Abstract
The uneven detection of prebiotic organic compounds in meteorites—where amino acids and nucleobases are commonly identified but sugars remain rare and poorly characterized—limits our understanding of extraterrestrial organic chemistry. This discrepancy is striking given that laboratory simulations of interstellar ice chemistry readily produce complex sugars. Here we report the simultaneous analysis of sugar and amino acid enantiomers in a meteorite sample. Multiple aldoses were detected in the Orgueil meteorite, including ribose, arabinose, xylose, lyxose, and the ketopentose ribulose, several of which display near-racemic distributions consistent with an extraterrestrial origin. Recovery experiments demonstrate that sugar abundances are severely underestimated. Despite this limitation, pentose abundances are comparable to those of some C4–C5 amino acid enantiomers, implying higher true concentrations. These results indicate efficient abiotic sugar formation in space and suggest that meteorites may have delivered a broader range of prebiotically relevant sugars to early Earth than previously recognized.
Similar content being viewed by others
Introduction
Nucleic acids (RNA, DNA), composed of sugars, nucleobases, and phosphates, and proteins, built from amino acids, are the fundamental biopolymers of life. A central question concerning the origin of life on Earth is how these essential biomolecular building blocks were synthesized, concentrated, and assembled into biopolymers, given their fragile nature, the planet’s extreme and fluctuating environmental conditions1,2,3 and the brief timescale for life’s emergence following Earth’s consolidation4. One plausible source of these biomolecular precursors to the terrestrial prebiotic inventory, strongly supported by both astronomical and geological evidence, is their exogenous delivery via extraterrestrial bodies such as interplanetary dust particles and meteorites1,5,6,7,8,9,10, supplementing the local prebiotic synthesis on early Earth.
However, while amino acids and nucleobases have been repeatedly detected in various carbonaceous chondrites11,12,13,14 and, more recently, in samples from the asteroids Ryugu15,16,17 and Bennu18, the detection of sugars—essential as components of nucleic acids but also as monomers of structural and energy‑storage polymers in all living organisms—remains remarkably scarce. To date, only a single study by Furukawa et al.19 has confirmed the presence of ribose and other aldopentoses in meteorite samples, highlighting the current limitations in our ability to detect and understand sugar inventories in extraterrestrial materials. Moreover, the relatively large sample masses required in that study (~2 g) further constrain the applicability of such analyses to scarce pristine materials such as those returned from asteroids (Ryugu, total = 5.4 g; Bennu, 121.6 g). However, sugars and sugar-related compounds have recently been identified in powdered aliquots of regolith returned from asteroid Bennu by the OSIRIS-REx mission20 (OREX-800107-108; 603.4 mg), demonstrating that such compounds can be detected in substantially smaller masses of pristine asteroidal material. Nevertheless, the paucity of data on extraterrestrial sugars contrasts sharply with numerous laboratory studies on interstellar ice21,22,23,24 and planetary or parent body mineral analogs25,26,27, which predict the widespread formation of a diverse range of C3 to C6 aldose sugars under extraterrestrial conditions. Moreover, glycolaldehyde, a potential precursor of larger carbohydrates, has been previously detected in the interstellar medium towards the Galactic center source Sagittarius B2(N)28 as well as on comet 67P/Churyumov-Gerasimenko29. From a prebiotic chemistry perspective, investigating the presence, abundance, and formation mechanisms of sugars under astrophysical conditions is a critical objective. Indeed, the potential delivery of sugars like ribose via meteorites could have provided a sustained source for the formation of primitive nucleotides in environments such as warm little ponds that may have facilitated their stabilization through rapid polymerization2. This is particularly significant given the inherent instability of sugars in aqueous environments6,30,31,32,33, where rapid degradation could otherwise limit their role in early biochemical pathways. The apparent scarcity of sugars in extraterrestrial samples has been attributed to their potential chemical transformation in space or within the meteorite mineral matrix, where they may be converted into sugar alcohols or sugar acids34. In addition, strong matrix effects—particularly the entrapment of sugars within clay mineral phases—could significantly reduce extraction efficiencies35,36.
A major challenge in analyzing prebiotically relevant organics such as amino acids and sugars from astrophysical samples lies in confirming their extraterrestrial origin, due to the ever-present risk of terrestrial contamination. While meteorites are often subject to terrestrial weathering and potential microbial colonization prior to analysis37,38, even returned samples—such as those from (162173) Ryugu and (101955) Bennu, despite being collected under meticulously controlled conditions by the JAXA’s Hayabusa2 and NASA’s OSIRIS-REx missions38,39,40,41 require meticulous handling and verification protocols to confidently assess indigenous organics. Contamination risks remain particularly relevant during post-collection sample processing37. Stable isotope ratios provide a powerful tool for addressing this issue, as they yield insights into the formation pathways and environmental conditions of organic compounds. Importantly, they can help distinguish biological from abiotic origins, since extraterrestrial organics are typically enriched in heavy isotopes relative to their terrestrial counterparts11,42. Notably, chiral amino acids found in meteorites have been found to be enriched in the heavy isotopes ¹³C, ¹⁵N, and D (deuterium)43,44,45. Similarly, the ¹³C isotopic enrichment of the detected sugars in Murchison (CM2) and NWA 801 (CR2) meteorites was used to support their extraterrestrial origin19. However, the feasibility and precision of compound-specific isotopic measurements are significantly constrained by the need for high analyte concentrations, requiring large sample sizes46. This constraint makes isotopic analysis of soluble organics impossible for many relevant extraterrestrial samples, for which very limited sample material is available. Furthermore, since isotopic measurements depend on prior chromatographic separations, the sample preparation process can introduce additional losses and analytical biases, further compromising the reliability of the measurements46. In particular, in the only confirmed detection of sugars in meteorites, Furukawa et al.19 reported that the accuracy of the carbon isotope measurements for arabinose in NWA 801 was limited by a significant co-eluting peak, preventing reliable δ¹³C determination.
As a result of this limitation, enantiomeric excess measurements of chiral organics have increasingly become crucial for evaluating terrestrial contamination, particularly in samples of limited mass available for analysis13,38. The molecular architecture of life on Earth is defined by a striking preference for specific chiral configurations—such as l-amino acids in proteins and d-sugars in nucleic acids—therefore, near-racemic mixtures or the presence of non-biological enantiomers suggest abiotic, and potentially extraterrestrial, origins.
In this study, we report the detection and enantiomeric separation of all four aldopentoses and ribulose, a ketopentose, in a fragment of the Orgueil carbonaceous meteorite weighing only 178 mg. Our analysis combined ultrasound-assisted extraction, ion-exchange chromatography for fractionation and purification, with enantioselective comprehensive two-dimensional gas chromatography coupled to time-of-flight mass spectrometry (GC×GC–TOF‑MS)47,48. The differences in enantiomeric distributions between the detected sugars and the procedural blanks, along with the near-racemic distribution of ribulose, provide evidence supporting their possible extraterrestrial origin.
Critically, enantiomeric resolution was essential for confirming the origin of the detected sugars, as the limited quantities targeted precluded isotopic analysis. Our findings challenge the widely held assumption that sugars are significantly less abundant than amino acids in carbonaceous chondrites. Incorporating compound-specific recovery rates indicates that the estimated quantities of individual sugar enantiomers are at least comparable to those of C4–C5 amino acid enantiomers in the same samples. These results support the hypothesis that meteorites delivered not only amino acids and nucleobases but also appreciable amounts of prebiotic sugars to early Earth—bridging a critical gap between their abiotic synthesis in interstellar ices and their potential role in the emergence of early biochemical systems.
Results
To characterize the sugar and amino acid content of the Orgueil sample while minimizing the degradation of labile organics, we developed an analytical workflow combining sequential extraction, fractionation, and derivatization steps optimized for the simultaneous recovery and separate enantioselective analysis of both compound classes (Fig. 1). Key to this methodology is a mild extraction protocol employing ultrasonic-assisted extraction (temperature <20 °C, neutral pH, and 10-min sonication intervals) to prevent thermal degradation of sugars32. An ion exchange chromatography (IEC) step was employed to remove inorganic ions (e.g., Fe²⁺, Fe³⁺, and Mg²⁺)—major components of chondritic mineral matrices that interfere with subsequent derivatization and chromatographic protocols49,50—and to separate neutral sugars from amino acid zwitterions49. This separation allows for tailored derivatization procedures and chromatographic conditions specific to each molecular class, enhancing both sensitivity and selectivity in their analyses. A detailed description of the sample preparation, extraction procedure, and chromatographic measurements is in the “Methods” section.
IEC = Ion Exchange Chromatography. GC×GC–TOF-MS = Two-dimensional gas chromatography coupled with time-of-flight mass spectrometry.
Detection of ribulose and aldopentoses in Orgueil
Building on the reported inverse relationship between sugar-related compound abundance and carbon number reported by Cooper et al.34, we focused on the detection of C3 to C5 sugar aldoses, including glyceraldehyde, erythrose, ribose, 2-deoxyribose, lyxose, arabinose, xylose, as well as the C5 ketose ribulose. These compounds were targeted as methyl boronic acid derivatives using GC×GC–TOF-MS, chosen for its high enantiomeric resolution power and detectability47.
Despite the limited sample quantity of 178.3 mg of meteorite material and the challenges associated with the extraction and analysis of sugars from extraterrestrial samples we unambiguously detected all four C5 aldoses at concentrations above procedural blank levels (Fig. 2, Supplementary Fig. 4). A detailed comparison of sugar enantiomers in the Orgueil sample and procedural blanks is provided in Supplementary Figs. 4–5 alongside their corresponding mass spectra (Supplementary Figs. 6–9). Arabinose and xylose detected in Orgueil exhibited much stronger bias towards the biologically abundant enantiomers l-arabinose and d-xylose, likely resulting at least partly from terrestrial contamination accumulated over the long curation history of this CI meteorite51. In contrast to arabinose and xylose, the prebiotically significant sugar ribose and its rare stereoisomer lyxose were both detected in nearly racemic proportions (Fig. 2), suggesting an abiotic origin.
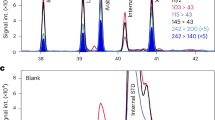
Enantioselective GC×GC–TOF-MS selected ion (m/z = 84 and 97) chromatograms of C5 sugar enantiomers as methylboronic acid esters detected in (A) the Orgueil meteorite sample compared to (B) the reference standard (RS). 1D RT and 2D RT correspond to the retention times in the first and second chromatographic dimensions, respectively.
Enantiomeric excess (ee) data for arabinose and xylose in the Orgueil sample are reported in Table 1. For the other detected sugars, rapid degradation of their derivatives in the presence of residual matrix material (Supplementary Fig. 10) hindered replicate analyses, rendering the corresponding ee values indicative rather than statistically robust. The %ee calculations were based on the peak areas of both enantiomers in the single ion chromatograms to minimize uncertainties associated with calibration curves, such as the conversion from ion counts to ppb. Moreover, matrix effects likely cause different slopes between standard and meteoritic sample curves, potentially leading to inaccurate concentrations. The same approach was applied for the amino acids reported below.
We also report the detection of the C5 ketose ribulose, found in near-racemic proportions. Figure 3 shows the GC×GC chromatograms and corresponding mass spectra of the ribulose enantiomers, along with procedural blanks and reference standards for comparison.
Enantioselective GC×GC–TOF-MS selected ion chromatograms (m/z = 97) and mass spectra of ribulose enantiomers as methylboronic acid esters detected in (A) the Orgueil fragment compared to (B) a procedural blank processed identically (PB) and (C) a reference standard (RS). 1D RT and 2D RT correspond to the retention times in the first and second chromatographic dimensions, respectively.
The concentrations of sugars measured in the Orgueil sample ranged from 0.1 to 6.8 ppb, corresponding to approximately 0.67 to 45 pmol/g (Table 1). Arabinose and xylose concentrations were 2–3 orders of magnitude lower than those reported for the CM2 meteorite Murchison by Furukawa et al.19. However, because the reported isotopic ratio of Murchison xylose (δ13C ≈ −1 ± 4‰) falls only slightly outside the terrestrial range (−32 to −1‰) and its abundance is relatively high compared with other sugars, a terrestrial contribution to the observed xylose signal cannot be fully excluded, particularly in the absence of enantiomeric resolution. Given the very limited dataset on meteoritic sugars, it is premature to attribute the apparent differences in sugar abundances between CI and CM chondrites to variations in parent-body histories or mineral compositions. Comparison of aldopentose distributions between Orgueil and Bennu20 reveals some quantitative differences that likely reflect either distinct parent-body processing histories or variations in matrix effects during extraction. Lyxose abundances are of the same order of magnitude in both samples, while ribose appears slightly less abundant in Orgueil; these comparisons do not yet account for the low recovery rates in our analyses (~20% for lyxose and ~5% for ribose). In contrast, arabinose and xylose show more substantial differences between Orgueil and Bennu.
To better constrain the origin of the detected sugars, we conducted a detailed assessment of potential contamination sources in our procedural blanks. We compared sugar levels in the reagent blank with those in procedural blanks prepared without and with the SiO₂ matrix, which indicated that the major contribution to contamination arose during the derivatization step, specifically from the derivatization reagents (Supplementary Note 2, Supplementary Fig. 4). Moreover, spiking experiments with several milligrams of a previously extracted Orgueil sample revealed a pronounced suppression of sugar recovery in the presence of a CI-type meteorite matrix (Supplementary Note 3), with recovery rates ranging from approximately 1% for ribulose to 30% for arabinose. Notably, these compound-specific recovery efficiencies align with the aldopentoses ultimately detected and plausibly explain the absence of others whose recovery rates were below our detection threshold for the limited sample available for analysis. Importantly, these recovery tests provide evidence of non-uniform behavior among individual aldopentoses, with variable recoveries (Table 1) and degradation patterns (Supplementary Fig. 10) under identical conditions, suggesting sugar-dependent interactions with the sample matrix and/or during analytical processing. This highlights the need for standardized protocols employing closely resembling meteorite matrices in procedural blanks to ensure accurate blank correction when quantifying extraterrestrial abundances and enantiomeric excesses, especially for inter–parent-body comparisons.
Aqueously altered amino acid signatures in Orgueil
We simultaneously analyzed amino acids in the same sample extract to (1) monitor potential biological contamination in the absence of isotopic measurements, (2) evaluate the efficiency of our modified methodology with previous analyses of Orgueil samples, and (3) contextualize sugar abundances relative to better-characterized organic molecules within the same fragment. Given the limited availability of sugar data in the literature, amino acids—abundant and well-documented in Orgueil (Table 2)—served as a valid reference point.
A total of 25 amino acids, including proteinogenic, rare, and non-biological types, were detected. The abundances of the identified amino acids are summarized in Table 2 and compared with previously reported data for Orgueil51,52. The concentrations of the most abundant amino acids, glycine and β-alanine, closely align with previous reports, reinforcing their status as characteristic amino acids in CI, CM1, and CR1 meteorites51,52. A similar agreement was observed for C3 and C4 amino acids, except for 4-ABA. However, the C5 amino acids were present at concentrations roughly ten times lower in our extract compared to those reported in previous studies. Our analysis also revealed several amino acids not previously identified in Orgueil, including four proteinogenic amino acids (threonine, proline, leucine, and isoleucine) and six rare amino acids (N-methylglycine, N-ethylglycine, 2,3-DAP, 2,4-DAB, norleucine, and allo-isoleucine).
The distribution of amino acid structural isomers in meteorites varies significantly across different groups, reflecting distinct parent body conditions and alteration histories12,49,53. Orgueil (CI1) samples display markedly lower total amino acid abundances compared to less aqueously altered CM2 (e.g., Murchison) and CR2-CR3 chondrites—a pattern attributed to extensive aqueous processing on their parent bodies54,55. This lower content is particularly pronounced for hydrophilic amino acids, likely resulting from destruction during prolonged aqueous alteration and analytical biases due to phyllosilicate-rich matrices (>90 vol.%), which trap amino acids and reduce extraction efficiencies55. The predominance of glycine and β-alanine aligns with CI chondrite alteration trends, suggesting formation via HCN polymerization and Michael addition in an ice-rich parent body containing ammonia, hydrogen cyanide, and cyanoacetylene52,53.
The enantiomeric ratios of selected chiral amino acids were calculated as l-enantiomeric excess (%eel) (Table 3) to evaluate potential contamination during the analysis. Alanine and aspartic acid showed rather small l-excesses, while other proteinogenic amino acids (serine and valine) exhibited significant l-excess. This aligns, however, with previous data from Orgueil16,51 (Table 3) where high l-excess for serine/aspartic acid (in unhydrolyzed samples) was reported. The non-proteinogenic amino acids 2-ABA and 3-AIB were found in nearly racemic proportions. Notably, isovaline exhibited minor l-excess, matching reported values for non-hydrolyzed samples51,52.
Discussion
Sugars are increasingly recognized as key prebiotic molecules owing to their central roles in biological systems. Although laboratory simulations suggest that sugars can form under astrophysically relevant conditions21,22,23,24,25,26,27, their inferred delivery to the early Earth is supported by only two observations to date: a report of ribose and related aldoses in meteorites19 and a recent detection in samples returned from asteroid Bennu20. Our detection of individual sugar enantiomers alongside amino acids in the Orgueil meteorite provides complementary evidence supporting this hypothesis. Despite its greater terrestrial exposure, Orgueil serves as a mineralogically close analog to carbonaceous asteorides Ryugu and Bennu, while offering a larger available mass. Previous results from the Hayabusa2 mission15,56,57,58,59 confirmed that asteroid (162173) Ryugu samples closely resemble CI (Ivuna-like) chondrites, with notable differences arising from their distinct alteration histories.
In the Orgueil (CI) chondrite, we identified five distinct sugars: arabinose, xylose, lyxose, ribose, and ribulose (Table 1). Ribulose is a C₅ ketose, and as a key intermediate in the formose reaction, it can isomerize to ribose, linking it directly to plausible prebiotic synthesis pathways60. Ribose, lyxose, and ribulose showed near-racemic distributions, consistent with an abiotic, extraterrestrial origin. The detection of racemic lyxose in particular strengthens earlier interpretations of its extraterrestrial provenance, given its rarity in biological systems19. By contrast, d-xylose and l-arabinose displayed significant enantiomeric excesses. While these can reflect terrestrial inputs, previous isotopic analyses in Murchison demonstrated that chiral sugar acids—such as xylonic, lyxonic, and ribonic acids—can exhibit large d-excesses61 despite being extraterrestrial in origin. This suggests that enantiomeric biases may be inherent in some abiotic synthesis pathways. Thus, the enantiomer distributions reported here should not be interpreted as definitive evidence of contamination only.
Quantitatively, arabinose concentrations in Orgueil were 2–3 orders of magnitude lower than those reported for CM2/CR2 meteorites19, whereas ribose and lyxose abundances were comparable. Xylose was less abundant than in Murchison, although a direct comparison remains limited due to the absence of reliable δ¹³C data. Given our modest recovery rates (1–30%), most likely due to strong interactions between sugars and the phyllosilicate-rich matrix36, the detected amounts clearly underestimate true sugar abundances. Therefore, the abundances reported here should be considered conservative estimates of the actual mass of extraterrestrial sugars. Moreover, our derivatization approach does not allow for the detection of hexoses like glucose. However, their presence—suggested by both interstellar ice analogs21,22,23,24 and planetary/mineral analogs25,26,27, cannot be excluded, and their detection may depend heavily on the analytical strategy used.
The accompanying chiral analysis of amino acids serves both as contamination control and as a contextual reference for the organic inventory of Orgueil. Near-racemic distributions in proteinogenic alanine and aspartic acid, as well as racemic ratios in rare amino acids such as 2-ABA and 3-AIB, support an abiotic origin for these compounds and, by extension, for the sugars detected. The amino acid profiles also provide insight into parent-body processes: their distributions reflect extensive low-temperature aqueous alteration, consistent with the phyllosilicate-, carbonate-, and ammonium-rich reservoirs characteristic of CI chondrites18,62,63. Such alteration likely occurred under mildly alkaline aqueous conditions (pH ~8–9)13,52 at temperatures below 150 °C. This mineralogical context is particularly relevant for understanding sugar formation and preservation. Although alkaline fluids can channel sugar precursors into degradation pathways, such as Maillard-type reactions that consume both amino acids and sugars64, they are also essential for driving sugar synthesis. This interplay encompasses: (1) formose-type reactions catalyzed on phyllosilicate surfaces in fluids enriched in Mg2+, Ca2+, and Fe2+ ions65,66; (2) base-catalyzed polymerization of interstellar aldehydes such as formaldehyde and glycolaldehyde25,26; and (3) stabilization through adsorption onto mineral surfaces or trapping within phyllosilicate interlayers36, which can mitigate degradation. Alternative formation routes include photochemical processes and non-photolytic cold-grain chemistry62 as shown in laboratory simulations of interstellar ice chemistries22,23,24.
Our combined enantiomeric analyses of sugars and amino acids thus provide a more comprehensive picture of the chiral organic inventory in primitive solar system materials. However, quantitative comparisons between these compound classes remain constrained by methodological factors—most notably, our low sugar recovery rates and the degradation of sugar derivatives in the presence of residual matrix material (Supplementary Fig. 10). These challenges complicate assessments of possible synthetic or competitive relationships between amino acids and sugars, as well as interpretations of whether the low reported sugar abundances reflect genuinely lower formation rates, enhanced degradation on the asteroid, or atmospheric entry losses relative to more stable meteoritic organics. Our data, together with the recovery experiments presented here, point strongly toward extraction inefficiencies rather than intrinsic rarity as the primary cause of the apparent scarcity of meteoritic sugars. Methanol, the solvent used in this study, extracts sugars poorly, and ongoing tests indicate that alternative solvents such as water, which is relevant to early Earth environments, substantially improve recovery. Thus, the low yields reported here should not be taken as evidence that sugars were unavailable for prebiotic chemistry. These findings highlight the need for improved extraction protocols and systematic recovery studies using representative matrices to more accurately constrain the true abundance and mobility of meteoritic sugars. We briefly note these limitations in the present work, and detailed experimental validation addressing these gaps is underway.
Crucially, this work demonstrates that successful detection and enantiomeric resolution of sugars are achievable using less than 200 mg of material—establishing Orgueil as a valuable analog and methodological test case for future analyses of returned asteroid samples such as those from Ryugu and Bennu. Optimization of recovery and derivatization protocols will further reduce the required sample mass, an essential consideration given the extreme rarity of these pristine extraterrestrial materials.
In summary, our results reveal a richer and more diverse sugar inventory in carbonaceous meteorites than previously recognized. However, our findings also underscore the importance of systematically assessing recovery efficiencies across representative mineral matrices to refine abundance estimates and to better resolve the diversity of chiral organics relevant to prebiotic chemistry. Ultimately, detecting indigenous sugar enantiomeric excesses consistent with biological homochirality would offer compelling evidence for an extraterrestrial contribution to life’s molecular handedness on early Earth67,68,69.
Methods
Sample preparation
A 178.3 mg fragment of the Orgueil meteorite, of a total of 1.0 g allocated by the Muséum national d’Histoire naturelle (MNHN) in Paris and stored in a plastic container (Supplementary Fig. 1), was analyzed in this study. The fragment was transferred into a pre-weighed 5 mL V-Vial (V-Vial® Wheaton) using clean tweezers inside a Class II laminar flow safety cabinet (Thermo Scientific MSC-Advantage Class II).
All glassware was cleaned with 96% ethanol and Milli-Q water, then wrapped in aluminum foil and heated at 500 °C for 5 h. Other labware, including needles, caps, spatulas, tweezers, PTFE frits (20 μm pore size, Supelco), and PTFE disposable liners (Supelco), were thoroughly cleaned with HPLC-grade ethanol and Milli-Q® water, dried with a stream of pure nitrogen, and wrapped in pre-burned aluminum foil until further use. Biopur tips (2–200 μL and 50–1000 μL Eppendorf epT.I.P.S. Biopur) were employed when needed.
Ultrasonic-assisted extraction
Ultrasonic extraction was performed using a Fisherbrand FB15049 (Elma Schmidbauer GmbH, Singen) at a fixed frequency of 37 kHz operating in sweep mode. Prior to extraction, the water bath was degassed for 1 to 5 min to remove entrapped air and dissolved gases. 3D printed holders were employed to fix the position of the vials containing the samples in a reproducible way. The temperature of the bath was maintained between 15 and 21 °C. For high-temperature extractions, an MS-100 Thermo Shaker incubator was employed. Additional instrumentation used for sample pre-treatment, including specifications and operating parameters, is detailed in the Supplementary Note 1.
The extraction protocol applied for analyzing the Orgueil sample is schematically illustrated in Supplementary Fig. 2. A procedural blank (PB), without a matrix, was prepared alongside the sample. Two distinct extracts were obtained: a methanolic (Sigma Aldrich, >99.9%) extract, expected to contain extracted sugars and minor quantities of amino acids, and an aqueous extract, primarily containing amino acids. Subsequently, multiple water (Milli‑Q Direct 8 apparatus, 18.2 MΩ cm at 25 °C, <2 ppb total organic carbon) extraction steps, including conventional hot-water extraction, were performed to enhance amino acid recovery. The detailed water extraction procedure for each step was as follows: 1. Cold methanol extraction (sugar fraction): The Orgueil fragment was crushed with 1.784 mL of methanol (MeOH) using a glass rod to ensure thorough solvent penetration. The sample was then vortex-mixed and sonicated for 10 min at 17–20 °C. After sonication, it was centrifuged at 9500 × g (9000 rpm) for 10 min with a Hettich Universal 320 centrifuge with an angle-fixed rotor model 1620 A, and the supernatant was transferred to a 25 mL glass bottle. This extraction process was repeated five times, with all extracts combined into the same 25 mL flask. To minimize the co-evaporation of volatile compounds during pre-concentration under low pressure, 5 mL of water was added to the methanol extracts. The sample was then stored in the fridge at 5 °C until subsequent purification and fractionation. 2a. Cold water extraction: After methanolic extraction, the extraction continued seven more times on the matrix residue, using water instead of methanol. The resulting supernatants were transferred into a separate 25 mL glass bottle. The sample was then stored in the fridge at 5 °C until subsequent purification and fractionation. 2b. Hot water extraction: Next, 3.6 mL of water was added to the 5 mL vial containing the meteorite residue for hot-water extraction at 100 °C for 21 h. The resulting extract was then centrifuged at 9500 × g and combined with the cold-water extracts from step 2a. 2c. Cold water extraction: Three additional cold-water extractions were performed on the remaining matrix using 1.784 mL of water per extraction. The resulting supernatants were combined with the previous aqueous extracts (2a and 2b) for purification and fractionation.
Ion exchange chromatography (IEC)
Purification and fractionation
The methanolic extracts (approximately 15 mL MeOH/H2O (2:1, v/v) were loaded onto the corresponding ion exchange chromatography (IEC) columns in 200 μL portions using a Pasteur pipette (Supplementary Note 1). Eluates were collected in a 25 mL pear-shaped flask. The 25 mL bottles containing the extracts were rinsed with 5 × 0.5 mL of H2O to optimize compound recovery. The rinses were then eluted through the columns and collected in the same 25 mL pear-shaped flask. The water extracts from both cold and hot extractions (approximately 21.5 mL) were subsequently eluted through the columns in 200 μL portions without prior acidification to prevent potential racemization of the α-amino acids (Section 1.5). One milliliter of water was used to wash the walls of the columns. The resulting eluate was collected in a separate 25 mL pear-shaped flask, as it was not expected to contain sugars or amino acids, the latter of which should remain retained onto the column. Finally, to recover the amino acid fraction, 18 × 1 mL of NH3(aq) 2 M were eluted through the column and collected in a third 25 mL pear-shaped flask.
Pre-concentration and drying
The resulting sugar and amino acid fractions from the Orgueil sample and the procedural blank without matrix were then pre-concentrated to a final volume of ~100 μL using a rotary evaporator. For this, the pressure was gradually reduced from 500 mbar to a final pressure of 25 mbar (25 °C) for the sugar fraction and 20 mbar for the amino acid fraction (26–27 °C). Each concentrated fraction was transferred to a separate 1 mL V-Vial, and the pear-shaped flasks were rinsed with 3 × 150 μL of H2O to recover any remaining analyte from the inner walls of the flasks. Finally, the samples were dried under a gentle flow of nitrogen before compound-specific derivatization. While the amino acid fraction could be dried to completion within approximately 3 h, the presence of matrix components in the sugar fraction delayed drying to up to 6 h and likely resulted in residual water remaining in the matrix (Supplementary Note 3).
Identification and quantification by enantioselective two-dimensional gas chromatography
Derivatization
The sugar fractions of the extracted samples and procedural blanks were derivatized into methylboronic acid esters following the protocol of Garcia et al.47. This involved adding 30 μL of a methylboronic acid (MBA) solution (10 mg mL–1 in pyridine) to the dried samples, vigorous vortex-mixing and heating at 60 °C for 30 min. After cooling for 10 min, the samples were then dried under a gentle stream of nitrogen, leaving approximately 2–3 μL of solvent to prevent the loss of volatile derivatives. Finally, the MBA derivatives were dissolved in 30 μL of ethyl acetate containing methyl laurate (ML) as an internal standard at a concentration of 10–6 M. Amino acids were derivatized into N-trifluoroacetyl-O-methyl (N-TFA-O-Me) esters following the protocol of Pepino et al.48. First, 120 μL of a methanol/acetyl chloride (MeOH/AcCl) solution (4:1, v/v) were added, and the mixture was vigorously stirred and heated at 110 °C for 1 h. After cooling for 10 min, the sample was dried under a gentle stream of nitrogen, leaving approximately 2 μL of solvent to prevent the loss of volatile derivatives. Next, 120 μL of a dichloromethane/trifluoroacetic acid anhydride (DCM/TFAA) solution (4:1, v/v) was added, stirred for 10 s, and heated at 100 °C for 20 min. The solution was then cooled and fully dried with nitrogen. Finally, the N-TFA-O-Me ester derivatives were dissolved in 30 μL of chloroform containing ML as an internal standard at a concentration of 10–6 M.
Analysis
The sugar and amino acid fractions from the Orgueil sample were analyzed using comprehensive two-dimensional gas chromatography coupled with reflectron time-of-flight mass spectrometry (GC×GC–TOF-MS) equipped with a dual-stage thermal jet modulator (Pegasus BT 4D, Leco Corp.). The injector and ion source temperatures were maintained at 230 °C, while the transfer line was set to 240 °C. Aliquots of 1 μL were injected in splitless mode. Hydrogen was used as carrier gas at a constant flow rate of 1.2 mL min–1. After each injection, the sample GC vial was immediately recapped and stored at 4 °C to prevent degradation of the amino acid and sugar derivatives before subsequent injections.
For sugar analysis, the GC×GC column configuration consisted of a CP-Chirasil-Dex CB primary column (27.15 m length × 0.25 mm inner diameter (ID), 0.25 μm film thickness, Agilent Technologies) coupled to a DB-WAX secondary column (1.61 m × 0.1 mm ID, 0.1 μm film thickness, Agilent Technologies). The primary oven was programmed as follows: an initial hold at 55 °C for 1 min, followed by a temperature increase of 10 °C min–1 to 90 °C, where it was held for 8 min. The temperature then increased at 2 °C min–1 to 190 °C, with a final isothermal hold at 190 °C for 15 min. The secondary oven and thermal modulator maintained constant positive temperature offsets of 30 °C and 15 °C, respectively. A modulation period of 5 s was applied. The reflectron TOF-MS operated at a 150 Hz storage rate, with a mass range of 50–500 amu and a microchannel plate detector voltage of approximately 2 kV and a solvent delay of 10 min.
For amino acid analysis, the column set-up included a Lipodex E primary column (24.95 m length × 0.25 mm inner diameter, Macherey-Nagel) coupled with a DB-WAX secondary column (1.7 m length × 0.12 mm inner diameter, 0.1 μm film thickness, Agilent). The primary oven was programmed as follows: an initial hold at 40 °C for 1 min, then heated to 80 °C at 10 °C min–1, holding for 10 min. The temperature was then increased to 120 °C at 1.3 °C min–1, followed by an increase to 152 °C at 0.8 °C min–1, and finally raised to 185 °C at 10 °C min–1, where it was held for 10 min. The secondary oven was operated with a constant positive temperature offset of 35 °C, and the thermal modulator hot jets had a temperature offset of 15 °C. The modulation period was 6 s. The reflectron TOF-MS operated at a 150 Hz storage rate, with a mass range of 40–500 amu, a microchannel plate detector voltage of approximately 2 kV, and a solvent delay of 9 min.
Analyte identification was performed by comparing retention times across both chromatographic dimensions and mass spectra (within a 500-ppm mass tolerance) with a mixed analyte standard. Quantification was performed using four-point calibration curves for the target analytes ranging from 5 × 10–8 M to 10–6 M, with manual integration conducted via LECO Corp. ChromaTOF™ software.
Data availability
The chromatographic and mass spectral data generated in this study using LECO Corp. ChromaTOF™ software (.peg format) are publicly available in the Zenodo repository at https://doi.org/10.5281/zenodo.1800271970.
References
Chyba, C. & Sagan, C. Endogenous production, exogenous delivery and impact-shock synthesis of organic molecules: an inventory for the origins of life. Nature 355, 125–132 (1992).
Pearce, B. K. D., Pudritz, R. E., Semenov, D. A. & Henning, T. K. Origin of the RNA world: The fate of nucleobases in warm little ponds. Proc. Natl. Acad. Sci. 114, 11327–11332 (2017).
Bland, P. A., Berry, F. J., Smith, T. B., Skinner, S. J. & Pillinger, C. T. The flux of meteorites to the Earth and weathering in hot desert ordinary chondrite finds. Geochim. Cosmochim. Acta 60, 2053–2059 (1996).
Pearce, B. K. D., Tupper, A. S., Pudritz, R. E. & Higgs, P. G. Constraining the time interval for the origin of life on Earth. Astrobiology 18, 343–364 (2018).
Pearce, B. K. D., Molaverdikhani, K., Pudritz, R. E., Henning, T. & Cerrillo, K. E. Toward RNAlife on early Earth: from atmospheric HCN to biomolecule production in warm little ponds. Astrophys. J. 932, 9 (2022).
Damer, B. & Deamer, D. The hot spring hypothesis for an origin of life. Astrobiology 20, 429–452 (2020).
Anders, E. Prebiotic organic matter from comets and asteroids. Nature 342, 255–257 (1989).
Kimura, H., Mann, I. & Jessberger, E. K. Composition, structure, and size distribution of dust in the local interstellar cloud. Astrophys. J. 583, 314 (2003).
Flynn, G. J., Keller, L. P., Jacobsen, C. & Wirick, S. An assessment of the amount and types of organic matter contributed to the Earth by interplanetary dust. Adv. Space Res. 33, 57–66 (2004).
Flynn, G. J., Keller, L. P., Wirick, S. & Jacobsen, C. Organic matter in interplanetary dust particles. Proc. Int. Astron. Union 4, 267–276 (2008).
Glavin, D. P., Burton, A. S., Elsila, J. E., Aponte, J. C. & Dworkin, J. P. The search for chiral asymmetry as a potential biosignature in our solar system. Chem. Rev. 120, 4660–4689 (2020).
Glavin, D. P. et al. Chapter 3 - The origin and evolution of organic matter in carbonaceous chondrites and links to their parent bodies. in Primitive Meteorites and Asteroids (ed. Abreu, N.) 205–271 https://doi.org/10.1016/B978-0-12-813325-5.00003-3 (Elsevier, 2018).
Martins, Z., Chan, Q. H. S., Bonal, L., King, A. & Yabuta, H. Organic matter in the solar system—implications for future on-site and sample return missions. Space Sci. Rev. 216, 54 (2020).
Oba, Y. et al. Identifying the wide diversity of extraterrestrial purine and pyrimidine nucleobases in carbonaceous meteorites. Nat. Commun. 13, 2008 (2022).
Naraoka, H. et al. Soluble organic molecules in samples of the carbonaceous asteroid (162173) Ryugu. Sci. 379, eabn9033 (2023).
Parker, E. T. et al. Extraterrestrial amino acids and amines identified in asteroid Ryugu samples returned by the Hayabusa2 mission. Geochim. Cosmochim. Acta 347, 42–57 (2023).
Oba, Y. et al. Uracil in the carbonaceous asteroid (162173) Ryugu. Nat. Commun. 14, 1292 (2023).
Glavin, D. P. et al. Abundant ammonia and nitrogen-rich soluble organic matter in samples from asteroid (101955) Bennu. Nat. Astron. 9, 199–210 (2025).
Furukawa, Y. et al. Extraterrestrial ribose and other sugars in primitive meteorites. Proc. Natl. Acad. Sci. 116, 24440–24445 (2019).
Furukawa, Y. et al. Bio-essential sugars in samples from asteroid Bennu. Nat. Geosci. 19, 19–24 (2026).
de Marcellus, P. et al. Aldehydes and sugars from evolved precometary ice analogs: Importance of ices in astrochemical and prebiotic evolution. Proc. Natl. Acad. Sci. 112, 965–970 (2015).
Nuevo, M., Cooper, G. & Sandford, S. A. Deoxyribose and deoxysugar derivatives from photoprocessed astrophysical ice analogues and comparison to meteorites. Nat. Commun. 9, 5276 (2018).
Meinert, C. et al. Ribose and related sugars from ultraviolet irradiation of interstellar ice analogs. Science 352, 208–212 (2016).
Zhang, C. et al. Ionizing radiation exposure on Arrokoth shapes a sugar world. Proc. Natl. Acad. Sci. 121, e2320215121 (2024).
Haas, M., Lamour, S., Christ, S. B. & Trapp, O. Mineral-mediated carbohydrate synthesis by mechanical forces in a primordial geochemical setting. Commun. Chem. 3, 1–6 (2020).
Vinogradoff, V. et al. Olivine-catalyzed glycolaldehyde and sugar synthesis under aqueous conditions: application to prebiotic chemistry. Earth Planet. Sci. Lett. 626, 118558 (2024).
Abe, S., Yoda, I., Kobayashi, K. & Kebukawa, Y. Gamma-ray-induced synthesis of sugars in meteorite parent bodies. ACS Earth Space Chem. 8, 1737–1744 (2024).
Hollis, J. M., Lovas, F. J. & Jewell, P. R. Interstellar glycolaldehyde: the first sugar. Astrophys. J. 540, L107 (2000).
Goesmann, F. et al. Organic compounds on comet 67P/Churyumov-Gerasimenko revealed by COSAC mass spectrometry. Science 349, aab0689 (2015).
Gilbert, W. Origin of life: the RNA world. Nature 319, 618–618 (1986).
Orgel, L. E. Prebiotic chemistry and the origin of the RNA world. Crit. Rev. Biochem. Mol. Biol. 39, 99–123 (2004).
Larralde, R., Robertson, M. P. & Miller, S. L. Rates of decomposition of ribose and other sugars: implications for chemical evolution. Proc. Natl. Acad. Sci. 92, 8158–8160 (1995).
Grew, E. S., Bada, J. L. & Hazen, R. M. Borate minerals and origin of the RNA world. Orig. Life Evol. Biosph. 41, 307–316 (2011).
Cooper, G. et al. Carbonaceous meteorites as a source of sugar-related organic compounds for the early Earth. Nature 414, 879–883 (2001).
Pearson, V. K. et al. Clay mineral-organic matter relationships in the early solar system. Meteorit. Planet. Sci. 37, 1829–1833 (2002).
Viennet, J.-C. et al. Interaction between clay minerals and organics in asteroid Ryugu. Geochem. Perspect. Lett. 25, 8–12 (2023).
Genge, M. J. et al. Rapid colonization of a space-returned Ryugu sample by terrestrial microorganisms. Meteorit. Planet. Sci. 60, 64–73 (2025).
Chan, Q. H. S., Stroud, R., Martins, Z. & Yabuta, H. Concerns of organic contamination for sample return space missions. Space Sci. Rev. 216, 56 (2020).
Sugita, S. et al. The geomorphology, color, and thermal properties of Ryugu: Implications for parent-body processes. Science 364, eaaw0422 (2019).
Yada, T. et al. Preliminary analysis of the Hayabusa2 samples returned from C-type asteroid Ryugu. Nat. Astron. 6, 214–220 (2022).
Dworkin, J. P. et al. OSIRIS-REx contamination control strategy and implementation. Space Sci. Rev. 214, 19 (2017).
Sephton, M. A. & Gilmour, I. Compound-specific isotope analysis of the organic constituents in carbonaceous chondrites. Mass Spectrom. Rev. 20, 111–120 (2001).
Elsila, J. E., Charnley, S. B., Burton, A. S., Glavin, D. P. & Dworkin, J. P. Compound-specific carbon, nitrogen, and hydrogen isotopic ratios for amino acids in CM and CR chondrites and their use in evaluating potential formation pathways. Meteorit. Planet. Sci. 47, 1517–1536 (2012).
Engel, M. H. & Macko, S. A. Isotopic evidence for extraterrestrial non-racemic amino acids in the Murchison meteorite. Nature 389, 265–268 (1997).
Pizzarello, S. & Huang, Y. The deuterium enrichment of individual amino acids in carbonaceous meteorites: a case for the presolar distribution of biomolecule precursors. Geochim. Cosmochim. Acta 69, 599–605 (2005).
Sessions, A. L. Isotope-ratio detection for gas chromatography. J. Sep. Sci. 29, 1946–1961 (2006).
Garcia, A. D., Leyva, V., Bocková, J., Pepino, R. L. & Meinert, C. Resolution and quantification of carbohydrates by enantioselective comprehensive two-dimensional gas chromatography. Talanta 271, 125728 (2024).
Pepino, R., Leyva, V., Garcia, A. D., Bocková, J. & Meinert, C. Resolution, quantification, and reliable determination of enantiomeric excess of proteinogenic and non-proteinogenic amino acids by comprehensive two-dimensional gas chromatography. J. Sep. Sci. 45, 4416–4426 (2022).
Burton, A. S., Stern, J. C., Elsila, J. E., Glavin, D. P. & Dworkin, J. P. Understanding prebiotic chemistry through the analysis of extraterrestrial amino acids and nucleobases in meteorites. Chem. Soc. Rev. 41, 5459–5472 (2012).
Simkus, D. N. et al. Methodologies for analyzing soluble organic compounds in extraterrestrial samples: amino acids, amines, monocarboxylic acids, aldehydes, and ketones. Life 9, 47 (2019).
Burton, A. S., Grunsfeld, S., Elsila, J. E., Glavin, D. P. & Dworkin, J. P. The effects of parent-body hydrothermal heating on amino acid abundances in CI-like chondrites. Polar Sci. 8, 255–263 (2014).
Glavin, D. P. & Dworkin, J. P. Enrichment of the amino acid l-isovaline by aqueous alteration on CI and CM meteorite parent bodies. Proc. Natl. Acad. Sci. 106, 5487–5492 (2009).
Ehrenfreund, P., Glavin, D. P., Botta, O., Cooper, G. & Bada, J. L. Extraterrestrial amino acids in Orgueil and Ivuna: tracing the parent body of CI type carbonaceous chondrites. Proc. Natl. Acad. Sci. 98, 2138–2141 (2001).
Takano, Y. et al. Primordial aqueous alteration recorded in water-soluble organic molecules from the carbonaceous asteroid (162173) Ryugu. Nat. Commun. 15, 5708 (2024).
Glavin, D. P., Callahan, M. P., Dworkin, J. P. & Elsila, J. E. The effects of parent body processes on amino acids in carbonaceous chondrites. Meteorit. Planet. Sci. 45, 1948–1972 (2010).
Yokoyama, T. et al. Samples returned from the asteroid Ryugu are similar to Ivuna-type carbonaceous meteorites. Science 379, eabn7850 (2022).
Okazaki, R. et al. Noble gases and nitrogen in samples of asteroid Ryugu record its volatile sources and recent surface evolution. Science 379, eabo0431 (2022).
Nakamura, T. et al. Formation and evolution of carbonaceous asteroid Ryugu: Direct evidence from returned samples. Science 379, eabn8671 (2022).
Yabuta, H. et al. Macromolecular organic matter in samples of the asteroid (162173) Ryugu. Sci. 379, eabn9057 (2023).
Breslow, R. On the mechanism of the formose reaction. Tetrahedron Lett. 1, 22–26 (1959).
Cooper, G. & Rios, A. C. Enantiomer excesses of rare and common sugar derivatives in carbonaceous meteorites. Proc. Natl. Acad. Sci. 113, E3322–E3331 (2016).
Laize-Générat, L. et al. Nitrogen in the Orgueil meteorite: Abundant ammonium among other reservoirs of variable isotopic compositions. Geochim. Cosmochim. Acta 387, 111–129 (2024).
Yoshimura, T. et al. Chemical evolution of primordial salts and organic sulfur molecules in the asteroid 162173 Ryugu. Nat. Commun. 14, 5284 (2023).
Garcia, A. et al. Molecular diversity and amino acid evolution in simulated carbonaceous chondrite parent bodies. ACS Earth Space Chem. 8, 606–615 (2024).
Brearley, A. J., Binzel, R. P., Walker, R. M. & Cameron, A. G. W. The action of water. in Meteorites and the Early Solar System II. (eds Lauretta, D. S. & McSween, H. Y.) 587–624 https://doi.org/10.2307/j.ctv1v7zdmm.35 (University of Arizona Press, 2006).
Alexander, C. M. O., Cody, G. D., De Gregorio, B. T., Nittler, L. R. & Stroud, R. M. The nature, origin and modification of insoluble organic matter in chondrites, the major source of Earth’s C and N. Geochemistry 77, 227–256 (2017).
Bocková, J., Garcia, A. D., Jones, N. C., Hoffmann, S. V. & Meinert, C. Chiroptical properties of membrane glycerophospholipids and their chiral backbones. Chirality 36, e23654 (2024).
Bocková, J., Jones, N. C., Topin, J., Hoffmann, S. V. & Meinert, C. Uncovering the chiral bias of meteoritic isovaline through asymmetric photochemistry. Nat. Commun. 14, 3381 (2023).
Meinert, C. et al. Photonenergy-controlled symmetry breaking with circularly polarized light. Angew. Chem. Int. Ed. 53, 210–214 (2014).
Meinert, C. GC×GC–TOF-MS data from Orgueil carbonaceous chondrite. zenodo. https://doi.org/10.5281/zenodo.18002719.
Acknowledgments
This study was supported by the European Research Council under the European Union’s Framework Program for Research and Innovation HORIZON Europe under ERC grant 101170457–ICE-EEVOLVE (C.M.) and the Simone and Cino Del Duca Foundation – Institut de France (C.M.). We gratefully acknowledge the Muséum national d’Histoire naturelle, Paris, for providing the Orgueil meteorite sample used in this study (C.M.).
Author information
Authors and Affiliations
Contributions
C.M. designed the experiments and directed the overall project. V.L. and M.R. performed the extraction and GC×GC–TOF‑MS analyses. R.P. and J.B. assisted with the experiments. V.L., J.B., and C.M. wrote the manuscript and Supplementary Information. All authors reviewed and edited the manuscript and Supplementary Information.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Leyva, V., Robert, M., Pepino, R. et al. Abiotic sugar enantiomers in the CI carbonaceous chondrite Orgueil. Nat Commun 17, 2060 (2026). https://doi.org/10.1038/s41467-026-68709-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68709-5