Abstract
Converging evidence indicates that dysregulated cortico-subcortical connectivity underpins core cognitive impairment in schizophrenia (SCZ). However, the mechanistic basis linking this disrupted brain function to cognitive deficits remains elusive. We hypothesized that dysfunction within the hippocampal-thalamocortical circuitry mediates the impact of peripheral inflammation on cognitive deficits in SCZ. We recruited 62 drug-native patients with SCZ and 44 age and sex-matched healthy controls (HCs), assessing: serum levels of cytokines, cognition using the MATRICS Consensus Cognitive Battery, and resting-state functional connectivity (FC) and functional connectivity network (FCN) features. Correlation and mediation analyses were employed to examine relationships among peripheral inflammation, brain functional alterations, and cognitive deficits. The findings revealed reduced FC in SCZ between the right subiculum of the hippocampus and the anterior division of the right parahippocampal gyrus (aPaHC), as well as the entorhinal cortex and the left lingual gyrus/precuneus. Conversely, the thalamus exhibited aberrant hyperconnectivity with widespread areas, including the Cornu Ammonis of the hippocampus, right lateral occipital cortex, intraparietal sulcus (IPS), and sensorimotor areas. Conjunction analysis identified distinct disruptions within hippocampal-thalamo-visual circuitry, suggesting that an imbalance in thalamo-visual and intra-hippocampal connectivity may mediate the relationship between peripheral inflammation (e.g., IL-4, CCL-2) and working memory dysfunction. This study advances our understanding of the role of peripheral inflammation in SCZ, revealing complex cross-domain interactions between pathophysiological changes (e.g., dysregulated peripheral immunity and circuit-specific dysfunction) and behavioral features. Our findings highlight a novel combinatorial therapeutic strategy—integrating cytokine-specific agents (e.g., for CCL-2 and IL-4) with circuit-directed neuromodulation of the dysfunctional hippocampal-thalamic-visual circuitry, thereby ameliorating cognitive deficits in SCZ.
Similar content being viewed by others
Introduction
Cognitive impairment, a core feature of schizophrenia (SCZ) that progressively worsens with disease severity, serves as a critical predictor of treatment response and functional outcomes1,2. Growing evidence suggests that neuroinflammation contributes significantly to cognitive impairment3,4. Patients with first-episode schizophrenia (FES) and those in relapse exhibit elevated levels of pro-inflammatory cytokines (e.g., IL-6, IL-12, IL-1β, TNF-α, and TGF-β)5,6,7,8,9, which are specifically associated with cognitive deficits, including impairments in working memory and processing speed10,11,12. However, the specific neural basis through which peripheral inflammation contributes to cognitive dysfunction remains poorly understood.
Preclinical studies indicate that elevated neuroinflammation—marked by microglial overactivation in cognition-critical regions (hippocampus, prefrontal cortex, thalamus)—directly contributes to cognitive deficits13,14,15. Recent research suggests that persistent or recurring peripheral inflammation plays a central role in driving microglial overactivation through several mechanisms, and a damaged blood–brain barrier (BBB) is one of the major causative factors16. Microglial overactivation promotes the release of pro-inflammatory cytokines, chemokines, and free radicals, leading to neuron damage, atrophy, and synaptic loss16. Clinically, such overactivation profoundly affects brain structure and function, manifesting as morphological abnormalities, white matter demyelination, and altered functional connectivity (FC)3,11,17,18,19,20,21,22,23,24. Meta-analyses further associate elevated inflammatory markers with reduced cortical thickness (CT) in frontal, temporal, and occipital regions, as well as smaller hippocampal/amygdala volumes in individuals with psychosis22. In particular, elevated levels of IL-6 and IL-8 predict widespread gray matter volume (GMV) reductions23. Our previous study demonstrated that elevated IL-4 is linked to cognitive impairment in SCZ, an effect partially mediated by GMV loss in the left superior frontal gyrus11. Together, these findings highlight the role of dysregulated inflammation in promoting neuroanatomical abnormalities and cognitive deterioration.
Despite accumulating evidence, inconsistencies persist across studies25,26,27. For instance, some studies reported elevated GMV and gray matter thickening in the hippocampal, amygdala, putamen, and thalamus in patients with elevated inflammation25. Similarly, Hoang et al. (2022) found increased CT in the right parahippocampal gyrus, caudal anterior cingulate, and banks of the superior sulcus in these patients, though these structural changes lacked cognitive or symptom correlations26. Proposed explanations for these discrepancies include differing temporal effects of inflammation—such as acute vascular changes versus chronic neurodegenerative processes28.
In contrast, limited functional neuroimaging studies have revealed neural signatures associated with inflammation and its role in cognitive impairment from a global perspective19,26,29,30. A data-driven research using independent component analysis demonstrated that elevated inflammatory markers correlate with reduced functional connectivity (FC) between the anterior default mode network (DMN) and the right frontoparietal network—disruptions linked to deficits in verbal fluency, memory, symbolic coding, and global cognition19. Other studies suggested that inflammation-mediated dysregulation of the DMN underpins social cognitive deficits29,30. A recent study linked peripheral inflammatory markers to reduced corticostriatal FC associated with avolition31. Given established evidence that cortico-subcortical circuitry, particularly frontotemporal/parietal-thalamic and hippocampal-prefrontal pathways, is critically involved in cognition deficits24,32,33,34,35, we focused on key subcortical nodes—the hippocampus, thalamus, nucleus accumbens, amygdala, and caudate. The inclusion of the caudate and nucleus accumbens was specifically motivated by their established roles in reward and motivation36. We included specific hippocampal subfields (e.g., DG, CA1, CA3) based not only on their susceptibility to inflammatory insults but richly expression of immune-related receptors37. This approach enables us to capture system-level dysregulation within key immunomodulatory pathways.
This study aimed to identify biomarkers mediating the relationship between inflammation and neurocognition, thereby informing combined intervention strategies to ameliorate cognitive deficits in SCZ. We hypothesized that dysfunction within the hippocampal-thalamocortical circuitry mediates the effect of dysregulated peripheral immunity on cognition. To test this hypothesis, our analytical strategy comprised three steps: (1) Seed-to-voxel whole-brain FC mapping of a priori subcortical regions to localize dysregulated cortico-subcortical connectivity in SCZ without spatial constraints; (2) ROI-to-ROI network analysis to validate these findings and identify disease-altered network components within a canonical macro-scale architecture; (3) Mediation analysis to explore whether specific circuit disruptions account for the relationship between inflammatory markers and cognitive deficits.
Materials and methods
Participants
This study recruited 65 drug-naïve patients with SCZ from the First Affiliated Hospital of Xi’an Jiaotong University, China. All enrolled patients met the following inclusion criteria: (1) Age 18–50 years; (2) DSM-IV SCZ diagnosis confirmed by two independent psychiatrists; (3) Positive and Negative Syndrome Scale (PANSS) total score > 60; (4) Antipsychotic-naïve prior to enrollment. Patients meeting the following criteria were excluded: (1) Major neurological disorders or unstable physical conditions, including the acute episode of somatic diseases, decompensated period of chronic somatic diseases, severe abnormality in laboratory tests, or recent major surgery/trauma; (2) History of loss of consciousness > 5 min; and (3) MRI contraindications. Forty-six age-/sex-matched healthy controls (HCs) were recruited from local communities through advertisements. HCs met identical inclusion/exclusion criteria as patients, with additional requirements: (1) No lifetime DSM-IV psychiatric disorders; (2) No psychiatric history in first-degree relatives. The procedures were explained, and written informed consent was obtained from participants before the study. All participants provided written informed consent after being fully informed of the study procedures. The study was approved by the Ethics Committee of the First Affiliated Hospital of Xi’an Jiaotong University (XJTY1AF2015LSL-079).
Measurements of circulating inflammatory factors
On the day of inclusion or the following morning, approximately 5 mL of fasting venous blood was collected from all participants. Nine inflammatory cytokines in serum, including interleukin 1β (IL-1β), IL-4, IL-6, IL-8, IL-10, IL-12P70, chemokine ligand 2 (CCL-2), IFN-γ, and TNF-α, were detected using enzyme-linked immunosorbent technology (R&D Systems Company, United States). The selection of these cytokines was guided by consistent evidence from meta-analyses and mechanistic frameworks38,39,40, including pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, IL-8), immunoregulatory cytokines (IL-4, IL-10), mediators of Th1 immunity (IL-12p70, IFN-γ), and the chemokine CCL-2, all of which have been previously associated with alterations in SCZ.
Cognition assessment
Cognitive function was assessed using the MATRICS Consensus Cognitive Suite Battery (MCCB), which was developed by the Measurement and Treatment Research to Improve Cognition in Schizophrenia group41. We selected seven tests across seven cognitive domains, including speed of processing (Brief Assessment of Cognition in SCZ [BACS], category fluency test: Animal naming [Fluency], and Trail Making Test [TMT]), working memory (Wechsler Memory Scale-Third Edition [WMS-III]), verbal learning (Hopkins Verbal Learning Test-Revised [HVLT-R]), visual learning (Brief Visuospatial Memory Test-Revised [BVMT-R]), reasoning and problem solving (Neuropsychological Assessment Battery [NAB]). The original cognitive test score was converted into a T score after adjusting for age, sex, and education, with higher scores indicating better cognitive function. A complete case analysis was used for the MCCB assessments. The representativeness of the analyzed sample was supported by demographic, clinical, and biological comparisons between patients with and without complete MCCB data.
Neuroimaging acquisition and processing
Neuroimaging data were collected using a 3-tesla (3T) MRI scanner, equipped with an 8-channel phase array radio-frequency head coil (Signa HDxt, General Electric Medical System, Milwaukee, WI, USA). Three-dimensional (3D) fast-spoiled gradient-recalled echo T1-weighted images were obtained with parameters of repetition time/echo time (TR/TE) = 10/4.6 ms, FA = 15°, field of view (FOV) = 180 × 180 mm2, matrix size = 256 × 256, voxel size = 1 × 1 × 1 mm, and 160 contiguous 1-mm sagittal slices. Whole-brain resting-state functional images were acquired using a gradient-recalled echo-planar imaging pulse sequence (TR/TE = 2000/30 ms, FA = 90°, FOV = 240 × 240, acquisition matrix = 64 × 64, total volumes = 185). The participants’ heads were positioned carefully with restraining foam pads to minimize head motion, and earplugs were used to reduce scanner noise.
All images were preprocessed and analyzed with the CONN 20b toolbox (https://web.conn-toolbox.org/) based on SPM 12. A total of 185 time points were preprocessed as the following steps including: slice-timing, realignment, outlier detection (scrubbing), coregistration, normalization into the MNI space, resampling at 3 mm3, and spatial smoothing with full-width half-maximum (FWHM) = 8 mm. Finally, the residual time series was temporally band-pass-filtered (0.01–0.08 Hz) to reduce the effect of low- and high-frequency physiological noise. We controlled for head motion first through regression of six head motion parameters plus their temporal first derivatives, followed by the so-called scrubbing method to remove outlier volumes, defined as frame-wise displacement (FD) of more than 0.5 mm or mean global signal intensity of more than 2 standard deviations (SDs).
Regions of interest (ROIs)
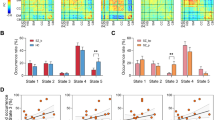
We defined core subcortical brain regions into 20 different ROIs to conduct seed-based resting-state FC (rs-FC) analysis. Firstly, five bilateral subcortical ROIs were defined from the FSL Harvard-Oxford Atlas maximum likelihood subcortical atlas42: hippocampus, nucleus accumbens, amygdala, caudate, and thalamus (see Fig. 1A, B). Secondly, five subfields of the hippocampus from the Anatomy toolbox’s cytoarchitectonic probabilistic maps43,44 were also included as ROIs (Cornu Ammonis [CA], Dentate gyrus [DG], Entorhinal cortex [EC], Hippocampal-amygdala-transition-area [HATA], and Subiculum [Subc])45 (see Fig. 1C).
A, B highlight five key subcortical regions in the hippocampal-thalamocortical circuitry. HIP hippocampus, THA thalamus, NAcc nucleus accumbens, AMYG amygdalal, CAU caudate. C represents five subfields of the hippocampus. Cornu Ammonis (CA), Dentate gyrus (DG), Entorhinal cortex (EC), Hippocampal-amygdala-transition-area (HATA), and Subiculum (Subc).
Seed-to-voxel rs-FC
We obtained FC maps for pre-selected subcortical regions and subfields of the hippocampus by calculating correlation coefficients between each subcortical seed and all other voxels to localize voxel-wise group differences in FC, thereby identifying the long-distance neural activity of the subcortical-cortical connectivity. The correlation coefficients were transformed to Fisher’s z values and subjected to subsequent statistical analyses. The resultant FC maps were compared between the SCZ and HC groups, adjusting for age, sex, and years of education. Group-level analyses were primarily thresholded at uncorrected P-values < 0.001 at the voxel level, followed by cluster-level inference with false discovery rate (FDR) correction at P < 0.05, applied to the whole-brain connectivity map of each seed46. An exploratory analysis was also conducted using a more lenient voxel-level threshold of P < 0.005, with cluster-level FDR maintained at P < 0.05, to identify potential group differences for hypothesis-generating purposes.
ROI-to-ROI rs-FCN
To contextualize the seed-to-voxel findings within a canonical macro-scale brain network architecture, we conducted an ROI-to-ROI analysis. This network-based approach served to validate the initial results and statistically assess the overall network integrity of the defined network matrix, thereby identifying coherent, multi-region network components altered in SCZ. In addition to the 20 subcortical seeds, we incorporated 30 cortical network-based ROIs (two ROIs for cerebellar networks were excluded) defined from CONN’s independent component analyses of the Human Connectome Project (HCP) dataset47. We extracted the average BOLD time series from all predefined ROIs (10 subcortical regions, 10 subregions of the Hip, and 30 cortical regions) to compute the Fisher-transformed bivariate correlation coefficient for each pair of ROIs. For each subject, a pairwise ROI-to-ROI connectivity (RRC) matrix was generated to characterize the functional connectivity networks (FCN) and used for group-level analyses (SCZ vs HC), where group-relevant functional connections were organized into significant network clusters with connection-level uncorrected P-values < 0.05 and cluster-level FDR-corrected P-values < 0.05.
Correlation and mediation analyses
To identify underlying mechanistic processes that may bridge the gap between inflammation and cognitive function, we first computed Spearman’s correlation coefficients between cognition, cytokines, and FC/FCN measures that showed significant group differences, and adjusted P-values using the Benjamini–Hochberg false discovery rate (FDR) procedure. Following this, we conducted a mediation analysis to explore the causal role of cytokines in contributing to cognitive impairment through brain functional alterations. The mediation analyses were carried out using the PROCESS v3.0 for SPSS (Model 4, 5000 bootstrapping samples to generate 95% confidence intervals [CIs]), a method that does not assume a normally distributed sampling distribution of the indirect effect48. Mediated effects were considered statistically significant at the 0.05 level if the 95% CI did not include zero.
Statistical analysis
Continuous, normally, and non-normally distributed variables were analyzed using the independent-sample t-test and the Wilcoxon signed-rank test, respectively. Categorical variables were compared between groups via the chi-square χ2 test. Statistical significance was set at P < 0.05. Serum cytokines underwent log10-transformation to achieve normality assumptions; subsequent group comparisons and association analyses utilized transformed data. Statistical maps of functional connectivity were generated after multiple comparison analysis (FDR corrected with P-values < 0.05). Statistical analyses were conducted using CONN 20b toolbox (https://web.conn-toolbox.org/), SPSS 23.0 (IBM, Armonk, NY, USA), and R 3.5.1 (http://cran.r-project.org). Visual representations of multiple omics correlations were made using the ggcor, ggplot2, and heatmap packages.
Results
Demographic and clinical characteristics of participants
After carefully examining the preprocessed neuroimaging data, three patients with SCZ and two HCs were excluded because of their deviant motion parameters. Finally, 62 drug-naïve patients and 44 HCs were enrolled in the analysis. Demographic and clinical characteristics were compared between the groups (Table 1). There were no significant differences between the two groups regarding age, sex, and body mass index (BMI). The patient group showed a significantly lower level of educational attainment than the HC group (t = 5.907, p < 0.001).
Clinically, the patient group exhibited a mean PANSS total score of 82.88 ± 26.36. Serum cytokine analysis revealed elevated levels of IL-1β, IL-4, IL-6, IL-8, TNF-α, and CCL-2 in the SCZ group compared to HCs, whereas IL-12p70 was slightly decreased (Fig. 4A). In the subset of participants who completed the MCCB, cognitive assessment showed significantly poorer performance across multiple domains in patients than in HCs (Table 1). No significant differences were observed in demographic, clinical, or biological markers—including serum cytokines and functional connectivity metrics—between patients with and without complete MCCB data (all p > 0.05; Supplementary Table S5 and Figs. S2 and S3), supporting the representativeness of the subsample for subsequent cognitive correlation and mediation analyses.
Seed-based rs-FC
Seed-based rs-FC analyses revealed significant group differences that surpassed the FDR multiple comparison correction with Pvoxel < 0.001(uncorrected) and Pcluster < 0.05 (FDR-corrected). Under this threshold, significant group differences were identified in hippocampal subfields (CA, EC, and Subiculum), the thalamus (THA), the amygdala (AMYG), and the nucleus accumbens (NAcc) (Table 2; Fig. 2). Specifically, the SCZ group exhibited increased FC between the left CA and left THA (Fig. 2A), as well as between the left THA and the superior division of the lateral occipital cortex (sLOC), relative to the HC group (Fig. 2D). Conversely, the patient group showed decreased FC between the left AMYG and bilateral precentral gyrus (PreCG), and between the right NAcc and posterior cingulate cortex (PCC) (Fig. 2F, G). In addition, a secondary sensitivity analysis was performed using a more lenient voxel-level threshold of Pvoxel < 0.005 and Pcluster < 0.05 (FDR-corrected) to identify potential effects of group differences. At this exploratory threshold, the SCZ group displayed reduced FC between the right EC and the left lingual gyrus/precuneus, as well as between the right subiculum and the right anterior parahippocampal gyrus (aPaHC) (Fig. 2B, C). Increased FC was also observed between the right THA and a cluster encompassing the right AMYG and hippocampus (Fig. 2E).
A–C depict FC alterations between hippocampus (HIP) subregions and the whole brain. D–G illustrate dysconnectivity in extra-hippocampal subcortical regions. Yellow and purple colors denote increased and decreased FC strength, respectively. H Box plots of seed-based FC comparisons between SCZ and HC groups. SCZ schizophrenia, HC healthy controls. FC functional connectivity. HIPCA the Cornu Ammonis of the Hippocampus, THA thalamus, HIPEC the entorhinal cortex of the hippocampus, LG/Precun the lingual gyrus/left precuneus cortex, HIPSubc the Subiculum of the Hippocampus, aPaHC parahippocampal gyrus, anterior division, sLOC lateral occipital cortex, superior division, AMYG amygdala, PreCG precentral gyrus, NAcc nucleus accumbens, PCC posterior cingulate cortex. l left, r right.
ROI-to-ROI rs-FCN
To build upon the seed-to-voxel findings, ROI-to-ROI network analyses were conducted to further identify altered, multi-region network components in SCZ and to elucidate specific patterns of subcortical-cortical dysconnectivity. Two thousand four hundred and fifty connections for all 50 ROIs (detailed in the “Materials and methods” section) were clustered based on CONN’s data-driven hierarchical clustering method of ROI-to-ROI spatial proximity and functional similarity metrics. A cluster-level FDR-corrected P-value of 0.05 was applied to control for multiple comparisons; however, no clusters survived after multiple comparisons. Due to the potential weakening of statistical effects from calculating multiple subregions of the hippocampus, we excluded 10 subregions, yielding 1560 pairwise functional connections. These connections were classified into 32 clusters; one cluster remained significant with 10 significant individual connections (FDR-corrected P-value = 0.044, Fig. 3A–C). Notably, all connections within this cluster were hyperfunctional in the SCZ group, involving bilateral thalamus and the lateral portion of the sensorimotor network (SMN), intraparietal sulcus (IPS), bilateral AMYG, and contralateral hippocampus connections.
A functional connectome circle showed significant group difference between SCZ and HC groups (FDR-corrected P-value < 0.05). The labels outside the circle stand for the 30 network-based cortical regions and 10 subcortical regions. Red curves indicate hyperconnectivity (SCZ > HC), with connection opacity scaled to t-statistic magnitude. B A 10 × 10 FC matrix visualizes dysregulated connections (cell opacity reflects t-statistics). C group comparisons of connectivity strength. SCZ schizophrenia, HC healthy controls. FC functional connectivity, FCN functional connectivity network. THA thalamus, AMYG amygdala, HIP hippocampus, DAN dorsal attention network, IPS intraparietal sulcus, SMN sensorimotor network. l left, r right.
Associations between brain FC/FCN, inflammatory markers, and cognitive domains
Spearman correlation analyses across the combined cohorts demonstrated that specific cytokines (IL-1β, IL-6, IL-4, CCL-2, TNF-α, and IL-8) were significantly negatively correlated with cortical-subcortical connectivity, including the EC-lingual gyrus/precuneus, NAcc-PCC, AMYG-PreCG, and HIPSubc-aPaHC connectivity. Conversely, these cytokines showed positive correlations with the thalamic-sLOC/CA and IPS/SMN connectivity (Fig. 4B, Spearman correlation, p < 0.05). Following FDR correction, IL-4, IL-1β, and CCL-2 maintained significant correlations with the THA-sLOC/CA connectivity, while IL-4 retained inverse correlation with the HIPSubc-aPaHC connectivity (FDR-corrected with p < 0.05). Furthermore, these dysregulated connectivity patterns were associated with poorer performance across multiple cognitive domains. Notably, dysfunctional connectivity in the THA-sLOC/IPS and HIPSubc-aPaHC pathways was significantly associated with working memory impairment (FDR-corrected with p < 0.05; Fig. 5A). For spatial visualization, BrainNet Viewer-generated connectivity mappings integrated these altered pathways associated with inflammatory markers and cognitive domains (Fig. 6A), with uncorrected statistical maps provided in Supplementary Fig. S1. Given that educational attainment significantly correlated with multiple cognitive domains (Table S1), we controlled for this variable when assessing associations between cognitive performance and brain FC. Scatter plots depicted significant relationships: (1) right HIPSubc-aPaHC coupling positively correlated with WMS (R = 0.45, P = 0.0002, adjusted for edu: R′ = 0.34, P = 0.007), and (2) left THA-right sLOC coupling negatively correlated with WMS (R = −0.30, P = 0.014, adjusted for edu: R′ = −0.27, P = 0.032) (Fig. 5B). In addition, analyses confined to the patient cohort revealed no significant associations between psychotic symptom severity and cognitive performance, neuroimaging metrics, or peripheral inflammation levels (Tables S2–S4).
A Serum cytokines differences between SCZ and HC groups. B Spearman correlations between serum cytokines and FC/FCN metrics. The black borders represent the remaining significant correlations after correcting for the P-value. FC functional connectivity, FCN functional connectivity network. HIPCA the Cornu Ammonis of the Hippocampus, HIPEC the entorhinal cortex of the Hippocampus, HIPSubc the subiculum of the hippocampus, aPaHC parahippocampal gyrus, anterior division, THA thalamus, LG/Precun the lingual gyrus/left precuneus cortex, sLOC lateral occipital cortex, superior division, AMYG amygdala, PreCG precentral gyrus, NAcc nucleus accumbens, PCC posterior cingulate cortex, IPS intraparietal sulcus, DAN dorsal attention network, SMN sensorimotor network. SCZ schizophrenia, HC healthy controls. l left, r right.
A Spearman correlations between cognitive scores and brain connectivity. The black borders represent the remaining significant correlations after correcting for the P-value. B Correlation between brain connectivity and cognition with and without adjusting for education. HIPCA the Cornu Ammonis of the Hippocampus, HIPEC the entorhinal cortex of the hippocampus, HIPSubc the subiculum of the hippocampus, aPaHC parahippocampal gyrus, anterior division, THA thalamus, LG/Precun the lingual gyrus/left precuneus cortex, sLOC lateral occipital cortex, superior division, AMYG amygdala, PreCG precentral gyrus, NAcc nucleus accumbens, PCC posterior cingulate cortex, IPS intraparietal sulcus, DAN dorsal attention network, SMN sensorimotor network. l left, r right. BACS brief assessment of cognition in SCZ, WMS-III Wechsler Memory Scale-Third Edition, Fluency category fluency test: animal naming, HVLT-R Hopkins Verbal Learning Test-Revised, BVMT-R Brief Visuospatial Memory Test-Revised, NAB Neuropsychological Assessment Battery, TMT Trail Making Test. SCZ schizophrenia, HC healthy controls.
A Spatial visualization of connectivity mappings associated with inflammatory markers and cognitive domains using BrainNet Viewer. B–D The mediation effect diagram in which the decreased FC of the THA-sLOC and HIPSubc-aPaHC couplings mediated inflammation and cognition (cytokines→brain FC→cognitive function). THA thalamus, sLOC lateral occipital cortex, superior division, HIPSubc the subiculum of the hippocampus, aPaHC parahippocampal gyrus, anterior division. WMS Wechsler Memory Scale. *P < 0.05, **P < 0.01, ***P < 0.001.
Mediation analyses
To evaluate whether brain functional alterations mediated the effects of peripheral inflammation on cognitive function, we performed mediation analyses. In this model, path a assessed the association between inflammatory markers and FC changes (mediator), path b measured the association between FC (mediator), and each dimension of cognitive function (outcome), path c represented the total effect of inflammation on cognitive outcomes, and path c′ represented the direct effect of inflammation on cognitive outcomes. The indirect effect was derived by multiplying the paths a and b49,50,51. In specific, after controlling for age, sex, and education, the left THA-right sLOC hyperfunction fully mediated the effects of IL-4 (indirect effect: B(ab) = −0.298, [95%CI: −0.205, −0.010]) and CCL-2 (indirect effect: B(ab) = −2.093, [95%CI: −0.225, −0.024]) on WMS (Fig. 6B, C). In addition, the right HIPSubc-aPaHC hypofunction also mediated the effects of IL-4 on WMS (indirect effect: B(ab) = −0.409, [95%CI: −0.216, −0.030], total effect: B(c) = −0.964, P = 0.046) after controlling for age, sex, and education (Fig. 6D). Similarly, we examined peripheral inflammation as a potential mediator in the relationship between brain FC and cognition using bootstrapped mediation analyses. However, no significant mediation effects were observed (all 95% CIs included zero).
Discussion
This study comprehensively characterized peripheral inflammatory markers, brain functional connectivity, and cognitive performance in drug-naïve patients with SCZ. By systematically examining interrelationships across these domains, we identified indirect associations consistent with the mediation of cognitive impairment. Specifically, we found distinct disruptions in hippocampal-thalamocortical circuitry that were concurrently associated with dysregulated peripheral immune profiles and cognitive deficits across multiple domains. Mediation analyses further indicated that thalamo-visual hyperconnectivity and intra-hippocampal hypoconnectivity may underlie the relationship between peripheral inflammation and working memory dysfunction. These findings add anatomical and functional specificity to the established link between neuroinflammation and large-scale network dysfunction in SCZ. While converging evidence indicates that inflammatory processes exert diffuse effects across thalamocortical, DMN-sensory, and corticostriatal networks, our findings refine this broader inflammatory phenotype by pinpointing the hippocampal (CA1)-thalamo-visual pathway as a particularly vulnerable circuit, potentially underlying specific cognitive deficits such as visual associative memory. These results advance the neuroinflammatory hypothesis of SCZ by delineating circuit-specific mechanisms, thereby informing future targeted therapeutic strategies. Methodologically, our inclusion of drug-naïve, early-stage patients (mean illness duration < 5 years) strengthens the biological inference by minimizing confounds from chronic medication and disease progression on both inflammatory markers and neural circuitry.
This study identified the thalamus as a key hub exhibiting hyperconnectivity with widespread cortical areas, including the CA subregion, the lateral occipital cortex, the intraparietal sulcus, and the lateral sensorimotor network. These hyperconnectivity patterns correlated with elevated peripheral cytokines and poorer cognitive performance. Our findings align with prior reports of thalamocortical imbalance in SCZ52, characterized by reduced prefrontal connectivity but increased sensorimotor cortex connectivity32,33,53,54. Recent work further described hyperconnected thalamo-cortico-cerebellar networks with aberrant thalamic nodal centrality in SCZ55. Building on prior evidence, we demonstrated thalamic hyperconnectivity specifically involving visual processing regions (the lateral occipital cortex and the intraparietal sulcus) that are critical for spatial perception and visual memory56,57. As a central hub for sensory filtering and cognitive resource allocation53, thalamocortical hyperactivity may overwhelm memory capacity by disrupting efficient sensory prioritization58. From an inflammation perspective, preclinical evidence has reported stress-induced microglial-mediated atrophy in the thalamus and visual cortex59,60,61. Although the present study does not directly measure microglial activity in thalamocortical circuits, the significant association between inflammatory markers and thalamo-visual hyperconnectivity indirectly supports the inflammation susceptibility of these pathways. To further identify specific immune mediators with potential clinical relevance, we performed mediation analyses, which revealed indirect associations consistent with CCL-2 and IL-4 being linked to working memory deficits through activation of the thalamo-visual pathway.
Traditionally, CCL-2 is regarded as a pro-inflammatory chemokine, whereas IL-4 is classified as an anti-inflammatory cytokine39,62. However, our mediation effects indicated that the associations between both cytokines and working memory deficits were linked via thalamo-visual hyperconnectivity, suggesting that the underlying neuroimmune interplay may involve mechanisms more complex than a simple pro‑/anti‑inflammatory dichotomy. Specifically, CCL-2 promotes the recruitment of immune cells to inflammatory sites, thereby activating classical inflammatory pathways that may indirectly drive aberrant thalamo-visual circuit activation and consequent cognitive impairment. In contrast, although elevated IL-4 is generally considered a compensatory response to pro‑inflammatory signals63, its association with cognitive deficits here indicates a “double-edged” role under pathological conditions. As a key regulator of microglial activity, persistent dysregulation of IL-4 signaling may polarize microglia toward an aberrant phenotype, leading to excessive or inaccurate synaptic pruning, which ultimately disrupts neural connectivity and contributes to cognitive deficits39,64. Furthermore, IL-4 can directly modulate neuronal network excitability; such interference may impair synaptic plasticity and memory encoding39,65,66. IL-4 has also been shown to increase neuronal vulnerability to oxidative stress, thereby exerting neurotoxic effects and potentially accelerating neuroprogressive pathways67.
These findings collectively emphasize a core principle: In the specific pathological context of SCZ, persistent aberrant IL-4 signaling—whether through distorting excitatory/inhibitory balance, dysregulating synaptic pruning, or exacerbating oxidative stress—can ultimately manifest as cognitive impairment. In summary, our study provides initial evidence that thalamo-visual hyperconnectivity may act as a neural substrate for inflammation-related cognitive impairment in SCZ. We propose that combining thalamo-visual neuromodulation with cytokine-targeted therapy may prove superior to either approach alone in ameliorating cognitive deficits.
Another key finding involved imbalanced hippocampal connectivity, primarily affecting the CA, EC, and subiculum subfields. First, we observed increased FC between the left CA and the thalamus in SCZ, consistent with prior reports documenting elevated left hippocampus-bilateral thalamus FC68 and enhanced hippocampal-thalamic synchronization during deep encoding tasks69. Structural evidence further supports this functional alteration, with studies reporting disrupted white matter integrity in thalamus-CA pathway and reduced gray matter volume in high-risk and SCZ populations24,68,70,71. While extending Cavdar et al.’s work on the thalamic nucleus reuniens innervation of the hippocampus72, our study specifically localized aberrant connectivity to the CA subfield. Subsequent correlation analyses support the susceptibility of hippocampal-thalamic circuitry to inflammatory/oxidative stress15,61,73,74, which correlated with cognitive impairments—though brain-cognition associations did not survive FDR correction.
Additionally, we observed reduced FC in EC-lingual gyrus/precuneus and Subiculum-aPaHC couplings in SCZ. The EC mediates hippocampal-neocortical information transfer between the hippocampus and neocortex and is central to episodic memory and spatial navigation75,76,77. As core nodes of the default mode network, the lingual gyrus and precuneus constitute the neural basis of episodic memory, suggesting that EC-lingual gyrus/precuneus hypoconnectivity may underlie memory impairment in SCZ. Similarly, the subiculum serves as the primary output hub of the hippocampus, consolidating information for long-term storage78,79,80. The anterior division of the parahippocampal gyrus, a region primarily comprising the EC81,82,83, integrates multimodal sensory information from association cortices, including visual and spatial inputs, and then transmits it to the subiculum via the perforant path83. Weakened Subiculum-aPaHC connectivity may therefore compromise sensory information integration by disrupting the role of the EC-subiculum circuit in cognitive mapping82,84,85.
Mediation analyses further suggest that intra-hippocampal disruptions are associated with persistently elevated IL-4 levels, with both factors concurrently linked to impairments in information transmission and memory. As previously noted, the immunoregulatory cytokine IL-4 is not invariably beneficial under conditions of sustained activation. In line with this, the current study indicates that elevated IL-4 levels may undermine the integrity of the entorhinal–subicular circuit, thereby mediating working memory impairment. The underlying mechanisms may be explained via two complementary pathways: first, persistently high IL-4 may disrupt excitatory/inhibitory balance through overactivation of inhibitory neurons, thereby impairing efficient information encoding65,66; second, despite its immunoregulatory properties, IL-4-induced M2 microglial polarizatio—when persistently or inappropriately activated, particularly during sensitive developmental or early disease windows—can lead to excessive microglial activation or aberrant synaptic pruning, ultimately impairing synaptic plasticity39,86. Together, these pathways outline the pathological basis by which IL-4 contributes to hippocampus-dependent cognitive deficits.
This study revealed potential pathways linking peripheral inflammation to cognitive dysfunction in SCZ, though several limitations warrant consideration. First, the modest sample size—particularly for cognitive correlation—may limit statistical robustness; larger cohorts are required to validate these results. Second, although we controlled for confounders (e.g., age, sex, and education), unmeasured variables such as smoking status or sleep disturbances could influence outcomes and should be systematically assessed in future studies. Third, the cross-sectional design precludes causal inference. Future longitudinal or within-subject designs tracking individuals over time, particularly across symptomatic and remissive phases, would help establish temporal precedence—and clarify the directionality of the observed associations. Notably, we observed no significant associations between inflammatory markers, functional brain alterations, cognitive performance, and symptom severity (as measured by PANSS) in our acute-phase SCZ cohort. This dissociation suggests that such neuroimmune and cognitive abnormalities may reflect stable, trait-like features of the illness, rather than state-dependent correlates of acute psychosis. These alterations appear more closely tied to core dimensions of SCZ—such as persistent cognitive impairment. Consequently, these findings highlight the value of targeting these fundamental pathophysiological processes in future therapies, irrespective of symptomatic state.
Conclusion
In conclusion, this study established significant associations among peripheral inflammatory markers, disrupted hippocampal-thalamocortical circuitry, and cognitive dysfunction in SCZ. Our findings specifically highlight hippocampal-thalamic-visual hyperactivity and intra-hippocampal weakening as key circuit-level pathways through which neuroinflammation may impair cognition. These findings thus provide a rationale for combinatorial strategies that concurrently address the specific immune dysregulation (e.g., cytokine-focused therapy for CCL-2 and IL-4) and its downstream impact on neural circuitry to ameliorate cognitive deficits in SCZ.
Data availability
The data used in this study are not publicly available due to participants’ privacy; interested researchers may contact the corresponding author to discuss potential collaborations and access to the data, subject to institutional and ethical approvals.
References
Lee, M. et al. Cognitive function and variability in antipsychotic drug-naive patients with first-episode psychosis: a systematic review and meta-analysis. JAMA Psychiatry 81, 468–476 (2024).
Jonas, K. et al. The course of general cognitive ability in individuals with psychotic disorders. JAMA Psychiatry 79, 659–666 (2022).
Khandaker, G. M. et al. Inflammation and immunity in schizophrenia: implications for pathophysiology and treatment. Lancet Psychiatry 2, 258–270 (2015).
Kogan, S., Ospina, L. H. & Kimhy, D. Inflammation in individuals with schizophrenia - Implications for neurocognition and daily function. Brain Behav. Immun. 74, 296–299 (2018).
Fond, G., Lançon, C., Korchia, T., Auquier, P. & Boyer, L. The role of inflammation in the treatment of schizophrenia. Front. Psychiatry 11, 160 (2020).
Sæther, L. S. et al. Inflammation and cognition in severe mental illness: patterns of covariation and subgroups. Mol. Psychiatry 28, 1284–1292 (2023).
Dunleavy, C., Elsworthy, R. J., Upthegrove, R., Wood, S. J. & Aldred, S. Inflammation in first-episode psychosis: the contribution of inflammatory biomarkers to the emergence of negative symptoms, a systematic review and meta-analysis. Acta Psychiatr. Scand. 146, 6–20 (2022).
Zhou, X., Tian, B. & Han, H. B. Serum interleukin-6 in schizophrenia: a system review and meta-analysis. Cytokine 141, 155441 (2021).
Momtazmanesh, S., Zare-Shahabadi, A. & Rezaei, N. Cytokine alterations in schizophrenia: an updated review. Front. Psychiatry 10, 892 (2019).
Ermakov, E. A., Melamud, M. M., Buneva, V. N. & Ivanova, S. A. Immune system abnormalities in schizophrenia: an integrative view and translational perspectives. Front. Psychiatry 13, 880568 (2022).
Fan, Y. et al. Grey matter volume and its association with cognitive impairment and peripheral cytokines in excited individuals with schizophrenia. Brain Imaging Behav. 16, 2618–2626 (2022).
Patlola, S. R., Donohoe, G. & McKernan, D. P. The relationship between inflammatory biomarkers and cognitive dysfunction in patients with schizophrenia: a systematic review and meta-analysis. Prog. Neuropsychopharmacol. Biol. Psychiatry 121, 110668 (2023).
Zhuo, C. et al. Microglia and cognitive impairment in schizophrenia: translating scientific progress into novel therapeutic interventions. Schizophrenia 9, 42 (2023).
Na, K. S., Jung, H. Y. & Kim, Y. K. The role of pro-inflammatory cytokines in the neuroinflammation and neurogenesis of schizophrenia. Prog. Neuropsychopharmacol. Biol. Psychiatry 48, 277–286 (2014).
Vasic, V. & Schmidt, M. H. H. Resilience and vulnerability to pain and inflammation in the hippocampus. Int. J. Mol. Sci. https://doi.org/10.3390/ijms18040739 (2017).
Blossom, V. et al. Implicating neuroinflammation in hippocampus, prefrontal cortex and amygdala with cognitive deficit: a narrative review. 3 Biotech 15, 320 (2025).
Cai, M. et al. Homotopic functional connectivity disruptions in schizophrenia and their associated gene expression. Neuroimage 289, 120551 (2024).
de Bartolomeis, A. et al. Linking Inflammation, Aberrant Glutamate-Dopamine Interaction, and Post-synaptic Changes: Translational Relevance for Schizophrenia and Antipsychotic Treatment: a Systematic Review. Mol. Neurobiol. 59, 6460–6501 (2022).
Lizano, P. et al. Peripheral inflammatory subgroup differences in anterior Default Mode network and multiplex functional network topology are associated with cognition in psychosis. Brain Behav. Immun. 114, 3–15 (2023).
Goldsmith, D. R., Bekhbat, M., Mehta, N. D. & Felger, J. C. Inflammation-related functional and structural dysconnectivity as a pathway to psychopathology. Biol. Psychiatry 93, 405–418 (2023).
Williams, J. A. et al. Inflammation and brain structure in schizophrenia and other neuropsychiatric disorders: a Mendelian randomization study. JAMA Psychiatry 79, 498–507 (2022).
Kose, M., Pariante, C. M., Dazzan, P. & Mondelli, V. The role of peripheral inflammation in clinical outcome and brain imaging abnormalities in psychosis: a systematic review. Front. Psychiatry 12, 612471 (2021).
Alexandros Lalousis, P. et al. Inflammatory subgroups of schizophrenia and their association with brain structure: a semi-supervised machine learning examination of heterogeneity. Brain Behav. Immun. 113, 166–175 (2023).
Wannan, C. M. J. et al. Disruptions in white matter microstructure associated with impaired visual associative memory in schizophrenia-spectrum illness. Eur. Arch. Psychiatry Clin. Neurosci. 272, 971–983 (2022).
Lizano, P. et al. Multivariate relationships between peripheral inflammatory marker subtypes and cognitive and brain structural measures in psychosis. Mol. Psychiatry 26, 3430–3443 (2021).
Hoang, D. et al. Inflammatory subtypes in antipsychotic-naïve first-episode schizophrenia are associated with altered brain morphology and topological organization. Brain Behav. Immun. 100, 297–308 (2022).
Mendelson, D., Mizrahi, R., Lepage, M. & Lavigne, K. M. C-reactive protein and cognition: Mediation analyses with brain morphology in the UK Biobank. Brain Behav. Immun. Health 31, 100664 (2023).
Jurcau, A. & Simion, A. Neuroinflammation in cerebral ischemia and ischemia/reperfusion injuries: from pathophysiology to therapeutic strategies. Int. J. Mol. Sci. https://doi.org/10.3390/ijms23010014 (2021).
King, S. et al. Early life stress, low-grade systemic inflammation and weaker suppression of the default mode network (DMN) during face processing in Schizophrenia. Transl. Psychiatry 13, 213 (2023).
King, S. et al. Childhood trauma, IL-6 and weaker suppression of the default mode network (DMN) during theory of mind (ToM) performance in schizophrenia. Brain Behav. Immun. Health 26, 100540 (2022).
Goldsmith, D. R. et al. Inflammation is associated with avolition and reduced resting state functional connectivity in corticostriatal reward circuitry in patients with schizophrenia. Neuropsychopharmacology 50, 1706–1714 (2025).
Wu, G. et al. Imbalance between prefronto-thalamic and sensorimotor-thalamic circuitries associated with working memory deficit in schizophrenia. Schizophr. Bull. 48, 251–261 (2022).
Xi, C. et al. Schizophrenia patients and their healthy siblings share decreased prefronto-thalamic connectivity but not increased sensorimotor-thalamic connectivity. Schizophr. Res. 222, 354–361 (2020).
Perry, B. A. L., Mendez, J. C. & Mitchell, A. S. Cortico-thalamocortical interactions for learning, memory and decision-making. J. Physiol. 601, 25–35 (2023).
Sui, J. et al. In search of multimodal neuroimaging biomarkers of cognitive deficits in schizophrenia. Biol. Psychiatry 78, 794–804 (2015).
Haynos, A. F. et al. Resting state hypoconnectivity of reward networks in binge eating disorder. Cereb. Cortex 31, 2494–2504 (2021).
Chen, J. et al. Neuroinflammation and disruption in working memory in aged mice after acute stimulation of the peripheral innate immune system. Brain Behav. Immun. 22, 301–311 (2008).
Halstead, S. et al. Alteration patterns of peripheral concentrations of cytokines and associated inflammatory proteins in acute and chronic stages of schizophrenia: a systematic review and network meta-analysis. Lancet Psychiatry 10, 260–271 (2023).
Roomruangwong, C. et al. The role of aberrations in the Immune-Inflammatory Response System (IRS) and the Compensatory Immune-Regulatory Reflex System (CIRS) in different phenotypes of schizophrenia: the IRS-CIRS theory of schizophrenia. Mol. Neurobiol. 57, 778–797 (2020).
Goldsmith, D. R., Rapaport, M. H. & Miller, B. J. A meta-analysis of blood cytokine network alterations in psychiatric patients: comparisons between schizophrenia, bipolar disorder and depression. Mol. Psychiatry 21, 1696–1709 (2016).
Nuechterlein, K. H. et al. The MATRICS Consensus Cognitive Battery, part 1: test selection, reliability, and validity. Am. J. Psychiatry 165, 203–213 (2008).
Desikan, R. S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 31, 968–980 (2006).
Eickhoff, S. B. et al. A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. Neuroimage 25, 1325–1335 (2005).
Amunts, K. et al. Cytoarchitectonic mapping of the human amygdala, hippocampal region and entorhinal cortex: intersubject variability and probability maps. Anat. Embryol. 210, 343–352 (2005).
Jung, J. et al. Altered hippocampal functional connectivity patterns in patients with cognitive impairments following ischaemic stroke: a resting-state fMRI study. Neuroimage. Clin. 32, 102742 (2021).
Chumbley, J. R. & Friston, K. J. False discovery rate revisited: FDR and topological inference using Gaussian random fields. Neuroimage 44, 62–70 (2009).
Whitfield-Gabrieli, S. & Nieto-Castanon, A. Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect. 2, 125–141 (2012).
Preacher, K. J. & Hayes, A. F. Asymptotic and resampling strategies for assessing and comparing indirect effects in multiple mediator models. Behav. Res. Methods 40, 879–891 (2008).
Kotackova, L. et al. Bariatric surgery and its impact on depressive symptoms, cognition, brain and inflammation. Front. Endocrinol. 14, 1171244 (2023).
Li, Y., Chen, M., Zhang, R. & Li, X. Experiencing happiness together facilitates dyadic coordination through the enhanced interpersonal neural synchronization. Soc. Cogn. Affect. Neurosci. 17, 447–460 (2022).
Crowley, S. J., Kanel, P., Roytman, S., Bohnen, N. I. & Hampstead, B. M. Basal forebrain integrity, cholinergic innervation and cognition in idiopathic Parkinson’s disease. Brain 147, 1799–1808 (2024).
Hwang, K., Bertolero, M. A., Liu, W. B. & D’Esposito, M. The human thalamus is an integrative hub for functional brain networks. J. Neurosci. 37, 5594–5607 (2017).
Giraldo-Chica, M. & Woodward, N. D. Review of thalamocortical resting-state fMRI studies in schizophrenia. Schizophr. Res. 180, 58–63 (2017).
Cao, H. et al. Cerebello-thalamo-cortical hyperconnectivity as a state-independent functional neural signature for psychosis prediction and characterization. Nat. Commun. 9, 3836 (2018).
Forlim, C. G., Klock, L., Gallinat, J. & Kühn, S. Altered resting-state functional connectivity in a thalamo-cortico-cerebellar network in patients with schizophrenia. Sci. Rep. 14, 26284 (2024).
Kravitz, D. J., Saleem, K. S., Baker, C. I. & Mishkin, M. A new neural framework for visuospatial processing. Nat. Rev. Neurosci. 12, 217–230 (2011).
Jiang, Y. & Kanwisher, N. Common neural mechanisms for response selection and perceptual processing. J. Cogn. Neurosci. 15, 1095–1110 (2003).
Anticevic, A. et al. Characterizing thalamo-cortical disturbances in schizophrenia and bipolar illness. Cereb. Cortex 24, 3116–3130 (2014).
Yoshii, T. et al. Brain atrophy in the visual cortex and thalamus induced by severe stress in animal model. Sci. Rep. 7, 12731 (2017).
Nakagawa, Y. & Chiba, K. Involvement of neuroinflammation during brain development in social cognitive deficits in autism spectrum disorder and schizophrenia. J. Pharmacol. Exp. Ther. 358, 504–515 (2016).
Sugama, S. Stress-induced microglial activation may facilitate the progression of neurodegenerative disorders. Med. Hypotheses 73, 1031–1034 (2009).
Woodward, E. A., Prêle, C. M., Nicholson, S. E., Kolesnik, T. B. & Hart, P. H. The anti-inflammatory effects of interleukin-4 are not mediated by suppressor of cytokine signalling-1 (SOCS1). Immunology 131, 118–127 (2010).
Carril Pardo, C., Oyarce Merino, K. & Vera-Montecinos, A. Neuroinflammatory loop in schizophrenia, is there a relationship with symptoms or cognition decline? Int. J. Mol. Sci. https://doi.org/10.3390/ijms26010310(2025).
Howes, O. D. & McCutcheon, R. Inflammation and the neural diathesis-stress hypothesis of schizophrenia: a reconceptualization. Transl. Psychiatry 7, e1024 (2017).
Herz, J. et al. GABAergic neuronal IL-4R mediates T cell effect on memory. Neuron 109, 3609–3618.e3609 (2021).
Hanuscheck, N. et al. Interleukin-4 receptor signaling modulates neuronal network activity. J. Exp. Med. https://doi.org/10.1084/jem.20211887 (2022).
Mori, S., Maher, P. & Conti, B. Neuroimmunology of the interleukins 13 and 4. Brain Sci. https://doi.org/10.3390/brainsci6020018 (2016).
Su, W. et al. Thalamo-hippocampal dysconnectivity is associated with serum cholesterol level in drug-naïve patients with first-episode schizophrenia. J. Psychiatr. Res. 151, 497–506 (2022).
Ragland, J. D. et al. Levels-of-processing effect on frontotemporal function in schizophrenia during word encoding and recognition. Am. J. Psychiatry 162, 1840–1848 (2005).
Bernard, J. A., Orr, J. M. & Mittal, V. A. Abnormal hippocampal-thalamic white matter tract development and positive symptom course in individuals at ultra-high risk for psychosis. NPJ Schizophr. 1, 15009 (2015).
Prasad, K. M., Muldoon, B., Theis, N., Iyengar, S. & Keshavan, M. S. Multipronged investigation of morphometry and connectivity of hippocampal network in relation to risk for psychosis using ultrahigh field MRI. Schizophr. Res. 256, 88–97 (2023).
Cavdar, S. et al. The pathways connecting the hippocampal formation, the thalamic reuniens nucleus and the thalamic reticular nucleus in the rat. J. Anat. 212, 249–256 (2008).
Prasad, K. M., Burgess, A. M., Keshavan, M. S., Nimgaonkar, V. L. & Stanley, J. A. Neuropil pruning in early-course schizophrenia: immunological, clinical, and neurocognitive correlates. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 1, 528–538 (2016).
Ramlackhansingh, A. F. et al. Inflammation after trauma: microglial activation and traumatic brain injury. Ann. Neurol. 70, 374–383 (2011).
Reznik, D., Margulies, D. S., Witter, M. P. & Doeller, C. F. Evidence for convergence of distributed cortical processing in band-like functional zones in human entorhinal cortex. Curr. Biol. https://doi.org/10.1016/j.cub.2024.10.020 (2024).
Nilssen, E. S., Doan, T. P., Nigro, M. J., Ohara, S. & Witter, M. P. Neurons and networks in the entorhinal cortex: a reappraisal of the lateral and medial entorhinal subdivisions mediating parallel cortical pathways. Hippocampus 29, 1238–1254 (2019).
Sargolini, F. et al. Conjunctive representation of position, direction, and velocity in entorhinal cortex. Science 312, 758–762 (2006).
de Melo, M. B., Daldegan-Bueno, D., Favaro, V. M. & Oliveira, M. G. M. The subiculum role on learning and memory tasks using rats and mice: a scoping review. Neurosci. Biobehav. Rev. 155, 105460 (2023).
Baset, A. & Huang, F. Shedding light on subiculum’s role in human brain disorders. Brain Res. Bull. 214, 110993 (2024).
Böhm, C., Peng, Y., Geiger, J. R. P. & Schmitz, D. Routes to, from and within the subiculum. Cell Tissue Res. 373, 557–563 (2018).
Maass, A. et al. Laminar activity in the hippocampus and entorhinal cortex related to novelty and episodic encoding. Nat. Commun. 5, 5547 (2014).
van Strien, N. M., Cappaert, N. L. & Witter, M. P. The anatomy of memory: an interactive overview of the parahippocampal-hippocampal network. Nat. Rev. Neurosci. 10, 272–282 (2009).
Raslau, F. D. et al. Memory part 2: the role of the medial temporal lobe. Ajnr. Am. J. Neuroradiol. 36, 846–849 (2015).
Canto, C. B., Wouterlood, F. G. & Witter, M. P. What does the anatomical organization of the entorhinal cortex tell us?. Neural Plast. 2008, 381243 (2008).
Makhdoum, R. M. & Alahmadi, A. A. S. Disturbances in resting state functional connectivity in schizophrenia: a study of hippocampal subregions, the parahippocampal gyrus and functional brain networks. Diagnostics https://doi.org/10.3390/diagnostics15151955 (2025).
Cherry, J. D., Olschowka, J. A. & O’Banion, M. K. Neuroinflammation and M2 microglia: the good, the bad, and the inflamed. J. Neuroinflammation 11, 98 (2014).
Acknowledgements
This work was supported by the Key Program of National Natural Science of China (No.82230044), the National Natural Science Foundation of China (NO.82201659).
Author information
Authors and Affiliations
Contributions
XC Ma, YJ Fan, and W.W. designed the project. QY Ma, M.J., and ZY Qi collected the clinical and MRI data. NZ Gou and YJ Fan analyzed the data, NZ Gou interpreted the results, and wrote the manuscript. YJ Fan and Y.G. revised the manuscript. All authors contributed to and have approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41537_2025_719_MOESM1_ESM.pdf (download PDF )
Peripheral Inflammation Mediates Cognitive Deficits in Drug-naive Schizophrenia through Hippocampal-thalamo-visual Circuitry Dysfunction
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Gou, N., Ma, Q., Jia, M. et al. Peripheral inflammation mediates cognitive deficits in drug-naive schizophrenia through hippocampal-thalamo-visual circuitry dysfunction. Schizophr 12, 15 (2026). https://doi.org/10.1038/s41537-025-00719-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41537-025-00719-6