Abstract
Visceral pain represents an unmet clinical need due to the lack of safe and generalizable therapies. Given the analgesic potential of music and the underlying crosstalk between auditory and nociceptive pathways, this study aims to delineate the neural circuits interfacing auditory processing and visceral pain modulation. Using a mouse model of early-life stress, we identify a cortico-cortical circuit linking the primary auditory cortex (Au1) to the anterior cingulate cortex (ACC) that mediates stress-induced visceral pain. Mechanistically, early-life stress suppresses the activity of Au1 inhibitory (GABAergic, Au1GABA) neurons, which disinhibits downstream ACC excitatory (glutamatergic, ACCGlu) neurons, thereby driving visceral hypersensitivity. Notably, music intervention alleviates visceral pain by restoring Au1GABA neuronal activity and suppressing ACCGlu activity. In conclusion, this study unveils the Au1-ACC circuit as a critical hub for stress-induced visceral hypersensitivity and music-induced analgesia, providing a mechanistic foundation for clinical translation of music therapy in visceral pain management.
Similar content being viewed by others
Introduction
Visceral pain represents a debilitating clinical manifestation associated with a variety of gastrointestinal disorders, affecting more than 20% of the global population and reducing their quality of life1. Despite three decades of research on functional gastrointestinal disorders, particularly irritable bowel syndrome (IBS) characterized by visceral hypersensitivity, the underlying pathogenesis remains incompletely understood, largely due to the inherent heterogeneity of these conditions. Therefore, current therapeutic options are limited and narrowly targeted, such as guanylate cyclase-C agonists linaclotide and plecanatide, 5-HT4 receptor agonist tegaserod for constipation-predominant IBS (IBS-C), opioid receptor agonist eluxadoline, 5-HT3 receptor antagonist alosetron, and antibiotic rifaximin for diarrhea-predominant IBS (IBS-D)2,3. The limitations of these medications are attributed to their adverse side effects and single-target mechanisms, which consequently restrict their applicability to only a small subpopulation of IBS patients. For instance, alosetron was withdrawn due to severe constipation and ischemic colitis; although reintroduced later, its use is strictly limited to female IBS-D patients4. This highlights a significant lack of safe and generalizable treatments for visceral pain management.
The Rome IV criteria introduced in 2016 redefined functional gastrointestinal disorders as disorders of gut-brain interaction, reflecting the evolving understanding of their neurobiological underpinnings. Emerging research now explores central neuromodulation approaches, such as sound, light, electrical and magnetic interventions, in attempts to modulate pain perception. Music therapy has been widely applied to improve mental health, cognitive function and physical wellbeing, given its cost-effectiveness, ease of use and lack of side-effects. Although clinical studies have demonstrated the efficacy of music intervention in alleviating various forms of pain5,6, the underlying neurobiological mechanisms remain elusive, and its specific effects on visceral pain are yet to be revealed.
Sensory hypersensitivity represents a common clinical manifestation across various neuropsychiatric disorders such as autism spectrum disorder, post-traumatic stress disorder, fibromyalgia, sensorineural hearing loss and migraine7. Notably, striking neurological parallels have been noticed between auditory and nociceptive hypersensitivity. Tinnitus (auditory perception occurring without external sounds) and hyperacusis (increased sensitivity to acoustic stimuli) are highly analogous to allodynia and hyperalgesia in pain hypersensitivity. Intriguingly, both auditory and pain hypersensitivity can be triggered by stress8,9,10,11, and a clinical correlation between tinnitus and chronic pain has been documented12,13. Furthermore, IBS patients exhibit hyperactivity to auditory stimuli14,15, and conversely, IBS is one of the comorbid conditions of hyperacusis16. This neurobiological convergence implies potential crosstalk between the auditory pathway and the visceral pain pathways. The primary auditory cortex (Au1), serving as the central hub for auditory processing, exhibits functional connectivity across the brain networks to regulate diverse physiological functions. Notably, clinical and preclinical evidence has revealed functional and neuroanatomical disturbance in the auditory cortex of depression patients and preclinical chronic mild stress model17, implying its unexplained role in stress response integration.
This study employed an early-life stress model, neonatal colonic inflammation (NCI), to recapitulate stress-induced visceral hypersensitivity in IBS, and aimed to investigate the role of Au1 in stress response and visceral pain modulation, and to identify the neural circuits linking auditory processing and visceral pain modulation. The findings of this study will shed light on the neurobiological mechanisms underlying cross-modal visceral pain modulation, and expand current therapeutic strategies for visceral pain management.
Results
NCI-induced suppression of Au1GABA neurons drives visceral hypersensitivity
The involvement of Au1 neurons in somatosensory representation remains unclear. An electrophysiological study indicates that Au1 neurons barely respond to tactile stimulation alone, with only approximately 11.6% responding to combined auditory and tactile stimuli18, suggesting limited involvement of the Au1 in direct processing of somatosensory inputs. To determine whether Au1 neurons directly encode visceral nociceptive stimuli, we used in vivo fibre photometry recording to assess their calcium responses to CRD stimuli. In agreement with prior research, our findings revealed that both Au1GABA and Au1Glu neurons exhibited minimal responsiveness to CRD (Fig. 1A–D), suggesting a marginal engagement of the Au1 in direct encoding of visceral pain.
A Experimental setup for fibre photometry recording. Summarized changes in calcium fluorescence (ΔF/F) showing minimal responsiveness to CRD at 60 mmHg in both Au1GABA (B) and Au1Glu (C). A brief cat meow stimulus was used as a positive control. The red and blue scale bars denote the 1-second duration of stimulation. D Quantitative comparisons of area under the curve of the ΔF/F indicate lower calcium responses to CRD compared to cat meow (F1,10 = 56.280, ***p < 0.0001, two-way ANOVA with Bonferroni test, n = 6) on both Au1GABA (**p = 0.018) and Au1Glu (***p = 0.0003). E A schematic showing the timeline of NCI model and EMG recording. F Representative EMG recording traces showing visceral pain responses to CRD on adolescent CON and NCI mice. G NCI significantly increased EMG responses to CRD (F1,16 = 8.496, *p = 0.010, two-way ANOVA with Bonferroni test, n = 9), particularly at 60 (*p = 0.010) and 80 mmHg (**p = 0.006). H Representative images showing c-Fos immunofluorescence in the Au1 of adolescent CON and NCI mice, scale bar = 100 µm. I–K NCI group exhibited increased total c-Fos+ neurons (***p = 0.0005, Mann–Whitney test, n = 47 sections from 6 CON mice and 44 sections from 6 NCI mice), fewer c-Fos++GABA+ neurons (*p = 0.026, unpaired t-test, n = 23 sections from 6 CON and 19 sections from 6 NCI mice) and more c-Fos++Glu+ neurons (***p = 0.0005, unpaired t-test, n = 23 sections from 6 mice in each group). L–M The ratio of c-Fos++GABA+ neurons in c-Fos+ neurons in the Au1 of NCI group was significantly reduced compared to CON (***p = 0.0003, unpaired t-test, n = 23 and 19 sections from 6 CON and NCI mice respectively), while there was no significant difference in the ratio of c-Fos++Glu+ in c-Fos+ neurons (p = 0.119, unpaired t-test, n = 23 sections from 6 mice). Representative fluorescent images showing co-localization of c-Fos with GABA (N) or Glu (O), scale bar = 50 µm. Yellow arrows indicate co-localization. P Experimental design of optogenetic manipulation of Au1GABA and EMG recordings, scale bar = 100 µm. Q, S Representative EMG responses to CRD. R, T Blue light exposure significantly reduced EMG responses to CRD in NCI mice (R, F1,5 = 99.150, ***p = 0.0002, two-way ANOVA with Bonferroni test, n = 6) at 20 (***p = 0.0002), 40 (*p = 0.014), 60 (***p = 0.0002) and 80 mmHg (***p < 0.0001) but not CON mice (T, p = 0.296, F1,6 = 1.309, two-way ANOVA with Bonferroni test, n = 7). U–V Experimental design of chemogenetic inhibition of Au1GABA on naïve mice and representative image of AAV expression, scale bar = 100 µm. W Chemogenetic inhibition of Au1GABA by CNO (3.33 mg/kg, IP injection) attenuated visceral pain threshold since day 1 after injection (F1,9 = 41.600, ***p = 0.0001, two-way ANOVA with Bonferroni test, n = 6 for hM4Di-mCherry and 5 for mCherry). EMG traces in this and all subsequent figures depict the EMG responses during the entire 10-second CRD stimulation. All data are presented as means ± SEM.
An early-life stress model, NCI (Fig. 1E), was employed to investigate the role of Au1 in stress response integration and visceral pain modulation. Compared to CON mice, visceral pain responses to CRD stimulation were significantly increased in adolescent NCI mice (Fig. 1F&G). To examine the role of Au1 in stress-induced visceral hypersensitivity, spontaneous neuronal activity in the Au1 from adolescent NCI and CON mice was assessed using c-Fos immunofluorescence. The number of c-Fos+ neurons in the Au1 of NCI group was significantly higher than CON group (Fig. 1H&I). To further identify the neuronal subtypes involved in this change, c-Fos colocalization with neuronal marker gamma-aminobutyric acid (GABA, Fig. 1N) or glutamate (Glu, Fig. 1O) was quantified. NCI mice exhibited fewer c-Fos++GABA+ neurons (Fig. 1J) and more c-Fos++Glu+ neurons (Fig. 1K) in the Au1. The proportion of GABAergic neurons in c-Fos+ neurons was decreased in NCI mice (Fig. 1L), while the proportion of glutamatergic neurons was not significantly changed (Fig. 1M), suggesting that Au1GABA may be more vulnerable to early-life stress.
To further determine the role of Au1GABA in stress-induced visceral hypersensitivity, optogenetic and chemogenetic techniques based on AAV vectors were utilized to selectively manipulate the excitability of Au1GABA (Fig. 1P&U). Blue light-induced activation of Au1GABA significantly attenuated visceral pain responses in NCI mice (Fig. 1Q&R) but not CON mice (Fig. 1S&T). In addition, chemogenetic inhibition of Au1GABA on naïve mice significantly reduced visceral pain threshold since Day 1 after CNO injection (Fig. 1U-W). These data suggest that suppression of Au1GABA caused by early-life stress drives visceral hypersensitivity.
Mapping the downstream nuclei of Au1 in visceral pain modulation
Although the Au1 contributed minimally to the encoding of visceral pain, it may shape pain perception by tuning relevant neural network through its projections. Indeed, a recent study has demonstrated that the auditory cortex dampens somatic pain via its descending projections to the thalamus19. To map the downstream projections of Au1, antegrade tracing was performed with AAV2/9-hSyn-EGFP and density of EGFP fluorescence was quantified using ImageJ software (Fig. 2A–C). Axons originating from the Au1 were observed throughout the brain, with prominent innervation in the auditory pathways (medial geniculate nucleus, MGB; inferior colliculus, IC), cerebral cortex (temporal association cortex, TeA; secondary visual cortex lateral area, V2L; contralateral Au1; ACC; insular cortex, ICx;), basal ganglia (caudate putamen, CPu; basolateral amygdaloid nucleus, BLA; claustrum, Cl) and thalamus (ventral posterior thalamic nucleus, VP; posterior thalamic nucleus, PO). Some of these regions have been reported to be implicated in visceral pain modulation by our previous studies, such as the ACC, Cl, ICx, and BLA20,21,22. Among them, the ACC received relatively more extensive projections from the Au1 (Fig. 2B–C). Furthermore, monosynaptic anterograde tracing using a Cre-DIO system revealed direct projections from the Au1 to the ACC (Fig. 2D). Therefore, we focused on the ACC as a presumed downstream effector of Au1-mediated visceral pain modulation.
A–C Anterograde tracing from the Au1 using AAV revealed axons across representative coronal sections spanning the brain, scale bar = 1 mm. EGFP-labelled regions were roughly grouped by projection routes depicted by the arrows, with the thickness of arrows indicating the magnitude of fibre tracts. Both the colours and sizes of the circles denote EGFP density. Representative images of the AAV injection site in the Au1 and downstream projections in the ACC were shown in panel C, n = 3, scale bar = 100 µm. D Monosynaptic anterograde tracing using a Cre-DIO system revealed direct projections from the Au1 to the ACC, n = 3, scale bar = 100 µm. Representative fluorescent images showing co-localization of CRD-induced c-Fos expression with glutamate (E) or GABA (F) in ACC, scale bar = 50 µm. Yellow arrows indicate co-localization. G CRD-induced c-Fos in the ACC predominantly colocalized with glutamate (***p < 0.0001, unpaired t-test, n = 18 sections for Glu and 10 sections for GABA, both from 6 mice). H Setup of optogenetic manipulation of ACCGlu in combination with EMG recording, scale bar = 100 µm. I–K Blue light-induced activation of ACCGlu in CON mice increased visceral pain responses (F1,5 = 35.320, **p = 0.002, two-way ANOVA with Bonferroni test, n = 6) at 20 (**p = 0.005), 40 (**p = 0.004), 60 (***p = 0.001) and 80 mmHg (**p = 0.002). L–N Yellow light-induced inhibition of ACCGlu in NCI mice significantly reduced the visceral pain responses (F1,4 = 29.920, **p = 0.005, two-way ANOVA with Bonferroni test, n = 5) at 40 (**p = 0.004) and 80 mmHg (***p = 0.0006). ACC, anterior cingulate cortex; Au1, primary auditory cortex; AuD, secondary auditory cortex dorsal area; AuV, secondary auditory cortex ventral area; BLA, basolateral amygdaloid nucleus; cc, corpus callosum; Cl, claustrum; Contra-Au1, contralateral primary auditory cortex; cp, cerebral peduncle; CPu, caudate putamen; DRN, dorsal raphe nucleus; Ect, ectorthinal cortex; Ent, entorhinal cortex; IC, inferior colliculus; ICx, insular cortex; LD, laterodorsal thalamic nucleus; LPB, lateral parabrachial nucleus; M2, secondary motor cortex; MGB, medial geniculate nucleus; NLL, nucleus of the lateral lemniscus; NTB, nucleus of the trapezoid; OC, orbital cortex; PAG, periaqueductal grey; PnC, pontine reticular nucleus caudal part; PO, posterior thalamic nucleus; PrL, prelimbic cortex; RMC, red nucleus magnocellular part; S1, primary somatosensory cortex; S2, secondary somatosensory cortex; SC, superior colliculus; SOC, superior olivary complex; TeA, temporal association cortex; V1B, primary visual cortex binocular area; V2L, secondary visual cortex lateral area; VCA, ventral cochlear nucleus; VP, ventral posterior thalamic nucleus. All data are presented as means ± SEM.
ACCGlu neurons encode visceral hypersensitivity in NCI mice
Colocalization analysis indicated that CRD-activated ACC neurons were predominantly glutamatergic (Fig. 2E–G). To examine the functional role of ACCGlu neurons in visceral hypersensitivity, they were optogenetically activated or inhibited in CON and NCI mice, respectively (Fig. 2H). Blue light-induced activation of ACCGlu in CON mice significantly increased visceral pain responses (Fig. 2I–K). Conversely, yellow light-induced inhibition of ACCGlu in NCI mice significantly mitigated visceral pain responses (Fig. 2L–N). In addition, optogenetic inhibition of ACCGlu in CON mice did not change visceral pain responses significantly (Fig. S2A–C). These results collectively demonstrate that ACCGlu neurons play a crucial role in visceral hypersensitivity.
Au1GABA neurons mediate visceral analgesia through enhanced GABAergic output to ACCGlu in synergy with attenuated glutamatergic transmission via Au1Glu
As we have demonstrated the essential role of Au1GABA and ACCGlu in stress-induced visceral hypersensitivity, we further investigated the involvement of the Au1GABA-ACCGlu neural circuitry. Monosynaptic retrograde tracing using RV was performed to identify presynaptic inputs to ACCGlu (Fig. 3A&B). Colocalization analysis of RV+ neurons in the Au1 identified approximately 53.8% as glutamatergic neurons and 28.5% as GABAergic (Fig. 3C–E), suggesting that both Au1GABA and Au1Glu projected to ACCGlu. While emerging evidence has documented the existence of cortico-cortical long-range GABAergic projections, their functional significance remains inadequately explored. To functionally validate our observation from RV tracing, in vivo extracellular recording using optical electrode arrays was performed to record neural activity in conjunction with optogenetic manipulation of Au1GABA projections in the ACC (Fig. 3F). 11 in 71 recorded ACC neurons responded to blue light stimulation with their firing rate attenuated by optogenetic activation of Au1GABA axons in the ACC. This effect was abolished in the presence of a GABAA receptor antagonist bicuculline (1 mg/kg, IP injection, Fig. 3G–H), confirming that the inhibitory effect of blue light was mediated via GABAA receptors and thus indicating the functional existence of GABAergic projections from the Au1 to the ACC. To examine which pathway contributed to modulation of visceral pain, optogenetic manipulation was conducted targeting axonal terminals of Au1GABA and Au1Glu in the ACC, respectively. Optogenetic activation of Au1GABA axonal terminals projecting to the ACC alleviated visceral pain responses in NCI mice (Fig. 3I-K). Interestingly, optogenetic inhibition of axonal terminals in the ACC originating from Au1Glu also mitigated visceral pain responses in NCI mice (Fig. 3L-N). These data suggest that Au1GABA and Au1Glu modulate visceral pain synergistically via downstream projections to the ACC.
A Schematic illustration of RV monosynaptic retrograde tracing to identify presynaptic inputs to ACCGlu. B Representative images showing the injection site in the ACC and input cells in the Au1, n = 3, scale bar = 100 µm. C–E Colocalization analysis of RV+ neurons in the Au1 revealed more glutamatergic than GABAergic neurons (C, ***p = 0.0002, unpaired t-test, n = 22 sections for Glu and 21 sections for GABA, both from 3 mice), scale bar = 50 µm. Yellow arrows indicate co-localization. F Experimental setup of in vivo extracellular recording using optical electrode arrays in NCI mice. G Representative raster diagrams and firing rate of an ACC neuron responsive to blue light. H Blue light attenuated firing rate in a proportion of ACC neurons, which was abolished by a GABAA receptor antagonist bicuculline (1 mg/kg, IP injection, **p = 0.001, one-way ANOVA with Bonferroni test, n = 10 neurons from 3 NCI mice). I–K Optogenetic activation of Au1GABA axonal terminals in the ACC alleviated visceral pain responses in NCI mice (K, F1,5 = 36.710, **p = 0.002, two-way ANOVA with Bonferroni test, n = 6) at 40 (*p = 0.032), 60 (*p = 0.023), and 80 mmHg (***p = 0.0002), scale bar = 100 µm. L–N Optogenetic inhibition of Au1Glu axonal terminals in the ACC significantly alleviated visceral pain responses in NCI mice (N, F1,5 = 8.941, *p = 0.030, two-way ANOVA with Bonferroni test, n = 6) at 60 (*p = 0.030) and 80 mmHg (***p < 0.0001), scale bar = 100 µm. O–S With optogenetic inhibition of Au1GABA projections in the ACC, administration of GABAA receptor agonist muscimol (1 mM, 1 μL) into the Au1 alleviated visceral pain responses in NCI mice (S, pink symbols: **p = 0.004, **p = 0.003,***p = 0.0001, two-way ANOVA with Bonferroni test, n = 7), while chemogenetic activation of Au1Glu reversed the analgesic effect of muscimol (blue symbols: *p = 0.033, **p = 0.005). Scale bar = 100 µm. All data are presented as means ± SEM.
It has been well documented that inhibitory neurons form reciprocal connections with local excitatory neurons in the auditory cortex and shape their response dynamic23. Given the potential involvement of local microcircuit in the Au1, we combined optogenetics and chemogenetics to examine the functional role of Au1GABA-Au1Glu-ACCGlu tripartite circuit in visceral pain modulation (Fig. 3O-P). Au1GABA axonal terminals in the ACC were suppressed by optogenetic inhibition (Fig. 3Q). A GABAA receptor agonist muscimol was infused into the Au1 to mimic activation of Au1GABA24. Following optogenetic blockade of direct Au1GABA signalling to the ACC, local administration of muscimol alleviated visceral pain responses in NCI mice, while chemogenetic activation of Au1Glu reversed the analgesic effect of muscimol (Fig. 3R&S), implying that muscimol-induced analgesia was mediated through Au1Glu pathway. In vivo extracellular recording was performed to demonstrate that Au1Glu neural activity was inhibited by muscimol and reversed by chemogenetic activation of Au1Glu (Fig. S3A-C). Collectively, these findings suggest that local microcircuits in the Au1 are involved in visceral pain modulation via both enhanced GABAergic output and reduced glutamatergic output to the ACC.
Music alleviates visceral hypersensitivity in NCI mice by enhancing Au1GABA neuronal activity
As the impact of music intervention on visceral pain remains unclear, we examined the analgesic potential of various music genres as well as white noise. Our findings revealed that classical music and electronic music, but not rock music, significantly increased visceral pain threshold in NCI mice (Fig. S4A–F), suggesting that varying acoustic parameters (such as the slower tempo of the rock music used here, as shown in Table S1) may influence their analgesic effects. To further investigate whether tempo is a critical parameter, we digitally modified the tempo of above-used classical and rock music. As a result, slower classical music lost its analgesic effect, whereas faster rock music acquired analgesic effect (Fig. S4H–I). Intriguingly, white noise also elevated visceral pain threshold (Fig. S4E), suggesting that this analgesic effect might be independent of musical structure. In addition, filtered low-frequency classical music (retaining <1 kHz components which fall below mouse’s hearing range of 1–100 kHz) failed to produce analgesia (Fig. S4C), indicating the necessity of the auditory pathways for this analgesic effect. Comparative quantification across all acoustic interventions identified classical music as the most potent analgesic intervention (Fig. S4G); consequently, classical music was used in subsequent experiments unless otherwise stated. To examine whether music induces stress responses in mice, elevated plus maze and open field test were performed to assess the effect of music on anxiety-like behaviours. Compared to CON mice, NCI mice exhibited reduced entries into the open arm, total distance travelled and mean speed, whereas a brief music intervention (15 min) did not alter these anxiety-like behaviours (Fig. S5A–H). In addition, 15-min music intervention did not change serum corticosterone levels (Fig. S5I–J).
Consistent with elevated visceral pain threshold, music significantly attenuated EMG responses to graded CRD stimulation in NCI mice (Fig. 4A–C), while had no effect on CON mice (Fig. 4D–E). To assess the effect of music on Au1 neural activity, in vivo extracellular recording was performed using microelectrode arrays (Fig. 4F). Compared to CON mice, spontaneous firing rate in the Au1 was elevated in NCI mice, and music attenuated Au1 neural activity in NCI mice only (Fig. 4G–H). Given our findings of suppressed Au1GABA activity in NCI mice (Fig. 1L), the impact of music on Au1GABA was assessed using immunofluorescence. Quantitative analysis revealed that music significantly increased the proportion of GABA+ neurons among c-Fos+ neurons in the Au1 of NCI mice (Fig. 4N) while concurrently reducing the ratio of Glu+ neurons in c-Fos+ neurons (Fig. 4O), indicating restoration of the excitation-inhibition balance. This finding aligns with the overall suppression of neural activity during music exposure observed in above electrophysiological experiment. Although music increased activation of both GABAergic and glutamatergic neurons in CON animals (Fig. 4L–M), their equilibrium was not altered (Fig. 4N–O). In addition, neither the NCI stress nor music intervention altered the density of GABA+ neurons and Glu+ neurons (Fig. S6). To functionally verify the role of Au1GABA in music-induced visceral analgesia, AAV vectors with or without yellow light-sensitive eNpHR3.0 were injected into the Au1 of NCI mice (Fig. 4P). In NCI mice injected with control virus, yellow light did not affect music-induced visceral analgesia (Fig. 4Q–R). On the contrary, in NCI mice infected with AAV containing eNpHR, silencing of Au1GABA by yellow light abolished music’s analgesic effect (Fig. 4S–T). These data suggest that music attenuates visceral hypersensitivity by enhancing Au1GABA activity.
A–E Music significantly reduced visceral pain responses to CRD in NCI mice (B&C, F1,9 = 9.943, *p = 0.012, two-way ANOVA with Bonferroni test, n = 10), particularly at 60 (**p = 0.001) and 80 mmHg (***p < 0.0001), but had no effect on CON mice (D&E, F1,9 = 4.448, p = 0.064, two-way ANOVA with Bonferroni test, n = 10). F–G Schematic illustration of in vivo extracellular recording using microelectrode arrays and representative neural activity in the Au1 of NCI and CON mice. H Spontaneous firing rate in the Au1 was increased in NCI mice, compared to CON mice (F1,127 = 6.546, ***p < 0.0001, two-way ANOVA with Bonferroni test, n = 56 neurons from 3 CON mice and 73 neurons from 4 NCI mice. Music attenuated firing rate in NCI mice (F1,127 = 34.430, ***p < 0.0001) but not CON mice (p > 0.999). Spontaneous firing rate was averaged using 20 sec time windows. Representative images showing co-localization of c-Fos with GABA (I) or glutamate (J) in NCI mice with or without music intervention, scale bar = 50 µm. Yellow arrows indicate co-localization. K Music increased the total number of c-Fos+ neurons in the Au1 of both NCI (***p = 0.0008) and CON mice (***p < 0.0001, unpaired t-test, n = 37 sections for CON, 42 for CON+Music, 35 for NCI, 32 for NCI+Music, each including 6 mice). L Music increased the number of c-Fos++GABA+ neurons in the Au1 of both groups (**p = 0.003, ***p <0.0001, unpaired t-test, n = 23, 25, 20 and 22 sections from 6 mice in CON, CON+Music, NCI, and NCI+Music groups, respectively). M Music increased the number of c-Fos++Glu+ neurons in the Au1 of CON group (**p = 0.002, unpaired t-test, n = 13 and 17 sections from 6 mice in CON and CON+Music groups), but had no significant effect in NCI mice (p = 0.940, unpaired t-test, n = 13 and 12 sections from 6 mice in NCI and NCI+Music groups). N Music increased the ratio of c-Fos++GABA+ in c-Fos+ neurons in the Au1 of NCI group (*p = 0.019, unpaired t-test), while had no effect on CON mice (p = 0.752, same n values as L). O Music significantly reduced the ratio of c-Fos++Glu+ in c-Fos+ neurons in the Au1 of NCI group (***p < 0.0001, unpaired t-test) but not CON mice (p = 0.073, same n as M). P Experimental design of optogenetic inhibition of Au1GABA, scale bar = 100 µm. Q–T In NCI mice injected with control AAV, yellow light did not affect music-induced visceral analgesia (R, F1,4 = 98.460, ***p = 0.0006, two-way ANOVA with Bonferroni test, n = 5). In NCI mice injected with AAV containing eNpHR, yellow light abolished the analgesic effect of music intervention (T, F1,4 = 0.205, p = 0.674, two-way ANOVA with Bonferroni test, n = 5). Data are presented as means ± SEM.
Music induces visceral analgesia in NCI mice by suppressing ACCGlu activity
The effect of music on ACC neuronal activity was assessed using c-Fos immunofluorescence. Compared to CON mice, NCI mice exhibited more prominent CRD-induced c-Fos expression in the ACC, which was effectively attenuated by music intervention in NCI but not CON mice (Fig. 5A&B). To further investigate the contribution of ACCGlu neurons to music-induced visceral analgesia, calcium responses of ACCGlu to CRD were assessed using in vivo fibre photometry (Fig. 5C&D). Music significantly attenuated calcium responses to CRD in ACCGlu, as evidenced by reduced AUC and peak of ΔF/F (Fig. 5E–H), whereas no significant effect was observed in CON mice. In agreement with calcium signals, ACC neuronal firings in response to CRD were significantly attenuated by music (Fig. 5I–K). Furthermore, the role of ACCGlu in music-induced visceral analgesia was verified by optogenetic activation of ACCGlu in EMG recordings (Fig. 5L). In NCI mice infected with the control virus lacking blue light-sensitive hChR2, music alleviated CRD-evoked visceral pain responses (Fig. 5M&N). Conversely, in NCI mice injected with AAV vectors containing hChR2, blue light activation of ACCGlu abolished music-induced visceral analgesia (Fig. 5O&P). These observations suggest that music mediates visceral analgesia by suppressing the hyperactivity of ACCGlu.
A Representative c-Fos immunofluorescence in the ACC from CON and NCI mice with or without CRD stimulation and music intervention, scale bar = 100 µm. B CRD significantly increased c-Fos expression in the ACC from both CON (**p = 0.004, one-way ANOVA with Bonferroni test, n = 10, 21, 12, 22, 15 and 25 sections from 6 mice in CON, CON + CRD, CON + CRD+Music, NCI, NCI + CRD, CON + CRD+Music groups respectively) and NCI mice (***p < 0.0001), and this increase was more pronounced in NCI mice (**p = 0.001). In addition, music intervention reduced CRD-induced c-Fos expression in NCI mice (***p < 0.0001) but not CON mice (p > 0.999). C–D A schematic showing the timeline and setup of in vivo fibre photometry recording and representative image of AAV expression in the ACC, scale bar = 100 µm. E–F Heatmaps and summarized changes in fluorescence (ΔF/F) showing calcium responses to CRD of 20 or 60 mmHg in ACCGlu of CON and NCI mice. (G) Music reduced the AUC of ΔF/F in response to 60 mmHg CRD in NCI mice (***p = 0.0004, two-way ANOVA with Bonferroni test, n = 6), while no significant difference was observed at 20 mmHg (p > 0.999) and CON mice (p = 0.088, n = 8). H Music attenuated the peak ΔF/F in response to 60 mmHg CRD in NCI mice (***p = 0.0002, two-way ANOVA with Bonferroni test, n = 6), while no significant difference was observed at 20 mmHg (p > 0.999) and CON mice (p = 0.302, n = 8). I–J Experimental setup of in vivo extracellular recording using microelectrode arrays and representative ACC neural activity in response to 60 mmHg CRD before and during music intervention in NCI and CON mice. K CRD responses of ACC neurons were increased in NCI mice, compared to CON mice (F1,49 = 0.844, *p = 0.024, two-way ANOVA with Bonferroni test, n = 24 neurons from 4 CON mice and 27 neurons from 4 NCI mice. Music attenuated CRD responses in NCI mice (F1,49 = 7.012, ***p = 0.0001) but not CON mice (p = 0.642). CRD responses were calculated as the mean firing rate during the 10-sec CRD stimulation period subtracted by the mean firing rate during the 10-sec baseline prior to CRD. L Timeline and setup of optogenetic activation of ACCGlu in NCI mice, scale bar = 100 µm. M–N In NCI mice injected with control virus, blue light did not affect music-induced alleviation in visceral pain responses (F1,6 = 24.680, **p = 0.003, two-way ANOVA with Bonferroni test, n = 7) at 20 (**p = 0.008), 40 (***p = 0.0007), 60 (***p = 0.0008) and 80 mmHg (***p = 0.0002). O–P In NCI mice injected with AAV containing hChR2, optogenetic activation of ACCGlu abolished music-induced visceral analgesia (F1,4 = 0.151, p = 0.717, two-way ANOVA with Bonferroni test, n = 5). All data are presented as means ± SEM.
Music induces visceral analgesia in female NCI mice by modulating the Au1-ACC circuit
To ascertain whether the sex of mice influences music’s analgesic effect on visceral hyperalgesia, we replicated the NCI protocol in female mice (Fig. S7A). As in males, female NCI mice exhibited lower visceral pain threshold than their CON littermates, and music intervention elevated these thresholds in female NCI but not CON mice (Fig. S7C). Furthermore, chemogenetic activation of ACCGlu neurons receiving Au1 input reversed music’s analgesic effect (Fig. S7C). Taken together, music alleviates visceral pain in female mice by modulating the same Au1-ACC circuit as males.
Au1-ACC circuit mediates music-induced somatic analgesia
While our findings thus far have demonstrated that music therapy mitigates visceral pain through modulation of the Au1-ACC circuit, a pivotal question remains: whether this is selective for the visceral pain modality. To strengthen the translational relevance of our study, we adopted the Complete Freund’s Adjuvant (CFA)-induced hind-paw inflammatory pain model for further investigation of somatic pain. We first compared the analgesic dynamics of acute and chronic music interventions between the two modalities. A single 15-min bout of classical music exposure increased visceral pain threshold in NCI mice, and this effect sustained more than 60 min (Fig. 6A); whereas identical acute music intervention increased paw withdrawal threshold in CFA mice after intervention, which diminished within 30 minutes (Fig. 6C). Chronic paradigm consisted of 3 daily sessions of 15-min classical music exposure, with pain thresholds assessed prior to music intervention in the first 3 days. Visceral analgesia emerged on Day 2 and 3 (Fig. 6B), whereas somatic analgesia was only present on day 1 (Fig. 6D). Although the two pain modalities may not be directly comparable, our observations suggest that music appears to produce a more sustained analgesic effect on visceral pain than somatic pain.
A A single 15-min bout of classical music increased visceral pain threshold in NCI mice immediately after intervention, and this effect peaked at approximately 30 min and diminished until 120 min (F1,9 = 30.41, ***p = 0.0004, two-way ANOVA with Bonferroni test, n = 6 for the music group and 5 for the ambient group). B Chronic paradigm consisted of 3 daily sessions of 15-min classical music exposure. Two consecutive sessions were sufficient to produce sustained visceral analgesia in the absence of ongoing music intervention (F1,36 = 19.58, ***p < 0.0001, two-way ANOVA with Bonferroni test, n = 6 and 5 for the music and ambient group). C Identical acute music intervention increased paw withdrawal threshold in CFA mice immediately after intervention but this effect diminished within 30 minutes (F1,12 = 2.769, *p = 0.029, two-way ANOVA with Bonferroni test, n = 7 for both groups). D The analgesic effect of chronic music intervention on somatic pain was observed only on day 1 (F1,12 = 5.744, *p = 0.042, two-way ANOVA with Bonferroni test, n = 7). This experiment commenced on day 1 after CFA injection. E Experimental design for chemogenetic activation of Au1GABA combined with von Frey test on mice injected with CFA or saline. F Representative image of AAV expression in the Au1, scale bar = 100 µm. G Chemogenetic activation of Au1GABA neurons increased paw withdrawal threshold in the CFA group (F1,14 = 8.161, *p = 0.012, two-way ANOVA with Bonferroni test, n = 10) but not the Saline group (p = 0.291, n = 6). H Experimental design for chemogenetic activation of ACCGlu neurons receiving Au1 projections. I Representative expression in the ACC, scale bar = 100 µm. J Music increased paw withdrawal threshold in CFA mice (F2,20 = 10.41, *p = 0.011, two-way ANOVA with Bonferroni test, n = 6), which was reversed by chemogenetic activation of ACCGlu neurons innervated by the Au1 (*p = 0.021). Music had no effect on saline-injected mice (p > 0.999, n = 6), while CNO decreased their paw withdrawal thresholds (*p = 0.017). K Experimental timeline for CRD and von Frey test on NCI + CFA model. L Music increased visceral pain threshold on NCI + CFA mice (F1,14 = 20.12, ***p < 0.0001, two-way ANOVA with Bonferroni test, n = 9 for NCI + CFA mice and 7 for CON + CFA mice) but not CON + CFA mice (p > 0.999). M Music elevated paw withdrawal threshold on both NCI + CFA mice (F1,13 = 13.58, *p = 0.034, two-way ANOVA with Bonferroni test, n = 8 for NCI + CFA mice and 7 for CON + CFA mice) and CON + CFA mice (*p = 0.034). All data are presented as means ± SEM.
To investigate the involvement of Au1GABA neurons and the Au1-ACC circuit in somatic pain modulation, chemogenetic approaches were used to manipulate Au1GABA neurons and ACCGlu neurons receiving Au1 projections (Fig. 6E&H). Chemogenetic activation of Au1GABA neurons mimicked the analgesic effect of music in CFA mice (Fig. 6G). Consistently, chemogenetic activation of ACCGlu neurons innervated by the Au1 reversed music’s analgesic effect (Fig. 6J). These data suggest that the Au1-ACC circuit also mediates music-induced somatic analgesia.
To determine whether music possesses analgesic efficacy in animals subjected to combined early-life and adult stress, CFA injection was performed on NCI and CON mice at 6 weeks of age (Fig. 6K). Only the NCI + CFA group exhibited visceral hyperalgesia, which was relieved by music therapy (Fig. 6L). Conversely, music attenuated somatic pain in both NCI + CFA and CON + CFA mice (Fig. 6M). Taken together, these data suggest that music exerts analgesic efficacy across different pain modalities.
Discussion
Music intervention exhibits the potential as a safe and generalizable therapeutic option required for the clinical management of visceral pain, yet its application has been hindered by insufficient mechanistic understanding of the crosstalk between the auditory and the visceral pain pathways. This study unravels a cortico-cortical Au1-ACC neural circuit, comprising parallel GABAergic and glutamatergic projections, as the critical interface underpinning early-life stress-induced visceral hypersensitivity and music-induced visceral analgesia (Fig. 7). Our findings reveal that early-life stress evokes the excitation-inhibition imbalance in the Au1, leading to hyperactivity of the ACC through attenuated Au1 GABAergic output and enhanced glutamatergic output, thereby driving visceral hypersensitivity. Notably, music intervention rectifies this imbalance in the Au1, and effectively alleviates visceral pain by suppressing the hyperactivity of the ACC. Moreover, the Au1-ACC circuit also mediates music-induced analgesia in somatic pain. These findings highlight a pivotal role of the Au1 in stress integration and pain modulation, and provide mechanistic insights into cross-modal pain modulation and a theoretical basis for clinical application of music therapy in pain management.
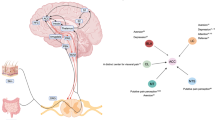
This study discovers an Au1-ACC neural circuit integrating both GABAergic and glutamatergic projections, functioning as a bridge linking stress response/auditory processing with visceral pain modulation. Early-life stress disrupts the balance between GABAergic and glutamatergic neuronal activity in the Au1, thereby inducing visceral hypersensitivity by driving hyperactivity in the ACC. Remarkably, music intervention restores the excitation-inhibition balance in the Au1, and effectively alleviates visceral pain by enhancing Au1 GABAergic output and suppressing glutamatergic output to the ACC. The interconnecting structure between the Au1 and the ACC is metaphorically represented by the silhouette of the Zhaozhou Bridge, the world’s oldest open-spandrel segmental arch bridge of stone construction, still standing after over 1400 years. GABA: gamma-aminobutyric acid; Glu: glutamate.
Non-auditory functions of Au1 in stress integration and pain modulation
Emerging evidence indicates non-auditory functions of the Au1, such as regulation in multimodal sensory integration, stress responses and mood disorders17,24,25. Notably, our findings demonstrate that early-life stress-induced visceral hypersensitivity in adolescence correlates with suppressed activity of Au1GABA neurons, and chemogenetic inhibition of Au1GABA is sufficient to drive visceral hypersensitivity in naïve mice. It has been well-documented that inhibitory interneurons in the auditory cortex shape the response dynamics of local excitatory neurons23,26. Physiological and psychological stress may create a neuroendocrine environment that elicits dysfunctions selectively in metabolically vulnerable cell types, particularly GABAergic neurons27. Consistent with this theory, restraint stress impairs GABAergic synaptic efficacy28 and the inhibitory gating29 in rat auditory cortex. In line with our observation, another study using a different early-life stress model, neonatal maternal separation, uncovers age-dependent alterations in excitation-inhibition balance in the infralimbic cortex, particularly reduced spontaneous GABAergic synaptic events in adulthood30. Imbalance in excitation and inhibition may be closely associated with several mental disorders including depression, autism, schizophrenia and epilepsy; moreover, this imbalance within the auditory cortex has been implicated in hearing loss, tinnitus and hyperacusis31,32. Collectively, these converging lines of evidence position the Au1 as a pivotal hub in stress response integration and visceral pain modulation, thereby expanding its role beyond canonical auditory processing.
Notably, in CON mice, neither optogenetic activation of Au1GABA, inhibition of ACCGlu, nor music therapy altered visceral pain responses. As discussed above, GABAergic neurons may be more vulnerable to stress; and consistently, we observed impaired Au1GABA neuronal activity in NCI mice relative to CON mice. As such, it may be inferred that spontaneous activity of Au1GABA in CON mice already imposes potent suppression over local and downstream excitatory neurons, leaving little room for further inhibition. Therefore, the optogenetic manipulation mentioned above or music therapy exhibits little analgesic effect. By contrast, this inhibitory brake is compromised in NCI mice, and the same interventions are capable of restoring the excitation-inhibition balance and achieving visceral analgesia. Furthermore, this selective relief of visceral pain in NCI but not CON mice offers a plausible explanation for why music therapy benefits only a subset of patients in the clinic.
The involvement of Au1 neurons in somatosensory encoding remains unclear. Electrophysiological evidence suggests that Au1 neurons barely respond to tactile stimulation alone, while approximately 11.6% of these neurons respond to combined auditory and tactile stimulation, likely contributing to multisensory integration18. This aligns with the natural occurrence of multisensory events, where most auditory events are accompanied by non-auditory signals, which may regulate auditory perception in a context-dependent manner33. Our in vivo fibre photometry recordings revealed minimal responsiveness of Au1 neurons to CRD stimulation, implying negligible contribution of Au1 neurons to direct encoding of visceral pain. Yet, a recent study has shown that auditory cortex dampens somatic pain via its projections to the thalamus19. Taken together, while Au1 contributes minimally to pain perception, it may shape the nociception by tuning network activity through its projections.
An Au1-ACC neural circuit underlying cross-modal visceral pain modulation
While previous anatomical investigations have implied potential connections between the Au1 and the ACC19,34, this study has provided comprehensive anatomical and functional characterization of this cortico-cortical circuit. We have demonstrated that this circuit, comprising parallel GABAergic and glutamatergic projections from the Au1 to the ACC, mediates early-life stress-induced visceral hypersensitivity and music-induced visceral analgesia. GABAergic neurons are traditionally thought to mediate local inhibition, whereas emerging evidence suggests that they also extend long-range axons to remote cortical and subcortical areas35, and in some cortical regions, GABAergic projections may be distributed more widely over ipsilateral neocortical areas than excitatory projections36,37. It has been reported that somatostatin (SOM)-positive neurons in the auditory cortex send long-range GABAergic projections to the lateral amygdala38. In addition, parvalbumin (PV)- and SOM-positive long-range GABAergic projections of the auditory cortex extend to the striatum39,40. Intriguingly, our findings have revealed that the Au1GABA-ACCGlu pathway and the Au1GABA-Au1Glu-ACCGlu pathway mediate the regulation of visceral pain by stress or music intervention in a synergistic manner, possibly implying the complexity and sophistication of the nervous system in terms of enhancing regulatory efficiency, enabling dynamic fine-tuning, and maintaining redundancy for evolutionary fitness. Given that inhibitory interneurons form reciprocal connections not only with the excitatory neurons, but also with each other23, local microcircuits in the Au1 may function in a highly intricate manner. Nevertheless, this study has provided preliminary insights into the mechanistic involvement of cortical microcircuits in cross-modal visceral pain modulation.
As a pivotal region in pain processing, the ACC is involved in the maintenance of pain hypersensitivity41. In line with studies on somatic pain, our previous work has elucidated the important role of the ACC in neonatal maternal deprivation-induced visceral hypersensitivity20, thereby corroborating the functional importance of the Au1-ACC circuitry in visceral pain modulation proposed herein. Consistently, an fMRI study with a small sample size has provided preliminary evidence that music exposure attenuates pain-related responses in the ACC42. On the other hand, the ACC may send reciprocal projections to the Au1. Emerging evidence indicates that the ACC enhances auditory responses through direct projections to the auditory cortex to facilitate flight behaviour43. Taken together, functionally reciprocal connections exist between the Au1 and the ACC, likely underpinning cross-modal interactions and forming part of a large-scale brain network.
The analogy between auditory hypersensitivity and pain hypersensitivity may lie in many aspects, including shared etiological factors, overlapping neurological mechanisms and frequent clinical comorbidity. Notably, tinnitus is correlated with altered excitation-inhibition balance with decreased inhibition and unleashed excitation in the Au132, resembling our observation in stress-induced visceral hypersensitivity herein. An fMRI study has revealed that the connectivity between the Au1 and rostral ACC is negatively correlated with tinnitus awareness44. These converging findings suggest that hypersensitivity in different modalities may be closely correlated through interconnected neural networks, and dysregulation in these networks may represent a shared pathogenesis across various neuropsychiatric conditions such as autism, fibromyalgia, migraine and IBS. It has been reported that abnormal brain networking may contribute to multimodal sensory hypersensitivity in fibromyalgia45. In addition, the salience network, a large-scale brain network primarily comprising the anterior insula cortex and ACC, is implicated in the evaluation of the importance of internal or external stimuli and the recruitment of relevant functional networks46. An fMRI study suggests that sensory hypersensitivity in patients with autism spectrum disorders is related to increased resting-state functional connectivity between salience network nodes and brain regions implicated in primary sensory processing, with the strength of this connectivity correlating with the extent of brain activity in response to auditory and tactile stimuli47. Our findings extend the insights of a recent report identifying the thalamus as a bridge for auditory-somatosensory processing19 to the cortical level, providing preliminary evidence towards understanding the intricate cross-modal interactions within large-scale neural networks.
Music effectively alleviates visceral and somatic pain
Music therapy has been widely used across various fields primarily due to its non-invasive nature, low cost and lack of side effects. Despite discrepancies in the literature regarding music-induced analgesia, extensive clinical evidence supports the efficacy of music in relieving various forms of pain5,6, although the lack of mechanistic understanding impedes its application. Until recently, a preclinical study revealed that attenuated glutamatergic output from the auditory cortex to the PO and VP nuclei in the thalamus mediate the analgesic effect of music and white noise on complete Freund’s adjuvant (CFA)-evoked inflammatory pain in mouse paws19. Our findings advance the insights of this report by providing an important mechanistic complement that music potentiates the activity of Au1 GABAergic neurons, which in turn suppresses local glutamatergic output. Nevertheless, the efficacy of music on visceral pain and underlying mechanisms remains unexplored. Our findings have demonstrated that music effectively alleviates visceral hypersensitivity in a mouse model of IBS via the Au1-ACC circuit, offering a complementary therapeutic strategy for clinical management of visceral pain. Intriguingly, we observed differential analgesic efficacy of various music genres, implying the relevance of acoustic parameters (frequency spectra, tempo, and timbre). Notably, given that we also observed visceral analgesia caused by white noise, the effectiveness of acoustic interventions in rodents may be independent of musical structure, in agreement with the recent study mentioned above which found no difference in analgesic effect between consonant music, dissonant music and white noise. In addition, low-pass filtered classical music (retaining <1 kHz components which fall below mouse’s hearing range of 1-100 kHz48) loses its analgesic effect, implying the necessity of functional auditory pathways. In line with this phenomenon, we observed increased neuronal activity in the Au1 from mice exposed to music, aligning with prior reports in rat49 and mouse50 auditory cortex. However, a conflicting report failed to replicate white noise-induced analgesia in a mouse CFA model51. This study neither specified the frequency range of the auditory intervention used nor validated whether such intervention altered neuronal activity of the auditory pathways, raising questions about the adequacy of neuronal engagement for potential neuromodulatory effects. Moreover, activation of the auditory cortex by acoustic stimuli is plastic, depending on the physiological state and the behavioural context of the receiving animals52. Furthermore, some clinical studies suggest that patient-selected music is critical for the efficacy of music therapy42,53. Additionally, music predictability, the listeners’ ability to anticipate upcoming events in a musical piece, is closely related to its rewarding effects54.
Given the well-established role of the ACC in both somatic and visceral pain modulation, the Au1-ACC neural circuit may not be specific to pain modalities. Indeed, supplementary studies on CFA-induced inflammatory pain model have revealed that music also alleviates somatic pain through the same Au1-ACC circuit. Thus, the circuit identified here appears to constitute a shared neural substrate for music therapy across diverse pain modalities and possibly other neuropsychiatric diseases, underscoring its broad and generalizable translational potential.
While some evidence suggests that mice exhibit music preference at early developmental stages (postnatal days 15-24)55 or when experiencing inflammatory pain19, it remains unresolved whether mice perceive and appreciate music in a manner comparable to humans. Given the more intricate neuroanatomical organization of human auditory fields in comparison to rodents, the processing of music in humans may be more context-dependent, involving cognition, emotion, and predictability components56. Future studies should verify the translatability of mechanistic insights derived from rodent studies. In addition, the anxiolytic effect of music has been well-documented in both clinical settings57,58 and rodent studies59,60, which typically requires multiple sessions of intervention. While the current study focused on the acute analgesic effect of music intervention, we observed no significant changes in anxiety-like behaviours in NCI mice following a single session of 15-min intervention. However, repeated music exposure may exert an analgesic effect via alleviating anxiety. Consistently, our anterograde tracing results revealed projections from the Au1 to the BLA, a pivotal neural hub for anxiety regulation. This potential mechanism warrants thorough investigation in future studies.
To sum up, this study reveals a cortico-cortical circuit comprising parallel GABAergic and glutamatergic projections from the Au1 to the ACC; through these projections, stress induces visceral hypersensitivity, while music intervention exerts analgesic effects. These findings not only elucidate the non-auditory functions of the Au1 in stress response integration and pain regulation but also provide mechanistic insights into auditory-pain crosstalk, paving the way for neuromodulatory interventions in the clinical management of pain.
Methods
Animals and ethical approval
Male and female C57BL/6 J mice were purchased from the Charles River Laboratories (Beijing, China), and housed at the Experimental Animal Centre of Soochow University under standard conditions (12-h light/dark cycle, temperature of 23–25 °C and humidity of 40–60%) with free access to food and water. Mice were given one week to acclimate to the new housing environment prior to any procedures. Male and female mice used in all experiments were 6–10 weeks old and weighed 20–25 g. All experiments were approved by the Animal Care Committee of Soochow University, and conducted in accordance with the guidelines of the International Association for the Study of Pain.
Neonatal colonic inflammation
An early-life stress model, NCI, was used to induce chronic visceral pain as described previously21. Briefly, on postnatal day 10, male or female mouse pups were randomized into NCI and CON groups, and injected with 30 μL of 0.5% acetic acid (diluted in saline) or equal volume of saline, respectively, into the colorectum 1 cm from the anus. Subsequent experiments commenced when the animals reached 6 weeks of age.
Abdominal withdrawal reflex (AWR)
AWR in response to colorectal distension (CRD) was used to evaluate visceral pain threshold21. A homemade balloon (1 cm in length) was lubricated with vaseline and gently inserted into the colorectum of anesthetized mice, with the end of the balloon positioned 0.5 cm from the anus. The catheter of the balloon was securely attached to the base of mouse’s tail, and mice were then placed under transparent acrylic restrainers to acclimate for 1 h. During the AWR test, the balloon catheter was connected to a sphygmomanometer, and the balloon was slowly inflated at a rate of 5 mmHg/sec until a sign of visceral pain (abdominal lifting or back arching) was observed or until a maximum pressure of 40 mmHg was reached. This procedure was repeated five times with 5-min intervals. The readings of distension pressure from the sphygmomanometer were recorded and averaged to determine the colorectal visceral pain threshold.
Electromyography (EMG) recording of visceromotor responses
Electrodes were implanted one week prior to EMG recording. After deep anesthesia induced by isoflurane, the abdominal skin of the mouse was carefully incised to expose underlying muscle tissue. Two electrode wires were implanted into the external oblique muscles on both sides of the abdomen, and were threaded subcutaneously to exit at the neck. The surgical incision was subsequently closed with 5-0 silk suture. Following 1-week recovery, mice were acclimatized in a restrainer for 1 h with a balloon inserted into the colorectum as described above. EMG activity was recorded via MP160 Data Acquisition System and digitized using AcqKnowledge software (BIOPAC Systems Inc., Goleta, CA, USA). EMG responses to graded CRD stimulation (20, 40, 60 and 80 mmHg in duplicate) were examined, with each distension lasting 10 s and a 3-min rest period in between. EMG responses to CRD were analysed as the area under the curve (AUC) of EMG activity during distension (10 s) subtracted by the AUC of baseline immediately prior to distension.
Acoustic interventions
A variety of music genres were chosen from the literature, including classical music (Mozart’s Sonata for Two Pianos, K.44861), rock music (“Still Loving You” by the Scorpions62) and electronic music (“Breathe” by the Prodigy63). In addition, radio noise (static between radio stations) was used as white noise64. All acoustic interventions were transposed up by one octave (the frequency of each note is doubled) using GarageBand (Version 10.4.7, Apple Inc., Cupertino, CA, USA) in order to better fit the hearing range of mice (1–100 kHz48). Detailed acoustic parameters and frequency distribution of interventions are shown in Table S1 and Fig. S1. All interventions were delivered via a portable audio player on a constant loop with sound pressure level at 50–55 dB in an environment with ambient noise level at 40-45 dB. Additionally, a brief cat meow (75 dB) stimulus was used as a positive control in fibre photometry.
Immunofluorescence
Mice were anaesthetized with xylazine (10 mg/kg, IP injection) and perfused transcardially with saline followed by 4% paraformaldehyde (PFA) in PBS. The brain specimens were carefully removed, post-fixed overnight in 4% PFA and dehydrated successively in 20% and 30% sucrose at 4 °C. The brains were embedded in OCT and sectioned into 30 μm-thick coronal slices using a Leica CM3050 S Cryostat (Leica Biosystem, Nussloch, Germany). These slices were collected onto adhesive slides (CITOTEST 188105) and stored at −20 °C. For immunofluorescence, the slices were rinsed three times with PBS (each lasting 10 min) and incubated in a blocking buffer (7% normal donkey serum and 0.3% Triton X-100 in PBS) for 1 hour at room temperature. The slices were then incubated with primary antibodies (1:300 dilution) at 4 °C for 24 h. Following three-time PBS washes, the sections were incubated with secondary antibodies (1:500 dilution) for 1 h at room temperature in the dark. After three additional PBS washes, the sections were mounted in a medium containing DAPI (Abcam, AB104139). Images were acquired using a fluorescent microscope (BX51, Olympus Corporation, Tokyo, Japan). For quantitative assessment and colocalization analysis, putative cell fragments and dim fluorescence not in the focal plane were excluded. When assessing c-Fos immunofluorescence following CRD or music stimulation, transcardiac perfusion was performed 90 min post-stimulation. Specifically, CRD was applied at 60 mmHg pressure for 5 times, each lasting 20 s separated by 3-min intervals; classical music was played continuously for 30 min following the same protocol as established in other experiments. To enhance the colorblind-friendliness of the fluorescent images, red-green contrast was adjusted to red-cyan or magenta-green colour schemes where necessary.
Stereotaxic surgery
Stereotaxic surgery was performed as previously described65,66. Mice were anesthetized with xylazine (10 mg/kg, IP injection), and firmly secured in a stereotaxic apparatus (RWD, 71000-M, Shenzhen, China). Following disinfection, a precise incision was made in the skull to expose the targeted brain regions (Au1 coordinates: AP −2.6 mm, ML −4.15 mm, DV −2.35 mm; ACC coordinates: AP 1.1 mm, ML − 0.35 mm, DV − 1.7 mm). Using a microsyringe pump (Legato 130, KD Scientific, Holliston, MA, USA), 200 nL of virus was injected into the targeted regions at a flow rate of 25 nL/min. The needle remained in situ for 15 min to ensure optimal diffusion. All viral injections were performed unilaterally in the right hemisphere of the mouse brain unless otherwise stated. The adeno-associated virus (AAV) and rabies virus (RV) were purchased from BrainVTA (as listed in Table S2).
For fibre photometry recordings and optogenetic manipulation, a ceramic insert (O.D. 200 μm, NA 0.37, length 2.5 mm, Thinker Tech Nanjing Bioscience, Nanjing, China) was implanted into the desired region, ensuring that the tip of the optical fibre was situated 100–200 μm above the viral injection site. This ceramic insert was stabilized on the skull using a cranial nail and dental cement. For local drug administration in the brain, a guide cannula (O.D. 0.48 mm, I.D. 0.34 mm, length 2.5 mm, RWD, Shenzhen, China) was implanted into the desired region. To prevent blockage, a mating cannula (O.D. 0.30 mm, I.D. 0.14 mm, RWD, Shenzhen, China) was inserted into the guide cannula.
Neural tracing
Anterograde tracing was performed via stereotaxic injection of AAV2/9 or AAV2/1 into targeted regions. Monosynaptic retrograde tracing using RV required a two-step injection process: AAV helpers were first injected to express RV glycoprotein and tumour virus A (TVA) receptors, followed by injection of RV-ΔG 3 weeks later. Images of entire coronal slices were acquired by a slide scanner (SLIDEVIEW VS200, Olympus Corporation, Tokyo, Japan). Fluorescence density was analysed using ImageJ software (Version 1.53k, National Institutes of Health, Bethesda, MD, USA). Each experiment was repeated on 3–5 mice.
Fibre photometry recording
In vivo fibre photometry was utilized to record calcium signals of selected neurons using the fluorescent calcium indicator GCaMP6s, which was delivered to targeted regions via AAV vectors. Three weeks after AAV injection and optical fibre implantation, GCaMP6s was excited by a 470-nm laser, with emitted fluorescence captured using a fibre photometry recording system (ThinkerTech Nanjing Bioscience Inc., Nanjing, China). Calcium responses to brief CRD or acoustic stimuli in conscious mice were analysed based on changes in fluorescence (ΔF/F) using custom MATLAB scripts (MATLAB R2017b, MathWorks, Natick, MA, USA). ΔF/F over the 2 s prior to the stimulation (−2 to 0 s) served as the baseline, and ΔF/F during 5 s after the onset of stimulation (0 to 5 s) was used to assess the responses.
Optogenetic and chemogenetic manipulation
To enable optogenetic manipulation of neuronal excitability, targeted neurons were engineered to express light-sensitive proteins via stereotaxic injection of AAV vectors 3 weeks prior to experiments. Humanized channelrhodopsin-2 (hChR2) and enhanced Natronomonas pharaonis halorhodopsin 3.0 (eNpHR3.0) can be activated by blue or yellow light to depolarize or hyperpolarize neurons, respectively. A laser source (Optogenetics-LED-STSI, Prizmatix Ltd., Holon, Israel) connected to an AlphaLab SnR Stimulation and Recording system (Alpha Omega Engineering, Nazareth Illit, Israel) was employed to deliver 5 mW yellow light (589 nm, continuous) or 3 mW blue light (473 nm, 20 Hz, 10 ms pulse width). Yellow light was continuously applied 15 min prior to CRD tests and sustained until the completion of graded CRD stimulation protocol (20, 40, 60 and 80 mmHg in duplicate); in contrast, blue light was synchronized with each 10-s distension only. Similarly, for chemogenetic manipulation, genetically engineered Designer Receptors Exclusively Activated by Designer Drugs (DREADDs) were delivered to targeted regions via AAV vectors 3 weeks prior to experiments. Human M3 muscarinic DREADD receptor coupled to Gq (hM3Dq) and human M4 muscarinic DREADD receptor coupled to Gi/o (hM4Di) were activated by the chemical ligand CNO (3.33 mg/kg, IP injection, CNO-02, BrainVTA). Visceral pain was assessed 30 minutes after CNO injection.
Microelectrode Array
In vivo neural activity was recorded using microwire electrode arrays and CereCube High-Throughput Neural Signal Acquisition System (NeuroXess, Shanghai, China). Electrodes were implanted into the targeted brain regions by stereotaxic surgery as described above. The mice were allowed to recover for one week after surgery. On the day of recording, the mice were briefly anesthetized by isoflurane inhalation and placed in an acrylic restrainer with their electrodes connected to an amplifier headstage (OMNETICS 2409). Neural signals were amplified, processed by a CereCube NSP1 processor, and recorded on a PC running MindExplorer software (Version 1.5.6, NeuroXess) with sampling rate at 30000 Hz. Spike sorting was performed offline based on V.seek algorithm using MindExplorer. CRD responses were calculated as mean firing frequency over the 10-sec distension period subtracted by the mean firing rate of the 10-s prior to distension. Spontaneous firing rate was averaged using 20 s time windows. Adjustable microwire electrode arrays (KD-MWA-AD-16, Kedou Brain-Computer Technology Co., Ltd., Suzhou, China) were used to record neural activity of the Au1 and the ACC. To record neural activity in conjunction with optogenetic manipulation, optrodes (KD-MWA-F-16) were implanted into the ACC. Additionally, cannulated microelectrode arrays (KD-MWA-A-4) were used to assess neural activity with pharmacological intervention.
Inflammatory pain model and von Frey test
Complete Freund’s Adjuvant (CFA)-induced inflammatory pain was adopted as a somatic pain model. 10 μL CFA was injected into the plantar surface of the right hind paw using a Hamilton syringe. The von Frey test was used to assess mechanical pain threshold on the inflamed paw. The 50% paw withdrawal threshold was determined using the up-down method of Dixon67.
Open field test and elevated plus maze
Open field test and elevated plus maze were performed using the ANY-maze acquisition system and software (Stoelting, Kiel, WI, USA). Mice were acclimated to the testing room 1 day prior to the test. Locomotion was recorded for 5 min in an open-top box (45 × 45 cm), and some key parameters were analysed, including total distance travelled, immobile time, mean speed without rest, and duration spent in the centre. For elevated plus maze, mice were placed at the centre of the apparatus and allowed to explore freely for 5 min. The number of entries into the open arms and time spent in the open arms were reported.
Statistical analyses
All data were expressed as means ± SEM unless otherwise stated. Significant differences were determined by student’s t-test, Mann–Whitney test, and one- or two-way ANOVA with Bonferroni test as appropriate using GraphPad Prism 9.5.0 (GraphPad Software, San Diego, CA, USA). All statistical tests were two-sided. Normality was checked with D’Agostino & Pearson test. When normality assumption was violated, the Mann-Whitney test was used. P < 0.05 was considered statistically significant with significance levels defined as follows: *p < 0.05, **p < 0.01, ***p < 0.001. Experiments and analyses were conducted in a blinded manner.
Key resources
Key resources, including antibodies, viruses and chemicals, are listed in Table S2.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All the data generated in the current study are presented in the figures and supplementary materials. The relevant raw data are provided as a Source Data file. Source data are provided with this paper.
Code availability
Calcium responses were analysed using a custom MATLAB script provided by ThinkerTech (Nanjing, China). The script with detailed instructions can be downloaded from the repository below: https://github.com/mdp10yy/TrippleColorMultiChannel-Script.git.
References
Grundy, L., Erickson, A. & Brierley, S. M. Visceral Pain. Annu. Rev. Physiol. 81, 261–284 (2019).
Papale, A. J., Flattau, R., Vithlani, N., Mahajan, D., & Nadella, S. A review of pharmacologic and non-pharmacologic therapies in the management of irritable bowel syndrome: current recommendations and evidence. J. Clin. Med. 13, 6948 (2024).
Ford, A. C., Vanner, S., Kashyap, P. C. & Nasser, Y. Chronic visceral pain: new peripheral mechanistic insights and resulting treatments. Gastroenterology 166, 976–994 (2024).
Ford, A. C. et al. Efficacy of 5-HT3 antagonists and 5-HT4 agonists in irritable bowel syndrome: systematic review and meta-analysis. Am. J. Gastroenterol. 104, 1831–1843 (2009).
Lee, J. H. The effects of music on pain: a meta-analysis. J. Music Ther. 53, 430–477 (2016).
Cepeda, M. S., Carr, D. B., Lau, J. & Alvarez, H. Music for pain relief. Cochrane Database Syst. Rev. CD004843 (2006).
McGill, M. et al. Neural signatures of auditory hypersensitivity following acoustic trauma. Elife 11, e80015 (2022).
Ciminelli, P. et al. Tinnitus: the sound of stress? Clin. Pract. Epidemiol. Ment. Health 14, 264–269 (2018).
Patil, J. D., Alrashid, M. A., Eltabbakh, A. & Fredericks, S. The association between stress, emotional states, and tinnitus: a mini-review. Front Aging Neurosci. 15, 1131979 (2023).
Manohar, S., Chen, G. D., Li, L., Liu, X. & Salvi, R. Chronic stress induced loudness hyperacusis, sound avoidance and auditory cortex hyperactivity. Hear Res 431, 108726 (2023).
Moloney, R. D. et al. Stress and the microbiota-gut-brain axis in visceral pain: relevance to irritable bowel syndrome. CNS Neurosci. Ther. 22, 102–117 (2016).
Ausland, J. H. et al. Tinnitus and associations with chronic pain: the population-based Tromso Study (2015-2016). PLoS One 16, e0247880 (2021).
De Meulemeester, K. et al. Comparing tinnitus, pain, psychosocial and cognitive factors between patients with tinnitus and pain: a systematic review. J. Psychosom. Res. 168, 111201 (2023).
Andresen, V. et al. Cerebral processing of auditory stimuli in patients with irritable bowel syndrome. World J. Gastroenterol. 12, 1723–1729 (2006).
Blomhoff, S., Jacobsen, M. B., Spetalen, S., Dahm, A. & Malt, U. F. Perceptual hyperreactivity to auditory stimuli in patients with irritable bowel syndrome. Scand. J. Gastroenterol. 35, 583–589 (2000).
Paulin, J., Andersson, L. & Nordin, S. Characteristics of hyperacusis in the general population. Noise Health 18, 178–184 (2016).
Khan, A. R., Geiger, L., Wiborg, O. & Czeh, B. Stress-induced morphological, cellular and molecular changes in the brain-lessons learned from the chronic mild stress model of depression. Cells 9, 1026 (2020).
Meredith, M. A. & Allman, B. L. Single-unit analysis of somatosensory processing in the core auditory cortex of hearing ferrets. Eur. J. Neurosci. 41, 686–698 (2015).
Zhou, W. et al. Sound induces analgesia through corticothalamic circuits. Science 377, 198–204 (2022).
Xu, Q. Y. et al. Identification of a glutamatergic claustrum-anterior cingulate cortex circuit for visceral pain processing. J. Neurosci. 42, 8154–8168 (2022).
Zhang, F. C. et al. Paraventricular thalamus-insular cortex circuit mediates colorectal visceral pain induced by neonatal colonic inflammation in mice. CNS Neurosci. Ther. 30, e14534 (2024).
Li, D. et al. The paraventricular thalamus mediates visceral pain and anxiety-like behaviors via two distinct pathways. Neuron 113, 2310–2324 (2025).
Blackwell, J. M. & Geffen, M. N. Progress and challenges for understanding the function of cortical microcircuits in auditory processing. Nat. Commun. 8, 2165 (2017).
Li, H. Y. et al. A thalamic-primary auditory cortex circuit mediates resilience to stress. Cell 186, 1352–1368 e18 (2023).
Zhou, Q., Wang, Y., Yi, L., Tan, Z. & Jiang, Y. Multisensory interplay within human auditory cortex: new evidence from intraoperative optical imaging of intrinsic signal. World Neurosurg. 98, 251–257 (2017).
Seybold, B. A. et al. Chronic reduction in inhibition reduces receptive field size in mouse auditory cortex. Proc. Natl. Acad. Sci. USA 109, 13829–13834 (2012).
Ibrahim, B. A. & Llano, D. A. Aging and central auditory disinhibition: is it a reflection of homeostatic downregulation or metabolic vulnerability? Brain Sci. 9, 351 (2019).
Perez, M. A. et al. Repeated restraint stress impairs auditory attention and GABAergic synaptic efficacy in the rat auditory cortex. Neuroscience 246, 94–107 (2013).
Ma, L., Li, W., Li, S., Wang, X. & Qin, L. Effect of chronic restraint stress on inhibitory gating in the auditory cortex of rats. Stress 20, 312–319 (2017).
Ayala-Rodriguez, J. D. & Garcia-Colunga, J. Maternal separation modifies spontaneous synaptic activity in the infralimbic cortex of stress-resilient male rats. PLoS One 18, e0294151 (2023).
Zhong, W., Zheng, W. & Ji, X. Spatial distribution of inhibitory innervations of excitatory pyramidal cells by major interneuron subtypes in the auditory cortex. Bioengineering (Basel), 10, 547 (2023).
Ghimire, M. et al. Increased pyramidal and VIP neuronal excitability in rat primary auditory cortex directly correlates with tinnitus behaviour. J. Physiol. 601, 2493–2511 (2023).
Musacchia, G. & Schroeder, C. E. Neuronal mechanisms, response dynamics and perceptual functions of multisensory interactions in auditory cortex. Hear Res 258, 72–79 (2009).
Fillinger, C., Yalcin, I., Barrot, M. & Veinante, P. Afferents to anterior cingulate areas 24a and 24b and midcingulate areas 24a’ and 24b’ in the mouse. Brain Struct. Funct. 222, 1509–1532 (2017).
Urrutia-Pinones, J., Morales-Moraga, C., Sanguinetti-Gonzalez, N., Escobar, A. P. & Chiu, C. Q. Long-range GABAergic projections of cortical origin in brain function. Front Syst. Neurosci. 16, 841869 (2022).
Tamamaki, N. & Tomioka, R. Long-range GABAergic connections distributed throughout the neocortex and their possible function. Front. Neurosci. 4, 2010 (2010).
Tomioka, R. et al. Demonstration of long-range GABAergic connections distributed throughout the mouse neocortex. Eur. J. Neurosci. 21, 1587–1600 (2005).
Bertero, A., Feyen, P. L. C., Zurita, H. & Apicella, A. J. A non-canonical cortico-amygdala inhibitory loop. J. Neurosci. 39, 8424–8438 (2019).
Bertero, A., Zurita, H., Normandin, M. & Apicella, A. J. Auditory long-range parvalbumin cortico-striatal neurons. Front Neural Circuits 14, 45 (2020).
Rock, C., Zurita, H., Wilson, C., & Apicella, A. J. An inhibitory corticostriatal pathway. Elife 5, e15890 (2016).
Lee, J. Y. et al. Role of anterior cingulate cortex inputs to periaqueductal gray for pain avoidance. Curr. Biol. 32, 2834–2847 e5 (2022).
Antioch, I. et al. Favorite music mediates pain-related responses in the anterior cingulate cortex and skin pain thresholds. J. Pain. Res. 13, 2729–2737 (2020).
Sun, W. et al. The anterior cingulate cortex directly enhances auditory cortical responses in air-puffing-facilitated flight behavior. Cell Rep. 38, 110506 (2022).
Song, J. J., Vanneste, S. & De Ridder, D. Dysfunctional noise cancelling of the rostral anterior cingulate cortex in tinnitus patients. PLoS One 10, e0123538 (2015).
Lee, U. et al. Functional brain network mechanism of hypersensitivity in chronic pain. Sci. Rep. 8, 243 (2018).
Menon, V. & Uddin, L. Q. Saliency, switching, attention and control: a network model of insula function. Brain Struct. Funct. 214, 655–667 (2010).
Green, S. A., Hernandez, L., Bookheimer, S. Y. & Dapretto, M. Salience network connectivity in autism is related to brain and behavioral markers of sensory overresponsivity. J. Am. Acad. Child Adolesc. Psychiatry 55, 618–626.e1 (2016).
Reynolds, R. P., Kinard, W. L., Degraff, J. J., Leverage, N. & Norton, J. N. Noise in a laboratory animal facility from the human and mouse perspectives. J. Am. Assoc. Lab Anim. Sci. 49, 592–597 (2010).
Nakamura, T., Tanida, M., Niijima, A. & Nagai, K. Effect of auditory stimulation on parasympathetic nerve activity in urethane-anesthetized rats. Vivo 23, 415–419 (2009).
Flores-Gutiérrez, E., Cabrera-Muñoz, E. A., Vega-Rivera, N. M., Ortiz-López, L. & Ramírez-Rodríguez, G. B. Exposure to patterned auditory stimuli during acute stress prevents despair-like behavior in adult mice that were previously housed in an enriched environment in combination with auditory stimuli. Neural Plast. 2018, 8205245 (2018).
Chen, Q.-Y., Wan, J., Wang, M., Hong, S. & Zhuo, M. Sound-induced analgesia cannot always be observed in adult mice. Mol. Pain. 19, 17448069231197158 (2023).
Geissler, D. B., Schmidt, H. S. & Ehret, G. Knowledge about sounds-context-specific meaning differently activates cortical hemispheres, auditory cortical fields, and layers in house mice. Front Neurosci. 10, 98 (2016).
Howlin, C. & Rooney, B. Cognitive agency in music interventions: Increased perceived control of music predicts increased pain tolerance. Eur. J. Pain. 25, 1712–1722 (2021).
Salimpoor, V. N. et al. Interactions between the nucleus accumbens and auditory cortices predict music reward value. Science 340, 216–219 (2013).
Yang, E. J., Lin, E. W. & Hensch, T. K. Critical period for acoustic preference in mice. Proc. Natl. Acad. Sci. USA 109, 17213–17220 (2012).
Lunde, S. J., Vuust, P., Garza-Villarreal, E. A. & Vase, L. Music-induced analgesia: how does music relieve pain? Pain 160, 989–993 (2019).
Nilsson, U. The anxiety- and pain-reducing effects of music interventions: a systematic review. AORN J. 87, 780–807 (2008).
Lu, G. et al. Effects of music therapy on anxiety: a meta-analysis of randomized controlled trials. Psychiatry Res. 304, 114137 (2021).
Fu, Q. et al. Music prevents stress-induced depression and anxiety-like behavior in mice. Transl. Psychiatry 13, 317 (2023).
Li, W. J. et al. Anxiolytic effect of music exposure on BDNFMet/Met transgenic mice. Brain Res. 1347, 71–79 (2010).
Kuhlmann, A. Y. R., de Rooij, A., Hunink, M. G. M., De Zeeuw, C. I. & Jeekel, J. Music affects rodents: a systematic review of experimental research. Front Behav. Neurosci. 12, 301 (2018).
Semyachkina-Glushkovskaya, O. et al. Phenomenon of music-induced opening of the blood-brain barrier in healthy mice. Proc. Biol. Sci. 2020 287, 20202337 (1941).
Morton, A. J., Hickey, M. A. & Dean, L. C. Methamphetamine toxicity in mice is potentiated by exposure to loud music. NeuroReport 12, 3277–3281 (2001).
Chikahisa, S., Sano, A., Kitaoka, K., Miyamoto, K. & Sei, H. Anxiolytic effect of music depends on ovarian steroid in female mice. Behav. Brain Res. 179, 50–59 (2007).
Li, Y. C. et al. Distinct circuits and molecular targets of the paraventricular hypothalamus decode visceral and somatic pain. Neuron 112, 3734–3749 (2024).
Zhang, F. C. et al. A vagus nerve dominant tetra-synaptic ascending pathway for gastric pain processing. Nat. Commun. 15, 9824 (2024).
Chaplan, S. R., Bach, F. W., Pogrel, J. W., Chung, J. M. & Yaksh, T. L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 53, 55–63 (1994).
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (grant numbers: 82530041 and 32230041 to G.Y.X., 82571396 to Y.Y.), the National Key Research and Development Programme of China (2024YFC3505200 and 2024YFC3505203 to G.Y.X.), the Chinese Red Cross Foundation National Brain Nutrition Research Fund (to G.Y.X.) and the Natural Science Foundation of Jiangsu Province (BK20255001 to G.Y.X.). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Y.Y., W.Q.K. and Y.H.H. acquired and analysed the data, prepared the figures and manuscript. Q.Q.C., Y.C.L. and F.C.Z. interpreted the data. HDN revised the manuscript. G.Y.X. designed the experiments, supervised the experiments and finalized the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Tao Chen, Xi Chen and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yu, Y., Kuang, WQ., He, YH. et al. A primary auditory cortex-anterior cingulate cortex circuit underlying cross-modal visceral pain modulation. Nat Commun 17, 2352 (2026). https://doi.org/10.1038/s41467-026-69135-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69135-3