Abstract
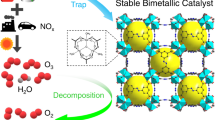
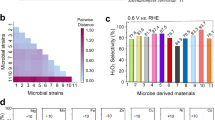
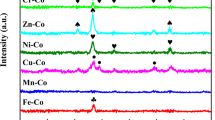
Ground-level ozone (O3) is a major air pollutant, and catalytic decomposition represents a promising strategy for its removal. However, maintaining high catalytic efficiency under humid conditions remains a considerable challenge. In this study, we encapsulate ultrafine metal oxides (UMOs; e.g., Co3O4, NiO) within the nanopores of an Fe3O-cluster-based metal–organic framework, PCN-333(Fe), for catalytic O3 decomposition. The optimized 30% Co3O4@PCN-333(Fe) catalyst achieves sustained 100% O3 conversion for over 120 hours in a continuous airflow containing 40 ppm O3 under high space velocity (1.75 × 105 h-1) and a broad range of humidity (10-90% RH). Mechanistic investigations reveal that the exceptional performance originates from an interfacial hydrogen-atom transfer process between Co3O4 and the Fe3O clusters of PCN-333(Fe), as confirmed by in situ diffuse reflectance infrared Fourier-transform spectroscopy (DRIFTS) and in situ Raman spectroscopy. This work proposes a general principle for designing humidity-immune catalytic interfaces between metal oxides and porous materials, thereby providing a practical foundation for sustainable control of pollutant emissions in complex environments.
Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the article and its Supplementary Information, or from the corresponding author upon reasonable request. Source data are provided with this paper.
References
Yeung, L. Y. et al. Isotopic constraint on the twentieth-century increase in tropospheric ozone. Nature 570, 224–227 (2019).
Zhao, Y. et al. Air pollution modulates trends and variability of the global methane budget. Nature 642, 369–375 (2025).
Finlayson-Pitts, B. J. et al. Tropospheric air pollution: ozone, airborne toxics, polycyclic aromatic hydrocarbons, and particles. Science 276, 1045–1051 (1997).
Wang, N. et al. Typhoon-boosted biogenic emission aggravates cross-regional ozone pollution in China. Sci. Adv. 8, eabl6166 (2022).
Feng, Z. et al. Ozone pollution threatens the production of major staple crops in East Asia. Nat. Food 3, 47–56 (2022).
Achebak, H. et al. Geographic sources of ozone air pollution and mortality burden in Europe. Nat. Med. 30, 1732–1738 (2024).
Simpkins, G. Ozone damage costs billions. Nat. Rev. Earth Environ. 3, 102–102 (2022).
Li, X. et al. Recent advances in catalytic decomposition of ozone. J. Environ. Sci. 94, 14–31 (2020).
Zhu, Y. et al. Rapid ozone decomposition over water-activated monolithic moo3/graphdiyne nanowalls under high humidity. Angew. Chem., Int. Ed. 62, e202309158 (2023).
Yang, J. et al. Defect-based lewis pairs on hydrophobic MnO mesocrystals for robust and efficient ozone decomposition. Nat. Commun. 16, 2922–2930 (2025).
Dai, W. et al. Exceptional ozone decomposition over δ-MnO2/AC under an entire humidity environment. Environ. Sci. Technol. 57, 17727–17736 (2023).
Deng, H. et al. Role of structural defects in MnOx promoted by Ag doping in the catalytic combustion of volatile organic compounds and ambient decomposition of O3. Environ. Sci. Technol. 53, 10871–10879 (2019).
Gong, S. et al. Heterostructured Ni/NiO nanocatalysts for ozone decomposition. ACS Appl. Nano Mater. 3, 597–607 (2019).
Zhu, G. et al. Encapsulate α-MnO2 nanofiber within graphene layer to tune surface electronic structure for efficient ozone decomposition. Nat. Commun. 12, 4152–4161 (2021).
Han, Y. et al. Topology-directed cage engineering in MOFs for Efficient C2H2/CO2/C2H4 separation. J. Am. Chem. Soc. 147, 19262–19271 (2025).
Wen, L. et al. Electronic state and microenvironment modulation of metal nanoparticles stabilized by MOFs for boosting electrocatalytic nitrogen reduction. Adv. Mater. 35, 2210669–2210676 (2023).
Feng, D. et al. Zirconium-metalloporphyrin PCN-222: mesoporous metal-organic frameworks with ultrahigh stability as biomimetic catalysts. Angew. Chem., Int. Ed. 51, 10307–10310 (2012).
Hu, S. et al. Selectivity control in the direct CO esterification over Pd@UiO-66: the Pd location matters. Angew. Chem. Int. Ed. 62, e202311625 (2023).
Ling, L.-L. et al. Light-assisted CO2 hydrogenation over Pd3Cu@UiO-66 promoted by active sites in close proximity. Angew. Chem., Int. Ed. 61, e202116396 (2022).
Ko, S. et al. Mesoporous CuO particles threaded with CNTs for high-performance lithium-ion battery anodes. Adv. Mater. 24, 4451–4456 (2012).
Kwon, H. J. et al. Large-scale synthesis and medical applications of uniform-sized metal oxide nanoparticles. Adv. Mater. 30, 1704290–1704313 (2018).
Zhou, T. et al. Ultrafine metal nanoparticles isolated on oxide nano-islands as exceptional sintering-resistant catalysts. Nat. Mater. 24, 891–899 (2025).
Qiao, G.-Y. et al. Perovskite quantum dots encapsulated in a mesoporous metal-organic framework as synergistic photocathode materials. J. Am. Chem. Soc. 143, 14253–14260 (2021).
Feng, D. et al. Stable Metal-organic frameworks containing single-molecule traps for enzyme encapsulation. Nat. Commun. 6, 5979–5986 (2015).
Jiang, Z. et al. Filling metal-organic framework mesopores with TiO2 for CO2 photoreduction. Nature 586, 549–554 (2020).
Wang, X. et al. Atomic-scale insights into surface lattice oxygen activation at the spinel/perovskite interface of Co3O4/La0.3Sr0.7CoO3. Angew. Chem., Int. Ed. 58, 11720–11725 (2019).
Wan, W. et al. Carbon-supported single Fe/Co/Ni atom catalysts for water oxidation: unveiling the dynamic active sites. Angew. Chem., Int. Ed. 64, e202424629 (2025).
Kim, M. et al. Reducing the barrier energy of self-reconstruction for anchored cobalt nanoparticles as highly active oxygen evolution electrocatalyst. Adv. Mater. 31, 1901977–1901984 (2019).
Aijaz, A. et al. Immobilizing highly catalytically active pt nanoparticles inside the pores of metal-organic framework: a double solvents approach. J. Am. Chem. Soc. 134, 13926–13929 (2012).
Liu, J. et al. In situ synthesis of highly dispersed and ultrafine metal nanoparticles from chalcogels. J. Am. Chem. Soc. 139, 2900–2903 (2017).
Xie, Y. et al. Modulation in spin state of Co3O4 decorated Fe single atom enables a superior rechargeable zinc-air battery performance. Adv. Mater. 37, 2414801–2414810 (2025).
Qian, J. et al. Enhanced stability and narrowed D-band gap of Ce-doped Co3O4 for rechargeable aqueous Zn-air battery. Adv. Funct. Mater. 33, 2212021–2212029 (2023).
Wang, H. et al. An iron-containing metal-organic framework as a highly efficient catalyst for ozone decomposition. Angew. Chem., Int. Ed. 57, 16416–16420 (2018).
Sun, Z. B. et al. Ozone decomposition by a manganese-organic framework over the entire humidity range. J. Am. Chem. Soc. 143, 5150–5157 (2021).
Dong, C. et al. Catalytic ozone decomposition and adsorptive VOCs removal in bimetallic metal-organic frameworks. Nat. Commun. 13, 4991–5000 (2022).
Wang, Z. et al. Layered double hydroxide catalysts for ozone decomposition: the synergic role of M2+ and M3+. Environ. Sci. Technol. 56, 1386–1394 (2022).
Li, X. et al. Oxygen vacancies induced by transition metal doping in γ-MnO2 for highly efficient ozone decomposition. Environ. Sci. Technol. 52, 12685–12696 (2018).
Wan, X. et al. Ozone decomposition below room temperature using Mn-based mullite YMn2O5. Environ. Sci. Technol. 56, 8746–8755 (2022).
Li, X. et al. Regulating the chemical state of silver via surface hydroxyl groups to enhance ozone decomposition performance of Ag/Fe2O3 catalyst. Catal. Today 410, 117–126 (2023).
Valdés, H. et al. New insight of the influence of acidic surface sites of zeolite on the ability to remove gaseous ozone using operando DRIFTS studies. Micropor. Mesopor. Mat. 294, 109912–109922 (2020).
Liang, X. et al. Mesoporous poorly crystalline α-Fe2O3 with abundant oxygen vacancies and acid sites for ozone decomposition. Sci. Total Environ. 804, 150161–150172 (2022).
Jing, C. et al. Electrocatalyst with dynamic formation of the dual-active site from the dual pathway observed by in situ Raman spectroscopy. ACS Catal. 12, 10276–10284 (2022).
Bo, X. et al. Operando Raman spectroscopy reveals Cr-induced-phase reconstruction of NiFe and CoFe oxyhydroxides for enhanced electrocatalytic water oxidation. Chem. Mater. 32, 4303–4311 (2020).
Niu, S. et al. Cu regulating the bifunctional activity of Co-O sites for the high-performance rechargeable zinc-air battery. ACS Appl. Mater. Interfaces 16, 36295–36303 (2024).
VandeVondele, J. et al. Quickstep: fast and accurate density functional calculations using a mixed Gaussian and plane waves approach. Comput. Phys. Commun. 167, 103–128 (2005).
Goedecker, S. et al. Separable dual-space Gaussian pseudopotentials. Phys. Rev. B 54, 1703–1710 (1996).
Hartwigsen, C. et al. Relativistic separable dual-space Gaussian pseudopotentials from H to Rn. Phys. Rev. B 58, 3641–3662 (1998).
Krack, M. et al. All-electron ab-initio molecular dynamics. Phys. Chem. Chem. Phys. 2, 2105–2112 (2000).
VandeVondele, J. et al. Gaussian basis sets for accurate calculations on molecular systems in gas and condensed phases. J. Chem. Phys. 127, 114105 (2007).
Perdew, J. P. et al. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S. et al. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22171121), the Applied Basic Research Plan of Liaoning Province (2023JH2/101300007).
Author information
Authors and Affiliations
Contributions
Z.H. conceived and designed this project. Y.L. and T.L. performed the experiments, Y.L. and Y.H. carried out the DFT calculation, Y.L. analyzed the data, Y.L., Z.H., and Z.-M.Z. wrote and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Yongfa Zhu, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lou, Y., Han, Y., Li, T. et al. Metal-organic framework-confined Co3O4 for humidity-immune ozone decomposition. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70324-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70324-3