,48,49,50,51,52,53, positioning Gd2B2MoO9 oxide among the leading ultra-low temperature magnetic refrigerants.
We further assessed the practical low-temperature cooling performances of Gd2B2MoO9 oxide by direct measure its adiabatic demagnetization cooling T(μ0H) curves (Fig. 6) using a custom-designed apparatus6, with initial conditions and field control provided by a PPMS-9T system and by indirect calculation of adiabatic temperature change (ΔTad) below 4 K (inset of Fig. 6) from C(T, μ0H) data, respectively. The resulting T(μ0H) curves, starting from 9 T, for both Gd2B2MoO9 and Gd3Ga5O12 oxides at various initial temperature (To), exhibit three distinct stages: initially, the temperature decreases continuously with decreasing μ0H and reaches a minimum temperature (Tmin) at a critical field (μ0Hcr); subsequently, the temperature increases slightly as the field is further reduced, indicating a transition from conventional to inverse MC effect. These behaviors are well consistent with those of indirectly calculated ΔTad (inset of Fig. 6) results, in which the values of ΔTad under low Δμ0H clearly change from positive to negative around 0.71(3) K, further proving the AFM ground state. Additionally, the deduced ΔTad values by direct measurement method (marked by star) are well-aligned with those by indirectly calculated from C(T, μ0H) data43, as illustrated in the inset of Fig. 6. The values of Tmin and μ0Hcr of Gd2B2MoO9 oxide are: 0.41 K and 0.62 T at T0 = 6 K; 0.28 K and 0.75 T at T0 = 4 K; 0.21 K and 0.84 T at T0 = 3 K; and 0.16 K and 1.04 T at T0 = 2 K, respectively. Under identical conditions, the Tmin values are obviously lower than those of commercial Gd3Ga5O12, making it promising for practical ultra-low temperature applications. Moreover, Gd2B2MoO9 oxide can maintain the minimum temperature for more than half an hour (Fig. S10), demonstrating significant cooling capacity. It is worth noting that the achieved temperature of 0.16 K is well below the operating temperature (~1 K) of state-of-the-art silicon-based quantum computers36,37, thereby enhancing the prospects for practical quantum computing applications. Additionally, Gd2B2MoO9 oxide can be synthesized via a simple solid-state reaction route, and possess high chemical stabilities, high density, and environmental friendliness. These attributes underscore the potential of Gd2B2MoO9 oxide as an excellent candidate for practical ultra-low temperature MR applications.
Direct measured the adiabatic demagnetization cooling T(μ0H) curves highlights a refrigeration down to 0.16 K, surpassing the commercial Gd3Ga5O12 refrigerant. Inset presents temperature dependence of adiabatic temperature change ΔTad(T) curves by indirect calculation (dashed lines) and by direct measurement (marked by stars).
Methods
Sample synthesis
Polycrystalline Gd2B2MoO9 oxide was fabricated by a solid-state reaction from high-purity (≥ 99.9 wt.%) MgCO3, H3BO3, and (NH4)6Mo7O24·4H2O powders. The raw materials, weighted in stoichiometric proportions, were thoroughly ground by hand for 2 h with the addition of a small amount of alcohol to facilitate mixing. To compensate for the volatilization of H3BO3, a 6% excess was included to improve reproducibility. The mixture was preheated at 700 °C for 10 h. The resulting precursor was reground, pressed into thin pellets, and sintered in air at 900 °C for 18 h, followed by furnace cooling.
Microstructural characterization
The phase purity and crystal structure of Gd2B2MoO9 oxide were examined by powder XRD at RT using a Rigaku-SmartLab diffractometer. The elemental chemical states were analyzed using XPS (Thermo-Scientific K-Alpha). The density was determined to be 5.76(5) g/cm3 by Archimedes method. Microscopic morphology and EDS mapping were assessed using SEM (SM-7800F), TEM (FEI-TalosF200s), and STEM (FEI Titan-T300kV).
Physical property measurements
Magnetization measurements were acquired by a SQUID magnetometer (MPMS-Q3, Quantum Design) equipped with a 3He refrigerator inset, using an irregular bulk sample with a mass of 3.65(4) mg. The field-dependent magnetization data used for the magnetic entropy change calculations were recorded during cooling and by incrementally measuring the field from 0 to 5 T. Specific heat measurements were performed using the adiabatic heat relaxation method with the PPMS-9T and a dilution refrigerator module; the sample mass in this case was 1.16(3) mg. All magnetic field-dependent addenda measurements were conducted under the magnetic fields of 0, 0.5, 1, 1.5, 2, and 5 T.
Calculation details
Atomic-level spin-polarized first-principles calculations were performed within the framework of DFT using commercial VASP software39,40,41. The valence electron contributions used [5s2 5p6 4f7 5d1 6s2] for Gd, [2s2 3p1] for B, [4d5 5s1] for Mo, and [2s2 2p4] for O in the PAW pseudopotentials. The k-point mesh for Gd2B2MoO9 oxide was set as 7 × 5 × 3 using the Monkhorst-Pack method. Structural optimization of Gd2B2MoO9 oxide was performed until the total energies and Hellman-Feynman forces on each atom were converged to below 0.01 eV/Å and 10−7 eV, respectively.
Quasi-adiabatic demagnetization device
The adiabatic demagnetization cooling μ0H(T) curves of Gd2B2MoO9 oxide were determined using a custom-built apparatus5, with initial conditions and field control provided by a PPMS-9T system. Approximately 3 g of powder sample was mixed with ~50 wt.% silver powder to enhance thermal conductivity and cold-pressed into a cylindrical pellet. Then, the sample pellet was mounted on a sapphire-plate holder, which was supported by a Vespel frame. A field-calibrated RuO2 thermometer with the uncertainty less than 0.001 K was attached to the sample pellet for temperature measurements. The sample pellet was precooled to T0 and stabilized in a chamber where residual gases were evacuated using a cryopump to ensure adiabatic conditions. The μ0H(T) curves were acquired by decreasing the external μ0H at a rate of 0.3 T/min from 9 to 0 T.
Data availability
The data that support the findings of this study are provided in the main text and the Supplementary Information. Source data are provided with this paper.
References
Xue, X. et al. CMOS-based cryogenic control of silicon quantum circuits. Nature 593, 205–210 (2021).
Dong, M. et al. High-speed programmable photonic circuits in a cryogenically compatible, visible–near-infrared 200 mm CMOS architecture. Nat. Photon. 16, 59–65 (2022).
Rowe, E. et al. Resonant enhancement of photo-induced superconductivity in K3C60. Nat. Phys. 19, 1821–1826 (2023).
Pappalardo, R. T. et al. Science overview of the Europa Clipper mission. Space Sci. Rev. 220, 40 (2024).
Xiang, J. S. et al. Giant magnetocaloric effect in spin supersolid candidate Na2BaCo(PO4)2. Nature 625, 270–275 (2024).
Zhu, P. et al. Emergent quantum Majorana metal from a chiral spin liquid. Nat. Commun. 16, 2420 (2025).
Liebster, N. et al. Supersolid-like sound modes in a driven quantum gas. Nat. Phys. 21, 1064–1070 (2025).
Cho, A. Helium-3 shortage could put the freeze on low-temperature research. Science 326, 778–779 (2009).
Moya, X., Kar-Narayan, S. & Mathur, N. D. Caloric materials near ferroic phase transitions. Nat. Mater. 13, 439–450 (2014).
Lin, J. C. et al. Colossal and reversible barocaloric effect in liquid-solid-transition materials n-alkanes. Nat. Commun. 13, 596 (2022).
Klinar, K., Law, J. Y., Franco, V., Moya, X. & Kitanovski, A. Perspectives and energy applications of magnetocaloric, pyromagnetic, electrocaloric, and pyroelectric materials. Adv. Energy Mater. 14, 2401739 (2024).
Zhang, Y. K. et al. Rare-Earth-Free Mn30Fe20-xCuxAl50 magnetocaloric materials with stable cubic cscl-type structure for room-temperature refrigeration. Adv. Funct. Mater. 33, 2310047 (2023).
Franco, V. et al. A. Magnetocaloric effect: from materials research to refrigeration devices. Prog. Mater. Sci. 93, 112–232 (2018).
Debye, P. Einige Bemerkungen zur Magnetisierung bei tiefer Temperatur. Ann. Phys. 386, 1154–1160 (1926).
Giauque, W. F. A thermodynamic treatment of certain magnetic effects. A proposed method of producing temperatures considerably below 1° absolute. J. Am. Chem. Soc. 49, 1864–1870 (1927).
Giauque, W. F. & MacDougall, D. P. Attainment of temperatures below 1° absolute by demagnetization of Gd2(SO4)3·8H2O. Phys. Rev. 43, 768 (1933).
Barclay, J. A., Rosenblum, S. S. & Steyert, W. A. Low temperature thermal conductivity of some composite materials. Cryogenics 16, 539–543 (1976).
Bulled, J. M. et al. Geometric frustration on the trillium lattice in a magnetic metal-organic framework. Phys. Rev. Lett. 128, 177201 (2022).
Law, J. Y. & Franco, V. Modern rare-earth-containing magnetocaloric materials: Standing on the shoulders of giant Gd5Si2Ge2. Handbook on the Physics and Chemistry of Rare Earths 64, 175–246 (2023).
Zhang, Y. et al. Investigation of the structural and magnetic properties of the GdCoC compound featuring excellent cryogenic magnetocaloric performance. Acta Mater. 276, 120128 (2024).
Zhong, H. et al. Design of excellent mechanical performances and magnetic refrigeration via in situ forming dual-phase alloys. Adv. Mater. 36, 2402046 (2024).
Tang, X. et al. Magnetic refrigeration material operating at a full temperature range required for hydrogen liquefaction. Nat. Commun. 13, 1817 (2022).
Chen, F. et al. Structural and magnetic characterization of weberite-type RE3NbO7 (RE = Gd, Dy, Ho and Er) ceramics featuring notable cryogenic magnetocaloric responses. Sci. China Mater. 68, 2828–2840 (2025).
Xu, Q. et al. Gd(OH)F₂: a promising cryogenic magnetic refrigerant. J. Am. Chem. Soc. 144, 13787–13793 (2022).
Ye, Z. X. et al. Coexistence of antiferromagnetic and ferromagnetic interactions in dimer-like arranged EuAl2O4 systems: regulatory strategy to giant low-field cryogenic magnetocaloric effects. Adv. Funct. Mater. 2509843 (2025).
Wang, B. J. et al. A record-high cryogenic magnetocaloric effect discovered in EuCl2 compound. J. Am. Chem. Soc. 146, 35016–35022 (2024).
Hamilton, A. C. S., Lampronti, G. I., Rowley, S. E. & Dutton, S. E. Enhancement of the magnetocaloric effect driven by changes in the crystal structure of Al-doped GGG, Gd3Ga5-xAlxO12 (0 ≤ x ≤ 5). J. Phys.: Condens. Matt. 26, 116001 (2014).
Kleinhans, M. et al. Magnetocaloric properties of R3Ga5O12 (R = Tb, Gd, Nd, Dy). Phys. Rev. Appl. 19, 014038 (2023).
Bramwell, S. T. & Gingras, M. J. P. Spin ice state in frustrated magnetic pyrochlore materials. Science 294, 1495–1501 (2001).
Gardner, J. S., Gingras, M. J. P. & Greedan, J. E. Magnetic pyrochlore oxides. Rev. Mod. Phys. 82, 53 (2010).
Mukherjee, P. & Dutton, S. E. Enhanced magnetocaloric effect from cr substitution in ising lanthanide gallium garnets Ln3CrGa4O12 (Ln = Tb, Dy, Ho). Adv. Funct. Mater. 27, 1701950 (2017).
Li, H. et al. Magnetocaloric effect of topological excitations in Kitaev magnets. Nat. Commun. 15, 7011 (2024).
Zhitomirsky, M. E. Enhanced magnetocaloric effect in frustrated magnets. Phys. Rev. B 67, 104421 (2003).
Koskelo, E. A. C. et al. Comparative Study of magnetocaloric properties for Gd3+ compounds with different frustrated lattice geometries. PRX Energy 2, 033005 (2023).
Zhang, Y., Hao, W., Lin, J., Li, H. F. & Li, L. Geometrically frustrated Gd2Ti2O7 oxide: a comprehensive exploration of structural, magnetic, and magnetocaloric properties for cryogenic magnetic cooling applications. Acta Mater 272, 119946 (2024).
Yang, C. H. et al. Operation of a silicon quantum processor unit cell above one kelvin. Nature 580, 350–354 (2020).
Noah, G. M. et al. CMOS on-chip thermometry at deep cryogenic temperatures. Appl. Phys. Rev. 11, 021414 (2024).
Rodriguez-Carvajal, J. FULLPROF: a Rietveld and pattern-matching analysis program. Laboratoire Léon Brillouin CEA-CNRS, France (2007).
Perdew, J., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865 (1996).
Evarestov, R. A. & Smirnov, V. P. Modification of the Monkhorst-Pack special points meshes in the Brillouin zone for density functional theory and Hartree-Fock calculations. Phys. Rev. B 70, 233101 (2004).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Ramirez, A. P. Strongly geometrically frustrated magnets. Annu. Rev. Mater. Sci. 24, 453–480 (1994).
Pecharsky, V. K. & Gschneidner, K. A. Jr. Magnetocaloric effect from indirect measurements: magnetization and heat capacity. J. Appl. Phys. 86, 565–575 (1999).
Tziotzi, T. G. et al. A {Gd12Na6} molecular quadruple-wheel with a record magnetocaloric effect at low magnetic fields and temperatures. J. Am. Chem. Soc. 145, 7743–7747 (2023).
Wang, Y. P. et al. Giant low-field cryogenic magnetocaloric effect in a polycrystalline EuB4O7 compound. J. Am. Chem. Soc. 146, 3315–3322 (2024).
Xu, N., Chen, W. M., Ding, Y. S. & Zheng, Z. P. A cubic Tinkertoy-like heterometallic cluster with a record magnetocaloric effect. J. Am. Chem. Soc. 146, 9506–9511 (2024).
Zhang, Y. K., Na, Y. Z., Hao, W. X., Gottschall, T. & Li, L. W. Enhanced cryogenic magnetocaloric effect from 4f-3d exchange interaction in B-Site ordered Gd2CuTiO6 double perovskite oxide. Adv. Funct. Mater. 34, 2409061 (2024).
Lorusso, G. et al. A dense metal-organic framework for enhanced magnetic refrigeration. Adv. Mater. 25, 4653–4656 (2013).
Zhou, C. Q. & Li, R. K. Gd3TeBO9: a rare-earth borate with significant magnetocaloric effect. Chem. Eur. J. 30, e202303048 (2024).
Yang, Z. Y. et al. Large magnetocaloric effect in gadolinium borotungstate Gd3BWO9. J. Mater. Chem. C. 8, 11866 (2020).
Mo, Z. J. et al. Magnetic properties and large low-field cryogenic magnetocaloric effect in the divalent europium borate Eu2B2O5 compound. Mater. Today Phys. 41, 101351 (2024).
Delacotte, C. et al. NaGdS2: a promising sulfide for cryogenic magnetic cooling. Chem. Mater. 34, 1829–1837 (2022).
Xu, Q. F. et al. Enhancing the performance of magnetic refrigerants through tuning their magnetism from antiferromagnetism to weak ferromagnetism. Sci. China Mater. 65, 3171–3174 (2022).
Zhang, Y. K., Li, A. S., Hao, W. X., Li, H. F. & Li, L. W. Apatite-type gadolinium-based dense MGd4Si3O13 (M = Mg, Ca, Sr) ceramics: an emerging class of sub-liquid helium temperature magnetic refrigerant. Acta Mater. 292, 121033 (2025).
Acknowledgments
The present work was supported by the National Natural Science Foundation of China (Grant Nos. 52472274 L.W.L. and 52572290 Y.K.Z.) and Zhejiang Provincial Natural Science Foundation of China (Grant No. LZ25E020002 Y.K.Z.). The authors of L.W.L. and Y.K.Z. also acknowledge Dr. Chao Zhang from the Instrumentation and Service Center for Physical Sciences at Westlake University for the magnetization measurements and the Supercomputing Center of Hangzhou Dianzi University for providing computing resources.
Author information
Authors and Affiliations
Contributions
Y.K.Z., X.F.Z., and L.W.L. raised the idea. L.W.L., H.F.L., P.J.S., and S.Q.Z. conceived the experiments. F.Y.C. and Y.K.Z. synthesized and characterized the sample. Y.Z.N., L.W.L., and Y.K.Z. performed the magnetization measurements and DFT calculations. X.Y.L. and J.S.X. performed the Quasi-Adiabatic Demagnetization measurements. All authors participated in the data analysis and manuscript preparation.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Sagar Ghorai and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, Y., Na, Y., Liu, X. et al. Refrigeration down to 0.16 K using a frustrated magnet Gd2B2MoO9. Nat Commun 17, 1554 (2026). https://doi.org/10.1038/s41467-025-68278-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-025-68278-z