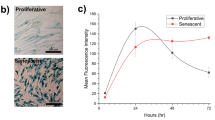
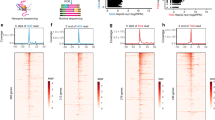
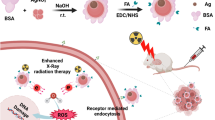
Abstract
Glioblastoma (GBM) patients have dismal survival due to resistance to initial ionizing radiation therapy (RT). Clonal evolution analysis reveals no dominant RT-resistant clones, prompting a genome-wide CRISPR screen to identify radiosensitizing targets. The screening highlights DNA damage response genes, validating the effectiveness of our approach. N-acylneuraminate-9-phosphatase (NANP), a critical enzyme in the sialic acid synthetic pathway, is top-ranked in the screening and associated with patient outcomes. After radiation, NANP-deficient cells exhibit more DNA damage, G2/M arrest and apoptosis, and impaired DNA repair by favoring non-homologous end-joining over homologous recombination. Mechanistically, NANP influences NF-κB signaling and the mesenchymal state by modulating sialylation and internalization of tumor necrosis factor receptor 1 (TNFR1), thereby affecting RT sensitivity. Intracranial orthotopic xenograft experiments validate the function of NANP in vivo. Here, we identify NANP as a radiosensitizing target dependent on TNFR1 sialylation and mesenchymal shift, providing a basis for developing RT sensitizers for GBM.
Similar content being viewed by others
Data availability
All RNA-seq data were deposited in the NCBI GEO repository under accession GSE274135. Raw data for the genome-wide CRISPR screen and clonal evolution analysis have been deposited in the NCBI Sequence Read Archive (SRA) repository under accession number PRJNA1432116 (https://www.ncbi.nlm.nih.gov/sra). Other data that support the findings of this research are available within the paper and its Supplementary Information. Source data are provided with this paper.
References
Stupp, R. et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 10, 459–466 (2009).
Stupp, R. et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 352, 987–996 (2005).
Walker, M. D. et al. Evaluation of BCNU and/or radiotherapy in the treatment of anaplastic gliomas. A cooperative clinical trial. J. Neurosurg. 49, 333–343 (1978).
Walker, M. D. et al. Randomized comparisons of radiotherapy and nitrosoureas for the treatment of malignant glioma after surgery. N. Engl. J. Med. 303, 1323–1329 (1980).
Walker, M. D., Strike, T. A. & Sheline, G. E. An analysis of dose-effect relationship in the radiotherapy of malignant gliomas. Int. J. Radiat. Oncol. Biol. Phys. 5, 1725–1731 (1979).
Stupp, R. et al. Maintenance therapy with tumor-treating fields plus temozolomide vs temozolomide alone for glioblastoma: a randomized clinical trial. JAMA 314, 2535–2543 (2015).
Bao, S. et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 444, 756–760 (2006).
Beier, D., Schulz, J. B. & Beier, C. P. Chemoresistance of glioblastoma cancer stem cells-much more complex than expected. Mol. Cancer 10, 128 (2011).
Gondi, V. et al. Radiotherapy (RT) dose-intensification (DI) using intensity modulated RT (IMRT) versus standard-dose (SD) RT with temozolomide (TMZ) in newly diagnosed glioblastoma (GBM): preliminary results of NRG Oncology BN001. Int. J. Radiat. Oncol., Biol., Phys. 108, S22–S23 (2020).
Wang, Q. et al. Tumor evolution of glioma-intrinsic gene expression subtypes associates with immunological changes in the microenvironment. Cancer Cell 33, 152 (2018).
Neftel, C. et al. An Integrative model of cellular states, plasticity, and genetics for glioblastoma. Cell https://doi.org/10.1016/j.cell.2019.06.024 (2019).
Bhat, K. P. L. et al. Mesenchymal differentiation mediated by NF-kappaB promotes radiation resistance in glioblastoma. Cancer Cell 24, 331–346 (2013).
Osuka, S. et al. N-cadherin upregulation mediates adaptive radioresistance in glioblastoma. J. Clin. Invest. 131 https://doi.org/10.1172/JCI136098 (2021).
Huang, T. et al. MST4 phosphorylation of ATG4B regulates autophagic activity, tumorigenicity, and radioresistance in glioblastoma. Cancer Cell 32, 840–855.e848 (2017).
Minata, M. et al. Phenotypic plasticity of invasive edge glioma stem-like cells in response to ionizing radiation. Cell Rep. 26, 1893–1905.e1897 (2019).
Peixoto, A., Relvas-Santos, M., Azevedo, R., Santos, L. L. & Ferreira, J. A. Protein glycosylation and tumor microenvironment alterations driving cancer hallmarks. Front Oncol. 9, 380 (2019).
Li, F. & Ding, J. Sialylation is involved in cell fate decision during development, reprogramming and cancer progression. Protein Cell 10, 550–565 (2019).
Munkley, J. Aberrant sialylation in cancer: therapeutic opportunities. Cancers (Basel) 14 https://doi.org/10.3390/cancers14174248 (2022).
Zhang, Z.-Y. et al. Lineage-coupled clonal capture identifies clonal evolution mechanisms and vulnerabilities of BRAFV600E inhibition resistance in melanoma. Cell Discov. 8, 102 (2022).
Zhou, W. et al. Purine metabolism regulates DNA repair and therapy resistance in glioblastoma. Nat. Commun. 11, 3811 (2020).
Fu, S. et al. Glutamine synthetase promotes radiation resistance via facilitating nucleotide metabolism and subsequent DNA damage repair. Cell Rep. 28, 1136–1143.e1134 (2019).
Jin, Y. et al. SREBP1/FASN/cholesterol axis facilitates radioresistance in colorectal cancer. FEBS Open Bio 11, 1343–1352 (2021).
Ovejero, S. et al. A sterol-PI(4)P exchanger modulates the Tel1/ATM axis of the DNA damage response. EMBO J. 42, e112684 (2023).
Zhang, Y. et al. Cholesterol depletion sensitizes gallbladder cancer to cisplatin by impairing DNA damage response. Cell Cycle 18, 3337–3350 (2019).
Zhao, H. et al. γ-Radiation-induced G2 delay, apoptosis, and p53 response as potential susceptibility markers for lung cancer. Cancer Res. 61, 7819–7824 (2001).
Bonner, W. M. et al. GammaH2AX and cancer. Nat. Rev. Cancer 8, 957–967 (2008).
Biau, J., Chautard, E., Verrelle, P. & Dutreix, M. Altering DNA repair to improve radiation therapy: specific and multiple pathway targeting. Front Oncol. 9, 1009 (2019).
Arnoult, N. et al. Regulation of DNA repair pathway choice in S and G2 phases by the NHEJ inhibitor CYREN. Nature 549, 548–552 (2017).
Yu, H., Lin, L., Zhang, Z., Zhang, H. & Hu, H. Targeting NF-kappaB pathway for the therapy of diseases: mechanism and clinical study. Signal Transduct. Target Ther. 5, 209 (2020).
Du, J. et al. Dynamic sialylation in transforming growth factor-beta (TGF-beta)-induced epithelial to mesenchymal transition. J. Biol. Chem. 290, 12000–12013 (2015).
Dobie, C. & Skropeta, D. Insights into the role of sialylation in cancer progression and metastasis. Br. J. Cancer 124, 76–90 (2021).
Willems, A. P. et al. Activity of N-acylneuraminate-9-phosphatase (NANP) is not essential for de novo sialic acid biosynthesis. Biochim Biophys. Acta Gen. Subj. 1863, 1471–1479 (2019).
Liu, Z. et al. ST6Gal-I regulates macrophage apoptosis via alpha2-6 sialylation of the TNFR1 death receptor. J. Biol. Chem. 286, 39654–39662 (2011).
Holdbrooks, A. T., Britain, C. M. & Bellis, S. L. ST6Gal-I sialyltransferase promotes tumor necrosis factor (TNF)-mediated cancer cell survival via sialylation of the TNF receptor 1 (TNFR1) death receptor. J. Biol. Chem. 293, 1610–1622 (2018).
D’Addio, M., Frey, J. & Otto, V. I. The manifold roles of sialic acid for the biological functions of endothelial glycoproteins. Glycobiology 30, 490–499 (2020).
Hwang, J. et al. Apoptotic signaling by TNFR1 is inhibited by the alpha2-6 sialylation, but not alpha2-3 sialylation, of the TNFR1 N-glycans. J. Biol. Chem. 301, 108043 (2025).
Barthel, F. P. et al. Longitudinal molecular trajectories of diffuse glioma in adults. Nature 576, 112–120 (2019).
Helleday, T., Petermann, E., Lundin, C., Hodgson, B. & Sharma, R. A. DNA repair pathways as targets for cancer therapy. Nat. Rev. Cancer 8, 193–204 (2008).
Yang, C. et al. A20/TNFAIP3 regulates the DNA damage response and mediates tumor cell resistance to DNA-damaging therapy. Cancer Res. 78, 1069–1082 (2018).
Allen, M., Bjerke, M., Edlund, H., Nelander, S. & Westermark, B. Origin of the U87MG glioma cell line: Good news and bad news. Sci. Transl. Med. 8, 354re353 (2016).
Gc, S. et al. alpha2,6 Sialylation mediated by ST6GAL1 promotes glioblastoma growth. JCI Insight 7 https://doi.org/10.1172/jci.insight.158799 (2022).
Lee, M., Lee, H. J., Bae, S. & Lee, Y. S. Protein sialylation by sialyltransferase involves radiation resistance. Mol. Cancer Res. 6, 1316–1325 (2008).
Lee, M., Park, J. J., Ko, Y. G. & Lee, Y. S. Cleavage of ST6Gal I by radiation-induced BACE1 inhibits golgi-anchored ST6Gal I-mediated sialylation of integrin beta1 and migration in colon cancer cells. Radiat. Oncol. 7, 47 (2012).
Park, J. J. et al. Sialylation of epidermal growth factor receptor regulates receptor activity and chemosensitivity to gefitinib in colon cancer cells. Biochem. Pharm. 83, 849–857 (2012).
Lopez-Sambrooks, C. et al. Oligosaccharyltransferase inhibition induces senescence in RTK-driven tumor cells. Nat. Chem. Biol. 12, 1023–1030 (2016).
Lampson, B. L. et al. Positive selection CRISPR screens reveal a druggable pocket in an oligosaccharyltransferase required for inflammatory signaling to NF-kappaB. Cell 187, 2209–2223.e2216 (2024).
Maliekal, P., Vertommen, D., Delpierre, G. & Van Schaftingen, E. Identification of the sequence encoding N-acetylneuraminate-9-phosphate phosphatase. Glycobiology 16, 165–172 (2006).
Kim, M., Lu, F. & Zhang, Y. Loss of HDAC-Mediated Repression and Gain of NF-kappaB Activation Underlie Cytokine Induction in ARID1A- and PIK3CA-Mutation-Driven Ovarian Cancer. Cell Rep. 17, 275–288 (2016).
Feng, J. et al. GFOLD: a generalized fold change for ranking differentially expressed genes from RNA-seq data. Bioinformatics 28, 2782–2788 (2012).
Doench, J. G. et al. Optimized sgRNA design to maximize activity and minimize off-target effects of CRISPR-Cas9. Nat. Biotechnol. 34, 184–191 (2016).
Li, W. et al. MAGeCK enables robust identification of essential genes from genome-scale CRISPR/Cas9 knockout screens. Genome Biol. 15, 554 (2014).
Wang, B. et al. Integrative analysis of pooled CRISPR genetic screens using MAGeCKFlute. Nat. Protoc. 14, 756–780 (2019).
Evangelista, J. E. et al. Enrichr-KG: bridging enrichment analysis across multiple libraries. Nucleic Acids Res. 51, W168–W179 (2023).
Sule, A. et al. Targeting IDH1/2 mutant cancers with combinations of ATR and PARP inhibitors. NAR Cancer 3, zcab018 (2021).
Gyori, B. M., Venkatachalam, G., Thiagarajan, P. S., Hsu, D. & Clement, M. V. OpenComet: an automated tool for comet assay image analysis. Redox Biol. 2, 457–465 (2014).
Lal, S. et al. An implantable guide-screw system for brain tumor studies in small animals. J. Neurosurg. 92, 326–333 (2000).
Shu, J., Dolman, G. E., Duan, J., Qiu, G., Ilyas, M. in International Conference on Image and Graphics (ICIG) 937–942 (2013).
Bowman, R. L., Wang, Q., Carro, A., Verhaak, R. G. & Squatrito, M. GlioVis data portal for visualization and analysis of brain tumor expression datasets. Neuro Oncol. 19, 139–141 (2017).
Gao, J. et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal 6, pl1 (2013).
Sherman, B. T. et al. DAVID: a web server for functional enrichment analysis and functional annotation of gene lists (2021 update). Nucleic Acids Res. 50, W216–W221 (2022).
Zhao, Z. et al. Chinese glioma genome Atlas (CGGA): a comprehensive resource with functional genomic data from Chinese Glioma Patients. Genomics Proteom. Bioinforma. 19, 1–12 (2021).
Gravendeel, L. A. et al. Intrinsic gene expression profiles of gliomas are a better predictor of survival than histology. Cancer Res. 69, 9065–9072 (2009).
Acknowledgements
We thank Dr. Michael Pacold at NYU Langone Health for helpful discussions. We thank the NYU Langone Health’s Cytometry and Cell Sorting Laboratory for cytometry support; Experimental Pathology Research Laboratory for support on IHC experiment; Microscopy Laboratory for microscope imaging support and Preclinical Imaging Laboratory for small animal imaging support. These cores are all partially supported by the Cancer Center Support Grant P30CA016087 at NYU Langone’s Laura and Isaac Perlmutter Cancer Center. The Preclinical Imaging Laboratory is also partially supported by NIBIB Biomedical Technology Resource Center Grant NIH P41 EB017183. This work is supported by R01CA282756 (Y.D., Z.-Y.Z., and E.P.S.). This project was also supported by a grant from the NYU Technology Opportunities & Ventures Therapeutics Alliances Fund (Y.D., Z.-Y.Z.).
Author information
Authors and Affiliations
Contributions
E.P.S., Z.-Y.Z., and Y.D. conceived and designed the study; Y.D. and Z.-Y.Z. designed, performed, and analyzed most experiments; Z.-Y.Z., R.E., and M.G. performed in vivo study. G.M. and A.J. provided help with the experiments during revision. A.M., J.K. provided critical suggestions; R.E. did administration and management. Y.D., Z.-Y.Z., and E.P.S. wrote the manuscript. All authors have reviewed and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Monica Venere and the other anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ding, Y., Zhang, ZY., Ezhilarasan, R. et al. NANP targeting radiosensitizes glioblastoma through TNFR1 sialylation-driven mesenchymal shift. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70853-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70853-x