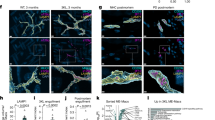
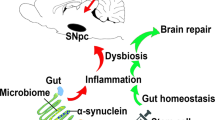
Abstract
Gastrointestinal dysfunction often precedes motor symptoms in Parkinson’s disease (PD), suggesting the enteric nervous system (ENS) is central to early pathogenesis. How α-synuclein contributes to ENS dysfunction, and how inflammation modulates this, remains unclear. Here we show that Tumor Necrosis Factor alpha enhances α-synuclein accumulation in induced pluripotent stem cell-derived enteric neurons and glia, and impairs the malate-aspartate shuttle, a key pathway for mitochondrial energy production. This drives a metabolic shift toward glutamine oxidation in patient cells. This metabolic impairment reduces overall mitochondrial function, which is partially rescued by the neuroprotective compound Chicago-Sky-Blue 6B. Furthermore, transcriptomic and histological analyses of human gut tissue from inflammatory bowel disease patients reveal that inflammation-associated metabolic suppression and α-synuclein upregulation occur beyond PD, representing general hallmarks of intestinal inflammation. These findings highlight a conserved metabolic vulnerability in the ENS and establish patient-derived enteric lineages as a robust platform to model inflammatory ENS pathology.
Similar content being viewed by others
Data availability
The scRNAseq data generated in this study have been deposited in the GEO database under accession code GSE301050. The proteomics data generated in this study have been deposited in the ProteomeXChange database under accession code PXD075048 and in the MassIVE database under accession code MSV000101003 [https://massive.ucsd.edu/ProteoSAFe/dataset.jsp?task=b41f61176673413694a7cf65448de9fe]. The metabolomics data generated in this study have been deposited in the MassIVE database under accession code MSV000098366 [https://massive.ucsd.edu/ProteoSAFe/dataset.jsp?task=85492deedc7c40a39a3568287f5d398b]. Source data are provided with this paper.
Code availability
All codes used in this publication, together with the Seurat object used to generate all our scRNAseq data are available at [Ghirotto, Bruno (2025), “ENS alpha synuclein paper 2025”, Mendeley Data, V2, [https://data.mendeley.com/datasets/v8dknj466y/2].
References
Poewe, W. et al. Parkinson disease. Nat. Rev. Dis. Prim. 3, 17013 (2017).
Warnecke, T., Schäfer, K. H., Claus, I., Del Tredici, K. & Jost, W. H. Gastrointestinal involvement in Parkinson’s disease: pathophysiology, diagnosis, and management. NPJ Parkinson’s. Dis. 8, 31 (2022).
Fasano, A., Visanji, N. P., Liu, L. W., Lang, A. E. & Pfeiffer, R. F. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 14, 625–639 (2015).
Cersosimo, M. G. et al. Gastrointestinal manifestations in Parkinson’s disease: prevalence and occurrence before motor symptoms. J. Neurol. 260, 1332–1338 (2013).
Schneider, S., Wright, C. M. & Heuckeroth, R. O. Unexpected roles for the second brain: enteric nervous system as master regulator of bowel function. Annu. Rev. Physiol. 81, 235–259 (2019).
Andersen, K. B. et al. Sympathetic and parasympathetic subtypes of body-first Lewy body disease observed in postmortem tissue from prediagnostic individuals. Nat. Neurosci. 28, 925–936 (2025).
Hijaz, B. A. & Volpicelli-Daley, L. A. Initiation and propagation of α-synuclein aggregation in the nervous system. Mol. Neurodegeneration 15, 19 (2020).
Emmi, A. et al. Duodenal alpha-synuclein pathology and enteric gliosis in advanced Parkinson’s disease. Mov. Disord. 38, 885–894 (2023).
Pellegrini, C. et al. Enteric α-synuclein impairs intestinal epithelial barrier through caspase-1-inflammasome signaling in Parkinson’s disease before brain pathology. NPJ Parkinson’s. Dis. 8, 9 (2022).
Pan-Montojo, F. et al. Environmental toxins trigger PD-like progression via increased alpha-synuclein release from enteric neurons in mice. Sci. Rep. 2, 898 (2012).
Garretti, F. et al. Interaction of an α-synuclein epitope with HLA-DRB1∗15:01 triggers enteric features in mice reminiscent of prodromal Parkinson’s disease. Neuron 111, 3397–3413.e5 (2023).
Tansey, M. G. et al. Inflammation and immune dysfunction in Parkinson disease. Nat. Rev. Immunol. 22, 657–673 (2022).
Gao, H. M. et al. Neuroinflammation and oxidation/nitration of alpha-synuclein linked to dopaminergic neurodegeneration. J. Neurosci. Off. J. Soc. Neurosci. 28, 7687–7698 (2008).
Bayati, A. et al. Modeling Parkinson’s disease pathology in human dopaminergic neurons by sequential exposure to α-synuclein fibrils and proinflammatory cytokines. Nat. Neurosci. 27, 2401–2416 (2024).
Sigutova, V., Xiang, W., Regensburger, M., Winner, B. & Prots, I. Alpha-synuclein fine-tunes neuronal response to pro-inflammatory cytokines. Brain, Behav., Immun. 122, 216–230 (2024).
Resnikoff, H. et al. Colonic inflammation affects myenteric alpha-synuclein in nonhuman primates. J. Inflamm. Res. 12, 113–126 (2019).
Jiang, W. et al. Involvement of abnormal p-α-syn sccumulation and TLR2-Mediated Inflammation of Schwann Cells in Enteric Autonomic Nerve Dysfunction of Parkinson’s Disease: an Animal Model Study. Mol. Neurobiol. 60, 4738–4752 (2023).
Li, Z. et al. Transcriptomics of Hirschsprung disease patient-derived enteric neural crest cells reveals a role for oxidative phosphorylation. Nat. Commun. 14, 2157 (2023).
Henrich, M. T., Oertel, W. H., Surmeier, D. J. & Geibl, F. F. Mitochondrial dysfunction in Parkinson’s disease - a key disease hallmark with therapeutic potential. Mol. Neurodegeneration 18, 83 (2023).
Peng, W., Schröder, L. F., Song, P., Wong, Y. C. & Krainc, D. Parkin regulates amino acid homeostasis at mitochondria-lysosome (M/L) contact sites in Parkinson’s disease. Sci. Adv. 9, eadh3347 (2023).
Marshall, L. J., Bailey, J., Cassotta, M., Herrmann, K. & Pistollato, F. Poor translatability of biomedical research using animals - a narrative review. Alternat Lab. animals: ATLA 51, 102–135 (2023).
Vadodaria, K. C., Jones, J. R., Linker, S. & Gage, F. H. Modeling brain disorders using induced pluripotent stem cells. Cold Spring Harb. Perspect. Biol. 12, a035659 (2020).
Barber, K., Studer, L. & Fattahi, F. Derivation of enteric neuron lineages from human pluripotent stem cells. Nat. Protoc. 14, 1261–1279 (2019).
Fattahi, F. et al. Deriving human ENS lineages for cell therapy and drug discovery in Hirschsprung disease. Nature 531, 105–109 (2016).
Stojkovska, I. et al. Rescue of α-synuclein aggregation in Parkinson’s patient neurons by synergistic enhancement of ER proteostasis and protein trafficking. Neuron 110, 436–451.e11 (2022).
Guyer, R. A. et al. Single-cell multiome sequencing clarifies enteric glial diversity and identifies an intraganglionic population poised for neurogenesis. Cell Rep. 42, 112194 (2023).
Drokhlyansky, E. et al. The human and mouse enteric nervous system at single-cell resolution. Cell 182, 1606–1622.e23 (2020).
Hamnett, R. et al. Enteric glutamatergic interneurons regulate intestinal motility. Neuron 113, 1019–1035.e6 (2025).
Patikas, N., Ansari, R. & Metzakopian, E. Single-cell transcriptomics identifies perturbed molecular pathways in midbrain organoids using α-synuclein triplication Parkinson’s disease patient-derived iPSCs. Neurosci. Res. 195, 13–28 (2023).
Jin, Y. et al. Modeling Lewy body disease with SNCA triplication iPSC-derived cortical organoids and identifying therapeutic drugs. Sci. Adv. 10, eadk3700 (2024).
Wang, X. et al. Pathogenic alpha-synuclein aggregates preferentially bind to mitochondria and affect cellular respiration. Acta Neuropathol. Commun. 7, 41 (2019).
Street, K. et al. Slingshot: cell lineage and pseudotime inference for single-cell transcriptomics. BMC Genom. 19, 477 (2018).
Fernandez-Vizarra, E. & Zeviani, M. Mitochondrial disorders of the OXPHOS system. FEBS Lett. 595, 1062–1106 (2021).
Fernandez-Marcos, P. J. & Auwerx, J. Regulation of PGC-1α, a nodal regulator of mitochondrial biogenesis. Am. J. Clin. Nutr. 93, 884S–890S (2011).
Jin, S. M. & Youle, R. J. PINK1- and Parkin-mediated mitophagy at a glance. J. Cell Sci. 125, 795–799 (2012).
Vanstone, J. R. et al. DNM1L-related mitochondrial fission defect presenting as refractory epilepsy. Eur. J. Hum. Genet. 24, 1084–1088 (2016).
Jin, S. et al. Inference and analysis of cell-cell communication using CellChat. Nat. Commun. 12, 1088 (2021).
Byrne, A. M., Bouchier-Hayes, D. J. & Harmey, J. H. Angiogenic and cell survival functions of vascular endothelial growth factor (VEGF). J. Cell Mol. Med 9, 777–794 (2005).
Rothhammer, V. et al. Microglial control of astrocytes in response to microbial metabolites. Nature 557, 724–728 (2018).
Cirac, A. et al. The aryl hydrocarbon receptor-dependent TGF-α/VEGF-B ratio correlates with disease subtype and prognosis in multiple sclerosis. Neurol. Neuroimmunol. Neuroinflamm 8, e1043 (2021).
Fu, M., Vohra, B. P., Wind, D. & Heuckeroth, R. O. BMP signaling regulates murine enteric nervous system precursor migration, neurite fasciculation, and patterning via altered Ncam1 polysialic acid addition. Dev. Biol. 299, 137–150 (2006).
Van Landeghem, L. et al. Enteric glia promote intestinal mucosal healing via activation of focal adhesion kinase and release of proEGF. Am. J. Physiol. Gastrointest. Liver Physiol. 300, G976–G987 (2011).
Wang, H., Foong, J. P. P., Harris, N. L. & Bornstein, J. C. Enteric neuroimmune interactions coordinate intestinal responses in health and disease. Mucosal Immunol. 15, 27–39 (2022).
Kowalczyk, A., Kleniewska, P., Kolodziejczyk, M., Skibska, B. & Goraca, A. The role of endothelin-1 and endothelin receptor antagonists in inflammatory response and sepsis. Archivum Immunol. Therapiae Experimentalis 63, 41–52 (2015).
Crisponi, L., Buers, I. & Rutsch, F. CRLF1 and CLCF1 in development, health and disease. Int. J. Mol. Sci. 23, 992 (2022).
Luo, J. K., Melland, H., Nithianantharajah, J. & Gordon, S. L. Postsynaptic neuroligin-1 mediates presynaptic endocytosis during neuronal activity. Front. Mol. Neurosci. 14, 744845 (2021).
Born, G. et al. Modulation of synaptic function through the α-neurexin-specific ligand neurexophilin-1. Proc. Natl. Acad. Sci. USA 111, E1274–E1283 (2014).
Qiu, P. et al. The Eph/ephrin system symphony of gut inflammation. Pharm. Res 197, 106976 (2023).
Diner, I., Nguyen, T. & Seyfried, N. T. Enrichment of detergent-insoluble protein aggregates from human postmortem brain. J. Visualized Exp JoVE 128, 55835 (2017).
Windster, J. D. et al. A combinatorial panel for flow cytometry-based isolation of enteric nervous system cells from human intestine. EMBO Rep. 24, e55789 (2023).
Sauvola, C. W. & Littleton, J. T. SNARE regulatory proteins in synaptic vesicle fusion and recycling. Front. Mol. Neurosci. 14, 733138 (2021).
Borst, P. The malate-aspartate shuttle (Borst cycle): How it started and developed into a major metabolic pathway. IUBMB Life 72, 2241–2259 (2020).
Plattner, C. et al. IBDome: an integrated molecular, histopathological, and clinical atlas of inflammatory bowel diseases. bioRxiv: the preprint server for biology, 2025.03.26.645544. https://doi.org/10.1101/2025.03.26.645544 (2025).
Majd, H. et al. A call for a unified and multimodal definition of cellular identity in the enteric nervous system. EMBO J. 44, 5622–5639 (2025).
Flierl, A. et al. Higher vulnerability and stress sensitivity of neuronal precursor cells carrying an alpha-synuclein gene triplication. PloS One 9, e112413 (2014).
Sharma, M. & Burré, J. α-Synuclein in synaptic function and dysfunction. Trends Neurosci. 46, 153–166 (2023).
Yi, S., Wang, L., Wang, H., Ho, M. S. & Zhang, S. Pathogenesis of α-synuclein in Parkinson’s disease: from a neuron-glia crosstalk perspective. Int. J. Mol. Sci. 23, 14753 (2022).
Corbillé, A. G., Neunlist, M. & Derkinderen, P. Cross-linking for the analysis of α-synuclein in the enteric nervous system. J. Neurochem. 139, 839–847 (2016).
Reale, M. et al. Peripheral cytokines profile in Parkinson’s disease. Brain Behav. Immun. 23, 55–63 (2009).
Brodacki, B. et al. Serum interleukin (IL-2, IL-10, IL-6, IL-4), TNFalpha, and INFgamma concentrations are elevated in patients with atypical and idiopathic Parkinsonism. Neurosci. Lett. 441, 158–162 (2008).
Roulis, M., Armaka, M., Manoloukos, M., Apostolaki, M. & Kollias, G. Intestinal epithelial cells as producers but not targets of chronic TNF suffice to cause murine Crohn-like pathology. Proc. Natl. Acad. Sci. USA 108, 5396–5401 (2011).
Peter, I. et al. Anti-tumor necrosis factor therapy and incidence of Parkinson disease among patients with inflammatory bowel disease. JAMA Neurol. 75, 939–946 (2018).
Xu, J., Wang, L., Chen, X. & Le, W. New Understanding on the Pathophysiology and Treatment of Constipation in Parkinson’s Disease. Front. Aging Neurosci. 14, 917499 (2022).
Schöndorf, D. C. et al. The NAD+ precursor nicotinamide riboside rescues mitochondrial defects and neuronal loss in iPSC and fly models of Parkinson’s disease. Cell Rep. 23, 2976–2988 (2018).
Lautrup, S., Sinclair, D. A., Mattson, M. P. & Fang, E. F. NAD+ in brain aging and neurodegenerative disorders. Cell Metab. 30, 630–655 (2019).
Perez, M. J. & Deleidi, M. New insights into the autophagy-NAD axis in brain disease. Cell Rep. 42, 112420 (2023).
Schmidt, S. et al. A reversible state of hypometabolism in a human cellular model of sporadic Parkinson’s disease. Nat. Commun. 14, 7674 (2023).
Li, W. X. et al. Systematic metabolic analysis of potential target, therapeutic drug, diagnostic method and animal model applicability in three neurodegenerative diseases. Aging 12, 9882–9914 (2020).
Graham, S. F. et al. Biochemical profiling of the brain and blood metabolome in a mouse model of prodromal Parkinson’s disease reveals distinct metabolic profiles. J. Proteome Res. 17, 2460–2469 (2018).
Di Maio, R. et al. α-Synuclein binds to TOM20 and inhibits mitochondrial protein import in Parkinson’s disease. Sci. Transl. Med. 8, 342ra78 (2016).
Pizarro-Galleguillos, B. M., Kunert, L., Brüggemann, N. & Prasuhn, J. Neuroinflammation and mitochondrial dysfunction in Parkinson’s disease: connecting neuroimaging with pathophysiology. Antioxidants (Basel, Switz.) 12, 1411 (2023).
Min, J. O., Strohäker, T., Jeong, B. C., Zweckstetter, M. & Lee, S. J. Chicago sky blue 6B inhibits α-synuclein aggregation and propagation. Mol. Brain 15, 27 (2022).
Yoo, H. C., Yu, Y. C., Sung, Y. & Han, J. M. Glutamine reliance in cell metabolism. Exp. Mol. Med. 52, 1496–1516 (2020).
Houten, S. M., Violante, S., Ventura, F. V. & Wanders, R. J. The biochemistry and physiology of mitochondrial fatty acid β-oxidation and its genetic disorders. Annu. Rev. Physiol. 78, 23–44 (2016).
Espinosa-Oliva, A. M. et al. Inflammatory bowel disease induces pathological α-synuclein aggregation in the human gut and brain. Neuropathol. Appl. Neurobiol. 50, e12962 (2024).
Jerber, J. et al. Population-scale single-cell RNA-seq profiling across dopaminergic neuron differentiation. Nat. Genet 53, 304–312 (2021).
Aran, D. et al. Reference-based analysis of lung single-cell sequencing reveals a transitional profibrotic macrophage. Nat. Immunol. 20, 163–172 (2019).
Miller, S. A. et al. LSD1 and aberrant DNA methylation mediate persistence of enteroendocrine progenitors that support BRAF-mutant colorectal cancer. Cancer Res. 81, 3791–3805 (2021).
Dann, E., Henderson, N. C., Teichmann, S. A., Morgan, M. D. & Marioni, J. C. Differential abundance testing on single-cell data using k-nearest neighbor graphs. Nat. Biotechnol. 40, 245–253 (2022).
Wagner, A. et al. Metabolic modeling of single Th17 cells reveals regulators of autoimmunity. Cell 184, 4168–4185.e21 (2021).
Gerez, J. A. et al. A cullin-RING ubiquitin ligase targets exogenous α-synuclein and inhibits Lewy body-like pathology. Sci. Transl. Med. 11, eaau6722 (2019).
Gessner, A. et al. A metabolomic analysis of sensitivity and specificity of 23 previously proposed biomarkers for renal transporter-mediated drug-drug interactions. Clin. Pharmacol. Therapeutics 114, 1058–1072 (2023).
Bierling, T. E. H. et al. GLUT1-mediated glucose import in B cells is critical for anaplerotic balance and humoral immunity. Cell Rep. 43, 113739 (2024).
Pang, Z. et al. MetaboAnalystR 4.0: a unified LC-MS workflow for global metabolomics. Nat. Commun. 15, 3675 (2024).
Hemel, I. M. G. M., Engelen, B. P. H., Luber, N. & Gerards, M. A hitchhiker’s guide to mitochondrial quantification. Mitochondrion 59, 216–224 (2021).
Acknowledgements
Funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – 505539112 – KFO 5024 (A01, A02, A04, B02, Z01). Further support came from the Bavarian Ministry of Science and the Arts in the framework of the ForInter network. The LC-MS system used for metabolomic analysis was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – INST 90/1048-1 FUGG. A.H. and D.B.B. were supported by the German Federal Ministry of Research, Technology and Space (BMFTR) – 031L0309A. M.S. and F.J.T.: Funded by the European Union (ERC, DeepCell − 101054957). Imaging was performed at the Optical Imaging Compentence Center Erlangen (OICE) using a DFG-funded microscope system (project number 522417173). P.G. was supported by the AI-PREDICT project (grant no. 01ZU2502) funded by the BMFTR, as well as by Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) through TRR 417 (Project-ID 540805631, Project S03) and SFB 1755 “CASCAID” (Project-ID 550296805, Project Z01). We thank the OICE staff, mainly Dr. Philipp Tripal and Dr. Benjamin Schmid for their technical support. We thank Daniel Beß for technical assistance with the tissue paraffin sections.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
B.G. designed the research, performed experiments, analyzed and interpreted the data, executed bioinformatics analyses, created the figures, and wrote the manuscript. L.E.G., V.R., C.J., E.G, A.G., J.G., N.C.P. performed experiments and analyzed the data. H.W. and M.F. maintained iPSC cultures. M.K., M.S. and A.H. performed bioinformatic analysis on the scRNAseq data. F.Z. generated the iPSC lines used in this study in the laboratory of J. R. Mazzulli, Northwestern University, Chicago. T.R., I.P., J.W., M.V., D.B.B., C.G., F.J.T., R.R. and M.N. provided resources and supervised the project. P.G. wrote the manuscript, analyzed and interpreted data and supervised the bioinformatics analyses. B.W. designed the research, provided resources, interpreted the data, wrote the manuscript, acquired funding and supervised the project. All authors critically reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
F.J.T. consults for Immunai Inc., CytoReason Ltd, Cellarity, BioTuring Inc., and Genbio.AI Inc., and has an ownership interest in Dermagnostix GmbH and Cellarity. The remaining authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Helle Bogetofte and the other, anonymous, reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ghirotto, B., Gonçalves, L.E., Ruder, V. et al. TNF alpha unmasks enteric malate aspartate shuttle dysfunction bridging Parkinson disease and intestinal inflammation. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71317-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71317-y