Abstract
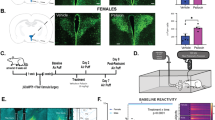
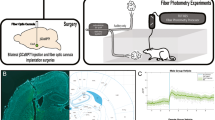
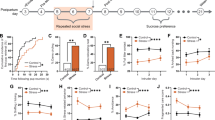
The psychedelic psilocybin has shown therapeutic potential, yet underlying neural mechanisms remain poorly understood. We investigated the impact of psilocin—the active metabolite of psilocybin—on basal activity and reactivity within the paraventricular nucleus of the thalamus (PVT) and PVT projections to central amygdala (CeA) in rats. Psilocin administration increased PVT c-Fos expression and selectively engaged PVT→CeA neurons in females, but not males. Psilocin enhanced PVT reactivity to an aversive air-puff stimulus, with effects primarily driven by passive responders. In PVT→CeA neurons, psilocin prevented time-dependent reductions in stimulus-evoked activity and maintained reactivity across timepoints in females but not males. The sustained engagement of PVT→CeA circuitry was driven by active responders. These findings identify sex-specific modulation of thalamic-limbic circuitry and behavior by psilocin, implicating PVT→CeA circuitry in the neural and behavioral effects of psychedelic compounds, advancing our understanding of how psychedelics modulate emotional brain circuits to further inform potential therapeutic mechanisms.
Similar content being viewed by others
Data availability
All data generated in this study, including a source data file containing raw data values for each figure, have been deposited in the Dryad database https://doi.org/10.5061/dryad.fbg79cp7b. Source data are provided with this paper.
Code availability
Custom-written MATLAB code and resulting MATLAB structures have been made available on the Dryad database https://doi.org/10.5061/dryad.fbg79cp7b.
References
Dinis-Oliveira, R. J. Metabolism of psilocybin and psilocin: clinical and forensic toxicological relevance. Drug Metab. Rev. 49, 84–91 (2017).
Hofmann, A. et al. Psilocybin und Psilocin, zwei psychotrope Wirkstoffe aus mexikanischen Rauschpilzen. Helv. Chim. Acta 42, 1557–1572 (1959).
Agrawal, M. et al. Assessment of psilocybin therapy for patients with cancer and major depression disorder. JAMA Oncol. 9, 864–866 (2023).
Bogenschutz, M. P. et al. Psilocybin-assisted treatment for alcohol dependence: a proof-of-concept study. J. Psychopharmacol. 29, 289–299 (2015).
Carhart-Harris, R. L. et al. Psilocybin with psychological support for treatment-resistant depression: six-month follow-up. Psychopharmacology 235, 399–408 (2018).
Carhart-Harris, R. L. et al. Psilocybin for treatment-resistant depression: fMRI-measured brain mechanisms. Sci. Rep. 7, 1–11 (2017).
Davis, A. K. et al. Effects of psilocybin-assisted therapy on major depressive disorder: a randomized clinical trial. JAMA Psychiatry 78, 481–489 (2021).
Goodwin, G. M. et al. Single-dose psilocybin for a treatment-resistant episode of major depression. N. Engl. J. Med. 387, 1637–1648 (2022).
Griffiths, R. R. et al. Psilocybin produces substantial and sustained decreases in depression and anxiety in patients with life-threatening cancer: a randomized double-blind trial. J. Psychopharmacol. 30, 1181–1197 (2016).
Johnson, M. W., Garcia-Romeu, A. & Griffiths, R. R. Long-term follow-up of psilocybin-facilitated smoking cessation. Am. J. Drug Alcohol Abus. 43, 55–60 (2017).
Johnson, M. W. & Griffiths, R. R. Potential therapeutic effects of psilocybin. Neurotherapeutics 14, 734–740 (2017).
Raison, C. L. et al. Single-dose psilocybin treatment for major depressive disorder: a randomized clinical trial. JAMA 330, 843–853 (2023).
Ross, S. et al. Rapid and sustained symptom reduction following psilocybin treatment for anxiety and depression in patients with life-threatening cancer: a randomized controlled trial. J. Psychopharmacol. 30, 1165–1180 (2016).
Barrett, F. S., Doss, M. K., Sepeda, N. D., Pekar, J. J. & Griffiths, R. R. Emotions and brain function are altered up to one month after a single high dose of psilocybin. Sci. Rep. 10, 1–14 (2020).
Barrett, F. S., Krimmel, S. R., Griffiths, R. R., Seminowicz, D. A. & Mathur, B. N. Psilocybin acutely alters the functional connectivity of the claustrum with brain networks that support perception, memory, and attention. NeuroImage 218, 116980 (2020).
Carhart-Harris, R. L. et al. Neural correlates of the psychedelic state as determined by fMRI studies with psilocybin. Proc. Natl. Acad. Sci. USA 109, 2138–2143 (2012).
Grimm, O., Kraehenmann, R., Preller, K., Seifritz, E. & Vollenweider, F. Psilocybin modulates functional connectivity of the amygdala during emotional face discrimination. Eur. Neuropsychopharmacol. 28, 691–700 (2018).
Kraehenmann, R. et al. The mixed serotonin receptor agonist psilocybin reduces threat-induced modulation of amygdala connectivity. NeuroImage Clin. 11, 53–60 (2016).
Mertens, L. J. et al. Therapeutic mechanisms of psilocybin: changes in amygdala and prefrontal functional connectivity during emotional processing after psilocybin for treatment-resistant depression. J. Psychopharmacol. 34, 167–180 (2020).
Preller, K. H. et al. Psilocybin induces time-dependent changes in global functional connectivity. Biol. Psychiatry 88, 197–207 (2020).
Roseman, L., Demetriou, L., Wall, M. B., Nutt, D. J. & Carhart-Harris, R. L. Increased amygdala responses to emotional faces after psilocybin for treatment-resistant depression. Neuropharmacology 142, 263–269 (2018).
Roseman, L., Leech, R., Feilding, A., Nutt, D. J. & Carhart-Harris, R. L. The effects of psilocybin and MDMA on between-network resting state functional connectivity in healthy volunteers. Front. Hum. Neurosci. 8, 204 (2014).
Hwang, K., Bertolero, M. A., Liu, W. B. & D’Esposito, M. The human thalamus is an integrative hub for functional brain networks. J. Neurosci. 37, 5594–5607 (2017).
Shine, J. M., Lewis, L. D., Garrett, D. D. & Hwang, K. The impact of the human thalamus on brain-wide information processing. Nat. Rev. Neurosci. 24, 416–430 (2023).
Jones, E. The Thalamus 2nd edn (Cambridge University Press, 2007).
Halassa, M. M. The Thalamus (Cambridge University Press, 2022).
Sherman, S. M. & Guillery, R. W. Functional Connections of Cortical Areas: A New View from the Thalamus (MIT Press, 2013).
Barson, J. R., Mack, N. R. & Gao, W.-J. The paraventricular nucleus of the thalamus is an important node in the emotional processing network. Front. Behav. Neurosci. 14, 598469 (2020).
Kirouac, G. J. Placing the paraventricular nucleus of the thalamus within the brain circuits that control behavior. Neurosci. Biobehav. Rev. 56, 315–329 (2015).
Penzo, M. A. & Gao, C. The paraventricular nucleus of the thalamus: an integrative node underlying homeostatic behavior. Trends Neurosci. 44, 538–549 (2021).
Arluison, M. et al. Demonstration of peptidergic afferents to the bed nucleus of the stria terminalis using local injections of colchicine. A combined immunohistochemical and retrograde tracing study. Brain Res. Bull. 34, 319–337 (1994).
Csaki, A., Kocsis, K., Halasz, B. & Kiss, J. Localization of glutamatergic/aspartatergic neurons projecting to the hypothalamic paraventricular nucleus studied by retrograde transport of [3H] D-aspartate autoradiography. Neuroscience 101, 637–655 (2000).
Parsons, M. P., Li, S. & Kirouac, G. J. Functional and anatomical connection between the paraventricular nucleus of the thalamus and dopamine fibers of the nucleus accumbens. J. Comp. Neurol. 500, 1050–1063 (2007).
Dong, X., Li, S. & Kirouac, G. J. Collateralization of projections from the paraventricular nucleus of the thalamus to the nucleus accumbens, bed nucleus of the stria terminalis, and central nucleus of the amygdala. Brain Struct. Funct. 222, 3927–3943 (2017).
Li, S. & Kirouac, G. J. Projections from the paraventricular nucleus of the thalamus to the forebrain, with special emphasis on the extended amygdala. J. Comp. Neurol. 506, 263–287 (2008).
Huang, H., Ghosh, P. & van den Pol, A. N. Prefrontal cortex–projecting glutamatergic thalamic paraventricular nucleus-excited by hypocretin: a feedforward circuit that may enhance cognitive arousal. J. Neurophysiol. 95, 1656–1668 (2006).
Ma, J. et al. Divergent projections of the paraventricular nucleus of the thalamus mediate the selection of passive and active defensive behaviors. Nat. Neurosci. 24, 1429–1440 (2021).
Do-Monte, F. H., Quinones-Laracuente, K. & Quirk, G. J. A temporal shift in the circuits mediating retrieval of fear memory. Nature 519, 460–463 (2015).
Penzo, M. A. et al. The paraventricular thalamus controls a central amygdala fear circuit. Nature 519, 455–459 (2015).
Pliota, P. et al. Stress peptides sensitize fear circuitry to promote passive coping. Mol. Psychiatry 25, 428–441 (2020).
Spencer, S. J., Fox, J. C. & Day, T. A. Thalamic paraventricular nucleus lesions facilitate central amygdala neuronal responses to acute psychological stress. Brain Res. 997, 234–237 (2004).
Pizzi, S. D. et al. LSD-induced changes in the functional connectivity of distinct thalamic nuclei. NeuroImage 283, 120414 (2023).
Effinger, D. P., Quadir, S. G., Ramage, M. C., Cone, M. G. & Herman, M. A. Sex-specific effects of psychedelic drug exposure on central amygdala reactivity and behavioral responding. Transl. Psychiatry 13, 119 (2023).
Aboharb, F. et al. Classification of psychedelics and psychoactive drugs based on brain-wide imaging of cellular c-Fos expression. Nat. Commun. 16, 1590 (2025).
Davoudian, P. A., Shao, L.-X. & Kwan, A. C. Shared and distinct brain regions targeted for immediate early gene expression by ketamine and psilocybin. ACS Chem. Neurosci. 14, 468–480 (2023).
Kiilerich, K. F. et al. Repeated low doses of psilocybin increase resilience to stress, lower compulsive actions, and strengthen cortical connections to the paraventricular thalamic nucleus in rats. Mol. Psychiatry 9, 1–13 (2023).
Effinger, D. P. et al. Increased reactivity of the paraventricular nucleus of the hypothalamus and decreased threat responding in male rats following psilocin administration. Nat. Commun. 15, 5321 (2024).
Gruene, T. M., Flick, K., Stefano, A., Shea, S. D. & Shansky, R. M. Sexually divergent expression of active and passive conditioned fear responses in rats. Elife 4, e11352 (2015).
Choi, E. A., Jean-Richard-dit-Bressel, P., Clifford, C. W. & McNally, G. P. Paraventricular thalamus controls behavior during motivational conflict. J. Neurosci. 39, 4945–4958 (2019).
Kato, T. M. et al. Presynaptic dysregulation of the paraventricular thalamic nucleus causes depression-like behavior. Sci. Rep. 9, 16506 (2019).
Zhao, D. et al. The paraventricular thalamus input to central amygdala controls depression-related behaviors. Exp. Neurol. 342, 113744 (2021).
Zhou, K. & Zhu, Y. The paraventricular thalamic nucleus: a key hub of neural circuits underlying drug addiction. Pharmacol. Res. 142, 70–76 (2019).
Hartmann, M. C. & Pleil, K. E. Circuit and neuropeptide mechanisms of the paraventricular thalamus across stages of alcohol and drug use. Neuropharmacology 198, 108748 (2021).
Hill-Bowen, L. D., Flannery, J. S. & Poudel, R. Paraventricular thalamus activity during motivational conflict highlights the nucleus as a potential constituent in the neurocircuitry of addiction. J. Neurosci. 40, 726–728 (2020).
Lawlor, V. M. et al. Dissecting the impact of depression on decision-making. Psychol. Med. 50, 1613–1622 (2020).
Harlé, K. M., Allen, J. J. & Sanfey, A. G. The impact of depression on social economic decision making. J. Abnorm. Psychol. 119, 440 (2010).
Paulus, M. P. & Angela, J. Y. Emotion and decision-making: affect-driven belief systems in anxiety and depression. Trends Cogn. Sci. 16, 476–483 (2012).
Dom, G., Sabbe, B., Hulstijn, W. & Van Den Brink, W. Substance use disorders and the orbitofrontal cortex: systematic review of behavioural decision-making and neuroimaging studies. Br. J. Psychiatry 187, 209–220 (2005).
Chen, S. et al. Risky decision-making in individuals with substance use disorder: a meta-analysis and meta-regression review. Psychopharmacology 237, 1893–1908 (2020).
Flynn, M. & Rudolph, K. D. The contribution of deficits in emotional clarity to stress responses and depression. J. Appl. Dev. Psychol. 31, 291–297 (2010).
Stikkelbroek, Y., Bodden, D. H., Kleinjan, M., Reijnders, M. & van Baar, A. L. Adolescent depression and negative life events, the mediating role of cognitive emotion regulation. PLoS ONE 11, e0161062 (2016).
Kober, H. & Bolling, D. Emotion regulation in substance use disorders. Handb. Emot. Regul. 2, 428–446 (2014).
Jones, E. G. The Thalamus (Springer Science & Business Media, 2012).
Temereanca, S. & Simons, D. J. Functional topography of corticothalamic feedback enhances thalamic spatial response tuning in the somatosensory whisker/barrel system. Neuron 41, 639–651 (2004).
Perry, B. A., Mendez, J. C. & Mitchell, A. S. Cortico-thalamocortical interactions for learning, memory and decision-making. J. Physiol. 601, 25–35 (2023).
Brown, E. C., Clark, D. L., Hassel, S., MacQueen, G. & Ramasubbu, R. Thalamocortical connectivity in major depressive disorder. J. Affect. Disord. 217, 125–131 (2017).
Kerr, C. C., Kemp, A. H., Rennie, C. J. & Robinson, P. A. Thalamocortical changes in major depression probed by deconvolution and physiology-based modeling. Neuroimage 54, 2672–2682 (2011).
Ma, J. et al. Convergent direct and indirect cortical streams shape avoidance decisions in mice via the midline thalamus. Nat. Commun. 15, 6598 (2024).
Vollenweider, F. X. et al. Positron emission tomography and fluorodeoxyglucose studies of metabolic hyperfrontality and psychopathology in the psilocybin model of psychosis. Neuropsychopharmacology 16, 357–372 (1997).
Gouzoulis-Mayfrank, E. et al. Neurometabolic effects of psilocybin, 3, 4-methylenedioxyethylamphetamine (MDE) and d-methamphetamine in healthy volunteers: a double-blind, placebo-controlled PET study with [18F] FDG. Neuropsychopharmacology 20, 565–581 (1999).
Lewis, C. R. et al. Two dose investigation of the 5-HT-agonist psilocybin on relative and global cerebral blood flow. Neuroimage 159, 70–78 (2017).
Mitchell, J. R. et al. Darting across space and time: parametric modulators of sex-biased conditioned fear responses. Learn. Mem. 29, 171–180 (2022).
Engelke, D. et al. A hypothalamic-thalamostriatal circuit that controls approach-avoidance conflict in rats. Nat. Commun. 12, 2517 (2021).
Bangasser, D. A. et al. Sex differences in corticotropin-releasing factor receptor signaling and trafficking: potential role in female vulnerability to stress-related psychopathology. Mol. Psychiatry 15, 896–904 (2010).
Hammack, S. E. et al. Roles for pituitary adenylate cyclase-activating peptide (PACAP) expression and signaling in the bed nucleus of the stria terminalis (BNST) in mediating the behavioral consequences of chronic stress. J. Mol. Neurosci. 42, 327–340 (2010).
Hashimoto, H. et al. PACAP is implicated in the stress axes. Curr. Pharm. Des. 17, 985–989 (2011).
Curtis, G. R. et al. Sex-related differences in endogenous pituitary adenylate cyclase-activating polypeptide (PACAP) in the thalamic paraventricular nucleus: implications for addiction neuroscience. Addict. Neurosci. 5, 100058 (2023).
Hayata-Takano, A. et al. PACAP–PAC1 signaling regulates serotonin 2A receptor internalization. Front. Endocrinol. 12, 732456 (2021).
Halberstadt, A. L. & Geyer, M. A. Multiple receptors contribute to the behavioral effects of indoleamine hallucinogens. Neuropharmacology 61, 364–381 (2011).
Halberstadt, A. L. & Geyer, M. A. Characterization of the head-twitch response induced by hallucinogens in mice: detection of the behavior based on the dynamics of head movement. Psychopharmacology 227, 727–739 (2013).
Halberstadt, A. L., Chatha, M., Klein, A. K., Wallach, J. & Brandt, S. D. Correlation between the potency of hallucinogens in the mouse head-twitch response assay and their behavioral and subjective effects in other species. Neuropharmacology 167, 107933 (2020).
Acknowledgements
This work was supported by National Institute of Health grants AA026858 (M.A.H.), GM135095 (D.P.E.), AA007573 (S.G.Q.), AA030493 (S.G.Q.), and by the Brain and Behavior Research Foundation NARSAD Young Investigator Award (M.A.H.).
Author information
Authors and Affiliations
Contributions
D.P.E. contributed to the initial conception, experimental design, apparatus design, surgical procedures, behavioral recording, histology, data analysis, custom code, data visualization, the initial draft of the manuscript, and manuscript editing. J.L.H. contributed to the behavioral recording, behavioral analysis, a section in methods, and manuscript editing. S.G.Q. contributed to histological analyses, schematic illustrations, and manuscript editing. C.S.R. contributed to histology. D.T. contributed to histology. M.E.S. contributed to histology. M.W.H. contributed to histology. C.W.H. contributed to the behavioral recording and manuscript editing. M.A.H. contributed to the initial conception, experimental design, behavioral assay, and acquisition of funds.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Mikael Palner and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Effinger, D.P., Hoffman, J.L., Quadir, S.G. et al. Sex-specific increased reactivity of the PVT and prolonged PVT→CeA circuit engagement following psilocin administration. Nat Commun (2026). https://doi.org/10.1038/s41467-026-71481-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-026-71481-1