Abstract
Major Depressive Disorder (MDD) affects around 20% of people globally and is often comorbid with anxiety. This study investigates prucalopride, a serotonin type 4 receptor (5-HT4R) agonist approved for constipation, as a fast-acting anxiolytic/antidepressant using a mouse model of stress, based on corticosterone (CORT) administration. Behavioral effects of prucalopride (0.5 and 1.5 mg/kg/day) were compared to fluoxetine, a common SSRI, over 7 (subchronic) and 28 (chronic) days. Prucalopride showed faster and more significant improvements in emotionality scores than fluoxetine, reversing CORT-induced behavioral changes within 7 days. Gut microbiota analysis revealed CORT-induced changes at the subchronic timepoint. While chronic prucalopride did not alter microbial alpha diversity, it significantly shifted microbial composition (beta-diversity). Notably, prucalopride restored levels of the genus Ruminococcus, which were depleted by CORT. Our findings highlight prucalopride’s rapid anxiolytic and antidepressant-like effects and its impact on gut microbiota, supporting the potential of 5-HT4R-targeting molecules as therapeutic options for psychiatric disorders.
Similar content being viewed by others
Introduction
Major Depressive Disorder (MDD) is one of the most common mental health disorders, affecting approximately 20% of individuals worldwide at some point in their lives1. During Major Depressive Episodes (MDE), about 2/3 of patients experience significant levels of anxiety1. Current treatment options for MDE, especially when comorbid with anxiety, face several challenges. Monoaminergic antidepressants for instance, show a delayed onset of action leaving patients vulnerable during the critical early weeks of treatment2. Benzodiazepines, rapid-acting GABAergic anxiolytics, primarily address anxiety but provide minimal relief for depressive symptoms3. Furthermore, their use is associated with significant adverse effects, including sedation, cognitive impairment, and a high risk of dependence4. Therefore, there is a pressing need for the innovation and development of new treatments that are both fast-acting and safe, representing a crucial step forward for public health.
Recent studies have identified the serotonin type 4 receptor (5-HT4R) as a promising target for treating depression and anxiety5,6,7. This metabotropic receptor is primarily expressed in the limbic system of both primates and rodents, including the hypothalamus, hippocampus, nucleus accumbens, the amygdala and the frontal cortex, consistent with its role in emotions regulation [for review5]. In clinical settings, a 5-HT4R genetic polymorphism has been associated with reduced response to conventional antidepressants8. Additionally, positron emission tomography scanning has highlighted a reduced expression of 5-HT4R in individuals diagnosed with MDD compared to healthy controls9. Overall, these findings emphasize the critical role of 5-HT4R in the pathophysiology of MDD and provide the fundamental bases to support ongoing research into 5-HT4R agonists as a promising direction for developing more effective treatments in the context of mood disorders. In this context, we have recently shown that a 5-HT4R agonist, RS67333, exhibits rapid anxiolytic and antidepressant effects in mouse models5,6,7. Nonetheless, studying these effects in humans has been constrained by the adverse side effects. Interestingly, prucalopride, a selective and a high-affinity 5-HT4R partial agonist (pKi=8.610), with good brain penetration11, is approved by both the Food and Drug Administration and the European Medicines Agency for the treatment of chronic idiopathic constipation (CIC)10. A recent study utilizing anonymized data from US electronic health records found that treatment with prucalopride was associated with significantly lower incidence of depression over the following year compared to two alternative anti-constipation medications12. In rodents, while a previous study demonstrated prucalopride’s prophylactics effect against stress13 and increases firing of serotonin (5-HT) neurons, a mechanism likely contributing to its antidepressant activity, its efficacy in a mouse model of anxiety/depression has not yet been implemented14.
In addition to its expression in the brain, 5-HT4Rs are highly abundant in the gastrointestinal tract, where they play a key role in regulating gut motility, secretion, and sensory functions15,16. As increasing evidence is highlighting a role for the microbiota-gut-brain axis in psychiatric disorders17,18, the question arises as to whether prucalopride might exert potential antidepressant/anxiolytic effects via the gut-brain axis. No studies to date have explored the effects of prucalopride on microbiota composition.
To address this gap in knowledge, the present study aimed to characterize the effects of prucalopride in the chronic corticosterone (CORT) stress mouse model, focusing on both central and peripheral outcomes. The central readout involved behavioral screening of emotion-related responses, while the peripheral investigation centered on the gut microbiota using 16S sequencing. This technique targets the 16S ribosomal RNA (rRNA) gene, which is highly conserved and present in all bacteria, making it ideal for identifying and characterizing each microorganism19. Observations along the gut-brain axis were conducted at two distinct timepoints, allowing a comparison of subchronic versus chronic effects. Overall, this research has the potential to provide novel insights, paving the way for future clinical investigations into the therapeutic role of 5-HT4R-targeting molecules for the treatment of MDD.
Results
Corticosterone administration increases plasma corticosterone levels and impacts the gut microbiota composition
To confirm the biological validity of our model, we measured circulating CORT levels following 5 and 8 weeks of CORT administration in the drinking water. Consistent with previous findings20, the CORT/Veh group showed an increase in CORT levels after 5 and 8 weeks of treatment compared to Veh-treated group. This increase was reversed in all other treatment groups (CORT/Flx, CORT/Pruca0.5, CORT/Pruca1.5; Fig. S2). As previously described, CORT treatment increased anxiety- and depressive-like behavior as observed with the increase in emotionality score after 5 and 8 weeks of treatment7,20,21. Indeed, at both time points, in the EPM, CORT decreased the time spent and the number of entries in the open arms the ratio of ambulatory distance in the open arms divided by total distance (Fig. 1A, B). Moreover, chronic CORT decreased the grooming duration in the ST (Fig. 1D) and increased latency to feed in the NSF (Fig. 1E, F).
While most research to date has focused on the microbiota’s ability to regulate CORT levels22,23, the impact of chronic CORT supplementation on the gut microbiota itself remains underexplored. To this end, we compared specifically the gut microbiota of Veh-treated and CORT-treated mice. CORT administration induced distinct, time-dependent alterations in the gut microbiota, as detailed in Fig. S3. Specifically, the Chao1 index (a species richness estimator that considers both the observed species and the contribution of rare species in the community) for alpha-diversity was significantly decreased by CORT after 5 weeks but not 8 weeks of treatment. Also, CORT-receiving mice clustered apart from Veh mice on the beta-diversity Bray-Curtis matrix, independent of the timepoint. The genera Clostridia UCG014 and Ruminococcus were significantly depleted by CORT supplementation at both timepoints.
The 5-HT4R agonist prucalopride induces differential dose-related anxiolytic/antidepressant effects following both subchronic and chronic treatments
To assess the potential fast anxiolytic/antidepressant effect of prucalopride, a battery of behavioral tests was conducted after 3 days of subchronic treatment. In the EPM, prucalopride at both doses significantly increased the time spent and the number of entries in the open arms, highlighting an anxiolytic-like effect, which was not observed for fluoxetine (Fig. 1A, B). The ratio of ambulatory distance in the open arms divided by total distance was increased by both doses of prucalopride, indicating an anxiolytic-like effect (Fig. 1C). The ST revealed a significant increase in grooming duration for both doses of prucalopride, an effect also observed in the fluoxetine group (Fig. 1D). Subsequently, we evaluated these mice in the NSF test to measure hyponeophagia, an index of anxiolytic/antidepressant-like activity. Prucalopride, at both tested doses, significantly decreased the latency to feed (Fig. 1E, F) without affecting the home-cage food consumption (Fig. S4). The NSF effect seemed independent of bodyweight changes across groups, which were not significant (Fig. S5), suggesting no changes in feeding behavior. Overall, prucalopride at both doses normalized the emotionality score increase induced by chronic CORT after a week of treatment (Fig. 1G). Notably, at the lowest dose tested, prucalopride elicited a more pronounced anxiolytic/antidepressant-like effect.
The same behavioral tests were administered following chronic treatment. In the EPM, prucalopride (1.5 but not 0.5 mg/kg) exhibited an anxiolytic effect (Fig. 1H–J). This effect was evident across several measures, including time spent in the open arms, entries into the open arms, and the ratio of ambulatory distance in the open arms to total distance. These results were comparable to those observed with fluoxetine. The ST revealed a dose-dependent significant increase in grooming duration for prucalopride as compared to Veh/CORT mice (Fig. 1K). In the NSF, both doses of prucalopride reduce the latency time to reach the food pellet (Fig. 1L, M) without affecting home-cage food consumption (Fig. S4). Overall, the anxiolytic/antidepressant-like effects observed after a week of treatment with prucalopride was sustained following chronic administration, as evidenced by the normalization of the emotionality score at both tested doses (Fig. 1N). Additionally, we confirmed that chronic treatment with fluoxetine reversed chronic CORT-induced increase in emotionality score23.
Effects of a subchronic (A–G) or chronic (H–N) prucalopride (Pruca0.5, Pruca1.5) or fluoxetine (Flx) administration on corticosterone (CORT) -induced anxiety-like behaviors in the Elevated Plus Maze (EPM). Anxiety is expressed as mean time spent in seconds (A or H) or number of entries (B or I) in the open arms or as mean ratio of ambulatory distance in the center/total ambulatory distance (C or J). Effect of a subchronic (D) or chronic (K) prucalopride (Pruca0.5, Pruca1.5) or fluoxetine (Flx) administration on corticosterone (CORT)-induced depression-related behaviors in the Splash Test (ST). Depression-related behavior is expressed as mean duration of grooming in seconds (D or K). Effects of a subchronic (E, F) or chronic (L, M) prucalopride (Pruca0.5, Pruca1.5) or fluoxetine (Flx) administration on corticosterone (CORT) -induced anxiety and depression-like behaviors in the Novelty Suppressed Feeding (NSF) paradigm. Results are cumulative survival of animals that have not eaten over 10 min (E or L) or mean of latency to feed in seconds (F or M). Effects of a subchronic (G) or chronic (N) prucalopride (Pruca0.5, Pruca1.5) or fluoxetine (Flx) administration on corticosterone (CORT)-induced anxiety/depression-like phenotype. Test Z-values (Elevated Plus maze, Splash test and Novelty Suppressed Feeding) are then calculated by averaging individual Z-scores and averaged to obtain emotionality Z-score. Data are expressed as mean ± SEM (Veh/Veh n = 5, CORT/Veh n = 9, CORT/Flx n = 9–10, CORT/Pruca0.5 n = 9, CORT/Pruca1.5 n = 9). *p < 0.05; **p < 0.01; ***p < 0.001 versus appropriate group.
Following a subchronic treatment gut microbiota shifts are not prucalopride-dependent but likely linked to the depressive- and anxious-like phenotype (CORT model)
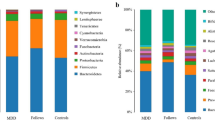
Two distinct timepoints, corresponding to behavioral assessments, were selected for microbiota compositional analysis. First, the gut microbiota was characterized after 1 week of fluoxetine or prucalopride treatment. At this timepoint, genus level analysis revealed no changes in alpha-diversity metrics (Fig. 2A), which reflects the diversity within each group. Specifically, neither Chao1, which focuses on the number of species, nor Shannon, which accounts for both species’ richness and evenness, were affected. However, beta-diversity analysis, which refers to the comparison of microbial composition between different environments, indicated that the microbiota composition in CORT-treated mice, regardless of fluoxetine or prucalopride administration, was significantly different from that of Veh/Veh controls (Fig. 2B). At the individual genus level, we observed a significant reduction in Clostridia UCG014, Intestinimonas, UBA1819, and Incertae Sedis in all CORT-treated groups compared to the Veh-treated group. Additionally, Muribaculum was specifically increased in the CORT/Flx and CORT/Pruca0.5 relative to Veh/Veh mice. Ruminococcus was depleted in CORT/Veh and CORT/Pruca1.5 groups as compared to Veh/Veh and was restored in the CORT/Flx and CORT/Pruca0.5 groups relative to the CORT/Veh group (Fig. 2C).
A Alpha-diversity: Kruskal-Wallis test for Chao1 and Shannon not significant. B Beta-diversity, principal coordinate analysis of Bray-Curtis compiled distance matrix of all genera. C Microbiota changes at genus level. *p < 0.05 vs Veh/Veh and $p < 0.05 vs CORT/Veh. Data are expressed as median + min-to-max values (Veh/Veh n = 4, CORT/Veh n = 9, CORT/Flx n = 10, CORT/Pruca0.5 n = 10, CORT/Pruca1.5 n = 9). Data is analyzed using Kruskal-Wallis non-parametric test followed by Mann–Whitney U-test and corrected for multiple comparisons using the Benjamin-Hochberg false discovery rate (FDR) method.
Following a chronic administration the genus Ruminococcus is restored in prucalopride-receiving mice
After 28 days of fluoxetine or prucalopride treatment, microbiota composition was re-analyzed. Consistent with the findings from the subchronic timepoint, no changes in alpha-diversity metrics were detected (Fig. 3A). However, beta-diversity analysis using the Bray-Curtis index revealed the same CORT-related effect as noted at earlier timepoint (compared to Veh/Veh), along with distinct separation between the CORT/Flx and CORT/Pruca1.5 groups, as well as between the CORT/Pruca0.5 and CORT/Pruca1.5 groups (Fig. 3B). At the genus level, a significant reduction in UBA1819 was observed across all CORT-treated groups compared to Veh-treated group. Lactobacillus levels were specifically increased in the CORT/Veh, CORT/Pruca0.5, and CORT/Pruca1.5 groups relative to Veh/Veh mice. The same genus was also increased in the CORT/Pruca1.5 compared to the CORT/Pruca0.5 mice. Ruminococcus remained depleted in the CORT/Veh group but was restored following chronic prucalopride treatment at both doses, an effect not observed with fluoxetine (Fig. 3C). These data suggest a prucalopride-dependent effect in restoring Ruminococcus levels.
A Alpha-diversity: Kruskal-Wallis test for Chao1 and Shannon not significant. B Beta-diversity, principal coordinate analysis of Bray-Curtis compiled distance matrix of all genera. C Microbiota changes at genus level. *p < 0.05 vs Veh/Veh and $p < 0.05 vs CORT/Veh. Data are expressed as median + min-to-max values (Veh/Veh n = 4, CORT/Veh n = 7, CORT/Flx n = 8, CORT/Pruca0.5 n = 8, CORT/Pruca1.5 n = 7). Data is analyzed using Kruskal-Wallis non-parametric test followed by Mann–Whitney U-test and corrected for multiple comparisons using the Benjamin-Hochberg false discovery rate (FDR) method.
Discussion
Here, using a mouse stress model, we sought to evaluate prucalopride, a clinically approved 5-HT4R agonist, as a rapid anxiolytic/antidepressant and to assess its effects on the gut microbiota. These data indicate that prucalopride produced rapid and robust anxiolytic- and antidepressant-like effects compared to fluoxetine, normalizing CORT-induced emotional alterations within a week. In parallel, CORT exposure impacted gut microbiota composition, and prucalopride selectively modulated beta-diversity without affecting alpha-diversity. Notably, prucalopride consistently restored Ruminococcus levels, highlighting this genus as a potential mediator of the treatment’s gut–brain axis effects.
Current antidepressant treatments face a high rate of non-responsiveness among patients24, a delayed onset of therapeutic effects, and a range of undesirable side effects25 underscoring the critical need for the development of novel antidepressants26. Although benzodiazepines, which enhance γ-aminobutyric acid (GABA) neurotransmission27,28, have proven effective in the treatment of most anxiety disorders, their long-term daily use is associated with side effect. Consequently, they are often replaced by monoaminergic antidepressants such as selective serotonin reuptake inhibitors (SSRIs), highlighting the central role of serotonin not only in depression but also in anxiety27,28. Emerging evidence6,7,29,30,31, along with the present findings, confirm that 5-HT4R agonists act as rapid anxiolytics and antidepressants. Our previous research demonstrated that RS-67,333, a 5-HT4R partial agonist, could induce rapid anxiolytic effects comparable to those of diazepam6 and achieve faster antidepressant-like effects than fluoxetine7. However, while the potential for investigating RS-67,333 in humans is constrained by its associated adverse side effects, prucalopride, a selective high-affinity partial agonist of the 5-HT4R with excellent brain penetration11, offers a promising alternative. As previously mentioned, prucalopride is currently approved for the treatment of CIC10 and a recent study based on electronic health records showed prucalopride’s association with reduced likelihood of experiencing a first depressive episode12. In our study, while a subchronic treatment with fluoxetine or prucalopride induced anxiolytic/antidepressant-like activity in the NSF and ST, only the 5-HT4R agonist resulted in anxiolytic-like activity in the EPM. Subchronic treatment with prucalopride effectively reversed the CORT-induced increase in emotionality after just one week of administration at both tested doses. Notably, the anxiolytic and antidepressant-like effects were more pronounced at the lower dose compared to those observed in fluoxetine-treated animals. A longer duration of treatment (28 days) was required for fluoxetine to exert anxiolytic-like effects comparable to 7 days of prucalopride treatment. Our study further confirmed that the anxiolytic and antidepressant-like effects observed following chronic 5-HT4R activation are comparable to those achieved with fluoxetine. To our knowledge, the present study is the first to clearly demonstrate fast anxiolytic-like activity of prucalopride in a mouse model of stress and provide novel insights on the potential repurposing of prucalopride for anxiety and depressive disorders.
Considering that prucalopride is used for the treatment of CIC10 and that 5-HT4Rs are highly abundant in the GI tract32, we explored the fast antidepressant/anxiolytic effects of the 5-HT4R agonist prucalopride and its effects on the gut microbiota, but also microbiota-targeting effects of chronic CORT, in the context of the gut-brain axis. Extensive human cohort studies have highlighted significant disruptions in gut microbiota composition associated with neuropsychiatric disorders, such as anxiety disorders and MDD33. Specifically, the gut microbiota plays an important role in the gut-brain axis, influencing depression through multiple pathways including the hypothalamic-pituitary-adrenal (HPA) axis. Interestingly, there seems to be a bidirectional link between the gut microbiota and the HPA axis, which can ultimately result in shifts in CORT levels. Stress-induced glucocorticoid release is modulated by the gut microbiota; for example, germ-free (GF) mice display stress-induced exaggerated glucocorticoid levels that can be recovered upon colonization with specific pathogen-free (SPF) feces34,35. Corroborating and expanding these findings, a recent paper showed that microbial depletion dysregulates not only glucocorticoid levels but also their circadian rhythmicity22. While the role of the gut microbiota on stress response is becoming more and more evident, few studies to date have looked at the effects of CORT administration on the gut microbiota. We took advantage of the chronic CORT stress model to explore the gut-targeting effects of CORT supplementation. Our results revealed specific time-point-dependent changes in microbial composition. At 5 weeks, the Chao1 index for alpha-diversity was decreased by CORT, and the beta-diversity clearly showed two separate clusters for vehicle versus CORT mice. Several taxa were also depleted by CORT. At 8 weeks, however, there were no differences in alpha-diversity and the separation between groups on the Bray-Curtis beta-diversity plot was less evident. At this timepoint, some individual changes at the genus level persisted (i.e., a decrease in Clostridia UCG014 and Ruminococcus) and others appeared. A recent study showed that a chronic exposure to CORT via i.p. in male C57BL6/J mice induced not only anxiety- and depressive-like behaviors, but also specific changes in the gut microbiota, such as increases in Lactobacillus, Bacillus, and Bifidobacterium36. It is interesting to note that we also reported an increase in Lactobacillus, but no similarities were found regarding the other two genera. The CORT administration protocol differed in terms of dose, duration, and route, which may account for variations in microbiota composition observed between studies. Overall, these findings show that CORT supplementation can impact not only circulating levels of CORT, as expected, but also the gut microbiota composition in a duration-dependent manner.
Recent findings emphasize that antidepressants can markedly alter the gut microbiota, with their effects surpassing those associated with the diagnosis of neuropsychiatric disorders alone37. Among psychotropic drugs, antidepressants’ ability to target the microbiota has been extensively studied both in vitro and in vivo [for review38]. Specifically, the SSRIs escitalopram and fluoxetine showed differential antimicrobial activity in vitro against E.coli and L. rhamnosus, two bacteria residing in the human gut39, but also against Staphylococcus and Enterococcus40,41. Consistent with these findings, it is not surprising to observe a fluoxetine-related effect on the gut microbiota, whether after subchronic or chronic treatment, even though the effect of the CORT model seems to be prevalent. Although SSRIs do not directly target serotonin receptors, they are considered indirect agonists; they might therefore act similarly to prucalopride in shifting 5-HT intestinal transmission and, in turn, microbial composition.
We next focused on prucalopride’s distal effects. While at the subchronic timepoint we mostly observed CORT-induced changes, after chronic administration, despite the absence of changes in global alpha-diversity metrics, we saw a specific CORT-induced depletion in the genus Ruminococcus which was restored following prucalopride treatment at both tested doses. Functionally, bacteria belonging to this genus serve to degrade and convert complex polysaccharides into a variety of nutrients for their host42. Interestingly, Ruminococcus was found to be depleted in rodent models of depression other than the CORT model, including the chronic unpredictable mild stress (CUMS) model43, the maternal separation model44, and the social defeat (SD) model45, suggesting that its depletion might be independent of the causal factor and stable across models. Conversely recent preclinical findings46,47 showed that (i) Ruminococcus was decreased following conventional antidepressant treatment (fluoxetine, duloxetine) and (ii) introduction in male BALB/c OlaHsd mice of a single Ruminococcus species, R. flavefaciens, induced cortical transcriptional changes, including the up-regulation of genes related to mitochondrial oxidative phosphorylation and down-regulation of genes associated with neuronal plasticity, which were interpreted as counteracting the antidepressant response. Taken together, these seemingly divergent results raise important questions about the genus Ruminococcus and its strain-specific effects on host physiology and antidepressant efficacy. One possibility is that different Ruminococcus strains may exert opposite influences on behavioral outcomes, depending on the pharmacological context and host state (e.g., chronic stress vs. pharmacological antidepressant exposure). Alternatively, prucalopride may engage gut-brain signaling pathways that leverage Ruminococcus restoration in a manner distinct from classical monoaminergic antidepressants. In our study, several other taxa were affected by subchronic and chronic treatments, but the observed shifts did not seem to be prucalopride-specific. A limitation of the current design is the absence of fluoxetine- and prucalopride-only groups. This restricts our ability to distinguish drug-specific microbiome effects from those occurring only in the context of chronic CORT exposure. Future studies incorporating these groups are essential to fully tease out treatment-specific versus stress-modulatory effects on the gut microbiota. It is also important to note that this type of grouped housing may introduce microbiome-related cage effects, which is a limitation of the current study. Although our current findings on Ruminococcus remain observational and do not supply functional readouts, they provide nonetheless a basis for future research aimed at exploring the potential causal role of this genus in prucalopride-induced fast anxiolytic/antidepressant effects. Importantly, these preclinical observations are supported by clinical evidence. To date, seven clinical studies have reported a depletion of Ruminococcus in depressed patients relative to healthy controls, although two studies observed an increase in this genus48. Discrepancies may stem from confounding factors such as diet and lifestyle. Notably, one clinical study reported that Ruminococcus depletion was specific to “severely depressed” patients49, suggesting that its reduction may be linked to disease severity. Taken together, the convergence of preclinical and clinical data highlights Ruminococcus as a potentially relevant genus in both depression pathophysiology and antidepressant response, warranting further investigation.
Overall, this study shows concomitant changes in the gut microbiota and behavioral outcomes relevant to depression and anxiety. It is important to highlight certain limitations of this study, some of which could be addressed in future experiments. First, 16S sequencing is limited to the genus levels as compared to a whole genome shotgun sequencing, which would further allow the functional classification of the microbiota and Ruminococcus-associated metabolic pathways. Secondly, prucalopride, a drug used to treat CIC, could induce GI side effects like diarrhea in mice, but this was not the case in our experiment. However, deeper measures of intestinal permeability could be useful in future experiments, especially considering that microbiota plays a role in regulating intestinal barrier function50 and GI comorbidities can often occur together with psychiatric disorders51. The absence of gut motility outcomes represent a limitation that should be addressed in future studies. In addition, prucalopride was administered via osmotic minipumps: the question arises as to whether an oral administration, which directly reaches the GI tract, would differently affect the microbiota of the mice; therefore, comparing different routes of administration would be informative. Finally, isoflurane anesthesia can impact the microbiota composition52, a factor that was not accounted for and represents a limitation of the present study. Future studies would be valuable to track within-subject microbial and behavioral dynamics over time, with the complementary goal to evaluate microbial stability.
These findings shed light on an additional mechanism through which prucalopride exerts its effects, highlighting the intricate and dynamic relationship between the gut and the brain. The dual behavioral and microbiota-modulating effects of the 5-HT4R agonist prucalopride provide a solid foundation for further research into the therapeutic potential of 5-HT4R-targeting molecules in the treatment of MDD. Notably, Ruminococcus emerges as a compelling bacterial candidate for exploring the microbiota’s role in mediating prucalopride’s central effects. The twofold benefits of prucalopride in improving both mood disorders and gut health underscore its potential as a novel therapeutic option. Future investigations could pave the way for its broader application in addressing the interconnected challenges of mental health and gastrointestinal well-being.
Methods
Animals
Adult male C57BL/6JRj mice were purchased from Janvier Labs (Le Genest-Saint-Isle, France). Mice were 7-8 weeks old and weighed 23–35 g at the beginning of the experiment, were maintained in a room with a controlled temperature (21 ± 1°C) on a 12-h/12-h light/dark cycle and were housed four per cage. Food and water were provided ad libitum. Behavioral testing occurred during the light phase. Protocols were approved by the Institutional Animal Care and Use Committee (IACUC) in France (Council directive n° 87-848, October 19, 1987, Ministère de l’Agriculture et de la Forêt, Service Vétérinaire de la Santé et de la Protection Animale, protocol APAFIS n° 39512) as well as with the European directive 2010/63/EU.
Drugs
Corticosterone [4-pregnen-11b-DIOL-3 20-DIONE 21-hemi-succinate, CORT (35 µg/mL)] purchased from Sigma-Aldrich (Saint-Quentin Fallavier, France) was dissolved in a vehicle [Veh = 0.45% hydroxypropyl-β-cyclodextrin (β-CD); Sigma-Aldrich]. Fluoxetine hydrochloride (Flx = 160 mg/L, equivalent to 18 mg/kg/day) was purchased from Anawa Trading (Zurich, Switzerland) and dissolved in 0.45% β-CD/CORT solution. Prucalopride (4-Amino-5-chloro-2,3-dihydro-N-[1-(3-methoxypropyl)-4-piperidinyl]-7-benzofurancarboxamide, 0.5 mg/kg/day and 1.5 mg/kg/day), dissolved in a mixture of 0.9% saline and 0.5% dimethyl sulfoxide (DMSO), was delivered by osmotic minipumps (28 days minipumps, 2004 model, Alzet, Cupertino, CA) implanted subcutaneously under light isoflurane anesthesia (3% induction and 1% maintenance) (Fig. S1). CORT-treated drinking water was replaced every 3 days to prevent any possible degradation as previously described7,20 and maintained throughout the experiment. Control animals (Veh/Veh and CORT/Veh groups) were also implanted with an Alzet minipump (model 2004) delivering 0.9% saline and 0.5% DMSO. CORT and Flx doses were calculated based an average fluid intake of 5 mL/mouse/day20. Instead of regular drinking water, mice were exposed for 8 weeks with Veh or CORT in the presence or absence of drugs (fluoxetine or prucalopride) administered during the last four weeks of the CORT protocol. Plasmatic drug levels were measured after subchronic (day 7) and chronic (day 28) treatments (Table S1).
Behavioral tests
All behavioral tests were conducted during the light phase (7 am–7 pm). The same animal was successively tested in three different behavioral paradigms [Elevated Plus Maze (EPM), the Novelty Suppressed Feeding (NSF) paradigm, and the Splash Test (ST)], from the least to the most aversive after a subchronic (days 3–7) and chronic treatment (days 24–28) with vehicle, fluoxetine or prucalopride (Fig. S1).
Elevated plus maze
The EPM is a widely used behavioral assay for rodents and it has been validated to assess the anti-anxiety effects of pharmacological agents53. This test was performed as described previously7. The maze was a plus-cross-shaped apparatus, with two open arms and two arms, each 45 cm long × 10 cm wide, linked by a central platform, 60 cm above the floor. Mice were individually put in the center of the maze facing an open arm and were allowed to explore the maze for a duration of 5 min. The time spent in the open arms and the number of entries into them were used as indexes of anxiety. The whole set-up was controlled using ANYMAZE version 7.4 video tracking software (Stoelting Co/Vivo-tech, Salon de Provence, France).
Novelty-suppressed feeding
The NSF is a conflict test that elicits competing motivations: the drive to eat and the fear of venturing into the center of a brightly lit arena54. Testing was performed as previously described20. Briefly, the NSF testing apparatus consisted of a plastic box (50 × 50 × 20 cm). The floor of which was covered with approximately 2 cm of wooden bedding and the arena was brightly lit. Mice were food restricted for 24 h prior to testing. At the time of testing, a single pellet of food (regular chow) was placed on a white paper platform in the center of the box. Each animal was placed in a corner of the box, and a stopwatch was immediately started. The NSF test was performed during a 10 min period as previously described20. The latency of the mice to begin eating in the arena was recorded. Immediately after the latency was recorded, the food pellet was removed from the arena. The mice were then placed back into their home cages. The latency to eat and the amount of food consumed in 5 min were measured (home cage consumption), followed by an assessment of post-fasting weight.
Splash test
This test consisted of squirting about 200 µL of a 10% sucrose solution on the mouse’s back55. This procedure induces grooming behaviors, due to the viscosity and palatability of the sucrose. The grooming behavior is sensitive to chronic stress or chronic CORT exposure and antidepressant treatment7. The total time spent grooming was recorded for 5 min in the home cage.
Emotionality score
The behavioral tests were used to compile behavioral emotionality of the mice (Z-score) as previously described56. Briefly, Z-scores are standardized scores (by the group mean and group standard deviation). They indicate how many standard deviations (σ) an observation (x) is above or below the mean of a control group (μ). Z-scores for behavioral measures were first averaged within the test, and then across the test for equal weighting of the three tests comprising the final emotionality score.
Fecal microbiota composition
Feces collection
For feces collection, mice were isolated in plastic boxes. The collection was performed in the morning when mice readily produce feces. Simple handling of the mice in the boxes was sufficient to produce feces. As soon as 3 droppings had been collected, and after a maximum of 10 min of isolation, the mice were returned to their original cages. After collection, feces were weighed and stored at –80 °C.
Bacterial DNA extraction
DNA was extracted using the Qiagen QIAmp Fast DNA Stool Mini Kit. The V3-V4 hypervariable region of the 16S rRNA gene was amplified and prepared for sequencing as outlined in the Illumina 16S Metagenomic Sequencing Library protocol. Samples were sequenced at (GenoToul, Toulouse) on the Illumina MiSeq platform using a (2 × 300 base pairs) as previously described by Picher and colleagues57. InhibitEX buffer was pre-heated and homogenized to eliminate crystals and safeguard functionality. Elution allowed efficient DNA purification. DNA concentrations were measured by a Nanodrop spectrophotometer.
V3-V4 PCR amplification of the 16S gene
The V3-V4 region of the 16S rRNA gene were targeted and amplified using the universal primers: PCR1F_460: 5′ CTITCCCTACACGACGCTCTTCCGATCTACGGRAGGCAGCAG3 ; PCR1R_460: 5′ GGAGITCAGACGTGTGCTCTTCCGATCTTACCAGGGTATCTAATCCT3′ and the conventional PCR protocol for 16S/18S MolTaq enzyme was followed (Alliance Bio Expertise, Breman, Germany). The number of PCR cycles was 30 and the concentration of bacterial DNA used was 50 ng/µL. The amplicons are migrated on % agarose gel and visualized under UV light to verify their quality. The 16S rRNA gene is a conserved phylogenetic marker among prokaryotes and allows the differentiation among the latter in a metagenomic sample, such as feces. The V3-V4 region allows discrimination among bacterial taxa and the calculation of relative abundances58.
Statistical analyses
All statistical tests and p values are listed in Supplemental Tables (Tables S2–S7).
Behavior analysis
Statistical analyses were performed using Prism Graphpad version 10.2.3. A one-way ANOVA followed by LSD multiple comparison test was employed for the EPM, ST and NSF (except for the survival analysis). The Fisher’s LSD test was chosen for its sensitivity in detecting pairwise differences following a significant ANOVA, particularly in small-sample exploratory analyses. However, follow-up studies with larger sample sizes would be required to confirm these findings. In the NSF, A Kaplan-Meier survival analysis was used due to the lack of normal distribution of data. The Mantel-Cox log-rank test was used to evaluate differences between experimental groups. All probabilities were two-sided with statistical significance set at p < 0.05.
16S rRNA sequencing data analysis
Data analysis was conducted using the QIIME2 bioinformatics package (version 2024.10) and the user-friendly software MicrobiomeAnalyst (version 2.0). The average number of reads was 24,418. Samples with less than 10k reads were excluded. Reads were denoised on a sequence base threshold of 240 which corresponded to a quality score ≥ 20, and rarefied to the median frequency of 14k. After quality control, the paired sequences that are 97% similar were clustered into Operational Taxonomic Units (OTUs) de novo. OTUs with low abundances were excluded from downstream analysis (maximum combined abundance <10). Bacterial taxa were assigned by aligning the OTUs to pre-defined sequences in the reference database classifier SILVA (silva-138-99-nb-classifier). Raw count data were transformed using the CLR (centered log ratio) transformation before conducting statistical analysis. Principal component analysis (PCoA), which evaluates the dissimilarity between samples, was performed on CLR values using the Bray-Curtis index and analyzed by a PERMANOVA. Non-parametric tests were used for compositional abundances (e.g., individual taxa, Kruskal-Wallis, and Mann-Whitney tests). To control for the False Discovery Rate (FDR) related to multiple testing, significant features were selected using the Benjamini-Hochberg procedure with a q-value of 0.2. All probabilities were two-sided with statistical significance set at p < 0.05.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Malhi, G. S. & Mann, J. J. Depression. Lancet 392, 2299–2312 (2018).
Gourion, D. Antidepressants and their onset of action: A major clinical, methodological and pronostical issue]. Encephale 34, 73–81 (2008).
Guina, J. & Merrill, B. Benzodiazepines I: Upping the care on downers: The evidence of risks, benefits and alternatives. J. Clin. Med. 7, 17 (2018).
Penninx, B. W., Pine, D. S., Holmes, E. A. & Reif, A. Anxiety disorders. Lancet 397, 914–927 (2021).
Samuels, B. A. et al. Serotonin 1A and serotonin 4 receptors: Essential mediators of the neurogenic and behavioral actions of antidepressants. Neuroscientist 22, 26–45 (2016).
Faye, C. et al. Rapid anxiolytic effects of RS67333, a serotonin type 4 receptor agonist, and diazepam, a benzodiazepine, are mediated by projections from the prefrontal cortex to the dorsal raphe nucleus. Biol. Psychiatry 87, 514–525 (2020).
Mendez-David, I. et al. Rapid anxiolytic effects of a 5-HT4 receptor agonist are mediated by a neurogenesis-independent mechanism. Neuropsychopharmacology 39, 1366–1378 (2014).
Poinsignon, V. et al. The GG genotype of the serotonin 4 receptor genetic polymorphism, rs1345697, is associated with lower remission rates after antidepressant treatment: Findings from the METADAP cohort. J. Affect Disord. 299, 335–343 (2022).
Köhler-Forsberg, K. et al. Serotonin 4 receptor brain binding in major depressive disorder and association with memory dysfunction. JAMA Psychiatry 80, 296–304 (2023).
Mahajan, R. Prucalopride: A recently approved drug by the food and drug administration for chronic idiopathic constipation. Int J. Appl Basic Med Res. 9, 1–2 (2019).
Johnson, D. et al. The 5-hydroxytryptamine4 receptor agonists prucalopride and PRX-03140 increase acetylcholine and histamine levels in the rat prefrontal cortex and the power of stimulated hippocampal oscillations. J. Pharmacol. Exp. Ther. 341, 681–691 (2012).
De Cates, A. N. et al. Association between a selective 5-HT4 receptor agonist and incidence of major depressive disorder: emulated target trial. Br. J. Psychiatry 225, 371–378.
Chen, B. K. et al. Prophylactic efficacy of 5-HT4R agonists against stress. Neuropsychopharmacology 45, 542–552 (2020).
Makovkina, E., Brody, B. D. & Shaffer, C. Possible association of 5-HT4 receptor agonist prucalopride in a 52-year-old man with an index manic episode. Bipolar Disord. 24, 767–769 (2022).
Kendig, D. M. & Grider, J. R. Serotonin and colonic motility. Neurogastroenterol. Motil. 27, 899–905 (2015).
Terry, N. & Margolis, K. G. Serotonergic mechanisms regulating the GI tract: Experimental evidence and therapeutic relevance. Handb. Exp. Pharm. 239, 319–342 (2017).
Cryan, J. F. et al. The microbiota-gut-brain axis. Physiol. Rev. 99, 1877–2013 (2019).
Cussotto, S., Sandhu, K. V., Dinan, T. G. & Cryan, J. F. The neuroendocrinology of the microbiota-gut-brain axis: A behavioural perspective. Front Neuroendocrinol. 51, 80–101 (2018).
Johnson, J. S. et al. Evaluation of 16S rRNA gene sequencing for species and strain-level microbiome analysis. Nat. Commun. 10, 5029 (2019).
David, D. J. et al. Neurogenesis-dependent and -independent effects of fluoxetine in an animal model of anxiety/depression. Neuron 62, 479–493 (2009).
Mendez-David, I. et al. A complex relation between levels of adult hippocampal neurogenesis and expression of the immature neuron marker doublecortin. Hippocampus 33, 1075–1093 (2023).
Tofani, G. S. S. et al. Gut microbiota regulates stress responsivity via the circadian system. Cell Metab. https://doi.org/10.1016/j.cmet.2024.10.003 (2024)
Ergang, P. et al. The gut microbiota affects corticosterone production in the murine small intestine. Int. J. Mol. Sci. 22, 4229 (2021).
Trivedi, M. H. et al. Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: Implications for clinical practice. Am. J. Psychiatry 163, 28–40 (2006).
Kato, M. & Serretti, A. Review and meta-analysis of antidepressant pharmacogenetic findings in major depressive disorder. Mol. Psychiatry 15, 473–500 (2010).
Wong, A., Taylor, D. M., Ashby, K. & Robinson, J. Changing epidemiology of intentional antidepressant drug overdose in Victoria, Australia. Aust. N. Z. J. Psychiatry 44, 759–764 (2010).
Griffin, C. E., Kaye, A. M., Bueno, F. R. & Kaye, A. D. Benzodiazepine pharmacology and central nervous system–mediated effects. Ochsner J. 13, 214–223 (2013).
Taylor, C., Fricker, A. D., Devi, L. A. & Gomes, I. Mechanisms of action of antidepressants: From neurotransmitter systems to signaling pathways. Cell Signal 17, 549–557 (2005).
Lucas, G. et al. Serotonin(4) (5-HT(4)) receptor agonists are putative antidepressants with a rapid onset of action. Neuron 55, 712–725 (2007).
Pascual-Brazo, J. et al. Modulation of neuroplasticity pathways and antidepressant-like behavioural responses following the short-term (3 and 7 days) administration of the 5-HT4 receptor agonist RS67333. Int. J. Neuropsychopharmacol. 15, 631–643 (2012).
Tamburella, A., Micale, V., Navarria, A. & Drago, F. Antidepressant properties of the 5-HT4 receptor partial agonist, SL65.0155: Behavioral and neurochemical studies in rats. Prog. Neuropsychopharmacol. Biol. Psychiatry 33, 1205–1210 (2009).
Yaakob, N. S., Chinkwo, K. A., Chetty, N., Coupar, I. M. & Irving, H. R. Distribution of 5-HT3, 5-HT4, and 5-HT7 receptors along the human colon. J. Neurogastroenterol. Motil. 21, 361 (2015).
Patel, R. A., Panche, A. N. & Harke, S. N. Gut microbiome-gut brain axis-depression: interconnection. World J. Biol. Psychiatry 26, 1–36 (2025).
Lyte, J. M. et al. Gut-brain axis serotonergic responses to acute stress exposure are microbiome-dependent. Neurogastroenterol. Motil. 32, e13881 (2020).
Sudo, N. et al. Postnatal microbial colonization programs the hypothalamic–pituitary–adrenal system for stress response in mice. J. Physiol. 558, 263–275 (2004).
Wang, G. et al. Gut microbiota dysbiosis-mediated ceramides elevation contributes to corticosterone-induced depression by impairing mitochondrial function. npj Biofilms Microbiomes 10, 111 (2024).
Dilmore, A. H. et al. Medication use is associated with distinct microbial features in anxiety and depression. Mol. Psychiatry 1–13 https://doi.org/10.1038/s41380-024-02857-2 (2025).
Cussotto, S., Clarke, G., Dinan, T. G. & Cryan, J. F. Psychotropics and the microbiome: A chamber of secrets. Psychopharmacol. (Berl.) 236, 1411–1432 (2019).
Cussotto, S. et al. Differential effects of psychotropic drugs on microbiome composition and gastrointestinal function. Psychopharmacol. (Berl.) 236, 1671–1685 (2019).
Ayaz, M. et al. Sertraline enhances the activity of antimicrobial agents against pathogens of clinical relevance. J. Biol. Res. (Thessalon.) 22, 4 (2015).
Coban, A. Y., Tanriverdi Cayci, Y., Keleş Uludağ, S. & Durupinar, B. Investigation of antibacterial activity of sertralin]. Mikrobiyol. Bul. 43, 651–656 (2009).
La Reau, A. J. & Suen, G. The Ruminococci: Key symbionts of the gut ecosystem. J. Microbiol 56, 199–208 (2018).
Li, Y. & Liu, P. Characteristics of oral-gut microbiota in model rats with CUMS-induced depression. Neuropsychiatr. Dis. Treat. 20, 221–232 (2024).
Kemp, K. M., Colson, J., Lorenz, R. G., Maynard, C. L. & Pollock, J. S. Early life stress in mice alters gut microbiota independent of maternal microbiota inheritance. Am. J. Physiol. Regul. Integr. Comp. Physiol. 320, R663–R674 (2021).
McGaughey, K. D. et al. Relative abundance of Akkermansia spp. and other bacterial phylotypes correlates with anxiety- and depressive-like behavior following social defeat in mice. Sci. Rep. 9, 3281 (2019).
Liu, L. et al. Gut microbiota and its metabolites in depression: From pathogenesis to treatment. eBioMedicine 90, 104527 (2023).
Lukić, I. et al. Antidepressants affect gut microbiota and Ruminococcus flavefaciens is able to abolish their effects on depressive-like behavior. Transl. Psychiatry 9, 133 (2019).
Gao, M. et al. Gut microbiota composition in depressive disorder: a systematic review, meta-analysis, and meta-regression. Transl. Psychiatry 13, 379 (2023).
Hu, X. et al. Changes of gut microbiota reflect the severity of major depressive disorder: a cross sectional study. Transl. Psychiatry 13, 1–9 (2023).
Ghosh, S., Whitley, C. S., Haribabu, B. & Jala, V. R. Regulation of intestinal barrier function by microbial metabolites. Cell Mol. Gastroenterol. Hepatol. 11, 1463–1482 (2021).
Person, H. & Keefer, L. Psychological comorbidity in gastrointestinal diseases: Update on the brain-gut-microbiome axis. Prog. Neuropsychopharmacol. Biol. Psychiatry 107, 110209 (2021).
Serbanescu, M. A. et al. General anesthesia alters the diversity and composition of the intestinal microbiota in mice. Anesth. Analg. 129, e126–e129 (2019).
Walf, A. A. & Frye, C. A. The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat. Protoc. 2, 322–328 (2007).
Francois, M., Canal Delgado, I., Shargorodsky, N., Leu, C.-S. & Zeltser, L. Assessing the effects of stress on feeding behaviors in laboratory mice. eLife 11, e70271 (2022).
Bouguiyoud, N. et al. Anxiety and depression assessments in a mouse model of congenital blindness. Front. Neurosci. 15, 807434 (2022).
Guilloux, J.-P., Seney, M., Edgar, N. & Sibille, E. Integrated behavioral z-scoring increases the sensitivity and reliability of behavioral phenotyping in mice: Relevance to emotionality and sex. J. Neurosci. Methods 197, 21–31 (2011).
Pichler, M. et al. A 16S rRNA gene sequencing and analysis protocol for the Illumina MiniSeq platform. Microbiologyopen 7, e00611 (2018).
Shahi, S. K., Zarei, K., Guseva, N. V. & Mangalam, A. K. Microbiota analysis using two-step PCR and next-generation 16S rRNA gene sequencing. J. Vis. Exp. https://doi.org/10.3791/59980 (2019).
Acknowledgements
We kindly thank V. Domergue and the staff of the animal care facility (UMS-IPSIT Animex, Université Paris-Saclay, Orsay, France) for their invaluable support. We thank Bienvenue Koffi and Lou Bomal for their technical support with fecal DNA extraction. IMD was supported by a National Alliance for Research on Schizophrenia and Depression (NARSAD) 2017 and 2022 Young Investigator Award from the Brain & Behavior Research Foundation, the Deniker Foundation and by the France 2030 program “ANR-11-IDEX-0003”, from the Head Core call from Health and Drug Sciences Graduate School of Université Paris-Saclay. DJD was supported by the France 2030 program “ANR-11-IDEX-0003”, the OI HEALTHI of Université Paris-Saclay, and a 2021 Schaefer Award Scholarship from Columbia University.
Author information
Authors and Affiliations
Contributions
I.M.D., D.J.D., C.A.D., and R.H. designed the experiment. I.M.D., S.R.A., I.E., and D.J.D. conducted experiments and prepared the figures. I.M.D., S.C., D.J.D., E.C., R.C. performed literature search and participated in data interpretation. S.R.A., S.C., I.E., J.C.A. were involved in data collection. I.M.D. and S.C. performed data analysis. S.C. wrote the initial draft of the manuscript. All authors critically revised the manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing interests
I.M.D., D.J.D., and C.A.D. are named on provisional and non-provisional patent applications for the use of novel serotonin type four receptor prophylactics against stress-induced psychiatric disease.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cussotto, S., Abdennebi, S.R., Etting, I. et al. Prucalopride, a serotonin type 4 receptor agonist, induces fast anxiolytic/antidepressant effects and concomitant changes in the gut microbiota. npj Biofilms Microbiomes 12, 62 (2026). https://doi.org/10.1038/s41522-026-00928-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41522-026-00928-6