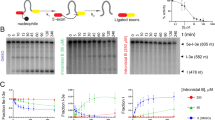
Abstract
Targeting RNA with small molecules offers a strategy to modulate gene expression at undruggable targets. Traditional screens favor thermodynamically stable, low-entropy RNA motifs with defined conformations, yet these provide limited energetic leverage for functional modulation. Many RNAs instead sample dynamic structural ensembles that small molecules can repartition. Using group I self-splicing introns as a model, we identified the antineoplastic drug Mitoxantrone as a competitive inhibitor of RNA self-splicing (IC50 = 4.3 μM) that stabilizes the native conformation of the T4 td intron. Structure-activity analysis showed that the anthraquinone scaffold alone is insufficient, and basic amine-containing side chains are required for RNA structural modulation. Transcriptome-wide chemical probing in human cells revealed preferential binding to GC-rich structured regions, although only a subset showed structural change. Furthermore, global analysis of 5′ UTR ensembles showed altered structural heterogeneity and translation, demonstrating functional repartitioning of RNA conformational landscapes.
Similar content being viewed by others
Data availability
The data supporting the findings of this study are available from the corresponding authors upon request. Sequencing data have been deposited to the Gene Expression Omnibus (GEO) database, under the accession GSE302505. Raw MM files for analysis with DRACO are available from Zenodo (https://doi.org/10.5281/zenodo.15874381). Additional processed data are available at https://www.incarnatolab.com/datasets/Mitoxantrone_Zhang_2026.php. Source data for the figures and Supplementary Figure are provided as a Source Data file. Source data are provided with this paper.
Code availability
The source codes of DRACO v1.3, and of the diffShape utility are freely available from GitHub, under the GPLv3 license (https://github.com/dincarnato/draco and https://github.com/dincarnato/papers).
References
Warner, K. D., Hajdin, C. E. & Weeks, K. M. Principles for targeting RNA with drug-like small molecules. Nat. Rev. Drug Discov. 17, 547–558 (2018).
Childs-Disney, J. L. et al. Targeting RNA structures with small molecules. Nat. Rev. Drug Discov. 21, 736–762 (2022).
Hewitt, W. M., Calabrese, D. R. & Schneekloth, J. S. Evidence for ligandable sites in structured RNA throughout the Protein Data Bank. Bioorg. Med. Chem. 27, 2253–2260 (2019).
Veenbaas, S. D., Koehn, J. T., Irving, P. S., Lama, N. N. & Weeks, K. M. Ligand-binding pockets in RNA and where to find them. Proc. Natl. Acad. Sci. USA 122, e2422346122 (2025).
Mustoe, A. M., Brooks, C. L. & Al-Hashimi, H. M. Hierarchy of RNA functional dynamics. Annu. Rev. Biochem. 83, 441–466 (2014).
Ganser, L. R., Kelly, M. L., Herschlag, D. & Al-Hashimi, H. M. The roles of structural dynamics in the cellular functions of RNAs. Nat. Rev. Mol. Cell Biol. 20, 474–489 (2019).
Spitale, R. C. & Incarnato, D. Probing the dynamic RNA structurome and its functions. Nat. Rev. Genet. 24, 178–196 (2023).
Bose, R., Saleem, I. & Mustoe, A. M. Causes, functions, and therapeutic possibilities of RNA secondary structure ensembles and alternative states. Cell Chem. Biol. 31, 17–35 (2024).
Bonilla, S. L., Jones, A. N. & Incarnato, D. Structural and biophysical dissection of RNA conformational ensembles. Curr. Opin. Struct. Biol. 88, 102908 (2024).
Siegfried, N. A., Busan, S., Rice, G. M., Nelson, J. A. E. & Weeks, K. M. RNA motif discovery by SHAPE and mutational profiling (SHAPE-MaP). Nat. Methods 11, 959–965 (2014).
Mustoe, A. M. et al. Pervasive regulatory functions of mRNA structure revealed by high-resolution SHAPEprobing. Cell 173, 181–195.e18 (2018).
Manfredonia, I. et al. Genome-wide mapping of SARS-CoV-2 RNA structures identifies therapeutically-relevant elements. Nucleic Acids Res. 48, 12436–12452 (2020).
Sztuba-Solinska, J., Chavez-Calvillo, G. & Cline, S. E. Unveiling the druggable RNA targets and small molecule therapeutics. Bioorg. Med. Chem. 27, 2149–2165 (2019).
Juru, A. U., Patwardhan, N. N. & Hargrove, A. E. Understanding the contributions of conformational changes, thermodynamics, and kinetics of RNA–small molecule interactions. ACS Chem. Biol. 14, 824–838 (2019).
Ottink, O. M. et al. Ligand-induced folding of the guanine-sensing riboswitch is controlled by a combined predetermined induced fit mechanism. RNA 13, 2202–2212 (2007).
Noeske, J. et al. Interplay of ‘induced fit’ and preorganization in the ligand induced folding of the aptamer domain of the guanine binding riboswitch. Nucleic Acids Res. 35, 572–583 (2007).
Vicens, Q., Mondragón, E. & Batey, R. T. Molecular sensing by the aptamer domain of the FMN riboswitch: a general model for ligand binding by conformational selection. Nucleic Acids Res. 39, 8586–8598 (2011).
Serganov, A. & Nudler, E. A decade of riboswitches. Cell 152, 17–24 (2013).
Kovachka, S. et al. Covalent probes reveal small-molecule binding pockets in structured RNA and enable bioactive compound design. J. Am. Chem. Soc. 147, 37460–37479 (2025).
Ratni, H. et al. Discovery of risdiplam, a selective survival of motor neuron-2 (SMN2) gene splicing modifier for the treatment of spinal muscular atrophy (SMA). J. Med. Chem. 61, 6501–6517 (2018).
Ishigami, Y. et al. Specificity, synergy, and mechanisms of splice-modifying drugs. Nat. Commun. 15, 1880 (2024).
Campagne, S. et al. Structural basis of a small molecule targeting RNA for a specific splicing correction. Nat. Chem. Biol. 15, 1191–1198 (2019).
Malard, F. et al. The diversity of splicing modifiers acting on A-1 bulged 5’-splice sites reveals rules for rational drug design. Nucleic Acids Res. 52, 4124–4136 (2024).
Childs-Disney, J. L., Wu, M., Pushechnikov, A., Aminova, O. & Disney, M. D. A small molecule microarray platform to select RNA internal loop-ligand interactions. ACS Chem. Biol. 2, 745–754 (2007).
Tran, T. & Disney, M. D. Two-dimensional combinatorial screening (2DCS) of a bacterial rRNA A-site-like motif library: defining privileged asymmetric internal loops that bind aminoglycosides. Biochemistry 49, 1833–1842 (2010).
Balaratnam, S. et al. Investigating the NRAS 5′ UTR as a target for small molecules. Cell Chem. Biol. 30, 643–657.e8 (2023).
Sidharthan, V. et al. Use of a small molecule microarray screen to identify inhibitors of the catalytic RNA subunit of Methanobrevibacter smithii RNase P. Nucleic Acids Res. 53, gkae1190 (2025).
Wicks, S. L. & Hargrove, A. E. Fluorescent indicator displacement assays to identify and characterize small molecule interactions with RNA. Methods 167, 3–14 (2019).
Davila-Calderon, J. et al. IRES-targeting small molecule inhibits enterovirus 71 replication via allosteric stabilization of a ternary complex. Nat. Commun. 11, 4775 (2020).
Binas, O. et al. 19 F NMR-based fragment screening for 14 different biologically active RNAs and 10 DNA and protein counter-screens. Chembiochem 22, 423–433 (2021).
Lundquist, K. P. et al. Design, synthesis, and screening of an RNA optimized fluorinated fragment library. SLAS Discov. 31, 100215 (2025).
Zeller, M. J. et al. SHAPE-enabled fragment-based ligand discovery for RNA. Proc. Natl. Acad. Sci. 119, e2122660119 (2022).
Rizvi, N. F. et al. Discovery of selective RNA-binding small molecules by affinity-selection mass spectrometry. ACS Chem. Biol. 13, 820–831 (2018).
Rizvi, N. F. et al. Targeting RNA with small molecules: identification of selective, RNA-binding small molecules occupying drug-like chemical space. SLAS Discov. 25, 384–396 (2020).
Nickbarg, E. B., Spencer, K. B., Mortison, J. D. & Lee, J. T. Targeting RNA with small molecules: lessons learned from Xist RNA. RNA 29, 463–472 (2023).
Childs-Disney, J. L. et al. A massively parallel selection of small molecule-RNA motif binding partners informs design of an antiviral from sequence. Chem 4, 2384–2404 (2018).
Hargrove, A. E. Small molecule-RNA targeting: starting with the fundamentals. Chem. Commun. 56, 14744–14756 (2020).
Donlic, A. et al. R-BIND 2.0: an updated database of bioactive RNA-targeting small molecules and associated RNA secondary structures. ACS Chem. Biol. 17, 1556–1566 (2022).
Yazdani, K. et al. Machine learning informs RNA-binding chemical space. Angew. Chem. Int. Ed. Engl. 62, e202211358 (2023).
Mortison, J. D. et al. Tetracyclines modify translation by targeting key human rRNA substructures. Cell Chem. Biol. 25, 1506–1518.e13 (2018).
Velagapudi, S. P., Li, Y. & Disney, M. D. A cross-linking approach to map small molecule-RNA binding sites in cells. Bioorg. Med, Chem. Lett. 29, 1532–1536 (2019).
Mukherjee, H. et al. PEARL-seq: a photoaffinity platform for the analysis of small molecule-RNA interactions. ACS Chem. Biol. 15, 2374–2381 (2020).
Balaratnam, S. et al. A chemical probe based on the PreQ1 metabolite enables transcriptome-wide mapping of binding sites. Nat. Commun. 12, 5856 (2021).
Shah, R. et al. Photoaffinity enabled transcriptome-wide identification of splice modulating small molecule–RNA binding events in native cells. RSC Chem. Biol. 6, 905–918 (2025).
Yang, X. et al. Mapping small molecule-RNA binding sites via Chem-CLIP synergized with capillary electrophoresis and nanopore sequencing. Nucleic Acids Res. 53, gkaf231 (2025).
Fang, L. et al. Pervasive transcriptome interactions of protein-targeted drugs. Nat. Chem. 15, 1374–1383 (2023).
Yesley, P., Poulladofonou, G., Incarnato, D. & Velema, W. A. Site-selective ligand selection by mutational profiling for covalent RNA targeting. Angew. Chem. Int Ed. Engl. 65, e17243 (2026).
Sengupta, A., Rice, G. M. & Weeks, K. M. Single-molecule correlated chemical probing reveals large-scale structural communication in the ribosome and the mechanism of the antibiotic spectinomycin in living cells. PLoS Biol. 17, e3000393 (2019).
Wang, Y., Parmar, S., Schneekloth, J. S. & Tiwary, P. Interrogating RNA-small molecule interactions with structure probing and artificial intelligence-augmented molecular simulations. ACS Cent. Sci. 8, 741–748 (2022).
Tomezsko, P. J. et al. Determination of RNA structural diversity and its role in HIV-1 RNA splicing. Nature 582, 438–442 (2020).
Morandi, E. et al. Genome-scale deconvolution of RNA structure ensembles. Nat. Methods 18, 249–252 (2021).
Olson, S. W. et al. Discovery of a large-scale, cell-state-responsive allosteric switch in the 7SK RNA using DANCE-MaP. Mol. Cell 82, 1708–1723.e10 (2022).
Yang, M. et al. In vivo single-molecule analysis reveals COOLAIR RNA structural diversity. Nature 609, 373–379 (2022).
Faulds, D., Balfour, J. A., Chrisp, P. & Langtry, H. D. Mitoxantrone. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in the chemotherapy of cancer. Drugs 41, 400–449 (1991).
Chu, F. K., Maley, G. F., Maley, F. & Belfort, M. Intervening sequence in the thymidylate synthase gene of bacteriophage T4. Proc. Natl. Acad. Sci. USA 81, 3049–3053 (1984).
Gott, J. M., Shub, D. A. & Belfort, M. Multiple self-splicing introns in bacteriophage T4: evidence from autocatalytic GTP labeling of RNA in vitro. Cell 47, 81–87 (1986).
Cech, T. R., Damberger, S. H. & Gutell, R. R. Representation of the secondary and tertiary structure of group I introns. Nat. Struct. Biol. 1, 273–280 (1994).
Janes, J. et al. The ReFRAME library as a comprehensive drug repurposing library and its application to the treatment of cryptosporidiosis. Proc. Natl. Acad. Sci. USA 115, 10750–10755 (2018).
Shortridge, M. D., Vidalala, V. & Varani, G. The kinase inhibitor palbociclib is a potent and specific RNA-binding molecule. Preprint at https://doi.org/10.1101/2022.01.20.477126 (2022).
Meyer, S. M. et al. Optimization of a protein-targeted medicine into an RNA-specific small molecule. ACS Chem. Biol. 18, 2336–2342 (2023).
von Ahsen, U., Davies, J. & Schroeder, R. Antibiotic inhibition of group I ribozyme function. Nature 353, 368–370 (1991).
von Ahsen, U., Davies, J. & Schroeder, R. Non-competitive inhibition of group I intron RNA self-splicing by aminoglycoside antibiotics. J. Mol. Biol. 226, 935–941 (1992).
von Ahsen, U. & Noller, H. F. Footprinting the sites of interaction of antibiotics with catalytic group I intron RNA. Science 260, 1500–1503 (1993).
Hoch, I., Berens, C., Westhof, E. & Schroeder, R. Antibiotic inhibition of RNA catalysis: neomycin B binds to the catalytic core of the td group I intron displacing essential metal ions. J. Mol. Biol. 282, 557–569 (1998).
Mercure, S., Montplaisir, S. & Lemay, G. Correlation between the presence of a self-splicing intron in the 25S rDNA of C.albicans and strains susceptibility to 5-fluorocytosine. Nucleic Acids Res. 21, 6020–6027 (1993).
Bass, B. L. & Cech, T. R. Ribozyme inhibitors: deoxyguanosine and dideoxyguanosine are competitive inhibitors of self-splicing of the Tetrahymena ribosomal ribonucleic acid precursor. Biochemistry 25, 4473–4477 (1986).
Yarus, M. A specific amino acid binding site composed of RNA. Science 240, 1751–1758 (1988).
von Ahsen, U. & Schroeder, R. Streptomycin and self-splicing. Nature 346, 801 (1990).
von Ahsen, U. & Schroeder, R. Streptomycin inhibits splicing of group I introns by competition with the guanosine substrate. Nucleic Acids Res. 19, 2261–2265 (1991).
Liu, T. et al. Molecular insights into de novo small-molecule recognition by an intron RNA structure. Proc. Natl. Acad. Sci. USA 122, e2502425122 (2025).
Pors, K. et al. Alchemix: a novel alkylating anthraquinone with potent activity against anthracycline- and cisplatin-resistant ovarian cancer. Mol. Cancer Ther. 2, 607–610 (2003).
Pors, K. et al. Synthesis and biological evaluation of novel chloroethylaminoanthraquinones with potent cytotoxic activity against cisplatin-resistant tumor cells. J. Med. Chem. 47, 1856–1859 (2004).
Pors, K. et al. Synthesis of DNA-directed pyrrolidinyl and piperidinyl confined alkylating chloroalkylaminoanthraquinones: potential for development of tumor-selective N-oxides. J. Med. Chem. 49, 7013–7023 (2006).
Abdallah, Q. M. A. et al. Minor structural modifications to alchemix influence mechanism of action and pharmacological activity. Biochem. Pharm. 83, 1514–1522 (2012).
Wright, E. P. et al. Mitoxantrone and analogues bind and stabilize i-motif forming DNA sequences. Sci. Rep. 6, 39456 (2016).
Errington, R. J. et al. Probing cytochrome P450 bioactivation and fluorescent properties with morpholinyl-tethered anthraquinones. Bioorg. Med. Chem. Lett. 28, 1274–1277 (2018).
Sugiura, Y., Shiraki, T., Konishi, M. & Oki, T. DNA intercalation and cleavage of an antitumor antibiotic dynemicin that contains anthracycline and enediyne cores. Proc. Natl. Acad. Sci. USA 87, 3831–3835 (1990).
Carlson, C. B., Vuyisich, M., Gooch, B. D. & Beal, P. A. Preferred RNA binding sites for a threading intercalator revealed by in vitro evolution. Chem. Biol. 10, 663–672 (2003).
Zheng, S., Chen, Y., Donahue, C. P., Wolfe, M. S. & Varani, G. Structural basis for stabilization of the tau pre-mRNA splicing regulatory element by novantrone (mitoxantrone). Chem. Biol. 16, 557–566 (2009).
Verebová, V. et al. Anthraquinones quinizarin and danthron unwind negatively supercoiled DNA and lengthen linear DNA. Biochem. Biophys. Res. Commun. 444, 50–55 (2014).
Mohammad, H. et al. Role of intercalation in the electrical properties of nucleic acids for use in molecular electronics. Nanoscale Horiz. 6, 651–660 (2021).
Sosic, A. et al. Multifaceted aspects of HIV-1 nucleocapsid inhibition by TAR-targeting peptidyl-anthraquinones bearing terminal aromatic moieties. Viruses 14, 2133 (2022).
Kapuscinski, J. & Darzynkiewicz, Z. Interactions of antitumor agents ametantrone and mitoxantrone (novatrone) with double-stranded DNA. Biochem. Pharm. 34, 4203–4213 (1985).
Pommier, Y., Leo, E., Zhang, H. & Marchand, C. DNA topoisomerases and their poisoning by anticancer and antibacterial drugs. Chem. Biol. 17, 421–433 (2010).
Feofanov, A., Sharonov, S., Kudelina, I., Fleury, F. & Nabiev, I. Localization and molecular interactions of mitoxantrone within living K562 cells as probed by confocal spectral imaging analysis. Biophys. J. 73, 3317–3327 (1997).
Gopinath, S. C. B., Matsugami, A., Katahira, M. & Kumar, P. K. R. Human vault-associated non-coding RNAs bind to mitoxantrone, a chemotherapeutic compound. Nucleic Acids Res. 33, 4874–4881 (2005).
Velagapudi, S. P. et al. Approved anti-cancer drugs target oncogenic non-coding RNAs. Cell Chem. Biol. 25, 1086–1094.e7 (2018).
Zwelling, L. A. et al. Activity of two novel anthracene-9,10-diones against human leukemia cells containing intercalator-sensitive or -resistant forms of topoisomerase II. Biochem. Pharm. 46, 265–271 (1993).
McKeown, S. R., Hejmadi, M. V., McIntyre, I. A., McAleer, J. J. & Patterson, L. H. AQ4N: an alkylaminoanthraquinone N-oxide showing bioreductive potential and positive interaction with radiation in vivo. Br. J. Cancer 72, 76–81 (1995).
Zubradt, M. et al. DMS-MaPseq for genome-wide or targeted RNA structure probing in vivo. Nat. Methods 14, 75–82 (2017).
Yazdani, K. et al. Decoding complexity in biomolecular recognition of DNA i-motifs with microarrays. Nucleic Acids Res. 51, 12020–12030 (2023).
Liu, T. & Pyle, A. M. Discovery of highly reactive self-splicing group II introns within the mitochondrial genomes of human pathogenic fungi. Nucleic Acids Res. 49, 12422–12432 (2021).
Walstrum, S. A. & Uhlenbeck, O. C. The self-splicing RNA of Tetrahymena is trapped in a less active conformation by gel purification. Biochemistry 29, 10573–10576 (1990).
Zhang, A., Derbyshire, V., Salvo, J. L. & Belfort, M. Escherichia coli protein StpA stimulates self-splicing by promoting RNA assembly in vitro. RNA 1, 783–793 (1995).
Pan, J., Thirumalai, D. & Woodson, S. A. Folding of RNA involves parallel pathways. J. Mol. Biol. 273, 7–13 (1997).
Treiber, D. K., Rook, M. S., Zarrinkar, P. P. & Williamson, J. R. Kinetic intermediates trapped by native interactions in RNA folding. Science 279, 1943–1946 (1998).
Russell, R. & Herschlag, D. Probing the folding landscape of the Tetrahymena ribozyme: commitment to form the native conformation is late in the folding pathway. J. Mol. Biol. 308, 839–851 (2001).
Waldsich, C., Masquida, B., Westhof, E. & Schroeder, R. Monitoring intermediate folding states of the td group I intron in vivo. EMBO J. 21, 5281–5291 (2002).
Sinan, S., Yuan, X. & Russell, R. The Azoarcus group I intron ribozyme misfolds and is accelerated for refolding by ATP-dependent RNA chaperone proteins. J. Biol. Chem. 286, 37304–37312 (2011).
Li, S. et al. Topological crossing in the misfolded Tetrahymena ribozyme resolved by cryo-EM. Proc. Natl. Acad. Sci. USA 119, e2209146119 (2022).
Bonilla, S. L., Vicens, Q. & Kieft, J. S. Cryo-EM reveals an entangled kinetic trap in the folding of a catalytic RNA. Sci. Adv. 8, eabq4144 (2022).
Marinus, T., Fessler, A. B., Ogle, C. A. & Incarnato, D. A novel SHAPE reagent enables the analysis of RNA structure in living cells with unprecedented accuracy. Nucleic Acids Res. 49, e34 (2021).
Incarnato, D. Sequencing-based analysis of RNA structures in living cells with 2A3 via SHAPE-MaP. Methods Enzymol. 691, 153–181 (2023).
Burns, C. P., Haugstad, B. N. & North, J. A. Membrane transport of mitoxantrone by L1210 leukemia cells. Biochem. Pharm. 36, 857–860 (1987).
Bell, D. H. Characterization of the fluorescence of the antitumor agent, mitoxantrone. Biochim. Biophys. Acta 949, 132–137 (1988).
Lan, T. C. T. et al. Secondary structural ensembles of the SARS-CoV-2 RNA genome in infected cells. Nat. Commun. 13, 1128 (2022).
Miglietta, G. et al. RNA G-quadruplexes in Kirsten Ras (KRAS) oncogene as targets for small molecules inhibiting translation. J. Med. Chem. 60, 9448–9461 (2017).
Zhao, J. et al. Enhanced transcriptome-wide RNA G-quadruplex sequencing for low RNA input samples with rG4-seq 2.0. BMC Biol. 20, 257 (2022).
Foye, W. O., Vajragupta, O. & Sengupta, S. K. DNA-binding specificity and RNA polymerase inhibitory activity of bis(aminoalkyl)anthraquinones and bis(methylthio)vinylquinolinium iodides. J. Pharm. Sci. 71, 253–257 (1982).
Lown, J. W., Hanstock, C. C., Bradley, R. D. & Scraba, D. G. Interactions of the antitumor agents mitoxantrone and bisantrene with deoxyribonucleic acids studied by electron microscopy. Mol. Pharm. 25, 178–184 (1984).
Panousis, C. & Phillips, D. R. DNA sequence specificity of mitoxantrone. Nucleic Acids Res. 22, 1342–1345 (1994).
Parker, B. S., Cutts, S. M., Cullinane, C. & Phillips, D. R. Formaldehyde activation of mitoxantrone yields CpG and CpA specific DNA adducts. Nucleic Acids Res. 28, 982–990 (2000).
Liaw, Y. C. et al. Antitumor drug nogalamycin binds DNA in both grooves simultaneously: molecular structure of nogalamycin-DNA complex. Biochemistry 28, 9913–9918 (1989).
Van Nostrand, E. L. et al. A large-scale binding and functional map of human RNA-binding proteins. Nature 583, 711–719 (2020).
Lambert, N. et al. RNA Bind-n-Seq: quantitative assessment of the sequence and structural binding specificity of RNA binding proteins. Mol. Cell 54, 887–900 (2014).
Orenstein, Y., Ohler, U. & Berger, B. Finding RNA structure in the unstructured RBPome. BMC Genomics 19, 154 (2018).
Jolma, A. et al. Binding specificities of human RNA-binding proteins toward structured and linear RNA sequences. Genome Res. 30, 962–973 (2020).
Laverty, K. U. et al. PRIESSTESS: interpretable, high-performing models of the sequence and structure preferences of RNA-binding proteins. Nucleic Acids Res. 50, e111 (2022).
Harris, S. E. et al. Dissecting RNA selectivity mediated by tandem RNA-binding domains. J. Biol. Chem. 301, 108435 (2025).
Borovská, I. et al. Identification of conserved RNA regulatory switches in living cells using RNA secondary structure ensemble mapping and covariation analysis. Nat. Biotechnol. 43, 842–856 (2025).
Clamer, M. et al. Active ribosome profiling with RiboLace. Cell Rep. 25, 1097–1108.e5 (2018).
Cohen, S. S. & Lichtenstein, J. Polyamines and ribosome structure. J. Biol. Chem. 235, 2112–2116 (1960).
Stevens, L. The binding of spermine to the ribosomes and ribosomal ribonucleic acid from Bacillus stearothermophilus. Biochem J. 113, 117–121 (1969).
Hardy, S. J. & Turnock, G. Stabilization of 70S ribosomes by spermidine. Nat. New Biol. 229, 17–19 (1971).
Yoshida, M., Kashiwagi, K., Kawai, G., Ishihama, A. & Igarashi, K. Polyamine enhancement of the synthesis of adenylate cyclase at the translational level and the consequential stimulation of the synthesis of the RNA polymerase sigma 28 subunit. J. Biol. Chem. 276, 16289–16295 (2001).
Koculi, E., Lee, N.-K., Thirumalai, D. & Woodson, S. A. Folding of the tetrahymena ribozyme by polyamines: importance of counterion valence and size. J. Mol. Biol. 341, 27–36 (2004).
Lightfoot, H. L. & Hall, J. Endogenous polyamine function—the RNA perspective. Nucleic Acids Res. 42, 11275–11290 (2014).
Neuner, E., Frener, M., Lusser, A. & Micura, R. Superior cellular activities of azido- over amino-functionalized ligands for engineered preQ1 riboswitches in E.coli. RNA Biol. 15, 1376–1383 (2018).
Sander, T., Freyss, J., von Korff, M. & Rufener, C. DataWarrior: an open-source program for chemistry aware data visualization and analysis. J. Chem. Inf. Model. 55, 460–473 (2015).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J. 17, 10–12 (2011).
Zhang, J., Kobert, K., Flouri, T. & Stamatakis, A. PEAR: a fast and accurate Illumina Paired-End reAd mergeR. Bioinformatics 30, 614–620 (2014).
Incarnato, D., Morandi, E., Simon, L. M. & Oliviero, S. RNA framework: an all-in-one toolkit for the analysis of RNA structures and post-transcriptional modifications. Nucleic Acids Res. 46, e97 (2018).
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Lorenz, R. et al. ViennaRNA package 2.0. Algorithms Mol. Biol. 6, 26 (2011).
Langmead, B., Trapnell, C., Pop, M. & Salzberg, S. L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 10, R25 (2009).
Acknowledgements
This work was supported by a grant from the European Research Council (European Union’s Horizon Europe research and innovation program), grant agreement number 101124787 (RNAStrEnD) to D.I., and partly supported by the Intramural Research Program of the National Institutes of Health (NIH) (ZIABC011585). The contributions of the NIH author(s) were made as part of their official duties as NIH federal employees, are in compliance with agency policy requirements, and are considered Works of the United States Government. However, the findings and conclusions presented in this paper are those of the author(s) and do not necessarily reflect the views of the NIH or the U.S. Department of Health and Human Services.
Author information
Authors and Affiliations
Contributions
C.Z. and D.I. designed the experiments, with input from J.S.S., K.P., and M.D.W.; C.Z., I.B., T.I., M.L., and O.S.O. performed the experiments; R.B. performed the microscopy; M.L., O.S.O., M.D.W., and K.P. performed synthesis and characterization of the anthraquinone analogs; E.M. and D.I. optimized the DRACO algorithm and performed the bioinformatic analyses; M.C. supported the RiboLace analysis; C.Z., I.B., J.S.S., and D.I. wrote the article with input from all the authors; D.I. supervised the research.
Corresponding author
Ethics declarations
Competing interests
M.C. is the founder, director of, and a shareholder in IMMAGINA Biotechnology S.r.l. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Yiliang Ding, and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, C., Borovská, I., Iobashvili, T. et al. RNA functional modulation by Mitoxantrone via RNA structural ensemble repartitioning. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70801-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70801-9