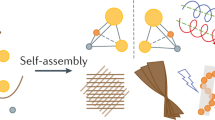
Abstract
Atomically precise nanoclusters can be assembled into ordered superlattices with unique electronic, magnetic, optical and catalytic properties. The co-crystallization of nanoclusters with functional organic molecules provides opportunities to access an even wider range of structures and properties, but can be challenging to control synthetically. Here we introduce a supramolecular approach to direct the assembly of atomically precise silver nanoclusters into a series of nanocluster‒organic ionic co-crystals with tunable structures and properties. By leveraging non-covalent interactions between anionic silver nanoclusters and cationic organic macrocycles of varying sizes, the orientation of nanocluster surface ligands can be manipulated to achieve in situ resolution of enantiopure nanocluster‒organic ionic co-crystals that feature large chiroptical effects. Beyond chirality, this co-crystal assembly approach provides a promising platform for designing functional solid-state nanomaterials through a combination of supramolecular chemistry and atomically precise nanochemistry.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data supporting the findings of this study are available within the Article and its Supplementary Information. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition nos. CCDC 2352972 (NOIC25-2C41C8-A), 2352973 (NOIC25-3C6-L), 2352974 (NOIC25-3C6-R1), 2352975 (NOIC25-3C6-R2), 2352976 (NOIC25-3C6-R3), 2352977 (NOIC25-3C6-R4), 2352978 (NOIC25-3C6-R5), 2352979 (NOIC25-3C7-L), 2352980 (NOIC25-3C7-R), 2352981 (NOIC25-3C8-L), 2352982 (NOIC25-3C8-R), 2342983 (NOIC25-2C61C8-L), 2352984 (NOIC25-2C61C8-R), 2352985 (NOIC25-2C81C6-L), 2352986 (NOIC25-2C81C6-R) and 2352987 (NOIC25-3C3-M). Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. Source data are provided with this paper.
References
Huang, R.-W. et al. Hypersensitive dual-function luminescence switching of a silver-chalcogenolate cluster-based metal–organic framework. Nat. Chem. 9, 689–697 (2017).
Alhilaly, M. J. et al. Assembly of atomically precise silver nanoclusters into nanocluster-based frameworks. J. Am. Chem. Soc. 141, 9585–9592 (2019).
Deng, G., Teo, B. K. & Zheng, N. Assembly of chiral cluster-based metal–organic frameworks and the chirality memory effect during their disassembly. J. Am. Chem. Soc. 143, 10214–10220 (2021).
Doud, E. A. et al. Superatoms in materials science. Nat. Rev. Mater. 5, 371–387 (2020).
Gunawardene, P. N., Corrigan, J. F. & Workentin, M. S. Golden opportunity: a clickable azide-functionalized [Au25(SR)18]− nanocluster platform for interfacial surface modifications. J. Am. Chem. Soc. 141, 11781–11785 (2019).
Zeng, C., Chen, Y., Kirschbaum, K., Lambright, K. J. & Jin, R. Emergence of hierarchical structural complexities in nanoparticles and their assembly. Science 354, 1580–1584 (2016).
Li, Y. et al. Double-helical assembly of heterodimeric nanoclusters into supercrystals. Nature 594, 380–384 (2021).
Yao, Q. et al. Supercrystal engineering of atomically precise gold nanoparticles promoted by surface dynamics. Nat. Chem. 15, 230–239 (2022).
Lehn, J.-M. Supramolecular chemistry—scope and perspectives molecules, supermolecules, and molecular devices (Nobel Lecture). Angew. Chem. Int. Ed. 27, 89–112 (1988).
Fyfe, M. C. T. & Stoddart, J. F. Synthetic supramolecular chemistry. Acc. Chem. Res. 30, 393–401 (1997).
Nag, A. & Pradeep, T. Assembling atomically precise noble metal nanoclusters using supramolecular interactions. ACS Nanosci. Au 2, 160–178 (2022).
Sergeyev, S., Pisula, W. & Geerts, Y. H. Discotic liquid crystals: a new generation of organic semiconductors. Chem. Soc. Rev. 36, 1902–1929 (2007).
Bosson, J., Gouin, J. & Lacour, J. Cationic triangulenes and helicenes: synthesis, chemical stability, optical properties and extended applications of these unusual dyes. Chem. Soc. Rev. 43, 2824–2840 (2014).
Baisch, B. et al. Mounting freestanding molecular functions onto surfaces: the platform approach. J. Am. Chem. Soc. 131, 442–443 (2009).
Hauptmann, N. et al. Surface control of alkyl chain conformations and 2D chiral amplification. J. Am. Chem. Soc. 135, 8814–8817 (2013).
Wei, Z. et al. Triazatriangulene as binding group for molecular electronics. Langmuir 30, 14868–14876 (2014).
Joshi, C. P., Bootharaju, M. S., Alhilaly, M. J. & Bakr, O. M. [Ag25(SR)18]−: the “golden” silver nanoparticle. J. Am. Chem. Soc. 137, 11578–11581 (2015).
Zhu, M., Aikens, C. M., Hollander, F. J., Schatz, G. C. & Jin, R. Correlating the crystal structure of a thiol-protected Au25 cluster and optical properties. J. Am. Chem. Soc. 130, 5883–5885 (2008).
Aikens, C. M. Origin of discrete optical absorption spectra of M25(SH)18− nanoparticles (M = Au, Ag). J. Phys. Chem. C 112, 19797–19800 (2008).
Jin, R., Zeng, C., Zhou, M. & Chen, Y. Atomically precise colloidal metal nanoclusters and nanoparticles: fundamentals and opportunities. Chem. Rev. 116, 10346–10413 (2016).
Chakraborty, I. & Pradeep, T. Atomically precise clusters of noble metals: emerging link between atoms and nanoparticles. Chem. Rev. 117, 8208–8271 (2017).
Sharma, J. et al. Control of self-assembly of DNA tubules through integration of gold nanoparticles. Science 323, 112–116 (2009).
Yeom, J. et al. Chiral templating of self-assembling nanostructures by circularly polarized light. Nat. Mater. 14, 66–72 (2015).
Lee, H.-E. et al. Amino-acid- and peptide-directed synthesis of chiral plasmonic gold nanoparticles. Nature 556, 360–365 (2018).
Cheng, Z. & Jones, M. R. Assembly of planar chiral superlattices from achiral building blocks. Nat. Commun. 13, 4207 (2022).
Li, Y., Higaki, T., Du, X. & Jin, R. Chirality and surface bonding correlation in atomically precise metal nanoclusters. Adv. Mater. 32, 1905488 (2020).
Dolamic, I., Knoppe, S., Dass, A. & Bürgi, T. First enantioseparation and circular dichroism spectra of Au38 clusters protected by achiral ligands. Nat. Commun. 3, 798 (2012).
Yan, J. et al. Asymmetric synthesis of chiral bimetallic [Ag28Cu12(SR)24]4− nanoclusters via ion pairing. J. Am. Chem. Soc. 138, 12751–12754 (2016).
Jin, Y. et al. Cations controlling the chiral assembly of luminescent atomically precise copper(I) clusters. Angew. Chem. Int. Ed. 58, 12143–12148 (2019).
Liang, X.-Q. et al. Revealing the chirality origin and homochirality crystallization of Ag14 nanocluster at the molecular level. Nat. Commun. 12, 4966 (2021).
W. Laursen, B. & C. Krebs, F. Synthesis, structure, and properties of azatriangulenium salts. Chem. Eur. J. 7, 1773–1783 (2001).
Johnson, E. R. et al. Revealing noncovalent interactions. J. Am. Chem. Soc. 132, 6498–6506 (2010).
Lu, H., Vardeny, Z. V. & Beard, M. C. Control of light, spin and charge with chiral metal halide semiconductors. Nat. Rev. Chem. 6, 470–485 (2022).
Ma, W. et al. Chiral inorganic nanostructures. Chem. Rev. 117, 8041–8093 (2017).
Bisoyi, H. K. & Li, Q. Light-directing chiral liquid crystal nanostructures: from 1D to 3D. Acc. Chem. Res. 47, 3184–3195 (2014).
Solomon, M. L. et al. Nanophotonic platforms for chiral sensing and separation. Acc. Chem. Res. 53, 588–598 (2020).
Turner, D. H., Tinoco, I. Jr. & Maestre, M. Fluorescence detected circular dichroism. J. Am. Chem. Soc. 96, 4340–4342 (1974).
Hassey, R., Swain, E. J., Hammer, N. I., Venkataraman, D. & Barnes, M. D. Probing the chiroptical response of a single molecule. Science 314, 1437–1439 (2006).
Chen, G. et al. Nucleation-mediated growth of chiral 3D organic–inorganic perovskite single crystals. Nat. Chem. 15, 1581–1590 (2023).
Solomon, M. L., Abendroth, J. M., Poulikakos, L. V., Hu, J. & Dionne, J. A. Fluorescence-detected circular dichroism of a chiral molecular monolayer with dielectric metasurfaces. J. Am. Chem. Soc. 142, 18304–18309 (2020).
Mori, T. Chiroptical properties of symmetric double, triple, and multiple helicenes. Chem. Rev. 121, 2373–2412 (2021).
Carr, R., Evans, N. H. & Parker, D. Lanthanide complexes as chiral probes exploiting circularly polarized luminescence. Chem. Soc. Rev. 41, 7673–7686 (2012).
Li, H. et al. Optical activity from anisotropic-nanocluster-assembled supercrystals in achiral crystallographic point groups. J. Am. Chem. Soc. 144, 4845–4852 (2022).
Kumar, J., Nakashima, T., Tsumatori, H. & Kawai, T. Circularly polarized luminescence in chiral aggregates: dependence of morphology on luminescence dissymmetry. J. Phys. Chem. Lett. 5, 316–321 (2014).
Telfer, S. G., McLean, T. M. & Waterland, M. R. Exciton coupling in coordination compounds. Dalton Trans. 40, 3097–3108 (2011).
Xiong, J.-B. et al. The fixed propeller-like conformation of tetraphenylethylene that reveals aggregation-induced emission effect, chiral recognition, and enhanced chiroptical property. J. Am. Chem. Soc. 138, 11469–11472 (2016).
Qiu, Z. et al. Amplification of dissymmetry factors in π-extended [7]- and [9]helicenes. J. Am. Chem. Soc. 143, 4661–4667 (2021).
Izquierdo-García, P. et al. Helical bilayer nanographenes: impact of the helicene length on the structural, electrochemical, photophysical, and chiroptical properties. J. Am. Chem. Soc. 145, 11599–11610 (2023).
Wang, Y., Niu, D., Ouyang, G. & Liu, M. Double helical π-aggregate nanoarchitectonics for amplified circularly polarized luminescence. Nat. Commun. 13, 1710 (2022).
Lu, G. et al. Semitransparent circularly polarized phosphorescent organic light-emitting diodes with external quantum efficiency over 30% and dissymmetry factor close to 10−2. Adv. Funct. Mater. 31, 2102898 (2021).
Brandt, J. R., Wang, X., Yang, Y., Campbell, A. J. & Fuchter, M. J. Circularly polarized phosphorescent electroluminescence with a high dissymmetry factor from PHOLEDs based on a platinahelicene. J. Am. Chem. Soc. 138, 9743–9746 (2016).
Bozoklu, G. et al. Metal-controlled diastereoselective self-assembly and circularly polarized luminescence of a chiral heptanuclear europium wheel. J. Am. Chem. Soc. 134, 8372–8375 (2012).
Ahn, J. et al. A new class of chiral semiconductors: chiral-organic-molecule-incorporating organic–inorganic hybrid perovskites. Mater. Horiz. 4, 851–856 (2017).
Ishii, A. & Miyasaka, T. Direct detection of circular polarized light in helical 1D perovskite-based photodiode. Sci. Adv. 6, eabd3274 (2020).
Wang, J. et al. Aqueous synthesis of low-dimensional lead halide perovskites for room-temperature circularly polarized light emission and detection. ACS Nano 13, 9473–9481 (2019).
Chen, S. et al. Assembly of the thiolated [Au1Ag22(S-Adm)12]3+ superatom complex into a framework material through direct linkage by SbF6− anions. Angew. Chem. Int. Ed. 59, 7542–7547 (2020).
Feng, W. et al. Assembly of mesoscale helices with near-unity enantiomeric excess and light–matter interactions for chiral semiconductors. Sci. Adv. 3, e1601159 (2017).
Guerrero-Martínez, A. et al. Intense optical activity from three-dimensional chiral ordering of plasmonic nanoantennas. Angew. Chem. Int. Ed. 50, 5499–5503 (2011).
Wang, Y., Liu, R., Zhang, Z., Wei, J. & Yang, Z. Large optical asymmetry in silver nanoparticle assemblies enabled by CH−π interaction-mediated chirality transfer. J. Am. Chem. Soc. 145, 4035–4044 (2023).
Lu, J. et al. Enhanced optical asymmetry in supramolecular chiroplasmonic assemblies with long-range order. Science 371, 1368–1374 (2021).
Noguchi, H., Hirose, T., Yokoyama, S. & Matsuda, K. Fluorescence behavior of 2,6,10-trisubstituted 4,8,12-triazatriangulene cations in solution and in the solid state. CrystEngComm 18, 7377–7383 (2016).
Mei, L., Veleta, J. M. & Gianetti, T. L. Helical carbenium ion: a versatile organic photoredox catalyst for red-light-mediated reactions. J. Am. Chem. Soc. 142, 12056–12061 (2020).
Dolomanov, O. V., Bourhis, L. J., Gildea, R. J., Howard, J. A. & Puschmann, H. OLEX2: a ccmplete structure solution, refinement and analysis program. J. Appl. Crystallogr. 42, 339–341 (2009).
Sheldrick, G. M. SHELXT—integrated space-group and crystal-structure determination. Acta Crystallogr. A71, 3–8 (2015).
Sheldrick, G. M. Crystal structure refinement with SHELXL. Acta Crystallogr. C C71, 3–8 (2015).
te Velde, G. et al. Chemistry with ADF. J. Comput. Chem. 22, 931–967 (2001).
Perdew, J. P. Density-functional approximation for the correlation energy of the inhomogeneous electron gas. Phys. Rev. B 33, 8822–8824 (1986).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H–Pu. J. Chem. Phys. 132, 154104 (2010).
van Lenthe, E., Ehlers, A. & Baerends, E.-J. Geometry optimizations in the zero order regular approximation for relativistic effects. J. Chem. Phys. 110, 8943–8953 (1999).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Graph. 14, 33–38 (1996).
Bannwarth, C. & Grimme, S. A simplified time-dependent density functional theory approach for electronic ultraviolet and circular dichroism spectra of very large molecules. Comput. Theor. Chem. 1040–1041, 45–53 (2014).
Grimme, S. & Bannwarth, C. Ultra-fast computation of electronic spectra for large systems by tight-binding based simplified Tamm–Dancoff approximation (sTDA-xTB). J. Chem. Phys. 145, 054103 (2016).
Barca, G. M. J. et al. Recent developments in the general atomic and molecular electronic structure system. J. Chem. Phys. 152, 154102 (2020).
Bannwarth, C., Ehlert, S. & Grimme, S. GFN2-xTB—an accurate and broadly parametrized self-consistent tight-binding quantum chemical method with multipole electrostatics and density-dependent dispersion contributions. J. Chem. Theory Comput. 15, 1652–1671 (2019).
Acknowledgements
We acknowledge support from the Major Research Instrumentation (MRI) Program of the National Science Foundation under NSF award no. 2216066 for crystallography experiments. This work was performed in part at the Harvard University Center for Nanoscale Systems (CNS), a member of the National Nanotechnology Coordinated Infrastructure Network (NNCI), which is supported by the National Science Foundation under NSF award no. ECCS-2025158. We thank the IBS Center for Catalytic Hydrocarbon Functionalizations for computational resources. This research was partially supported by the Arnold and Mabel Beckman Foundation through a Beckman Young Investigator grant awarded to J.A.M. and a National Science Foundation Graduate Research Fellowship awarded to G.J.S.
Author information
Authors and Affiliations
Contributions
J.A.M., Y.L. and G.J.S. conceived the initial idea. G.J.S. and S.T. synthesized and characterized the organic molecules. Y.L. prepared and characterized the co-crystals. G.J.S. and Y.L. collected X-ray diffraction data and solved the crystal structures with the help of S.-L.Z. G.J.S. and Y.L conducted FDCD measurements with the help of A.M. Y.L. and G.J.S. analysed the FDCD data. H.K.K. and G.J.S performed DFT calculations. Y.L., G.J.S., H.K.K. and J.A.M. wrote the paper, and all authors contributed to editing the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Tobias Kraus and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Stability of Ag25 clusters with different counterions.
UV-vis-NIR absorption spectra of a, Ag25(SR)18−PPh4+, b, Ag25(SR)18−TATA3C3+, and c, Ag25(SR)18−TATA3C7+ in CH2Cl2 solution at different time points. Spectra in b and c are offset vertically for clarity.
Extended Data Fig. 2 Representative optical images of NOIC25−3Cm (m = 3–8).
Scale bar = 100 μm.
Extended Data Fig. 3 Calculations of non-covalent interactions.
“Marionette” interactions (top) and C‒H‧‧‧π interactions (bottom) between a TATA+ ion and a Ag25(SR)18− nanocluster in a, NOIC25−3C6‒R, b, NOIC25−3C7‒L, c, NOIC25−3C7‒R, and d, NOIC25−3C8‒R.
Extended Data Fig. 4 Calculated association energy of TATA+ and Ag25(SR)18−–R.
a, Schematic illustration of rotational analysis of a TATA3C7+ residing on a Ag25(SR)18−–R nanocluster at every 10°. b, Relative E(SCF) energies (black circles) of the TATA3C7+ and Ag25(SR)18−–R pair in NOIC25‒3C7‒R as a function of torsional angle and a fit to the values (red dotted line).
Extended Data Fig. 5 Lattice structures of NOICs with symmetric TATAs.
Structures of a, NOIC25−3C6‒L/R, b, NOIC25−3C7‒L/R and c, NOIC25−3C8‒L/R. In TATA3C8+ (panel c), the three C8 chains are disordered among three conformations (marked in different colors) with occupancies of each disorder type = 0.33 for NOIC25−3C8‒L; the C5‒C8 atoms of the three C8 chains (highlighted in orange) are disordered between two conformations with occupancies of both disorder type I/II = 0.5(3) for NOIC25−3C8‒R. Color codes: light blue = N, blue/magenta/yellow/orange/pink = C in TATA+; purple = Ag, yellow = S, grey = C. Note that H atoms and CH2Cl2 solvent molecules in the crystal structures are omitted for clarity.
Extended Data Fig. 6 Photoluminescence (PL) spectra (linearly polarized).
Linearly polarized photoluminescence spectra for Ag25(SR)18−PPh4+ single crystal (black line), NOIC25−3C7 (maroon line), and TATA3C7+BF4‒ single crystal (magenta line). PL intensities are normalized to the peak maximum. λex = 490 nm.
Extended Data Fig. 7 Photoluminescence spectra of NOIC25−3C7 (circular polarized).
Averaged photoluminescence spectra of a, NOIC25−3C7‒L crystal #1, b, NOIC25−3C7‒R crystal #1, c, NOIC25−3C7‒L crystal #2, and d, NOIC25−3C7‒R crystal #2 excited by LCP light (left) and RCP light (right), λex = 490 nm. Four spectra obtained at different positions on the same crystal were averaged (solid lines), and shaded regions correspond to ±1 s.d. e, FDCD IL–IR of NOIC25−3C7‒L (#1 and #2) and NOIC25−3C7‒R (#1 and #2), and f, corresponding FDCD gabs-lum.
Extended Data Fig. 8 Photoluminescence spectra of NOIC25−3C7 (circular polarized).
Averaged photoluminescence spectra of a, NOIC25−3C7‒L crystal #3, b, NOIC25−3C7‒R crystal #3, c, NOIC25−3C7‒L crystal #4, and d, NOIC25−3C7‒R crystal #4 excited by LCP light (left) and RCP light (right), λex = 490 nm. Four spectra obtained at different positions on the same crystal were averaged (solid lines), and shaded regions correspond to ±1 s.d. e, FDCD IL–IR of NOIC25−3C7‒L (#3 and #4) and NOIC25−3C7‒R (#3 and #4), and f, corresponding FDCD gabs-lum.
Extended Data Fig. 9 Effect of intermolecular exciton coupling on the dissymmetry factor.
a, A one-dimensional chain of TATA+ molecules with fixed 20.6 Å spacing and a variable angle, θ, between the transition dipole \(\mathop{{\rm{\mu }}}\limits^{ \rightharpoonup }\) (red) and vector connecting TATA+ centers (blue). b, Rotational strength vs. number of TATAs in the one-dimensional chain for TATA+ molecules rotated 10° clockwise (θ = 100°, red) or 10° counterclockwise (θ = 80°, blue) away from perpendicular. c, Periodic variation of dissymmetry factor as the angle between the intermolecular vector and transition dipole vector changes. The simulated dissymmetry factor corresponding to the experimental θ is indicated in pink.
Extended Data Fig. 10 Effect of crystal orientation on FDCD of NOIC25−3C7‒L/R.
FDCD gabs-lum of a, NOIC25−3C7‒R and b, NOIC25−3C7‒L measured at different crystal rotation angles relative to the incident excitation light. Average values across all rotation angles are shown as solid lines, and shaded regions correspond to ±1 standard deviation. The excitation wavelength was 490 nm for all measurements.
Supplementary information
Supplementary Information (download PDF )
Further Methods and discussion, and Supplementary Figs. 1–17 and Tables 1–18.
Supplementary Data 1
Crystallographic data for NOIC25-2C41C8-A (CCDC no. 2352972).
Supplementary Data 2
Crystallographic data for NOIC25-3C6-L (CCDC no. 2352973).
Supplementary Data 3
Crystallographic data for NOIC25-3C6-R1 (CCDC no. 2352974).
Supplementary Data 4
Crystallographic data for NOIC25-3C6-R2 (CCDC no. 2352975).
Supplementary Data 5
Crystallographic data for NOIC25-3C6-R3 (CCDC no. 2352976).
Supplementary Data 6
Crystallographic data for NOIC25-3C6-R4 (CCDC no. 2352977).
Supplementary Data 7
Crystallographic data for NOIC25-3C6-R5 (CCDC no. 2352978).
Supplementary Data 8
Crystallographic data for NOIC25-3C7-L (CCDC no. 2352979).
Supplementary Data 9
Crystallographic data for NOIC25-3C7-R (CCDC no. 2352980).
Supplementary Data 10
Crystallographic data for NOIC25-3C8-L (CCDC no. 2352981).
Supplementary Data 11
Crystallographic data for NOIC25-3C8-R (CCDC no. 2352982).
Supplementary Data 12
Crystallographic data for NOIC25-2C61C8-L (CCDC no. 2352983).
Supplementary Data 13
Crystallographic data for NOIC25-2C61C8-R (CCDC no. 2352984).
Supplementary Data 14
Crystallographic data for NOIC25-2C81C6-L (CCDC no. 2352985).
Supplementary Data 15
Crystallographic data for NOIC25-2C81C6-R (CCDC no. 2352986).
Supplementary Data 16
Crystallographic data for NOIC25-3C3-M (CCDC no. 2352987).
Supplementary Data 17 (download XLSX )
Statistical source data for Supplementary Figs. 1 and 13–17.
Source data
Source Data Fig. 4 (download XLSX )
Statistical source data for Fig. 4.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data for Extended Data Fig. 1.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data for Extended Data Fig. 6.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data for Extended Data Fig. 7.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical source data for Extended Data Fig. 8.
Source Data Extended Data Fig. 10 (download XLSX )
Statistical source data for Extended Data Fig. 10.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, Y., Stec, G.J., Kim, H.K. et al. Self-assembly of chiroptical ionic co-crystals from silver nanoclusters and organic macrocycles. Nat. Chem. 17, 169–176 (2025). https://doi.org/10.1038/s41557-024-01696-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41557-024-01696-6
This article is cited by
-
Inverse palladocenes
Nature Communications (2026)
-
A bowl-to-bowl programmable hierarchical self-assembly into multidimensional superstructures
Science China Chemistry (2026)
-
Photo-controlled vacancies and conductivity within single crystals of silver chalcogenolate cluster-based MOFs
Nature Communications (2025)
-
Regulation of the photophysical dynamics of metal nanoclusters by manipulating single-point defects
Nature Communications (2025)