Abstract
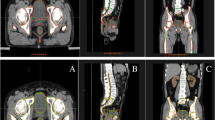
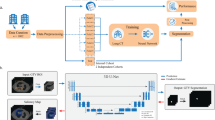
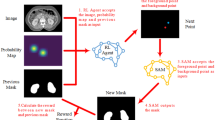
Widespread clinical implementation of rapidly evolving auto-segmentation tools remains constrained by a scarcity of high-quality prospective evidence. Here we show the results of a prospective, multicenter, observational trial (NCT05787522) evaluating the clinical performance of a deep learning model (iCurveE) for artificial intelligence (AI)-assisted delineation of organs at risk (OARs) in thoracic and breast cancer radiotherapy. Computed tomography images from 500 patients across five centers are annotated by 37 physicians using manual, AI-generated, and AI-assisted methods. Eleven thoracic OARs are evaluated based on the primary endpoints of volumetric Dice similarity coefficient (vDSC) and contouring time, alongside secondary metrics including 95% Hausdorff Distance (HD95). We prospectively annotate 2,483 OAR sets (27,043 OARs): 993 manual, 497 AI-generated, and 993 AI-assisted. AI-assisted delineation achieves significantly better vDSC (mean, 0.902) and HD95 (mean, 5.20 mm) than manual delineation (mean vDSC, 0.857; mean HD95, 8.01 mm; p < 0.0001) while improving time efficiency by 81.63% (median: 10.0 vs. 55.0 min; p < 0.0001). AI-assisted delineation reduces performance variability across centers and physicians with varying expertise. This study validates the clinical applicability of AI-assisted delineation in improving delineation performance and promoting healthcare equity.
Similar content being viewed by others
Data availability
The source data underlying the figures and tables generated in this study are provided in the Supplementary Materials and Source Data file. The CT imaging datasets and delineation results generated in this study are available under restricted access due to utilization in ongoing research projects and patient privacy protection; access can be obtained by contacting the corresponding author, Z.Y.Yu. (email: zyuan@tmu.edu.cn), with a research protocol and proof of ethical approval. The corresponding author will respond to access requests within 15 working days and data will be available for 6 months. Source data are provided with this paper.
Code availability
The iCurveE auto-segmentation model used in this study is a commercial software product developed by Perception Vision Medical Technology (PVmed), and its source code is proprietary and not publicly available. Further information regarding the model and its use can be obtained from the corresponding author (Z.Y.Yu.) with a research protocol and proof of ethical approval.
References
Hendriks, L. E. L. et al. Non-small-cell lung cancer. Nat. Rev. Dis. Prim. 10, 71 (2024).
Benitez Fuentes, J. D. et al. Global stage distribution of breast cancer at diagnosis: a systematic review and meta-analysis. JAMA Oncol. 10, 71–78 (2024).
Yang, H., Wang, F., Hallemeier, C. L., Lerut, T. & Fu, J. Oesophageal cancer. Lancet (Lond., Engl.) 404, 1991–2005 (2024).
Kim, S. Y., Park, H. S. & Chiang, A. C. Small cell lung cancer: a review. J. Am. Med. Assoc. 333, 1906–1917 (2025).
Sung, H. et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 71, 209–249 (2021).
Miller, K. D. et al. Cancer treatment and survivorship statistics, 2022. CA Cancer J. Clin. 72, 409–436 (2022).
Kroeze, S. G. C. et al. Metastases-directed stereotactic body radiotherapy in combination with targeted therapy or immunotherapy: systematic review and consensus recommendations by the EORTC-ESTRO OligoCare consortium. Lancet Oncol. 24, e121–e132 (2023).
Palma, D. A. et al. Stereotactic ablative radiotherapy for the comprehensive treatment of oligometastatic cancers: long-term results of the SABR-COMET phase II randomized trial. J. Clin. Oncol. 38, 2830–2838 (2020).
Lynch, C., Pitroda, S. P. & Weichselbaum, R. R. Radiotherapy, immunity, and immune checkpoint inhibitors. Lancet Oncol. 25, e352–e362 (2024).
Vinod, S. K., Jameson, M. G., Min, M. & Holloway, L. C. Uncertainties in volume delineation in radiation oncology: a systematic review and recommendations for future studies. Radiother. Oncol. 121, 169–179 (2016).
Niu, G. M. et al. Dosimetric analysis of brachial plexopathy after stereotactic body radiotherapy: Significance of organ delineation. Radiother. Oncol. 190, 110023 (2024).
Wang, K. & Tepper, J. E. Radiation therapy-associated toxicity: etiology, management, and prevention. CA Cancer J. Clin. 71, 437–454 (2021).
Podobnik, G., Ibragimov, B., Peterlin, P., Strojan, P. & Vrtovec, T. vOARiability: Interobserver and intermodality variability analysis in OAR contouring from head and neck CT and MR images. Med. Phys. 51, 2175–2186 (2024).
Yan, Y. et al. Edge roughness quantifies impact of physician variation on training and performance of deep learning auto-segmentation models for the esophagus. Sci. Rep. 14, 2536 (2024).
Ni, R., Han, K., Haibe-Kains, B. & Rink, A. Generalizability of deep learning in organ-at-risk segmentation: A transfer learning study in cervical brachytherapy. Radiother. Oncol. 197, 110332 (2024).
Vagni, M. et al. Auto-segmentation of pelvic organs at risk on 0.35T MRI using 2D and 3D Generative Adversarial Network models. Phys. Med. 119, 103297 (2024).
Liao, W. et al. Comprehensive evaluation of a deep learning model for automatic organs-at-risk segmentation on heterogeneous computed tomography images for abdominal radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 117, 994–1006 (2023).
Song, Y. et al. Automatic delineation of the clinical target volume and organs at risk by deep learning for rectal cancer postoperative radiotherapy. Radiother. Oncol. 145, 186–192 (2020).
Chen, X. et al. A deep learning-based auto-segmentation system for organs-at-risk on whole-body computed tomography images for radiation therapy. Radiother. Oncol. 160, 175–184 (2021).
Shi, F. et al. Deep learning empowered volume delineation of whole-body organs-at-risk for accelerated radiotherapy. Nat. Commun. 13, 6566 (2022).
Wasserthal, J. et al. TotalSegmentator: robust Segmentation of 104 Anatomic Structures in CT Images. Radio. Artif. Intell. 5, e230024 (2023).
Li, G., Wu, X. & Ma, X. Artificial intelligence in radiotherapy. Semin Cancer Biol. 86, 160–171 (2022).
Busch, F. et al. Navigating the European Union Artificial Intelligence Act for Healthcare. NPJ Digital Med. 7, 210 (2024).
Lekadir, K. et al. FUTURE-AI: international consensus guideline for trustworthy and deployable artificial intelligence in healthcare. Br. Med. J. 388, e081554 (2025).
Pham, T. Ethical and legal considerations in healthcare AI: innovation and policy for safe and fair use. R. Soc. Open Sci. 12, 241873 (2025).
Ye, X. et al. Comprehensive and clinically accurate head and neck cancer organs-at-risk delineation on a multi-institutional study. Nat. Commun. 13, 6137 (2022).
Chen, Z. H. et al. Deep learning-based delineation of whole-body organs at risk empowering adaptive radiotherapy. BMC Med Inf. Decis. Mak. 25, 268 (2025).
Poel, R. et al. A comprehensive multifaceted technical evaluation framework for implementation of auto-segmentation models in radiotherapy. Commun. Med. (Lond.) 5, 319 (2025).
Abdel-Wahab, M. et al. Addressing challenges in low-income and middle-income countries through novel radiotherapy research opportunities. Lancet Oncol. 25, e270–e280 (2024).
LaVigne, A. W. et al. Radiotherapy deserts: the impact of race, poverty, and the rural-urban continuum on density of providers and the use of radiation therapy in the US. Int. J. Radiat. Oncol. Biol. Phys. 116, 17–27 (2023).
Lu, S. L. et al. Randomized multi-reader evaluation of automated detection and segmentation of brain tumors in stereotactic radiosurgery with deep neural networks. Neurooncology 23, 1560–1568 (2021).
Tikhomirov, L. et al. Medical artificial intelligence for clinicians: the lost cognitive perspective. Lancet Digit Health 6, e589–e594 (2024).
Isensee, F., Jaeger, P. F., Kohl, S. A. A., Petersen, J. & Maier-Hein, K. H. nnU-Net: a self-configuring method for deep learning-based biomedical image segmentation. Nat. Methods 18, 203–211 (2021).
Vuong, Q.H. et al. How AI’s self-prolongation influences people’s perceptions of its autonomous mind: the case of U.S. residents. Behav. Sci. (Basel) 13, 470 (2023).
Rong, Y. et al. NRG oncology assessment of artificial intelligence deep learning-based auto-segmentation for radiation therapy: current developments, clinical considerations, and future directions. Int. J. Radiat. Oncol. Biol. Phys. 119, 261–280 (2024).
Zhao, W. et al. MMCA-NET: a multimodal cross attention transformer network for nasopharyngeal carcinoma tumor segmentation based on a total-body PET/CT system. IEEE J. Biomed. Health Inform. 28, 5447–5458 (2024).
Zhu, H. et al. Global radiotherapy demands and corresponding radiotherapy-professional workforce requirements in 2022 and predicted to 2050: a population-based study. Lancet Glob. Health 12, e1945–e1953 (2024).
Cao, M. et al. Current cancer burden in China: epidemiology, etiology, and prevention. Cancer Biol. Med. 19, 1121–1138 (2022).
Hosny, A. et al. Clinical validation of deep learning algorithms for radiotherapy targeting of non-small-cell lung cancer: an observational study. Lancet Digital Health 4, e657–e666 (2022).
Wong, J. et al. Training and validation of deep learning-based auto-segmentation models for lung stereotactic ablative radiotherapy using retrospective radiotherapy planning contours. Front. Oncol. 11, 626499 (2021).
Costea, M. et al. Comparison of atlas-based and deep learning methods for organs at risk delineation on head-and-neck CT images using an automated treatment planning system. Radiother. Oncol. 177, 61–70 (2022).
Prabhu, S. P. Ethical challenges of machine learning and deep learning algorithms. Lancet Oncol. 20, 621–622 (2019).
Watson, D. S. et al. Clinical applications of machine learning algorithms: beyond the black box. Br. Med. J. 364, l886 (2019).
Huang, K. et al. Impact of slice thickness, pixel size, and CT dose on the performance of automatic contouring algorithms. J. Appl. Clin. Med. Phys. 22, 168–174 (2021).
Monnin, P., Sfameni, N., Gianoli, A. & Ding, S. Optimal slice thickness for object detection with longitudinal partial volume effects in computed tomography. J. Appl. Clin. Med. Phys. 18, 251–259 (2017).
Oh, Y. et al. LLM-driven multimodal target volume contouring in radiation oncology. Nat. Commun. 15, 9186 (2024).
Kong, F. M. et al. Consideration of dose limits for organs at risk of thoracic radiotherapy: atlas for lung, proximal bronchial tree, esophagus, spinal cord, ribs, and brachial plexus. Int. J. Radiat. Oncol. Biol. Phys. 81, 1442–1457 (2011).
Jabbour, S. K. et al. Upper abdominal normal organ contouring guidelines and atlas: a Radiation Therapy Oncology Group consensus. Pract. Radiat. Oncol. 4, 82–89 (2014).
Weir, J. & Abrahams, P.H. Weir & Abrahams’ Imaging Atlas of Human Anatomy (Elsevier, 2020).
Bentzen, S. M. et al. Quantitative Analyses of Normal Tissue Effects in the Clinic (QUANTEC): an introduction to the scientific issues. Int. J. Radiat. Oncol. Biol. Phys. 76, S3–S9 (2010).
Timmerman, R. A story of hypofractionation and the table on the wall. Int. J. Radiat. Oncol. Biol. Phys. 112, 4–21 (2022).
Yeghiazaryan, V. & Voiculescu, I. Family of boundary overlap metrics for the evaluation of medical image segmentation. J. Med. Imaging (Bellingham) 5, 015006 (2018).
Acknowledgements
We would like to thank all the patients and medical and non-medical participants across the centers for their invaluable contribution. We also sincerely thank PVmed for providing the Res-SE net (iCurveE) auto-segmentation model and PVmed Contouring system, which significantly supported this research. This research was supported by grants from the National Natural Science Foundation of China (No. 82473240 to Z.Y.Yu.), the Tianjin Key Medical Discipline Construction Project (No. TJYXZDXK-3-004B to Z.Y.Yu.), the Tianjin Key Medical Discipline (Specialty) Construction Project (TJYXZDXK-009A to Z.Y.Yu.), the National Key Research and Development Program of China (No. 2023YFE0204300 to Z.Y.Yu.), Tianjin Medical University Cancer Hospital 358 project (No. 358-2022-5 to Y.G.) and the Guilin Technology Application and Promotion Project (No. 20210227-9-4 to D.M.W.).
Author information
Authors and Affiliations
Contributions
Z.Y.Yu., Y.G., and G.M.N. conceived and designed the study. Z.Y.Yu. and Y.C.S. supervised the study. Y.G., Y.F.Z., Y.C.S., S.H., K.Y.Y., H.Q.Y., X.F.P., Y.N., Y.M.L., J.J.C., Y.T.F., D.H.Z., and L.J.Z. collected and curated the data. G.M.N., M.Y., X.F.W., and J.R.C. conducted data analysis and statistical tests. G.M.N., S.F.L., and J.R.C. designed the figures and tables. Y.L., Z.Y.Ya., and T. L. contributed to model development. Y.C.S., W.C.Z., M.B.M., J.H., J.Z.C., S.S.G., and D.M.W. were responsible for project administration. G.M.N., Y.G., and Y.F.Z., wrote the manuscript. Y.C.S., L.J.Z., and Z.Y.Yu. discussed and reviewed the manuscript. These authors contributed equally: G.M.N., Y.G., Y.F.Z., and Y.C.S. All authors had access to the raw datasets and were responsible for the decision to submit the manuscript for publication.
Corresponding author
Ethics declarations
Competing interests
T.L., Z.Y.Ya., and Y.L. are employees of Perception Vision Medical Technologies (PVmed) and contributed to software development. PVmed did not participate in study design, data collection, analysis, or manuscript writing. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Jia Wu, who co-reviewed with Eman Showkatian; and Nanna Sijtsema for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Niu, G., Guan, Y., Zhang, Y. et al. A prospective multicenter trial of deep learning auto-segmentation for organs at risk in thoracic radiotherapy. Nat Commun (2026). https://doi.org/10.1038/s41467-026-70863-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-70863-9