Abstract
Schizophrenia is highly heritable, and polygenic risk score for schizophrenia (PRSSCZ) has been associated with brain and behavior in healthy populations. However, the full associations of PRSSCZ with brain white matter microstructure and cognitive and mental health outcomes, the potential effects of sex and areal deprivation on these associations, and the mediation of white matter microstructure for the associations between PRSSCZ and behavioral outcomes remain largely unknown. In up to 300,000 participants from the UK Biobank, we investigated the associations of PRSSCZ with eight white matter microstructure metrics of 48 tracts and 14 cognitive and mental health phenotypes, and we further tested the moderation of sex and index of multiple deprivation (IMD) on these associations and the mediation of brain white matter phenotypes for the associations between PRSSCZ and behavioral outcomes. We found that higher PRSSCZ was associated with decreased white matter integrity in 26 tracts, such as cingulum, corona radiata, and fornix. We also found that higher PRSSCZ was associated with poorer mental health and worse cognitive performance. These associations were not significantly moderated by sex and IMD. Causal mediation analyses revealed that these adverse effects of PRSSCZ on cognitive and mental health outcomes were partially mediated by brain white matter phenotypes. These results indicate that genetic risk for schizophrenia affects the integrity of white matter tracts, which may account for its adverse effects on cognitive and mental health outcomes.
Similar content being viewed by others
Introduction
Schizophrenia is a chronic disabling mental disorder characterized by hallucinations, delusions, confusion, disorganization, and disturbances in motivation, cognition and socialization1,2. It is highly heritable and polygenic, with genetic factors contributing significantly to its etiology, evidenced by an estimated twin heritability of around 80%3,4,5 and single-nucleotide polymorphism heritability of ~24%6. For instance, a recent genome-wide association study (GWAS) has identified 287 genomic loci associated with schizophrenia6. An individual’s lifetime genetic risk for schizophrenia can be assessed by a polygenic risk score (PRS). Since an individual’s PRS for schizophrenia is innate and remains stable throughout their lifetime, genetic risk factors may lead to brain and behavioral changes much earlier before the onset of the disorder.
Brain structure, function, and connection can be assessed by magnetic resonance imaging (MRI) techniques. Patients with schizophrenia have demonstrated extensive structural, functional, and connectivity impairments compared to healthy controls7,8,9,10,11,12,13,14,15,16,17,18,19. In addition to classical symptoms, patients with schizophrenia also demonstrate other cognitive and mental impairments, such as the decline in memory, attention, and execution as well as the symptoms of anxiety and depression20,21,22,23,24. Furthermore, a large portion of these brain and behavioral impairments have been observed in offsprings of parents with schizophrenia25,26,27,28, indicating that genetic risk for schizophrenia may at least partially contribute to these impairments. Therefore, researchers have explored whether PRS for schizophrenia (PRSSCZ) could affect brain and behavior in healthy populations. Although many studies have revealed the associations of PRSSCZ with brain and behavioral phenotypes29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57, the sequential effects of PRSSCZ on proximal white matter microstructure and then distal behavior remain largely unknown.
As disconnection is a key feature of schizophrenia58, it is essential to explore the effects of PRSSCZ on the microstructure integrity of brain white matter tracts, which serve as the substrate of brain connections. The white matter microstructure can be assessed using diffusion MRI metrics, and thus several studies have investigated the associations between PRSSCZ and diffusion MRI phenotypes29,30,31,32,33. However, the evidence for an association between PRSSCZ and white matter phenotypes remains inconclusive, with limited findings primarily derived from a large community dataset and small sample sizes from younger healthy individuals and first episode patients with schizophrenia. The most comprehensive study has constructed the PRSSCZ using GWAS data from 69,369 cases and 236,642 controls, investigating the associations of PRSSCZ with fractional anisotropy (FA), mean diffusivity (MD), neurite density index, and orientation dispersion index of 15 major white matter tracts in the UK Biobank30. Although significant associations were identified between PRSSCZ and white matter microstructure, several issues still need clarification. First, as patients with schizophrenia also show abnormalities in other diffusion MRI metrics and other white matter tracts, it is plausible to investigate the associations of PRSSCZ with more diffusion MRI metrics and more tracts. Second, although PRSSCZ has been associated with both brain and behavioral outcomes, it remains unclear whether white matter microstructure damage mediates the effects of PRSSCZ on cognitive and mental health outcomes. Third, the influence of PRSSCZ on brain and behavior may not be consistent across all individuals. Potential moderators such as sex and socioeconomic conditions need to be considered. For instance, a sex-specific association between PRSSCZ and schizophrenia-related traits has been observed59. Furthermore, areal deprivation is a critical environmental risk factor for schizophrenia. Studies have consistently shown that living in more deprived areas correlates with a higher incidence and prevalence of the disorder60,61. This link cannot be solely attributable to social drift, suggesting that factors related to deprived environments likely play a causal role in the development of illness. Such environmental adversity may interact with genetic vulnerability. However, it is unknown whether the impact of PRSSCZ on white matter microstructure and subsequent cognitive and mental health outcomes is more severe in individuals exposed to higher levels of areal deprivation.
Based on the PRSSCZ scores provided by UK Biobank (UKB)62,63, we explored the effects of PRSSCZ on eight diffusion MRI metrics of 48 white matter tracts in 23,468 UKB participants and 14 cognitive and mental health phenotypes in up to 299,846 UKB participants. Then, we identified the interactions between PRSSCZ and sex as well as between PRSSCZ and multiple deprivation (IMD) to determine whether PRSSCZ effects on brain and behavioral outcomes differ between male and female as well as between higher and lower levels of areal deprivation. Finally, we investigated the mediation of white matter phenotypes for the associations between PRSSCZ and behavioral outcomes.
Methods
Study population
We used the data provided by the UKB cohort (https://www.ukbiobank.ac.uk/), a population-based prospective study that recruited over 500,000 people aged between 37 and 73 years at the baseline assessments in 2006–2010 from across the UK62. The UKB study was approved by the National Health Service National Research Ethics Service (21/NW/0157), and written informed consent was obtained from all participants. We accessed UKB data under the granted application number (75556).
PRSSCZ
PRSSCZ was used to estimate an individual’s cumulative genetic risk for schizophrenia. A standard set of PRSSCZ scores was downloaded from the UKB PRS Release (field ID 26275, https://biobank.ndph.ox.ac.uk/ukb/field.cgi?id=26275)64. To calculate the PRSSCZ, the summary statistics of a GWAS meta-analysis involving three external GWASs for schizophrenia (97,456 patients and 334,331 healthy controls) were treated as the training dataset, while all UKB participants were used as the testing dataset. A Bayesian approach that effectively combines data from multiple ancestries and related traits was utilized to model the posterior effect size for each variant across the entire genome in the training dataset. The PRSSCZ score of each UKB participant was then calculated as the genome-wide sum of the per-variant posterior effect size multiplied by allele dosage. Genetic variants used to generate PRS weights were required to have an INFO score >0.8, not display large differences (<0.2) in allele frequency between UKB inferred ancestry and 1000 Genomes Project, and not display large departures from Hardy–Weinberg Equilibrium (P > 1 × 10−10). The variants also needed to have a one-to-one mapping between Genome Builds 37 and 38. Indels, the pseudoautosomal regions, and variants with minor allele frequency <0.05 in the 1000 Genomes Project were also excluded. Following the generation of a raw PRS value, a centering and standardization procedure was applied to produce a corrected PRS value that reflects the ancestry distribution.
Imaging-derived phenotypes (IDPs)
In UKB, brain diffusion MRI data was acquired at four imaging centers with 3.0-Tesla MRI scanners (Siemens Skyra) using standard 32-channel radiofrequency receive head coils. The details about scanning parameters and data preprocessing are provided at the website (https://biobank.ctsu.ox.ac.uk/crystal/crystal/docs/brain_mri.pdf). In this study, we used eight diffusion MRI metrics to assess the microstructural properties of 48 white matter tracts and defined each metric of each tract as an IDP. Specifically, we used several metrics derived from the standard Diffusion Tensor Imaging (DTI) model. These included FA to reflect diffusion anisotropy, where higher values indicate more organized fiber structures and thus better white matter integrity; MD, the average diffusivity, where lower values suggest more restricted water movement and denser tissue; and mode of anisotropy (MO) to reflect the anisotropy type. Diffusivity was further decomposed into axial diffusivity (AD), reflecting diffusion along the principal axis (L1), and radial diffusivity (RD), the mean diffusivity of the two minor axes (L2 and L3), which is particularly sensitive to myelin status, with higher values suggesting poorer myelin integrity. To complement these DTI metrics and capture more specific biological properties, we also incorporated three metrics from the advanced Neurite Orientation Dispersion and Density Imaging (NODDI) model: orientation dispersion (OD), where higher values indicate more complex or disorganized fiber arrangements; intracellular volume fraction (ICVF), an index of neurite density where higher values reflect healthier tissue; and isotropic volume fraction (ISOVF), which quantifies extracellular free water, with elevated levels potentially indicating processes like neuroinflammation or atrophy. The tract-based spatial statistics pipeline65 was applied to preprocess brain diffusion MRI data to create a white matter skeleton in standard space. The 48 brain white matter tracts were defined by an atlas (http://bmap.ucla.edu/portfolio/atlases/ICBM_DTI-81_Atlas/) created by researchers at Johns Hopkins University. After aligning individual metric map to the white matter skeleton, the IDP of the metric for each tract was extracted by intersecting the skeleton with the tract mask in the standard space. As UKB only provided IDPs for L2 and L3, we used the mean value of L2 and L3 to represent RD of this IDP, while other IDPs were directly downloaded from UKB (category ID 134). For each of the 384 finally included IDPs, we calculated the median absolute deviation (MAD) of this IDP across the participants and removed the participants with values larger than six MADs. Brain IDPs were then normalized using rankTransPheno function in FRGEpistasis R66. Details about these brain white matter IDPs are provided in Supplementary Table S1.
Covariates
Age (field ID 21003) was assessed at the baseline and the time of MRI data acquisition (instance-2). Sex (field ID 22001) was determined using genetic sex, which provides sex classification based on genotyping analysis. The first four genetic principal components (PCs; field ID 26201) were used to measure genetic population stratification64. The imaging sites (field ID 54) were obtained at instance-2. In association analyses of PRSSCZ with behavioral phenotypes, we adjusted for baseline age, sex, age2, age × sex, age2 × sex, and four PCs. In association analyses of PRSSCZ with brain IDPs, we additionally adjusted for imaging site and used the age assessed at the instance-2. All continuous covariates were scaled by z-scoring.
Cognitive function
At the baseline, UKB participants underwent six cognitive function tests, please see details at the website (https://biobank.ndph.ox.ac.uk/showcase/label.cgi?id=116). From these tests, we included six cognitive assessments: fluid intelligence (field ID 20191), trail making (field ID 20156), symbol-digit substitution (field ID 20159), pairs matching (field ID 20131), reaction time (field ID 20023), and numeric memory (field ID 20240). The six tests showed correlations with their corresponding reference tests, indicating concurrent validity and test-retest reliability67. Details about these cognitive function assessments are provided in Supplementary Table S2.
Mental health
At the baseline, the Mental Health Questionnaire (MHQ) was used to assess mental health symptoms (https://biobank.ndph.ox.ac.uk/showcase/label.cgi?id=136). Items used for assessing mental health symptoms are summarized in Supplementary Table S3. We included eight mental health assessments: anxiety symptoms, depressive symptoms, mania symptoms, mental distress, psychotic experience, self-harm, trauma, and well-being. The item scores in each subcategory were adjusted in the same direction and then normalized to a range of 0–1 using the function “mapminmax” and finally averaged to generate an overall measure for the subcategory. Higher scores of these assessments indicate more pronounced mental symptoms and unhappiness.
Areal deprivation
Areal deprivation was assessed by IMD (field ID 26410). IMD was assessed at baseline, which was derived from a government qualitative study of deprived areas in British local councils. The study is conducted in England.
Statistical analysis
Linear regression analysis
We performed a linear regression analysis to investigate the associations of PRSSCZ with 384 brain IDPs and 14 cognitive and mental health phenotypes while controlling for age, sex, age2, age × sex, age2 × sex, and four genetic PCs. We further adjusted for imaging sites in the IDP analyses. The baseline age was used in behavioral analyses and the age at the instance-2 was used in IDP analyses. The linear regression models for IDPs were constructed using Eq. (1):
The linear regression models for cognitive and mental health phenotypes were fitted using Eq. (2):
where β0 is the intercept, β1 is the regression coefficient for PRSSCZ, β2 - β11 are the regression coefficients for covariates, ε is the residual error. The Bonferroni method was used to correct for the total number of neuroimaging (n = 384), cognitive (n = 6), and mental health (n = 8) phenotypes (P < 0.05/398 = 1.26 × 10−4).
Sex effect
We investigated the potential sex differences in the associations of PRSSCZ with brain IDPs and behavioral phenotypes by testing the interactions between PRSSCZ and sex using Eq. (3):
where Y represents each phenotype. β3 is the regression coefficients for the interaction item, Cov represents covariates including age, age2, age × sex, age2 × sex, and genetic PC1-4. Imaging sites were also included as a covariate if Y is the brain IDPs.
IMD effect
We also investigated the IMD effects on the associations of PRSSCZ with brain IDPs and behavioral phenotypes by testing the interactions between PRSSCZ and IMD using Eq. (4):
where Y represents each phenotype. β3 is the regression coefficients for the interaction item, Cov represents covariates including age, sex, age2, age × sex, age2 × sex, and genetic PC1-4. Imaging sites were also included as a covariate if Y is the brain IDPs.
Causal mediation analysis (CMA)
For the brain IDPs and behavioral phenotypes that were significantly associated with PRSSCZ, causal mediation analysis (CMA) was conducted to test whether brain IDPs (mediator, M) mediated the association between PRSSCZ (treatment, X) and behavioral phenotypes (outcome, Y) using the mediation R package68, while controlling for the covariates (age, sex, age2, age × sex, age2 × sex, four genetic PCs and imaging sites). CMA was performed on the participants with complete data by running two separate linear regression models. The average direct effect (ADE) and average causal mediation effect (ACME) were estimated for each CMA model. A non-parametric bootstrap process with 2000 resamples was employed to estimate 95% confidence interval (CI) of ACME. The significant mediation was defined as 95% CI of ACME not including zero and Pc < 0.05 (Bonferroni correction for tested models). As a sensitivity analysis to explore the potential for reverse causation, we also tested an alternative mediation model. In this reverse model, we examined whether the behavioral phenotypes (M) mediated the association between PRSSCZ (X) and the brain IDPs (Y). The analytical procedure, including bootstrapping and significance criteria, was identical to that of the primary analysis.
Sensitivity analyses
To mitigate the bias caused by multicollinearity among dMRI metrics, we conducted Principal Component Analysis (PCA). For each tract with significant association in our primary analysis, we applied PCA to its eight standardized microstructure metrics. We then tested for associations between PRSSCZ and the first two principal components (PC1 and PC2), which consistently captured a substantial portion (typically >80%) of the variance. PC1 generally represents a “white matter integrity” factor, while PC2 captures more specific geometric properties. A Bonferroni correction was applied to these tests (Pc < 0.05).
We further performed two analyses to assess the generalizability of our primary findings, considering replication if they showed a consistent direction of effects and reached nominal significance (P < 0.05). First, we repeated the primary analyses in non-Caucasian participants (N = 58,219), further adjusting for self-reported ethnicity. Second, we repeated the analyses in 1486 patients with schizophrenia (ICD-10 codes: F20-F29). These analyses were conducted on subsets of these participants with brain imaging or behavioral data.
Results
Demographics
We included 301,236 healthy UKB participants in the main analyses after excluding those with incomplete PRSSCZ data, withdrawn from the study, non-Caucasians, and having a diagnosis of any neuropsychiatric disorders (Supplementary Table S4). Detailed inclusion and exclusion criteria for the participants are presented in Supplementary Figs. S1–S3. These participants included 137,699 males and 163,537 females, their baseline ages were 58.00 ± 8.01 years (ranged 39–73 years), and their PRSSCZ were -0.36 ± 0.99 (ranged -5.54–3.95). In addition, we performed sensitivity analyses in non-Caucasian participants and individuals with schizophrenia. Demographic characteristics are provided in Supplementary Table S5.
Associations between PRSSCZ and brain white matter IDPs
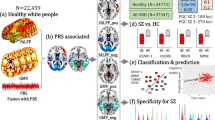
We used 384 IDPs to assess the brain white matter microstructure of 48 tracts with eight diffusion MRI metrics (FA, ICVF, ISOVF, MD, MO, OD, AD, and RD), and then used the linear regression to identify IDPs that were significantly associated with PRSSCZ (P < 1.26 × 10−4, Bonferroni correction for all brain and behavioral phenotypes), while controlling for age, sex, age2, age × sex, age2 × sex, four genetic PCs, and imaging sites in 23,468 UKB participants. We found that PRSSCZ showed significant associations with 61 IDPs (Fig. 1A, and Supplementary Table S6), involving eight diffusion MRI metrics and 26 tracts (Fig. 1B). According to the biological meanings of these metrics, converging evidence indicates that higher PRSSCZ are associated with reduced white matter integrity. For instance, PRSSCZ was associated with six IDPs of fornix, including FA (β = −0.032, P = 5.71 × 10−9), ICVF (β = −0.026, P = 5.49 × 10-5), ISOVF (β = 0.021, P = 9.49 × 10−5), MD (β = 0.023, P = 1.80 × 10−5), MO (β = −0.026, P = 3.64 × 10−5), and RD (β = 0.026, P = 1.33 × 10−6). PRSSCZ was associated with eight IDPs of cingulum, including FA (Left: β = −0.025, P = 7.33 × 10−5; Right: β = −0.028, P = 9.68 × 10-6), ICVF (Left: β = −0.024, P = 1.02 × 10−4; Right: β = −0.025, P = 4.70 × 10−5), MD (Left: β = 0.031, P = 1.31 × 10−6), MO (Right: β = −0.029, P = 5.21 × 10−6), and RD (Left: β = 0.033, P = 1.68 × 10−7; Right: β = 0.033, P = 1.93 × 10−7).
A Brain white matter tracts whose microstructure metrics (FA, MD, MO, OD, ICVF, ISOVF, AD, and RD) demonstrate significant positive (warm color) or negative (cold color) correlations (P < 1.26 × 10−4) with PRSSCZ. B Bubble plot shows the associations between PRSSCZ and 384 brain white matter IDPs. A bubble represents an association between PRSSCZ and IDP. The x-axis shows 48 white matter tracts, while the y-axis shows eight metrics. The bubble size indicates the -log10(P) value and the bubble color indicates the T value of this association. ACR anterior corona radiata, AD axial diffusivity, ALIC anterior limb of internal capsule, BCC body of corpus callosum, CP cerebral peduncle, CST corticospinal tract, CgC cingulum cingulate gyrus, CgH cingulum hippocampus, EC external capsule, FA fractional anisotropy, FX fornix, FX/ST fornix cres and stria terminalis, GC genu of corpus callosum, IC inferior cerebellar peduncle, ICVF intracellular volume fraction, ISOVF isotropic volume fraction, L left, MCP middle cerebellar peduncle, MD mean diffusivity, ML medial lemniscus, MO mode of anisotropy, OD orientation dispersion, PCR posterior corona radiata, PCT pontine crossing tract, PLIC posterior limb of internal capsule, PTR posterior thalamic radiation, R right, RD radial diffusivity, RLIC retrolenticular part of internal capsule, SCC splenium of corpus callosum, SCP superior cerebellar peduncle, SCR superior corona radiata, SFO superior fronto-occipital fasciculus, SLF superior longitudinal fasciculus, SS sagittal stratum, TAP tapetum, UNC uncinate fasciculus.
In the sensitivity analyses, we first conducted PCA on the eight dMRI metrics for each of the 26 tracts with significant associations in our primary analyses. We then tested for associations (P < 0.05/26/2 = 9.62 × 10−4) of PRSSCZ with PC1 and PC2 of each tract. The PRSSCZ was significantly associated with PCs in 23 of the 26 tracts. Specifically, the PRSSCZ showed significant associations with PC1 in 18 tracts and with PC2 in 6 tracts, indicating a widespread influence of schizophrenia genetic risk on distinct dimensions of white matter microstructure (Supplementary Table S7). The detailed results, including explained variance and factor loadings for all components, are provided in Supplementary Table S8. Next, we assessed the generalizability of our primary findings in two independent cohorts. In 3577 non-Caucasian participants, we replicated 10 of the 61 associations (P < 0.05 with a consistent direction of effects) (Supplementary Table S9). In 37 Caucasian participants with schizophrenia, we failed to replicate any associations (Supplementary Table S10).
Associations between PRSSCZ and behavioral phenotypes
We used six measures (fluid intelligence, trail making, symbol-digit substitution, pairs matching, reaction time, and numeric memory) to assess cognitive function and eight measures (anxiety symptoms, depressive symptoms, mania symptoms, mental distress, psychotic experience, self-harm, trauma, and well-being) to assess mental health. Then, we used the linear regression to identify behavioral phenotypes that were significantly associated with PRSSCZ (P < 1.26 × 10−4, Bonferroni correction for the total number of brain and behavioral phenotypes), while controlling for age, sex, age2, age × sex, age2 × sex, and four genetic PCs in up to 299,846 UKB participants. We found that PRSSCZ showed associations with 5/6 cognitive phenotypes: fluid intelligence (n = 82,613; β = −0.058, P = 2.83 × 10−67), trail making (n = 70,260; β = 0.041, P = 1.15 × 10−31), symbol-digit substitution (n = 79,405; β = −0.063, P = 1.47 × 10−90), pairs matching (n = 79,434; β = −0.006, P = 1.20 × 10−6), and reaction time (n = 299,846; β = 0.040, P = 5.22 × 10−121). We also found that PRSSCZ showed associations with 6/8 mental health symptoms: anxiety (n = 104,114; β = 0.034, P = 2.20 × 10-41), depression (n = 31,336; β = 0.048, P = 9.92 × 10−19), mania (n = 19,739; β = 0.029, P = 2.13 × 10−5), mental distress (n = 103,956; β = 0.045, P = 4.11 × 10−76), trauma (n = 41,307; β = 0.050, P = 3.29 × 10−25), and well-being (n = 102,601; β = 0.026, P = 2.90 × 10−10). The correlations between PRSSCZ and behavior are presented in Fig. 2 and Supplementary Table S11.
A The -log10(P) values of associations between PRSSCZ and 14 behavioral phenotypes (six cognitive and eight mental health phenotypes). The size of the dots in the lollipop plot represents the absolute standardized beta coefficients value. The dashed line represents P = 1.26 × 10−4. B The bar chart shows the numbers of participants for these association analyses.
In sensitivity analyses, we replicated all associations (P < 0.05 with a consistent direction of effects) in non-Caucasian participants (Supplementary Table S12). We also verified two associations in patients with schizophrenia (Supplementary Table S13). Specifically, a higher PRSSCZ was associated with greater symptoms of mania (β = 0.2941, P = 7.16 × 10−3) and mental distress (β = 0.1511, P = 1.75 × 10−2).
Sex effect
For each of the significant associations of PRSSCZ with 61 brain IDPs and five cognitive and six mental health phenotypes, we investigated the sex effect on this association by identifying the significant interaction (P < 0.05/72 = 6.94 × 10−4) between PRSSCZ and sex in the linear regression model (Eq. 3). However, we failed to identify any significant PRSSCZ × sex interactions (all P > 0.05; Supplementary Table S14).
IMD effect
For the 72 significant associations between PRSSCZ and phenotypes, we also tested the influence of IMD on these associations by identifying the significant interactions (P < 6.94 × 10−4) between PRSSCZ and IMD in the corresponding linear regression models (Eq. 4). Although we found three nominally significant PRSSCZ × IMD interactions (P = 0.011−0.031), none of these interactions survived at the Bonferroni-corrected P < 6.94 × 10−4 (Supplementary Table S15).
Brain IDPs mediated the effects of PRSSCZ on behavioral phenotypes
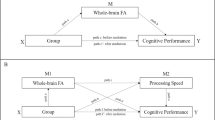
Based on the significant associations of PRSSCZ with 61 brain IDPs and 11 behavioral phenotypes, CMA was conducted to investigate whether these brain IDPs could mediate the associations between PRSSCZ and behavioral phenotypes. We controlled for age, sex, age2, age × sex, age2 × sex, four genetic PCs, and imaging sites, and defined significant ACME as P < 0.05/61/11 = 7.45 × 10−5. From the 671 mediation analyses, we found 81 significant mediations (Fig. 3A), including 67 for the associations between PRSSCZ and four cognitive phenotypes (fluid intelligence, reaction time, symbol-digit substitution, and trail making) and 14 for the associations between PRSSCZ and two mental health symptoms (mental distress and well-being). The total effects of PRSSCZ on these six behavioral phenotypes are presented in Fig. 3B. We provided the most significant CMA results for each association between PRSSCZ and behavioral phenotype in Fig. 3C–H and all CMA results in Supplementary Table S16. The association between PRSSCZ and fluid intelligence was mediated by the FA of left fornix cres and stria terminalis (FX/ST; explained portion = 3.34%, ACME: β = −0.0019, 95% CI: [−0.0032, −0.0008]) (Fig. 3C); (2) The association between PRSSCZ and reaction time was mediated by the RD of right FX/ST (explained portion = 4.52%, ACME: β = 0.0014, 95% CI: [0.0007, 0.0022]) (Fig. 3D); (3) The association between PRSSCZ and symbol-digit substitution was mediated by the FA of left posterior thalamic radiation (PTR; explained portion = 1.75%, ACME: β = −0.0015, 95% CI: [−0.0025, −0.0006]) (Fig. 3E); (4) The association between PRSSCZ and trail making was mediated by the RD of right FX/ST (explained portion = 3.12%, ACME: β = 0.0017, 95% CI: [0.0007, 0.0029]) (Fig. 3F); (5) The association between PRSSCZ and mental distress was mediated by the FA of left PTR (explained portion = 2.10%, ACME: β = 0.0009, 95% CI: [0.0004, 0.0016]) (Fig. 3G); (6) The association between PRSSCZ and well-being was mediated by the OD of left cerebral peduncle (CP; explained portion = 3.70%, β = 0.0012, 95% CI: [0.0004, 0.0024]) (Fig. 3H). Furthermore, to explore the bidirectional relationship between brain and behavior, we conducted a reverse mediation analysis. This analysis revealed several significant mediation pathways, suggesting that behavioral phenotypes can also mediate the effect of genetic risk on brain structure (Supplementary Table S17). These findings highlight the complex and potentially reciprocal interplay between brain structure and behavior.
A The distribution of the 81 significant CMA results across the six behavioral phenotypes. B Total effects between PRSSCZ and behavioral phenotypes. C–H Top CMA results for associations between PRSSCZ and six behavioral phenotypes. ACME average causal mediation effects, ADE average direct effects, CP cerebral peduncle, FX/ST fornix cres and stria terminalis, L left, OD orientation dispersion, PRSSCZ polygenetic risk score for schizophrenia, PTR posterior thalamic radiation, R right, RD radial diffusivity.
Discussion
In UKB participants, we investigated the associations of PRSSCZ with 384 brain white matter microstructure IDPs and 14 cognitive and mental health phenotypes and found that higher PRSSCZ was associated with extensive white matter impairments and poorer cognitive and mental health outcomes. We confirmed that these associations were not influenced by sex and areal deprivation, indicating relatively consistent effects. We also found significant mediations of white matter IDPs for the associations between PRSSCZ and behavioral outcomes. These findings may improve our understanding of complex relationships among the genetic risk for schizophrenia, brain white matter integrity, and cognitive and mental health outcomes.
Based on the disconnection hypothesis of schizophrenia58, we aimed to investigate whether PRSSCZ is associated with the microstructural changes in white matter tracts. Compared to previous studies that were focused on few imaging metrics of a few white matter tracts (the most comprehensive study included four metrics and 15 tracts)30 we included eight white matter microstructure metrics of 48 tracts, including diffusion MRI metrics derived from NODDI that can assess both intracellular and extracellular diffusion of water molecules69. We found that higher PRSSCZ was associated with widespread alterations in white matter microstructure, affecting 26 of the 48 tracts (54.2%). The consistent negative correlations with FA and ICVF, along with positive correlations with MD, AD, and RD, suggest that a higher genetic risk for schizophrenia is linked to extensive impairments in white matter integrity70,71,72. PCA not only validated the robustness of these findings against metric collinearity but also offered deeper insight into the nature of these microstructural alterations. PC1 serves as a general indicator of white matter integrity, whereas PC2 captures more specific geometric properties. Thus, our results suggest that genetic risk may impact both the overall health of white matter tracts and more subtle, distinct aspects of their microstructural organization.
In addition to psychotic symptoms, cognitive impairments are also commonly seen in patients with schizophrenia20,21,22. More importantly, cognitive decline is also observed before the onset of schizophrenia, such as in the health individuals with a family history of schizophrenia26. We also found that PRSSCZ was associated with cognitive deficits in healthy people. As UKB participants were middle-aged and elderly people, our results suggest that genetic risk for schizophrenia may be related to cognitive deficits in a portion of these people. We also identified significant associations of higher PRSSCZ with the increased levels of anxiety, depression, mania, mental distress, and trauma, and the decreased well-being. Most of these symptoms are also observed in patients with schizophrenia23,24. These findings may be explained by the genetic architecture shared by schizophrenia and these mental symptoms or disorders73, which is also related to clinical overlap74 and comorbidity between psychiatric disorders75.
Compared to most previous studies that focus only on the main effects of PRSSCZ on brain and behavioral phenotypes29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57, we also investigated whether sex and areal deprivation could influence these associations. Although sex-specific PRSSCZ effects on subclinical schizophrenia-related traits are reported59 and higher areal deprivation is a risk for schizophrenia60,61, we found no significant interactions between PRSSCZ and sex, nor between PRSSCZ and IMD regarding the PRSSCZ effects on phenotypes. These null findings should be interpreted with caution, since various methodological factors may influence our results. Regarding socioeconomic factors, IMD, being an area-level assessment, may not accurately capture the true socioeconomic conditions of each participant. Future studies should consider individual-level socioeconomic factors, such as education, income, and occupation, as potential moderators. The null findings may be also attributed to demographic factors, including age, sex, diagnosis, and the homogeneity of the sample. Furthermore, the current sample size may possess limited statistical power to identify interactions, because interaction analyses typically necessitate larger sample sizes than those required for main effect analysis. Therefore, we cannot definitively rule out the existence of such moderation. If these results are verified, we can assert that schizophrenia-related genetic risk factors may have stable effects on white matter development across sex and socioeconomic factors.
Although previous studies have separately reported the effects of PRSSCZ on brain and behavioral phenotypes29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57, no studies have been conducted to investigate whether the brain IDPs could mediate the effects of PRSSCZ on behavioral phenotypes. We found 81 significant mediations of brain white matter IDPs for the associations of PRSSCZ with four cognitive and two mental health phenotypes. These results may provide a plausible explanation for the cognitive and mental health impairments caused by the genetic risk of schizophrenia.
The strengths of this study included: (1) we used the PRSSCZ model trained using three external GWASs (97,456 schizophrenia patients and 334,331 healthy controls)64, representing the largest dataset for the PRSSCZ model construction; (2) we included more diffusion MRI metrics and white matter tracts than previous studies to provide a full picture of the associations between PRSSCZ and brain white matter microstructure; (3) we included brain and behavioral phenotypes in the same study, allowing us to test the mediation of brain white matter IDPs for the associations between PRSSCZ and behavioral phenotypes; and (4) we investigated the effects of sex and areal deprivation on the associations of PRSSCZ with brain and behavioral phenotypes to determine the potential stratification of the effects. However, this study also had several limitations: (1) the sample size of GWAS meta-analysis for PRSSCZ model construction is far from enough to discover all genetic risk variants associated with schizophrenia, which may affect the precise of the estimation of individual’s PRSSCZ score; (2) while we attempted to verify our primary findings in non-Caucasians and in patients with schizophrenia, the smaller sample sizes prevent us from replicating all the findings, highlighting additional validation for the generalizability of our findings; (3) the large variability in the sample sizes of behavioral phenotypes may reduce the comparability between their effects; (4) we only tested the modulation of sex and IMD on the associations of PRSSCZ with brain and behavioral phenotypes, the modulation of other factors also deserves investigation; and (5) although our large sample size provided excellent statistical power, the effect sizes of the observed associations were generally small. This is a well-known characteristic in the field of imaging genetics, where individual common genetic variants or even polygenic scores for complex traits typically explain only a small fraction of the variance in brain phenotypes. Therefore, while our findings are statistically robust, their clinical or predictive utility at an individual level may be limited, and they should be interpreted as highlighting biological pathways rather than as strong predictive markers.
In conclusion, we systematically investigated the effects of PRSSCZ on brain white matter microstructure, neurocognition, and mental health, and found highly consistent adverse effects, indicating that higher genetic risk for schizophrenia may impair brain and behavior in the healthy population. We also found that the PRSSCZ effects on brain and behavior were not modulated by sex and areal deprivation, suggesting stable effects. We further found that brain white matter phenotypes mediated the associations between PRSSCZ and behavioral outcomes, providing the neural mechanisms for PRSSCZ effects on behavioral outcomes. These findings may improve our understanding of the relations among genetic risk for schizophrenia, white matter microstructure, and cognitive and mental health phenotypes.
Data availability
All the UKB data used in the study are available at the UKB (https://www.ukbiobank.ac.uk). The granted application access number for UKB cohort is 75556. The UK Biobank has approval from the North West Multi-centre Research Ethics Committee (MREC) as a Research Tissue Bank (RTB) approval. All participants provided written informed consent before enrolment in the study. The research conformed to the principles of the Helsinki Declaration.
Code availability
For the analyses conducted in R (version 4.1.3), R function ‘glm’ (version 3.4.0) was used to perform linear regression analyses, and R package ‘mediation’ (version 4.5.0) was used to conduct causal mediation analyses. The R scripts used for the analyses in this study are publicly available in our GitHub repository: https://github.com/QianQian1213/PRSscz_brainhealth.
References
Owen, M. J., Sawa, A. & Mortensen, P. B. Schizophrenia. Lancet 388, 86–97 (2016).
Jauhar, S., Johnstone, M. & McKenna, P. J. Schizophrenia. Lancet 399, 473–486 (2022).
Cardno, A. G. et al. Heritability estimates for psychotic disorders: the Maudsley twin psychosis series. Arch. Gen. Psychiatry 56, 162–168 (1999).
Sullivan, P. F., Kendler, K. S. & Neale, M. C. Schizophrenia as a complex trait: evidence from a meta-analysis of twin studies. Arch. Gen. Psychiatry 60, 1187–1192 (2003).
Hilker, R. et al. Heritability of schizophrenia and schizophrenia spectrum based on the Nationwide Danish Twin Register. Biol. Psychiatry 83, 492–498 (2018).
Trubetskoy, V. et al. Mapping genomic loci implicates genes and synaptic biology in schizophrenia. Nature 604, 502–508 (2022).
Kubicki, M., McCarley, R. W. & Shenton, M. E. Evidence for white matter abnormalities in schizophrenia. Curr. Opin. Psychiatry 18, 121–134 (2005).
Zalesky, A. et al. Disrupted axonal fiber connectivity in schizophrenia. Biol. Psychiatry 69, 80–89 (2011).
Kelly, S. et al. Widespread white matter microstructural differences in schizophrenia across 4322 individuals: results from the ENIGMA Schizophrenia DTI Working Group. Mol. Psychiatry 23, 1261–1269 (2018).
Koshiyama, D. et al. White matter microstructural alterations across four major psychiatric disorders: mega-analysis study in 2937 individuals. Mol. Psychiatry 25, 883–895 (2020).
Lee, D. K. et al. Common gray and white matter abnormalities in schizophrenia and bipolar disorder. PLoS ONE 15, e0232826 (2020).
Keshavan, M. S. et al. Neuroimaging in Schizophrenia. Neuroimaging Clin. N. Am. 30, 73–83 (2020).
van Erp, T. G. M. et al. Cortical brain abnormalities in 4474 individuals with schizophrenia and 5098 control subjects via the enhancing neuro imaging genetics through meta analysis (ENIGMA) consortium. Biol. Psychiatry 84, 644–654 (2018).
Shenton, M. E. et al. Abnormalities of the left temporal lobe and thought disorder in schizophrenia. A quantitative magnetic resonance imaging study. N. Engl. J. Med. 327, 604–612 (1992).
Barta, P. E., Pearlson, G. D., Powers, R. E., Richards, S. S. & Tune, L. E. Auditory hallucinations and smaller superior temporal gyral volume in schizophrenia. Am. J. Psychiatry 147, 1457–1462 (1990).
Rimol, L. M. et al. Cortical thickness and subcortical volumes in schizophrenia and bipolar disorder. Biol. Psychiatry 68, 41–50 (2010).
van Erp, T. G. et al. Subcortical brain volume abnormalities in 2028 individuals with schizophrenia and 2540 healthy controls via the ENIGMA consortium. Mol. Psychiatry 21, 585 (2016).
Kuhn, S. & Gallinat, J. Resting-state brain activity in schizophrenia and major depression: a quantitative meta-analysis. Schizophr. Bull. 39, 358–365 (2013).
Zhao, J. et al. Structure-function coupling in white matter uncovers the abnormal brain connectivity in Schizophrenia. Transl. Psychiatry 13, 214 (2023).
Kahn, R. S. & Keefe, R. S. Schizophrenia is a cognitive illness: time for a change in focus. JAMA Psychiatry 70, 1107–1112 (2013).
Heinrichs, R. W. & Zakzanis, K. K. Neurocognitive deficit in schizophrenia: a quantitative review of the evidence. Neuropsychology 12, 426–445 (1998).
Guo, J. Y., Ragland, J. D. & Carter, C. S. Memory and cognition in schizophrenia. Mol. Psychiatry 24, 633–642 (2019).
Braga, R. J., Reynolds, G. P. & Siris, S. G. Anxiety comorbidity in schizophrenia. Psychiatry Res. 210, 1–7 (2013).
Krynicki, C. R., Upthegrove, R., Deakin, J. F. W. & Barnes, T. R. E. The relationship between negative symptoms and depression in schizophrenia: a systematic review. Acta Psychiatr. Scand. 137, 380–390 (2018).
Vink, M. et al. Diminishing striatal activation across adolescent development during reward anticipation in offspring of schizophrenia patients. Schizophr. Res. 170, 73–79 (2016).
van Haren, N. E. M. et al. Brain structure, IQ, and psychopathology in young offspring of patients with schizophrenia or bipolar disorder. Eur. Psychiatry 63, e5 (2020).
Sugranyes, G. et al. Gray matter volume decrease distinguishes schizophrenia from bipolar offspring during childhood and adolescence. J. Am. Acad. Child Adolesc. Psychiatry 54, 677–684.e672 (2015).
de Leeuw, M. et al. Changes in white matter organization in adolescent offspring of schizophrenia patients. Neuropsychopharmacology 42, 495–501 (2017).
Alloza, C. et al. Polygenic risk score for schizophrenia and structural brain connectivity in older age: a longitudinal connectome and tractography study. NeuroImage 183, 884–896 (2018).
Stauffer, E. M. et al. Grey and white matter microstructure is associated with polygenic risk for schizophrenia. Mol. Psychiatry 26, 7709–7718 (2021).
Bolhuis, K. et al. Interaction of schizophrenia polygenic risk and cortisol level on pre-adolescent brain structure. Psychoneuroendocrinology 101, 295–303 (2019).
Su, W. et al. Effects of polygenic risk of schizophrenia on interhemispheric callosal white matter integrity and frontotemporal functional connectivity in first-episode schizophrenia. Psychol. Med. 53, 2868–2877 (2023).
Taquet, M. et al. A structural brain network of genetic vulnerability to psychiatric illness. Mol. Psychiatry 26, 2089–2100 (2021).
Abe, C. et al. Genetic risk for bipolar disorder and schizophrenia predicts structure and function of the ventromedial prefrontal cortex. J. Psychiatry Neurosci. 46, E441–E450 (2021).
Alloza, C. et al. Psychotic-like experiences, polygenic risk scores for schizophrenia, and structural properties of the salience, default mode, and central-executive networks in healthy participants from UK Biobank. Transl. Psychiatry 10, 122 (2020).
Alnaes, D. et al. Brain heterogeneity in schizophrenia and its association with polygenic risk. JAMA Psychiatry 76, 739–748 (2019).
French, L. et al. Early cannabis use, polygenic risk score for schizophrenia and brain maturation in adolescence. JAMA Psychiatry 72, 1002–1011 (2015).
Harris, M. A. et al. Stratifying major depressive disorder by polygenic risk for schizophrenia in relation to structural brain measures. Psychol. Med. 50, 1653–1662 (2020).
Harrisberger, F. et al. Impact of polygenic schizophrenia-related risk and hippocampal volumes on the onset of psychosis. Transl. Psychiatry 6, e868 (2016).
Lieslehto, J. et al. Polygenic risk score for schizophrenia and face-processing network in young adulthood. Schizophr. Bull. 45, 835–845 (2019).
Liu, S. et al. Polygenic effects of schizophrenia on hippocampal grey matter volume and hippocampus-medial prefrontal cortex functional connectivity. Br. J. Psychiatry 216, 267–274 (2020).
Lv, J. et al. Individual deviations from normative models of brain structure in a large cross-sectional schizophrenia cohort. Mol. Psychiatry 26, 3512–3523 (2021).
Neilson, E. et al. Effects of environmental risks and polygenic loading for schizophrenia on cortical thickness. Schizophr. Res. 184, 128–136 (2017).
Neilson, E. et al. Impact of polygenic risk for schizophrenia on cortical structure in UK Biobank. Biol. Psychiatry 86, 536–544 (2019).
Sha, Z., Schijven, D. & Francks, C. Patterns of brain asymmetry associated with polygenic risks for autism and schizophrenia implicate language and executive functions but not brain masculinization. Mol. Psychiatry 26, 7652–7660 (2021).
Rashid, B. et al. A framework for linking resting-state chronnectome/genome features in schizophrenia: a pilot study. NeuroImage 184, 843–854 (2019).
McIntosh, A. M. et al. Polygenic risk for schizophrenia is associated with cognitive change between childhood and old age. Biol. Psychiatry 73, 938–943 (2013).
Hatzimanolis, A. et al. Common genetic variation and schizophrenia polygenic risk influence neurocognitive performance in young adulthood. Am. J. Med. Genet. B Neuropsychiatr. Genet. 168B, 392–401 (2015).
Kauppi, K. et al. Polygenic risk for schizophrenia associated with working memory-related prefrontal brain activation in patients with schizophrenia and healthy controls. Schizophr. Bull. 41, 736–743 (2015).
Germine, L. et al. Association between polygenic risk for schizophrenia, neurocognition and social cognition across development. Transl. Psychiatry 6, e924 (2016).
Hagenaars, S. P. et al. Shared genetic aetiology between cognitive functions and physical and mental health in UK Biobank (N=112 151) and 24 GWAS consortia. Mol. Psychiatry 21, 1624–1632 (2016).
Liebers, D. T. et al. Polygenic risk of schizophrenia and cognition in a population-based survey of older adults. Schizophr. Bull. 42, 984–991 (2016).
Cosgrove, D. et al. MiR-137-derived polygenic risk: effects on cognitive performance in patients with schizophrenia and controls. Transl. Psychiatry 7, e1012 (2017).
Ranlund, S. et al. A polygenic risk score analysis of psychosis endophenotypes across brain functional, structural, and cognitive domains. Am. J. Med. Genet. B Neuropsychiatr. Genet. 177, 21–34 (2018).
Rampino, A. et al. A Polygenic Risk Score of glutamatergic SNPs associated with schizophrenia predicts attentional behavior and related brain activity in healthy humans. Eur. Neuropsychopharmacol. 27, 928–939 (2017).
Richards, A. et al. Associations between schizophrenia genetic risk, anxiety disorders and manic/hypomanic episode in a longitudinal population cohort study. Br. J. Psychiatry 214, 96–102 (2019).
Riglin, L. et al. Schizophrenia risk alleles and neurodevelopmental outcomes in childhood: a population-based cohort study. Lancet Psychiatry 4, 57–62 (2017).
Crow, T. J. Schizophrenia as a transcallosal misconnection syndrome. Schizophr. Res. 30, 111–114 (1998).
Mas-Bermejo, P. et al. Sex-specific association between schizophrenia polygenic risk and subclinical schizophrenia-related traits. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 136, 111161 (2025).
Lee, S. C. et al. Area deprivation, urbanicity, severe mental illness and social drift - A population-based linkage study using routinely collected primary and secondary care data. Schizophr. Res. 220, 130–140 (2020).
Harrison, G., Gunnell, D., Glazebrook, C., Page, K. & Kwiecinski, R. Association between schizophrenia and social inequality at birth: case-control study. Br. J. Psychiatry 179, 346–350 (2001).
Palmer, L. J. UK Biobank: bank on it. Lancet 369, 1980–1982 (2007).
Miller, K. L. et al. Multimodal population brain imaging in the UK Biobank prospective epidemiological study. Nat. Neurosci. 19, 1523–1536 (2016).
Thompson, D. J. et al. A systematic evaluation of the performance and properties of the UK Biobank Polygenic Risk Score (PRS) Release. PLoS One 19, e0307270 (2024).
Smith, S. M. et al. Tract-based spatial statistics: voxelwise analysis of multi-subject diffusion data. NeuroImage 31, 1487–1505 (2006).
Zhang, F., Boerwinkle, E. & Xiong, M. Epistasis analysis for quantitative traits by functional regression model. Genome Res. 24, 989–998 (2014).
Fawns-Ritchie, C. & Deary, I. J. Reliability and validity of the UK Biobank cognitive tests. PLoS ONE 15, e0231627 (2020).
Tingley, D., Yamamoto, T., Hirose, K., Keele, L. & Imai, K. mediation: r package for causal mediation analysis. J. Stat. Softw. 59, 1–38 (2014).
Zhang, H., Schneider, T., Wheeler-Kingshott, C. A. & Alexander, D. C. NODDI: practical in vivo neurite orientation dispersion and density imaging of the human brain. NeuroImage 61, 1000–1016 (2012).
Stinear, C. M. et al. Functional potential in chronic stroke patients depends on corticospinal tract integrity. Brain 130, 170–180 (2007).
Fjell, A. M., Westlye, L. T., Amlien, I. K. & Walhovd, K. B. Reduced white matter integrity is related to cognitive instability. J. Neurosci. 31, 18060–18072 (2011).
Acosta, J. N. et al. Blood pressure-related white matter microstructural disintegrity and associated cognitive function impairment in asymptomatic adults. Stroke Vasc. Neurol. 8, 358–367 (2023).
Brainstorm, C. et al. Analysis of shared heritability in common disorders of the brain. Science 360, 6395 (2018).
Caspi, A. et al. The p factor: one general psychopathology factor in the structure of psychiatric disorders?. Clin. Psychol. Sci. 2, 119–137 (2014).
Plana-Ripoll, O. et al. Exploring comorbidity within mental disorders among a Danish National Population. JAMA Psychiatry 76, 259–270 (2019).
Acknowledgements
The authors report no biomedical financial interests or potential conflicts of interest. The founders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. This work was supported by the National Natural Science Foundation of China to C.Y (Grant No. 82030053 and 82430063), the National Natural Science Foundation of China to J.X (Grant No. 82371924), the National Key Project of “Inter-governmental International Scientific and Technological Innovation Cooperation” to J.X (Grant No. 2023YFE0199700), the Natural Science Foundation of Tianjin to J.X (Grant No. 25JCZDJC00640), the Tianjin Young Talents in Science and Technology for J.X (Grant No. QN20230336), the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences to J.X (Grant No. 2024-JKCS-18), the Tianjin Science and Technology Commission Major Special Project in Public Health Science and Technology to J.X (Grant No. 24ZXGQSY00050) and the Tianjin Medical University “Clinical Talent Training 123 Climbing Plan” to J.X.
Author information
Authors and Affiliations
Contributions
C.Y. and J.X. designed and led the project. Q.Q. contributed substantially to the analysis and interpretation of the data. Q.Q. created the figures. J.X. provided statistical advice. G.Z. and S.W. provided genetic advice. N.Z. advised on the neuroimaging analyses. J.S. advised on the areal deprivation analyses. Q.Q. wrote the manuscript. C.Y. and J.X. revised the manuscript. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Qian, Q., Zhao, G., Zhang, N. et al. Polygenic risk for schizophrenia is associated with white matter microstructure, cognitive and mental health. Schizophr 12, 12 (2026). https://doi.org/10.1038/s41537-025-00714-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41537-025-00714-x