Abstract
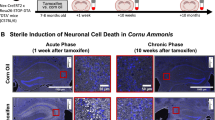
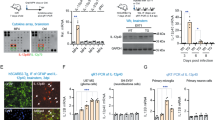
The cellular interactions that drive the neuropathology of most neuroinvasive viruses remain elusive. We used Japanese encephalitis virus (JEV) to infect female BALB/c mice and applied Stereo-seq to simultaneously capture host and viral transcriptomes in situ, thereby constructing a comprehensive spatiotemporal atlas of Japanese encephalitis (JE) pathogenesis. Our analysis pinpoints Ly6c2+ monocytes as the primary virus carrier, the most abundant infiltrating immune cell type, and a major source of IFN-γ in the infected mouse brain. Following infection, Ackr1+ endothelial cell activation is linked to blood-brain barrier disruption and immune cell chemotaxis, particularly Ly6c2+ monocytes. The crosstalk between these two cell types appears to orchestrate the pronounced inflammation and cell death, including pyroptosis and necroptosis, which radiate from the vasculature to different brain regions. Disrupting the activation and interactions of these two cell types may help mitigate JE progression. This study also provides a technical framework for investigating the pathogenesis of other neurotropic viruses.
Similar content being viewed by others
Data availability
All the Stereo-seq data supporting the findings of this study have been deposited in CNGBdb (https://db.cngb.org/cnsa/) under the accession number of STT0000076 (https://db.cngb.org/stomics/). The raw sequencing data have been deposited in the Genome Sequence Archive in National Genomics Data Center129,130, China National Center for Bioinformation under the GSA number of CRA035980 (https://ngdc.cncb.ac.cn/gsa/). The gene expression matrix and associated metadata have been deposited in Zenodo (https://doi.org/10.5281/zenodo.17958448). All the experimental validation data of this study have been deposited in the Figshare database (https://doi.org/10.6084/m9.figshare.30382759) and the Zenodo database (https://doi.org/10.5281/zenodo.18323401). Source data are provided with this paper.
References
Auerswald, H., Maquart, P.-O., Chevalier, V. & Boyer, S. Mosquito vector competence for Japanese encephalitis virus. Viruses 13, 1154 (2021).
Heffelfinger, J. D. et al. Japanese encephalitis surveillance and immunization—Asia and Western Pacific Regions, 2016. MMWR Morb. Mortal. Wkly Rep. 66, 579–583 (2017).
Campbell, G. L. et al. Estimated global incidence of Japanese encephalitis: a systematic review. Bull. World Health Organ 89, 766–774 (2011). 774A-774E.
Fischer, M. et al. Japanese encephalitis prevention and control: advances, challenges, and new initiatives. in Emerging Infections Vol.8 (eds Michael Scheld, W., Hammer, S. M. & Hughes, J. M.) 93–124. https://doi.org/10.1128/9781555815592.ch6 (ASM Press, 2014).
Quan, T. M., Thao, T. T. N., Duy, N. M., Nhat, T. M. & Clapham, H. Estimates of the global burden of Japanese encephalitis and the impact of vaccination from 2000-2015. eLife 9, e51027 (2020).
Baldwin, Z. et al. The seroprevalence of antibodies to Japanese encephalitis virus in five New South Wales towns at high risk of infection, 2022: a cross-sectional serosurvey. Med. J. Aust. 220, 561–565 (2024).
Wang, G. et al. Peripheral nerve injury associated with JEV infection in high endemic regions, 2016-2020: a multicenter retrospective study in China. Emerg. Microbes Infect. 13, 2337677 (2024).
Zhang, X. et al. Adults in Northwest China experienced the largest outbreak of Japanese encephalitis in history 10 years after the Japanese encephalitis vaccine was included in the national immunization program: A retrospective epidemiological study. J. Med. Virol. 95, e28782 (2023).
Wang, P. et al. DC-SIGN as an attachment factor mediates Japanese encephalitis virus infection of human dendritic cells via interaction with a single high-mannose residue of viral E glycoprotein. Virology 488, 108–119 (2016).
Wang, K. & Deubel, V. Mice with different susceptibility to Japanese encephalitis virus infection show selective neutralizing antibody response and myeloid cell infectivity. PLoS ONE 6, e24744 (2011).
Sharma, K. B., Chhabra, S. & Kalia, M. Japanese encephalitis virus-infected cells. in Virus Infected Cells Vol. 106 (eds Vijayakrishnan, S., Jiu, Y. & Harris, J. R.) 251–281 (Springer International Publishing, 2023).
Yang, K. D. et al. A model to study neurotropism and persistency of Japanese encephalitis virus infection in human neuroblastoma cells and leukocytes. J. Gen. Virol. 85, 635–642 (2004).
Byrne, S. N., Halliday, G. M., Johnston, L. J. & King, N. J. C. Interleukin-1β but not tumor necrosis factor is involved in west nile virus-induced Langerhans cell migration from the skin in C57BL/6 mice. J. Investig. Dermatol. 117, 702–709 (2001).
Wang, P. et al. DC-SIGN promotes Japanese encephalitis virus transmission from dendritic cells to T cells via virological synapses. Virol. Sin. 32, 495–502 (2017).
Malissen, B., Tamoutounour, S. & Henri, S. The origins and functions of dendritic cells and macrophages in the skin. Nat. Rev. Immunol. 14, 417–428 (2014).
Pingen, M., Schmid, M. A., Harris, E. & McKimmie, C. S. Mosquito biting modulates skin response to virus infection. Trends Parasitol. 33, 645–657 (2017).
Lai, C. et al. Endothelial Japanese encephalitis virus infection enhances migration and adhesion of leukocytes to brain microvascular endothelia via MEK-dependent expression of ICAM 1 and the CINC and RANTES chemokines. J. Neurochem. 123, 250–261 (2012).
Chang, C. et al. Disruption of in vitro endothelial barrier integrity by Japanese encephalitis virus-Infected astrocytes. Glia 63, 1915–1932 (2015).
Wang, K. et al. IP-10 promotes blood–brain barrier damage by inducing tumor necrosis factor alpha production in Japanese encephalitis. Front. Immunol. 9, 1148 (2018).
Cain, M. D., Salimi, H., Diamond, M. S. & Klein, R. S. Mechanisms of pathogen invasion into the central nervous system. Neuron 103, 771–783 (2019).
Desai, A., Shankar, S. K., Ravi, V., Chandramuki, A. & Gourie-Devi, M. Japanese encephalitis virus antigen in the human brain and its topographic distribution. Acta Neuropathol. 89, 368–373 (1995).
Fu, T. L., Ong, K. C., Tan, S. H. & Wong, K. T. Japanese encephalitis virus infects the thalamus early followed by sensory-associated cortex and other parts of the central and peripheral nervous systems. J. Neuropathol. Exp. Neurol. 78, 1160–1170 (2019).
Ghoshal, A. et al. Proinflammatory mediators released by activated microglia induces neuronal death in Japanese encephalitis. Glia 55, 483–496 (2007).
Yang, L. et al. Single-cell RNA sequencing reveals the immune features and viral tropism in the central nervous system of mice infected with Japanese encephalitis virus. J. Neuroinflammation 21, 76 (2024).
Kim, J. H. et al. CCL 2, but not its receptor, is essential to restrict immune privileged central nervous system-invasion of Japanese encephalitis virus via regulating accumulation of CD 11b + Ly-6C hi monocytes. Immunology 149, 186–203 (2016).
Patil, A. M. et al. Type I IFN signaling limits hemorrhage-like disease after infection with Japanese encephalitis virus through modulating a prerequisite infection of CD11b+Ly-6C+ monocytes. J. Neuroinflammation 18, 136 (2021).
Howe, C. L., LaFrance-Corey, R. G., Goddery, E. N., Johnson, R. K. & Mirchia, K. Neuronal CCL2 expression drives inflammatory monocyte infiltration into the brain during acute virus infection. J. Neuroinflammation 14, 238 (2017).
Terry, R. L. et al. Inflammatory monocytes and the pathogenesis of viral encephalitis. J. Neuroinflammation 9, 270 (2012).
de Vries, L. & Harding, A. T. Mechanisms of neuroinvasion and neuropathogenesis by pathologic flaviviruses. Viruses 15, 261 (2023).
Zou, S.-S. et al. Brain microvascular endothelial cell-derived HMGB1 facilitates monocyte adhesion and transmigration to promote JEV neuroinvasion. Front. Cell. Infect. Microbiol. 11, 701820 (2021).
Xu, H. et al. The choroid plexus synergizes with immune cells during neuroinflammation. Cell 187, 4946–4963.e17 (2024).
Rua, R. & McGavern, D. B. Advances in meningeal immunity. Trends Mol. Med. 24, 542–559 (2018).
Myint, K. S. Neuropathogenesis of Japanese encephalitis in a primate model. PLoS Negl. Trop. Dis. 8, 2980 (2014).
Lauer, A. N., Tenenbaum, T., Schroten, H. & Schwerk, C. The diverse cellular responses of the choroid plexus during infection of the central nervous system. Am. J. Physiol.-Cell Physiol. 314, C152–C165 (2018).
Shwetank, Date, O. S., Kim, K. S. & Manjunath, R. Infection of human endothelial cells by japanese encephalitis virus: increased expression and release of soluble HLA-E. PLoS ONE 8, e79197 (2013).
Uyar, O. et al. Single-cell transcriptomics of the ventral posterolateral nucleus-enriched thalamic regions from HSV-1-infected mice reveal a novel microglia/microglia-like transcriptional response. J. Neuroinflammation 19, 81 (2022).
Qiu, M. et al. Decoding dengue’s neurological assault: insights from single-cell CNS analysis in an immunocompromised mouse model. J. Neuroinflammation 22, 62 (2025).
Ding, X. et al. Temporally resolved single-cell RNA sequencing reveals protective and pathological responses during herpes simplex virus CNS infection. J. Neuroinflammation 22, 146 (2025).
Piwecka, M., Rajewsky, N. & Rybak-Wolf, A. Single-cell and spatial transcriptomics: deciphering brain complexity in health and disease. Nat. Rev. Neurol. 19, 346–362 (2023).
Moses, L. & Pachter, L. Museum of spatial transcriptomics. Nat. Methods 19, 534–546 (2022).
Chen, A. et al. Spatiotemporal transcriptomic atlas of mouse organogenesis using DNA nanoball-patterned arrays. Cell 185, 1777–1792.e21 (2022).
Han, W. et al. Precise localization and dynamic distribution of Japanese encephalitis virus in the rain nuclei of infected mice. PLoS Negl. Trop. Dis. 15, e0008442 (2021).
Aran, D. et al. Reference-based analysis of lung single-cell sequencing reveals a transitional profibrotic macrophage. Nat. Immunol. 20, 163–172 (2019).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. Usa. 102, 15545–15550 (2005).
Liberzon, A. et al. The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst. 1, 417–425 (2015).
Castanza, A. S. et al. Extending support for mouse data in the Molecular Signatures Database (MSigDB). Nat. Methods 20, 1619–1620 (2023).
Kak, G., Raza, M. & Tiwari, B. K. Interferon-gamma (IFN-γ): exploring its implications in infectious diseases. Biomol. Concepts 9, 64–79 (2018).
Tripathi, A. et al. Lack of interferon (IFN) regulatory factor 8 associated with restricted IFN-γ response augmented Japanese encephalitis virus replication in the mouse brain. J. Virol. 95, e0040621 (2021).
Li, F. Viral infection of the central nervous system and neuroinflammation precede blood-brain barrier disruption during Japanese encephalitis virus infection. J. Virol. 89, 5602–5614 (2015).
Kraaij, M. D. et al. Human monocytes produce interferon-gamma upon stimulation with LPS. Cytokine 67, 7–12 (2014).
Frey, T. et al. Monocyte production of IFN-γ is interleukin-12 dependent in a model of mevalonate kinase deficiency. J. Interferon Cytokine Res. 39, 364–374 (2019).
Hu, C. et al. CellMarker 2.0: an updated database of manually curated cell markers in human/mouse and web tools based on scRNA-seq data. Nucleic Acids Res. 51, D870–D876 (2023).
Jin, S., Plikus, M. V. & Nie, Q. CellChat for systematic analysis of cell–cell communication from single-cell transcriptomics. Nat. Protoc. https://doi.org/10.1038/s41596-024-01045-4 (2024).
Browaeys, R., Saelens, W. & Saeys, Y. NicheNet: modeling intercellular communication by linking ligands to target genes. Nat. Methods 17, 159–162 (2020).
Fadnis, P. R., Ravi, V., Desai, A., Turtle, L. & Solomon, T. Innate immune mechanisms in japanese encephalitis virus infection: effect on transcription of pattern recognition receptors in mouse neuronal cells and brain tissue. Viral Immunol. 26, 366–377 (2013).
Arpaia, N. & Barton, G. M. Toll-like receptors: key players in antiviral immunity. Curr. Opin. Virol. 1, 447–454 (2011).
Li, D. & Wu, M. Pattern recognition receptors in health and diseases. Sig Transduct. Target. Ther. 6, 291 (2021).
Van De Sande, B. et al. A scalable SCENIC workflow for single-cell gene regulatory network analysis. Nat. Protoc. 15, 2247–2276 (2020).
Lawson, L. J., Perry, V. H., Dri, P. & Gordon, S. Heterogeneity in the distribution and morphology of microglia in the normal adult mouse brain. Neuroscience 39, 151–170 (1990).
Morabito, S., Reese, F., Rahimzadeh, N., Miyoshi, E. & Swarup, V. hdWGCNA identifies co-expression networks in high-dimensional transcriptomics data. Cell Rep. Methods 3, 100498 (2023).
Yang, X., Yang, S., Li, Q., Wuchty, S. & Zhang, Z. Prediction of human-virus protein-protein interactions through a sequence embedding-based machine learning method. Comput. Struct. Biotechnol. J. 18, 153–161 (2020).
Shah, P. S. et al. Comparative flavivirus-host protein interaction mapping reveals mechanisms of dengue and Zika virus pathogenesis. Cell 175, 1931–1945.e18 (2018).
WHO Japanese encephalitis vaccines: WHO position paper, February 2015—Recommendations. Vaccine 34, 302–303 (2016).
Mathur, A., Arora, K. L. & Chaturvedi, U. C. Host defence mechanisms against Japanese encephalitis virus infection in mice. J. Gen. Virol. 64, 805–811 (1983).
Larena, M., Regner, M., Lee, E. & Lobigs, M. Pivotal role of antibody and subsidiary contribution of CD8+ T cells to recovery from infection in a murine model of Japanese encephalitis. J. Virol. 85, 5446–5455 (2011).
Song, Z.-R. et al. The slow progression of Japanese encephalitis in aged mice is likely associated to B cell recruitment in the brain. Virol. Sin. 40, 546–559 (2025).
Srivastava, S. et al. Degradation of Japanese encephalitis virus by neutrophils. Int J. Exp. Pathol. 80, 17–24 (1999).
Lindsay, H. G., Hendrix, C. J., Gonzalez Murcia, J. D., Haynie, C. & Weber, K. S. The role of atypical chemokine receptors in neuroinflammation and neurodegenerative disorders. Int. J. Mol. Sci. 24, 16493 (2023).
Minten, C. et al. DARC shuttles inflammatory chemokines across the blood-brain barrier during autoimmune central nervous system inflammation. Brain 137, 1454–1469 (2014).
Marchetti, L. et al. ACKR1 favors transcellular over paracellular T-cell diapedesis across the blood-brain barrier in neuroinflammation in vitro. Eur. J. Immunol. 52, 161–177 (2022).
Lee, E.-S. et al. Inflammatory risk contributes to post-COVID endothelial dysfunction through anti-ACKR1 autoantibody. Life Sci. Alliance 7, e202402598 (2024).
Pham, A. M., Langlois, R. A. & tenOever, B. R. Replication in cells of hematopoietic origin is necessary for dengue virus dissemination. PLoS Pathog. 8, e1002465 (2012).
Quicke, K. M. et al. Zika virus infects human placental macrophages. Cell Host Microbe 20, 83–90 (2016).
Ma, Y. et al. Avian flavivirus infection of monocytes/macrophages by extensive subversion of host antiviral innate immune responses. J. Virol. 93, e00978-19 (2019).
Spiteri, A. G. et al. Temporal tracking of microglial and monocyte single-cell transcriptomics in lethal flavivirus infection. Acta Neuropathol. Commun. 11, 60 (2023).
Chauhan, S. et al. Japanese encephalitis virus infected human monocyte-derived dendritic cells activate a transcriptional network leading to an antiviral inflammatory response. Front. Immunol. 12, 638694 (2021).
Getts, D. R. et al. Targeted blockade in lethal West Nile virus encephalitis indicates a crucial role for very late antigen (VLA)-4-dependent recruitment of nitric oxide-producing macrophages. J. Neuroinflammation 9, 246 (2012).
Ge, S. et al. The CCL2 synthesis inhibitor bindarit targets cells of the neurovascular unit, and suppresses experimental autoimmune encephalomyelitis. J. Neuroinflammation 9, 171 (2012).
Mirolo, M. et al. Impact of the anti-inflammatory agent bindarit on the chemokinome: selective inhibition of the monocyte chemotactic proteins. Eur. Cytokine Netw. 19, 119–122 (2008).
Grassia, G. et al. The anti-inflammatory agent bindarit inhibits neointima formation in both rats and hyperlipidaemic mice. Cardiovasc. Res. 84, 485–493 (2009).
Chiu, T.-M. et al. CCR8/CCL1 and CXCR3/CXCL10 axis-mediated memory T-cell activation in patients with recalcitrant drug-induced hypersensitivity. Br. J. Dermatol. 192, 293–305 (2025).
Casanova, J.-L., MacMicking, J. D. & Nathan, C. F. Interferon-γ and infectious diseases: lessons and prospects. Science 384, eadl2016 (2024).
Li, Q. & Barres, B. A. Microglia and macrophages in brain homeostasis and disease. Nat. Rev. Immunol. 18, 225–242 (2018).
Liao, C. L. et al. Effect of enforced expression of human bcl-2 on Japanese encephalitis virus-induced apoptosis in cultured cells. J. Virol. 71, 5963–5971 (1997).
Mukherjee, S. et al. Japanese encephalitis virus induces human neural stem/progenitor cell death by elevating GRP78, PHB and hnRNPC through ER stress. Cell Death Dis. 8, e2556–e2556 (2017).
Zhu, W. et al. Ferroptosis contributes to JEV-induced neuronal damage and neuroinflammation. Virol. Sin. S1995820X23001566. https://doi.org/10.1016/j.virs.2023.12.004 (2023).
Wang, Z.-Y., Zhen, Z.-D., Fan, D.-Y., Wang, P.-G. & An, J. Transcriptomic analysis suggests the M1 polarization and launch of diverse programmed cell death pathways in japanese encephalitis virus-infected macrophages. Viruses 12, 356 (2020).
Yu, P. et al. Pyroptosis: mechanisms and diseases. Sig Transduct. Target. Ther. 6, 128 (2021).
Wei, Y. et al. GSDME-mediated pyroptosis promotes the progression and associated inflammation of atherosclerosis. Nat. Commun. 14, 929 (2023).
Wei, C. et al. Brain endothelial GSDMD activation mediates inflammatory BBB breakdown. Nature. https://doi.org/10.1038/s41586-024-07314-2 (2024).
Hu, J. J. et al. FDA-approved disulfiram inhibits pyroptosis by blocking gasdermin D pore formation. Nat. Immunol. 21, 736–745 (2020).
Xu, M. et al. A systematic integrated analysis of brain expression profiles reveals YAP1 and other prioritized hub genes as important upstream regulators in Alzheimer’s disease. Alzheimers Dement. 14, 215–229 (2018).
You, Y. et al. ATP1A3 as a target for isolating neuron-specific extracellular vesicles from human brain and biofluids. Sci. Adv. 9, eadi3647 (2023).
Liu, H. et al. Biomarker identification for gender specificity of Alzheimer’s disease based on the glial transcriptome profiles. J. Vis. Exp. https://doi.org/10.3791/66552 (2024).
Yamamoto, H. et al. NDRG4 protein-deficient mice exhibit spatial learning deficits and vulnerabilities to cerebral ischemia. J. Biol. Chem. 286, 26158–26165 (2011).
Wen, L. et al. NDRG4 prevents cerebral ischemia/reperfusion injury by inhibiting neuronal apoptosis. Genes Dis. 6, 448–454 (2019).
Qu, M. et al. Histone deacetylase 6’s function in viral infection, innate immunity, and disease: latest advances. Front Immunol. 14, 1216548 (2023).
Banerjee, I. et al. Influenza A virus uses the aggresome processing machinery for host cell entry. Science 346, 473–477 (2014).
Zan, J. et al. Rabies virus infection induces microtubule depolymerization to facilitate viral RNA synthesis by upregulating HDAC6. Front. Cell. Infect. Microbiol. 7, 146 (2017).
Zhu, J., Coyne, C. B. & Sarkar, S. N. PKC alpha regulates Sendai virus-mediated interferon induction through HDAC6 and β-catenin: PKCα-HDAC6-β-catenin controls interferon induction. EMBO J. 30, 4838–4849 (2011).
Youn, G. S., Cho, H., Kim, D., Choi, S. Y. & Park, J. Crosstalk between HDAC6 and Nox2-based NADPH oxidase mediates HIV-1 Tat-induced pro-inflammatory responses in astrocytes. Redox Biol. 12, 978–986 (2017).
Ripamonti, C. et al. HDAC inhibition as potential therapeutic strategy to restore the deregulated immune response in severe COVID-19. Front. Immunol. 13, 841716 (2022).
Adhya, D., Dutta, K., Kundu, K. & Basu, A. Histone deacetylase inhibition by Japanese encephalitis virus in monocyte/macrophages: a novel viral immune evasion strategy. Immunobiology 218, 1235–1247 (2013).
Lu, C.-Y. et al. Tubacin, an HDAC6 selective inhibitor, reduces the replication of the Japanese encephalitis virus via the decrease of viral RNA synthesis. Int. J. Mol. Sci. 18, 954 (2017).
Klein, R. S. et al. Neuroinflammation during RNA viral infections. Annu. Rev. Immunol. 37, 73–95 (2019).
Fekete, R. et al. Microglia control the spread of neurotropic virus infection via P2Y12 signalling and recruit monocytes through P2Y12-independent mechanisms. Acta Neuropathol. 136, 461–482 (2018).
Wang, R. et al. Maternal immunization with a DNA vaccine candidate elicits specific passive protection against post-natal Zika virus infection in immunocompetent BALB/c mice. Vaccine 36, 3522–3532 (2018).
Frank, J. C., Song, B.-H. & Lee, Y.-M. Mice as an animal model for Japanese encephalitis virus research: mouse susceptibility, infection route, and viral pathogenesis. Pathogens 12, 715 (2023).
Sarkar, S. & Heise, M. T. Mouse models as resources for studying infectious diseases. Clin. Ther. 41, 1912–1922 (2019).
Bharucha, T. et al. Mouse models of Japanese encephalitis virus infection: a systematic review and meta-analysis using a meta-regression approach. PLoS Negl. Trop. Dis. 16, e0010116 (2022).
Saxena, V., Mathur, A., Krishnani, N. & Dhole, T. N. Kinetics of cytokine profile during intraperitoneal inoculation of Japanese encephalitis virus in BALB/c mice model. Microbes Infect. 10, 1210–1217 (2008).
Yang, H. et al. Peripheral nerve injury induced by Japanese encephalitis virus in C57BL/6 mouse. J. Virol. 97, e0165822 (2023).
Ma, X., Ju, L., Zheng, J., Li, Z. & Gao, L. Japanese encephalitis virus infection modulates gut microbiota of immunocompetent mice. Vet. Microbiol 311, 110779 (2025).
Wang, Z.-Y. et al. Axl deficiency promotes the neuroinvasion of Japanese encephalitis virus by enhancing IL-1α production from pyroptotic macrophages. J. Virol. 94, e00602-20 (2020).
Yang, Z.-Z. et al. Inhibition of GZMB activity ameliorates cognitive dysfunction by reducing demyelination in diabetic mice. Free Radic. Biol. Med. 225, 53–62 (2024).
Gong, C. et al. SAW: An efficient and accurate data analysis workflow for stereo-seq spatial transcriptomics. GigaByte 2024, gigabyte111 (2024).
Wolf, F. A., Angerer, P. & Theis, F. J. SCANPY: large-scale single-cell gene expression data analysis. Genome Biol. 19, 15 (2018).
Zhang, B. et al. Generating single-cell gene expression profiles for high-resolution spatial transcriptomics based on cell boundary images. GigaByte 2024, gigabyte110 (2024).
Hao, Y. et al. Integrated analysis of multimodal single-cell data. Cell 184, 3573–3587.e29 (2021).
Korsunsky, I. et al. Fast, sensitive and accurate integration of single-cell data with Harmony. Nat. Methods 16, 1289–1296 (2019).
Xu, S. et al. Using clusterProfiler to characterize multiomics data. Nat. Protoc. https://doi.org/10.1038/s41596-024-01020-z (2024).
Yu, G., Wang, L.-G., Han, Y. & He, Q.-Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16, 284–287 (2012).
Hänzelmann, S., Castelo, R. & Guinney, J. GSVA: gene set variation analysis for microarray and RNA-Seq data. BMC Bioinform. 14, 7 (2013).
Mishra, A. P. et al. Programmed cell death, from a cancer perspective: an overview. Mol. Diagn. Ther. 22, 281–295 (2018).
Seo, J., Nam, Y. W., Kim, S., Oh, D.-B. & Song, J. Necroptosis molecular mechanisms: recent findings regarding novel necroptosis regulators. Exp. Mol. Med. 53, 1007–1017 (2021).
Xie, Y. et al. Ferroptosis: process and function. Cell Death Differ. 23, 369–379 (2016).
Hao, Y. et al. Dictionary learning for integrative, multimodal and scalable single-cell analysis. Nat. Biotechnol. 42, 293–304 (2024).
Aibar, S. et al. SCENIC: single-cell regulatory network inference and clustering. Nat. Methods 14, 1083–1086 (2017).
Zhang, S. et al. The GSA family in 2025: A broadened sharing platform for multi-omics and multimodal data. Genom. Proteom. Bioinform. 23, qzaf072 (2025).
CNCB-NGDC Members and Partners Database resources of the national genomics data center, China National Center for Bioinformation in 2025. Nucleic Acids Res. 53, D30–D44 (2025).
Acknowledgements
This study was supported by grants from the Science and Technology Project of Southwest United Graduate School of Yunnan (202302AO370010 to G.C.), Shenzhen Medical Research Fund (B2404002 and B2402011 to G.C.), Yunnan Major Scientific and Technological Projects (202502AU100001 to G.C.), the National Key Research and Development Plan of China (2023YFA1801000 to G.C.), the National Natural Science Foundation of China (82341118 and 82341082 to Z.O., 82502710 to Z.W., 32188101 and 82422049 to G.C.), the Shenzhen San Ming Project for Prevention and Research on Vector-borne Diseases (SZSM202211023 to G.C.), the New Cornerstone Science Foundation through the New Cornerstone Investigator Program to G.C., and the XPLORER PRIZE to G.C. We thank China National GeneBank for providing sequencing services for this project. We would also like to thank DCS Cloud (https://cloud.stomics.tech) for providing the computational resources and software support.
Author information
Authors and Affiliations
Contributions
G.C., J.L., Z.O., and Z.W. conceived the study and designed the research. Z.W. constructed the animal models, collected samples, and performed all validation experiments, including flow cytometry, qPCR, and immunofluorescence assays. Y.L. and O.Z. assisted Z.W. with the validation experiments. Z.O., P.R., S.L., A.C., and J.Z. designed and optimized the orthoflavivirus-specific Stereo-seq chips. P.R., Q.C., and Ying’an L. conducted Stereo-seq experiments. Z.P., W.L., and G.H. performed sequencing of Stereo-seq libraries. Z.O., Q.C., X.H., Ying’an L., J.W., D.W., and J.S. carried out bioinformatic analyses. Z.O., Z.W., Q.C., and X.H. wrote and revised the manuscript. G.C., J.L., Z.O., X.X., X.J., and Z.D. supervised the project and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
X.X., A.C., and S.L. are the co-inventors of Stereo-seq technology. Employees of BGI have stock holdings in BGI. The other authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Juan Quintana and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Ou, Z., Wang, Z., Chen, Q. et al. Spatial transcriptomics uncovers vasculature-centered cellular interactions driving Japanese encephalitis progression in a mouse model. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70047-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70047-5