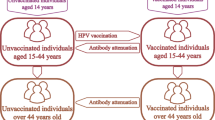
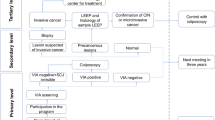
Abstract
Though cervical cancer is largely preventable, Africa faces a high burden with thousands of deaths. Highlighting the urgent need for action toward the WHO’s 90-70-90 elimination strategy, this study presents the epidemiological burden of HPV and cervical cancer in Africa and advocates for decisive measures to improve screening and implement effective prevention through vaccination. Here, we discuss which is the better option for Africa: a single-dose or a two-dose vaccine regimen. Furthermore, we explore screening methods, such as methylation markers and AI-based screening, as well as treatment-related side effects and the prognosis of cervical cancer in Africa.
Similar content being viewed by others
Data availability
The compiled and processed dataset generated from these sources for the analysis and visualizations in this study has been deposited in the Zenodo repository under accession code https://doi.org/10.5281/zenodo.17722637 (https://doi.org/10.5281/zenodo.17512055). Source data are provided with this paper.
Code availability
R software (version 4.4.2) was used for all analyses and visualization. The custom code used in this study has been deposited in the Zenodo repository under accession code (https://doi.org/10.5281/zenodo.17534613) (https://doi.org/10.5281/zenodo.17511134).
References
WHO. Cervical cancer. https://www.who.int/news-room/fact-sheets/detail/cervical-cancer#:~:text=Key%20facts,%2Dincome%20countries%20 (2024).
Sundström, K. & Elfström, K. M. Advances in cervical cancer prevention: efficacy, effectiveness, elimination?. PLoS Med 17, e1003035 (2020).
Patel, C. et al. The impact of 10 years of human papillomavirus (HPV) vaccination in Australia: what additional disease burden will a nonavalent vaccine prevent? Euro Surveill 23, 1700737 (2018).
Hall, M. T. et al. The projected timeframe until cervical cancer elimination in Australia: a modelling study. Lancet Public Health 4, e19–e27 (2019).
Falcaro, M. et al. The effects of the national HPV vaccination programme in England, UK, on cervical cancer and grade 3 cervical intraepithelial neoplasia incidence: a register-based observational study. Lancet 398, 2084–2092 (2021).
WHO. in Global Strategy To Accelerate The Elimination Of Cervical Cancer As A Public Health Problem 7 (Geneva, 2020).
Ferlay J. et al. Global Cancer Observatory: Cancer Today. 2024 Lyon, France: International Agency for Research on Cancer. https://gco.iarc.who.int/today (2024).
Akanda, R., Kawale, P. & Moucheraud, C. Cervical cancer prevention in Africa: a policy analysis. J. Cancer Policy 32, 100321 (2022).
Singh, D. et al. Global estimates of incidence and mortality of cervical cancer in 2020: a baseline analysis of the WHO Global Cervical Cancer Elimination Initiative. Lancet Glob. Health 11, e197–e206 (2023).
Campos, N. G. et al. The health and economic impact of scaling cervical cancer prevention in 50 low- and lower-middle-income countries. Int J. Gynaecol. Obstet. 138, 47–56 (2017).
Bruni, L. et al. Cervical human papillomavirus prevalence in 5 continents: meta-analysis of 1 million women with normal cytological findings. J. Infect. Dis. 202, 1789–1799 (2010).
Mbulawa, Z. Z. A., Somdyala, N. I., Mabunda, S. A. & Williamson, A. L. High human papillomavirus prevalence among females attending high school in the Eastern Cape Province of South Africa. PLoS One 16, e0253074 (2021).
Obeid, D. et al. Sexually transmitted infections in the middle east and North Africa: comprehensive systematic review and meta-analysis. BMC Infect. Dis. 24, 1229 (2024).
Obeid, D. A., Almatrrouk, S. A., Alfageeh, M. B., Al-Ahdal, M. N. A. & Alhamlan, F. S. Human papillomavirus epidemiology in populations with normal or abnormal cervical cytology or cervical cancer in the Middle East and North Africa: A systematic review and meta-analysis. J. Infect. Public Health 13, 1304–1313 (2020).
Pimple, S. & Mishra, G. Cancer cervix: epidemiology and disease burden. Cytojournal 19, 21 (2022).
Derbie, A. et al. Human papillomavirus genotype distribution in Ethiopia: an updated systematic review. Virol. J. 19, 13 (2022).
Okoye, J. O. et al. Racial disparities associated with the prevalence of vaccine and non-vaccine HPV types and multiple HPV infections between asia and africa: a systematic review and meta-analysis. Asian Pac. J. Cancer Prev. 22, 2729–2741 (2021).
Seyoum, A. et al. Prevalence and genotype distribution of high-risk human papillomavirus infection among sub-saharan african women: a systematic review and meta-analysis. Front Public Health 10, 890880 (2022).
Chachage, M. et al. High-risk human papillomavirus genotype distribution among women living with and at risk for HIV in Africa. Aids 37, 625–635 (2023).
Seyoum, A. et al. High rate of non-vaccine targeted high-risk HPV genotypes circulate among women in Eastern Ethiopia. Sci. Rep. 14, 958 (2024).
Happel, A. U. et al. Natural history of high-risk human papillomavirus in kenyan and south african women: implications for vaccination campaigns and cervical cancer screening programs. Open Forum Infect. Dis. 11, ofae690 (2024).
Boublenza, L. et al. Prevalence of human papillomavirus infection among women at a gynaecological clinic in Tlemcen, Algeria. Afr. J. Reprod. Health 27, 56–63 (2023).
Tounkara, F. K. et al. Type-specific incidence, persistence and factors associated with human papillomavirus infection among female sex workers in Benin and Mali, West Africa. Int J. Infect. Dis. 106, 348–357 (2021).
Tawe, L. et al. Human papillomavirus genotypes in women with invasive cervical cancer with and without human immunodeficiency virus infection in Botswana. Int J. Cancer 146, 1667–1673 (2020).
Grover, S. et al. Characterization of HPV subtypes in invasive cervical cancer in Botswana patients using a pan-pathogen microarray technology. Tumour Virus Res 15, 200262 (2023).
Kelly, H. A. et al. Diagnostic accuracy of cervical cancer screening and screening-triage strategies among women living with HIV-1 in Burkina Faso and South Africa: A cohort study. PLoS Med 18, e1003528 (2021).
Meda, Z. C. et al. Factors associated with positive human papillomavirus (HPV) test results in cervical precancer screening: a cross-sectional study at Souro Sanou National Teaching Hospital (SSNTH) in Bobo Dioulasso, Burkina Faso. BMC Public Health 24, 2900 (2024).
Vassilakos, P. et al. A cross-sectional study exploring triage of human papillomavirus (HPV)-positive women by visual assessment, manual and computer-interpreted cytology, and HPV-16/18-45 genotyping in Cameroon. Int J. Gynecol. Cancer 31, 808–816 (2021).
Manga, S. M. et al. Human papillomavirus types and cervical cancer screening among female sex workers in Cameroon. Cancers (Basel) 16, 243 (2024).
Vieira, R., Montezuma, D., Barbosa, C. & Macedo Pinto, I. Cervical cytology and HPV distribution in Cape Verde: A snapshot of a country taken during its first HPV nation-wide vaccination campaign. Tumour Virus Res 17, 200280 (2024).
Tchounga, B. et al. Human papilloma viruses infection among adolescent females perinatally infected with HIV in Côte d’Ivoire. Sex. Transm. Infect. 97, 238–243 (2021).
A. Ibrahim, S., A. Abdel-Salam, S., I. Amer, M., A. El-Sayed, A. & A. Hassan, R. Prevalence of human papilloma virus (HPV) and type distribution of genotypes (6, 11, 16 and 18) among Egyptian women. Egypt. J. Med. Microbiol.EJMM. 29, 45–50 (2020).
Ashry, M. et al. Prevalence and risk factors of human papilloma virus infection among women living with HIV, Egypt, a cross sectional study. BMC Public Health 24, 1821 (2024).
Ginindza, T. G., Forestier, M. & Almonte, M. Cervical cancer screening by visual inspection and HPV testing in Eswatini. Prev. Med 161, 107144 (2022).
Derbie, A. et al. High-risk human papillomavirus genotype distribution among women with gynecology complaints in northwest Ethiopia. Infect. Agent Cancer 18, 4 (2023).
Teka, B. et al. Population-based human papillomavirus infection and genotype distribution among women in rural areas of South Central Ethiopia. Int J. Cancer 148, 723–730 (2021).
Tesfaye, E. et al. Prevalence of human papillomavirus infection and associated factors among women attending cervical cancer screening in setting of Addis Ababa, Ethiopia. Sci. Rep. 14, 4053 (2024).
Moussavou-Boundzanga, P. et al. HPV genotypes in high-grade cervical lesions and invasive cervical carcinoma detected in Gabonese women. Infect. Agent Cancer 18, 16 (2023).
Bah, H. et al. Human papillomavirus type-specific distribution in cervical intraepithelial neoplasia and cancer in The Gambia prior to HPV immunization programme: a baseline for monitoring the quadrivalent vaccine. Infect. Agent Cancer 19, 44 (2024).
Nartey, Y. et al. Human papillomavirus genotype distribution among women with and without cervical cancer: Implication for vaccination and screening in Ghana. PLoS One 18, e0280437 (2023).
Akakpo, P. K. et al. The distribution of hrHPV genotypes among cervical cancer cases diagnosed across Ghana: a cross-sectional study. BMC Infect. Dis. 24, 356 (2024).
Kaba, G. et al. Diversity of cervicovaginal human papillomavirus (HPV) genotypes and naturally occurring E6/E7 DNA polymorphisms of HPV-16 in Ghana. Tumour Virus Res 15, 200261 (2023).
Tekpor, E. et al. Prevalence of high-risk human papillomavirus infection and cervical lesions among female migrant head porters (kayayei) in Accra, Ghana: a pilot cross-sectional study. BMC Public Health 24, 659 (2024).
Effah, K. et al. Nationwide cervical precancer screening in Ghana: concurrent HPV DNA testing and visual inspection under an expanded hub-and-spoke model. Sci. Rep. 15, 1744 (2025).
Effah, K. et al. Cervical precancer screening using self-sampling, HPV DNA testing, and mobile colposcopy in a hard-to-reach community in Ghana: a pilot study. BMC Cancer 24, 1367 (2024).
Guthrie, B. L. et al. Human papillomavirus and abnormal cervical lesions among HIV-infected women in HIV-discordant couples from Kenya. Sex. Transm. Infect. 96, 457–463 (2020).
Sweet, K. et al. Prevalence, incidence, and distribution of human papillomavirus types in female sex workers in Kenya. Int J. STD AIDS 31, 109–118 (2020).
Benski, A. C. et al. HPV self-testing for primary cervical cancer screening in Madagascar: VIA/VILI triage compliance in HPV-positive women. PLoS One 14, e0220632 (2019).
Mungo, C. et al. Comparison of the ScreenFire and Xpert HPV assays for the detection of human papillomavirus and cervical precancer among women living with HIV in Malawi. Infect. Agent Cancer 19, 24 (2024).
Jary, A. et al. Prevalence of cervical HPV infection, sexually transmitted infections and associated antimicrobial resistance in women attending cervical cancer screening in Mali. Int J. Infect. Dis. 108, 610–616 (2021).
Abdoudaim, M. S. et al. Human Papillomavirus genotypes distribution in high-grade cervical lesions and invasive cervical carcinoma in women living in Mauritania: implications for cervical cancer prevention and hpv prophylactic vaccination. Diagnostics (Basel) 14, 1986 (2024).
Rogua, H. et al. Human Papillomavirus genotypes distribution and associated risk factors among women living in Southern Morocco. Heliyon 9, e22497 (2023).
Tadlaoui, K. A. et al. Prevalence of specific human papillomavirus genotypes among Moroccan women with invasive cervical cancer. Exploration Med. 5, 76–88 (2024).
Maueia, C. et al. Identification of the human papillomavirus genotypes, according to the human immunodeficiency virus status in a cohort of women from Maputo, Mozambique. Viruses 14, 24 (2021).
Salcedo, M. P. et al. The Capulana study: a prospective evaluation of cervical cancer screening using human papillomavirus testing in Mozambique. Int J. Gynecol. Cancer 30, 1292–1297 (2020).
Nejo, Y. T., Olaleye, D. O. & Odaibo, G. N. Molecular characterisation of genital human papillomavirus among women in Southwestern, Nigeria. PLoS One 14, e0224748 (2019).
Cosmas, N. T. et al. Prevalence of vaginal HPV infection among adolescent and early adult girls in Jos, North-Central Nigeria. BMC Infect. Dis. 22, 340 (2022).
Mpunga, T. et al. Human papillomavirus genotypes in cervical and other HPV-related anogenital cancer in Rwanda, according to HIV status. Int J. Cancer 146, 1514–1522 (2020).
Muhimpundu, M. A. et al. Screen, notify, see, and treat: initial results of cervical cancer screening and treatment in Rwanda. JCO Glob. Oncol. 7, 632–638 (2021).
Diop-Ndiaye, H. et al. Respective prevalence of high-risk HPV genotypes in cervical neoplasia in Senegal. J. Med Virol. 93, 5110–5117 (2021).
Downham, L. et al. Field experience with the 8-HPV-type oncoprotein test for cervical cancer screening among HPV-positive women living with and without HIV in LMICs. Int J. Cancer 155, 816–827 (2024).
Denny, L. et al. Point-of-care testing with Xpert HPV for single-visit, screen-and-treat for cervical cancer prevention: a demonstration study in South Africa. Sci. Rep. 13, 16182 (2023).
Dreyer, G., Maske, C. & Stander, M. Clinical evaluation and budget impact analysis of cervical cancer screening using cobas 4800 HPV screening technology in the public sector of South Africa. PLoS One 14, e0221495 (2019).
Dreyer, G. et al. The performance of single and combination test strategies using visual inspection, cytology, high-risk HPV DNA and HPV16/18 to screen South African women with and without HIV-infection. Infect. Agent Cancer 19, 22 (2024).
Hopkins, K. L. et al. Assessing national cervical cancer screening guidelines: results from an HIV testing clinic also screening for cervical cancer and HPV in Soweto, South Africa. PLoS One 16, e0255124 (2021).
Vink, F. J. et al. Validation of ASCL1 and LHX8 methylation analysis as primary cervical cancer screening strategy in South African women with human immunodeficiency virus. Clin. Infect. Dis. 76, 416–423 (2023).
Saidu, R. et al. Performance of xpert HPV on self-collected vaginal samples for cervical cancer screening among women in South Africa. J. Low. Genit. Trac. Dis. 25, 15–21 (2021).
Taku, O. et al. Distribution of Human Papillomavirus (HPV) genotypes in HIV-negative and HIV-positive women with cervical intraepithelial lesions in the Eastern Cape Province, South Africa. Viruses 13, 280 (2021).
Travill, D. I. et al. High prevalence of human papillomavirus (HPV) in unvaccinated adolescent girls in South Africa, particularly those living with HIV. Vaccine 42, 126442 (2024).
Mbulawa, Z. Z. A., Kondlo, S., Toni, S., Faye, L. M. & Businge, C. B. Prevalence, characteristics, and distribution of human papillomavirus according to age and HIV status in women of Eastern Cape Province, South Africa. Viruses 16, 1751 (2024).
Baisley, K. J. et al. HPV prevalence around the time of sexual debut in adolescent girls in Tanzania. Sex. Transm. Infect. 96, 211–219 (2020).
McHaro, R. et al. HPV type distribution in HIV positive and negative women with or without cervical dysplasia or cancer in East Africa. Front Oncol. 11, 763717 (2021).
McHome, B. L. et al. HPV types, cervical high-grade lesions and risk factors for oncogenic human papillomavirus infection among 3416 Tanzanian women. Sex. Transm. Infect. 97, 56–62 (2021).
Ardhaoui, M. et al. The prevalence, genotype distribution and risk factors of human papillomavirus in Tunisia: a national-based study. Viruses 14, 2175 (2022).
Nakigozi, H., Ategyeka, P. M., Nabadda, S. & Ndejjo, R. Positivity rates, trends and experiences of health workers on human papillomavirus screened using genexpert in Uganda: a three-year retrospective cohort study. BMC Cancer 24, 375 (2024).
Nakigozi, H. et al. Prevalence of genital high-risk human papillomavirus infections and associated factors among women living with human immunodeficiency virus in Uganda. BMC Cancer 24, 243 (2024).
Muchaili, L. et al. Prevalence and correlates of human papillomavirus infection in females from Southern Province, Zambia: a cross-sectional study. PLoS One 19, e0299963 (2024).
Kuguyo, O. et al. High-risk HPV genotypes in Zimbabwean women with cervical cancer: Comparative analyses between HIV-negative and HIV-positive women. PLoS One 16, e0257324 (2021).
Thistle, P. et al. Prevalence and subtype distribution of high-risk human papillomavirus among women presenting for cervical cancer screening at Karanda Mission Hospital. JCO Glob. Oncol. 6, 1276–1281 (2020).
Onwuamah, C. K. et al. Prevalence and risk factors for high-risk human papillomavirus infection among women from three southern geopolitical zones of Nigeria. Front Oncol. 13, 1254304 (2023).
Taku, O. et al. Human papillomavirus prevalence and risk factors among HIV-negative and HIV-positive women residing in rural Eastern Cape, South Africa. Int J. Infect. Dis. 95, 176–182 (2020).
Ibrahim Khalil, A. et al. Age-specific burden of cervical cancer associated with HIV: A global analysis with a focus on sub-Saharan Africa. Int J. Cancer 150, 761–772 (2022).
Chibwesha, C. J. & Stringer, J. S. A. Cervical cancer as a global concern: contributions of the dual epidemics of HPV and HIV. Jama 322, 1558–1560 (2019).
Okoye, J. O., Ofodile, C. A., Adeleke, O. K. & Obioma, O. Prevalence of high-risk HPV genotypes in sub-Saharan Africa according to HIV status: a 20-year systematic review. Epidemiol. Health 43, e2021039 (2021).
Akakpo, P. K. et al. High-risk human papillomavirus genotype distribution among women living with HIV; implication for cervical cancer prevention in a resource limited setting. Infect. Agent Cancer 18, 33 (2023).
Tchouaket, M. C. T. et al. Variability of high-risk human papillomavirus and associated factors among women in sub-Saharan Africa: a systematic review and meta-analysis. Pathogens 12, 1032 (2023).
Denny, L. et al. Human papillomavirus prevalence and type distribution in invasive cervical cancer in sub-Saharan Africa. Int J. Cancer 134, 1389–1398 (2014).
Gilles, C. et al. HPV genotyping in biopsies of HSIL and invasive cervical cancers in women living with HIV: a cohort- and a nested -case control study. Vaccine 40, 7230–7237 (2022).
Das, M. WHO launches strategy to accelerate elimination of cervical cancer. Lancet Oncol. 22, 20–21 (2021).
Boily, M. C. et al. Estimating the effect of HIV on cervical cancer elimination in South Africa: Comparative modelling of the impact of vaccination and screening. EClinicalMedicine 54, 101754 (2022).
WHO-AFRO. African Health Ministers, Partners Urge Equitable Access To Critical Tools To Curb Cervical Cancer Threat., https://www.afro.who.int/news/african-health-ministers-partners-urge-equitable-access-critical-tools-curb-cervical-cancer. (2024).
WHO-AFRO. in Regional Immunisation Technical Advisory Group (RITAG) meeting report 21–22 (Brazzavile, 2023).
Okolie, E. A. & Nwadike, B. I. Spotlight on human papillomavirus vaccination coverage: is Nigeria making any progress?. JCO Glob. Oncol. 9, e2300088 (2023).
WHO. Introduction of HPV (Human Papilloma Virus) vaccine. 2024, https://immunizationdata.who.int/global/wiise-detail-page/introduction-of-hpv-(2025).
WHO. Human Papillomavirus (HPV) vaccination coverage. 2024, https://immunizationdata.who.int/global/wiise-detail-page/human-papillomavirus-(2025).
GAVI. Vaccine Support Type Data. 2025, <https://www.gavi.org/programmes-impact/country-hub> (Accessed 10 March 2025).
Malagón, T., Franco, E. L., Tejada, R. & Vaccarella, S. Epidemiology of HPV-associated cancers past, present and future: towards prevention and elimination. Nat. Rev. Clin. Oncol. 21, 522–538 (2024).
Rahangdale, L., Mungo, C., O’Connor, S., Chibwesha, C. J. & Brewer, N. T. Human papillomavirus vaccination and cervical cancer risk. Bmj 379, e070115 (2022).
Casey, R. M. et al. National introduction of HPV vaccination in Senegal-Successes, challenges, and lessons learned. Vaccine 40, A10–a16 (2022).
Ndiaye, C. et al. Integrating HPV vaccine service delivery with adolescent health programmes - experiences and perspectives from selected countries in Africa. Vaccine 42, S45–s48 (2024).
Rosen, J. G. et al. Feasibility and sustainability of a school-based platform for integrated delivery of HPV vaccination with adolescent health services in Tanzania: qualitative insights from stakeholders. Health Policy Plan 38, 486–495 (2023).
Simuyemba, M. C. et al. An evaluation of the cost of human papilloma virus (HPV) vaccine delivery in Zambia. BMC Infect. Dis. 24, 369 (2024).
Dreyer, G. et al. Combining cervical cancer screening for mothers with schoolgirl vaccination during human papillomavirus (HPV) vaccine implementation in South Africa: results from the VACCS1 and VACCS2 trials. Int J. Gynecol. Cancer 32, 592–598 (2022).
Kouassi, K. S. et al. Pandemic-related resilience in HPV vaccination programmes - Perspectives from selected countries in Africa on what it will take to vaccinate 90 % of girls by 2030. Vaccine 42, S33–s36 (2024).
Asangbeh-Kerman, S. L. et al. Cervical cancer prevention and care in HIV clinics across sub-Saharan Africa: results of a facility-based survey. J. Int AIDS Soc. 27, e26303 (2024).
Muñoz, N. et al. Safety, immunogenicity, and efficacy of quadrivalent human papillomavirus (types 6, 11, 16, 18) recombinant vaccine in women aged 24-45 years: a randomised, double-blind trial. Lancet 373, 1949–1957 (2009).
Sharma, H. et al. Immunogenicity and safety of a new quadrivalent HPV vaccine in girls and boys aged 9-14 years versus an established quadrivalent HPV vaccine in women aged 15-26 years in India: a randomised, active-controlled, multicentre, phase 2/3 trial. Lancet Oncol. 24, 1321–1333 (2023).
Sayinzoga, F. et al. Human papillomavirus vaccine effect against human papillomavirus infection in Rwanda: evidence from repeated cross-sectional cervical-cell-based surveys. Lancet Glob. Health 11, e1096–e1104 (2023).
Baussano, I. et al. Impact of human papillomavirus vaccination, Rwanda and Bhutan. Emerg. Infect. Dis. 27, 1–9 (2021).
Dorji, T., Nopsopon, T., Tamang, S. T. & Pongpirul, K. Human papillomavirus vaccination uptake in low-and middle-income countries: a meta-analysis. EClinicalMedicine 34, 100836 (2021).
Mphuru, A. et al. National introduction of human papillomavirus (HPV) vaccine in Tanzania: Programmatic decision-making and implementation. Vaccine 40, A2–a9 (2022).
LaMontagne, D. S. et al. Progress in HPV vaccination in low- and lower-middle-income countries. Int J. Gynaecol. Obstet. 138, 7–14 (2017).
Nabirye, J. et al. Health system factors influencing uptake of Human Papillomavirus (HPV) vaccine among adolescent girls 9-15 years in Mbale District, Uganda. BMC Public Health 20, 171 (2020).
El Mansouri, N. et al. Awareness and knowledge associated to Human papillomavirus infection among university students in Morocco: A cross-sectional study. PLoS One 17, e0271222 (2022).
Bencherit, D. et al. Knowledge and awareness of algerian students about cervical cancer, HPV and HPV vaccines: a cross-sectional study. Vaccines (Basel) https://doi.org/10.3390/vaccines10091420 (2022).
Haddison, E., Tambasho, A., Kouamen, G. & Ngwafor, R. Vaccinators’ perception of HPV vaccination in the Saa health district of Cameroon. Front Public Health 9, 748910 (2021).
Oduwole, E. O., Mahomed, H., Laurenzi, C. A., Larson, H. J. & Wiysonge, C. S. Point-of-care vaccinators’ perceptions of vaccine hesitancy drivers: a qualitative study from the Cape Metropolitan District, South Africa. Vaccine 39, 5506–5512 (2021).
Okunade, K. S. et al. Impact of mobile health technologies on human papillomavirus vaccination uptake among mothers of unvaccinated girls aged 9-14 years in Lagos, Nigeria (mHealth-HPVac): study protocol of a randomised controlled trial. BMC Cancer 24, 751 (2024).
Fokom Domgue, J. et al. HPV vaccination in Africa in the COVID-19 era: a cross-sectional survey of healthcare providers’ knowledge, training, and recommendation practices. Front Public Health 12, 1343064 (2024).
Sharma, K. et al. No woman left behind: achieving cervical cancer elimination among women living with HIV. Lancet HIV 10, e412–e420 (2023).
Barnabas, R. V. et al. Durability of single-dose HPV vaccination in young Kenyan women: randomized controlled trial 3-year results. Nat. Med 29, 3224–3232 (2023).
Baandrup, L., Dehlendorff, C. & Kjaer, S. K. One-dose human papillomavirus vaccination and the risk of genital warts: a danish nationwide population-based study. Clin. Infect. Dis. 73, e3220–e3226 (2021).
Bénard, É et al. Potential population-level effectiveness of one-dose HPV vaccination in low-income and middle-income countries: a mathematical modelling analysis. Lancet Public Health 8, e788–e799 (2023).
Watson-Jones, D. et al. Immunogenicity and safety of one-dose human papillomavirus vaccine compared with two or three doses in Tanzanian girls (DoRIS): an open-label, randomised, non-inferiority trial. Lancet Glob. Health 10, e1473–e1484 (2022).
Baisley, K. et al. Comparing one dose of HPV vaccine in girls aged 9-14 years in Tanzania (DoRIS) with one dose of HPV vaccine in historical cohorts: an immunobridging analysis of a randomised controlled trial. Lancet Glob. Health 10, e1485–e1493 (2022).
Baisley, K. et al. Comparing one dose of HPV vaccine in girls aged 9-14 years in Tanzania (DoRIS) with one dose in young women aged 15-20 years in Kenya (KEN SHE): an immunobridging analysis of randomised controlled trials. Lancet Glob. Health 12, e491–e499 (2024).
Delany-Moretlwe, S. et al. Impact of single-dose HPV vaccination on HPV 16 and 18 prevalence in South African adolescent girls with and without HIV. J. Natl. Cancer Inst. Monogr. 2024, 337–345 (2024).
Bénard, É et al. Potential benefit of extended dose schedules of human papillomavirus vaccination in the context of scarce resources and COVID-19 disruptions in low-income and middle-income countries: a mathematical modelling analysis. Lancet Glob. Health 11, e48–e58 (2023).
Sabahelzain, M. M., Dwyer, H., Abimbola, S. & Leask, J. Implications of conflict on vaccination in the Sahel region. BMJ Glob Health 10, e016496 (2025).
Sonawane, K. et al. Human papillomavirus vaccination among young adults before and during the COVID-19 pandemic. JAMA Netw. Open 7, e2356875 (2024).
Kutz, J. M., Rausche, P., Gheit, T., Puradiredja, D. I. & Fusco, D. Barriers and facilitators of HPV vaccination in sub-saharan Africa: a systematic review. BMC Public Health 23, 974 (2023).
Burd, E. M. Human papillomavirus and cervical cancer. Clin. Microbiol Rev. 16, 1–17 (2003).
Fontham, E. T. H. et al. Cervical cancer screening for individuals at average risk: 2020 guideline update from the American Cancer Society. CA Cancer J. Clin. 70, 321–346 (2020).
Bouvard, V. et al. The IARC perspective on cervical cancer screening. N. Engl. J. Med 385, 1908–1918 (2021).
WHO. in WHO Guideline For Screening And Treatment Of Cervical Pre-cancer Lesions For Cervical Cancer Prevention, second edition 24–25 (Geneva, 2021).
Stoler, M. H., Parvu, V., Yanson, K., Andrews, J. & Vaughan, L. Risk stratification of HPV-positive results using extended genotyping and cytology: Data from the baseline phase of the Onclarity trial. Gynecol. Oncol. 174, 68–75 (2023).
WHO. in WHO Guideline For Screening And Treatment Of Cervical Pre-cancer Lesions For Cervical Cancer Prevention, second edition 3 (Geneva, 2021).
Salcedo, M. P. et al. The Mulher Study: cervical cancer screening with primary HPV testing in Mozambique. Int J. Gynecol. Cancer 33, 1869–1874 (2023).
Dzinamarira, T., Moyo, E., Dzobo, M., Mbunge, E. & Murewanhema, G. Cervical cancer in sub-Saharan Africa: an urgent call for improving accessibility and use of preventive services. Int J. Gynecol. Cancer 33, 592–597 (2023).
Pry, J. M. et al. Cervical cancer screening outcomes in Zambia, 2010-19: a cohort study. Lancet Glob. Health 9, e832–e840 (2021).
Fokom Domgue, J. et al. Feasibility of a community-based cervical cancer screening with “test and treat” strategy using self-sample for an HPV test: experience from rural Cameroon, Africa. Int J. Cancer 147, 128–138 (2020).
Asgary, R. et al. Evaluating smartphone strategies for reliability, reproducibility, and quality of VIA for cervical cancer screening in the Shiselweni region of Eswatini: a cohort study. PLoS Med 17, e1003378 (2020).
Kabukye, J. K. et al. Implementing smartphone-based telemedicine for cervical cancer screening in Uganda: qualitative study of stakeholders’ perceptions. J. Med Internet Res 25, e45132 (2023).
Jonnalagedda-Cattin, M. et al. Barriers and facilitators to the preadoption of a computer-aided diagnosis tool for cervical cancer: qualitative study on health care providers’ perspectives in Western Cameroon. JMIR Cancer 11, e50124 (2025).
Champlin, C., Bell, D. & Schocken, C. AI medicine comes to Africa’s rural clinics. IEEE Spectr. 54, 42–48 (2017).
Xue, Z. et al. A demonstration of automated visual evaluation of cervical images taken with a smartphone camera. Int J. Cancer 147, 2416–2423 (2020).
Hu, L. et al. Internal validation of automated visual evaluation (AVE) on smartphone images for cervical cancer screening in a prospective study in Zambia. Cancer Med 13, e7355 (2024).
Barrett, C. et al. Performance of an artificial intelligence-based tool for cervical precancer screening in five countries in Africa: a prospective, observational, diagnostic accuracy study. Lancet Glob. Health 14, e49–e60 (2026).
Holmström, O. et al. Point-of-care digital cytology with artificial intelligence for cervical cancer screening in a resource-limited setting. JAMA Netw. Open 4, e211740 (2021).
Kuhn, L. et al. Clinical evaluation of modifications to a human papillomavirus assay to optimise its utility for cervical cancer screening in low-resource settings: a diagnostic accuracy study. Lancet Glob. Health 8, e296–e304 (2020).
Steenbergen, R. D., Snijders, P. J., Heideman, D. A. & Meijer, C. J. Clinical implications of (epi)genetic changes in HPV-induced cervical precancerous lesions. Nat. Rev. Cancer 14, 395–405 (2014).
Schreiberhuber, L. et al. Cervical cancer screening using DNA methylation triage in a real-world population. Nat. Med 30, 2251–2257 (2024).
Kelly, H. et al. Performance of DNA methylation assays for detection of high-grade cervical intraepithelial neoplasia (CIN2+): a systematic review and meta-analysis. Br. J. Cancer 121, 954–965 (2019).
Kremer, W. W., Steenbergen, R., Heideman, D., Kenter, G. G. & Meijer, C. The use of host cell DNA methylation analysis in the detection and management of women with advanced cervical intraepithelial neoplasia: a review. Bjog 128, 504–514 (2021).
Xu, W. et al. Integrative analysis of DNA methylation and gene expression identified cervical cancer-specific diagnostic biomarkers. Signal Transduct. Target Ther. 4, 55 (2019).
Kremer, W. W. et al. Clinical regression of high-grade cervical intraepithelial neoplasia is associated with absence of FAM19A4/miR124-2 DNA methylation (CONCERVE Study). J. Clin. Oncol. 40, 3037–3046 (2022).
van den Helder, R. et al. HPV and DNA Methylation Testing in Urine for Cervical Intraepithelial Neoplasia and Cervical Cancer Detection. Clin. Cancer Res 28, 2061–2068 (2022).
Verlaat, W. et al. Identification and validation of a 3-gene methylation classifier for HPV-based cervical screening on self-samples. Clin. Cancer Res 24, 3456–3464 (2018).
Vink, F. J. et al. Posttreatment monitoring by ASCL1/LHX8 methylation analysis in women with HIV treated for cervical intraepithelial neoplasia grade 2/3. Aids 36, 953–961 (2022).
Lince-Deroche, N., Phiri, J., Michelow, P., Smith, J. S. & Firnhaber, C. Costs and cost effectiveness of three approaches for cervical cancer screening among HIV-Positive Women in Johannesburg, South Africa. PLoS One 10, e0141969 (2015).
Sefuthi, T. & Nkonki, L. A systematic review of economic evaluations of cervical cancer screening methods. Syst. Rev. 11, 162 (2022).
Tran, J. et al. Cost-effectiveness of single-visit cervical cancer screening in KwaZulu-Natal, South Africa: a model-based analysis accounting for the HIV epidemic. Front Oncol. 14, 1382599 (2024).
Joseph, J. et al. Human papillomavirus testing using existing nucleic acid testing platforms to screen women for cervical cancer: implementation studies from five sub-Saharan African countries. BMJ Open 13, e065074 (2023).
Zimmermann, M. R. et al. Cost-effectiveness of cervical cancer screening and preventative cryotherapy at an HIV treatment clinic in Kenya. Cost. Eff. Resour. Alloc. 15, 13 (2017).
Kahesa, C. et al. Comparison of human papillomavirus-based cervical cancer screening strategies in Tanzania among women with and without HIV. Int J. Cancer 152, 686–696 (2023).
Arechkik, A., Lahlou, L., Obtel, M., Kharbach, A. & Razine, R. Cervical cancer in Morocco: a systematic review. Rev. Epidemiol. Sante Publique 70, 230–242 (2022).
Plaisy, M. K. et al. Barriers to early diagnosis of cervical cancer: a mixed-method study in Côte d’Ivoire, West Africa. BMC Women’s. Health 23, 135 (2023).
Zewdie, A. et al. Determinants of late-stage cervical cancer presentation in Ethiopia: a systematic review and meta-analysis. BMC Cancer 23, 1228 (2023).
Mwenda, V. et al. Factors associated with late diagnosis of cervical cancer at two national referral hospitals, Kenya 2017: A case control study. Gynecol. Oncol. Rep. 52, 101355 (2024).
Anakwenze, C. et al. Cervical cancer stage distribution and survival outcomes in Africa: a systematic review and meta-analysis. Int J. Gynecol. Cancer 35, 100008 (2025).
Téguété, I. et al. A population-based combination strategy to improve the cervical cancer screening coverage rate in Bamako, Mali. Acta Obstet. Gynecol. Scand. 100, 794–801 (2021).
Hailegebireal, A. H., Bizuayehu, H. M. & Tirore, L. L. Far behind 90-70-90’s screening target: the prevalence and determinants of cervical cancer screening among Sub-Saharan African women: evidence from Demographic and Health Survey. BMC Cancer 24, 1050 (2024).
Yang, L. et al. Regional and country-level trends in cervical cancer screening coverage in sub-Saharan Africa: A systematic analysis of population-based surveys (2000-2020). PLoS Med 20, e1004143 (2023).
Ahmed, H. A. A. et al. Cervical cancer screening uptake in Arab countries: a systematic review with meta-analysis. BMC Cancer 24, 1438 (2024).
Ortashi, O. & Alkalbani, M. in Handbook of Healthcare in the Arab World (ed Ismail Laher) 413-433 (Springer International Publishing, 2021).
Liang, J., Wolsiefer, K., Zestcott, C. A., Chase, D. & Stone, J. Implicit bias toward cervical cancer: Provider and training differences. Gynecol. Oncol. 153, 80–86 (2019).
Gök, M. et al. HPV testing on self collected cervicovaginal lavage specimens as screening method for women who do not attend cervical screening: cohort study. Bmj 340, c1040 (2010).
Yeh, P. T., Kennedy, C. E., de Vuyst, H. & Narasimhan, M. Self-sampling for human papillomavirus (HPV) testing: a systematic review and meta-analysis. BMJ Glob. Health 4, e001351 (2019).
Costa, S., Verberckmoes, B., Castle, P. E. & Arbyn, M. Offering HPV self-sampling kits: an updated meta-analysis of the effectiveness of strategies to increase participation in cervical cancer screening. Br. J. Cancer 128, 805–813 (2023).
Asare, M., Abah, E., Obiri-Yeboah, D., Lowenstein, L. & Lanning, B. HPV self-sampling for cervical cancer screening among women living with HIV in low- and middle-income countries: what do we know and what can be done? Healthcare (Basel) 10, 1270 (2022).
Effah, K. et al. Self-sampling and HPV DNA testing for cervical precancer screening in a cohort of nuns in Ghana: a cross-sectional cohort study. EClinicalMedicine 63, 102183 (2023).
Gibert, M. J., Sánchez-Contador, C. & Artigues, G. Validity and acceptance of self vs conventional sampling for the analysis of human papillomavirus and Pap smear. Sci. Rep. 13, 2809 (2023).
Daponte, N. et al. HPV-based self-sampling in cervical cancer screening: an updated review of the current evidence in the literature. Cancers (Basel) 15, 1669 (2023).
Polman, N. J. et al. Performance of human papillomavirus testing on self-collected versus clinician-collected samples for the detection of cervical intraepithelial neoplasia of grade 2 or worse: a randomised, paired screen-positive, non-inferiority trial. Lancet Oncol. 20, 229–238 (2019).
Cooper, E. C. et al. Implementation of human papillomavirus video education for women participating in mass cervical cancer screening in Tanzania. Am. J. Obstet. Gynecol. 224, 105.e101–105.e109 (2021).
Alfaro, K., Maza, M., Cremer, M., Masch, R. & Soler, M. Removing global barriers to cervical cancer prevention and moving towards elimination. Nat. Rev. Cancer 21, 607–608 (2021).
Lakew, G. et al. Cervical cancer screening practice and associated factors among female health care professionals in Ethiopia 2024: a systematic review and meta-analysis. BMC Cancer 24, 986 (2024).
WHO. in Comprehensive Cervical Cancer Control: A Guide To Essential Practice – 2nd ed 93-104 (2014).
Raffoul, M., Bartlett-Esquilant, G. & Phillips, R. L. Jr. Recruiting and training a health professions workforce to meet the needs of tomorrow’s health care system. Acad. Med 94, 651–655 (2019).
Subramanian, S. et al. Acceptability, utility, and cost of a mobile health cancer screening education application for training primary care physicians in India. Oncologist 26, e2192–e2199 (2021).
Thomas, M. P. et al. Development of a simple and effective online training for health workers: results from a pilot in Nigeria. BMC Public Health 22, 551 (2022).
Fokom Domgue, J. et al. Development, implementation, and evaluation of a distance learning and telementoring program for cervical cancer prevention in Cameroon. JAMA Netw. Open 5, e2240801 (2022).
Cancedda, C. et al. Maximizing the impact of training initiatives for health professionals in low-income countries: frameworks, challenges, and best practices. PLoS Med 12, e1001840 (2015).
Johnson, L. G., Armstrong, A., Joyce, C. M., Teitelman, A. M. & Buttenheim, A. M. Implementation strategies to improve cervical cancer prevention in sub-Saharan Africa: a systematic review. Implement Sci. 13, 28 (2018).
Levin, A. et al. Comprehensive approach to costing cervical cancer prevention and control: a case study in the united republic of tanzania using the cervical cancer prevention and control costing (C4P) tool. BMC Med 20, 384 (2022).
WHO. in Comprehensive cervical cancer control: a guide to essential practice – 2nd ed 154–159 (2014).
Shin, M. B. et al. Implementation and scale-up of a single-visit, screen-and-treat approach with thermal ablation for sustainable cervical cancer prevention services: a protocol for a stepped-wedge cluster randomized trial in Kenya. Implement Sci. 18, 26 (2023).
Batman, S. et al. Expanding Cervical Cancer Screening in Mozambique: Challenges Associated With Diagnosing and Treating Cervical Cancer. JCO Glob. Oncol. 9, e2300139 (2023).
WHO. in WHO Technical Guidance And Specifications Of Medical Devices For Screening And Treatment Of Precancerous Lesions In The Prevention Of Cervical Cancer 68 (Geneva, 2020).
Mungo, C. et al. Safety and acceptability of thermal ablation for treatment of human papillomavirus among women living with HIV in Western Kenya. JCO Glob. Oncol. 6, 1024–1033 (2020).
Metaxas, T. et al. Acceptability and safety of thermal ablation to prevent cervical cancer in sub-Saharan Africa. BMC Cancer 22, 132 (2022).
Sauvaget, C. et al. Evaluation of effectiveness, acceptability and safety of thermal ablation in the treatment of cervical neoplasia in Burundi. Int J. Cancer 151, 1120–1126 (2022).
Mungo, C., Osongo, C. O., Ambaka, J., Omoto, J. & Cohen, C. R. Efficacy of thermal ablation for treatment of biopsy-confirmed high-grade cervical precancer among women living with HIV in Kenya. Int J. Cancer 153, 1971–1977 (2023).
Sebitloane, H. M. et al. Cervical cancer screening and treatment algorithms using human papillomavirus testing-lessons learnt from a south african pilot randomized controlled trial. Cancer Epidemiol. Biomark. Prev. 33, 779–787 (2024).
Chinula, L. et al. Uptake and safety of community-based “screen-and-treat” with thermal ablation preventive therapy for cervical cancer prevention in rural Lilongwe, Malawi. Int J. Cancer 149, 371–377 (2021).
Gottschlich, A. et al. Experiences with thermal ablation for cervical precancer treatment after self-collection HPV-based screening in the ASPIRE Mayuge randomized trial. Int J. Cancer 152, 1630–1639 (2023).
DeBoer, R. J. et al. Cervical cancer treatment in Rwanda: resource-driven adaptations, quality indicators, and patient outcomes. Gynecol. Oncol. 164, 370–378 (2022).
Vulpe, H. et al. External beam radiation therapy and brachytherapy for cervical cancer: the experience of the national centre for radiotherapy in Accra, Ghana. Int J. Radiat. Oncol. Biol. Phys. 100, 1246–1253 (2018).
Chibonda, S. et al. Working title: high dose rate intra-cavitary brachytherapy with cobalt 60 source for locally advanced cervical cancer: the Zimbabwean experience. Infect. Agent Cancer 16, 1 (2021).
Rubagumya, F. et al. Financial toxicity: unveiling the burden of cancer care on patients in Rwanda. Oncologist 29, e345–e350 (2024).
Beltrán Ponce, S. E. et al. Access to radiation therapy and related clinical outcomes in patients with cervical and breast cancer across sub-Saharan Africa: a systematic review. JCO Glob. Oncol. 9, e2200218 (2023).
Dereje, N. et al. Association between waiting time for radiotherapy initiation and disease progression among women with cervical cancer in Addis Ababa, Ethiopia. Int J. Cancer 149, 1284–1289 (2021).
Kuguyo, O. et al. Prevalence and predictors for cisplatin-induced toxicities in Zimbabwean women with cervical cancer. Future Oncol. 20, 1909–1924 (2024).
Paken, J., Govender, C. D., Pillay, M., Ayele, B. T. & Sewram, V. Feasibility and first results of a prospective cohort study to investigate cisplatin-associated ototoxicity amongst cancer patients in South Africa. BMC Cancer 21, 822 (2021).
Sengayi-Muchengeti, M. et al. Cervical cancer survival in sub-Saharan Africa by age, stage at diagnosis and Human Development Index: A population-based registry study. Int J. Cancer 147, 3037–3048 (2020).
Griesel, M. et al. Cervical cancer in Sub-Saharan Africa: a multinational population-based cohort study of care and guideline adherence. Oncologist 26, e807–e816 (2021).
Grover, S. et al. Chemoradiation versus radiation alone in stage IIIB cervical cancer patients with or without human immunodeficiency virus. Int J. Gynecol. Cancer 31, 1220–1227 (2021).
Kassa, R. et al. Survival of women with cervical cancer in East Africa: a systematic review and meta-analysis. J. Obstet. Gynaecol. 43, 2253308 (2023).
MacDuffie, E. et al. Five-year overall survival following chemoradiation therapy for locally advanced cervical carcinoma in women living with and without HIV infection in Botswana. Infect. Agent Cancer 16, 55 (2021).
Meghani, K. et al. Significance of HIV status in cervical cancer patients receiving curative chemoradiation therapy, definitive radiation alone, or palliative radiation in Botswana. Cancer 130, 2462–2471 (2024).
Acknowledgements
The authors express their profound gratitude to Wenyu Lin, Yuxuan Huang, Jingxuan Ye, Afia Boadu Poku, Andoh Sheikh A., Dingjie Wang, and Yashi Shi for their additions to this work. Also, thanks to Bamisope Steve Basimile (PhD) for his guidance in editing this work. The following grants support this work: 1. The National Natural Science Foundation of China (grant no. 82271658)(P.M.S). 2. The Major Scientific Research Program for Young and Middle-aged Health Professionals of Fujian Province, China (grant no. 2021ZQNZD011) (P.M.S). 3. National High-Level Hospital Clinical Research Funding under Grant 2022-PUMCH-C-060 (H.Y). 4. Beijing Natural Science Foundation (grant no. L232074) (H.Y).
Author information
Authors and Affiliations
Contributions
K.S.O.–conceptualization, drafting, tables, and figures, revision; Y.Z.–drafting, tables, and revision; D.O.Y.– revision; B.S.–revision; H.Y.–revision; P.M.S.–conceptualization and revision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Tafadzwa Dzinamarira who co-reviewed with Mathias Dzobo; Ritu Salani, Joël Fokom-Domgue, Isaac F. Adewole, Patrick Petignat, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Osafo, K.S., Zhang, Y., Obiri-Yeboah, D. et al. Advancing the elimination of cervical cancer across Africa. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70450-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70450-y