Abstract
Aromatase inhibitors are widely used in the treatment of hormone-sensitive breast cancer, but their suppression of estrogen production accelerates bone loss, increases fracture risk, and negatively impacts muscle and fat metabolism. Here, we demonstrate that daily low intensity vibration, serving as a non-drug mimetic for exercise, protects musculoskeletal health in skeletally immature, female mice under complete estrogen deprivation. Subsequent improvements in vertebral bone density are paralleled by greater and leaner skeletal muscle mass and function alongside reduced fat accretion and circulating metabolites. In mature, estrogen deprived mice, vibration enhances weekly bisphosphonate treatment, improving bone density, cortical thickness, and mechanical resistance to fracture. These findings support the proposed hypothesis that low intensity vibration reduces musculoskeletal frailty in estrogen deprived mice, with stronger effects observed in younger cohorts, while in skeletally mature mice combination therapy with anti-resorptive treatment is necessary to suppress cancer-treatment induced musculoskeletal degradation.
Similar content being viewed by others
Introduction
Breast cancer is among the most prevalent malignancies afflicting individuals in the United States1,2. Approximately 80% of individuals with breast cancer have estrogen (E2) receptor-positive disease3,4, with the majority of cases occurring in early- to postmenopausal women. Untreated E2-receptor-positive breast cancer exhibits a high affinity to metastasize to bone, liver, and lung5,6. The skeleton is a favorable site of metastasis due to the high concentration and co-localization of growth factors; some of which are derived from the bone matrix, such as transforming growth factor-β (TGF-β)7, and others are directly secreted by the tumor, including parathyroid hormone-related peptide8,9. By virtue of proximity, these growth factors are locally available to drive tumor progression7,10,11,12,13. Suppressing tumor advancement through pharmacologic intervention and/or radiologic modalities is integral to prolonging patient lives14,15,16. Clinical strategies to inhibit breast cancer progression include selective E2-receptor (ER) modulators (e.g., tamoxifen)17 for pre-menopausal patients and hormone deprivation therapies (e.g., aromatase inhibitors; AI)18 for postmenopausal patients. Third-generation AIs (e.g., letrozole) curtail peripheral estradiol synthesis from adrenal tissue by inhibiting aromatase production19,20. These anti-E2 therapies are highly effective in decreasing tumor-associated mortality and increasing disease-free survival.
While the inhibition of estradiol synthesis by aromatase inhibitors (AIs) confers therapeutic efficacy in postmenopausal patients, this intervention concurrently promotes osteoclast-mediated bone resorption, thereby exacerbating skeletal degeneration and increasing the risk of secondary osteoporotic complications21,22,23. Such bone loss not only fuels tumor growth24 but leads to increased release of TGF-β, which exacerbates muscle weakness25,26,27 through dysregulation of ryanodine receptor-1 (RyR-1), a critical Ca2+-dependent Ca2+-release channel within the sarcoplasmic reticulum28. Denosumab, an antibody recognizing receptor-activator of nuclear-factor Kappa-β ligand (RANKL), is the first line of treatment prescribed to inhibit osteoclast-mediated bone resorption in postmenopausal women29, as well as in patients with hormone receptor-positive breast cancer treated with AIs30,31. Including bisphosphonates [e.g., zoledronic acid (ZA)] in the treatment regimen for breast cancer also effectively inhibits osteoclast activity and reduces cancer recurrence, improving survival in postmenopausal ER+ breast cancer patients32. Bisphosphonates prevent adhesion of metastatic cells to bone and induce osteoclast apoptosis in vivo33 and in vitro34,35. Thus, clinical challenges persist following treatment with AIs, including muscle weakness36, progressive adiposity, and imbalanced bone remodeling37,38. As these toxicities reduce quality of life39, lead to poor patient adherence, and result in treatment discontinuation40,41,42, innovative approaches are needed to reduce the musculoskeletal burden on these individuals to ultimately maintain adherence to AI-mediated therapies and to increase survival.
Daily physical activity is universally recommended as an alternative or supplemental therapy in standards-of-care to prevent bone and muscle loss43,44,45, including in clinical cancer-treatment guidelines. Exercise is known to have anabolic effects on bone and muscle while suppressing fat formation46,47. Additionally, exercise reduces inflammation48 and regulates glucose metabolism. Despite its well-recognized benefits and patient willingness, breast cancer patients often find it difficult to participate in typical exercise regimens due to decreased energy and muscle strength49. Cancer patients also experience heightened pain and frailty; thus, incorporating strenuous loads, such as those induced by exercise, may elevate fracture risk, therein precipitating the fractures that the exercise was intended to prevent50,51.
As an alternative to the high-intensity loads generated by daily locomotion let alone physical activity or exercise regimens52, low-intensity vibration (LIV) provides a low-magnitude (<1 g acceleration, where 1 g = Earth’s gravitational field or 9.8 m/s2) mechanical signal delivered through vertical (uniaxial) acceleration at a physiologically high frequency (~20–100 Hz) but low-magnitude force that is anabolic to bone in both translational and clinical studies53,54. LIV does not require strenuous engagement by the patient, providing a fraction of the dynamic mechanical energy used in postural muscle contraction55 to maintain upright balance, which is itself a significant contributor to bone homeostasis and wanes with age55,56 or as a result of dystrophy or disease54. We previously showed that LIV safely preserves bone and reduces tumor burden in murine ovarian and myeloma cancer models57,58,59. Additionally, LIV inhibits adiposity in mice fed a high-fat diet60,61 and increases the density of muscle-bound satellite cells in ovariectomized mice62. In mesenchymal stem cells, LIV induces osteogenic differentiation while attenuating adipogenesis63,64. We’ve also shown that the timing of LIV dosing affects outcomes, where delivering multiple bouts of LIV separated by a rest period results in beneficial effects in both bone tissue60 and cells65. We recently showed that direct application of LIV to MDA-MB-231 breast cancer cells resulted in decreased invasion and impaired the ability to secrete paracrine factors that promote osteoclast differentiation66. In the clinic, LIV improved bone formation in young women with low bone mineral density67 and in adolescent patients with anorexia nervosa68 and cerebral palsy69, as well as increased t-scores of childhood cancer survivors after 1 year of LIV treatment when compared with placebo-treated patients70.
In the current study, we utilize mouse models to simulate the extreme bone loss, muscle weakness, and increased adiposity experienced by patients undergoing AI treatment to induce complete E2 deprivation. As LIV exhibits protective effects on bone and muscle in preclinical studies of high-fat diet and cancer models and in patients with cancer remission and suppressed E2 production, we hypothesized that daily administration of LIV would preserve critical bone and muscle endpoints in the context of treatment-induced E2 deprivation. LIV protects bone and muscle function from degradation and adverse accumulation of adiposity and short-chain fatty acids in younger mice. Coupling LIV with a standard anti-resorptive agent enhances bone strength more than either treatment alone and partially mitigates muscle fatigue in skeletally mature mice.
Results
Experiment I
The following experiment was designed to determine the effects of LIV in young, skeletally immature E2-deprived mice. Results from this experiment are detailed below.
LIV increased trabecular bone microarchitecture and histomorphometric markers
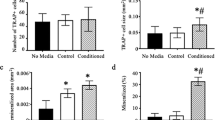
Trabecular bone volume fraction (Tb.BV/TV) and trabecular connectivity density (Tb.Conn.D) in L5 vertebrae of OVX/AI + LIV(y) mice were greater (p = 0.048, p = 0.017, respectively) than in OVX/AI(y) mice (Fig. 1a). (Data from micro-computed tomography (µCT) analyses are presented in their entirety in Supplementary Table S2). The number of osteoclasts (N.OC/BS) and osteoclast surface area (OC.S/BS) in OVX/AI + LIV(y) L5 vertebrae were lower (p = 0.02, p = 0.002, respectively) than in OVX/AI mice (Fig. 1b). The number of osteoblasts (OB/BS) in OVX/AI + LIV(y) mice was greater (p = 0.01) than in OVX/AI(y) mice (Fig. 1b). Dynamic histomorphometric analysis of vertebral trabecular bone indicated that bone formation rate (BFR/BS) was greater (p = 0.01) in OVX/AI + LIV(y) mice relative to OVX/AI(y) mice (Fig. 1c). Dynamic histomorphometry of femoral trabecular bone showed an increase (p = 0.001) in bone formation rate (BFR/BS) and in mineral apposition rate (MAR) (p = 0.003) of OVX/AI + LIV(y) mice relative to that of OVX/AI(y) mice (Fig. 1c). No differences were observed in N.OC/BS or OC.S/BS across the femoral metaphysis.
Results from experiment I (the younger mouse cohort, n = 10) are shown for differences in a trabecular bone microarchitecture (Tb.BV/TV: p = 0.048; Tb.Conn.D: p = 0.017; scale bar = 100 µm) and b static (N.OC/BS: p = 0.018; OC/BS: p = 0.014; OB/BS: p = 0.004; scale bar = 100 µm) and c dynamic histomorphometry (Tb.BFR/BS: p = 0.048; Tb.MAR: p = 0.016; scale bar = 100 µm). Significant decreases in N.OC/BS and OC.S/BS were observed in OVX/AI + LIV(y) mice as confirmed by tartrate-resistant acid phosphatase–stained sections exhibiting a reduced number of positively-stained OC. Histomorphometry of OVX/AI + LIV(y) L5 vertebral body showed significant increases in trabecular BFR/BS and OB/BS, as indicated in the images (magnification 10×) by the greater fluorescent double-labeled surface area, as compared to OVX/AI(y). Differences between OVX/AI(y) and OVX/AI + LIV(y) are shown in terms of d longitudinal muscle function (i.e., forelimb grip strength measured weekly: p = 0.044) and e % whole-body lean mass (as quantified by dual-energy X-ray absorptiometry (DXA) (p = 0.005) to accompany analysis of f myofiber cross-sectional area (p = 0.034; scale bar = 100 µm) and number. g Whole-body weight was longitudinally tracked (weekly) with the rate of increase performed from week 14 until euthanasia. Adiposity was longitudinally quantified by h whole-body fat mass (as quantified by DXA) and i perigonadal fat pad weight (p = 0.022). j Regression analyses were performed to determine the correlation of % fat mass to serum leptin (R2 = 0.827: p = 0.001; graphical color key: datapoints = blue). k Osmium tetroxide staining was used to quantify tibial marrow adiposity from a single hindlimb; scale bar = 1000 µm. Metabolism was quantified by l GTT. AUC area under curve, BFR/BS bone formation rate, BV/TV bone volume fraction, Conn.D connectivity density, CSA cross-sectional area, GTT glucose tolerance test, MAR mineral apposition rate, OVX ovariectomy, OB/BS number of osteoblasts, N.OC/BS number of osteoclasts, OC/BS osteoclast surface, AI aromatase inhibitor, LIV low-intensity vibration, [OVX/AI(y)]. *p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001; ****p ≤ 0.0001, as measured by Student’s two-tailed t-test for (a–c, f, i, and k); repeated measures ANOVA for (d, e, g, h); total fat mass and leptin were correlated by Pearson correlations using regression analysis to generate R2 for (j); area under the curve for (l). All data are presented as mean values ± SEM. All values are derived from biological replicates (Graphical color key: OVX/AI = black; OVX/AI + LIV = green).
LIV increased muscle strength and function following E2 deprivation
From 8 weeks of age (after OVX) until euthanasia (32-week-old), longitudinal change in forelimb grip strength measurements relative to baseline readings were significantly greater (p = 0.05) in OVX/AI + LIV(y) mice than in OVX/AI(y) mice (Fig. 1d). By 26 weeks after OVX, OVX/AI + LIV(y) mice exhibited greater (p = 0.03) grip strength than OVX/AI(y) mice (Fig. 1d). Throughout the study, significant effects were observed as a result of treatment with LIV (p = 0.05), time (p = 0.0001), and between subjects (p = 0.0001), and an interaction effect (p = 0.0001) was also observed (Fig.1d). Longitudinal DXA scans showed that OVX/AI + LIV(y) mice had greater (p = 0.03) lean mass relative to OVX/AI(y) mice by week 28 (Fig. 1e). Muscle from LIV-treated mice had increased myofiber size with decreased spacing between individual fibers (Fig. 1f). Histological sections of OVX/AI + LIV(y) quadratus femores showed increased myofiber cross-sectional area (CSA; p = 0.034) by 24 weeks compared with OVX/AI(y) mice (Fig. 1f). No differences in the number of myofibers were observed between the two groups (Fig. 1f). Tetanic stimulation of the EDL at euthanasia exhibited an interaction (p = 0.01), time (p = 0.001), and subject-related (p = 0.001) effect on muscle fatigue between OVX/AI(y) and OVX/AI + LIV(y) mice (Supplementary Fig. 2). Although there were time and subject-related effects, contractility of the EDL did not exhibit any differences in muscle-specific force between OVX/AI(y) and OVX/AI + LIV(y) mice (Supplementary Fig. 2).
LIV suppressed body weight gain, adiposity, and increased glucose metabolism associated with E2 deprivation
Longitudinal measurements of body weight in young mice in experiment I showed no differences between the OVX/AI(y) and OVX/AI + LIV(y) groups (Fig. 1g). However, linear regression analysis from week 14 to week 28 after OVX showed a significant difference between slopes, with OVX/AI + LIV(y) mice gaining weight at half the rate of OVX/AI(y) mice (Fig. 1g). OVX/AI + LIV(y) mice had lower (p = 0.0001) fat mass relative to OVX/AI(y) mice by week 28 (Fig. 1h). The weight of the fat pad harvested from the perigonadal depots at euthanasia was significantly (p = 0.05) lower in OVX/AI + LIV(y) mice than in OVX/AI(y) mice after 28 weeks of treatment with LIV (Fig. 1i). Serum leptin exhibited a decreasing trend in OVX/AI + LIV(y) mice at 24 weeks post-OVX that, when compared with total fat mass at euthanasia, resulted in a significant correlation (R2 = 0.827, p = 0.001; Fig. 1j). Marrow adipose tissue quantified from the tibiae of younger mice was 6.5% lower in OVX/AI + LIV(y) mice than in OVX/AI(y) mice when normalized to medullary cavity volume, but this was not significantly different (Fig. 1k). Blood glucose readings in young mice were recorded using a glucometer at 0, 15, 30, 60, and 120 min from blood draw at the tail vein. OVX/AI + LIV(y) mice exhibited a trend of lower serum blood glucose (p = 0.09) than OVX/AI(y) mice, as quantified by the area under the curve (Fig. 1l).
Analysis of serum fatty acid oxidation intermediates (acylcarnitines) in young mice prior to and 24 weeks after commencing E2 deprivation identified significant increases in short-chain and medium-chain acylcarnitines. Specifically, 24 weeks of E2 deprivation induced 61% greater (p = 0.0002) circulating short-chain acylcarnitine (2–7 carbon chains; C2–C7) concentrations in OVX/AI(y) compared to baseline (Supplementary Fig. 3c), which increased by just 26% (p = 0.025) from baseline mice when compared to OVX/AI + LIV(y). Among the C2–C7 acylcarnitine groups, acetylcarnitine (C2), propionylcarnitine (C3), butyrylcarnitine/isobutyrylcarnitine (C4/Ci4), and hexanoylcarnitine (C6) contributed to the increased concentration in OVX/AI observed at 24 weeks post-E2 deprivation relative to baseline in experiment I (Supplementary Fig. 3c). C2 species in BL mice significantly (p = 0.0005) increased in OVX/AI(y) but did not differ as compared to OVX/AI + LIV(y), while OVX/AI(y) decreased by 24% (p = 0.0154) in OVX/AI + LIV(y) (Supplementary Fig. 3c). C3 species significantly (p = 0.0013) increased in OVX/AI + LIV(y) mice relative to baseline mice but did not differ compared to OVX/AI(y) mice (Supplementary Fig. 3c). Both OVX/AI(y) and OVX/AI + LIV(y) mice increased (p = 0.00005, p = 0.000067, respectively) C4/Ci4 species as compared to baseline mice (Supplementary Fig. 3c). C6 species exhibited 9x (p = 0.00004) and 5.5x increases (p = 0.00117) in OVX/AI(y) and OVX/AI + LIV(y), respectively, as compared to baseline mice (Supplementary Fig. 3c). by Medium-chain (C8–C14) acylcarnitines significantly increased (p = 0.006) after 28 weeks of E2 deprivation relative to baseline mice, but these levels did not decrease in OVX/AI + LIV(y) relative to baseline nor OVX/AI(y) mice (Supplementary Fig. 3d). Relative to baseline mice, C14:2 (tetradecadienoylcarnitine) significantly increased in OVX/AI(y) and OVX/AI + LIV(y) (p = 0.017, p = 0.002, respectively), while no differences were observed between either group at 28w. No differences in long-chain (C16–C22) species were observed (Supplementary Fig. 3e).
E2 deprivation confirmed from uterine weights
Complete E2 deprivation was performed by ovariectomy surgery and maintained by an aromatase inhibitor. At euthanasia, uterine weights from either group, both E2-deprived, were not statistically different and were comparable to our reported prior data71 (Supplementary Fig. 1).
Experiment II
This experiment was designed to determine the effects of LIV in combination with zoledronic acid in skeletally mature E2-deprived mice. Results from this experiment are detailed below.
Bone mineral density and cortical and trabecular microarchitecture are enhanced when LIV is administered in combination with ZA
Data from µCT analyses are presented in their entirety in Supplementary Table S2. DXA-generated whole-body BMD in the older mice subject to E2 deprivation (OVX/AI) was decreased (p < 0.00001) compared to SH mice (Fig.2a). OVX/AI + LIV mice exhibited no differences relative to OVX/AI mice (Fig. 2a). OVX/AI + ZA mice exhibited increased whole-body BMD (p < 0.00001) relative to OVX/AI mice but lower whole-body BMD (p = 0.0029) than SH mice (Fig. 2a). However, whole-body BMD was increased (p < 0.00001) in OVX/AI + LIV + ZA mice compared with OVX/AI mice, an increase when compared with SH mice (Fig. 2a). BMD analysis of the lumbar spine in the skeletally mature mice using DXA exhibited time, group, and interaction effects (p = 0.0001; Fig. 2b). OVX/AI and OVX/AI + LIV mice exhibited a reduction (p = 0.0001) in lumbar BMD relative to SH mice as early as 4 weeks post-OVX and this effect continued throughout the study (Fig. 2b). However, at 4 weeks post-OVX, lumbar BMD in OVX/AI + ZA mice were lower (p = 0.01) than in SH mice (Fig. 2b). Lumbar BMD in OVX/AI + ZA mice was greater compared to OVX/AI and OVX/AI + LIV mice (Fig. 2b). Additionally, OVX/AI + LIV + ZA mice exhibited no differences in lumbar BMD 4 weeks post-OVX relative to SH mice, which was greater (p = 0.0001) than the difference observed between OVX/AI and OVX/AI + LIV mice (Fig. 2b). By 15 weeks after OVX, BMD of the lumbar vertebrae in OVX-AI + ZA mice were higher (p = 0.000039) than in OVX/AI mice but only mildly greater (p = 0.01) than in OVX/AI + LIV mice (Fig. 2b). However, lumbar BMD of OVX/AI + LIV + ZA mice were greater (p < 0.00001) than in OVX/AI mice at 15 weeks post-OVX (Fig. 2b).
Longitudinal dual-energy X-ray absorptiometry (DXA) scans were performed to quantify a whole-body (SH vs. OVX/AI, OVX/AI + LIV: p < 0.000001; SH vs. OVX/AI + ZA: p = 0.0029; OVX/AI vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p < 0.000001; OVX/AI + LIV vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p < 0.000001) and b lumbar spine BMD (SH vs. OVX/AI, OVX/AI + LIV: p < 0.000001; SH vs. OVX/AI + ZA: p = 0.00013; OVX/AI vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p = 0.000039, p < 0.000001; OVX/AI + LIV vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p = 0.0013, p < 0.000001) for regional measurements. c Longitudinal µCT scans of L5 vertebral bodies were used to quantify Tb.BV/TV (SH vs. OVX/AI, OVX/AI + LIV: p = 0.0034, p = 0.0039; SH vs. OVX/AI + LIV + ZA: p = 0.039; OVX/AI vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p = 0.00009, p < 0.000001; OVX/AI + LIV vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p = 0.0001, p < 0.000001; scale bar = 1000 µm). d Dynamic histomorphometry was performed to quantify differences in double-labeled surface dLS (SH vs. OVX/AI + LIV, OVX/AI + ZA: p = 0.03, p = 0.0018), BFR/BS (SH vs. OVX/AI + ZA: p = 0.0062), MS/BS (SH vs. OVX/AI + ZA: p = 0.002), Ir.L.Wi, and MAR. Static histomorphometry was performed to quantify OB/BS (SH vs. OVX/AI + ZA: p = 0.008; OVX/AI + LIV vs. OVX/AI + ZA: p = 0.027), N.OC/BS, and OC.S/BS. e Ultimate force derived from compression testing of the L5 vertebrae (SH vs. OVX/AI, OVX-AI + LIV: p = 0.0022, p = 0.011; OVX/AI vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p = 0.0001, p < 0.00001; OVX/AI + LIV vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p = 0.0009, p = 0.00007). f 3-point bending of the femora to measure yield force (SH vs. OVX/AI: p = 0.038; OVX/AI vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p = 0.043, p = 0.0048), resilience (SH vs. OVX/AI + LIV + ZA: p = 0.0017; OVX/AI vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p = 0.0068, p = 0.0001), ultimate force (SH vs. OVX/AI, OVX/AI + LIV: p = 0.0014, p = 0.026; OVX/AI vs. OVX/AI + ZA, OVX/AI + LIV + ZA: p = 0.0005; OVX/AI + LIV vs. OVX/AI + LIV + ZA (p = 0.012), and toughness (OVX/AI vs. OVX/AI + LIV + ZA: p = 0.036). BMD bone mineral density, OVX ovariectomy, AI aromatase inhibitor, LIV low-intensity vibration, SH sham-OVX and saline vehicle (control), ZA zoledronic acid, w weeks after OVX, Tb trabecular, BFR/BS bone formation rate, OB/BS number of osteoblasts, N.OC/BS number of osteoclasts, OC.S/BS osteoclast surface, dLS double-label fluorescent surface area, MS/BS mineralizing surface area, Ir.L.Wi Interlabel Width, MAR Mineral Apposition Rate, Tb. trabecular, BV/TV bone volume fraction. *p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001; ****p ≤ 0.0001; One-way ANOVA, Tukey’s post hoc was used for bone histomorphometry and mechanical testing data; repeated measures ANOVA, Tukey’s post hoc was used for longitudinal DXA for BMD and µCT for vertebral Tb.BV/TV. All data are presented as mean values ± SEM. (Graphical color key: SH = black; OVX/AI = yellow; OVX/AI + LIV = green; OVX/AI + ZA = blue; OVX/AI + LIV + ZA = pink).
L5 vertebrae in OVX/AI and OVX/AI + LIV mice exhibited a significant (p = 0.0034, p = 0.0039, respectively) decrease in Tb.BV/TV compared with those of SH mice by 15 weeks post-OVX (Fig. 2c). A trend of increased BV/TV was observed in OVX/AI + LIV + ZA mice relative to SH. In OVX/AI + ZA and OVX/AI + LIV + ZA mice, Tb.BV/TV increased (p = 0.0001, p < 0.000001, respectively) compared with untreated OVX/AI mice (Fig. 2c). Interaction (p = 0.0001) and time (p = 0.01) effects were found between groups throughout the study as a function of percentage change in L5 Tb.BV/TV (Fig. 2c). Untreated OVX/AI mice had decreased Tb.BV/TV (p = 0.0001) relative to SH mice (Fig. 2c). OVX/AI + ZA mice increased (p = 0.0001) in Tb.BV/TV relative to OVX/AI mice, and in OVX/AI + LIV mice, Tb.BV/TV was greater (p = 0.0001) than in untreated OVX/AI mice; a similar effect was also observed (p = 0.01) in OVX/AI + LIV + ZA mice (p < 0.000001) (Fig. 2c). These findings were paralleled by similar differences in Tb.N and Tb.Th, each showing decreases (p = 0.05, p = 0.01, respectively) in OVX/AI mice relative to SH mice and increases (p = 0.05, p = 0.001, respectively) in mice treated with ZA (Supplementary Table S2). OVX/AI + LIV + ZA mice exhibited increased Tb.N and Tb.Th (p = 0.001, p = 0.0001, respectively) compared with OVX/AI mice, as well as increases relative to OVX/AI + ZA mice (p = 0.05, p = 0.05, respectively; Supplementary Table S2).
Femoral Ct.BA/TA exhibited significant interaction, time, and treatment (ZA and LIV)-related effects (p = 0.0001) in older mice (Supplementary Table S2). Ct.BA/TA decreased (p = 0.0001) in OVX/AI mice compared to SH mice by 15 weeks after OVX (Supplementary Table S2). Although OVX/AI + LIV mice did not show improved Ct.BA/TA, Ct.BA/TA was increased (p = 0.001) in OVX/AI + ZA mice (Supplementary Table S2). OVX/AI + ZA mice had increased (p = 0.0001) Ct.BA/TA, and OVX/AI + LIV mice had increased (p = 0.0001) Ct.BA/TA as well (Supplementary Table S2). Ct.BA/TA in OVX/AI + LIV + ZA mice increased (p = 0.0001) compared with that of OVX/AI mice, thereby improving on treatment with ZA alone (Supplementary Table S2). Corresponding decreases in Ct.Th (p = 0.0001) and Ct.pMOI (p = 0.05) were observed in OVX/AI mice compared with SH mice (Supplementary Table S2). OVX/AI + ZA mice showed increased (p = 0.001) Ct.Th relative to OVX/AI mice, and Ct.Th increased further (p = 0.0001) in OVX/AI + LIV + ZA mice (Supplementary Table S2). Ct.pMOI was not statistically different in OVX/AI + ZA or OVX/AI + LIV + ZA mice relative to OVX/AI or SH mice (Supplementary Table S2).
Decreased bone formation and mineralization in E2-deprived mice due to ZA is attenuated in LIV-treated mice
dLS was reduced in OVX/AI + LIV and OVX/AI + ZA mice (p = 0.03, p = 0.0018, respectively) compared to SH mice (Fig. 2d); however, dLS of OVX/AI + LIV + ZA mice, although not statistically different from SH mice, was 2 times greater than that observed in OVX/AI + ZA mice (Fig. 2d). BFR/BS was lower (p = 0.0062) in the L5 of OVX/AI + ZA mice than in SH mice, while OVX/AI + LIV + ZA mice exhibited trends of lower BFR/BS than SH mice. Mineralizing surface area (MS/BS) was significantly reduced (p = 0.002) in OVX/AI + ZA mice relative to SH mice, while OVX/AI + LIV + ZA mice exhibited trends of lower MS/BS compared to SH mice (Fig. 2d). OB/BS was significantly decreased (p = 0.008) in OVX/AI + ZA mice compared with SH mice and was also lower (p = 0.027) compared to OVX/AI + LIV mice (Fig. 2d). No differences were observed in Ir.L.Wi., OC.S/BS, or N.OC/BS (Fig. 2d). Sections stained with tartrate-resistant acid phosphatase exhibited no differences in the number of osteoclasts or osteoclast surface area between groups; however, there were fewer osteoclasts in OVX/AI + LIV mice at a similar proportion to that observed with OVX/AI + ZA mice relative to untreated OVX/AI mice.
The mechanical properties of vertebral and diaphyseal bone degraded by E2 deprivation are normalized with ZA but improved further when combined with LIV
Mechanical testing of bone data are presented in their entirety in Supplementary Table S3. Compression testing of L5 vertebrae of OVX/AI and OVX/AI + LIV mice exhibited decreased (p = 0.0022, p = 0.011, respectively) ultimate force compared to SH mice (Fig. 2e). Ultimate force increased in OVX/AI + ZA mice L5 vertebrae (p = 0.0001) compared to untreated OVX/AI mice (Fig. 2e). Ultimate force of L5 in OVX/AI + LIV + ZA mice improved (p < 0.00001) relative to OVX/AI mice, a 43% increase compared with OVX/AI + ZA (Fig. 2e). In comparison to OVX/AI + LIV mice, OVX/AI + ZA and OVX/AI + LIV + ZA mice showed greater ultimate force (p = 0.0009, p = 0.00007, respectively).
As measured by 3-point bending, femora of OVX/AI mice exhibited a reduction (p = 0.038) in yield force compared to SH mice, but which was increased (p = 0.001) in OVX/AI + ZA and OVX/AI + LIV + ZA mice relative to OVX/AI mice (p = 0.043, p = 0.0038, respectively) (Fig. 2f). Yield force in OVX/AI + LIV + ZA mice improved (p = 0.012) relative to OVX/AI mice (Fig. 2f). Similarly, stiffness was also reduced (p = 0.002) in OVX/AI mice relative to SH mice, but no significant differences were observed in OVX/AI + LIV, OVX/AI + ZA, or OVX/AI + LIV + ZA mice relative to either SH or OVX/AI mice (Fig. 2f). Tissue-level properties indicated similar decreases in yield stress, ultimate stress, resilience, and toughness in OVX/AI mice compared with SH mice (Fig. 2f). Improvements (p = 0.007) were observed in OVX/AI + ZA mice compared with OVX/AI mice, while nonsignificant increases were observed in resilience (47%), toughness (38%), and yield stress (18%) in OVX/AI + LIV mice relative to untreated OVX/AI mice (Fig. 2e and Supplementary Table S2). OVX/AI + LIV + ZA mice exhibited increased resilience (p = 0.0001), toughness (p = 0.036), yield stress (p = 0.04), and ultimate stress (p = 0.02) compared with OVX/AI mice (Fig. 2e and Supplementary Table S2).
Muscle function improved following either LIV or ZA treatment
Peak percentage change in grip strength was observed 4 weeks following sham-OVX in the SH mice (Fig. 3a). Although all groups exhibited increased grip strength throughout the initial 2 weeks after surgery (OVX), OVX/AI + LIV and OVX/AI + LIV + ZA mice exhibited the sharpest increase among the groups, peaking at an average 125 g of force (Fig. 3a), but these data were not significant. At the initial 2 weeks post-surgery, OVX/AI + LIV mice had greater grip strength (p = 0.05) than OVX/AI mice, and OVX/AI + LIV + ZA mice trended toward increased grip strength (p = 0.06) relative to OVX/AI mice (Fig. 3a). At 5 weeks post-OVX, grip strength began to decline in all groups (Fig. 3a). By week 9, OVX/AI + LIV mice exhibited greater grip strength (p = 0.05) than did OVX/AI mice (Fig. 3a). This margin increased (p = 0.007) by week 11, at which point OVX/AI + LIV + ZA mice also exhibited increased grip strength (p = 0.03) relative to OVX/AI mice (Fig. 3a). Grip strength of the OVX/AI + LIV mice remained consistently higher than all groups throughout the course of the study following E2 deprivation (Fig. 3a). Throughout the post-OVX period, an effect (p = 0.01) was observed between groups (Fig. 3a).
a As a measure of muscle function, forelimb grip strength was quantified weekly. Ex vivo muscle contractility force testing was performed at euthanasia on extensor digitorum longus muscles resected from the hindlimbs. Through tetanic stimulation, b fatigue (SH vs. OVX/AI + LIV: p = 0.0071; SH vs. OVX/AI + ZA: p = 0.00004; SH vs. OVX/AI + LIV + ZA: p = 0.0032; OVX/AI vs. OVX/AI + ZA: p = 0.12) and c maximum muscle-specific forces were determined. d Histologic sections (magnification 10×; hematoxylin and eosin; scale bar = 100 µm) of quadratus femorum were analyzed to quantify average myofiber CSA and number of myofibers. No significant differences between groups were observed in e CSA or f number of myofibrils. CSA cross-sectional area, EDL extensor digitorum longus, OVX ovariectomy, AI aromatase inhibitor, LIV low-intensity vibration, SH sham-OVX and saline vehicle (control), ZA zoledronic acid. *p ≤ 0.05; **p ≤ 0.01; ****p ≤ 0.0001; One-way ANOVA, Tukey’s post hoc was used for muscle CSA and myofibril number. Repeated measures ANOVA, Tukey’s post hoc was used for longitudinal grip strength data and ex vivo muscle fatigue and contractility readings. All data are presented as mean values ± SEM. (Graphical color key: SH = black; OVX/AI = yellow; OVX/AI + LIV = green; OVX/AI + ZA = blue; OVX/AI + LIV + ZA = pink).
Percentage decrease from baseline to the first tetanus (30 Hz stimulation) during ex vivo muscle contractility of the EDL yielded significant improvement in fatigue in OVX/AI + LIV mice (p = 0.028) and OVX/AI + LIV + ZA mice (p = 0.041) relative to SH mice (Fig. 3b). At the next frequency (40 Hz) in the stimulation protocol, OVX/AI + LIV and OVX/AI + LIV + ZA mice exhibited less muscle fatigue (p = 0.0065, p = 0.0017, respectively) than SH mice (Fig. 3b). Additionally, OVX/AI + ZA mice had reduced muscle fatigue (p = 0.045) in contrast to SH mice; however, none of the groups differed from OVX/AI mice (Fig. 3b). At 50 Hz stimulation, treatment with LIV and LIV + ZA in OVX/AI mice both led to reduced muscle fatigue (p = 0.0071, p = 0.0032, respectively) than that seen in SH mice, whereas OVX/AI + ZA mice had 10% more muscle fatigue (p = 0.00004) than that observed in SH mice (Fig. 3b). At this stimulation frequency, OVX/AI + ZA mice had less muscle fatigue (p = 0.012) than did untreated OVX/AI mice (Fig. 3b). No differences were observed in muscle-specific force at any of the frequencies used to stimulate the muscle (Fig. 3c). Untreated OVX/AI mice showed lower myofiber CSA compared with SH mice (Fig. 3d); however, no significant differences in myofiber CSA or number of myofibers were observed in the skeletally mature mice (Fig. 3e, f, respectively).
E2 deprivation increased perigonadal and bone marrow adipose accrual, whereas either LIV or ZA alone reduced body weight and decreased adiposity
DXA analysis indicated significant increases in % fat mass of OVX/AI mice (p = 0.01) and OVX/AI + ZA mice (p = 0.02) relative to SH mice at 9 weeks post-OVX (Fig. 4a). By 14 weeks post-OVX, all E2-deprived mice had greater (p = 0.0001) percent fat mass relative to SH mice (Fig. 4a). Neither LIV treatment nor ZA alone or in combination significantly altered percent fat mass compared to E2-deprived mice (Fig. 4a). No differences were observed in percent lean mass between groups. A significant effect (p = 0.0001) was found in all groups with respect to time over the course of the study (Fig. 4a). Throughout experiment II (older cohort), body weight increased the least in SH mice, and OVX/AI mice had significantly greater (p = 0.01) weight than SH mice (Fig. 4b). OVX/AI + LIV and OVX/AI + LIV + ZA mice exhibited greater (p = 0.0001) body weight than SH mice by 20 weeks post-OVX (Fig. 4b). Body weight of OVX/AI + ZA mice was lower (p = 0.05) than that of OVX/AI + LIV + ZA mice by the same endpoint (Fig. 4b). Taken together, these results indicated that young mice treated with LIV had a significantly slower rate of weight and fat gain compared with their untreated counterparts, but older E2-deprived mice gained significantly more weight than did their E2-replete, SH counterparts.
a Total % fat mass measured by dual-energy X-ray absorptiometry (DXA) was significantly increased in all E2-deprived mice when compared to SH mice at 14w (p = 0.0001). b Body weight of E2-deprived mice quantified throughout the study relative to SH mice (SH vs. OVX/AI: p = 0.001; SH vs. OVX/AI + LIV, OVX/AI + ZA: p = 0.00008, p = 0.000001; OVX/AI + ZA vs. OVX/AI + LIV + ZA: p = 0.027). c Gonadal fat pad (bilateral) weights were collected at euthanasia and weighed together (SH vs. OVX/AI, OVX/AI + LIV + ZA: p = 0.021, p = 0.017. Generally, E2-deprived mice treated with just LIV had lower fad pad mass than untreated OVX/AI mice. d Osmium tetroxide staining using µCT showed representative MAT across the length of the tibial marrow cavity (SH vs. OVX/AI, OVX/AI + LIV, OVX/AI + ZA, OVX/AI + LIV + ZA: p = 0.000001; scale bar = 1000 µm). e Histological quantification of Ad.Ar./Ma.Ar. (SH vs. OVX/AI: p = 0.0051) and f N.Ad./Ma.Ar. in untreated OVX/AI mice compared with SH mice (SH vs. OVX/AI: p = 0.0312). OVX ovariectomy, AI aromatase inhibitor, LIV low-intensity vibration, SH sham-OVX and saline vehicle (control), ZA zoledronic acid, Ad.Ar./Ma.Ar. total adipocyte volume encased within a given marrow volume, N.Ad./Ma.Ar. total number of adipocytes within a given marrow volume, MAT marrow adipose tissue, Me.Cavity medullary cavity volume. *p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001; ****p ≤ 0.0001; One-way ANOVA, Tukey’s post hoc was employed to compare gonadal fat pad weights and marrow adiposity; Repeated measures ANOVA, Tukey’s post hoc was employed to quantify longitudinal % change of fat mass as measured by DXA. All data are presented as mean values ± SEM. (Graphical color key: SH = black; OVX/AI = yellow; OVX/AI + LIV = green; OVX/AI + ZA = blue; OVX/AI + LIV + ZA = pink).
At euthanasia, both OVX/AI and OVX/AI + LIV + ZA mice had significantly more gonadal fat pad tissue (p = 0.02) than did SH mice (Fig. 4e). OVX/AI + LIV mice had more gonadal fat pad tissue than SH mice, but fat pad tissue did not differ between OVX/AI-LIV mice and untreated OVX/AI mice (Fig. 4c). None of the treatment groups in experiment II significantly differed from OVX/AI mice in terms of gonadal fat pad tissue weight (Fig. 4c).
Marrow adipose tissue normalized to medullary cavity volume, as quantified by µCT, was greater (p = 0.0001) in OVX/AI mice than in SH mice (Fig. 4d). OVX/AI + LIV mouse tibiae were not significantly different from those of OVX/AI mice, nor were those of OVX/AI + ZA mice (Fig. 4d). Marrow adipose tissue decreased in OVX/AI + LIV + ZA mice relative to OVX/AI mice, by 36% compared with SH mice, but this was not significant (Fig. 4d).
Ad.Ar./Ma.Ar. in the tibial metaphysis was increased (p = 0.01) in OVX/AI mice relative to SH mice, and these levels were decreased by 15% in OVX/AI + LIV mice (Fig. 4e). Ad.Ar./Ma.Ar. was decreased in OVX/AI + ZA mice by 10% relative to OVX/AI mice, and a nonsignificant reduction was also observed in OVX/AI + LIV + ZA mice (p = 0.08; Fig. 4e). N.Ad./Ma.Ar. was increased (p = 0.04) in OVX/AI mice relative to SH mice (Fig. 4f). Treatment with LIV resulted in a 33% reduction compared with OVX/AI mice, while N.Ad./Ma.Ar. in OVX/AI + ZA mice decreased by 26% (Fig. 4f). However, a 62% reduction was observed in OVX/AI + LIV + ZA mice as compared to OVX/AI mice (Fig. 4f).
E2 deprivation confirmed from uterine weights
Complete E2 deprivation was performed by ovariectomy surgery. Uterine weights measured at euthanasia in all OVX/AI experimental groups weighed significantly (p ≤ 0.0001) less than SH mice (Supplementary Fig. 1) and were not statistically different from uterine weights from Experiment I.
Discussion
Estrogens serve a central regulatory role in bone remodeling in order to maintain bone health. Aging coupled with menopause contributes to a natural decline in E2 synthesis, causing dysregulation in bone remodeling while leading to increased fat and an accompanying decline in muscle strength. Cancer and its many treatment strategies either directly or secondarily disrupt E2 homeostasis, and, when combined with the effects of aging and menopause, offset bone remodeling to favor bone loss while also upregulating the production of fat, which in turn heightens inflammation and crowds out bone remodeling cells in the marrow. Our group has shown that patients with breast cancer bone metastases have both bone loss and muscle weakness attributed to the release of sequestered TGF-β from the bone matrix27. Therefore, limiting bone loss is critical to the stabilization of disease progression and improving patient outcomes. Our overarching objectives in this study were to quantify the effects of applying a low-magnitude, high-frequency vibratory signal to the whole body as a countermeasure to the negative effects of complete E2 deprivation therapy and to determine whether, when coupled with a bisphosphonate (e.g., ZA), LIV could complement the current standard-of-care for patients diagnosed with breast cancer bone metastases.
E2-receptor-positive breast cancers progress through a feed-forward pathway whereby estrogens synthesized from the follicle-stimulating cells of the ovary bind to receptors on tumorigenic cells, driving division. Transformed tumor cells enter the circulation and then have the propensity to seed metastases in the brain, liver, kidney, and bone, inducing osteolytic resorption pits at the endosteal surface in bones. This mechanism is analogous to the androgen-driven effects on tumor progression in prostate cancers, where bone metastases predominantly manifest as sclerotic lesions. Secondary E2 sources derive from androgens synthesized by adrenal glands and converted into β-estradiol through aromatase secreted by adipocytes housed in the viscera and the liver. Thus, the use of AI therapy in postmenopausal patients induces complete E2 deprivation, significantly decreasing patient mortality by eliminating E2, a major accelerator of E2-receptor-positive breast cancer progression. Despite improvements in tumor-associated morbidity, E2 deprivation exacerbates the deleterious effects of aging and menopause on bone, muscle, and body composition in this patient population. Increased bone fragility is observed in breast cancer patients in remission undergoing AI therapy, raising patient susceptibility to developing fractures, with the highest incidences occurring at the femoral neck and across the lumbar spine72,73. Moreover, the efficacy of long-term anti-hormone treatments in preventing cancer from recurring often wanes with time as patients experience increased resistance to treatment74.
Mechanical signals are fundamentally inseparable from bone maintenance, a reliance perhaps most evident in the setting of physiological unloading (e.g., chronic bed rest, disuse), a condition that aggressively upregulates osteoclast-mediated bone resorption. In patients, age-related osteopenia is differentiated from the extensive cortical lesions derived from bone metastases. Clinical treatment to inhibit osteolysis is achieved through bisphosphonate therapy, but rarely do these lesions heal. Simulated unloading in murine models (e.g., hindlimb suspension models) induces bone catabolism75 and muscle atrophy76, but these effects have been reversed through administration of LIV77. The vertebrae have been a target for LIV in ameliorating intervertebral disc degeneration78 in rats with simulated hindlimb unloading. Furthermore, clinical studies have demonstrated that daily administration of LIV can restore the bone mass lost during simulated periods of extended bed rest and improve z-scores for childhood cancer survivors70. Additionally, our previous studies showed that treatment of mice bearing spontaneous granulosa cell ovarian cancer57 and myeloma58 resulted in suppression of bone loss induced by both endogenous and aggressive cancers.
As anticipated, ZA significantly improved trabecular bone mass and mechanical integrity in the trabecular-rich lumbar vertebral body of the L5 vertebrae when challenged by the OVX/AI phenotype. However, the effects of ZA were significantly enhanced to levels of E2-replete mice when the treatment was complemented with LIV, improving trabecular bone across the lumbar spine, whereas treatment with LIV alone did not impart any benefit in retaining cancellous bone of the appendicular skeleton. Improvements in trabecular bone quantity coincided with improved resistance to fracture from compression testing. These effects on bone, however, were not isolated to cancellous bone, because the cortical bone improved in terms of both quantity and quality (i.e., mechanical strength). What is of specific interest is the greater effect observed when the LIV regimen was combined with ZA, resulting in an outcome that was more effective than ZA alone and which persisted throughout the entirety of the study. We speculate that the mechanism is driven by ZA-induced suppression of osteoclast activity.
Recently, a great deal of attention has focused on the crosstalk between bone and muscle, recognizing that dysfunction in one tissue is likely to translate to the other. With regards to the present study, compounding the negative effects of AI and breast cancer metastases in swaying bone remodeling toward heightened focal resorption, patients experience reduced muscle strength79 and elevated body fat79. Heightened bone-derived TGF-β, in addition to its autocrine feedback function in metastatic growth, enters the circulation and perpetuates RyR dysfunction and disrupts Ca2+ signaling, inducing muscle weakness. Our murine model highlights the accelerated and near-systemic loss of bone associated with E2 deprivation. Concomitant loss of lean mass, myofiber number, and myofiber CSA suggests a morphologic relationship between bone and muscle. Histological sections quantified from the quadratus femoris in the younger mouse cohort showed that incorporating daily mechanical loads for an extended period led to muscle fiber hypertrophy in the CSA of hindlimb muscles. These findings may be due to reduced fibrosis or adipocyte deposition, and the increased spacing observed between muscle fibers may stem from reduced oxidative capacity of the muscle fibers or decreased satellite cell maintenance of muscle. Whether the increase in myofiber area is a byproduct of muscle preservation following E2 loss or an anabolic response derived from LIV is not fully understood.
Expanding on the functional outcomes, the EDL fatigability observed in OVX/AI mice was similar to the amount detected in SH mice. The inclusion of LIV, regardless of ZA administration, lessened fatigue in the treated mice relative to the muscle fatigue observed in SH mice. However, treatment with ZA alone significantly reduced fatigue in the EDL relative to the values observed in SH mice, but not relative to mice treated with LIV. The reduced fatigue observed in OVX/AI + LIV + ZA mice provides evidence supporting these results. Despite these findings, fatigue in the EDL was unchanged in younger mice exposed to LIV once per day. Reduced EDL fatigue following treatment with LIV + ZA in the older mice suggests that inhibition of skeletal resorption and subtle exposure to LIV elicited a greater response in older mice than in the younger mice. Perhaps the capacity of ZA to prevent TGF-β release contributed to the protection of Ca2+ signaling in the muscle. Alternatively, the second bout of daily treatment with LIV could have served to bolster the signal that was only mildly effective in the younger cohort (treated only once per day). The increased grip strength observed in the younger mice supports the clinical observation that mechanical loading (e.g., exercise, physical activity) builds stronger bone and muscle if introduced early in skeletal development. Together, these LIV effects on muscle, either alone or in tandem with ZA, appear to exert multiple effects on composition and functionality. The varied efficacy of the treatment may stem from the recruitment of different muscle groups and be a consequence of differing ages. The possible extension of these findings into other muscles, suggested by the increased grip strength observed throughout the study, is of interest.
Bone loss ensuing from E2 deprivation therapy illustrates the reliance of bone homeostasis on E2 availability while also highlighting the reciprocal relationship between bone and fat. The association of obesity with cancer is gaining more traction80; excess fatty tissue is highly correlated with breast cancer incidence81. An adipose-enriched phenotype, as paralleled by a phenotype in individuals with obesity, has been associated with a list of maladies, including a body habitus permissive to breast cancers81,82,83. As a consequence of age and diminished E2 synthesis, postmenopausal women (and aging men) accumulate fat in visceral tissues and marrow cavities. β-oxidation of excess fatty acids (lipolysis), a secondary means of ATP production, disrupts normal glycolysis in the mitochondria of insulin-sensitive tissues, including muscle and liver84. Intermediate metabolites of different carbon chain lengths derived from incomplete transformation are termed acylcarnitines, and their accumulation is associated with insulin resistance85,86. Age-related increases in adiposity are exacerbated by AI therapy, causing adipocyte hypertrophy and infiltration of muscle fibers, displacement of healthy marrow, encasement of critical organs, inflammation, and occlusion of vasculature.
We observed significantly increased gonadal fat deposition in the older mice in response to OVX and daily AI. Additionally, tibial marrow adipose tissue reflected similar increases in adipocyte density and hypertrophy in response to complete E2 deprivation. Treatment with LIV drastically reduced both the total area and number of marrow adipocytes, and, to a lesser degree, we observed the same effect following treatment with ZA. These results would translate to clinical standard-of-care, including anti-resorptive drugs, and if paired with LIV, these drugs could significantly reduce the adiposity (and adverse localized activity) wrought by disease and primary treatments. Therefore, suppressing adiposity in various depots could reduce inflammation and inhibit bone resorption, consistent with upregulation in the lipid profile exhibited by the postmenopausal patient cohort being treated with AIs in a previous study87.
Quantitative serum acylcarnitine analysis identified significant increases in short-chain (C2–C7) acylcarnitine intermediates, specifically acetylcarnitine (C2), hexanoylcarnitine (C6), in the mice in our study, and these findings served as biomarkers of E2 deprivation-related muscle and bone disease, as well as their response and subsequent decrease following treatment with LIV. Acetylcarnitine has been proposed as a biomarker of hepatocellular carcinoma88. Age-dependent increases in acetylcarnitine in mouse and human muscle have been reported89. Significant increases in circulating hexanoylcarnitine were identified in response to a 12-week low-calorie diet to induce weight reduction, along with increases in medium- and long-chain acylcarnitines and free fatty acids90. The specific source of these acylcarnitines appears to be from fat deposits; however, the specificity of acetylcarnitine and hexanoylcarnitine is not known. Our findings support possible benefits from LIV by suppressing systemic fat mass loss and across various deposits. Peroxisome-proliferator alpha receptor-gamma (Pparg), a transcriptional regulator of adipocyte differentiation91, also regulates acylcarnitine metabolism92. Previous evidence suggests that LIV directs MSC differentiation towards Runx2-directed pathways and, while deterring from Pparg61,93 and induces fat reduction in models of metabolic dysfunction94,95. This is consistent with the data observed in our OVX/AI + LIV mice, suggesting a possible mechanistic connection to LIV-mediated reduction in adiposity and serum acylcarnitines. However, no differences in fat pad weights were observed between all E2-deprived mice in the older mouse cohort; therefore, metabolomics was not performed in this cohort, as it did not track with E2 deprivation.
The complex mechanisms responsible for driving LIV effects on adiposity may lie, in part, in the ability of mechanical loading to drive the differentiation of the mesenchymal stem cell towards an osteogenic phenotype, thus deterring adipogenic differentiation96. To what degree this affects the synthesis of the fatty acids of different classes and across critical tissues remains to be addressed in future experiments. However, the reduction in bone marrow adiposity in the long bones of mice undergoing an extreme and acute deficit in E2 without the aid of pharmacologic intervention is a promising finding.
Combining LIV with ZA significantly improved lumbar trabecular bone volume fraction and cortical bone CSA in the femur above either treatment alone, a finding that could be of specific benefit to patients undergoing similar treatments, given that femoral head and vertebral compression fractures are common in patients undergoing anti-E2 therapy. Patients who experience fractures have increased recovery times and morbidity. LIV is advantageous as a potential additional arm of treatment, as it does not interrupt other therapeutic efforts. In addition to improved bone measures, a significant reduction in the fatigability of the EDL via muscle contractility testing was observed in mice jointly treated with LIV and ZA. In short, the benefits to bone were accompanied by improvements in muscle phenotype and strength.
The current study has some limitations deriving from inherent differences in the modeling of E2 deprivation and the route of LIV administration in mice compared with humans. E2 deprivation results in a substantial reduction in trabecular bone across the proximal tibiae and distal femur in mice, with mild traces remaining, necessitating the quantitation of the trabecular-rich vertebral bodies of the lumbar spine. Muscle strength was measured with a grip meter using tail suspension over a grip mesh, a method that recruits different muscle groups than a grip test performed in humans, which is quantified exclusively via forelimb grip. Furthermore, that EDL muscle contractility did not improve following either treatment may suggest muscle fiber type and/or location specificity for treatment-induced effects. Perhaps the most important consideration in the study design is the degree of daily handling stress endured by the mice. Despite this, all mice experienced similar handling and subsequently similar downstream stress effects between groups. Additionally, the mechanical signal used in these studies was administered with a singularly defined set of parameters. Thus, by varying spatiotemporal parameters, differing effects on the target tissues may be observed. Extending or limiting treatment duration, modulating the waveform frequency or magnitude, or changing the number of treatments could improve outcomes even further. Lastly, the dose of letrozole was determined to be adequate to limit E2 synthesis, but these effects, initially observed in younger mice, may not translate completely to skeletally mature and/or aging mice further burdened by accumulating visceral adiposity and waning resident marrow-bound progenitors.
Our study demonstrates that LIV can protect and/or increase lean mass, reduce fat mass, and improve bone endpoints as otherwise compromised by complete E2 deprivation, and when LIV is administered twice daily in combination with zoledronic acid, these effects are additive in preserving vertebral trabecular bone. Combining LIV with ZA is unique in the benefit imparted on multiple systems at once. Inhibition of bone resorption with ZA combined with increased bone formation following LIV may aid in patient recovery after treatment or be considered as a pre-surgical strategy to improve patient outcomes for those undergoing anti-hormone treatment.
In conclusion, our study quantified the negative effects of E2 deprivation, a therapeutic modality employed to stop the progression of breast cancer in E2-receptor-positive patients, on bone and muscle, while demonstrating the potential of utilizing mechanical signals in the form of LIV and the anti-resorptive drug ZA to inhibit the degree of musculoskeletal deterioration (Supplementary Fig. 3). These findings highlight the combined deleterious impact of diminished E2 synthesis and anti-E2 treatments, modeled via OVX in AI-treated mice, in decreasing bone mass and muscle strength and increasing fat due to the absence of circulating E2. Although ZA inhibited bone loss, our findings demonstrate the added positive effects of combining low-magnitude mechanical signals with ZA on enhancing preservation and/or increasing lean mass while reducing fat mass, and most impressively, improving trabecular and cortical bone endpoints. Further, we identified 4 unique biomarkers (acetylcarnitine, O-butanoylcarnitine, hexanoylcarnitine, and tetradecadienoylcarnitine) that are increased with E2 deprivation-associated musculoskeletal defects. In addition, acetylcarnitine, O-butanoylcarnitine, and hexanoylcarnitine decreased with a successful clinical intervention (e.g., LIV). Under these conditions, our data highlight the benefits of exercise, but more specifically, mechanical loads induced by LIV, in protecting muscle and skeletal tissue, and provide the basis to direct future clinical therapies towards combinatorial strategies that incorporate both pharmacologic and non-pharmacologic treatments.
Methods
All experimental procedures were approved by the Institutional Animal Care and Use Committee guidelines at Indiana University and complied with all relevant ethical regulations in strict accordance with the guidelines of the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals and the ARRIVE guidelines.
Study design
The overarching objective of this study was intended to assess the suitability and impact of low-magnitude mechanical signals, administered using low-intensity vibrations (LIV), either alone or in combination with an anti-resorptive agent, on musculoskeletal parameters and systemic or regional adiposity in a murine model of complete E2 deprivation. Our initial aim was to contrast the effects of daily exposure to LIV on mice experiencing aggressive bone and muscle degeneration and accretion of adiposity in skeletally immature mice while addressing effects on the quantity and functionality of these tissues. Following this aim, complete E2 deprivation was modeled in skeletally mature mice. All C57BL/6 mice underwent complete E2 deprivation through surgical ovariectomy and were maintained through daily letrozole injection to ensure inhibition of both ovarian and adrenal androgen sources, as this model had been previously validated by our group and translates clinically. µCT analysis and 3-point and compressive mechanical testing of femora and vertebrae were performed to quantify bone microarchitecture and resistance to fracture as a measure of bone quantity and quality. Body composition was measured from DXA scans. Histological analyses were performed to visualize and measure bone histomorphometry and muscle morphology.
Animal model
C57BL/6 mice (Envigo, Indianapolis, IN, USA) were monitored daily and maintained in accordance with the Institutional Animal Care and Use Committee guidelines at Indiana University and the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals. Sample sizes were determined based on prior studies and power analyses (80% power, α = 0.05), an n = 10 was calculated for the preliminary experiment (I) a total of 20 mice per group was allocated to account for various downstream assays. Mice were housed five per cage at standard room conditions of 22 °C and 40–60% humidity and allowed ad libitum access to standard diet-chow (catalog # 2018SX, Envigo) and water. All mice were monitored daily for signs of ill health, and humane endpoints were applied in accordance with institutional guidelines. A period of 1 week was allotted at the beginning of the study to acclimate the mice to the handling procedures and LIV and mock-LIV treatment protocol. Group designations are detailed in Supplementary Table S1. For experiment I, 4-week-old female C57BL/6 mice were divided into two groups (n = 10/group): a. ovariectomized (OVX) and treated with AI only [OVX/AI(y)] and b. OVX and treated with AI and LIV [OVX/AI + LIV(y)]. For experiment II, 15-week-old female C57BL/6 mice were divided into five groups (n = 20/group): a. sham-ovariectomized age-matched controls given saline vehicle (SH); b. OVX and treated with AI only (OVX/AI); c. OVX and treated with AI and LIV (OVX/AI + LIV); d. OVX and treated with AI and ZA (OVX/AI + ZA); and e. OVX and treated with AI, LIV, and ZA (OVX/AI + LIV + ZA).
Complete E2 deprivation
Sham-surgery and vehicle (phosphate-buffered saline) injection were performed on SH mice, and all other treatment groups underwent OVX supplemented with subcutaneous injection of AI letrozole (Novartis International AG, Basel, Switzerland) administered at a dose of 5 µg/kg per day, 5 times per week. Serum β-estradiol was significantly decreased (p = 0.004) in 28-week-old OVX/AI(y) mice and (p = 0.0002) in 28-week-old OVX/AI + LIV(y) mice relative to 4-week-old mice at baseline. No differences in serum β-estradiol were detected between OVX/AI(y) and OVX/AI + LIV(y) mice at 28 weeks of age. This model, characterized in experiment I, was used in a similar fashion for experiment II.
Bisphosphonate administration
Low-dose (2.5 ng/kg) ZA (Zometa, Novartis) was subcutaneously injected in the skeletally mature mice (OVX/AI + ZA and OVX/AI + LIV + ZA groups) from experiment II once per week commencing the day following OVX surgery.
Mechanical loading protocol
Mechanical loading (LIV and mock-LIV) regimens commenced at 4 weeks of age for the younger cohort (experiment I) and at 16 weeks of age for the older cohort (experiment II). Mice assigned to the mechanical loading regimen were subjected to LIV (0.7 ± 0.025 g, 90 Hz sine wave) for 20 min per treatment, administered once per day, 5 days per week over 28 weeks (experiment I), or twice per day, 5 days per week over 24 weeks (experiment II). OVX/AI(y) mice underwent identical handling and loading protocols as OVX/AI + LIV(y) mice but without activation of the platform (mock-LIV). Daily loading consisted of placing mice into individual compartments within a larger container placed on a vertically oscillating platform (modified from Marodyne Medical, LLC) to administer the LIV signal. Displacements required to produce accelerations at 90 Hz induce membrane deformations of approximately 5 microstrain. The lead investigator was not blinded to the experimental groups during the LIV treatment windows.
Dual-energy X-ray absorptiometry (DXA)
Longitudinal in vivo measurements of bone mineral density (BMD, g/cm2), % whole-body fat mass (grams), and % whole-body lean mass (grams) were collected via DXA scanning (Piximus 2, Lunar Corporation, Madison, WI, USA) to determine body composition. Prior to each scan, mice were anesthetized with an intraperitoneal injection of a ketamine/xylene cocktail (100 mg/kg + 10 mg/kg). Whole-body regions of interest excluded the head and tail. DXA analysis of the lumbar spine was performed using a region of interest situated above the sacral spine and extending superior to L1.
Tissue harvest and preservation
At the termination of the study, each mouse was anesthetized using isoflurane inhalation. Euthanasia was confirmed by cervical dislocation. Whole blood collected via cardiac puncture was stored in serum-separation tubes, with serum aliquots stored at −80 °C. Bones for histologic processing, including the right femora and tibiae, were stripped of muscle and residual soft tissue and fixed in 10% neutral buffered formalin, which was replaced at 48 h with 70% ethanol. At euthanasia, lumbar vertebrae and femora harvested for mechanical testing were wrapped in saline-soaked gauze and frozen at −20 °C. The quadratus femoris muscle was formalin-fixed for 48 h following euthanasia and then transferred to 70% ethanol and preserved for downstream processing.
Micro-computed tomography (µCT)
In vivo and ex vivo scans were performed to quantify bone microarchitecture and visualize reconstructed 3-dimensional representations. Distal femora and L5 vertebrae were measured and evaluated ex vivo using X-ray µCT (μCT40, OpenVMS, Scanco Medical, Wayne, PA, USA). X-ray parameters were set to the following: in vivo source voltage E = 55 kVp, current = 145 μA, integration time = 300 ms, and voxel size = 19 µm; ex vivo source voltage E = 55 kVp, current = 145 μA, integration time = 300 ms, and voxel size = 10 μm. Cancellous (trabecular) bone parameters were identified using the following nomenclature: Tb.BV/TV, Tb.Conn.D, Tb.N, trabecular thickness (Tb.Th), trabecular separation (Tb.Sp), trabecular BMD (Tb.BMD), and trabecular structure model index (Tb.SMI). Cortical bone parameters were identified using the following nomenclature: cortical bone area fraction (Ct.BA/TA), cortical thickness (Ct.Th), and cortical polar moment of inertia (Ct.pMOI). Starting 700 μm proximal to the femoral growth plate, 1000 μm of metaphyseal trabecular bone was evaluated. A 500-μm uniform length of trabecular bone (excluding the growth plate) was evaluated across the L5 vertebral bodies; 500 µm of cortical bone was quantified about the femoral mid-diaphysis.
Mechanical testing of bones
Mechanical testing was performed using a 3-point bending jig to quantify both structural and material-level cortical bone properties at the femoral mid-diaphysis, and vertical compression jigs were used to quantify the structural properties resisting fracture in the lumbar vertebrae. Force and displacement data were recorded (Rcontroller, Test Resources, Shakopee, MN, USA), and structural and material properties were derived from force curves using a customized MATLAB (MathWorks, Natick, MA, USA) script.
Spines were thawed, then the L5 vertebrae were isolated using a scalpel and cleaned of intervertebral disks and cartilaginous tissue from proximal and distal ends. Pedicles and spinous processes were removed, leaving only the vertebral body. Sample vertebrae were loaded along the vertical axis with 2 N of preload force between platens using 11 lb-f load cells. Position-controlled rate of compression was applied until failure. Samples that slipped during compression were omitted from the dataset.
Femora were thawed, excess soft tissue was removed, and the femora were scanned using µCT to obtain the CSA. For 3-point mechanical testing of the femora, the distance between the lower supports (∆x = 9 mm) was maintained throughout the analyses, while the upper support was centered between the bottom supports. 150 lb-f load cells were used for testing with 1 N of preload force, and the position-controlled rate was applied to each sample (femora were positioned anterior side facing up) until failure.
Bone histology/histomorphometry
The lengths of long bones were measured ex vivo using digital calipers. Formalin-fixed bone specimens for paraffin embedding and osmium tetroxide staining were decalcified in ethylenediaminetetraacetic acid (10%; pH = 7.32) for 2 weeks. 5 μm paraffin-embedded sagittal cross sections were stained with hematoxylin and eosin. PMMA-embedded intact lumbar vertebrae were sectioned (4.5 µm) and were subsequently either left unstained (i.e., dynamic histomorphometry) or stained with tartrate-resistant acid phosphatase to quantify osteoclasts or with von Kossa-McNeil to quantify osteoblasts. Bone analysis software (v.18.1, BIOQUANT Osteo, Bioquant Image Analysis Corporation, Nashville, TN, USA) was used to analyze the results.
Forelimb grip strength
Muscle function was quantified with the use of a grip strength meter (Bioseb, Pinellas Park, FL, USA) to report maximum applied force (grams). Prior to each pass, the researcher suspended mice individually by their tails, placing forelimbs within reaching distance of the front of the mesh grid. A constant horizontal pulling force was applied until the mice released their grip from the mesh. Triplicate readings per mouse were averaged, and longitudinal measurements were taken throughout the study.
Muscle histology
Paraffin-embedded histologic sections (4.5 µm) of the quadratus femoris were stained with hematoxylin and eosin. Dedicated software (v.18.1 BIOQUANT Osteo) was used to acquire 5 fields-of-view at 10× magnification and then used to quantify myofiber CSA by contouring each myofiber within the field of view, thresholding out the space between the fibers, and normalizing to the total tissue area.
Muscle-specific force/muscle fatigue
Ex vivo contractility of the extensor digitorum longus (EDL) muscle was performed via tetanic stimulation as previously described27. Briefly, whole EDL muscles were dissected from hindlimbs and placed in Tyrode buffer to maintain Ca2+ potential. Muscles were mounted to force transducers (Aurora Scientific, Ontario, Canada) using stainless steel hooks tied to the proximal and distal tendons, while remaining hydrated in a stimulation chamber containing O2/CO2 (95/5%) bubbled into Tyrode buffer. The excised muscle was stimulated to contract using a supramaximal stimulus between two platinum electrodes. Force–frequency and fatigue–frequency relationships were determined by triggering contraction using incremental stimulation frequencies (specific force: 0.5 ms pulses at 1–150 Hz for 350 ms at maximal voltage; fatigue: 0.5 ms pulses at 1–50 Hz for 350 ms at maximal voltage). Data were collected using proprietary “Dynamic Muscle Control/Data Acquisition” and “Dynamic Muscle Control Data Analysis” programs (Aurora Scientific). Wet tissue weights and lengths of gastrocnemius, EDL, soleus, and tibialis anterior muscles were recorded with digital calipers at euthanasia. For quantification of the specific force and fatigue of the EDL, the absolute force was normalized to muscle size and calculated by dividing muscle weight by muscle length using a muscle density constant (1.056 kg/m−3)97. Investigators performing the measurements were blinded to the treatment groups.
Marrow adipose histology
Histologic sections (20×, hematoxylin and eosin) of the distal tibial metaphysis were analyzed starting from the growth plate for 2000 µm distally to quantify the total adipocyte volume encased within a given marrow volume (Ad.Ar./Ma.Ar.) and total number of adipocytes within this space (N.Ad./Ma.Ar.). Briefly, the region of interest across the marrow space was contoured, followed by binary thresholding of the marrow and adipocytes independently, which were each quantified using Osteo (v.19.2).
Osmium tetroxide staining of marrow adipose tissue
Marrow adipose tissue was quantified as described previously98. Formalin-fixed tibiae were scanned via µCT. Bones were decalcified in ethylenediaminetetraacetic acid (pH = 7.32) for 10 days, rinsed with tap water, and transferred to Eppendorf tubes. A 1:1 ratio of osmium tetroxide (OsO4) to sodium bicarbonate mixture was added to each sample, allowing the OsO4 to permeate the marrow for 48 h. After discarding the waste solution, bones were washed with tap water again and stored in phosphate-buffered saline at 4 °C. Samples were scanned using µCT imaging (E = 55 kVa, I = 72 µA, voxel size = 12.5 µm). Values were produced by thresholding tissue that absorbed OsO4 and normalizing this value to the medullary cavity volume across 6000 µm from the epiphysis to the inferior tibiofibular joint.
Immunoassays
Serum was collected at the termination of the study and stored at −80 °C. Thawed samples were used to quantify leptin (Quantikine, catalog #MOB00, Minneapolis, MN, USA) as analyzed by enzyme-linked immunosorbent assay per manufacturer guidelines.
Serum metabolite analyses
Serum isolated from whole blood at baseline (n = 10) and euthanasia (OVX/AI(y): n = 5, OVX/AI + LIV(y): n = 10) was used to quantify fatty acid metabolite intermediates (acylcarnitines). Acylcarnitines were analyzed by flow injection electrospray ionization tandem mass spectrometry and quantified by isotope or pseudo-isotope dilution using methods described previously99,100,101. Tissue homogenates were prepared at 50 mg/ml in 50% acetonitrile/0.3% formic acid, and serum samples were spiked with a cocktail of heavy-isotope internal standards (Cambridge Isotope Laboratories, MA, USA; CDN Isotopes, Canada) and deproteinated with methanol. The supernatants were dried and esterified with acidified methanol. Mass spectra for acylcarnitines were obtained in the positive mode using a precursor ion scan of m/z 99. The spectra were acquired in a multi-channel analyzer (MCA) mode to improve signal-to-noise. The data were generated using a Waters Xevo TQD mass spectrometer equipped with an AcquityTM UPLC system and a data system controlled by MassLynx 4.1 operating system (Waters, Milford, MA). Ion ratios of analyte to respective internal standard computed from centroided spectra were converted to concentrations using calibrators constructed from authentic aliphatic acylcarnitines (Sigma, MO, USA; Larodan Sweden) and dialyzed fetal bovine serum (Sigma, MO, USA). Acylcarnitine concentrations were analyzed using Metaboanalyst (v.2.0, McGill, Quebec, Canada)102,103,104 and run in the statistical package R (v.2.14.0). Initial unsupervised evaluation using principal component analysis identified the mouse strain as the primary source of variance; therefore, the metabolites were mean-centered per strain after log transformation of concentrations, resulting in a Gaussian distribution. All metabolite analyses were performed blinded to group, experimental treatment, and all other identifying data. After acylcarnitine values were determined, metabolomics analysis of grouped data was performed with the user blinded to all group designations. All metabolomics data has been uploaded to an online repository, Metabolomics Workbench105.
Glucose tolerance testing
Mice aged to 26 weeks were fasted overnight and intraperitoneally injected the following morning with a bolus of glucose (20% glucose solution). Subsequent blood glucose samples drawn from the tail vein were read using a glucometer at 0, 15, 30, 60, and 120 min intervals. Serum glucose was not measured in the older mice in order to minimize potential stress.
Statistical analyses
All graphical data are represented by means ± SEM. Repeated measures analysis of variance was performed for longitudinal measurements of body weight, grip strength, body composition, and bone mineral density as measured by DXA, muscle contractility, and µCT, followed by Tukey’s HSD procedure to adjust for multiple comparisons among groups (Prism, GraphPad Software v.7.0c, San Diego, CA, USA). Pearson correlations were performed to compare total adipose tissue mass with serum leptin and reported along with 95% confidence intervals. The student’s two-tailed t-test was performed on single-timepoint data to compare groups. Single-timepoint analyses of these groups were quantified by one-way ANOVA and Tukey’s post hoc procedure. A moderated t-statistic model (SAM)106 was used to identify metabolites that differed between groups at a false discovery rate <5% with Fisher’s least significant difference used for post-test comparisons.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Supplementary Information accompanies this paper and includes Supplementary Figs. 1–4, and Supplementary Tables 1–3, which provides extended methodological details and additional statistical analyses. A Reporting Summary is supplied as a separate file describing experimental design, animal use, and statistical approaches. Source Data supporting the main and Supplementary Figures are provided as an accompanying Excel file. The metabolomics datasets have been deposited in the Metabolomics Workbench under accession number ST004315 and are accessible at https://doi.org/10.21228/M8S55N. Source data are provided with this paper.
Code availability
The code for analyzing the mechanical data was developed by Dr. Joseph Wallace. Anyone wishing to use the code can contact him for training and access (jwalla78@purdue.edu). Dr. Wallace was not involved in the research presented here and is not responsible for any of the data.
References
Kingsley, L. A., Fournier, P. G., Chirgwin, J. M. & Guise, T. A. Molecular biology of bone metastasis. Mol. Cancer Ther. 6, 2609–2617 (2007).
Siegel, R. L., Miller, K. D., Fuchs, H. E. & Jemal, A. Cancer statistics, 2022. CA Cancer J. Clin. 72, 7–33 (2022).
Kohler, B. A. et al. Annual report to the nation on the status of cancer, 1975-2011, featuring incidence of breast cancer subtypes by race/ethnicity, poverty, and state. J. Natl. Cancer Inst. 107, djv048 (2015).
Clark, G. M., Osborne, C. K. & McGuire, W. L. Correlations between estrogen receptor, progesterone receptor, and patient characteristics in human breast cancer. J. Clin. Oncol. 2, 1102–1109 (1984).
Weilbaecher, K. N., Guise, T. A. & McCauley, L. K. Cancer to bone: a fatal attraction. Nat. Rev. Cancer 11, 411–425 (2011).
Tubiana-Hulin, M. Incidence, prevalence and distribution of bone metastases. Bone 12, S9–S10 (1991).
Buijs, J. T., Stayrook, K. R. & Guise, T. A. TGF-beta in the bone microenvironment: role in breast cancer metastases. Cancer Microenviron. 4, 261–281 (2011).
Guise, T. A., Yoneda, T., Yates, A. J. & Mundy, G. R. The combined effect of tumor-produced parathyroid hormone-related protein and transforming growth factor-alpha enhance hypercalcemia in vivo and bone resorption in vitro. J. Clin. Endocrinol. Metab. 77, 40–45 (1993).
Guise, T. A. et al. Evidence for a causal role of parathyroid hormone-related protein in the pathogenesis of human breast cancer-mediated osteolysis. J. Clin. Invest. 98, 1544–1549 (1996).
Guo, D., Huang, J. & Gong, J. Bone morphogenetic protein 4 (BMP4) is required for migration and invasion of breast cancer. Mol. Cell Biochem. 363, 179–190 (2012).
Mohan, S. & Baylink, D. J. Bone growth factors. Clin. Orthop. Relat. Res. 263, 30–48 (1991).
Javelaud, D. et al. TGF-beta/SMAD/GLI2 signaling axis in cancer progression and metastasis. Cancer Res. 71, 5606–5610 (2011).
Roodman, G. D. Mechanisms of bone metastasis. N. Engl. J. Med. 350, 1655–1664 (2004).
Love, R. R. et al. Effects of tamoxifen on bone mineral density in postmenopausal women with breast cancer. N. Engl. J. Med. 326, 852–856 (1992).
Fisher, B. et al. A randomized clinical trial evaluating tamoxifen in the treatment of patients with node-negative breast cancer who have estrogen-receptor-positive tumors. N. Engl. J. Med. 320, 479–484 (1989).
Burstein, H. J. et al. American Society of Clinical Oncology clinical practice guideline: update on adjuvant endocrine therapy for women with hormone receptor-positive breast cancer. J. Clin. Oncol. 28, 3784–3796 (2010).
Shiau, A. K. et al. The structural basis of estrogen receptor/coactivator recognition and the antagonism of this interaction by tamoxifen. Cell 95, 927–937 (1998).
Dowsett, M. et al. Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J. Clin. Oncol. 28, 509–518 (2010).
Simpson, E. R. & Davis, S. R. Minireview: aromatase and the regulation of estrogen biosynthesis-some new perspectives. Endocrinology 142, 4589–4594 (2001).
Goss, P. E. et al. Exemestane for breast-cancer prevention in postmenopausal women. N. Engl. J. Med. 364, 2381–2391 (2011).
Coates, A. S. et al. Five years of letrozole compared with tamoxifen as initial adjuvant therapy for postmenopausal women with endocrine-responsive early breast cancer: update of study BIG 1-98. J. Clin. Oncol. 25, 486–492 (2007).
Saad, F. et al. Cancer treatment-induced bone loss in breast and prostate cancer. J. Clin. Oncol. 26, 5465–5476 (2008).
Rabaglio, M. et al. Bone fractures among postmenopausal patients with endocrine-responsive early breast cancer treated with 5 years of letrozole or tamoxifen in the BIG 1-98 trial. Ann. Oncol. 20, 1489–1498 (2009).
Schneider, A. et al. Bone turnover mediates preferential localization of prostate cancer in the skeleton. Endocrinology 146, 1727–1736 (2005).
Bellinger, A. M. et al. Remodeling of ryanodine receptor complex causes “leaky” channels: a molecular mechanism for decreased exercise capacity. Proc. Natl. Acad. Sci. USA 105, 2198–2202 (2008).
Andersson, D. C. et al. Ryanodine receptor oxidation causes intracellular calcium leak and muscle weakness in aging. Cell Metab. 14, 196–207 (2011).
Waning, D. L. et al. Excess TGF-beta mediates muscle weakness associated with bone metastases in mice. Nat. Med. 21, 1262–1271 (2015).
McPherson, P. S. & Campbell, K. P. The ryanodine receptor/Ca2+ release channel. J. Biol. Chem. 268, 13765–13768 (1993).
Bekker, P. J. et al. A single-dose placebo-controlled study of AMG 162, a fully human monoclonal antibody to RANKL, in postmenopausal women. 2004. J. Bone Min. Res. 20, 2275–2282 (2005).
Lipton, A. et al. Randomized active-controlled phase II study of denosumab efficacy and safety in patients with breast cancer-related bone metastases. J. Clin. Oncol. 25, 4431–4437 (2007).
Gnant, M. et al. Adjuvant denosumab in breast cancer (ABCSG-18): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet 386, 433–443 (2015).
Ottewell, P. D. et al. Zoledronic acid has differential antitumor activity in the pre- and postmenopausal bone microenvironment in vivo. Clin. Cancer Res. 20, 2922–2932 (2014).
Hughes, D. E. et al. Bisphosphonates promote apoptosis in murine osteoclasts in vitro and in vivo. J. Bone Min. Res. 10, 1478–1487 (1995).
Jagdev, S. P., Coleman, R. E., Shipman, C. M., Rostami, H. A. & Croucher, P. I. The bisphosphonate, zoledronic acid, induces apoptosis of breast cancer cells: evidence for synergy with paclitaxel. Br. J. Cancer 84, 1126–1134 (2001).
Senaratne, S. G., Pirianov, G., Mansi, J. L., Arnett, T. R. & Colston, K. W. Bisphosphonates induce apoptosis in human breast cancer cell lines. Br. J. Cancer 82, 1459–1468 (2000).
Lintermans, A. et al. Aromatase inhibitor-induced loss of grip strength is body mass index dependent: hypothesis-generating findings for its pathogenesis. Ann. Oncol. 22, 1763–1769 (2011).
Henry, N. L., Giles, J. T. & Stearns, V. Aromatase inhibitor-associated musculoskeletal symptoms: etiology and strategies for management. Oncology 22, 1401–1408 (2008).
Brown, S. A. & Guise, T. A. Cancer treatment-related bone disease. Crit. Rev. Eukaryot. Gene Expr. 19, 47–60 (2009).
Donnellan, P. P., Douglas, S. L., Cameron, D. A. & Leonard, R. C. Aromatase inhibitors and arthralgia. J. Clin. Oncol. 19, 2767 (2001).
Henry, N. L. et al. Predictors of aromatase inhibitor discontinuation as a result of treatment-emergent symptoms in early-stage breast cancer. J. Clin. Oncol. 30, 936–942 (2012).
Henry, N. L. et al. Prospective characterization of musculoskeletal symptoms in early stage breast cancer patients treated with aromatase inhibitors. Breast Cancer Res. Treat. 111, 365–372 (2008).
Dent, S. F., Gaspo, R., Kissner, M. & Pritchard, K. I. Aromatase inhibitor therapy: toxicities and management strategies in the treatment of postmenopausal women with hormone-sensitive early breast cancer. Breast Cancer Res. Treat. 126, 295–310 (2011).
Stattin, K., Michaelsson, K., Larsson, S. C., Wolk, A. & Byberg, L. Leisure-time physical activity and risk of fracture: a cohort study of 66,940 men and women. J. Bone Min. Res. 32, 1599–1606 (2017).
Hinton, P. S., Nigh, P. & Thyfault, J. Effectiveness of resistance training or jumping-exercise to increase bone mineral density in men with low bone mass: a 12-month randomized, clinical trial. Bone 79, 203–212 (2015).
Dalsky, G. P. et al. Weight-bearing exercise training and lumbar bone mineral content in postmenopausal women. Ann. Intern. Med. 108, 824–828 (1988).
Romijn, J. A. et al. Regulation of endogenous fat and carbohydrate metabolism in relation to exercise intensity and duration. Am. J. Physiol. 265, E380–E391 (1993).
Styner, M. et al. Bone marrow fat accumulation accelerated by high fat diet is suppressed by exercise. Bone 64, 39–46 (2014).
Nicklas, B. J. et al. Diet-induced weight loss, exercise, and chronic inflammation in older, obese adults: a randomized controlled clinical trial. Am. J. Clin. Nutr. 79, 544–551 (2004).
Huang, H. P. et al. Adherence to prescribed exercise time and intensity declines as the exercise program proceeds: findings from women under treatment for breast cancer. Support Care Cancer 23, 2061–2071 (2015).
Ness, K. K. et al. Skeletal, neuromuscular and fitness impairments among children with newly diagnosed acute lymphoblastic leukemia. Leuk. Lymphoma 56, 1004–1011 (2015).
Ness, K. K. et al. Physiologic frailty as a sign of accelerated aging among adult survivors of childhood cancer: a report from the St Jude Lifetime cohort study. J. Clin. Oncol. 31, 4496–4503 (2013).
Fritton, S. P., McLeod, K. J. & Rubin, C. T. Quantifying the strain history of bone: spatial uniformity and self-similarity of low-magnitude strains. J. Biomech. 33, 317–325 (2000).
Rubin, C., Turner, A. S., Bain, S., Mallinckrodt, C. & McLeod, K. Anabolism. Low mechanical signals strengthen long bones. Nature 412, 603–604 (2001).
Pagnotti, G. M. et al. Combating osteoporosis and obesity with exercise: leveraging cell mechanosensitivity. Nat. Rev. Endocrinol. 15, 339–355 (2019).
Huang, R. P., Rubin, C. T. & McLeod, K. J. Changes in postural muscle dynamics as a function of age. J. Gerontol. A Biol. Sci. Med. Sci. 54, B352–B357 (1999).
Morse, C. I., Thom, J. M., Reeves, N. D., Birch, K. M. & Narici, M. V. In vivo physiological cross-sectional area and specific force are reduced in the gastrocnemius of elderly men. J. Appl. Physiol. 99, 1050–1055 (2005).
Pagnotti, G. M. et al. Low magnitude mechanical signals mitigate osteopenia without compromising longevity in an aged murine model of spontaneous granulosa cell ovarian cancer. Bone 51, 570–577 (2012).
Pagnotti, G. M. et al. Low intensity vibration mitigates tumor progression and protects bone quantity and quality in a murine model of myeloma. Bone 90, 69–79 (2016).
van Heuvelen, M. J. G. et al. Reporting guidelines for whole-body vibration studies in humans, animals and cell cultures: a consensus statement from an international group of experts. Biology https://doi.org/10.3390/biology10100965 (2021).
Patel, V. S. et al. Incorporating refractory period in mechanical stimulation mitigates obesity-induced adipose tissue dysfunction in adult mice. Obesity 25, 1745–1753 (2017).
Luu, Y. K. et al. Mechanical stimulation of mesenchymal stem cell proliferation and differentiation promotes osteogenesis while preventing dietary-induced obesity. J. Bone Min. Res. 24, 50–61 (2009).
Frechette, D. M., Krishnamoorthy, D., Adler, B. J., Chan, M. E. & Rubin, C. T. Diminished satellite cells and elevated adipogenic gene expression in muscle as caused by ovariectomy are averted by low-magnitude mechanical signals. J. Appl. Physiol. 119, 27–36 (2015).
Uzer, G. et al. Cell mechanosensitivity to extremely low-magnitude signals is enabled by a LINCed nucleus. Stem Cells 33, 2063–2076 (2015).
Uzer, G., Fuchs, R. K., Rubin, J. & Thompson, W. R. Concise review: plasma and nuclear membranes convey mechanical information to regulate mesenchymal stem cell lineage. Stem Cells 34, 1455–1463 (2016).
Sen, B. et al. Mechanical signal influence on mesenchymal stem cell fate is enhanced by incorporation of refractory periods into the loading regimen. J. Biomech. 44, 593–599 (2011).
Yi, X. et al. Mechanical suppression of breast cancer cell invasion and paracrine signaling to osteoclasts requires nucleo-cytoskeletal connectivity. Bone Res. 8, 40 (2020).
Gilsanz, V. et al. Low-level, high-frequency mechanical signals enhance musculoskeletal development of young women with low BMD. J. Bone Min. Res. 21, 1464–1474 (2006).
DiVasta, A. D. et al. The ability of low-magnitude mechanical signals to normalize bone turnover in adolescents hospitalized for anorexia nervosa. Osteoporos. Int. 28, 1255–1263 (2017).
Wren, T. A. et al. Effect of high-frequency, low-magnitude vibration on bone and muscle in children with cerebral palsy. J. Pediatr. Orthop. 30, 732–738 (2010).
Mogil, R. J. et al. Effect of low-magnitude, high-frequency mechanical stimulation on BMD among young childhood cancer survivors: a randomized clinical trial. JAMA Oncol. 2, 908–914 (2016).
Wright, L. E. et al. Aromatase inhibitor-induced bone loss increases the progression of estrogen receptor-negative breast cancer in bone and exacerbates muscle weakness in vivo. Oncotarget 8, 8406–8419 (2017).
Bouvard, B. et al. High prevalence of vertebral fractures in women with breast cancer starting aromatase inhibitor therapy. Ann. Oncol. 23, 1151–1156 (2012).
Villa, P. et al. Impact of aromatase inhibitor treatment on vertebral morphology and bone mineral density in postmenopausal women with breast cancer. Menopause 23, 33–39 (2016).
Ma, C. X., Reinert, T., Chmielewska, I. & Ellis, M. J. Mechanisms of aromatase inhibitor resistance. Nat. Rev. Cancer 15, 261–275 (2015).
Gupta, S., Vijayaraghavan, S., Uzer, G. & Judex, S. Multiple exposures to unloading decrease bone’s responsivity but compound skeletal losses in C57BL/6 mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 303, R159–R167 (2012).
Lawler, J. M., Song, W. & Demaree, S. R. Hindlimb unloading increases oxidative stress and disrupts antioxidant capacity in skeletal muscle. Free Radic. Biol. Med. 35, 9–16 (2003).
Ozcivici, E. & Judex, S. Trabecular bone recovers from mechanical unloading primarily by restoring its mechanical function rather than its morphology. Bone 67, 122–129 (2014).
Holguin, N., Uzer, G., Chiang, F. P., Rubin, C. & Judex, S. Brief daily exposure to low-intensity vibration mitigates the degradation of the intervertebral disc in a frequency-specific manner. J. Appl. Physiol. 111, 1846–1853 (2011).
Ballinger, T. J. et al. Impact of primary breast cancer therapy on energetic capacity and body composition. Breast Cancer Res. Treat. 172, 445–452 (2018).
Quail, D. F. et al. Obesity alters the lung myeloid cell landscape to enhance breast cancer metastasis through IL5 and GM-CSF. Nat. Cell Biol. 19, 974–987 (2017).
Iyengar, N. M. et al. Association of body fat and risk of breast cancer in postmenopausal women with normal body mass index: a secondary analysis of a randomized clinical trial and observational study. JAMA Oncol. 5, 155–163 (2019).
Chan, D. S. et al. Body mass index and survival in women with breast cancer-systematic literature review and meta-analysis of 82 follow-up studies. Ann. Oncol. 25, 1901–1914 (2014).
Calle, E. E. & Kaaks, R. Overweight, obesity and cancer: epidemiological evidence and proposed mechanisms. Nat. Rev. Cancer 4, 579–591 (2004).
Eaton, S. Control of mitochondrial beta-oxidation flux. Prog. Lipid Res. 41, 197–239 (2002).
Hoppel, C. L. & Genuth, S. M. Carnitine metabolism in normal-weight and obese human subjects during fasting. Am. J. Physiol. 238, E409–E415 (1980).
Mihalik, S. J. et al. Increased levels of plasma acylcarnitines in obesity and type 2 diabetes and identification of a marker of glucolipotoxicity. Obesity 18, 1695–1700 (2010).
Markopoulos, C. J., Tsaroucha, A. K. & Gogas, H. J. Effect of aromatase inhibitors on the lipid profile of postmenopausal breast cancer patients. Clin. Lipido. 5, 245–254 (2010).
Lu, Y. et al. Acetylcarnitine is a candidate diagnostic and prognostic biomarker of hepatocellular carcinoma. Cancer Res. 76, 2912–2920 (2016).
Costell, M., O’Connor, J. E. & Grisolia, S. Age-dependent decrease of carnitine content in muscle of mice and humans. Biochem. Biophys. Res. Commun. 161, 1135–1143 (1989).
Kang, M., Yoo, H. J., Kim, M., Kim, M. & Lee, J. H. Metabolomics identifies increases in the acylcarnitine profiles in the plasma of overweight subjects in response to mild weight loss: a randomized, controlled design study. Lipids Health Dis. 17, 237 (2018).
Hansen, J. B. et al. Peroxisome proliferator-activated receptor delta (PPARdelta)-mediated regulation of preadipocyte proliferation and gene expression is dependent on cAMP signaling. J. Biol. Chem. 276, 3175–3182 (2001).
van Vlies, N., Ferdinandusse, S., Turkenburg, M., Wanders, R. J. & Vaz, F. M. PPAR alpha-activation results in enhanced carnitine biosynthesis and OCTN2-mediated hepatic carnitine accumulation. Biochim. Biophys. Acta 1767, 1134–1142 (2007).
Sen, B. et al. Mechanical strain inhibits adipogenesis in mesenchymal stem cells by stimulating a durable beta-catenin signal. Endocrinology 149, 6065–6075 (2008).
Luu, Y. K. et al. Development of diet-induced fatty liver disease in the aging mouse is suppressed by brief daily exposure to low-magnitude mechanical signals. Int. J. Obes. 34, 401–405 (2010).
Krishnamoorthy, D. et al. Marrow adipogenesis and bone loss that parallels estrogen deficiency is slowed by low-intensity mechanical signals. Osteoporos. Int. 27, 747–756 (2016).
Luu, Y. K., Pessin, J. E., Judex, S., Rubin, J. & Rubin, C. T. Mechanical signals as a non-invasive means to influence mesenchymal stem cell fate, promoting bone and suppressing the fat phenotype. Bonekey Osteovision 6, 132–149 (2009).
Yamada, T. et al. Impaired myofibrillar function in the soleus muscle of mice with collagen-induced arthritis. Arthritis Rheum. 60, 3280–3289 (2009).
Scheller, E. L. et al. Use of osmium tetroxide staining with microcomputerized tomography to visualize and quantify bone marrow adipose tissue in vivo. Methods Enzymol. 537, 123–139 (2014).
An, J. et al. Hepatic expression of malonyl-CoA decarboxylase reverses muscle, liver and whole-animal insulin resistance. Nat. Med. 10, 268–274 (2004).
Wu, J. Y. et al. ENU mutagenesis identifies mice with mitochondrial branched-chain aminotransferase deficiency resembling human maple syrup urine disease. J. Clin. Invest. 113, 434–440 (2004).
Jensen, M. V. et al. Compensatory responses to pyruvate carboxylase suppression in islet beta-cells. Preservation of glucose-stimulated insulin secretion. J. Biol. Chem. 281, 22342–22351 (2006).
Xia, J., Psychogios, N., Young, N. & Wishart, D. S. MetaboAnalyst: a web server for metabolomic data analysis and interpretation. Nucleic Acids Res. 37, W652–W660 (2009).
Xia, J. & Wishart, D. S. Web-based inference of biological patterns, functions and pathways from metabolomic data using MetaboAnalyst. Nat. Protoc. 6, 743–760 (2011).
Xia, J., Mandal, R., Sinelnikov, I. V., Broadhurst, D. & Wishart, D. S. MetaboAnalyst 2.0-a comprehensive server for metabolomic data analysis. Nucleic Acids Res. 40, W127–W133 (2012).
Sud, M. et al. Metabolomics Workbench: an international repository for metabolomics data and metadata, metabolite standards, protocols, tutorials and training, and analysis tools. Nucleic Acids Res. 44, D463–D470 (2016).
Tusher, V. G., Tibshirani, R. & Chu, G. Significance analysis of microarrays applied to the ionizing radiation response. Proc. Natl. Acad. Sci. USA 98, 5116–5121 (2001).
Acknowledgements
The authors would like to express their gratitude to the Indiana University Laboratory Animal Resource Center; the Matthew Allen Lab personnel at IUSM, specifically, Mohammad W. Aref, Elizabeth A. Swallow, and Corrine E. Metzger, for their assistance during post-processing and access to mechanical testing equipment; Erica Goodoff, a from The University of Texas MD Anderson Cancer Center Research Medical Library; Rachel A. Davidowitz from The University of Texas MD Anderson Cancer Center CCSG and Extramural Research Development; Roland Bassett, Jr. from The University of Texas MD Anderson Cancer Center Department of Biostatistics; Thomas M. O’Connell from Indiana University Department of Otolaryngology; and Olga Ilkayeva from the Duke University Department of Medicine and Metabolomics Core. This study was developed and supported by Department of Defense Grants W81XWH−16-1-0040 (T.A.G., PI) and BC150678P1 (T.A.G., PI); the Cancer Prevention and Research Institute of Texas Grant 00011633 (T.A.G., PI); (Scholar of CPRIT Established Investigator Award RR190108: T.A.G., PI; Theresa A. Guise, MD is a CPRIT scholar in Cancer Research); Indiana Clinical and Translational Sciences Institute TL1 Grant TR002531 (A. Shekhar, PI; G.M.P., Subawardee) and UL1 TR002529 (A. Shekhar, PI; G.M.P., Subawardee); Dive into the Pink (T.A.G., PI); the Jerry W. and Peggy S. Throgmartin Endowment (T.A.G.); and the Indiana University School of Medicine (IUSM) Center for Musculoskeletal Health (ICMH); and the Lawrence Family Bone Disease Program of Texas (G.M.P., PI). The work of the Metabolomics Workbench repository is supported by NIH grant U2C-DK119886 and OT2-OD030544.
Author information
Authors and Affiliations
Contributions
G.M.P., T.T., L.E.W., K.S.M., and T.A.G. conceptualized the experiments. G.M.P., T.T., S.K.J., and Y.S. provided husbandry, logging, and technical care with the mice. G.M.P., T.T., R.R.P., S.M., A.F.T., K.S.M., and T.A.G. provided histological and bone histomorphometry expertise and analyses. G.M.P., S.K.J., R.R.P., and S.S. performed µCT scanning and analysis. G.M.P., L.E.W., and R.R.P. performed MAT staining and analysis. G.M.P. and T.T. performed glucose metabolism studies and analysis. M.W. provided technical advice and performed an analysis of metabolomics data. G.M.P., T.T., and L.E.W. performed ex vivo analysis of muscle contractility. G.M.P., T.T., S.K.J., R.R.P., and Y.S. performed and analyzed DXA scans. G.M.P. performed mechanical testing on femora and vertebrae and subsequent analyses. G.M.P., W.R.T., and C.T.R. contributed expertise on mechanical loading. T.T., L.E.W., K.S.M., and T.A.G. provided expertise on breast cancer treatment and complete estrogen deprivation procedures and outcomes. G.M.P. and T.A.G. wrote the manuscript with input from all authors. All authors contributed to the discussion of the results over the course of the experiments and provided a critical review of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare the following competing interests: Dr. Clinton T. Rubin is a founder of Marodyne Medical, LLC and Lahara Bio, LLC and has several patents issued and pending related to the ability of mechanical signals to control musculoskeletal and metabolic disorders.
Peer review
Peer review information
Nature Communications thanks Mário Bernardo-Filho, Penelope Ottewell, and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Pagnotti, G.M., Trivedi, T., Wright, L.E. et al. Low intensity vibration with zoledronate reduces musculoskeletal weakness and adiposity in estrogen deprived female mice. Nat Commun 17, 1808 (2026). https://doi.org/10.1038/s41467-026-68522-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68522-0