Abstract
The Breast Cancer Index (BCI) was previously shown to identify ~20% of postmenopausal patients with early stage, hormone receptor positive (HR+), node negative (N0) breast cancer with minimal (<5%) risk of 10-year distant recurrence (DR) even without receiving adjuvant endocrine therapy (ET). This prospective-retrospective study further validated the BCI minimal risk classification in postmenopausal patients with early-stage, HR + HER2– N0 breast cancer from the Netherlands Cancer Registry (NCR) and the Tamoxifen and Exemestane Adjuvant Multinational (TEAM, NCT00279448, NCT00032136) randomized trial who received 5 years of primary adjuvant ET. BCI classified approximately 15% of patients as minimal risk. In the NCR cohort (n = 1264 out of 15,053 HR+ patients in the registry), risks of DR in the minimal, low, intermediate, and high groups were 4.8%, 3.3%, 8.0%, and 12.4%, respectively (P < 0.001). In the TEAM cohort (n = 978 out of 3544 in the BCI study), DR risks were 3.8%, 8.3%, 12.6% and 22.7% (P < 0.001). In multivariate analyses, BCI risk scores provided independent information over standard prognostic factors (P < 0.001). This study confirmed the ability of the adjusted BCI model to identify postmenopausal women with HR + HER2– N0 breast cancer who are at minimal risk of DR and may consider de-escalating adjuvant ET.
Similar content being viewed by others
Introduction
The standard of care treatment for hormone receptor–positive (HR+) breast cancer includes at least 5 years of adjuvant endocrine therapy (ET) with aromatase inhibitors and/or tamoxifen1,2,3,4,5, administered with the goal of reducing disease recurrence. However, the selection of the optimal therapy regimen and duration can be complex and requires consideration of the patient’s recurrence risk, comorbidities, and potential benefits and risks of adjuvant ET1. Accurate assessment of recurrence risk, to identify patients at very low risk of recurrence, can potentially spare many patients from overtreatment with adjuvant ET and the associated adverse effects such as musculoskeletal symptoms and bone toxicities with aromatase inhibitors and uterine cancer and deep vein thrombosis with tamoxifen1,3,4,5,6. Genomic biomarkers assessing the risk of recurrence may be useful to identify very low-risk patients at the time of diagnosis to guide treatment selection with the option of de-escalating adjuvant ET.
The Breast Cancer Index (BCI), a gene expression–based signature, reports both predictive7,8,9,10 and prognostic results9,10,11,12 to aid in clinical decision-making for extended adjuvant (post–year 5) ET in patients with early-stage, HR+ breast cancer. BCI predicts the likelihood of benefit from extended ET using the HOXB13/IL17BR expression ratio (H/I), an endocrine response biomarker that interrogates estrogen signaling in HR+ breast cancer7,9,12,13. BCI prognostic scores provide the individualized risk of overall (0–10 years after diagnosis) and late (>5 years after diagnosis) distant recurrence, calculated using an algorithm that combines BCI (H/I) and the Molecular Grade Index, which assesses tumor proliferative status based on the expression levels of 5 cell cycle–associated genes10,12,14,15. The prognostic component of BCI has been validated to predict the risk of overall and late distant recurrence in patients with HR + N0 breast cancer12,14. BCI is recommended in the National Comprehensive Cancer Network (NCCN) and American Society of Clinical Oncology (ASCO) clinical practice guidelines for clinical decision-making regarding extended ET1,16.
Recently, an additional BCI cutpoint was identified that stratified postmenopausal patients with early-stage, HR+, N0 breast cancer with minimal (<5%) risk of 10-year distant recurrence17. The study used patients from the Stockholm STO-3 trial and real-world data from the Netherlands Cancer Registry (NCR) that did not receive any adjuvant systemic therapy (ET or chemotherapy) to identify approximately 20% of patients in both cohorts with a minimal risk of 10-year distant recurrence. These patients classified as minimal risk are considered appropriate candidates for de-escalation of adjuvant ET.
Here we report further validation of the BCI minimal risk cutpoint in postmenopausal patients from the NCR and TEAM trial with early-stage, HER2–, N0 breast cancer who received primary adjuvant ET.
Results
Patient characteristics
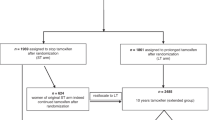
Tumor specimens were collected from 1305 patients in the NCR cohort (Supplemental Fig. 1). After excluding 41 specimens that had insufficient tumor content or did not meet eligibility criteria, the final NCR dataset included 1264 patients who received adjuvant ET.
Table 1 summarizes clinicopathological characteristics of the NCR and TEAM cohorts. All patients in the NCR cohort were ≥70 years old, while most (63.8%) in the TEAM cohort were <70 years old. A higher proportion of patients in the NCR cohort had well-differentiated tumors compared with the TEAM cohort (21.5% vs 3.9%), and a lower proportion had poorly differentiated tumors (NCR, 17.6%; TEAM, 36.4%). The median follow-up in the NCR and TEAM cohorts was 12.2 and 9.4 years, respectively.
Validation of the BCI minimal risk cutpoint in the NCR cohort
The prognostic performance of the adjusted BCI model with the additional minimal risk cutpoint was evaluated first in patients from the NCR cohort (n = 1264), all of whom received up to 5 years of adjuvant ET. BCI classified 173 (13.7%), 496 (39.2%), 320 (25.3%), and 275 (21.8%) patients as minimal, low, intermediate, and high risk with 10-year risks of distant recurrence of 4.8% (95% CI, 2.3–8.9), 3.3% (95% CI, 2.0–5.2), 8.0% (95% CI, 5.3–11.4), and 12.4% (95% CI, 8.8–16.7), respectively (Fig. 1A, Table 2). The sHRs were 0.69 (95% CI, 0.30–1.60) for low risk, 1.73 (95% CI, 0.79–3.81) for intermediate risk, and 2.74 (95% CI, 1.28-5.87) for high risk versus minimal risk, with death as a competing risk event (P < .001) (Table 2).
A NCR cohort overall. B, C Clinical subsets of the NCR cohort. BCI, Breast Cancer Index; G2/G3, tumor grade 2/3; NCR, Netherlands Cancer Registry; T1/T2, T stage 1/2; TAM, tamoxifen; TEAM, Tamoxifen and Exemestane Adjuvant Multinational trial.
The prognostic performance of the adjusted BCI model was further evaluated in relevant clinical subgroups. Among patients with T1/T2 disease (n = 1205), the 10-year risk of distant recurrence was 4.5% (95% CI, 2.0–8.7) for minimal risk, 3.5% (95% CI, 2.1–5.4) for low risk, 8.4% (95% CI, 5.6–11.9) for intermediate risk, and 12.7% (95% CI, 9.0–17.1) for high risk (Fig. 1B). The sHRs were 0.78 (95% CI, 0.32–1.87) for low risk, 1.98 (95% CI, 0.86–4.53) for intermediate risk, and 3.04 (95% CI, 1.36–6.81) for high risk versus minimal risk (P < .001).
Among patients with G2/G3 disease (n = 992), the 10-year risk of distant recurrence was 3.9% (95% CI, 1.3–9.1) for minimal risk, 2.5% (95% CI, 1.2–4.5) for low risk, 8.9% (95% CI, 5.8–12.7) for intermediate risk, and 11.7% (95% CI, 8.0–16.0) for high risk (Fig. 1C). The sHRs were 0.67 (95% CI, 0.21–2.16) for low risk, 2.50 (95% CI, 0.88–7.11) for intermediate risk, and 3.31 (95% CI, 1.19–9.25) for high risk versus minimal risk (P < 0.001).
To investigate whether BCI provided independent prognostic information over traditional clinicopatho-logical factors, univariate and multivariate Fine-Gray models were performed. Only BCI risk scores were significantly prognostic for 10-year distant recurrence in both univariate and multivariate analysis (Table 3), providing additional independent prognostic information over standard prognostic factors, with a sHR of 3.42 (1.87–6.26; likelihood ratio χ2 = 14.58; P < .001).
Validation of the BCI minimal risk cutpoint in the TEAM cohort
The prognostic performance of the adjusted BCI model was further validated in patients from the TEAM trial (n = 978). BCI categorized 151 (15.4%), 347 (35.5%), 280 (28.6%), and 200 (20.4%) patients as minimal, low, intermediate, and high risk, respectively (Fig. 2A, Table 2). The corresponding 10-year risks of distant recurrence were 3.8% (95% CI, 0.4–7.0), 8.3% (95% CI, 4.1–12.3), 12.6% (95% CI, 8.2–16.8), and 22.7% (95% CI, 16.1–28.9), respectively (Fig. 2A, Table 2). The HRs were 1.62 (95% CI, 0.60–4.37) for low risk, 3.36 (95% CI, 1.30–8.65) for intermediate risk, and 6.51 (95% CI, 2.57–16.51) for high risk versus minimal risk (P < 0.001) (Table 2).
A TEAM cohort overall. B–D Clinical subsets from the TEAM cohort. BCI, Breast Cancer Index; NCR, Netherlands Cancer Registry; T1/T2, T stage 1/2; G2/G3, grade 2/3; TEAM, Tamoxifen and Exemestane Adjuvant Multinational trial.
Among patients with T1/T2 disease (n = 946), the 10-year risk of distant recurrence was 3.1% (95% CI, 0.0–6.0) for minimal risk, 7.5% (95% CI, 3.4–11.4) for low risk, 12.1% (95% CI, 7.7–16.3) for intermediate risk, and 23.1% (95% CI, 16.3–29.3) for high risk (Fig. 2B). The HRs were 1.81 (95% CI, 0.60–5.41) for low risk, 3.89 (95% CI, 1.36–11.08) for intermediate risk, and 7.98 (95% CI, 2.85–22.30) for high risk versus minimal risk (P < .001).
Among patients with G2/3 disease (n = 900), the 10-year risk of distant recurrence was 3.4% (95% CI, 0.0–6.6) for minimal risk, 8.3% (95% CI, 3.5–12.8) for low risk, 12.8% (95% CI, 8.3–17.1) for intermediate risk, and 21.2% (95% CI, 14.6–27.3) for high risk (Fig. 2C). The HRs were 1.75 (95% CI, 0.58–5.27) for low risk, 3.88 (95% CI, 1.36–11.03) for intermediate risk, and 6.80 (95% CI, 2.42–19.11) for high risk versus minimal risk (P < .001).
Among patients <70 years old (n = 624), the 10-year risk of distant recurrence was 3.5% (95% CI, 0.0–7.4) for minimal risk, 5.9% (95% CI, 1.0–10.6) for low risk, 11.1% (95% CI, 5.8–16.2) for intermediate risk, and 21.0% (95% CI, 13.1–28.2) for high risk (Fig. 2D). The HRs were 1.05 (95% CI, 0.27–4.04) for low risk, 3.16 (95% CI, 0.92–10.84) for intermediate risk, and 6.44 (95% CI, 1.94–21.34) for high risk versus minimal risk (P < .001).
In univariate and multivariate analyses, both tumor size and BCI risk scores were significantly prognostic for 10-year distant recurrence (Table 3). In multivariate analysis, the additional independent prognostic information that BCI risk scores provided over standard prognostic factors was associated with an HR of 7.49 (95% CI, 3.97–14.11; likelihood ratio χ2 = 41.31; P < 0.001).
Discussion
Five years of adjuvant ET is part of the standard of care treatment for HR+ breast cancer, with the goal of reducing the risk of disease recurrence1,2. However, the rate of DR has declined in recent decades following improvements in adjuvant therapies and biomarker tests to better characterize the disease18. It is now understood that the risk of recurrence substantially varies among patients but can be predicted on an individual patient basis using clinicopathological risk factors and genomic classifiers16,19,20. Among patients with early-stage breast cancer, distant or multiple recurrences represent 71% of first recurrence events21. Many local recurrences are eventually followed by a DR event, which is associated with breast cancer–specific mortality21,22. DR is therefore the most clinically relevant endpoint when considering treatment decisions for early-stage breast cancer. Patients at minimal risk of distant recurrence may consider shortening ET treatment duration and avoiding the potentially serious toxicities associated with long-term adjuvant ET.
The prognostic ability of BCI to stratify patients with early-stage ER+ breast cancer based on the risk of overall (0–10 years) and late (5–10 years) DR has been demonstrated previously12,14. These initial studies classified patients according to low, intermediate, and high recurrence risk, allowing for the identification of patients most and least likely to benefit from adjuvant systemic therapy. Recently, an additional BCI cutpoint was developed and validated to identify a subgroup of postmenopausal patients with HR + N0 disease at minimal risk ( < 5%) of 10-year distant recurrence even without receiving any adjuvant ET17. In that study, which included patients from the prospective randomized Stockholm trial (untreated vs. 2 or 5 years of tamoxifen) and an untreated cohort from NCR, 16–22% of patients were classified as minimal risk of recurrence, with 10-year DR rates of 2.3–4.5% across cohorts. Notably, when analyzing patients in the two treatment arms (untreated vs. 2 or 5 years of tamoxifen) from the Stockholm trial, albeit the limited sample size, all patients categorized as low, intermediate and high risk derived substantial absolute benefit from adjuvant tamoxifen but not those in the minimal risk group. The reductions in risks of distant recurrence for minimal, low, intermediate and high risk patients were −2.0%, 10.5%, 8.1%, and 14.8%, respectively. As a result of the differential benefit across BCI risk groups, the minimal and low risk group displayed a similar risk profile during follow-up in the treated Stockholm cohort17.
The current analysis further validated the BCI minimal risk cutpoint in patients who received primary adjuvant ET. Similar to the previous validation17, approximately 15% of patients were identified as minimal risk (<5%) of 10-year DR. At year 10, 4.8% of minimal risk patients in the NCR cohort and 3.8% in the TEAM cohort had experienced a DR event. Notably, similar to what was observed in the Stockholm treated cohort, patients categorized as BCI minimal and low risk from both NCR and TEAM cohorts who were treated with adjuvant ET shared a similar risk profile across the 10 years of follow-up, suggesting that patients categorized as low risk may derive benefit from adjuvant ET, while those classified as minimal risk may not.
The immediate clinical utility of the minimal risk classification may mainly lie in patients deemed clinically low risk, as these patients are more likely to be considered for de-escalation of adjuvant ET. To that end, this study demonstrated that those patients with T1 or T2 tumors indeed experienced <5% risk of DR over 10 years (4.5% in the NCR cohort and 3.1% in the TEAM cohort) and thus could consider shorter duration of adjuvant ET to balance the potential benefit and harm of such treatment. Finally, since the NCR cohorts only included patients who were at least 70 years old, the subset analysis of patients from the TEAM trial who were younger than 70 years confirmed the prognostic performance of the minimal risk group in postmenopausal women regardless of age.
Study strengths include the use of a large, prospectively collected real-world patient cohort and data from a large, randomized controlled trial. In the NCR cohort, the estimated 10-year risk of distant recurrence rates are based on the assumption of having completed 5 years of ET, however the actual duration of ET may have been shorter, therefore the rates could be an overestimation of the true 10-year risk of DR. One limitation was the exclusion of patients ≤70 years old from the NCR cohort; however this was balanced by including the TEAM cohort with 64% of patients <70 years old. A high mortality rate in the NCR cohort (79%; not breast cancer–specific) during the 10-year follow-up posed another limitation to the study. This was addressed by considering death as a competing risk event in the analyses but might still lead to an underestimation of recurrence risk, especially in patients categorized as high risk. Additional studies may further elucidate the prognostic ability of the BCI minimal risk cutpoint beyond 10 years and in premenopausal patients who face the challenging clinical decision of whether to add ovarian function suppression to adjuvant ET.
In conclusion, the results of this study further validate BCI as a biomarker for personalized adjuvant ET decision-making to avoid over- and undertreatment of patients with early-stage, HR+ breast cancer. The use of BCI at the time of diagnosis or within the first 5 years can inform the risk of distant recurrence and, for patients with minimal risk of recurrence, may support decisions to consider de-escalating adjuvant ET.
Methods
Study design and patient selection
The primary study objective was to examine whether the adjusted BCI risk model would identify a group of women with HR + HER2– N0 breast cancer who received adjuvant ET and were at minimal risk (<5%) of 10-year distant recurrence. Secondary objectives were to evaluate the prognostic performance of the adjusted BCI model in patient subgroups according to tumor size, grade and age. The study endpoint was time to distant recurrence, defined as time from randomization (TEAM trial) or date of diagnosis (NCR cohort) to first recurrence at distant sites. Contralateral disease, locoregional recurrence, and other secondary primary cancers were not counted as events.
The phase 3 randomized TEAM trial evaluated disease-free survival in postmenopausal women with HR+ breast cancer who received 5 years of adjuvant treatment with exemestane alone or tamoxifen for 2.5-3 years followed by exemestane (NCT00279448, NCT00032136, and NCT00036270)23. The previous BCI translational study evaluated 3544 patients from the TEAM trial who had available RNA samples for BCI testing24. The current analysis included patients with HER2− N0 breast cancer who received adjuvant ET but no chemotherapy. The cutoff level for ER and PR positivity was set to 1% as previously described24.
The NCR cohort included patients ≥70 years old with a diagnosis of invasive, ER + HER2-, N0 breast cancer who had undergone surgery and received adjuvant ET but no chemotherapy from 45 hospitals throughout the Netherlands25. Specially trained data managers from NCR collected clinical data on diagnosis, staging, and treatment directly from medical records using international coding rules. In compliance with Dutch national breast cancer pathology guidelines, the cutoff level for ER positivity was set to 10%26. Information on disease recurrence was retrospectively collected from electronic medical records. Registration and distribution of formalin-fixed, paraffin-embedded tumor blocks acquired during routine treatment were coordinated by Palga, the nationwide network and registry of histo- and cytopathology in the Netherlands.
The TEAM trial and NCR study were conducted in accordance with the Declaration of Helsinki and the International Council for Harmonisation guidelines, as well as Good Clinical Practice. The collection and analysis of tumor samples from the TEAM trial were approved by the institutional review boards, and all patients provided written informed consent. The TEAM trial is registered with ClinicalTrials.gov, NCT00279448 and NCT00032136. The NCR study was ethically approved by the Institutional Review Board of the Leiden University Medical Center. Due to the large number of deceased participants, the study was granted a waiver for informed consent.
BCI cutpoints and molecular testing
The traditional BCI prognostic model stratifies patients into low, intermediate, and high risk groups using validated cutpoints of 5.1 and 6.512. Recently, an additional minimal risk cutpoint of 3.0 was developed and validated to identify patients who had less than a 5% risk of 10-year distant recurrence even without receiving adjuvant ET17. These cutpoints would stratify patients into four prognostic risk groups (minimal, low, intermediate, high risk). In the current study, this adjusted BCI model was further validated using patients in the TEAM trial and the NCR cohort who received adjuvant ET.
The BCI analysis of the TEAM cohort was described previously24. For the NCR cohort, analysis of BCI gene expression was done using RT-qPCR with RNA extracted from primary tumor specimens. Tumor sections were prepared at Leiden University Medical Center, shipped to Biotheranostics Inc., a Hologic company, and tested in the CLIA-certified, CAP-accredited laboratory. Testing was blinded to clinical outcome and done in accordance with Good Clinical Practice. Total extracted RNA was reverse transcribed. The resulting cDNA was pre-amplified using PCR with the PreAmp Master Mix Kit (Thermo Fisher Scientific; Carlsbad, CA) and was analyzed using TaqMan RT-PCR. The final analysis included all samples that met predetermined quality control criteria requiring a mean cycle threshold of 4 reference genes of <26.512. BCI scores were calculated and patients were stratified into the four prognostic risk groups as described above.
Statistical considerations
In the TEAM cohort, Kaplan-Meier survival analysis was used to graphically present survival curves for the adjusted BCI risk groups; equality of the survival curves was evaluated using a log-rank test. Cox proportional hazards models were used to calculate hazard ratios (HRs) and 95% CIs. In the NCR cohort, to account for the death of 79% of subjects, cumulative incidence analysis and sub-distribution HRs (sHRs) from Fine-Gray models with likelihood ratio tests were used, both analyses considering death as a competing risk event. For both cohorts, univariate and multivariate Cox or Fine-Gray models with likelihood ratio tests were used to evaluate whether BCI as a continuous risk index (analyzed in 5-unit increments) provided additional prognostic information independent of clinical and pathologic factors. For all analyses, follow-up was truncated at 10 years. A two-sided P ≤ .05 was considered statistically significant. Statistical analyses were done using R (version 4.3.1).
Data availability
Qualified researchers may request access to de-identified individual participant data by submitting a proposal to the corresponding author, which will be reviewed for scientific merit and feasibility.
References
National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines). Breast cancer. Version 4.2025 (2025).
Loibl, S. et al. Early breast cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 35, 159–182 (2024).
Early Breast Cancer Trialists’ Collaborative Group Tamoxifen for early breast cancer: an overview of the randomised trials. Early Breast Cancer Trialists’ Collaborative Group. Lancet 351, 1451–1467 (1998).
Arimidex Tamoxifen Alone or in Combination Trialists’ Group et al. Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 100-month analysis of the ATAC trial. Lancet Oncol. 9, 45–53 (2008).
Breast International Group 1-98 Collaborative Group et al. A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N. Engl. J. Med. 353, 2747–2757 (2005).
Davies, C. et al. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet 381, 805–816 (2013).
Bartlett, J. M. S. et al. Breast Cancer Index Is a predictive biomarker of treatment benefit and outcome from extended tamoxifen therapy: final analysis of the Trans-aTTom study. Clin. Cancer Res. 28, 1871–1880 (2022).
Noordhoek, I. et al. Breast Cancer Index predicts extended endocrine benefit to individualize selection of patients with HR(+) early-stage breast cancer for 10 years of endocrine therapy. Clin. Cancer Res. 27, 311–319 (2021).
Sgroi, D. C. et al. Prediction of late disease recurrence and extended adjuvant letrozole benefit by the HOXB13/IL17BR biomarker. J. Natl. Cancer Inst. 105, 1036–1042 (2013).
Zhang, Y. et al. A novel Breast Cancer Index for prediction of distant recurrence in HR(+) early-stage breast cancer with one to three positive nodes. Clin. Cancer Res. 23, 7217–7224 (2017).
Bartlett, J. M. et al. Prognostic performance of Breast Cancer Index (BCI) in postmenopausal women with early-stage HR+ breast cancer in the TEAM trial. Abstr. 138MO. Ann. Oncol. 33, S602 (2022).
Zhang, Y. et al. Breast Cancer Index identifies early-stage estrogen receptor-positive breast cancer patients at risk for early- and late-distant recurrence. Clin. Cancer Res. 19, 4196–4205 (2013).
Bartlett, J. M. S. et al. Breast Cancer Index and prediction of benefit from extended endocrine therapy in breast cancer patients treated in the Adjuvant Tamoxifen-To Offer More? (aTTom) trial. Ann. Oncol. 30, 1776–1783 (2019).
Sgroi, D. C. et al. Prediction of late distant recurrence in patients with oestrogen-receptor-positive breast cancer: a prospective comparison of the breast-cancer index (BCI) assay, 21-gene recurrence score, and IHC4 in the TransATAC study population. Lancet Oncol. 14, 1067–1076 (2013).
Habel, L. A. et al. HOXB13:IL17BR and molecular grade index and risk of breast cancer death among patients with lymph node-negative invasive disease. Breast Cancer Res. 15, R24 (2013).
Andre, F. et al. Biomarkers for adjuvant endocrine and chemotherapy in early-stage breast cancer: ASCO Guideline update. J. Clin. Oncol. 40, 1816–1837 (2022).
Jilderda, M. F. et al. Identification of Early-Stage Breast Cancer with a Minimal Risk of Recurrence by the Breast Cancer Index. Clin. Cancer Res. 31, 2222–2229 (2025).
Early Breast Cancer Trialists’ Collaborative Group Reductions in recurrence in women with early breast cancer entering clinical trials between 1990 and 2009: a pooled analysis of 155 746 women in 151 trials. Lancet 404, 1407–1418 (2024).
Sheffield, K. M. et al. A real-world US study of recurrence risks using combined clinicopathological features in HR-positive, HER2-negative early breast cancer. Future Oncol. 18, 2667–2682 (2022).
Volkel, V. et al. Improved risk estimation of locoregional recurrence, secondary contralateral tumors and distant metastases in early breast cancer: the INFLUENCE 2.0 model. Breast Cancer Res. Treat. 189, 817–826 (2021).
Early Breast Cancer Trialists’ Collaborative, G. et al. Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet 378, 771–784 (2011).
Vrieling, C. et al. The impact of isolated local recurrence on long-term outcome in early-breast cancer patients after breast-conserving therapy. Eur. J. Cancer 155, 28–37 (2021).
van de Velde, C. J. et al. Adjuvant tamoxifen and exemestane in early breast cancer (TEAM): a randomised phase 3 trial. Lancet 377, 321–331 (2011).
Bartlett, J. M. S. et al. Validation of the Prognostic Performance of Breast Cancer Index in Hormone Receptor-Positive Postmenopausal Breast Cancer Patients in the TEAM Trial. Clin. Cancer Res. 30, 1509–1517 (2024).
de Boer, A. Z. et al. Impact of older age and comorbidity on locoregional and distant breast cancer recurrence: a large population-based study. Oncologist 25, e24–e30 (2020).
Federatie Medisch Specialisten (Federation of Medical Specialists). Breast Cancer Guidelines. Section 4.7: Pathology, Receptor Determination (2012) https://richtlijnendatabase.nl/richtlijn/borstkanker/pathologie/receptorbepaling.html.
Acknowledgements
This study was funded by Biotheranostics Inc., a Hologic company. The authors thank the registration team of the Netherlands Comprehensive Cancer Organisation (IKNL) for the collection of data for the Netherlands Cancer Registry. Also, the authors greatly thank the Research Laboratory of the Department of Surgery of the Leiden University Medical Center for their help with the laboratory work.
Author information
Authors and Affiliations
Contributions
M.-F. Jilderda: data curation, supervision, writing—original draft, writing—review & editing. J.M.S. Bartlett: data curation, supervision, writing—original draft, writing—review & editing. G.-J. Liefers: conceptualization, data curation, funding acquisition, supervision, visualization, writing—original draft, writing—review & editing. Y. Zhang: conceptualization, data curation, formal analysis, visualization, writing—original draft, writing—review & editing. H. Dunn-Davies: writing—review & editing. V. Rebattu: conceptualization, data curation, funding acquisition, visualization, writing—review & editing. R. Salunga: data curation, writing—review & editing. E.M-K Kranenbarg: data curation, writing—review & editing. L. de Munck: data curation, writing—review & editing. A. Hasenburg: writing—review & editing C. Markopoulos: writing—review & editing L. Dirix: writing—review & editing C. J. H. van de Velde: writing—review & editing D. Rea: writing – review & editing. A.K.L. Anderson: writing – review & editing— E. Bastiaannet: data curation, formal analysis, writing—review & editing. K. Treuner: conceptualization, data curation, supervision, visualization, writing—original draft, writing—review & editing. K.J. Taylor: data curation, supervision, writing—review & editing. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare the following competing interests (non-financial and financial): M.-F. Jilderda reports an unrestricted research grant from Biotheranostics to LUMC, and employment at LUMC. Y. Zhang reports a pending patent for BCI, an issued patent for BCI from Biotheranostics, A Hologic Company, and employment and stock ownership of Biotheranostics. V. Rebattu reports an unrestricted research grant from Biotheranostics to LUMC, and employment at LUMC. R. Salunga reports employment and stock ownership at Biotheranostics, A Hologic company. A.K.L. Anderson reports employment and stock ownership of Biotheranostics, A Hologic company. K. Treuner reports a pending patent for BCI and employment and stock ownership of Biotheranostics, A Hologic company. G.-J. Liefers reports an unrestricted research grant from Biotheranostics to LUMC, and employment at LUMC. No competing interests were reported by the other authors.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jilderda, MF., Bartlett, J.M.S., Liefers, GJ. et al. Validation of minimal risk of recurrence classification by the Breast Cancer Index in early stage breast cancer. npj Breast Cancer 12, 21 (2026). https://doi.org/10.1038/s41523-025-00885-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41523-025-00885-x