Abstract
Sleep disturbances are both prodromes and potential risk factors for Parkinson’s disease (PD), yet associations between long-term sleep patterns and PD remain unclear. We analyzed data from two online cohorts—PPMI-Online (n = 15,905) and Fox Insight (n = 1929)—to examine associations between self-reported sleep duration trajectories across life stages and PD risk and age at onset (AAO). Latent class growth analysis identified distinct trajectories, linear and logistic regression examined their associations with PD risk and AAO, adjusting for demographics, lifestyle, and comorbidities. In PPMI-Online, sleep duration was generally stable in early adulthood with divergence in midlife. Compared to stable sleepers (7–8 h/day), individuals with midlife reductions (6–7 to ≤5–6 h/day: OR = 1.90; 7–8 to ≤6–7 h/day: OR = 1.64) and persistent short sleep (≤6 h/day: OR = 1.41) had higher PD risk, independent of comorbidities. Similar patterns were observed in probable prodromal PD. Persistent short sleep and sleep decline were also linked to earlier AAO (up to –4.23 years) across cohorts. These findings suggest that midlife sleep reduction may signal early PD, while chronic short sleep may represent a modifiable risk factor, highlighting the need for prospective studies to explore early detection and prevention potential.
Similar content being viewed by others
Introduction
Sleep disturbances are common nonmotor symptoms of Parkinson’s disease (PD), affecting 60% to 90% of patients1,2. Evidence indicates a bidirectional relationship between PD and altered sleep, even before symptomatic onset3,4. Sleep deprivation exacerbates synucleinopathy in rodent models5, and poor sleep in human patients is associated with faster motor progression6, indicating the role of sleep disturbances in PD pathogenesis. Furthermore, PD pathology disrupts the sleep/wake neural circuitry before overt clinical motor symptoms3. This disruption is exemplified by rapid eye movement sleep behavioral disorder (RBD), a well-established prodromal marker of PD7.
Population-based studies report higher rates of insomnia or hypersomnia8,9,10, and variations in sleep duration11,12 preceding PD diagnosis. However, these studies typically focus on isolated time points, primarily the decade before diagnosis, overlooking potential life course changes in sleep duration13. Sleep trajectories have been associated with adverse neurological outcomes, such as dementia14,15,16, but no study has examined the association between life course sleep duration and PD. Given that prodromal PD can span decades17 and early-life sleep patterns may contribute to neurodegeneration later in life18, it is crucial to investigate early-life sleep trajectories and their connection to PD onset. Such research is critical for disentangling the role of sleep disturbances as early or risk factors for PD, offering opportunities for early detection and intervention.
In this study, we identified self-reported sleep duration trajectories from adolesence to late adulthood using data from the Parkinson’s Progression Marker Initiative Online (PPMI-Online) and Fox Insight (FI). We assessed their relationship with PD risk and age at onset (AAO) in PPMI-Online, validating AAO findings in FI. We hypothesized that midlife sleep changes could serve as early markers of PD and that sleep durations below recommendations (<7 h/night19,20) in early adulthood or midlife would be associated with higher risk and earlier AAO of PD. We further examined whether these patterns were also associated with probable prodromal PD, as defined by the MDS research criteria21. Sensitivity analyses excluded participants with self-reported RBD, anxiety or depression, dementia, and examined sex as a potential effect modifier.
Results
Study participants
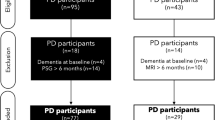
A flowchart of study participants is presented in Fig. 1. The final sample in the discovery cohort included 11,728 participants from PPMI-Online (5660 with PD and 10,245 without PD). Among the latter, 6922 had complete data and were classified as “probable prodromal PD (pPD)” (n = 1104) or “no probable pPD” (n = 5818) based on the Movement Disorders Society criteria21 (80% probability cutoff22). The validation FI sample included 1929 participants with PD; the 338 participants without PD were excluded due to limited sample size.
PD Parkinson’s disease, pPD prodromal Parkinson’s disease.
For the current analysis, we applied identical exclusion criteria to both cohorts, including age at registration <50 years, young-onset PD (AAO < 50 years23), diagnosis changed during follow-up, missing age at diagnosis, reported age at diagnosis older than the participant’s current age, and all sleep duration responses marked as “prefer not to answer” or “don’t know”.
Demographic and clinical characteristics of participants is summarized in Table 1: 15,905 from PPMI-Online [mean age at sleep report: 67.2 ± 7.94 years; 8795 (55.3%) female; mean follow-up: 22.7 ± 8.71 months] and 1929 from FI [mean age at sleep report: 67.5 ± 6.87 years; 940 (48.7%) female; mean follow-up: 68.1 ± 10.1 months]. PD mean AAO was 65.3 ± 7.57 in PPMI-Online and 62.9 ± 6.96 in FI. In PPMI-Online, participants without PD were younger, more likely female, and had higher education, income, and positive PD family history compared to those with PD. Across subgroups (“no probable pPD,” “probable pPD,” and “PD”; Supplmentary Table 1), motor and non-motor symptoms showed a clear progression, supporting the validity of the pPD criteria.
Sleep duration trajectories (PPMI-Online)
PPMI-Online sleep duration trajectories (Fig. 2A) remained relatively stable from the 20s to the 40s, with distinct trends emerging after the 50s (depicted in separate panels). Groups were labeled according to both baseline sleep in early adulthood (with categories: >8 h, 7–8 h, 6–7 h, ≤6 h) and changes after age 50 (categorized as: stable, decrease, or increase). These trajectory shapes were largely consistent in sensitivity analyses. The “≤6 h stable” group had a lower age at sleep report, smaller percentage of White individuals, fewer years of education, and lower household income. Groups with changes after 50s (“7–8 h increase”, “6–7 h decrease”, “7–8 h decrease”) had higher age at sleep report and lower income. The “>8 h stable” group included more females (Supplementary Table 2).
PD Parkinson’s disease, pPD prodromal Parkinson’s disease. Panel A: Sleep duration trajectory groups identified through latent class growth analysis, shown in separate panels based on trends emerging after age 50. Dots and shaded areas represent mean trajectories with 95% confidence intervals. The accompanying table shows the count and proportion of participants for each trajectory pattern. Panel B: Logistic regression results for the association between sleep duration trajectory patterns and risk of PD, using participants without a PD diagnosis as the reference group. Panel C: Logistic regression of sleep duration trajectory patterns and risk of “Probable pPD”, with the “No probable pPD” group as the reference. Model 1 was unadjusted. Model 2 adjusted for age at sleep report, sex, race, family history of PD, education, and income. Model 3 further adjusted for history of anxiety, depression, brain injury, diabetes, hypertension; Rapid Eye Movement Sleep Behavior Disorder Single-Question Screen at the time of sleep report; lifetime caffeine intake, smoking status, and lifetime physical inactivity.
Sleep duration trajectories and PD risk (PPMI-Online)
Compared to the “7–8 h stable” group, those with consistently shorter sleep (“≤6 h stable”: OR = 1.41, 95% CI 1.19–1.67, P < 0.001, all ORs from Model 3) or decreasing sleep after age 50 (“6–7 h decrease”: OR = 1.90, 95% CI 1.61–2.24, P < 0.001, “7–8 h decrease”: OR = 1.64, 95% CI 1.40–1.91, P < 0.001) showed increased PD risk. The associations remained consistent across all adjustments (Fig. 2B). The “6–7 h stable” group also showed a modestly higher risk of PD after adjustment (OR = 1.15, 95% CI 1.02–1.30, P = 0.02).
For probable pPD, consistently shorter sleep (“≤6 h stable”: OR = 1.61, 95% CI 1.10–2.32, P = 0.01) and decreasing sleep after 50 (“6–7 h decrease”: OR = 1.94, 95% CI 1.35–2.77, P < 0.001; “7–8 h decrease”: OR = 1.85, 95% CI 1.33–2.56, P < 0.001) were similarly associated with higher risks, consistent with the findings for PD risk. In addition, the “≤6 h increase” group also exhibited elevated pPD risk (OR = 1.73, 95% CI 1.12–2.62, P = 0.01).
Sleep duration trajectories and PD AAO (PPMI-Online)
Compared to the “7–8 h stable” group, short sleep in early adulthood or decreasing sleep in midlife was associated with earlier PD AAO (Fig. 3). After full adjustment, the largest effect size was observed in the “≤6 h stable” group (β = −2.45 years, 95% CI −3.33 to −1.56, P = < 0.001) and “6–7 h decrease” group (β = −2.37 years, 95% CI −3.19 to −1.56, P = < 0.001), followed by the “≤6 h increase” (β = −1.73 years, 95% CI −2.85 to −0.62, P = 0.002) and “6–7 h stable” (β = −0.91 years, 95% CI −1.57 to −0.26, P = 0.006).
Model 1 was unadjusted. Model 2 adjusted for sex, race, family history of PD, education, and income. Model 3 further adjusted for history of anxiety, depression, brain injury, diabetes, hypertension; Rapid Eye Movement Sleep Behavior Disorder Single-Question Screen at the time of sleep report; lifetime caffeine intake, smoking status, and lifetime physical inactivity.
Sleep duration trajectories and associations with PD AAO (FI)
Most FI trajectories (Fig. 4A) exhibited moderate sleep decline from adolescence to adulthood. Among them, four groups (labeled “>8 h”, “7–8 h”, “6–7 h”, “≤6 h”) maintained relatively stable during midlife. Two groups showed continuous declines: “decrease-1,” with decreases earlier in life, and “decrease-2,” with declines more evident during midlife. The “increase” group exhibited a sleep increase after the 40s, with early adulthood sleep duration between 6–7 h, resembling the “6–7 h increase” group in PPMI-Online. Groups with short sleep (“≤6 h”, “6–7 h”) and decreasing sleep (“decrease-1”, “decrease-2”) had lower education levels. The decrease groups also had a younger age at sleep report (Supplmentary Table 3).
Panel A: Dots and shaded areas represent mean trajectories for each class with 95% confidence intervals. The table displays the count and proportion of each trajectory pattern. Panel B: Sleep duration trajectory patterns and age at onset of PD. Model 1 was unadjusted. Model 2 adjusted for sex, race, family history of PD, education, and income. Model 3 further adjusted for history of anxiety, depression, brain injury, diabetes, hypertension; Rapid Eye Movement Sleep Behavior Disorder Single-Question Screen at the time of sleep report; lifetime caffeine intake, smoking status, and lifetime physical inactivity.
Compared to the “7–8 h” group, groups with less sleep exhibited earlier AAO (Fig. 4B). The effect size was most pronounced in the decrease groups (“decrease-1”: β = −4.23 years, 95% CI −5.52 to −2.93, P < 0.001; “decrease-2” (β = −3.49 years, 95% CI −4.46 to −2.51, P < 0.001), followed by “≤6 h” (β = −2.70 years, 95% CI −3.80 to −1.59, P < 0.001), “increase” (β = −1.65 years, 95% CI −3.04 to −0.26, P = 0.02), and “6–7 h” (β = −1.37 years, 95% CI −2.16 to −0.57, P < 0.001).
Sensitivity analyses
The associations remained consistent across sexes and were robust among participants without RBD symptoms or diagnoses throughout follow-up, those without a history of anxiety or depression, and after excluding individuals with possible dementia (Supplementary Figs. 1–7). Additionally, shorter baseline sleep duration and decreasing sleep during midlife were independently linked to an increased PD risk and earlierer AAO (Supplementary Fig. 8).
Discussion
In two large, PD-centered online cohorts, we identified distinct self-reported life course sleep duration trajectories and examined their associations with PD risk and AAO. PPMI-Online results highlighted midlife, particularly around age 50, as a critical turning point in sleep duration. Declining sleep during midlife was associated with up to a 90% higher risk of PD and pPD. Chronic short sleep from early adulthood was associated with a 1.4-fold increased PD risk. Additionally, both PPMI-Online and FI cohorts indicated that shorter-than-recommended sleep in early adulthood or midlife was associated with an earlier AAO, with such patterns associated with earlier AAO by up to 2.5 to 4.2 years.
PPMI-Online and FI trajectories exhibited both shared and distinct patterns, likely reflecting differences in life stage coverage and participant demographics. Around 50% of PPMI-Online participants were in trajectory groups showing changes after age 50, and 60% of FI participants belonged to groups showing declines from adolescence to the 30s. These findings align with previously identified turning points in sleep duration at ages 33 and 5313. However, stable groups might be overestimated due to categorical presentation, potentially missing subtle changes.
Midlife (ages 40–60) is increasingly recognized as a critical period marked by accelerated declines in gene expressions, brain connectivity, and cognitive function24,25. We observed that decreasing sleep after age 50 was associated with increased PD and pPD risk, suggesting that declining sleep is evident in prodromal PD and continues into clinical PD. This association remained robust even among participants without self-reported RBD, anxiety, or depression, indicating that decreasing sleep may be an early manifestation of PD itself, rather than a consequence of common prediagnostic comorbidities observed in a subset of patients26. Conversely, the “≤6 h increase” group showed an increased risk of pPD but not PD, suggesting that increasing sleep during midlife, particularly after chronic short sleep, may indicate prodromal PD. However, our study lacked an ultra-long sleep group (>8 h/day), limiting comparisons to previous reports linking ≥9 h/day with higher PD risk27,28.
Given the long prodromal window, it is challenging to determine the direction of the relationship between sleep disturbances and PD. Our study indicates that short sleep in early adulthood or midlife is associated with increased risk and earlier AAO of PD, potentially preceding the onset of PD by up to six decades. These findings suggest that chronic short sleep might be a risk factor, with prolonged short sleep likely preceding neurodegeneration. Our results also underscore the public health relevance of addressing insufficient sleep, particularly in minoritized populations29, and its adverse effects on aging-related outcomes30. As primary or secondary preventive trials for PD are being planned31, promoting healthy sleep hygiene may represent a scalable strategy for reducing risk or delaying onset.
Our findings align with mechanistic evidence from animal and human studies. Sleep disturbances may arise from early PD pathology affecting the locus coeruleus32, hypothalamus33, and dopaminergic systems34. Conversely, sleep deprivation exacerbates synucleinopathy5, possibly through mitochondrial and oxidative stress35, neuroinflammation36, impaired glymphatic clearance37, and synaptic homeostasis38. Notably, the locus coeruleus is particularly vulnerable to insufficient sleep and may contribute to PD pathogenesis39,40. Furthermore, chronic sleep loss in young adult rodents has long-lasting neurological effects41, and is associated with neurodegeneration later in life18, supporting our findings that short sleep in early adulthood might contribute to PD in later years.
This study offers a novel life course perspective by leveraging two large, PD-centered online cohorts with a large participant pool and sleep duration data spanning adolescence to age 80. However, several limitations should be considered. First, the retrospective self-reported sleep duration may introduce recall bias, although evidence suggests such information can be accurate42,43. In this study, participants could select “don’t know” for uncertain responses, and results remained robust after excluding possible dementia. Additionally, the sleep trajectory turning points aligned with a previous large-scale study13, enhancing the credibility of self-reported sleep. While self-reported sleep duration modestly correlates with objectively measured sleep44, subjective pereptions may still capture clinically relevant perceptual aspects of sleep disruption45,46. A related concern is that recall may differ by diagnostic status, potentially biasing associations. Notably, similar patterns were seen in participants with prodromal PD, even without a PD diagnosis, supporting a link to underlying disease rather than recall bias. Second, pPD was classified solely using self-reported markers, without confirmation from clinician-based evaluations or objective biomarkers such as DAT-SPECT or synuclein-based assays. This limitation may increase the risk of both false-positive and false-negative classifications. However, the observed stepwise progression of motor and non-motor scores across the “no probable pPD”, “probable pPD”, and “PD” groups supports the internal validity of the pPD classification in our study. Furthermore, the study population was predominantly White, with high socioeconomic status and education, and participants without PD were enriched for PD risk factors. Further studies are needed to assess generalizability. Finally, while the findings suggest short sleep as a potential PD risk factor, the observational nature of this study limits causal inferences. Future research should also consider the potential of major events like the COVID-19 pandemic, which may have affected sleep patterns differently across patients and age groups.
In this large-scale study involving individuals with and without PD, we found that the 50s represent a significant turning point in sleep duration. Monitoring self-perceived sleep duration changes, particularly after age 50, is crucial, as declining sleep duration at this stage may signal a higher risk and earlier onset of PD. Additionally, chronic short sleep was associated with an increased risk and earlier AAO of PD, suggesting short sleep since early adulthood as a potential risk factor and intervention targets. These findings support a life course framework for understanding the role of sleep in PD pathogenesis and underscore the need for future prospective studies to validate these associations and examine other sleep dimensions for early detection and prevention strategies across the life span.
Methods
PPMI-online and FI study design
PPMI-Online and FI are ongoing online longitudinal observational cohorts involving participants with and without PD. Convenience sampling was used to recruit participants aged 18 and older with minimal eligibility criteria. Recruitment was conducted through online channels, such as social media and email outreach, and on-site methods, including clinician referral and PD-related research events. Recruitment began in 2017 for FI47,48 and 2021 for PPMI-Online49. Both cohorts rely on repeated web-based assessments of PD status, symptoms, comorbidities, and lifestyle factors, thereby broadening the representativeness of PD research beyond clinic-based samples. Participants report PD status and symptoms quarterly via an online platform and complete occasional questionnaires, including those on sleep duration. The sleep questionnaire was administered during the 3rd and 15th months for PPMI-Online and between October 2017 and March 2019 for the FI study. For participants with multiple sleep reports, the most recent data were used for analysis. Study protocols were approved by institutional review boards (FI: New England IRB# 120160179; PPMI-Online: WCG IRB# 20211908), and all participants provided online informed consent. This study was conducted in accordance with the Declaration of Helsinki.
Self-perceived sleep duration across the lifespan
Participants reported their typical sleep duration during current and prior age periods. Response options were categorical and included: less than 5 h, 5–6 h, 6–7 h, 7–8 h, more than 8 h, don’t know, and prefer not to answer. Responses of “don’t know” or “prefer not to answer” were treated as missing values.The PPMI-Online study captured data spanning adulthood, with more granular age categories during late adulthood (18–29, 30–39, 40–49, 50–59, 60–64, 65–69, 70–74, 75–79, and 80 or older). The FI study focused on earlier life stages (12–17, 18–25, 26–35, 36–45, 46–55, 56–65, and 66 or older), capturing transitions from adolescence to adulthood.
Outcome measures
PD status was based on self-reported physician diagnosis at registration and follow-up, with previous studies confirming the reliability of self-reported PD in an online setting50. The self-reported age at first PD diagnosis was used as a proxy for PD AAO, as these measures were highly correlated51.
Motor and non-motor symptom measures
Several standardized instruments were collected to characterize the clinical profile of study participants, and their descriptive results are summarized in Table 1. The Movement Disorder Society–Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) parts I and II evaluate non-motor and motor aspects of daily living, respectively52. The Penn Parkinson’s Daily Activities Questionnaire–15 (PDAQ-15) measures daily cognitive functioning in PD53. The Geriatric Depression Scale–15 (GDS-15)54 and the Parkinson Anxiety Scale (PAS)55 assess depressive and anxiety symptoms. The Epworth Sleepiness Scale (ESS)56 evaluates excessive daytime sleepiness, while the Parkinson’s Disease Sleep Scale–2 (PDSS-2)57 captures PD-specific sleep disturbances. The RBD Single-Question Screen (RBD1Q)58 was used to assess dream enactment behavior suggestive of REM sleep behavior disorder. Higher scores indicate greater symptom burden for all instruments, except for the PDAQ-15, where higher scores reflect worse cognition.
For the present analysis, we used the assessment conducted at the same visit as the sleep duration questionnaire (or the closest available visit if not collected at the same time) to align symptom measures with the timing of the sleep report.
Statistical analysis
We used latent class growth analysis (LCGA) to classify individuals into distinct sleep duration trajectory groups separately for PPMI-Online and FI, incorporating both linear and quadratic time components. LCGA identifies latent classes with similar trajectory patterns over time, assuming homogeneity within each class59. In the initial phase, we identified trajectory groups in participants with complete sleep duration data from ages 18–29 to 75–79 for PPMI-Online (n = 2643) and ages 12–17 to 56–65 for FI (n = 1668), with the midpoint of each age range approximating age. We excluded the “80 or older” and “66 or older” groups due to limited sample sizes and broad age ranges. Participants lacking sleep data for all required periods were excluded to avoid overrepresentation of younger groups and sparse data for older groups. Those who selected “prefer not to answer” or “not sure” for any age period were also excluded to avoid potential bias from recall errors. In the next phase, all participants with at least one valid data point were assigned to the trajectory class with the highest membership probability. To assess trajectory consistency and robustness, we visually compared trajectory groups from (1) participants with complete data across age periods (main analysis) versus all participants, (2) those with PD versus without PD, and (3) those with RBD symptoms/diagnoses versus those without RBD. We tested models with 1–9 classes, selecting the optimal model based on lower Bayesian Information Criteria (BIC) and interpretability60. BIC decreased as the number of classes increased from 1 to 9 for both PPMI-Online and FI. We selected the 9-class models for PPMI-Online and the 7-class model for FI, as additional classes showed similar patterns with no meaningful differences and limited sample size. Further LCGA details are provided in Supplementary.
We analyzed associations between sleep duration trajectories and PD risk using logistic regression, and PD AAO using linear regression. Given possible non-monotonic changes during the prodromal phase61,62, we performed seperate binomial logistic regressions for “PD” (with “No PD” as the refrence) and “probable pPD” (with “no probable pPD” as the reference). We built three models: Model 1 (unadjusted), Model 2 (adjusted for age at sleep report [for risk analyses only], sex, race, family history of PD, education, and income), and Model 3 (further adjusted for self-reported lifetime history of anxiety, depression, brain injury, diabetes, hypertension; RBD Single-Question Screen63 at the time of sleep report; lifetime caffeine intake, smoking status, and lifetime physical inactivity). We did not include age at sleep report as a covariate for AAO as AAO typically precedes the sleep report and is minimally influenced by the age at sleep report. Covariates, selected based on their known or suspected associations with sleep and PD64, were self-reported, with the closest report to sleep report used for repeated measures. All models met assumptions, with no variance inflation factor exceeding 5, indicating minimal multicollinearity.
Sensitivity analyses included: (1) restricting to participants without self-reported RBD (n = 8395), given its association with shorter sleep65, (2) restricting to participants without history of anxiety or depression (n = 8566), given the divergent effects of these conditions and their treatments on sleep, (3) excluding participants with possible dementia (Penn Parkinson’s Daily Activities Questionnaire-15 < 4353) to enhance recall accuracy (n = 14,405), and (4) exploring sex as a possible effect modifier. Furthermore, as baseline sleep during early adulthood and trends during midlife are critical components of the sleep duration trajectories, we also investigated whether these two factors are independently associated with PD risk and AAO.
We performed data analysis from January to December 2024 using R (version 4.3.2). Major packages included “lcmm”66 (version 2.1.0), “stats” (version 4.3.2), “ggplot2” (version 3.4.4). All tests were 2-sided, with statistical significance set at P < 0.05. This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline.
Data availability
Final data used in this article were obtained from the PPMI study (tier1 data downloaded on 03 July 2024, [https://www.ppmi-info.org/access-data-specimens/download-data](https:/www.ppmi-info.org/access-data-specimens/download-data), RRID:SCR\_006431) and the FI study (version: December 2024 archived monthly data, [https://foxinsight-info.michaeljfox.org/insight/explore/insight.jsp](https:/foxinsight-info.michaeljfox.org/insight/explore/insight.jsp)). For up-todate information on the PPMI study, visit [www.ppmi-info.org](http:/www.ppmi-info.org). For up-to-date information on the FI study, visit [https://foxinsightinfo.michaeljfox.org/insight/explore/insight.jsp](https:/foxinsightinfo.michaeljfox.org/insight/explore/insight.jsp).
References
Lajoie, A. C., Lafontaine, A.-L. & Kaminska, M. The spectrum of sleep disorders in Parkinson disease: a review. Chest 159, 818–827 (2021).
Iranzo, A., Cock, V. C. D., Fantini, M. L., Pérez-Carbonell, L. & Trotti, L. M. Sleep and sleep disorders in people with Parkinson’s disease. Lancet Neurol. 23, 925–937(2024).
Al-Qassabi, A., Fereshtehnejad, S.-M. & Postuma, R. B. Sleep disturbances in the prodromal stage of Parkinson disease. Curr. Treat. Options Neurol. 19, 22 (2017).
Leng, Y. et al. Association of circadian abnormalities in older adults with an increased risk of developing Parkinson disease. JAMA Neurol. 77, 1270–1278 (2020).
Morawska, M. M. et al. Slow-wave sleep affects synucleinopathy and regulates proteostatic processes in mouse models of Parkinson’s disease. Sci. Transl. Med. 13, eabe7099 (2021).
Schreiner, S. J. et al. Slow-wave sleep and motor progression in Parkinson disease. Ann. Neurol. 85, 765–770 (2019).
Postuma, R. B. et al. Risk and predictors of dementia and parkinsonism in idiopathic REM sleep behaviour disorder: a multicentre study. Brain 142, 744–759 (2019).
Schrag, A., Horsfall, L., Walters, K., Noyce, A. & Petersen, I. Prediagnostic presentations of Parkinson’s disease in primary care: a case-control study. Lancet Neurol. 14, 57–64 (2015).
Simonet, C. et al. Assessment of risk factors and early presentations of Parkinson disease in primary care in a diverse UK population. JAMA Neurol. 79, 359 (2022).
Schrag, A. et al. Widening the spectrum of risk factors, comorbidities, and prodromal features of Parkinson disease. JAMA Neurol. 80, 161–171 (2023).
Otaiku, A. I. Association of sleep abnormalities in older adults with risk of developing Parkinson’s disease. Sleep 45, zsac206 (2022).
Lysen, T. S., Darweesh, S. K. L., Ikram, M. K., Luik, A. I. & Ikram, M. A. Sleep and risk of parkinsonism and Parkinson’s disease: a population-based study. Brain 142, 2013–2022 (2019).
Coutrot, A. et al. Reported sleep duration reveals segmentation of the adult life-course into three phases. Nat. Commun. 13, 7697 (2022).
Cavaillès, C. et al. Trajectories of sleep duration and timing before dementia: a 14-year follow-up study. Age Ageing 51, afac186 (2022).
Lu, Y., Sugawara, Y., Zhang, S., Tomata, Y. & Tsuji, I. Changes in sleep duration and the risk of incident dementia in the elderly Japanese: the Ohsaki Cohort 2006 Study. Sleep 41, zsy143 (2018).
Sabia, S. et al. Association of sleep duration in middle and old age with incidence of dementia. Nat. Commun. 12, 2289 (2021).
Berg, D. et al. Prodromal Parkinson disease subtypes—key to understanding heterogeneity. Nat. Rev. Neurol. 17, 349–361 (2021).
Owen, J. E. et al. Late-in-life neurodegeneration after chronic sleep loss in young adult mice. Sleep 44, zsab057 (2021).
Watson, N. F. et al. Recommended amount of sleep for a healthy adult: a joint consensus statement of the American Academy of Sleep Medicine and Sleep Research Society. SLEEP https://doi.org/10.5665/sleep.4716 (2015).
Hirshkowitz, M. et al. National Sleep Foundation’s updated sleep duration recommendations: final report. Sleep. Health 1, 233–243 (2015).
Heinzel, S. et al. Update of the MDS research criteria for prodromal Parkinson’s disease. Mov. Disord. 34, 1464–1470 (2019).
Berg, D. et al. MDS research criteria for prodromal Parkinson’s disease. Mov. Disord. 30, 1600–1611 (2015).
Mehanna, R. et al. Age cutoff for early-onset Parkinson’s disease: recommendations from the International Parkinson and Movement Disorder Society Task Force on early onset Parkinson’s disease. Mov. Disord. Clin. Pract. 9, 869–878 (2022).
Dohm-Hansen, S. et al. The ‘middle-aging’ brain. Trends Neurosci. 47, 259–272 (2024).
Yaffe, K. et al. Cardiovascular risk factors and accelerated cognitive decline in midlife: the CARDIA Study. Neurology 95, e839–e846 (2020).
Blesa, J., Foffani, G., Dehay, B., Bezard, E. & Obeso, J. A. Motor and non-motor circuit disturbances in early Parkinson disease: which happens first? Nat. Rev. Neurosci. 23, 115–128 (2022).
Beydoun, H. A. et al. Association of sleep disturbance with Parkinson disease: evidence from the Women’s Health Initiative. Menopause 29, 255–263 (2022).
Chen, Y. et al. Association between sleep factors and Parkinson’s disease: a prospective study based on 409,923 UK biobank participants. Neuroepidemiology 57, 293–303 (2023).
Jean-Louis, G., Grandner, M. A. & Seixas, A. A. Social determinants and health disparities affecting sleep. Lancet Neurol. 21, 864–865 (2022).
Leng, Y., Knutson, K., Carnethon, M. R. & Yaffe, K. Association between sleep quantity and quality in early adulthood with cognitive function in midlife. Neurology 102, e208056 (2024).
Berg, D. et al. Path to Parkinson disease prevention. Neurology 99, 76–83 (2022).
Braak, E. et al. α-Synuclein immunopositive Parkinson’s disease-related inclusion bodies in lower brain stem nuclei. Acta Neuropathol. 101, 195–201 (2001).
De Pablo-Fernández, E. & Warner, T. T. Hypothalamic α-synuclein and its relation to autonomic symptoms and neuroendocrine abnormalities in Parkinson disease. in Handbook of Clinical Neurology, Vol. 182 223–233 (Elsevier, 2021).
Holst, S. C. et al. Dopaminergic role in regulating neurophysiological markers of sleep homeostasis in humans. J. Neurosci. 34, 566–573 (2014).
Zamore, Z. & Veasey, S. C. Neural consequences of chronic sleep disruption. Trends Neurosci. 45, 678–691 (2022).
Zhu, B. et al. Sleep disturbance induces neuroinflammation and impairment of learning and memory. Neurobiol. Dis. 48, 348–355 (2012).
Xie, L. et al. Sleep drives metabolite clearance from the adult brain. Science 342, 373–377 (2013).
Zahed, H. et al. The Neurophysiology of sleep in Parkinson’s disease. Mov. Disord. 36, 1526–1542 (2021).
Butkovich, L. M. et al. Transgenic mice expressing human α-synuclein in noradrenergic neurons develop locus ceruleus pathology and nonmotor features of Parkinson’s disease. J. Neurosci. 40, 7559–7576 (2020).
Ray Chaudhuri, K., Leta, V., Bannister, K., Brooks, D. J. & Svenningsson, P. The noradrenergic subtype of Parkinson disease: from animal models to clinical practice. Nat. Rev. Neurol. 19, 333–345 (2023).
Yuan, R. K. et al. Differential effect of sleep deprivation on place cell representations, sleep architecture, and memory in young and old mice. Cell Rep. 35, 109234 (2021).
Berney, L. R. & Blane, D. B. Collecting retrospective data: accuracy of recall after 50 years judged against historical records. Soc. Sci. Med. 45, 1519–1525 (1997).
Havari, E. & Mazzonna, F. Can we trust older people’s statements on their childhood circumstances? Evidence from SHARELIFE. Eur. J. Popul. 31, 233–257 (2015).
Lauderdale, D. S., Knutson, K. L., Yan, L. L., Liu, K. & Rathouz, P. J. Self-reported and measured sleep duration: how similar are they? Epidemiology 19, 838–845 (2008).
Edinger, J. D. & Fins, A. I. The distribution and clinical significance of sleep time misperceptions among insomniacs. Sleep 18, 232–239 (1995).
Trimmel, K. et al. The (mis)perception of sleep: factors influencing the discrepancy between self-reported and objective sleep parameters. J. Clin. Sleep Med. 17, 917–924 (2021).
Smolensky, L. et al. Fox Insight collects online, longitudinal patient-reported outcomes and genetic data on Parkinson’s disease. Sci. Data 7, 67 (2020).
Gottesman, J. et al. Fox Insight at 5 years—a cohort of 54,000 participants contributing longitudinal patient-reported outcome, genetic, and microbiome data relating to Parkinson’s disease. Sci. Data 11, 615 (2024).
M Tanner, C. & Marek, K. Parkinson’s progression markers initiative online study (PPMI online study). Preprint at https://doi.org/10.17504/protocols.io.q26g718y9gwz/v2 (2025).
Myers, T. L. et al. Video-based Parkinson’s disease assessments in a nationwide cohort of Fox Insight participants. Clin. Parkinsonism Relat. Disord. 4, 100094 (2021).
Pihlstrøm, L. et al. Genetic stratification of age-dependent Parkinson’s disease risk by polygenic hazard score. Mov. Disord. 37, 62–69 (2022).
Goetz, C. G. et al. Movement disorder society-sponsored revision of the unified Parkinson’s disease rating scale (MDS-UPDRS): process, format, and clinimetric testing plan. Mov. Disord. 22, 41–47 (2007).
Brennan, L. et al. The Penn Parkinson’s Daily Activities Questionnaire-15: psychometric properties of a brief assessment of cognitive instrumental activities of daily living in Parkinson’s disease. Parkinsonism Relat. Disord. 25, 21–26 (2016).
Yesavage, J. A. & Sheikh, J. I. Geriatric depression scale (GDS): recent evidence and development of a shorter version. Clin. Gerontologist 5, 165–173 (1986).
Leentjens, A. F. G. et al. The Parkinson anxiety scale (PAS): development and validation of a new anxiety scale. Mov. Disord. 29, 1035–1043 (2014).
Johns, M. W. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 14, 540–545 (1991).
Trenkwalder, C. et al. Parkinson’s disease sleep scale—validation of the revised version PDSS-2. Mov. Disord. 26, 644–652 (2011).
Postuma, R. B. et al. A single-question screen for REM sleep behavior disorder: a multicenter validation study. Mov. Disord. 27, 913–916 (2012).
Jung, T. & Wickrama, K. A. S. An introduction to latent class growth analysis and growth mixture modeling. Soc. Personal. Psychol. Compass 2, 302–317 (2008).
Lennon, H. et al. Framework to construct and interpret latent class trajectory modelling. BMJ Open 8, e020683 (2018).
Postuma, R. B., Gagnon, J.-F., Pelletier, A. & Montplaisir, J. Y. Insomnia and somnolence in idiopathic RBD: a prospective cohort study. npj Parkinson’s Dis. 3, 1–6 (2017).
Joza, S. et al. Progression of clinical markers in prodromal Parkinson’s disease and dementia with Lewy bodies: a multicentre study. Brain 146, 3258–3272 (2023).
Postuma, R. B. et al. A single-question screen for rapid eye movement sleep behavior disorder: a multicenter validation study. Mov. Disord. 27, 913–916 (2012).
Ben-Shlomo, Y. et al. The epidemiology of Parkinson’s disease. Lancet 403, 283–292 (2024).
Zhang, Y., Ren, R., Yang, L., Sanford, L. D. & Tang, X. Polysomnographically measured sleep changes in idiopathic REM sleep behavior disorder: A systematic review and meta-analysis. Sleep. Med. Rev. 54, 101362 (2020).
Proust-Lima, C., Philipps, V. & Liquet, B. Estimation of extended mixed models using latent classes and latent processes: the R package lcmm. J. Stat. Softw. 78, 1–56 (2017).
Acknowledgements
This study is supported by NIH NIA R01AG083836 (PI: Y.L.).We would like to thank the Parkinson’s community for participating in the Fox Insight and PPMI-Online study to make this research possible. The Fox Insight Study is funded by The Michael J. Fox Foundation for Parkinson’s Research. PPMI – a public-private partnership – is funded by the Michael J. Fox Foundation for Parkinson’s Research and funding partners, including 4D Pharma, Abbvie, AcureX, Allergan, Amathus Therapeutics, Aligning Science Across Parkinson's, AskBio, Avid Radiopharmaceuticals, BIAL, BioArctic, Biogen, Biohaven, BioLegend, BlueRock Therapeutics, Bristol-Myers Squibb, Calico Labs, Capsida Biotherapeutics, Celgene, Cerevel Therapeutics, Coave Therapeutics, DaCapo Brainscience, Denali, Edmond J. Safra Foundation, Eli Lilly, Gain Therapeutics, GE HealthCare, Genentech, GSK, Golub Capital, Handl Therapeutics, Insitro, Jazz Pharmaceuticals, Johnson & Johnson Innovative Medicine, Lundbeck, Merck, Meso Scale Discovery, Mission Therapeutics, Neurocrine Biosciences, Neuron23, Neuropore, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi, Servier, Sun Pharma Advanced Research Company, Takeda, Teva, UCB, Vanqua Bio, Verily, Voyager Therapeutics, the Weston Family Foundation and Yumanity Therapeutics.
Author information
Authors and Affiliations
Contributions
Y.F. and Y.L. designed the study. Y.F. performed the analysis and drafted the manuscript. All authors contributed to data interpretation and critical revision of the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
SL has previously been consulted for Iota Biosciences. CMT declares consultancies for CNS Ratings, Australian Parkinson’s Mission, Biogen, Evidera, Cadent (data safety monitoring board), Adamas (steering committee), Biogen (via the Parkinson Study Group steering committee), Praxis (via the International Parkinsons and Movement Disorders Society), Kyowa Kirin (advisory board), Lundbeck (advisory board), Jazz/Cavion (steering committee), Acorda (advisory board), Bial (DMC) and Genentech. CMT also declares grant support to her institution from The Michael J. Fox Foundation, National Institutes of Health, Gateway LLC, Department of Defense, Roche Genentech, Biogen, Parkinson Foundation and Marcus Program in Precision Medicine. CMT declares membership on the editorial boards for the Journal of Parkinson’s Disease and the Journal of Neurology. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fang, Y., Hardy, R., Yaffe, K. et al. Self-perceived life course sleep duration trajectories and risk and age at onset of Parkinson’s disease. npj Parkinsons Dis. 11, 357 (2025). https://doi.org/10.1038/s41531-025-01202-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41531-025-01202-w