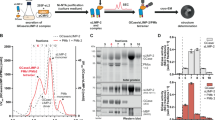
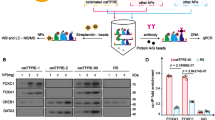
Abstract
In vertebrates, newly synthesized lysosomal enzymes traffic to lysosomes through the mannose-6-phosphate (M6P) pathway. The Golgi membrane protein LYSET was recently discovered to regulate lysosome biogenesis by controlling the level of GlcNAc-1-phosphotransferase (GNPT). However, its working mechanism remained unclear. In this study, we demonstrate that LYSET is a two-transmembrane protein essential for GNPT stability, cleavage by Site-1 Protease (S1P), and enzymatic activity. We reconcile conflicting models by showing that LYSET enhances GNPT cleavage and prevents its mislocalization to lysosomes for degradation. We further establish that LYSET achieves this by interacting with GOLPH3 and retromer complexes to anchor the LYSET-GNPT complex at the Golgi. Alanine mutagenesis identified an F4XXR7 motif in LYSET’s N-tail for GOLPH3 binding. The retromer further promotes Golgi retention by binding to the C-terminal of LYSET and recycling it from endolysosomes. Together, our findings reveal LYSET’s multifaceted role in stabilizing GNPT, retaining it at the Golgi, and ensuring the fidelity of the M6P pathway, thereby providing insights into its molecular function.
Similar content being viewed by others
Data availability
Data supporting this study is provided within the paper and supplementary files. Source data are provided with this paper.
References
Ghosh, P., Dahms, N. M. & Kornfeld, S. Mannose 6-phosphate receptors: new twists in the tale. Nat. Rev. Mol. Cell Biol. 4, 202–212 (2003).
Settembre, C. & Perera, R. M. Lysosomes as coordinators of cellular catabolism, metabolic signalling and organ physiology. Nat. Rev. Mol. Cell Biol. https://doi.org/10.1038/s41580-023-00676-x (2023).
Khan, S. A. & Tomatsu, S. C. Mucolipidoses overview: past, present, and future. Int. J. Mol. Sci. https://doi.org/10.3390/ijms21186812 (2020).
Zhang, B., Yang, X. & Li, M. LYSET/TMEM251/GCAF is critical for autophagy and lysosomal function by regulating the mannose-6-phosphate (M6P) pathway. Autophagy, 19, 1596–1598 (2023).
Zhang, W. et al. GCAF(TMEM251) regulates lysosome biogenesis by activating the mannose-6-phosphate pathway. Nat. Commun. 13, 5351 (2022).
Pechincha, C. et al. Lysosomal enzyme trafficking factor LYSET enables nutritional usage of extracellular proteins. Science 378, eabn5637 (2022).
Richards, C. M. et al. The human disease gene LYSET is essential for lysosomal enzyme transport and viral infection. Science 378, eabn5648 (2022).
Ain, N. U. et al. Biallelic TMEM251 variants in patients with severe skeletal dysplasia and extreme short stature. Hum. Mutat. 42, 89–101 (2021).
Braulke, T., Carette, J. E. & Palm, W. Lysosomal enzyme trafficking: from molecular mechanisms to human diseases. Trends Cell Biol. https://doi.org/10.1016/j.tcb.2023.06.005 (2023).
Qiao, W., Richards, C. M. & Jabs, S. LYSET/TMEM251- a novel key component of the mannose 6-phosphate pathway. Autophagy 19, 2143–2145 (2023).
van Meel, E., Qian, Y. & Kornfeld, S. A. Mislocalization of phosphotransferase as a cause of mucolipidosis III αβ. Proc. Natl. Acad. Sci.USA 111, 3532–3537 (2014).
Muller-Loennies, S., Galliciotti, G., Kollmann, K., Glatzel, M. & Braulke, T. A novel single-chain antibody fragment for detection of mannose 6-phosphate-containing proteins: application in mucolipidosis type II patients and mice. Am. J. Pathol. 177, 240–247 (2010).
van Meel, E. et al. Multiple Domains of GlcNAc-1-phosphotransferase mediate recognition of lysosomal enzymes. J. Biol. Chem. 291, 8295–8307 (2016).
Li, H. et al. Structure of the human GlcNAc-1-phosphotransferase alphabeta subunits reveals regulatory mechanism for lysosomal enzyme glycan phosphorylation. Nat. Struct. Mol. Biol. https://doi.org/10.1038/s41594-022-00748-0 (2022).
Thomas, G. Furin at the cutting edge: from protein traffic to embryogenesis and disease. Nat. Rev. Mol. Cell Biol. 3, 753–766 (2002).
Kisselev, A. F., Akopian, T. N. & Goldberg, A. L. Range of sizes of peptide products generated during degradation of different proteins by archaeal proteasomes. J. Biol. Chem. 273, 1982–1989 (1998).
Kisselev, A. F., Akopian, T. N., Woo, K. M. & Goldberg, A. L. The sizes of peptides generated from protein by mammalian 26 and 20S proteasomes. Implications for understanding the degradative mechanism and antigen presentation. J. Biol. Chem. 274, 3363–3371 (1999).
Jumper, J. et al. Highly accurate protein structure prediction with AlphaFold. Nature 596, 583–589 (2021).
Tsirigos, K. D., Peters, C., Shu, N., Kall, L. & Elofsson, A. The TOPCONS web server for consensus prediction of membrane protein topology and signal peptides. Nucleic Acids Res. 43, W401–W407 (2015).
Viklund, H. & Elofsson, A. OCTOPUS: improving topology prediction by two-track ANN-based preference scores and an extended topological grammar. Bioinformatics 24, 1662–1668 (2008).
Liu, L., Doray, B. & Kornfeld, S. Recycling of Golgi glycosyltransferases requires direct binding to coatomer. Proc. Natl. Acad. Sci. USA 115, 8984–8989 (2018).
Welch, L. G., Peak-Chew, S. Y., Begum, F., Stevens, T. J. & Munro, S. GOLPH3 and GOLPH3L are broad-spectrum COPI adaptors for sorting into intra-Golgi transport vesicles. J. Cell Biol. https://doi.org/10.1083/jcb.202106115 (2021).
Tanenbaum, M. E., Gilbert, L. A., Qi, L. S., Weissman, J. S. & Vale, R. D. A protein-tagging system for signal amplification in gene expression and fluorescence imaging. Cell 159, 635–646 (2014).
Ali, M. F., Chachadi, V. B., Petrosyan, A. & Cheng, P. W. Golgi phosphoprotein 3 determines cell binding properties under dynamic flow by controlling Golgi localization of core 2 N-acetylglucosaminyltransferase 1. J. Biol. Chem. 287, 39564–39577 (2012).
Tu, L., Tai, W. C., Chen, L. & Banfield, D. K. Signal-mediated dynamic retention of glycosyltransferases in the Golgi. Science 321, 404–407 (2008).
Rizzo, R. et al. Golgi maturation-dependent glycoenzyme recycling controls glycosphingolipid biosynthesis and cell growth via GOLPH3. EMBO J. 40, e107238 (2021).
Seaman, M. N., Burd, C. G. & Emr, S. D. Receptor signalling and the regulation of endocytic membrane transport. Curr. Opin. Cell Biol. 8, 549–556 (1996).
Seaman, M. N., Marcusson, E. G., Cereghino, J. L. & Emr, S. D. Endosome to Golgi retrieval of the vacuolar protein sorting receptor, Vps10p, requires the function of the VPS29, VPS30, and VPS35 gene products. J. Cell Biol. 137, 79–92 (1997).
Suzuki, S. W., Chuang, Y. S., Li, M., Seaman, M. N. J. & Emr, S. D. A bipartite sorting signal ensures specificity of retromer complex in membrane protein recycling. J. Cell Biol. 218, 2876–2886 (2019).
Seaman, M. N. The retromer complex - endosomal protein recycling and beyond. J. Cell Sci. 125, 4693–4702 (2012).
Arighi, C. N., Hartnell, L. M., Aguilar, R. C., Haft, C. R. & Bonifacino, J. S. Role of the mammalian retromer in sorting of the cation-independent mannose 6-phosphate receptor. J. Cell Biol. 165, 123–133 (2004).
Humphrey, J. S., Peters, P. J., Yuan, L. C. & Bonifacino, J. S. Localization of TGN38 to the trans-Golgi network: involvement of a cytoplasmic tyrosine-containing sequence. J. Cell Biol. 120, 1123–1135 (1993).
Gorelik, A., Illes, K., Bui, K. H. & Nagar, B. Structures of the mannose-6-phosphate pathway enzyme, GlcNAc-1-phosphotransferase. Proc. Natl. Acad. Sci. USA 119, e2203518119 (2022).
Du, S. et al. Structural insights into how GlcNAc-1-phosphotransferase directs lysosomal protein transport. J. Biol. Chem. 298, 101702 (2022).
Doray, B. et al. LYSET facilitates integration of both the N- and C-terminal transmembrane helices/cytoplasmic domains of GlcNAc-1-phosphotransferase. Mol. Biol. Cell 36, br12 (2025).
Brauer, B. K. et al. GOLPH3 and GOLPH3L maintain Golgi localization of LYSET and a functional mannose 6-phosphate transport pathway. EMBO. J. https://doi.org/10.1038/s44318-024-00305-z (2024).
Ran, F. A. et al. Genome engineering using the CRISPR-Cas9 system. Nat. Protoc. 8, 2281–2308 (2013).
Zhang, W. et al. A conserved ubiquitin- and ESCRT-dependent pathway internalizes human lysosomal membrane proteins for degradation. PLoS Biol. 19, e3001361 (2021).
Shao, W. & Espenshade, P. J. Sterol regulatory element-binding protein (SREBP) cleavage regulates Golgi-to-endoplasmic reticulum recycling of SREBP cleavage-activating protein (SCAP). J. Biol. Chem. 289, 7547–7557 (2014).
Valenzano, K. J., Remmler, J. & Lobel, P. Soluble insulin-like growth factor II/mannose 6-phosphate receptor carries multiple high molecular weight forms of insulin-like growth factor II in fetal bovine serum. J. Biol. Chem. 270, 16441–16448 (1995).
Venkatarangan, V. et al. ER-associated degradation in cystinosis pathogenesis and the prospects of precision medicine. J Clin. Invest. https://doi.org/10.1172/JCI169551 (2023).
Yang, X. et al. ESCRT, not intralumenal fragments, sorts ubiquitinated vacuole membrane proteins for degradation. J. Cell Biol. https://doi.org/10.1083/jcb.202012104 (2021).
Acknowledgements
We thank the Li and Kornfeld laboratory members for their helpful discussion and technical support. This research is supported by the Protein Folding and Diseases Initiative, a MICHR Pathway Pilot grant, and NIH grants R01HD109346 to M. Li and R01CA008759 to S. Kornfeld.
Author information
Authors and Affiliations
Contributions
Conceptualization: X.Y., B.D., D.H, V.V., S.K. and M.L.; methodology: X.Y., V.V., D.H, B.D., B.J., L.C. and W.Z.; investigation: X.Y., B.D., D.H., V.V., Z.D, W.Z., L.C., B.J., L.Y., J.L. and B.Z.; writing & editing: V.V., B.D., D.H., X.Y. and M.L.; funding acquisition: M.L. and S.K.; resources & supervision: M.L and S.K.; X.Y., BD., D.H. and V.V. contributed equally, and all four have the right to list themselves first in bibliographic documents.
Corresponding author
Ethics declarations
Competing interests
S.K. was co-founder of M6P Therapeutics and held stock options in the company. The rest of the authors declare no competing interests.
Dedication
We wish to dedicate this paper to the memory of Stuart Kornfeld (1936-2025) who devoted his life to advancing our understanding of the mannose-6-phosphate pathway.
Peer review
Peer review information
Nature Communications thanks Alberto Luini who co-reviewed with Domenico Russo; Vladimir Lupashin and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, X., Doray, B., Henn, D. et al. Molecular insights into the regulation of GNPTαβ by LYSET. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70402-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70402-6