Abstract
Small-cell lung cancer (SCLC) is the most lethal type of lung cancer. Paradoxically, this tumor displays a high mutation burden; however, a modest response to immunotherapy. Improving Immunotherapy response in SCLC patients remains an unmet need. Here, we report that across 24 tumor types, including over 179,000 real-world patient tumors, SCLC has the highest expression of nonhomologous end joining (NHEJ) DNA repair regulator PRKDC (DNAPKcs). High PRKDC expression predicts poor response to immunotherapy in SCLC. DNAPKcs depletion causes activation of cGAS/STING pathway due to cytoplasmic accumulation of double-stranded DNA, inducing immunogenicity and enhancing sensitivity of SCLC models to immunotherapy. Analyses in SCLC cell lines and mouse models shows that depletion of DNAPKcs leads to proteasomal degradation of MYC via GSK3β pathway. We show that DNAPKcs upregulation contributes to immunotherapy resistance and DNAPKcs inhibition represents a promising therapeutic strategy to induce antitumor immunity and potentiate immunotherapy efficacy in immunologically suppressed SCLC.
Similar content being viewed by others
Introduction
Small cell lung cancers (SCLCs) are highly aggressive neuroendocrine cancers that account for 15% of all lung cancer cases, with a median overall survival of only 9–11 months1,2. SCLC accounts for 30,000 new cases diagnosed annually in the United States and has an overall 5-year survival rate of about 7%3. While treatment options have recently broadened, however, overall outcomes only modestly improved, indicating a pressing need for novel therapies. The recent addition of surface-targeted drugs shows promise but is also accompanied by severe toxicities4. Efforts to expand the therapeutic arsenal toward SCLC have focused on immune checkpoint blockade (ICB) using agents targeting programmed cell death protein 1 (PD-1) and its ligand (programmed death ligand 1 [PD-L1]). One particularly understudied but common feature of SCLC is an immunosuppressed phenotype characterized by relatively low levels of infiltrating T-cells, suppression of class I major histocompatibility complex (MHC-I), or reduced antigen presentation5. Consistent with a compromised tumor microenvironment immune milieu, only a few patients with SCLC have a deep and durable response to ICB. The approved first-line treatment of platinum doublet and PD-L1 blockade leads to a modest increase in overall survival by approximately 2 months2,6,7,8. The mechanisms preventing ICB from reactivating antitumor immunity in SCLC are not fully understood.
Identifying therapeutic targets in SCLC has been challenging, partly because primary alterations are loss-of-function mutations in tumor suppressor genes and patients with SCLC exhibit heterogeneous transcriptomic and proteomic profiles9. SCLC is characterized by the ubiquitous loss of TP53 and RB1 and amplification of MYC family genes in a subset of SCLC2,9. Observed in approximately 20% of patients with SCLC, amplification of the MYC family genes (MYCL, MYCN, and MYC) has been reported and associated with worse prognosis10. MYC is often termed “undruggable” due to its critical role and complex protein structure11. Owing to the alterations in the genes involved in replication stress, we have previously established that targeting the replication stress response pathways is an effective therapeutic strategy in SCLC2,12,13,14,15,16.
Nonhomologous DNA end joining (NHEJ) is the primary pathway for repair of double-strand DNA breaks in human cells, and disruption of this pathway has been implicated in tumorigenesis and immune evasion. NHEJ confers resistance to chemotherapy in multiple cancers17. KU and DNA-PKcs are overexpressed in multiple cancers and repair DNA double-strand breaks (DSBs)18. DNA-dependent protein kinase catalytic subunit (DNAPKcs, gene name: PRKDC) is a multifunctional serine–threonine protein kinase that orchestrates the NHEJ-mediated repair upon detection of double-strand break in DNA19. DNAPKcs overexpression is often associated with a poor prognosis in patients with non-SCLC (NSCLC) and other solid tumors20,21. DNAPKcs can also drive antiviral responses via a STING-independent pathway21,22. These reports suggest that DNAPKcs is a potential regulator of the innate immune response in cancer cells. Multiple DNAPKcs inhibitors are undergoing clinical trials as monotherapies or in combination with other therapies23,24,25 for cancer. However, the biological significance of NHEJ signaling and the effects of targeting NHEJ signaling via DNAPKcs inhibition has not been previously explored in SCLC.
In this work, we demonstrate that PRKDC is significantly overexpressed in SCLC and predicts poor prognosis. DNAPKcs inhibition in vivo leads to increased immunogenicity and augmentes the response to chemotherapy and PD-L1 blockade. Therefore, we establish the biological effects of targeting NHEJ in SCLC and highlight the role DNAPKcs inhibition as a promising therapeutic strategy to augment the efficacy of chemotherapy and immunotherapy in SCLC.
Results
SCLC tumors have a significantly higher expression of PRKDC compared to other cancers and PRKDC expression predicts worse prognosis in patients with SCLC
NHEJ is a major DNA double-strand break repair pathway whose effect has been previously unexplored in SCLC. Therefore, we investigated the expression of PRKDC (key regulator of the NHEJ pathway) in 179,508 real-world tumor samples across 24 tumor types, including SCLC. Encouragingly, SCLC had the highest expression of PRKDC among the 24 types of cancer (Fig. 1A and Supplementary Table S1). This is a significant finding as this reports of the significantly high expression of the NHEJ gene, PRKDC in SCLC.
A–C The boxplots represent common distribution metrics, with the center line representing the median, the top and bottom bounds of the boxes representing the 25th and 75th percentiles, and whiskers length of 1.5*IQR (interquartile range) beyond the 25th or 75th percentiles, represents the minimum or maximum value if no outliers beyond the 1.5*IQR are present. A Boxplots showing comparative mRNA expression of PRKDC in 24 cancer types (n > 179,000 tumor samples). Statistical details in Supplementary Table 1. B Boxplots showing comparative mRNA expression of PRKDC in 5 major classes of lung cancer in patient tumor samples. Statistical details in Supplementary Table 2. C Boxplots showing comparative mRNA expression of PRKDC in patient tumor samples with subtypes of SCLC. Statistical significance: Pairwise Mann-Whitney U test test, P-values: TF-neg/Mixed, P = 7.5e-30; TF-neg/ASCL1, P = 2.9475e-16; YAP1/TF-neg, P = 3.41e-12; TF-neg/NEUROD1, P = 3.63375e-7; Mixed/ASCL1, P = 0.000002919; NEUROD1/Mixed, P = 0.00000685; POU2F3/Mixed, P = 0.0000852857; YAP1/Mixed, P = 0.000384375; TF-neg/POU2F3, P = 0.022666667; YAP1/POU2F3, P = 0.060136364; POU2F3/ASCL1, P = 0.060136364; YAP1/NEUROD1, P = 0.305769231; NEUROD1/ASCL1, P = 0.305769231; POU2F3/NEUROD1, P = 0.3075; YAP1/ASCL1, P = 0.845. Detailed in Supplementary Table 3.D Kaplan-Meier survival curve of patients with SCLC with high versus low PRKDC expression (n = 21). Cox regression was used to obtain significance (P: 0.041). E Enrichment curves showing the top correlated immune pathways in real-world patients with SCLC with lower expression of PRKDC (top 25%). Statistical details in Supplementary Table 4. F Enrichment curves showing correlation hallmark pathway in SCLC patient population with the highest expression of PRKDC (top 25%). G Kaplan-Meier curve showing OS of patients with SCLC with high (top 25%) versus low (bottom 25%) PRKDC expression (n = 423 patients with SCLC). For statistical analysis, the log-rank test was used (P = 0.033). H Survival curve showing the association of PRKDC expression with OS for patients with SCLC from the IMpower133 dataset falling into the top (high) and bottom (low) 25th percentiles; data are shown splitting into Atezo and Plazebo. Statistical significance tested using Cox regression. I PFS curve showing the association of PRKDC expression with time to disease progression for patients with SCLC falling into the top (high) and bottom (low) 25th percentiles of PRKDC expression. Data is further split into Atezo and Plazebo. P-value was calculated using Cox regression.
Since there is a substantial biological and therapeutic heterogeneity within lung cancer subtypes, we next examined PRKDC expression in clinical samples (n = 36,896) from multiple lung cancer subtypes, including lung carcinoid (n = 177), NSCLC (n = 35,324), atypical lung carcinoid (n = 19), large cell lung carcinoma (n = 432), and SCLC (n = 944). As expected, SCLC clinical samples had significantly higher expression of PRKDC compared with other lung cancer subtypes (Fig. 1B and Supplementary Table S2). To confirm the finding in cell lines, we next assessed PRKDC expression in NSCLC and SCLC cell lines using the Cancer Cell Line Encyclopedia database. Consistent with our observation in clinical samples, PRKDC expression was significantly higher (p = 0.00026) in SCLC compared with that in other lung cancer cell lines (Supplementary Fig. S1A).
SCLC has been subdivided into four major subtypes based on relative expression of transcription factors like ASCL1 (SCLC-A), NEUROD1 (SCLC-N) and POU2F3 (SCLC-P)2,26,27,28. A fourth subtype defined by relatively higher expression of immune-related genes (inflamed) and YAP1 (SCLC-Y). Hence, we next investigated whether there are any subtype-specific differences in the expression of PRKDC in over 900 SCLC patient tumors. In addition to the four well-known subtypes (ASCL1, NEUROD1, POU2F3, and YAP1) our cohort also included cases with no known lineage determining transcription factors (TFs; TF negative) and tumors with more than one TFs (mixed tumors). Interestingly, TF-negative tumors had the lowest expression, and mixed TF tumors had the highest expression of PRKDC (statistical significance detailed in figure legends) (Fig. 1C and Supplementary Table S3). Among the well-known subtypes, the ASCL1-driven subtype had the highest expression of PRKDC (TF-neg/ASCL1, p = 2.9475e-16). This is the largest SCLC dataset that has reported the transcriptomic expression of PRKDC in SCLC.
Since the role of the NHEJ pathway in the prognosis of patients with SCLC is obscure, we next investigated whether higher PRKDC expression affects the overall survival (OS) of patients with SCLC. Higher PRKDC expression predicted significantly shorter OS in 21 patients with SCLC29 (Fig. 1D). To further investigate, we leveraged the IMpower133 Phase 3 clinical trial dataset (NCT02763579)7, which to our knowledge, is the largest available clinical cohort with transcriptomic and survival data for SCLC. We stratified patients into quartiles based on PRKDC expression and then compared survival between the highest and lowest quartiles (n = 68 patients per group). Patients in the lowest PRKDC expression quartile had significantly better overall survival (OS) (Supplementary Fig. S1B) as well as progression-free survival (PFS) (Supplementary Fig. S1C) compared to those in the highest quartile.
Our results indicate that SCLC tumors and cell lines have significantly higher expression of NHEJ master regulator, PRKDC, and that PRKDC expression predicts a worse prognosis in SCLC.
PRKDC expression is correlated with lower expression of immune-related genes and predicts resistance to several drugs targeting the replication stress proteins
To understand the biological implications of high baseline expression of PRKDC in SCLC, we investigated the genes significantly correlated to PRKDC expression in SCLC cell lines and patient tumors. Multiple immunogenic pathways like hallmark inflammatory response, IFNγ response, IFNα response, were found to be enriched in SCLC clinical samples with low baseline expression of PRKDC (Fig. 1E). Whereas E2F target genes, G2M checkpoint and MYC target genes were observed to be enriched in SCLC patient samples with high expression of PRKDC (Fig. 1F). Furthermore, high PRKDC expression was positively correlated with DNA damage repair genes and MYC expression (Supplementary Fig. S1D).
To confirm our findings in preclinical models, we investigated the top genes that were positively or negatively correlated with PRKDC in SCLC cell lines. We demonstrated that replication stress genes, such as CHEK1, CHEK2, and PARP1 were positively correlated with PRKDC expression (Supplementary Fig. S1E). We also observed that the inhibitory NOTCH ligand, DLL3, which is an important therapeutic target in SCLC along with mTOR were positively correlated with PRKDC expression (Supplementary Fig. S1E). Interestingly, the chemokine C-X-C motif chemokine ligand 5 (CXCL5), TMEM173 (STING), and CD274 (PD-L1) were among the top negatively correlated genes with PRKDC expression (Supplementary Fig. S1F). Other immune-related genes, TGF-beta-I, and TNFRSF1A were also significantly negatively correlated with PRKDC expression (Supplementary Fig. S1F) in SCLC. These results indicate that PRKDC is positively correlated with replication stress genes and other SCLC-related oncogenes and is negatively correlated with immune-related genes in SCLC clinical samples and preclinical models.
Primary and acquired resistance to drugs targeting the DNA damage response pathways has been one of the major hurdles in the clinical translation of this class of drugs in SCLC. Therefore, to investigate whether PRKDC expression has any implications in drug resistance in SCLC, we compared the baseline expression of PRKDC in 63 SCLC cell lines that were treated with 103 US FDA-approved drugs and 423 investigational agents30. Interestingly, higher PRKDC expression was correlated with significantly higher IC50 of drugs targeting HSP90 (A1A7-AAG, A1A7-DMAG), PARP (BMN-673), IMPDH (AVN944), and topoisomerase (irinotecan, teniposide) (Supplementary Fig. S1G). Interestingly, all of these drugs target proteins linked to DNA repair and replication stress, which are key vulnerabilities in SCLC.
Therefore, we demonstrate that higher PRKDC expression is correlated with lower expression of key immune-related genes and is a potential biomarker of resistance to drugs targeting the DNA damage response pathways.
SCLC tumors with low PRKDC expression have better prognosis and derive greater benefit from chemotherapy and immunotherapy
Next, we investigated the role of NHEJ regulator, DNAPKcs, in the prognosis of patients treated with cisplatin in SCLC patients. High PRKDC expression predicted significantly poorer OS in a total of 423 patients treated with cisplatin (HR = 1.529 (95% CI: 1.032 - 2.265, p = 0.033,) (Fig. 1G). We further extended our analysis on OS and PFS (Progression free survival) using the IMPower133 dataset, which was the first randomized trial to demonstrate PFS or OS improvements with ICB in SCLC. We used the controlled access IMPower133 dataset (Phase 3 clinical trial dataset evaluating efficacy of atezolizumab in combination with chemotherapy), which includes 271 patients of various ages, genders, treated with either atezolizumab or placebo. First, we set out to test for the association of PRKDC expression with progression-free survival (OS). We used a cox regression model with censored time-to-disease progression (in months) as the dependent variable, and PRKDC as the independent variable (continuous, tpm), exploring possible covariates such as age, sex, and treatment, as well as interaction terms. This analysis revealed a significant association between OS and PRKDC (p-value = 0.0193), after adjusting for treatment as a covariate. Indeed, treatment (Atezo or Placebo) was also significantly associated with OS (p = 0.0193) (Fig. 1H). Next, we utilized the same statistical approach to test for the association of PRKDC expression with progression free survival (PFS) of SCLC patients. As expected, similar to OS, we demonstarte a significant association between PFS and PRKDC (p-value = 0.000302) (Fig. 1I). We demonstrate that high baseline PRKDC expression may be predictive of ICB benefit in SCLC.
DNAPKCs inhibition decreases the viability of SCLC cells and causes tumor regression in SCLC PDX models
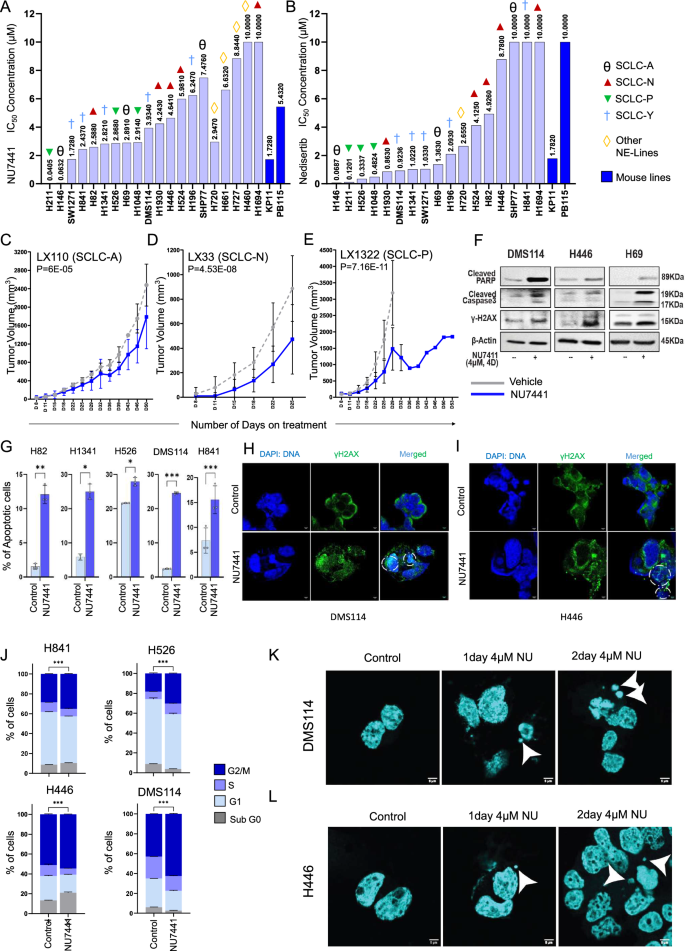
Next, we investigated the effect of DNAPKcs inhibition on SCLC cells. We used a panel of 16 SCLC cell lines (including 14 human-derived and 2 mouse-derived models), representing all four subtypes of SCLC. We treated the cells with two different DNAPKcs inhibitors (NU7441 and nedisertib) that are currently in clinical trials for other cancers31. Cells were treated with either NU7441 or nedisertib for 5 days with multiple doses and the resulting change in cell viability was assessed by cell titer glo. SCLC cells showed a range of sensitivity to NU7441 and nedisertib (Fig. 2A, B), with IC50s ranging starting at 40 nM to 10 µM for NU7441 and 68 nM to 10 µM for nedisertib. The IC50 for nedisertib for several cell lines was as low as 68 nM. To further extend the scope of our study, we treated four thoracic neuroendocrine cell lines with NU7441. Our data indicated that the thoracic neuroendocrine lines were also sensitive to NU7441 treatment, with IC50 concentration starting at 2.947 µM (Fig. 2A, B). This provides compelling evidence for the sensitivity of SCLC and other thoracic neuroendocrine cells to DNAPKcs inhibition.
A IC-50 values of 22 SCLC cell lines (20 human and 2 murine-derived) representing all major subtypes of SCLC and NETs, treated with NU7441. B IC-50 values of 17 SCLC cell lines (15 human and 2 murine-derived) representing all major subtypes of SCLC, treated with nedisertib. SCLC-A: ASCL1 High subtype, SCLC-N: NEUROD1 High Subtype, SCLC-P: POU2F3 High Subtype, and SCLC-Y: YAP1 High Subtype of SCLC. C–E Tumor growth curve data of 3 patient-derived xenograft models: LX110 (C), LX33 (D), and LX1322 (E) in response to vehicle and NU7441 treatment. The data represents the means ± SD (n = 12); P-values calculated using linear mixed-effects regression models. F Western blot analysis showing protein expression levels of cleaved PARP, cleaved caspase3, and phospho-H2AX upon 4 µM NU7441 in DMS114, H446, and H69 cells compared to untreated control cells. G Bar diagrams showing percentage of total-apoptotic H82, H1342, H526, DMS114, and H841 cells upon 4 µM NU7441 (4 d) treatment compared to control. The data represent the means ± SE (n = 3) of biological replicates; P-values calculated by unpaired 2-tailed student’s t test (ns > 0.05, ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001). P values: H82 P = 0.003; H1341 P = 0.19; H526 P = 0.012; DMS114 P < 0.001; H841 P < 0.001. H, I Confocal microscopy image of DMS114 (H) and H446 (I) cells showing increased puncta of γH2AX in response to NU7441 treatment compared to control. Cells imaged with Leica STED 3X at 63X lens zoomed at 2X. Scale-bar of 5 µm. J Bar graphs showing cell cycle analysis. Cell cycle phase profiles of H841, H526, H446, and DMS114 cells treated with 4 µM NU7441 for 4 days and control cells. The data represent the means ± SE (n = 3) of biological replicates; P-values calculated by unpaired 2-tailed student’s t test (ns > 0.05, ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001). K, L Confocal microscopy image of DMS114 (K) and H446 (L) cells showing increased micronuclei formation in response to NU7441 treatment compared to control cells. Cells imaged with Leica STED 3X at 63X lens zoomed at 2X. Scale-bar of 5 µm.
We next investigated the antitumor effect of NU7441 in SCLC PDX models representing the three major subtypes. We included three PDX models previously characterized using genomic, transcriptomic, and proteomic assays with correlative clinical data32. The three PDX models had high expression of ASCL1 (LX110), NEUROD1 (LX33), and POU2F3 (LX1322). Tumor bearing mice from each model were treated with either vehicle, or single agent NU7441 (10 mg/kg, PO, 5 out of 7, QD). We measured the rate of tumor reduction when the vehicle treated group reached maximum tumor volume.
We observed the modest anti-tumor efficacy in the ASCL1 high PDX model (LX110) with 28% reduction in tumor growth upon NU7441 treatment relative to control at day 50 (Fig. 2C). In NEUROD1 high (LX33) PDX model, we observed a higher degree of tumor growth inhibition with a 58% reduction in tumor volume post-NU7441 treatment relative to control at day 50 (Fig. 2D). Encouragingly, in the POU2F3 high (LX1322) we observed a remarkable ~ 76% reduction of tumor growth post-NU7441 treatment relative to control by day 29 (Fig. 2E). Together these results demonstrate that a subgroup of SCLC preclinical models is sensitive to DNAPKcs targeting.
To optimize patient selection for clinical trials, we next investigated the biomarkers that predict the sensitivity of SCLC cell lines to DNAPKcs inhibition. We compare the cell line IC50 to the baseline gene expression of the cell lines. Pathway analysis with GSEA demonstrated MYC targets, G2M checkpoint pathway, and E2F pathway to be the top pathways correlated with sensitivity to both NU7441 and nedisertib in SCLC cell lines (Supplementary Fig. S2A, S2B). Next, we investigated the probable correlation between any of the top three lineage-defining transcription factors (LDTFs) (ASCL1, NEUROD1 and POU2F3) with PRKDC expression. Our results did not show a strong correlation of PRKDC expression with basal expression of the three LDTFs (Supplementary Fig. S2C).
Next, we looked at individual genes that may predict sensitivity to DNAPKcs inhibition. Interestingly, high baseline expression of SLFN11 predicted lower IC50 of nedisertib (p = 0.036, Supplementary Fig. S2D). We and others have demonstrated the role of SLFN11 as a predictive biomarker to chemotherapy and PARP inhibition in SCLC33. This is translationally very relevant as SLFN11 can be detected by IHC, and a recent clinical trial (SWOG S1929) has demonstrated the feasibility of including SLFN11 expression as a predictive biomarker in selecting ES-SCLC patients for treatment with PARP inhibitors and immunotherapy in the maintenance setting. Therefore, if validated in clinical samples, SLFN11 can be a predictive biomarker for DNAPKcs inhibition in SCLC clinical trials.
DNAPKcs inhibition induced DNA damage, apoptosis and micronuclei formation in SCLC models
To establish the downstream effect of DNAPKcs inhibition in SCLC, we treated DMS114, H446, and H69 cells with the concentration of NU7441, where we observed appreciable target inhibition (4 µM NU7441 for 4 days). Western blotting analysis showed an increase in apoptosis markers, cleaved PARP, and cleaved caspase-3, in post-NU7441 treatment compared to control in all three cell lines (Fig. 2F), indicating apoptotic induction. We performed annexin V-PI based flow cytometry to further validate apoptosis induction post-NU7441 treatment (4 µM NU7441 treatment for 4 days). We included five SCLC models that showed differential IC50 to NU7441 treatment (Fig. 2Gand Supplementary Fig. S2E). Interestingly, DMS114 representing the SCLC-Y subtype showed highest induction of apoptosis ( ~ 24%) post-NU7441 treatment followed by H1341 ( ~ 20% increase) (Fig. 2Gand Supplementary Fig. S2E). SCLC-N subtype, H82 cells showed increase in apoptosis, from 1.5% to ~12% (Fig. 2Gand Supplementary Fig. S2E). However, H841 (SCLC-Y) and H526 (SCLC-P) cell lines showed modest yet statistically significant increase in apoptotic population ( ~ 6–8%) post-NU7441 treatment (Fig. 2Gand Supplementary Fig. S2E). These findings establish that DNAPKcs inhibition causes appreciable apoptosis in SCLC models.
Next, we determined whether this apoptotic induction is a drug-specific event or due to the inhibition of the NHEJ regulator, DNAPKcs. Therefore, we performed siRNA-mediated knockdown of PRKDC in SCLC cell lines using 2 different siRNA equences. PRKDC knockdown also resulted in an appreciable increase in cleaved PARP (Supplementary Fig. S2F). Moreover, both pharmacological inhibition (with NU7441) of DNAPKcs, or genetic knockdown of PRKDC led to an notable increase in phospho-H2AX protein level indicating increased double-stranded DNA damage in SCLC models (Fig. 2Fand Supplementary Fig. S2F). To further validate double stranded DNA damage, we performed confocal microscopy in multiple SCLC models pre- and post-NU7441 treatment. We observed significant increase of γH2AX puncta formation inside the nucleus in both DMS114 and H446 cell lines post 4 days of NU7441 treatment (Fig. 2H, Iand Supplementary Fig. S2G).
Next, we investigated the effect of DNAPKcs targeting cell cycle progression in SCLC cell lines. Four SCLC cell lines with a range of sensitivity to NU7441 were treated with 4 µM NU7441 for 4 days. Flow cytometry-based cell cycle analysis showed an appreciable G2/M arrest in H526, and DMS114 cell lines (more than 1.5-fold change, p < 0.001) (Fig. 2J) and only a modest increase in H841 and H446 cells.
The induction of DNA damage is associated with aberrant breaks in DNA and the presence of dsDNA in the cytoplasm. Hence, we investigated the presence of DNA in the cytoplasm. Western blotting analysis showed increased levels of cytosolic histone H3 in DMS114, H446, and H69 cells, most prominently at day 4 after NU7441 treatment, indicating the presence of cytosolic DNA (Supplementary Fig. S3A). Unresolved DNA damage leads to nuclear envelope rupture, exposing double-stranded DNA to the cytosol. The presence of this dsDNA in cytoplasm can invoke inflammatory signaling in the cell34. To confirm micronuclei formation, we performed confocal microscopy in multiple SCLC models pre- and post-NU7441 treatment. We observed the presence of micronuclei in both DMS114 and H446 cell lines after 1 and 2 days of NU7441 treatment (Fig. 2K, Land Supplementary Fig. S3B).
Taken together, we demonstrate that pharmacologic and genetic targeting of DNAPKcs leads to appreciable DNA damage, apoptosis induction and formation of micronuclei in SCLC models.
DNAPKcs inhibition activates the cGAS-STING pathway and increases IFN signaling in SCLC
Cancers often evade immune detection by silencing the Stimulator of Interferon Genes (STING)-Interferon (IFN) signaling. We have previously shown that therapeutic reactivation of STING by epigenetic therapies and DNA damage response inhibitors can restore antitumor immunity in multiple preclinical models. Moreover, we and others have shown that DNA damage itself leads to the formation of micronuclei and cytosolic GMP-AMP synthase (cGAS) in the cytoplasm can colocalize with and recognize broken DNA in micronuclei12,35.
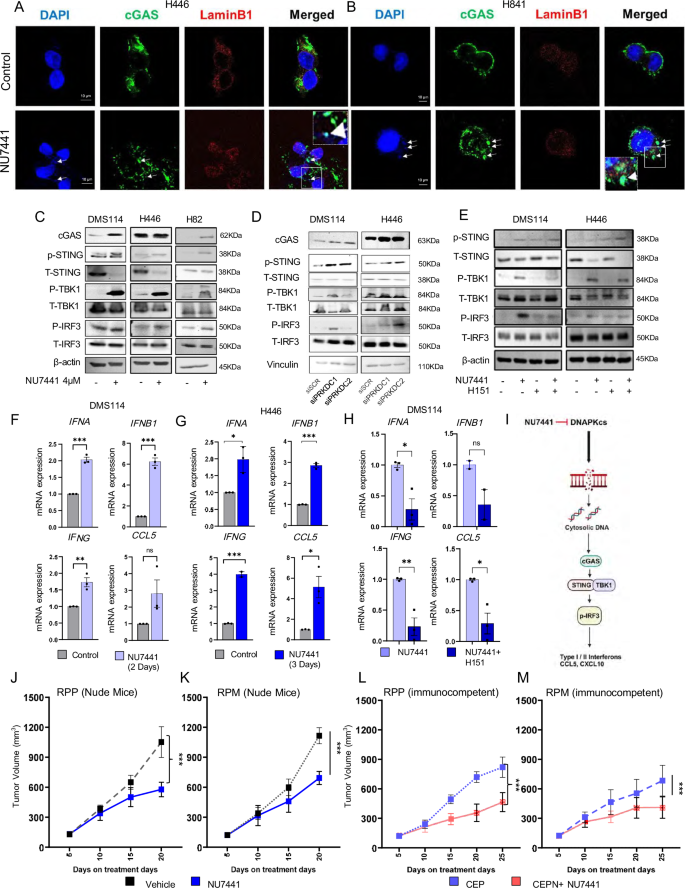
Hence, to further investigate the effect of DNAPKcs targeting on cGAS/STING pathway we performed confocal microscopy in H446 and H841 with or with treatment without NU7441 treatment (4 µM, 24 h treatment). We stained the cells with DAPI for micronuclei, cGAS, and lamin B1 to stain the nuclear envelope. We observed appreciable micronuclei formation in the treated group, and colocalization of cGAS to the micronuclei in both models after NU7441 treatment. Therefore, we conclude that DNAPKcs targeting leads to colocalization of the cytoplasmic cGAS with the DNA in micronuclei (Fig. 3A, Band Supplementary Fig. S3C).
A, B Immunofluorescence showing co-localization of micronuclei (blue) and cGAS (green) upon NU7441 treatment compared to control. Cells were imaged with Leica STED 3X. Scale-bar of 10 µm. C Western blots of cGAS, pSTING-S366, total-STING, pTBK1-S172, total-TBK1, pIRF3-S396, and total-IRF3 upon 4µM-NU7441 treatment for 4 days in DMS114, H446, and H82 cells compared to control. D Western blots of DNAPKcs, pSTING-S366, total-STING, pTBK1-S172, total-TBK1, pIRF3-S396, and total-IRF3 upon 2-independent si-RNA in DMS114, H446 cells compared to scrambled-si-RNA treatment. E Western blots of cGAS, pSTING-S366, total-STING, pTBK1-S172, total-TBK1, pIRF3-S396, and total-IRF3 upon 4µM-NU7441 ± H151 for 4 days in DMS114, H446, and H82 cells compared to control. F, G RT-PCR showing mRNA fold-change of type I/II interferon and CCL5 upon NU7441 treatment compared to control. Data represents SE (n = 3) of biological replicates, P-values calculated using unpaired 2-tailed student’s t test. P-values for 3 F: IFNA = 0.0002, IFNB1 = 0.0001, IFNG = 0.0064, CCL5 = 0.0002; P-values for 3 G IFNA = 0.0116, IFNB1 < 0.0001, IFNG = 0.0001, CCL5 = 0.0167. H RT-PCR showing mRNA fold change of type I/II interferon and CCL5 upon 4 µM NU7441 ± H151 in DMS114 cells. Data represents SE (n = 3) of biological replicates, P-values calculated using unpaired 2-tailed Student’s t test. P-values for 3H: IFNA = 0.015, IFNB1 (n = 2) = 0.125, IFNG = 0.006, CCL5 = 0.014. I Schematic diagram showing the mechanism of cGAS-STING pathway activation upon NU7441 treatment. This figure was created in BioRender. Sen, T. (2025) https://BioRender.com/abxm28o. J, K Tumor growth curve data of 2 immunocompromised GEMMS: RPP (J) and RPM (K) in response to cisplatin-etoposide + NU7441 treatment compared to standard-of-care cisplatin-etoposide. Data represented as mean ± SD (n = 7), P-values calculated using linear mixed-effects regression models. For 3 J: P = 1.41E-08; 3 K: P = 3.32E-11. L, M Tumor growth curve data of 2 immunocompetent GEMMS: RPP (L) and RPM (M) in response to cisplatin-etoposide+NU7441 treatment compared to standard of care. Data represents mean ± SD (n = 8 for CEP and n = 10 for CEPN treatment), P-values calculated using linear mixed-effects regression models. 3 L: P = 1.81E-17; 3 M: P = 1.442E-07. (ns > 0.05, ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001).
Colocalization of cytoplasmic cGAS with DNA in micronuclei can lead to the activation of the cGAS-STING–dependent immune response12,35. Next, we investigated whether DNAPKcs inhibition leads to the activation of the STING pathway. Pharmacological inhibition of DNAPKcs (by NU7441 treatment) increased protein expressions of cGAS, and other key components of the STING pathway like phosphorylated (p-) STING, p--TBK1, and phospho- IRF3, in three SCLC cell lines, DMS114 (SCLC-Y), H446 (SCLC-N), and H82 (SCLC-N) (Fig. 3C), and the neuro-endocrine tumor cells (NETs) cell line H720 (Supplementary Fig. S4A). This demonstrates the activation of the cGAS–STING pathway upon DNAPKcs inhibition. Next, to exclude the possibility of off-target drug effects, we performed siRNA-mediated PRKDC knockdown in DMS114 and H446 cells and assessed the activation of the cGAS–STING pathway by western blot (Fig. 3D). As with pharmacological inhibition, genetic knockdown of PRKDC by 2 independent siRNA sequences also increased expression of p-STING, p-TBK1, and p-IRF3 (Fig. 3D), confirming STING pathway activation.
Next, to confirm the importance of STING activation on the NU7441-mediated downstream effects, we treated DMS114 and H446 cells with NU7441 in the presence or absence of the STING inhibitor H151 (Fig. 3E). As before, NU7441 treatment led to an increase in p-STING, p-TBK1, and p-IRF3 protein expressions, which was successfully reversed in the presence of STING inhibition in either the DMS114 or H446 cell line. This confirms STING-mediated activation of the TBK1/IRF3 signaling after NU7441 treatment (Fig. 3E). Thus, our data indicate the activation of the cGAS–STING pathway in response to DNAPKcs inhibition in SCLC and other neuroendocrine tumor cell lines, extending the scope and utility of DNAPKcs inhibition.
We and others have shown that activation of IRF3 leads to the transcriptional upregulation of the interferon genes. Therefore, we next investigated the expression of the expression of interferons (IFNs; type I/II IFNs) and pro-inflammatory chemokines following NU7441 treatment. NU7441 treatment led to a significant time-dependent increase in the mRNA expression of IFNA, IFNB1, IFNG, and CCL5 in DMS114 (Fig. 3F), H446 (Fig. 3G) and H720 (atypical neuroendocrine carcinoid) cells (Supplementary Fig. S4B).
Next, we investigated the importance of STING on transcriptional upregulation of type I/II IFNs and chemokines in SCLC. As before, we performed pharmacological inhibition of STING in SCLC models. Treatment with the STING inhibitor, H151, successfully abrogated NU7441-mediated increased mRNA expression of IFNA, IFNG, and CCL5 in DMS114 cells (Fig. 3H). Next, we investigated whether increase mRNA expression of type-I interferons and chemokines is recapitulated in protein expression level. We performed ELISA both with cell free protein lysate and condition media of H446 and DMS114 to study the internal protein level as well as secreted protein level upon 3days of NU7441 treatment. Our results indicates increase in IFNα in cell free protein lysate upon NU7441 treatment in both H446 and DMS114 cell lines (Supplementary Fig. S4C). Whereas CCL5 internal protein level as well as secreted protein level increase was only found in DMS114 cell line (Supplementary Fig. S4D). Interestingly in DMS114 cell we further found increased secreted protein expression of another chemokine CXCL10 (Supplementary Fig. S4E).
We demonstrate that DNAPKcs inhibition activates the cGAS/STING pathway in SCLC cells, leading to increased expression of type I and II interferons IFNα, IFNβ, and IFNγ and the pro-inflammatory chemokine, CCL5, in SCLC cells. We further establish the crucial role of STING in TBK1/IRF3 activation and the increase in type I/II interferons and CCL5 in response to DNAPKcs inhibition (Fig. 3I).
Since DNAPKcs activated the cGAS/STING mediated increase in IFN pathway, next we wanted to assess the importance of the intact immune system in DNAPKcs-mediated anti-tumor response. For this we selected GEMM-derived cell lines derived from Rb1-/-, Trp53-/-, and Rbl2-/- (RPP) and Rb1-/-, Trp53-/-, and MYCT58A (RPM) models. RPP and RPM cells were subcutaneously injected in immunocompromised athymic nude mice. Tumor bearing mice were treated with vehicle or NU7441 (10 mg/kg, i.p. 5/7 days). NU7441 treatment caused significant tumor growth delay but not tumor regression in either RPP or RPM tumors grown in immunocompromised nude mice (Fig. 3J, K). This indicates the antitumor response of NU7441 in SCLC.
DNAPKcs inhibition augments the anti-tumor response to chemotherapy in SCLC
Cisplatin-etoposide based chemotherapy has been as the cornerstone of a first-line therapy for SCLC patients, but its activity is limited due to acquired resistance, highlighting the limitations of this treatment paradigm. As cisplatin-etoposide induces replication stress, we hypothesized that DNAPKcs may augment the anti-tumor response of cisplatin-etoposide, inducing potent tumor cell death while also avoiding the potential for overlapping toxicities. To explore this combination further, we tested the effect of NU7441 and cisplatin-etoposide combination on RPP and RPM tumors grown in immunocompetent SCLC in vivo models.
Tumor bearing mice were treated with: (1) Cisplatin-etoposide (3 mg/kg, 3 out of 7 days) alone (CEP) and (2) NU7441 (10 mg/kg, i.p. 5/7 days) and CEP combination (CEPN). NU7441 + CEPN combination treatment significantly delayed tumor growth in the RPP model as compared with vehicle treated mice 25 days after treatment. In the RPP model, NU7441 in combination with CEP treatment resulted in 65% regression in average tumor volume compared with the CEP treated group (Fig. 3L). Similarly, in the RPM model, CEP treatment resulted in a 46.4% decrease in tumor volume 25 days after treatment, compared with 67.95% reduction in tumor burden compared to post-treatment in CEPN treatment at day 25 (Fig. 3M). We did not observe any significant changes in body weight in respect to treatment with either CEP or CEPN combination (Supplementary Fig. S4F).
We demonstrate that DNAPKcs inhibition augments the anti-tumor response of cisplatin-etoposide in immunocompetent SCLC models.
DNAPKcs inhibition induces GSK3β-mediated MYC degradation that leads to immune activation
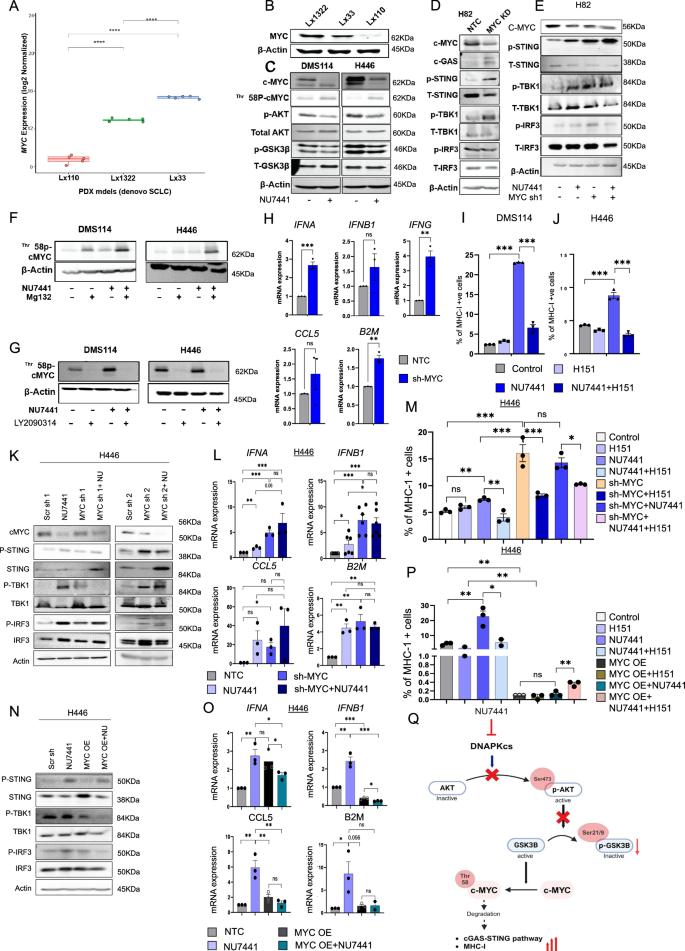
We next wanted to further explore mechanisms by which DNAPKcs inhibition might lead to immune activation in tumors under selective pressure of targeted therapy. As shown before, NU7441 caused better tumor reduction in two out of three PDX models, LX33 (NEUROD1 high) and LX1322 (POU2F3 high) (Fig. 2C–E). We have previously shown that NEUROD1- and POU2F3-driven SCLC models have high MYC expression36,37,38,39,40. We performed bulk transcriptomic analyses on tumors collected from the three PDXs (LX110, LX1322, LX33). In agreement with our previous findings, LX110 showed the lowest mRNA expression of MYC, followed by LX33, and LX1322 had the highest baseline expression of MYC (Fig. 4A), with consistent results observed for MYC protein expression by immunoblot analysis (Fig. 4B). Therefore, the baseline mRNA and protein expression of MYC correlated with the anti-tumor efficacy of DNAPKcs inhibition, i.e., we speculated that high MYC expression might predict improved efficacy of DNAPKcs inhibition.
A, B Boxplot (A) and Western blot (B) showing comparative MYC mRNA (A) and protein (B) levels in 3 PDX models of SCLC. Statistical significance from unpaired t test (n = 5): L x 110/L x 1322: 9.9e-06, L x 110/Lx33: 6.5e-07, L x 1322/Lx33: 5.8e-09. Boxplots defined as median (center), IQR (box), whiskers ( ± 1.5 x IQR). For Fig. 4A we have utilized our published publicly available dataset, GSE223372. C Western blot showing c-MYC, p-cMYC-Thr158, p-AKT-S473, total AKT, p-GSK3β-S21/9, total GSK3β in DMS114, H446 treated with 4 µM NU7441 (4 d). D, E Western blot showing c-MYC, cGAS, pSTING-S366, STING, pTBK1-S172, TBK1, pIRF3-S396, IRF3 with MYC sh-RNA ± NU7441 in H82. F, G Western blot of p-cMYC-Thr158, b-actin in DMS114, H446 ± NU7441 with Mg132 (F) or LY2090314 (G). H RT-PCR showing fold change of type I/II IFN, CCL5, B2M ± MYC sh-RNA in H82 (n = 3, biological replicates). P-values: IFNA < 0.001, IFNB1 = 0.226, IFNG = 0.002, CCL5 = 0.276, B2M = 0.001. I, J Flow cytometry showing %MHC-I + cells in DMS114 (I) and H446 (J) ± 4 µM NU7441 ± H151 (n = 3, biological replicates). P-values: (I) C/NU < 0.001, NU/NU + H151 < 0.001. J C/NU = 0.0007, NU/NU + H151 = 0.0003. K Western blot of MYC, cGAS, STING, TBK1, IRF3 ± 2 MYC sh-RNA ± NU7441 in H446. L RT-PCR in H446 ± sh-RNA ± NU7441 (n = 3 biological replicates, IFNB1 n = 6, biological replicates). P-values: IFNA, NTC/NU = 0.005, NTC/sh = 0.001, NU/Combo = 0.06, sh/combo = 0.39; IFNB1, NTC/NU = 0.005, NTC/sh < 0.001, NU/Combo = 0.01, sh/combo = 0.68; CCL5, NTC/NU = 0.007, NTC/sh = 0.02, NU/Combo = 0.51, sh/combo=0.3; B2M, NTC/NU = 0.002, NTC/sh = 0.007, NU/Combo = 0.82, sh/combo = 0.499. M. Flow cytometry in H446 ± NU7441 ± H151 ± MYC sh-RNA (n = 3, biological replicates). P-values: C/H151 = 0.083, C/NU = 0.0013, NU/NU + H151 = 0.0084, C/sh = 0.0024, NU/sh = 0.0055, sh/sh+H151 = 0.0079, sh/sh + NU = 0.38, sh+NU/sh + NU + H151 = 0.012. N Western blot showing MYC, cGAS, STING, TBK1, IRF3 ± MYC overexpression ± NU7441 in H446. O RT-PCR in H446 ± MYC OE ± NU7441 (n = 3, biological replicates). P values: IFNA, NTC/NU = 0.062, NTC/OE = 0.0118, NU/Combo=0.042, OE/combo=0.105; IFNB1, NTC/NU = 0.0029, NTC/OE < 0.0001, NU/Combo=0.0006, OE/combo=0.0109; CCL5, NTC/NU = 0.006, NTC/OE = 0.05, NU/Combo=0.008, OE/combo=0.17; B2M, NTC/NU = 0.05, NTC/OE = 0.17, NU/Combo=0.93, OE/combo=0.93. P Flow cytometry in H446 ± NU7441 ± H151 ± MYC OE. ANOVA and t-test for statistics (n = 3, biological replicates). P-values: C/H151 = 0.05, C/NU = 0.007, NU/NU + H151 = 0.04, C/OE = 0.001, NU/NU + OE = 0.003, OE + NU/OE + NU + H151 = 0.008, OE/OE + NU = 0.37. Q Schematic showing DNAPKcs inhibition activates cGAS-STING and MHC-I via MYC degradation in AKT-GSK3β–dependent manner. This figure was created in BioRender. Sen, T. (2025) https://BioRender.com/abxm28o.
DNAPKcs directly phosphorylates AKT, which in turn phosphorylates GSK3β to repress its activity41,42. The AKT-GSK3β pathway can regulate phosphorylation of MYC at Thr-58, resulting in the proteasomal degradation of MYC in SCLC42,43. We found that DNAPKcs inhibition led to a significant decrease in phospho-AKT (Ser473) (activating phosphorylation) and phospho-GSK3β (Ser21/9) (inhibitory phosphorylation) (Fig. 4C) in SCLC models. The decrease in GSK3β phosphorylation at the inhibitory Ser21/9 site indicated GSK3β activation. Active GSK3β enhances Thr-58 phosphorylation and ubiquitination of MYC, mediating rapid proteolysis42,44. Immunoblot analysis of DMS114 and H446 cells before and after 4 µM NU7441 treatment for 4 days showed a significant decrease in MYC protein expression after DNAPKcs inhibition in both cell lines (Fig. 4C). Furthermore, DNAPKcs inhibition by NU7441 treatment led to the increased expression of phospho-MYC (Thr-58) (Fig. 4C).
We next investigated the role of MYC on STING pathway activation. We performed shRNA-mediated MYC knockdown in H82 cells and examined the expression of cGAS–STING pathway proteins. MYC knockdown led to a notable upregulation of cGAS, p-STING, and p-TBK1 (Fig. 4D). Next, to determine if NU7441-mediated cGAS–STING activation was due to MYC depletion or was independent of MYC, we treated non-targeting control (NTC) and MYC-knockdown cells with NU7441 cells for 24 h. NU7441 treatment led to an appreciable increase in the cGAS–STING pathway markers, p-STING (Ser 396) and p-TBK1 (Ser 172) in both NTC and MYC KD cells (Fig. 4E).
Next, to confirm the proteasomal degradation of phosphorylated MYC, we treated the cells with MG132 (proteosome inhibitor) and NU7441. We observed a significant increase in phospho-MYC (Thr58) and total cMYC when proteasomal degradation was blocked (Fig. 4Fand Supplementary Fig. S5A). We also found an increase in PD-L1 expression in the proteasome degradation-inhibited condition in response to DNAPKcs inhibition in DMS114 and H446 cell lines (Supplementary Fig. S5B). Next, we aimed to confirm the involvement of GSK3β in the proteasomal degradation of MYC. We treated the two SCLC cell lines with LY2090314 (GSK3 inhibitor) in the presence or absence of NU7441. We observed almost complete depletion of phospho-MYC in the presence of GSK3 inhibitor, confirming GSK3 as the key molecular regulator of proteasomal degradation of MYC, thus affecting MYC stability (Fig. 4G). Further, investigating the protein level expression of total c-MYC we found increased expression in GSK3i treated condition in both DMS114 and H446 (Supplementary Fig. S5A). Interestingly, we further observed an increase in PD-L1 expression when GSK3 was pharmacologically inhibited (Supplementary Fig. S5B).
We next investigated the contribution of MYC in DNAPKcs mediated upregulation of IFNs and chemokines in SCLC models. shRNA-mediated MYC knockdown in H82 upregulated type I/II interferons (IFNA, IFNB, and IFNG); pro-inflammatory chemokine, CCL5; and HLA class-I gene, B2M (Fig. 4H).
Taken together, our results confirm that DNAPKcs inhibition activates GSK3β by inhibiting AKT phosphorylation, which decreases MYC stability, leading to activation of the cGAS/STING pathway and upregulation of IFNs and chemokines.
DNAPKcs inhibition leads to STING-mediated upregulation of MHC-I in SCLC models
One of the major contributing factors leading to the immunosuppressive phenotype of SCLC is the downregulation of MHC class I/II. The majority of SCLC tumors have been shown to have low/ no expression of MHC-I when IHC was performed which is in part due to epigenetic programming5. Hence, we next investigated the effect of DNAPKcs inhibition on HLA genes.
We found that, at baseline, high PRKDC expression is negatively correlated with the expression of two key MHC-I related genes, HLA-B and B2M in SCLC models (Supplementary Fig. S5C). Next, we performed bulk RNA sequencing of DMS114 and H446 cells pre- and post- NU7441 treatment. We observed an appreciable upregulation of HLA class I genes HLA-A, HLA-B, HLA-C, and B2M after NU7441 treatment in DMS114 and H446 cell lines (Supplementary Fig. S5D). This suggests that high PRKDC expression may predict MHC-I suppression and therapeutic targeting of DNAPKcs may increase expression of MHC-I related genes in SCLC.
Next, to assess the effect of DNAPKcs inhibition on the surface expression of the MHC-I in SCLC models, we treated the DMS114 and H446 cell lines with NU7441. Flow cytometry-based analysis indicated a significant increase in the percentage of MHC-I-positive cells post-NU7441 treatment in both models (Fig. 4I, J).
We next focused on the underlying mechanism of how DNAPKcs inhibition led to the upregulation of the MHC-I genes in SCLC models. DMS114 and H446 cells were treated with NU7441 with or without STING inhibitor, H151. We demonstrate that NU7441 treatment-mediated upregulation of MHC-I surface expression can be appreciably abrogated upon STING inhibition in SCLC (Fig. 4I, J). Since we found MYC-degradation mediated STING activation as a key mechanism of DNAPKcs mediated immune response, we next assessed the role of MYC in MHC-I induction in H82 cells. SCLC cells with either MYC knockdown or pharmacological inhibition of DNAPKcs were treated with STING inhibitor, H151. Flow cytometry-based analysis demonstrated an increase in the surface expression of MHC-I-upon DNAPKcs targeting or MYC knockdown (Supplementary Fig. S5E). However, NU7441 treatment had no added effect on the surface expression of MHC-I in cells which had MYC knockdown. Finally, STING depletion could successfully reverse the enhanced surface expression of MHC-I post NU7441-treatment or MYC- knockdown (Supplementary Fig. S5E). To further validate, we used a second shRNA sequence targeting MYC in H82 cell and treated the MYC KD cell with NU7441. Our data indicated increase in MHC-I positive cells with both NU7441 and MYC sh seq.2 treatment (Supplementary Fig. S5F). Most encouragingly NU7441 treatment upon MYC KD condition further increased the percentage of MHC-I positive cells compared to both only NU7441 treatment and MYC sh treatment alone (Supplementary Fig. S5F). Thus, our results demonstrate that MYC knockdown can induce MHC-I expression, and DNAPKcs inhibition increases MHC-I expression in a MYC-STING dependent manner.
As IFN signaling is a primary regulator of chemokine expression and MHC-I expression12,45, we further examined the expression of IFNs upon DNAPKcs inhibition in SCLC models. SCLC cells with or without MYC knockdown were treated with NU7441. The mRNA expression of IFNA, IFNB1, CCL5, and B2M was significantly enhanced in H82 cells upon treatment with NU7441 (Supplementary Fig. S5G). Similarly, MYC knockdown alone significantly increased the mRNA expression of IFNA and IFNB1, (Supplementary Fig. S5G), confirming our previous data (Fig. 4H). However, IFNA mRNA expressions did not increase further upon NU7441 treatment in SCLC cells (H82) with MYC knockdown (Supplementary Fig. S5G). This indicates that DNAPKcs inhibition increases the mRNA expression of IFNs in a MYC-dependent manner. However, IFNB1 expression further increased upon DNAPKcs inhibition in MYC knockdown cells, suggesting MYC-dependent and MYC-independent transcriptional regulation of IFNβ in SCLC models (Supplementary Fig. S5G).
Next, to ascertain the MYC mediated mechanism of cGAS-STING activation and MHC-I induction we knocked down MYC with 2 independent sh-RNA in H446 cell line. To determine if NU7441-mediated cGAS–STING activation was due to MYC depletion or was independent of MYC, we treated non-targeting control (NTC) and MYC-knockdown cells with NU7441 cells for 96 h. Western blot analysis shows significant decrease of MYC protein expression upon MYC shRNA treatment (Fig. 4K and Supplementary Fig. S6A). NU7441 treatment led to an appreciable increase in the cGAS–STING pathway markers, p-STING (Ser 396),p-TBK1 (Ser 172) and p-IRF3 in NTC (Fig. 4K). Interestingly, both MYC shRNA sequence showed a similar increase in the protein level expression of all the major molecular players of cGAS-STING pathway (Fig. 4K). Interestingly, NU7441 treatment upon MYC KD did not show further appreciable increase in the protein level (Fig. 4K), indicative of a threshold of cGAS-STING activation. Next, we studied the mRNA expression of type-I interferons along with chemokine CCL5 and B2M (Fig. 4L). Our q-rt-PCR results indicated increase in IFNA, IFNB1, CCL5 and B2M mRNA expression upon both MYC sh1 treated condition as well as NU7441 treated condition (Fig. 4L). Similar to our western blot results NU7441 treatment upon MYC KD failed to further induce these mRNA expressions with MYC shRNA sequence1. Interestingly, treating the cells with a second MYC sh RNA sequence indicated significant increase in the mRNA expression of IFNB1 and CCL5 upon combination treatment compared to NU7441 alone or MYC shRNA seq2 treatment alone (Supplementary Fig. S6B). Next, we investigated the effect of these 2 independent MYC shRNA sequence upon MHC-I surface expression. We observed increase in the percentage of MHC-I positive H446 cells both upon NU7441 treatment as well as 2 independent MYC shRNA treatment (Fig. 4M and Supplementary Fig. S6C) which were abrogated by STING inhibitor H151 co-treatment. Treating the MYC KD H446 cells with NU7441 did not show further increase in MHC-I positive cells with MYC shRNA sequence1 but for MYC shRNA seq2 (Fig. 4M and Supplementary Fig. S6C).
MYC over-expression reverses NU7441 mediated increase in cGAS-STING, interferon signaling as well as MHC-I induction
Next, we asked whether MYC over expression will reduce the immunogenic efficacy of NU7441 treatment. To investigate we over expressed MYC in H446 cells (Supplementary Fig. S6A). When we treated the non-targeting control and MYC overexpressing cells with NU7441 we observed a significant decrease in the protein expression of p-STING, p-TBK1 as well as the p-IRF3 upon MYC over expression (OE) (Fig. 4N). Furthermore, NU7441 treatment could not increase the p-TBK1 as well as p-IRF3 expression when MYC was over expressed in H446 cells (Fig. 4N). Similar decrease in mRNA expression of IFNA, IFNB1, CCL5 and B2M was observed upon MYC overexpression (Fig. 4O). NU7441 treatment could increase the mRNA expression of these genes in NTC but not when MYC was overexpressed (Fig. 4O).
Next we investigated the MHC-I suface expression phenotype of H446 cells in both NTC as well as MYC OE cells. Similar to our previous finding NU7441 treatment significantly increased the percentage of MHC-I H446 NTC cells (Fig. 4P). Interestingly, Upon MYC OE percentage of MHC-I cells decreased significantly (Fig. 4P). NU7441 treatment upon MYC overexpression could not significantly increase the MHC-I positive cell percentage indicating complete reversal of the immunogenic phenotype (Fig. 4P).
Taken together, our results indicate that DNAPKcs inhibition results in MYC degradation and subsequently in STING activation in SCLC. Furthermore, STING activation in response to DNAPKcs inhibition leads to MYC-dependent increase in type I/II interferons and MHC-I proteins [Fig. 4Q].
DNAPKcs inhibition decreases tumor growth, induces immune cell infiltration, and enhances response to ICB therapy in immunocompetent SCLC models
In SCLC clinical samples, PRKDC expression was significantly correlated with CD274 (PD-L1) expression (Supplementary Fig. S6D), suggesting that such immune signaling may also impact the tumor microenvironment and augment the antitumor immune responses of ICB in SCLC.
Therefore, we investigated whether the pharmacological inhibition of DNAPKcs by NU7441 leads to tumor regression and enhances the anti-tumor immune response of anti-PD-L1 antibody in two immunocompetent GEMMs of SCLC. We used cells derived from RPP and RPM models. RPP GEMM closely resembles the transcriptional landscape of the ASCL1-driven subtype of SCLC tumors, and RPM GEMMs closely resemble the transcriptional landscape of the NEUROD1-driven subtype of SCLC tumors. These two major NE subtypes of SCLCs are relatively more immunosuppressed with a low CD8 to regulatory T-cell ratio.
RPP or RPM subcutaneous tumor-bearing mice were randomized into 4 treatment arms: (1) vehicle, (2) PD-L1 alone (300 µg/animal, IP, 1/7) (3) NU7441 (10 mg/kg, intraperitoneal (IP) 5/7 days), and (4) NU7441 and PD-L1 combination treatment. In agreement with our previous work12,46 PD-L1 blockade did not show any anti-tumor effect in either RPP or RPM models when compared with the vehicle control (Fig. 5A and C).
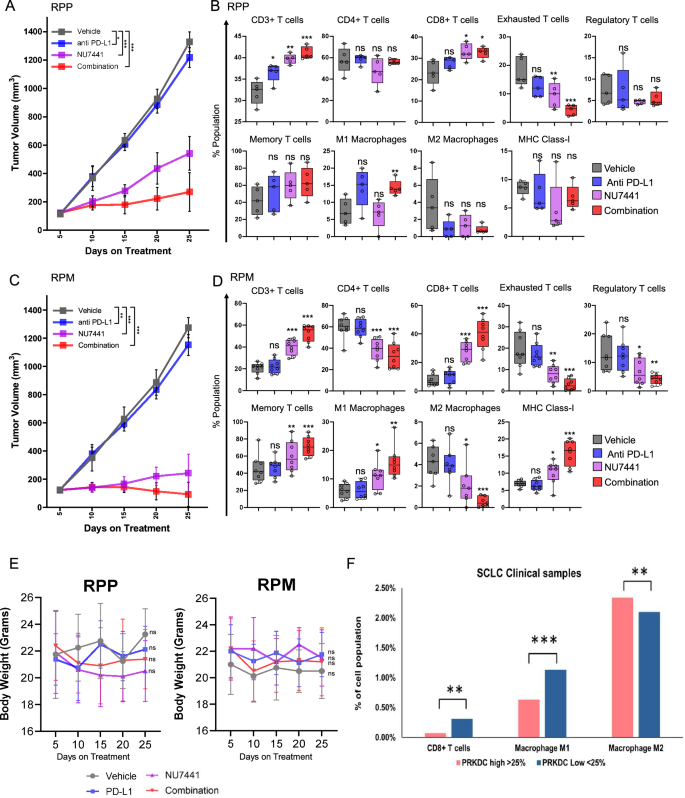
A, C Tumor growth curve data of 2 GEMMS: RPP (A) and RPM (C) in response to vehicle, anti-PD-L1 antibody, NU7441, or combination treatment. The data represent the means ± SD (n = 8 for vehicle and PD-L1 group and n = 10 for NU and combination treated group); P-values were calculated using linear mixed-effects regression models. For 5 A: P-values: vehicle/(PD-L1) = 0.030784159, vehicle/NU = 1.27E-14, vehicle/(NU-PD-L1) = 1.27E-14, (PD-L1)/NU = 1.27E-14, (PD-L1)/(NU-PD-L1) = 1.27E-14, NU/(NU-PD-L1) = 5.57E-13. For 5 C: P-values: vehicle/(PD-L1) = 0.00214126, vehicle/NU = 1.27E-14, vehicle/(NU-PD-L1) = 1.27E-14, (PD-L1)/NU = 1.27E-14, (PD-L1)/(NU-PD-L1) = 1.27E-14, NU/(NU-PD-L1) = 5.18E-06. B, D Boxplots of flow-cytometry analysis of harvested tumors showing % population of CD3+CD45+ total T cells, CD3+CD45+CD4+ helper T cells, CD3+CD45+CD8+ cytotoxic T cells, PD1+Tim3+ CD44high/CD62Low exhausted T cells, CD25+ve/FOXP3+ve regulatory T cells, CD45+CD3+CD8+CD44highCD62Llow effector-memory T cells; M1-macrophages, M2-macrophage population and MHC-class-I positive cells at the endpoint in RPP and RPM GEMMs. Each box represents the interquartile range (IQR) or the middle 50% of values; the horizontal line inside each box represents the median of that group, whiskers show the range of the data, and individual dots represent actual data points in each group. The data represents the means ± SE (n = 5); statistical summary is shown as unpaired 2-tailed students’ t test. E Graph showing changes in average body weight of RPP and RPM GEMM mice with different drug treatment. Statistical significance was calculated based on unpaired two-tailed students’ t test. ns, no significance; *, P < 0.05; **, P < 0.001; ***, P < 0.0001 (n = 8 for vehicle and PD-L1 group and n = 10 for NU and combination treated group). F Bar graphs showing the % cell population of CD8 + T cells, M1 macrophages and M2 macrophages in patients with SCLC with high (top 25%) versus low expression of PRKDC (bottom 25%) PRKDC high (n = 1296) and PRKDC low (n = 455). Data represented as mean ± SE of individual patient samples, T cell CD8 + : PRKDC-high ( > 25%) = 0.07%, PRKDC-low ( < 25%) = 0.31%, low/high ratio=4.43, P-value = 0.0004, M1 macrophage: PRKDC-high ( > 25%) = 0. 0.63%, PRKDC-low ( < 25%) = 1.13%, low/high ratio=1.79, P-value = 0.0, M2 macrophage: PRKDC-high ( > 25%) = 2.34%, PRKDC-low ( < 25%) = 2.10%, low/high ratio = 0.90, P-value = 0.0011. ns, no significance; *, P < 0.05; **, P < 0.001; ***, P < 0.0001.
In the RPP model, NU7441 treatment alone caused significant tumor regression, with mean tumor volume of 542 mm3 on day 25 (59.2% reduction in tumor burden compared with the vehicle group) (Fig. 5A). Most importantly, NU7441 in combination with anti-PD-L1 therapy reduced the tumor volume to an average of 269.6 mm3 on day 25, compared with the vehicle tumor volume of 1328.75 mm3 on day 25 post-treatment (79.7% reduction in tumor burden) (Fig. 5A). Similar results were obtained for RPM GEMM. NU7441 treatment alone showed a significant reduction in tumor growth, reaching an average tumor volume of 242 mm3 compared with 1275 mm3 in the vehicle-treated group on day 25 (80% reduction in tumor volume) (Fig. 5C). The combination treatment of NU7441 with anti-PD-L1 therapy showed remarkable tumor regression, reaching a volume of 92.8 mm3 post day 25 (Fig. 5C), indicating almost a 92% tumor regression at day 25 post-treatment compared to vehicle or PD-L1 alone. In the RPM models, 1 out of 10 mice showed complete tumor regression on day 25 post-treatment, and 6 out of 10 mice showed less tumor burden than on day 5 (first tumor measurement).
We next assessed the effect of NU7441, either as a single agent or in combination with PD-L1 blockade, on immune subsets in resected tumors using distinct T cell and antigen presenting cell panels (Supplementary Fig. S7A, B). Multicolor flow cytometry demonstrated that NU7441 treatment, either as a single agent or in combination with PD-L1 blockade, led to a significant increase in CD3+ total T-cells, and CD8+ cytotoxic T-cells (Fig. 5B, D) in RPP and RPM GEMMs. Interestingly, PD-1 + /TIM3+ exhausted T-cells and CD25 + /FOXP3+ regulatory T-cells, known immunosuppressive immune subsets, were significantly decreased in both the RPP and RPM models after NU7441 and NU7441 and PD-L1 combination treatment (Fig. 5B, D). We further observed that NU7441 + /- anti-PD-L1 treatment led to an increase in the CD62L/CD44 + memory-effector T-cells in the RPP and RPM models (Fig. 5B, D). Taken together, our data showed that NU7441 treatment, either as a single agent or in combination with a PD-L1 antibody, significantly enhanced CD8 + T-cell infiltration and decreased T-cell exhaustion and levels in two unique immunocompetent mouse models of SCLC.
Tumor-associated macrophages (TAMS), including M1 and M2 macrophages, are important components of the TME. Pro-inflammatory M1 macrophages play an antitumor role, whereas anti-inflammatory M2 macrophages have been reported to play immunosuppressive roles in lung cancer47. Multicolor flow cytometry of tumors from RPP GEMMs demonstrated that NU7441 treatment in combination with PD-L1 blockade significantly increased M1 macrophages (Fig. 5B). Interestingly, in the MYC high RPM GEMMs, the M1 macrophage population was upregulated upon NU7441 treatment alone or in combination of NU7441 and PD-L1 (Fig. 5D). Moreover, treatment with NU7441 alone or in combination with anti-PDL-1 significantly decreased the percentage of M2 macrophages in the RPM model (Fig. 5D). In addition, we observed a significant increase in MHC-I in NU7441 treated tumors of the RPM model (Fig. 5D). Encouragingly, none of the treatment groups showed any significant toxicity as demonstrated by the non-significant changes in body weight in both RPP and RPM GEM models (Fig. 5E).
Next, we demonstrate that in 423 SCLC real world clinical samples that PRKDC low tumors have significantly higher percentage of CD8 + T-cells, M1 macrophages as compared to tumors expressing high PRKDC (Fig. 5F). On the other hand, M2 macrophage was significantly decreased in PRKDC low SCLC patient tumors (Fig. 5F).
Taken together our data shows that DNAPKcs inhibition alone or in combination with PD-L1 blockade leads to significant tumor regression and NU7441 treatment augments the anti-tumor immune response in two immunocompetent SCLC GEMMs. Moreover, DNAPKcs inhibition alone or in combination with PD-L1 blockade led to an increase in cytotoxic T-cells, M1 macrophages and MHC-I in SCLC in vivo.
STING depletion reverses the DNAPKcs-mediated antitumor immune response, emphasizing the critical role of STING activation in SCLC
Our in vitro studies showed NU7441 treatment-mediated activation of the innate immune cGAS–STING pathway in a MYC-dependent manner. Hence, next, we assessed the role of the STING pathway in NU7441 treatment-mediated anti-tumor immune response in SCLC models. We silenced STING in RPP and RPM cells (Supplementary Fig. S7C–F). shRNA mediated STING-silenced RPP and RPM cells were injected into immunocompetent mice and such tumor bearing mice were then randomized into four treatment arms: (1) vehicle, (2) PD-L1 alone (300 µg/animal, IP, 1/7), (3) only NU7441 (10 mg/kg, IP 5/7 days), and (4) combination of NU7441 and PD-L1 treatment.
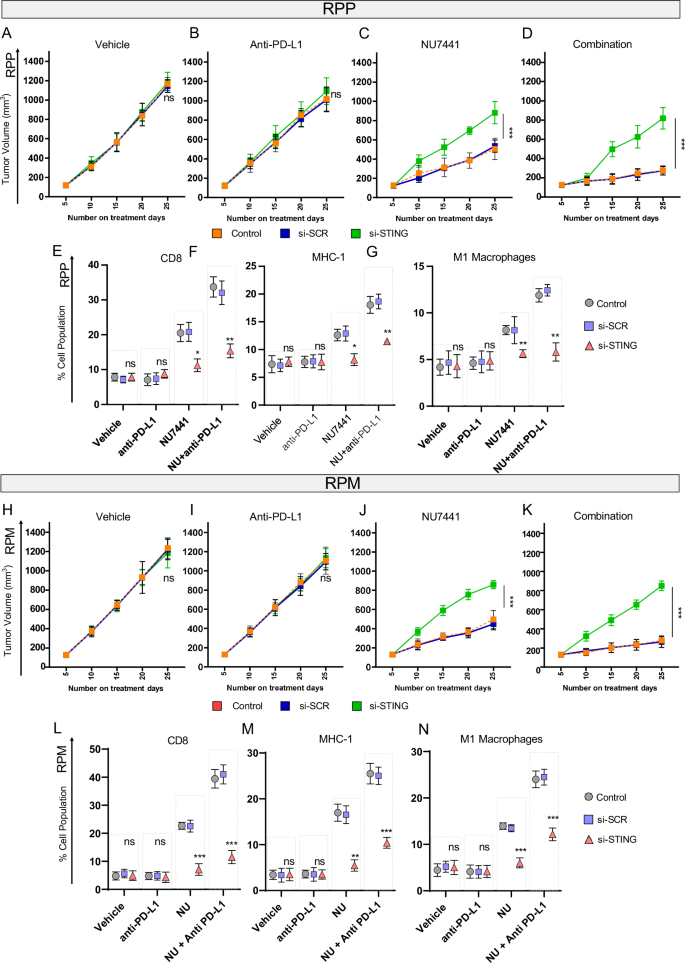
In both RPP and RPM tumors harboring knockdown of STING, we observed a significantly decreased degree of tumor shrinkage relative to control arms (Fig. 6A, B, H, I) upon NU7441 treatment. Moreover, contrary to scrambled control (SCR) tumors, all tumors with STING knockdown (shSTING-RPP or shSTING RPM) progressed even with combined NU7441 with or without PD-L1 blockade (Fig. 6C, D and Fig. 6J, K). Our finding confirms the vital role of the STING pathway in DNAPKcs-mediated anti-tumor response in two immunocompetent SCLC models.
A–D Tumor growth-curve data RPP GEMMs in response to vehicle, anti-PD-L1 antibody, NU7441, or combination in the presence of vehicle, scrambled RNA, and STING-sh-RNA. The data represents means ± SD (n = 6); statistical summary shown using linear mixed-effects regression models. P-values for 6 C: Control/SCR = 0.501200604, control/STING KD = 0, SCR/STING KD = 7.34E-11. P-values for 6D: Control/SCR = 0.983380796, control/STING KD = 1.46E-12, SCR/STING KD = 1.46E-12. E–G Plots showing percentage population of CD-8 + T cells, MHC-class-I positive cells, M1-macrophages in the resected tumors from the RPP model in response to vehicle, anti-PD-L1 antibody, NU7441, or combination in the presence of vehicle, scrambled RNA, and STING-sh-RNA. Data represents means ± SEM (n = 5), statistical summary is shown as per unpaired 2-tailed students’ t test. ns, no significance; *, P < 0.05; **, P < 0.001; ***, P < 0.0001. H–K Tumor growth-curve data of GEM model RPM in response to vehicle, anti-PD-L1 antibody, NU7441, or combination in the presence of vehicle, scrambled RNA, and STING-sh-RNA. Data represents means ± SD (n = 6); statistical summary is shown using linear mixed-effects regression models. P-values for 6H: Control/SCR = 0.967537553, Control/STING KD = 0.698809929, SCR/STING KD = 0.839526201. P-values for 6I: Control/SCR = 0.898053137, control/STING KD = 0.955496087, SCR/STING KD = 0.746875387. P-values for 6 J: Control/SCR = 0.467944098, control/STING KD = 0.0, SCR/STING KD = 0.0. P-values for 6 J: Control/SCR = 0.741193564, control/STING KD = 1.46E-12, SCR/STING KD = 1.46E-12. L–N Data showing percentage population of CD-8+ T cells, MHC class-I positive cells, M1-macrophages in the resected tumors from the RPM model in response to vehicle, anti-PD-L1 antibody, NU7441, or combination in the presence of vehicle, scrambled RNA, and STING-sh-RNA. Data represents means ± SEM (n = 5); statistical summary is shown students’ t test. ns, no significance; *, P < 0.05; **, P < 0.001; ***, P < 0.0001.
The knockdown of STING (TMEM173) was confirmed at the beginning of the treatment in RPP and RPM cells by qRT-PCR (Supplementary Fig S7C, E). STING knockdown was also confirmed at the end of the experiment in tumors resected from all treatment arms. RT-PCR analysis shows significantly lower expression of STING at day 20 in all treatment groups (Supplementary Fig. S7D, F).
Next, to assess the role of STING depletion on immune subsets that were significantly modulated upon NU7441 treatment (i.e., CD8, MHC-I and M1 macrophages) we performed multicolor flow cytometry of resected tumors at Day 20. STING knockdown reversed the increase in CD8 + T-cells upon NU7441 treatment with or without PD-L1 blockade in both RPP and RPM models (Fig. 6E, L). Furthermore, STING knockdown also led to the abrogation of NU7441-mediated upregulation of MHC-I and M1 macrophages in RPP and RPM models (Fig. 6F, G, M, N).
Taken together, we demonstrate that inhibition of DNAPKcs leads to a significant increase in CD8+ cytotoxic T-lymphocytes, MHC-I, and M1 macrophages in a STING-dependent manner and STING depletion can successfully abrogate the anti-tumor immune response of NU7441 with or without PD-L1 blockade.
The anti-tumor immune response of DNAPKcs inhibition can be reversed by CD8 depletion
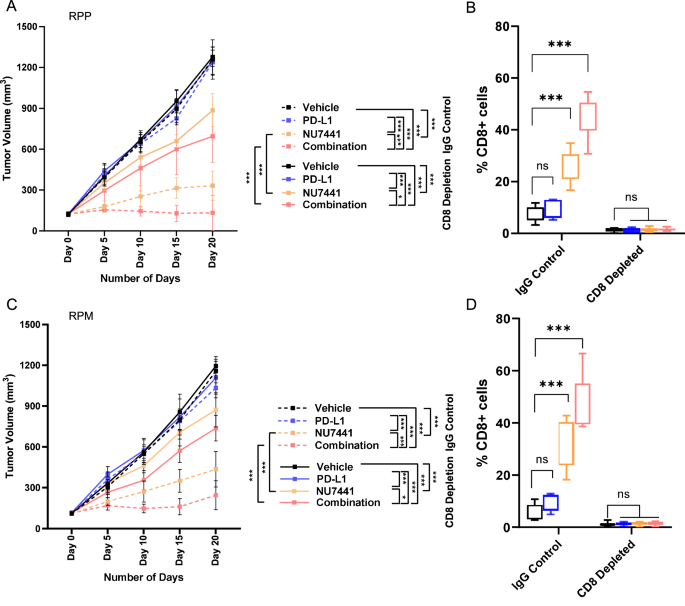
NU7441 treatment with or without PD-L1 blockade increased the cytotoxic T-cells. Hence, we next tested the role of CD8 cells in the NU7441 effect with or without PD-L1 inhibition-mediated antitumor response. RPP and RPM subcutaneous tumors with or without CD8 depletion were randomized into groups and treated with NU7441 with or without PD-L1 blockade. As before, NU7441 treatment with or without PD-L1 blockade in IgG antibody control groups led to significant tumor regression in both RPP and RPM models (Fig. 7A, C). However, as shown in Fig. 7A, C, in CD8-depleted mice NU7441 treatment with or without PD-L1 blockade led to tumor growth compared to mice treated with an IgG antibody control. CD8 levels in both RPP and RPM models were confirmed by flow cytometry which demonstrates successful depletion of CD8 in RPP and RPM models (Fig. 7B, D).
A, C Tumor growth curves of mean tumor volume +/− SD from vehicle, anti-PD-L1 alone (300 µg/ Animal, I.P. route), NU7441 (10 mg/kg, i.p. 5/7 days), and NU7441+anti-PD-L1 treatment groups in RPP and RPM GEMM mice in IgG control and CD8-depleted (anti-CD8, 200 μg, 2/7 days) groups. The data represent the means ± SD (n ≥ 8); statistical summary is shown using linear mixed-effects regression models. For 7 A: n = 8 for vehicle, 7 for PD-L1, 8 for NU, and 9 for combination treated groups. For 7B: n = 7 for vehicle, PD-L1, NU, and n = 8 for combination treated groups. ns, no significance; *, P < 0.05; **, P < 0.001; ***, P < 0.0001. B, D Percentage of CD8+ T cells measured by flow-cytometric analysis in single-cell suspensions prepared from tumors (n = 6, individual mice tumor samples, biological replicate) in CD8-depleted groups 20 days post-treatment as compared to IgG control groups. Each box represents the interquartile range (IQR) or the middle 50% of values; the horizontal line inside each box represents the median of that group, whiskers show the range of the data, and individual dots represent actual data points in each group. The data represent the means ± SD (n = 6); statistical summary shows students’ t test following 1-way ANOVA test. ns, no significance; *, P < 0.05; **, P < 0.001; ***, P < 0.0001.
Therefore, we demonstrate that NU7441 treatment leads to significant tumor regression through CD8+ cytotoxic T-cell infiltration and that depletion of CD8 can reverse the anti-tumor effect by NU7441 with or without anti-PD-L1 antibody.
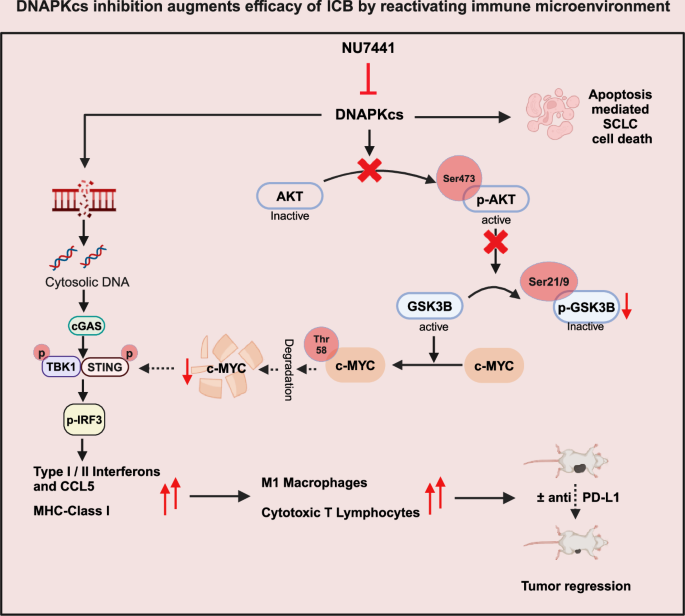
Taken together, we demonstrate that pharmacological inhibition of DNAPKcs leads to apoptosis, MYC degradation and micronuclei formation. Downregulation of MYC and micronuclei formation can lead to cGAS/STING pathway activation, interferon pathway upregulation, increase in pro-inflammatory chemokines and MHC-I. DNAPKcs inhibition with or without PD-L1 blockade leads to significant anti-tumor response in SCLC models (Fig. 8).
We demonstrate that pharmacological inhibition of DNAPKcs leads to apoptosis, MYC degradation, and micronuclei formation. Downregulation of MYC and micronuclei formation can lead to cGAS/STING pathway activation, interferon pathway upregulation, and increased pro-inflammatory chemokines and MHC-I. DNAPKcs inhibition with or without PD-L1 blockade leads to significant anti-tumor response in SCLC models. This figure was created in BioRender. Sen, T. (2025) https://BioRender.com/k1za30a.
Discussion
There is an urgent need to identify more effective and durable therapeutic options to improve outcomes of patients with SCLC, the most lethal type of lung cancer. We and others have shown that the induction of the innate immune response pathway is a promising strategy to activate the immune microenvironment and augment the antitumor immune response of ICB in immunologically “cold tumors” like SCLC. In the present work, we identify that the master regulator of the NHEJ pathway, DNAPKcs, is a potential repressor of immune response and define DNAPKcs inhibition as a potential therapeutic approach to activate the antitumor immune response of immunotherapy in SCLC.
DNAPKcs, the cell cycle regulator involved in DSB repair and replication stress response is upregulated in SCLC, consistent with the high genomic instability of these high-grade NE tumors. Our study demonstrated that genetic and pharmacological inhibition of DNAPKcs induces micronuclei formation, immunogenic double stranded DNA (dsDNA), causing tumor cell–intrinsic innate immune signaling. Activation of the cGAS-STING pathway induced interferon signaling, and triggering these tumor-intrinsic immune modulatory pathways significantly improve the efficacy of ICB treatment in SCLC. Notably, DNA damage, as evidenced by elevated p-H2AX levels, was detectable as early as 1 day post-treatment (data not shown). Given the rapid proliferation rate of SCLC cells, we hypothesize that micronuclei formation—an early trigger of cGAS activation—occurs within 1 to 2 days of treatment. Although prior studies have shown that micronuclei formation may not launch an immunological effect48, our findings strongly suggest that dsDNA formation and the co-localization of cGAS to the micronuclei after DNAPKcs depletion directly contribute to the induction of innate immune responses and replication stress in cancer cells, highlighting its potential as a target to enhance antitumor immunity and boost the antitumor effect of ICB. Given that cytoplasmic double-stranded DNA is actively cleared by cellular mechanisms such as DNases (e.g., DNase2 and TREX1), autophagy, and lysosomal degradation, we do not expect a continuous time-dependent accumulation of micronuclei. Rather, we believe the key factor is the presence of cytoplasmic ds-DNA at a threshold sufficient to trigger cGAS-STING signaling. Our study strongly suggests that targeting the NHEJ pathway might be a therapeutic strategy to effectively enhance antitumor immunity while concurrently inducing DNA damage and compromising cell viability in SCLC. Multiple contradictory reports have suggested close association of DNAPKcs with the cGAS-STING pathway. In some reports, DNAPKcs have been shown to be essential for cGAS-STING dependant IFN-I response during stimulation with exogenous viral DNA49. On the other hand, DNA-PK has been known to phosphorylate cGAS to suppress its enzymatic activity. Thus, DNA-PK deficiency reduces cGAS phosphorylation and promotes antiviral innate immune responses50. In our study, we found DNAPKcs inhibition leads to activation of cGAS_STING mediated anti-tumor immune response using both in vitro as well as in vivo GEM models.
SCLC is characterized by an almost ubiquitous inactivation of TP53 and RB1, key tumor suppressor genes and crucial regulators of replication stress response and cell cycle2. Moreover, in approximately 20% of SCLCs, members of the MYC family of oncogenes (MYCL, MYCN, and MYC) are amplified and are associated with worse survival10. Paradoxically, although SCLC has one of the highest somatic mutation burdens among all malignancies, it is highly immunosuppressed, which explains the modest response of SCLC to ICB. Therefore, continuing to identify effective treatment strategies to improve the effect of immunotherapy in SCLC remains an urgent unmet need. High chromosomal instability and mutational burden render SCLC tumors more reliant on DNA damage repair systems for proliferation. We have previously shown that targeting members of the DNA damage response pathway, such as PARP, CHK1, WEE1 and ATR2,12,14,15,46,51,52 can be an effective strategy to reduce SCLC tumor growth. In this report, we demonstrate the role of the NHEJ pathway as a targetable vulnerability in SCLC. SCLC has significantly higher expression of DNAPKcs, which predicted worse overall survival and increased resistance to chemotherapy and targeted therapy of SCLC tumors. This makes DNAPKcs an important target in SCLC to induce cell death and overcome drug resistance. To more deeply profile the potential of DNAPKcs inhibition in SCLC, we treated an array of PDXs derived from tumors of different SCLC subtypes. Inhibiting DNAPKcs showed a remarkable anti-tumor effect in POU2F3 and NEUROD1- driven PDXs without evident additional toxicity. Notably, MYC expression is high in NEUROD1- and POU2F3-driven SCLC subtypes and MYC acts as a pleiotropic regulator of multiple oncogenic processes53. In addition, DNAPKcs inhibition increased the anti-tumor efficacy of chemotherapy in immunocompetent SCLC models, suggesting that DNAPKcs inhibition may be an interesting therapeutic approach in combination with chemotherapeutic agents in SCLC. DDR inhibitors have been previously shown to augment the anti-tumor immune response of ICB in SCLC preclinical models12,14,15,46,51. Importantly, we and others have shown that targeting the DDR proteins, or DNA damage, induces dsRNA and R-loop production, increases IFN response and activates the immune microenvironment12,14,15,46,51. Interestingly, we found that DNAPKcs depletion in SCLC leads to micronuclei formation, and the co-localization of dsDNA and cGAS, triggering the cGAS-STING pathway. Mechanistically, we provide evidence that DNAPKCs inhibition leads to cGAS–STING pathway activation, transcriptional upregulation of type I and II interferons and the recruitment of pro-inflammatory chemokines in SCLC models, representing DNAPKcs as a targeted vulnerability for SCLC cells.
The primary reasons that contribute to inherent immune evasion and poor response of SCLC to immunotherapy are relatively low levels of infiltrating T-cells and reduced antigen presentation. Our data in two immunocompetent SCLC mouse models indicate that pharmacological inhibition of DNAPKcs induced significant tumor regression as a single agent, and led to the recruitment of multiple immune cells into the TMA, including CD8+ T-cells, M1 macrophages and MHC-I, dramatically sensitizing SCLC tumors to anti–PD-L1 immunotherapy. Pro-inflammatory M1 macrophages play an antitumor role, whereas anti-inflammatory M2 macrophages play immunosuppressive roles in lung cancer47. In the used mouse models, STING depletion could reverse the anti-tumor effect of NU7441 with or without PD-L1 blockade. Furthermore, STING depletion also reversed the DNAPKcs inhibition-mediated upregulation of MHC-I and M1 macrophages in SCLC immunocompetent models. Taken together, our study suggests DNAPKcs as a therapeutic target for SCLC that potentiates immune activation and improves the efficacy of anti-PD-L1 ICB therapy in a STING-dependent manner. It is important to note that, antitumor efficacy by NU7441 contributed through two complementary mechanisms: one through its cell intrinsic mechanism and second through activating immunogenicity. Considering RPP and RPM immunocompromised models showed tumor regression of ~38% and 45% whereas same models in immune-competent set up showed a ~ 59% and ~ 80% tumor reduction respectively. This indicates cell intrinsic, and cell extrinsic immune mechanism acts complementarily to provide a better anti-tumor efficacy in our in vivo models. Furthermore, it is important to mention the context-dependent role of CD8+ T cells in antitumor efficacy. IN vehicle or anti-PD-L1 treated groups, we observed minimal impact of CD8 + T cell depletion due to the minimal anti-tumor activity of these treatments in our SCLC model. Whereas, in the NU7441 plus anti–PD-L1 group, where tumor regression was robust, CD8+ T cell depletion markedly reduced therapeutic efficacy, confirming the essential role of CD8 + T cells in mediating response to DNAPKcs inhibition combined with checkpoint blockade.
We next investigated the underlying mechanism of DNAPKcs mediated STING modulation in SCLC. Our data shows that MYC stabilization by proteasomal inhibition is a potential mechanism of DNAPKcs-mediated immune evasion in SCLC. MYC is a transcription factor related to cancer cell proliferation54, previously implicated in immune evasion in multiple cancer types55, and as a driver of the NE differentiation53. MYC amplification is a crucial mechanism in immune evasion and chemoresistance43,56,57. In agreement with previous data, MYC expression was higher in immunosuppressed SCLCs and suppressed by DNAPKcs inhibition, concurrent to activation of the immune pathways. MYC is often termed “un-druggable” owing to its critical role and complex protein structure11. DNAPKcs depletion affected MYC protein stability via modulating proteasomal degradation. DNAPKcs inhibition decreased pAKT_Ser473 and pGSK3β_Ser 21/9. DNAPKcs directly phosphorylates AKT, which phosphorylates GSK3β to repress its activity31,42. Active GSK3β enhances Thr-58 phosphorylation and ubiquitination of MYC, mediating rapid proteolysis44. We show that DNAPKcs inhibition increased phosphorylation of MYC (Thr-58), which leads to MYC degradation. Therefore, DNAPKcs inhibition may lead to the activation of DNA damage repair pathways and MYC degradation and consequently activation of the immune response in SCLC. Using two independent shRNA sequence against MYC our results provide further evidence that MYC downregulation can induce activation of the cGAS–STING pathway and transcriptional induction of type I/II interferons. Furthermore, we provide compelling evidence that overexpression of MYC in our cell line model completely reversed the immunogenic phenotype upon NU7441 treatment. Given the reported role of MYC-mediated downregulation of the STING pathway, interferons, and MHC-I58, we propose that NU7441 mediates MYC destabilization as a crucial mechanism of immune activation and the appreciable antitumor efficacy of DNAPKcs inhibition. Our in vivo tumor regression and flow cytometry-based immune studies also showed a better response in the MYC-amplified RPM GEM models, further strengthening our hypothesis. Now it is important to note, the RPM immune competent GEM model harbors the MYCT58A mutation, which stabilizes MYC by preventing phosphorylation at threonine 58, a key step required for proteasomal degradation. Despite this stabilization, treatment with NU7441 leads to a robust activation of the STING pathway, as evidenced by significant TIME remodeling and anti-tumor efficacy. This suggests that although MYC is mutated to resist degradation, DNAPKcs inhibition by NU7441 may still reduce MYC levels, possibly through alternative pathways. These findings highlight that STING-IFN pathway activation and immune remodeling can occur even in the presence of stabilized but suppressed levels of MYC, further supporting a multifaceted mechanism of action for DNAPKcs inhibition. Our findings clearly demonstrate that DNAPKcs inhibition by NU7441 induces immune activation in a MYC-dependent manner across MYC-high SCLC models. But it is important to note that MYC mRNA levels in LX33 and LX1322 do not fully correspond with protein levels. This observation highlights the well-established concept that mRNA expression does not always predict protein abundance due to several layers of regulation. mRNA stability, translational efficiency, and post-translational modifications all play critical roles in determining final protein levels. In addition, protein turnover rates and degradation pathways further contribute to these discrepancies.
Although, multiple DNA damage inhibitors have been shown to have promising results in pre-clinical studies but only few have demonstrated significant clinical efficacy. Among the multiple reasons a major concern being off-target effect of the drugs. In our study, though NU7441 works selectively on DNAPKcs but at a higher concentration it can inhibit PI3K and mTOR. Though investigating its effect on PI3K and mTOR proteins was out of the scope of this study, PI3K and mTOR is involved in multiple signaling pathways. One of the discrepancy found in this study in respect to pharmacological and genetic inhibition of DNAPKcs was protein expression of total STING. Given the dynamics signaling proteins, the stability of STING not only depends on transcriptional and translational induction of STING but also degradation mechanism. ESCRT-mediated micro-autophagy has been reported to be a significant contributor to STING degradation pathway59,60. Both mTOR and PI3K has been known to play crucial role in initiating autophagy and lysosome mediated degradation of long lived / impaired proteins. Thus, it is possible that upon NU7441 mediated pharmacological inhibition of DNAPKcs mTOR or PI3K signaling may play a role in decreasing STING expression which is not found in case of siRNA mediated genetic inhibition of DNAPKcs. In another, surprising result, especially with MYC shRNA sequence1, we did not observe any significant increase in the expression of cGAS-STING pathway proteins, or type-I interferons/chemokines, as well as MHC-I positive cell population upon NU7441 treatment. From our results, we concluded it can be MYC shRNA sequence specific event or there can be threshold of increase of these proteins and mRNAs. With an already increased phenotype NU7441 may not significantly upregulate these molecular players significantly further. But deciphering the exact mechanism behind this inconsistency was out of the scope of the current manuscript and provides an interesting future direction of this work. Though, subcutaneous models remain a widely used and informative approach for mechanistic and therapeutic studies in small cell lung cancer, especially given the difficulty of modeling this aggressive and nonsurgical disease in its native environment. We recognize that subcutaneous tumors do not fully capture the immune and stromal context of lung tumors that arise in their native site.
In summary, our study identified DNAPKcs as a vulnerability in SCLC, an immunologically ‘cold’ tumor with inherent vulnerability in the replication stress response pathway. We show PRKDC expression predicts improved OS and PFS as well as response to immunotherapy. Furthermore, we show that targeting DNAPKcs leads to DNA damage response and apoptosis, and the pharmacological or genetic perturbation of DNAPKcs leads to MYC-mediated innate immune cGAS-STING pathway activation as well as induction of IFN signaling. DNAPKcs abrogation leads to STING-mediated increased cytotoxic T-cell infiltration, pro-inflammatory M1 macrophage population, and MHC-I upregulation. Moreover, we found that these tumor-intrinsic effects can turn immune ‘cold’ tumors into immune ‘hot’ tumors, and consequently, DNAPKcs inhibition dramatically enhances the anti-tumor immune response of ICB. Our findings highlight the role of DNAPKcs as a potential therapeutic target to augment the response of ICB in SCLC and potentially other immunologically ‘cold’ tumors.
Methods
All studies conducted as part of this work were reviewed by the institutional committee of the Icahn School of Medicine at Mount Sinai. The animal study was approved by the Institutional Animal Care and Use Committee (IACUC). The identifying number of the approved protocol is 13-07-007. The maximal tumor size/burden permitted by the IACUC ethics committee was 1600 mm3. We confirm that this maximal tumor size/burden was never exceeded for the immune competent models, whereas given the highly aggressive nature of these specific small-cell lung cancer PDX models specially LX110 and LX1322, the committee granted a model-specific exception allowing a modest extension of the tumor endpoint criteria. This approval was provided strictly for the LX110 and LX1322 cohorts and was contingent upon enhanced monitoring to ensure animal welfare was fully maintained.
Clinical samples cohort
Real-world patient tumor specimens (N = 179,508) were submitted for comprehensive molecular profiling to a Clinical Laboratory Improvement Amendments (CLIA)-certified laboratory (Caris Life Sciences; Phoenix, AZ). The present study was conducted in accordance with the guidelines of the Declaration of Helsinki, Belmont Report, and US Common Rule and in compliance with policy 45 CFR 46.101(b). The study was conducted using retrospective, de-identified clinical data, and patient consent was therefore not required. All statistical analyses were performed with JMP V13.2.1 (SAS Institute) [Detailed in Supplementary Tables S1–S4]. This study was conducted in accordance with guidelines of the Declaration of Helsinki, Belmont report, and U.S. Common rule. In keeping with 45 CFR 46.104(d)(4), this study was performed utilizing retrospective, deidentified clinical data, and is considered IRB exempt with waivers of patient informed consent. Waiver of patient consent and exempt status was determined by the WCG IRB.
RNA Whole transcriptome sequencing (WTS)
WTS was performed by a hybrid-capture method to pull down the full transcriptome from full formalin-fixed paraffin-embedded (FFPE) tumor samples using the Agilent SureSelect Human All Exon V7 bait panel (Agilent Technologies, Santa Clara, CA) and the Illumina NovaSeq platform (Illumina, Inc., San Diego, CA). FFPE specimens underwent pathology review to discern the percent tumor content and tumor size; a minimum of 10% tumor content in the area selected for microdissection was required to enable enrichment and extraction of tumor-specific RNA. Qiagen RNA FFPE tissue extraction kit was used for extraction, and the RNA quality and quantity were determined using the Agilent TapeStation. Biotinylated RNA baits were hybridized to the synthesized and purified cDNA targets, and the bait-target complexes were amplified in a post-capture PCR reaction. The resultant libraries were quantified and normalized, and the pooled libraries were denatured, diluted, and sequenced. Raw data was demultiplexed using the Illumina DRAGEN FFPE accelerator. FASTQ files were aligned with the STAR aligner (Alex Dobin, release 2.7.4a, GitHub). A full 22,948-gene dataset of expression data was produced by the Salmon, which provides fast and bias-aware quantification of transcript expression21. BAM files from the STAR aligner were further processed for RNA variants using a proprietary custom detection pipeline. The reference genome used was GRCh37/hg19, and analytical validation of this test demonstrated ≥ 97% Positive Percent Agreement (PPA), ≥ 99% Negative Percent Agreement (NPA), and ≥ 99% Overall Percent Agreement (OPA) with a validated comparator method.
Gene expression profiling for SCLC subtyping
For stratification of patient samples (N = 944) into SCLC subtypes, RNA expression values for established and putative lineage-defining transcription factors ASCL1 (A), NEUROD1 (N), POU2F3 (P), and YAP1 (Y) were standardized to Z-scores. Samples with a single positive Z-score among A/N/P/Y were assigned to the respective gene-associated subgroups, samples with multiple positive Z-scores were classified as ‘Mixed’, and samples with negative Z-scores for all 4 genes were classified as ‘Transcription Factor-Negative’ (TF-Neg).
Cell culture
Roswell Park Memorial Institute (RPMI) 1640 medium (gibco, #A10491-01) supplemented with 10% Fetal Bovine Serum, penicillin (100 U/mL), streptomycin (50 g/mL) and 2 mM L-Glutamine was used to culture SCLC cells that were obtained from the American Type Culture Collection (ATCC), [cell line details have been provided in Supplementary Table S5]. The GEMM-derived SCLC cell lines derived from a conditional triple-knockout model of SCLC, Trp53−/−, p130−/−, Rb1−/− (RPP) were provided by J. Sage, Stanford University California. Rb1−/−, Trp53−/−, and MYCT58A (RPM) cells were provided by T. G. Oliver, University of Utah. We inclubated the cells in 5% carbon dioxide at 37 °C with a relative humidity above 90%. We tested and authenticated the cell lines via short tandem repeat profiling (DNA fingerprinting) and routinely tested for mycoplasma species before any we performed any experiments. In general, the cells were used within the first 3-5 passages for experiments.
Drugs used in this study
We obtained NU7441 (Selleckchem, # S2638) and Nedisertib (Selleckchem, # S8586) from Selleckchem. We also obtained STING inhibitor H-151 from Selleckchem (Catalog No.S6652).
Cell viability assay
SCLC cell lines were plated in 96-well plates (2000 cells/well) in triplicate for each concentration. Cells were kept in complete RPMI media containing 10% fetal bovine serum, penicillin (100 U/mL), and streptomycin (50 g/mL) and 2 mM L-Glutamine overnight, and then they were treated with dimethyl sulfoxide (DMSO) for control or different concentrations of NU7441 for 110 hours (5 Days). The cell viability was assessed with CellTiter-Glo luminescent cell viability assay reagent (Promega, Madison, WI) according to the manufacturer’s protocol Half-maximal inhibitory concentrations (IC50s) were calculated using GraphPad Prism Ver. 9.0 (GraphPad Software, Inc., San Diego, CA, USA).
Animal studies
The Institutional Animal Care and Use Committee approved all animal experiments. We measured all tumors twice weekly using calipers. To calculated volume, we used the formula width2 x length x 0.5. Twice per week, we measured body weights. When tumor volumes exceeded 2 cm in any dimension, suffered consistent body weight loss exceeding 10%, or in the case of severe tumor ulceration, mice were euthanized. To estimate the tumor growth rate in each treatment group and to test for significant growth attenuation between treatment groups, linear mixed-effects regression models were used.
Nude/PDX mouse model
2 × 106 cells of the respective PDX models, i.e., LX110, LX33 and LX1322, were mixed with a 1:1 mixture of phosphate-buffered saline (PBS) and Matrigel (#CB40234, Fisher) and were injected subcutaneously into the flanks of 6-week-old female nude mice obtained from ENVIGO (Boyertown, PA). Mice were randomized into treatment groups of control and NU7441 treatment groups (n ≥ 8) once the mean tumor volume reached 100–150 mm3. When tumors reached a mean volume of 100–150 mm3, the mice were randomized into two groups and treated with either vehicle (5% glucose + 0.5%HPMC, IV) or NU7441 (10 mg/kg, i.p. 5/7 days) [n = 7 per group].
Immunocompetent mice model
Single-cell suspensions of 2 × 106 RPP (Trp53−/−, p130−/−, Rb1−/−) or RPM (Rb1−/−, Trp53−/−, MYCT58A) cells derived from GEMMs were injected into 4–6 week-old female B6129F1 mice (for RPP) and B6FVBF1/J mice (for RPM) (The Jackson Laboratory). When tumors reached the mean volume of 100–150 mm3, tumor bearing mice were randomized into four different groups. They were treated with either of the four drug regiments: vehicle (n = 8 for RPP and RPM) (5% glucose+0.5%HPMC, IV), anti-PD-L1 antibody (n = 8 for RPP and RPM) (300 µg/ animal, once weekly, intraperitoneal injection), NU7441 (n = 10 for RPP and RPM) (10 mg/kg, i.p. 5/7 days), or in combination of NU7441 and anti-PD-L1 antibody (n = 10 for RPP and RPM. Mice with tumor volumes around 1600 mm3, consistent loss more than 10% body weight or severe ulceration were euthanized.
Flow cytometry analysis
Flow cytometry analysis was performed as per standard protocol. LSRFortessa X-20 and FACSDiva software were used to obtain data and analysis, respectively. For Murine tumors, single-cell suspensions were prepared using the gentleMACS dissociator following the manufacturer’s instructions. Cells were stained with anti-CD16/32 monoclonal antibody and other fluorochrome-conjugated antibodies (Supplementary Table 5) for 30 min followed by fixation with 1% Paraformaldehyde. Cytek Aurora (Cytek, Bethesda, MD) flow cytometer was used to obtain data and data analysis was done with FlowJo software (version 10.6, BD Biosciences, Franklin Lakes, NJ). Details of the flow antibodies are provided in supplementary Table S6. For the cell cycle assay, cells were washed once with ice cold PBS and fixed with 70% ice cold ethanol overnight in − 20 ⁰C. Next, day cells were washed again with PBS and stained with FxCycleTMPI/RNase staining solution (Invitrogen) for 15 mins at room temperature and analyzed with FACSymphony A5 SE. For in vitro MHC-I surface staining single cell suspensions were prepared, and TruFCX (for human cells) was used to block nonspecific binding. Following the blocking, cells were stained (30 min) with anti-HLA-ABC antibody tagged with fluorescent dye. The cells were then washed twice with DPBS and then analyzed by FACSymphony A5 SE.
Annexin V-propidium iodide (PI) assay
Apoptosis was detected by Annexin-V – PI assay according to the manufacturer’s protocol in the FITC annexin V Apoptosis Detection Kit I (BD Biosciences). Briefly, in six-well plates, 4 × 105 cells were seeded and treated with or without 4 μM of NU7441 for 4 days for each cell line. After the treatment, harvested cells were stained with annexin-V and propidium iodide (PI) and analyzed using the FACSymphony A5 SE flow cytometer and with FlowJo software (version 10.6, BD Biosciences).
In vivo STING knockdown study
Single-cell suspensions of 2 × 106 STING knocked-down cells were injected into the right flank of RPP and RPM GEMM mice, respectively to establish a tumor. When the tumors reached its mean volume of 100–150 mm3, mice were randomized into groups into 4 groups and treated as following: (1) vehicle (n = 6), (2) anti PD-L1 antibody alone (300 µg/animal, IP, 1/7) (n = 6) (3) only NU7441 (10 mg/kg, i.p. 5/7 days) (n = 6) (4) combination of NU7441 and anti PD-L1 antibody (n = 6).
RNA sequencing and analysis
Freshly prepared cell pellets were sent for RNA isolation, followed by library preparation, and RNA sequencing to GENEWIZ. Total RNA extraction was done using RNeasy Plus Universal mini kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions. To prepare RNA seq. library for Illumina, NEBNext Ultra II RNA Library Prep Kit (NEB, Ipswich, MA, USA) was used. Validation and assessment of RNA samples and seq. libraries were performed using Agilent TapeStation (Agilent Technologies, Palo Alto, CA, USA). Library quantification was done by q-PCR (KAPA Biosystems, Wilmington, MA, USA) and by Qubit 2.0 Fluorometer (Invitrogen, Carlsbad, CA).
Illumina HiSeq instrument (4000 or equivalent) was used to load libraries following the manufacturer’s protocol after clustering on one flowcell lane. Samples were sequenced using a 2 x 150bp paired end (PE) configuration. Image analysis and base calling were performed using HiSeq Control Software (HCS). Illumina’s bcl2fastq 2.17 software was used to convert raw sequence data (.bclfiles) into fastq files, and were de-multiplexed allowing one mismatch for index sequence identification.
A total of 36 RNA-Seq libraries from six separate cell lines (3 control replicates and 3 NU7441 treatment replicates each) representing three different SCLC subtypes are processed using the same pipeline for compatibility. Quality control has been performed using FastQC (v0.11.8) (Andrews 2010) [https://www.bioinformatics.babraham.ac.uk/projects/fastqc/]. Trim Galore! (version 0.6.5) has been used to trim the adapter sequences with a quality threshold of 20. The human genome reference used was GRCh38.p13, and GENCODE release 36 was used as the transcriptome reference. Samples DMS114 control replicate 3, H196 NU replicate 1, H720 control replicate 3, and H1341 control replicate 2 were removed from the analysis due to poor clustering in the PCA. The alignment is performed by using the STAR aligner (v2.7.5b)61. Gene level read counts are obtained by using Salmon (v1.2.1) for all libraries62. Remaining samples have passed the quality control requirements with > 50% of reads uniquely mapping ( > 20 M uniquely mapped reads for each library) using the STAR aligner.
Differential expression and functional analysis
Differential expression analysis was performed using the gene-level read counts and the DESeq2 (v1.28.1) R package63. Genes with less than 5 reads in total across all samples are filtered as inactive genes. Differentially expressed genes were defined as those with an adjusted p-value below 0.05 and an absolute log2 fold change greater than 1, with p-values adjusted using the Benjamin-Hochberg method in DESeq2. The over-representation and gene set enrichment analysis for functional enrichment are both performed using the clusterProfiler R package (v3.16.0)64. The gene sets used for functional analysis are obtained from The Molecular Signatures Database (MSigDB)65,66,67. The Fisher test was used to determine whether the overlap between the DEGs and the genes in the term is statistically significant (p < 0.05). The bold terms with an asterisk in front are the terms that are significantly enriched (p-adjusted <0.05).
Principal component analysis, heatmaps and boxplots
Samples were normalized with the use of a variance stabilizing transformation (VST)-DESeq2 package. Principal component analysis (PCA) and Euclidean distance calculations between samples were based on the 1000 most variable genes. Gene expression heatmaps display z-scored VST-normalized gene-level read counts. Heatmaps in Supplementary Figs. S2A, B and S5C are generated using the R package pheatmaps (v1.0.12). Heatmaps in S2 A-B represent z-scores of GSEA calculated enrichment scores. The genes were ranked based on correlation with IC50 values of NU7441 and Nedisertib in Supplementary Fig. S2A and S2B respectively to perform GSEA.
Heatmaps of Supplementary Figs. S5C shows z-scored DESeq2-normalized gene-level counts for the specified genes in each marker group. Boxplots in Supplementary Figs. S1A, 4A are generated using ggplot2 (v3.4.2). Boxplot is Supplementary Fig.S1A represents public data available from CellMinerDB specifying the Cancer Cell Line Encyclopedia dataset and boxplots in 4 A represents expression counts from current and previous bulk RNA-seq datasets. Boxplots depict normalized gene expression counts for labeled genes across sample conditions. Except for heatmaps, all visualizations are generated using the plotly R package (v4.9.2.1). R (v4.1.0) is used to perform all bioinformatics analysis.
Survival analysis
Survival analysis was performed using a publicly available dataset of small-cell lung cancer samples from study GSE30219. Samples were split into high or low categories by ranking the average expression of PRKDC and dividing the samples into the two groups. P-value is calculated using Cox regression. Plots and analysis were done using R packages Survminer (v0.4.9) and Survival (v3.2-11).
For survival analysis with the IMPower133 data set which includes 271 patients of various ages, genders, treated with either Atezo or placebo. First, we set out to test for the association of PRKDC expression with progression-free survival (PFS). We used a Cox regression model with censored time-to-disease progression (in months) as the dependent variable, and PRKDC as the independent variable (continuous, tpm), exploring possible covariates such as age, sex, and treatment, as well as interaction terms. Examination of the scaled Schoenfeld residuals indicated that the proportional hazards assumption had been met. Next, we utilized the same statistical approach to test for the association of PRKDC expression with the overall survival (OS) of SCLC patients. Statistical analysis was performed using the “coxph”, “cox.zph”, and “ggsurvplot” functions from the R packages “survminer” version 0.4.9 and “survival” version 3.7.0.
Scatter plots and correlation analysis
Scatter plots and correlation analyses were performed using R packages ggplot2 (v3.4.2) and ggpubr (v0.4.0). Each dot in the plot represents one sample, the blue line represents a linear model applied to the dataset, and the gray shading depicts the standard error of the model. R value and p value are reported on plot, calculated as Pearson correlations. Expression data used in Supplementary Figs. S1C, S1D, and S5A, B are normalized counts from publicly available data from CellMinerCBD representing NCI-DTP SCLC cell lines. IC-50 values used in Supplementary Figs. are from publicly available data from GSE7316230.
Western blot
Protein extraction was done in a previously described method68. Briefly, post-treatment cells were collected in ice-cold PBS followed by centrifugation at 1000 × g for 3 min. Cell pellets were resuspended in 1X RIPA buffer (Thermo Fisher, #89901) containing 5 µM of Sodium Fluoride and 1X protease - phosphatase inhibitor cocktail (Thermo Fisher, #78446) and incubated on ice for 1 h with intermittent vortexing every 5 min followed by centrifugation at 13000 × g for 20 min at 4 °C. Supernatant was transferred to fresh tubes were quantified using Coomassie Protein Assay reagent (Thermo Scientific, #1856209). Laemmli Sample Buffer (Bio-Rad, #1610747) was used to prepare samples and were run on 10-15% SDS-PAGE followed by transfer on to 0.45 μm Immobilon-FL PVDF membrane (Millipore, #IPFL00010). Membrane was blocked with Blocker Casein (Thermo Scientific, #37532) in TBS (Fisher bioreagents, #BP2471-1) containing Tween 20 (Fisher bioreagents, #BP337-500) for 1 hour at room temperature. Then the membranes were probed with specific primary antibodies [listed in Supplementary Table S7] at 1:1000 dilution and incubated overnight at 4 °C. Following 2 TBST washes for 5 mins each in shaking condition the blots were incubated with 1:10000 dilution of secondary antibodies containing horseradish peroxidase enzyme for 1 hour at room temperature. Following 5 times washes with TBST, each for 5 mins in shaking condition, blots were developed in iBright Western Blot Imaging Systems (Thermo Fisher) using SuperSignal West Pico PLUS Chemiluminescent Substrate (Thermo Scientific, #34577).
Micronuclei assay
Cells were seeded in 8 chambered slides and were treated with either vehicle [DMSO] or 4 µM NU7441 for 1 day. 4 hours before the endpoint 3 μg/ml Cytochalasin B was added. After the endpoint of treatment slides were washed twice with DPBS. The cells were lysed using a weak hypotonic solution (0.075 M KCl/0.9 % saline, 1:9) by incubating at 37 °C for five min. 3:1 methanol: acetic acid solution was used to fix the cells followed by washing with DPBS. The cells were blocked with 1% BSA by incubating at 37 ⁰C for 45 mins followed by 2 washes with PBS each for 5 mins. After washing, cells were incubated with cGAS and Lamin B1 primary antibody (1:300 dilution) at 4 °C in a humidified chamber for 12–16 h. Then the cells were washed with PBS twice, each for 5 mins and incubated with secondary antibodies (Alexa Fluor 488 Rabbit Monoclonal and Alexa Fluor 647 mouse Monoclonal antibodies) (1:500 dilution) for an hour at 37 °C in a humidified chamber and washed twice with PBS, each for 5 mins. Following the wash cells were counterstained with DAPI (2ug/ml) for 30 mins at room temp. Finally, stained cells were imaged with Leica STED 3X at 63X lens.
Transfection of siRNAs
10 nM of Silencer Select siRNAs (Invitrogen) against TMEM173 (STING) or PRKDC was transfected into cells using lipofectamine RNAi-MAX (Invitrogen) per the manufacturer’s protocol. As the scrambled control, Silencer Select siRNA for Negative Control no.1 (AM4611, Invitrogen) was used. We confirmed knockdown with quantitative RT-PCR.
shRNA mediated knock down
MYC shRNA1 construct ref. seq. NM_002467 (Target seq: CCTGAGACAGATCAGCAACAA) was obtained from Sigma Aldrich, shRNA sequence 2 (HSH088534) was bought from Genecopoeia and transduced into SCLC cell lines using lenti-viral vector according to the manufacturer’s instructions.
RNA isolation and cDNA preparation
Trizol method was used for RNA isolation. Post-treatment, cells were harvested in PBS, centrifuged and the pellets were resuspended in Trizol (ambion by life technologies #15596026).
To 1 ml of trizol, 0.2 ml of 1-Bromo-3-chloropropane (Thermo Scientific, #106862500) was added and was mixed by inversion. The tubes were then incubated on ice for 2 min and then centrifugated for 15 mins at 12000 × g in 4 °C. The top layer (aqueous phase) was carefully taken out and transferred to new tubes. Following the transfer, 0.5 ml of isopropanol was added to the solution and incubated for 10 min followed by centrifugation at 12000 g for 10 minutes at 4 °C. The RNA pellets were then washed with 1 ml of 75% ethanol and centrifuged at 7500 × g for 5 minutes at 4 °C. This washing step was repeated twice and supernatants were discarded. The pellets were dissolved in DEPC water after air-drying the pellets. The dissolved RNA was incubated at 58 °C for 10–12 min then immidiately chilled on ice. RNA concentrations were measured with Thermo Scientific Nanodrop one, and cDNA was prepared with 2 µg of RNA with Verso cDNA synthesis kit (Thermo Scientific #AB-1452/B).
Quantitative rt-PCR
To perform Quantitative PCR, prepared cDNA were diluted with DEPC treated water (1:4 or 1:5 ratio). MAstermix were prepared with diluted cDNA, PowerUp SYBR Green Master Mix (applied biosystems, #A25742) and forward and reverse primers to final volume of 10 µl. The PCR cycle contained the following steps: initial denaturation at 95 °C for 10 min followed by 40 cycles of denaturation (95 °C for 30 s), annealing (60 °C for 30 s) and extension (72 °C 30 s) and a final extension for 10 mins at 72 °C. After the final extension, melting curve analysis was performed. Detailed primer list of the genes along with sequences are provided in Supplementary Table S8.
ELISA
SCLC cells (0.5 × 106) were seeded in 6-well plate in complete RPMI-1640 media. The next day cells were treated with either DMSO or 4 µM NU7441 for 3days. After the treatment condition media as well as the cell-free protein extract were collected. Elisa was performed to quantify INFa, IFNb, CCL5, CXCL10 (R&D Systems, Minneapolis, MN, USA) [Kits detailed in Supplementary Table S8] as per manufacturer’s instruction. Protein concentrations of each protein (pg/ml) was assessed from the standard curve.
Statistics and reproducibility
Linear mixed-effects regression models were used to estimate the association between time and tumor size of each treatment group for all the in vivo experiments (Figs. 2C–E, 3J–M, 5A, C, 6A–D, H–K, 7A, D). These models included tumor-volume as the dependent variable (Y), while considering time (days) and Treatment Group (Vehicle, NU7441 etc) as independent variables (X) along with interaction terms between Time and Treatment. To address the repeated measurement over time of the same mouse, a random intercept and and random slope for MouseID were included assuming an auto-regressive (AR) covariance structure across time.
Quantitative rt-PCR, flow cytometry and ELISA data were expressed as means ± standard error of mean (SE). Whereas tumor volume data of the in vivo studies were expressed as means ± standard deviation (SD). For the comparison between two groups paired two-tailed Students’ t test was performed following ANOVA or dunnett’s test to assess significance among multiple group using GraphPad Prism Ver. 9.0 software. p-values of students’ t-test less than 0.05 was considered to be statistically significant (ns > 0.05, ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001). Data are expressed as the mean ± SEM. All the in vitro experiments have been repeated at least twice for reproducibility.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
RNA-seq data have been deposited at GEO and are publicly available. The accession number is GSE273409. Along with this, we have also re-analyzed the following publicly available datasets: GSE30219, GSE73162 and GSE223372. For the human samples (WTS), the deidentified raw sequencing data are owned by Caris Life Sciences and can be made available to researchers for replication and verification purposes through a letter of intent to Caris Life Sciences (https://www.carislifesciences.com/letter-of-intent/). This study did not generate new, unique reagents. All unique/stable reagents generated in this study are available from the corresponding author with a completed Materials Transfer Agreement. Further information and requests for resources and reagents should be directed to and will be fulfilled by Triparna Sen (triparna.sen@osumc.edu). Source data are provided in this paper.
References
Saltos, A., Shafique, M. & Chiappori, A. Update on the biology, management, and treatment of small cell lung cancer (SCLC). Front. Oncol. 10, 1074 (2020).
Sen, T. et al. Emerging advances in defining the molecular and therapeutic landscape of small-cell lung cancer. Nat. Rev. Clin. Oncol. 21, 610–627 (2024).
Jeon, D. S. et al. Five-year overall survival and prognostic factors in patients with lung cancer: results from the korean association of lung cancer registry (KALC-R) 2015. Cancer Res. Treat. 55, 103–111 (2023).
McNamee, N., da Silva, I. P., Nagrial, A., Gao, B. Small-cell lung cancer-an update on targeted and immunotherapies. Int. J. Mol. Sci. 24, https://doi.org/10.3390/ijms24098129 (2023).
Mahadevan, N. R. et al. Intrinsic immunogenicity of small cell lung carcinoma revealed by its cellular plasticity. Cancer Discov. 11, 1952–1969 (2021).
Mamdani, H., Matosevic, S., Khalid, A. B., Durm, G. & Jalal, S. I. Immunotherapy in lung cancer: current landscape and future directions. Front. Immunol. 13, 823618 (2022).
Horn, L. et al. First-line atezolizumab plus chemotherapy in extensive-stage small-cell lung cancer. N. Engl. J. Med. 379, 2220–2229 (2018).
Paz-Ares, L. et al. Durvalumab plus platinum-etoposide versus platinum-etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): a randomised, controlled, open-label, phase 3 trial. Lancet 394, 1929–1939 (2019).
Rudin, C. M. et al. Molecular subtypes of small cell lung cancer: a synthesis of human and mouse model data. Nat. Rev. Cancer 19, 289–297 (2019).
Lang, C. et al. C-Myc protein expression indicates unfavorable clinical outcome in surgically resected small cell lung cancer. World J. Surg. Oncol. 22, 57 (2024).
Llombart, V. & Mansour, M. R. Therapeutic targeting of “undruggable” MYC. EBioMedicine 75, 103756 (2022).
Taniguchi, H. et al. ATR inhibition activates cancer cell cGAS/STING-interferon signaling and promotes antitumor immunity in small-cell lung cancer. Sci. Adv. 10, eado4618 (2024).
Sen T. Identifying and targeting the Achilles heel of a recalcitrant cancer. Sci. Transl. Med. 13, https://doi.org/10.1126/scitranslmed.abj6946 (2021).
Sen, T. et al. Targeting DNA damage response promotes antitumor immunity through STING-mediated T-cell activation in small cell lung cancer. Cancer Discov. 9, 646–661 (2019).
Sen, T. et al. Combination treatment of the Oral CHK1 inhibitor, SRA737, and low-dose gemcitabine enhances the effect of programmed death ligand 1 blockade by modulating the immune microenvironment in SCLC. J. Thorac. Oncol. 14, 2152–2163 (2019).
Panagiotou, E. et al. Biological and therapeutic implications of the cancer-related germline mutation landscape in lung cancer. Lancet Respir Med. 12, 997–1005(2024).
Begg, A. C., Stewart, F. A. & Vens, C. Strategies to improve radiotherapy with targeted drugs. Nat. Rev. Cancer 11, 239–253 (2011).
Srivastava, M. & Raghavan, S. C. DNA double-strand break repair inhibitors as cancer therapeutics. Chem. Biol. 22, 17–29 (2015).
Mohiuddin, I. S. & Kang, M. H. DNA-PK as an Emerging therapeutic target in cancer. Front. Oncol. 9, 635 (2019).
Xing, J., Wu, X., Vaporciyan, A. A., Spitz, M. R. & Gu, J. Prognostic significance of ataxia-telangiectasia mutated, DNA-dependent protein kinase catalytic subunit, and Ku heterodimeric regulatory complex 86-kD subunit expression in patients with nonsmall cell lung cancer. Cancer 112, 2756–2764 (2008).
Yang H., Yao F., Marti T. M., Schmid R. A. & Peng R. W. Beyond DNA repair: DNA-PKcs in tumor metastasis, metabolism and immunity Cancers 12, https://doi.org/10.3390/cancers12113389 (2020).
Burleigh, K. et al. Human DNA-PK activates a STING-independent DNA sensing pathway. Sci. Immunol.5, https://doi.org/10.1126/sciimmunol.aba4219 (2020).
Dylgjeri, E. & Knudsen, K. E. DNA-PKcs: A targetable protumorigenic protein kinase. Cancer Res. 82, 523–533 (2022).
van Bussel, M. T. J. et al. A first-in-man phase 1 study of the DNA-dependent protein kinase inhibitor peposertib (formerly M3814) in patients with advanced solid tumours. Br. J. Cancer 124, 728–735 (2021).
Rasco, D. W. et al. A first-in-human study of novel cereblon modulator avadomide (CC-122) in advanced malignancies. Clin. Cancer Res. 25, 90–98 (2019).
Huang, Y. H. et al. POU2F3 is a master regulator of a tuft cell-like variant of small cell lung cancer. Genes Dev. 32, 915–928 (2018).
Borromeo, M. D. et al. ASCL1 and NEUROD1 reveal heterogeneity in pulmonary neuroendocrine tumors and regulate distinct genetic programs. Cell Rep. 16, 1259–1272 (2016).
Baine, M. K. et al. SCLC Subtypes defined by ASCL1, NEUROD1, POU2F3, and YAP1: A comprehensive immunohistochemical and histopathologic characterization. J. Thorac. Oncol. 15, 1823–1835 (2020).
Rousseaux, S. et al. Ectopic activation of germline and placental genes identifies aggressive metastasis-prone lung cancers. Sci. Transl. Med. 5, 186ra66 (2013).
Polley, E. et al. Small cell lung cancer screen of oncology drugs, investigational agents, and gene and microRNA expression. J. Natl. Cancer Inst. 108, https://doi.org/10.1093/jnci/djw122 (2016).
Camfield, S. et al. Secrets of DNA-PKcs beyond DNA repair. NPJ Precis. Oncol. 8, 154 (2024).
Caeser, R. et al. Genomic and transcriptomic analysis of a library of small cell lung cancer patient-derived xenografts. Nat. Commun. 13, 2144 (2022).
Coleman, N., Zhang, B., Byers, L. A. & Yap, T. A. The role of Schlafen 11 (SLFN11) as a predictive biomarker for targeting the DNA damage response. Br. J. Cancer 124, 857–859 (2021).
MacDonald, K. M., Benguerfi, S. & Harding, S. M. Alerting the immune system to DNA damage: micronuclei as mediators. Essays Biochem. 64, 753–764 (2020).
MacDonald, K. M. et al. Antecedent chromatin organization determines cGAS recruitment to ruptured micronuclei. Nat. Commun. 14, 556 (2023).
Tlemsani, C. et al. SCLC-CellMiner: a resource for small cell lung cancer cell line genomics and pharmacology based on genomic signatures. Cell Rep. 33, 108296 (2020).
Rudin, C. M., Brambilla, E., Faivre-Finn, C. & Sage, J. Small-cell lung cancer. Nat. Rev. Dis. Primers 7, 3 (2021).
Mollaoglu, G. et al. MYC Drives progression of small cell lung cancer to a variant neuroendocrine subtype with vulnerability to aurora kinase inhibition. Cancer Cell 31, 270–285 (2017).
Ahmadi, S. E., Rahimi, S., Zarandi, B., Chegeni, R. & Safa, M. MYC: a multipurpose oncogene with prognostic and therapeutic implications in blood malignancies. J. Hematol. Oncol. 14, 121 (2021).
Garcia-Gutierrez, L., Delgado, M. D. & Leon, J. MYC Oncogene Contributions to Release of Cell Cycle Brakes. Genes 10, https://doi.org/10.3390/genes10030244 (2019).
Stronach, E. A. et al. DNA-PK mediates AKT activation and apoptosis inhibition in clinically acquired platinum resistance. Neoplasia 13, 1069–1080 (2011).
An, J. et al. DNA-dependent protein kinase catalytic subunit modulates the stability of c-Myc oncoprotein. Mol. Cancer 7, 32 (2008).
Chen, J. et al. MYC drives platinum resistant SCLC that is overcome by the dual PI3K-HDAC inhibitor fimepinostat. J. Exp. Clin. Cancer Res. 42, 100 (2023).
Gregory, M. A., Qi, Y. & Hann, S. R. Phosphorylation by glycogen synthase kinase-3 controls c-myc proteolysis and subnuclear localization. J. Biol. Chem. 278, 51606–51612 (2003).
Bhanpattanakul, S. et al. Modulation of MHC expression by interferon-gamma and its influence on PBMC-mediated cytotoxicity in canine mast cell tumour cells. Sci. Rep. 14, 17837 (2024).
Taniguchi, H. et al. WEE1 inhibition enhances the antitumor immune response to PD-L1 blockade by the concomitant activation of STING and STAT1 pathways in SCLC. Cell Rep. 39, 110814 (2022).
Sedighzadeh, S. S., Khoshbin, A. P., Razi, S., Keshavarz-Fathi, M. & Rezaei, N. A narrative review of tumor-associated macrophages in lung cancer: regulation of macrophage polarization and therapeutic implications. Transl. Lung Cancer Res. 10, 1889–1916 (2021).
Takaki, T., Millar, R., Hiley, C. T. & Boulton, S. J. Micronuclei induced by radiation, replication stress, or chromosome segregation errors do not activate cGAS-STING. Mol Cell 84, 2203–13.e5 (2024).
Hristova, D. B. et al. DNA-PKcs is required for cGAS/STING-dependent viral DNA sensing in human cells. iScience 27, 108760 (2023).
Sun, X. et al. DNA-PK deficiency potentiates cGAS-mediated antiviral innate immunity. Nat. Commun. 11, 6182 (2020).
Sen, T. et al. CHK1 Inhibition in small-cell lung cancer produces single-agent activity in biomarker-defined disease subsets and combination activity with cisplatin or olaparib. Cancer Res. 77, 3870–3884 (2017).
Sen, T., Gay, C. M. & Byers, L. A. Targeting DNA damage repair in small cell lung cancer and the biomarker landscape. Transl. Lung Cancer Res. 7, 50–68 (2018).
Ireland, A. S. et al. MYC Drives temporal evolution of small cell lung cancer subtypes by reprogramming neuroendocrine fate. Cancer Cell 38, 60–78 (2020).
Gabay, M., Li, Y., Felsher, D. W. MYC activation is a hallmark of cancer initiation and maintenance. Cold Spring Harb. Perspect. Med. 4, https://doi.org/10.1101/cshperspect.a014241 (2014).
Dhanasekaran, R. et al. The MYC oncogene - the grand orchestrator of cancer growth and immune evasion. Nat. Rev. Clin. Oncol. 19, 23–36 (2022).
Donati, G. & Amati, B. MYC and therapy resistance in cancer: risks and opportunities. Mol. Oncol. 16, 3828–3854 (2022).
Li, J., Dong, T., Wu, Z., Zhu, D. & Gu, H. The effects of MYC on tumor immunity and immunotherapy. Cell Death Discov. 9, 103 (2023).
Wu, S. Y. et al. MYC suppresses STING-dependent innate immunity by transcriptionally upregulating DNMT1 in triple-negative breast cancer. J. Immunother. Cancer 9, https://doi.org/10.1136/jitc-2021-002528 (2021).
Kuchitsu, Y. et al. STING signalling is terminated through ESCRT-dependent microautophagy of vesicles originating from recycling endosomes. Nat. Cell Biol. 25, 453–466 (2023).
Gentili, M. et al. ESCRT-dependent STING degradation inhibits steady-state and cGAMP-induced signalling. Nat. Commun. 14, 611 (2023).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Patro, R., Duggal, G., Love, M. I., Irizarry, R. A. & Kingsford, C. Salmon provides fast and bias-aware quantification of transcript expression. Nat. Methods 14, 417–419 (2017).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16, 284–287 (2012).
Liberzon, A. et al. Molecular signatures database (MSigDB) 3.0. Bioinformatics 27, 1739–1740 (2011).
Liberzon, A. et al. The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst. 1, 417–425 (2015).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 102, 15545–15550 (2005).
Chakraborty, S. et al. De Novo and histologically transformed small-cell lung cancer is sensitive to lurbinectedin treatment through the modulation of EMT and NOTCH signaling pathways. Clin. Cancer Res. 29, 3526–3540 (2023).
Acknowledgements
This work is supported by NIH/NCI R01 CA258784 (TS); Congressionally Directed Medical Research Programs (DOD-IITRA) LC190161 (TS). This work was supported in part by the Bioinformatics for Next Generation Sequencing (BiNGS) shared resource facility within the Tisch Cancer Institute at the Icahn School of Medicine at Mount Sinai, which is partially supported by NIH grant P30CA196521. This work was also supported in part through the computational and data resources and staff expertise provided by Scientific Computing and Data at the Icahn School of Medicine at Mount Sinai and supported by the Clinical and Translational Science Awards (CTSA) grant UL1TR004419 from the National Center for Advancing Translational Sciences. Research reported in this publication was also supported by the Office of Research Infrastructure of the National Institutes of Health under award numbers S10OD026880 and S10OD030463.
Author information
Authors and Affiliations
Contributions
Conceptualization: T.S.; Methodology: S.C., U.S., K.V., V.J., C.C., A.B., Y.M., D.D., D.H., and T.S.; Investigation: S.C., U.S., K.V., V.J., Y.M., C.C., A.B., F.S., E.S., D.D., D.H., T.S.; Validation: S.C., U.S., A.E., C.C., D.D., D.H., T.S.; Formal Analysis: S.C., U.S., K.V., C.C., F.S., D.D., A.E., U.S., D.H., H.T., C.W.F., A.V., B.H., T.S.; Writing – Original Draft: S.C., T.S.; Review & Editing: All authors; Supervision: T.S.; Funding acquisition: T.S.
Corresponding author
Ethics declarations
Competing interests
T.S. has received research grants from Jazz Pharmaceuticals. B.H. receives clinical research funding from Boehringer Ingelheim, AstraZeneca, Merck, BMS, Advaxis, Amgen, AbbVie, Daiichi, Pfizer, GSK, Beigene, Janssen, Black Diamond Therapeutics, Forward Pharma, Numab, Arrivent and receives Honoraria from AstraZeneca, Boehringer Ingelheim, Apollomics, Janssen, Takeda, Merck, BMS, Genentech, Pfizer, Eli-Lilly, Arcus, Merus, Daiichi, and Precede. He also serves on the Data Safety Monitoring Board/Advisory board for BMS, TPT, Apollomics, eFECTOR, and City of Hope. H.T. has received lecture fees from AstraZeneca, Chugai Pharmaceutical Co., Daiichi Sankyo, Taiho Pharmaceutical Co., and MSD. A.E. and A.V. are employees of Caris Life Sciences. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Hongbin Ji, Gurkan Mollaoglu and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chakraborty, S., Elliott, A., Sen, U. et al. Targeting NHEJ activates STING signaling through MYC degradation to boost antitumor immunity in SCLC. Nat Commun 17, 2597 (2026). https://doi.org/10.1038/s41467-026-69262-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-69262-x