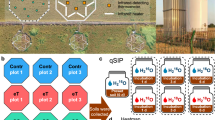
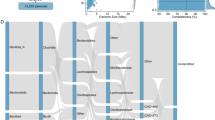
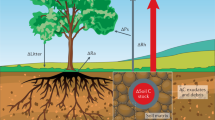
Abstract
Understanding the implications of global warming on the spread of antibiotic resistance genes (ARGs) and virulence factor genes (VFGs) within soil ecosystems is crucial for safeguarding human well-being and sustaining ecosystem health. However, there is currently a lack of large-scale, systematic underpinning data needed to examine this issue. Here, using an integrative approach that combines field experiments, extensive global metagenomic data and microbial culturing, we show that warming enriches bacteria with ARGs and VFGs, increases metabolic complexity and adaptability in bacteria, and accelerates genetic alterations related to ARG and VFGs development. Our validation experiments confirm that the warming effect is more pronounced in colder regions. Machine learning predictions further suggest that warming will increase the soil ARG abundance, especially in some areas that rely heavily on fossil fuels. These results suggest another major negative consequence of global warming, highlighting the importance of developing and implementing sustainability policies that simultaneously combat climate change and antibiotic resistance.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Metagenomic sequencing and metatranscriptomics sequencing data produced in this study were deposited in the NCBI Sequence Read Archive database under accession numbers PRJNA1131773 and PRJNA1131776. Supplementary Tables 1–12 in this study are also publicly available via figshare at https://doi.org/10.6084/m9.figshare.28706258 (ref. 95). Source data are provided with this paper.
Code availability
The R script is available via GitHub at https://github.com/DaLin-lab/Warming-ARGs (ref. 96).
References
The Lancet. The antimicrobial crisis: enough advocacy, more action. Lancet 395, 247 (2020).
Murray, C. J. et al. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet 399, 629–655 (2022).
Allen, J. P., Snitkin, E., Pincus, N. B. & Hauser, A. R. Forest and trees: exploring bacterial virulence with genome-wide association studies and machine learning. Trends Microbiol. 29, 621–633 (2021).
Zheng, D. et al. Global biogeography and projection of soil antibiotic resistance genes. Sci. Adv. 8, eabq8015 (2022).
Zhang, Q. et al. Metagenomic insight into the global dissemination of the antibiotic resistome. Adv. Sci. 10, 202303925 (2023).
Liu, X. et al. Assessing foodborne health risks from dietary exposure to antibiotic resistance genes and opportunistic pathogens in three types of vegetables: an in vitro simulation of gastrointestinal digestion. J. Hazard. Mater. 484, 136731 (2025).
Banerjee, S. & van der Heijden, M. G. A. Soil microbiomes and one health. Nat. Rev. Microbiol. 21, 6–20 (2023).
Chen, Q. L., Cui, H. L., Su, J. Q., Penuelas, J. & Zhu, Y. G. Antibiotic resistomes in plant microbiomes. Trends Plant Sci. 24, 530–541 (2019).
Singh, B. K., Yan, Z. Z., Whittaker, M., Vargas, R. & Abdelfattah, A. Soil microbiomes must be explicitly included in One Health policy. Nat. Microbiol. 8, 1367–1372 (2023).
Speißer, B., Wilschut, R. A. & van Kleunen, M. Number of simultaneously acting global change factors affects composition, diversity and productivity of grassland plant communities. Nat. Commun. 13, 7811 (2022).
Rillig, M. C. et al. The role of multiple global change factors in driving soil functions and microbial biodiversity. Science 366, 886–890 (2019).
The Core Writing Team IPCC. Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change 151 (IPCC, 2015).
Tao, X. et al. Experimental warming accelerates positive soil priming in a temperate grassland ecosystem. Nat. Commun. 15, 1178 (2024).
Delgado-Baquerizo, M. et al. The proportion of soil-borne pathogens increases with warming at the global scale. Nat. Clim. Chang. 10, 550–554 (2020).
Crowther, T. W. et al. Quantifying global soil carbon losses in response to warming. Nature 540, 104–108 (2016).
Dai, Z. et al. Elevated temperature shifts soil N cycling from microbial immobilization to enhanced mineralization, nitrification and denitrification across global terrestrial ecosystems. Glob. Chang. Biol. 26, 5267–5276 (2020).
Wu, L. et al. Reduction of microbial diversity in grassland soil is driven by long-term climate warming. Nat. Microbiol. 7, 1054–1062 (2022).
Yuan, M. M. et al. Climate warming enhances microbial network complexity and stability. Nat. Clim. Chang. 11, 343–348 (2021).
MacFadden, D. R., McGough, S. F., Fisman, D., Santillana, M. & Brownstein, J. S. Antibiotic resistance increases with local temperature. Nat. Clim. Chang. 8, 510–514 (2018).
Li, Z. et al. Climate warming increases the proportions of specific antibiotic resistance genes in natural soil ecosystems. J. Hazard. Mater. 430, 128442 (2022).
Nottingham, A. T. et al. Microbial diversity declines in warmed tropical soil and respiration rise exceed predictions as communities adapt. Nat. Microbiol. 7, 1650–1660 (2022).
Metze, D. et al. Soil warming increases the number of growing bacterial taxa but not their growth rates. Sci. Adv. 10, eadk6295 (2024).
Rodríguez-Verdugo, A., Lozano-Huntelman, N., Cruz-Loya, M., Savage, V. & Yeh, P. Compounding effects of climate warming and antibiotic resistance. iScience 23, 101024 (2020).
Ruan, Y. et al. Elevated temperature and CO2 strongly affect the growth strategies of soil bacteria. Nat. Commun. 14, 391 (2023).
Bay, R. A. et al. Genomic signals of selection predict climate-driven population declines in a migratory bird. Science 359, 83–86 (2018).
Cruz-Loya, M. et al. Stressor interaction networks suggest antibiotic resistance co-opted from stress responses to temperature. ISME J. 13, 12–23 (2019).
Rodríguez-Verdugo, A., Gaut, B. S. & Tenaillon, O. Evolution of Escherichia coli rifampicin resistance in an antibiotic-free environment during thermal stress. BMC Evol. Biol. 13, 50 (2013).
Zhao, W., Zheng, S., Ye, C., Li, J. & Yu, X. Nonlinear impacts of temperature on antibiotic resistance in Escherichia coli. Environ. Sci. Ecotechnol. 22, 100475 (2024).
Zhao, W. et al. High temperatures promote antibiotic resistance genes conjugative transfer under residual chlorine: mechanisms and risks. J. Hazard. Mater. 483, 136675 (2025).
Walsh, T. R., Weeks, J., Livermore, D. M. & Toleman, M. A. Dissemination of NDM-1 positive bacteria in the New Delhi environment and its implications for human health: an environmental point prevalence study. Lancet Infect. Dis. 11, 355–362 (2011).
Delgado-Baquerizo, M. et al. The global distribution and environmental drivers of the soil antibiotic resistome. Microbiome 10, 219 (2022).
Zhang, Z. et al. Assessment of global health risk of antibiotic resistance genes. Nat. Commun. 13, 1553 (2022).
Sáez-Sandino, T. et al. The soil microbiome governs the response of microbial respiration to warming across the globe. Nat. Clim. Chang. 13, 1382–1387 (2023).
Li, Q. et al. Different climate response persistence causes warming trend unevenness at continental scales. Nat. Clim. Chang. 12, 343–349 (2022).
Huang, X. et al. Temperature-dependent trophic associations modulate soil bacterial communities along latitudinal gradients. ISME J. 18, wrae145 (2024).
Wang, M. et al. Responses of soil organic carbon to climate extremes under warming across global biomes. Nat. Clim. Chang. 14, 98–105 (2024).
Kossin, J. P. A global slowdown of tropical-cyclone translation speed. Nature 558, 104–107 (2018).
Wang, M. et al. Global soil profiles indicate depth-dependent soil carbon losses under a warmer climate. Nat. Commun. 13, 5514 (2022).
Kang, L. et al. Metagenomic insights into microbial community structure and metabolism in alpine permafrost on the Tibetan Plateau. Nat. Commun. 15, 5920 (2024).
Lin, D., Xu, J. Y., Wang, L., Du, S. & Zhu, D. Long-term application of organic fertilizer prompting the dispersal of antibiotic resistance genes and their health risks in the soil plastisphere. Environ. Int. 183, 108431 (2024).
Wood, J. L. et al. Rethinking CSR theory to incorporate microbial metabolic diversity and foraging traits. ISME J. 17, 1793–1797 (2023).
Zhang, S. et al. Distribution of ARGs and MGEs among glacial soil, permafrost, and sediment using metagenomic analysis. Environ. Pollut. 234, 339–346 (2018).
Qian, X. et al. Metagenomic analysis reveals the shared and distinct features of the soil resistome across tundra, temperate prairie, and tropical ecosystems. Microbiome 9, 108 (2021).
McDonald, M. D. et al. What is microbial dormancy? Trends Microbiol. 32, 142–150 (2024).
Littlejohn, P. T. et al. Multiple micronutrient deficiencies in early life cause multi-kingdom alterations in the gut microbiome and intrinsic antibiotic resistance genes in mice. Nat. Microbiol. 8, 2392–2405 (2023).
Wang, J. et al. Supercarriers of antibiotic resistome in a world’s large river. Microbiome 10, 111 (2022).
Dai, D. et al. Long-read metagenomic sequencing reveals shifts in associations of antibiotic resistance genes with mobile genetic elements from sewage to activated sludge. Microbiome 10, 20 (2022).
Johnston, E. R. et al. Responses of tundra soil microbial communities to half a decade of experimental warming at two critical depths. Proc. Natl Acad. Sci. USA 116, 15096–15105 (2019).
Yang, S. et al. Effects of a long-term anoxic warming scenario on microbial community structure and functional potential of permafrost-affected soil. Permafr. Periglac. Process. 32, 641–656 (2021).
Wang, C. et al. Bacterial genome size and gene functional diversity negatively correlate with taxonomic diversity along a pH gradient. Nat. Commun. 14, 32 (2023).
Westoby, M. et al. Trait dimensions in bacteria and archaea compared to vascular plants. Ecol. Lett. 24, 1487–1504 (2021).
Fierer, N. Embracing the unknown: disentangling the complexities of the soil microbiome. Nat. Rev. Microbiol. 15, 579–590 (2017).
Krause, S. et al. Trait-based approaches for understanding microbial biodiversity and ecosystem functioning. Front. Microbiol. 5, 251 (2014).
Zhang, H. Y., Bissett, A., Aguilar-Trigueros, C. A., Liu, H. W. & Powell, J. R. Fungal genome size and composition reflect ecological strategies along soil fertility gradients. Ecol. Lett. 26, 1108–1118 (2023).
Darby, E. M. et al. Molecular mechanisms of antibiotic resistance revisited. Nat. Rev. Microbiol. 21, 280–295 (2023).
Cordero, M., Mitarai, N. & Jauffred, L. Motility mediates satellite formation in confined biofilms. ISME J. 17, 1819–1827 (2023).
Ciofu, O., Moser, C., Jensen, P. Ø. & Høiby, N. Tolerance and resistance of microbial biofilms. Nat. Rev. Microbiol. 20, 621–635 (2022).
Rosas, N. C. & Lithgow, T. Targeting bacterial outer-membrane remodelling to impact antimicrobial drug resistance. Trends Microbiol. 30, 544–552 (2022).
Andersson, D. I. & Hughes, D. Microbiological effects of sublethal levels of antibiotics. Nat. Rev. Microbiol. 12, 465–478 (2014).
Li, Z. et al. Integrated human skin bacteria genome catalog reveals extensive unexplored habitat-specific microbiome diversity and function. Adv. Sci. 10, 2300050 (2023).
Du, D. et al. Multidrug efflux pumps: structure, function and regulation. Nat. Rev. Microbiol. 16, 523–539 (2018).
El Meouche, I. & Dunlop, M. J. Heterogeneity in efflux pump expression predisposes antibiotic-resistant cells to mutation. Science 362, 686–690 (2018).
Wang, Y. et al. Antidepressants can induce mutation and enhance persistence toward multiple antibiotics. Proc. Natl Acad. Sci. USA 120, e2208344120 (2023).
Stalder, T. et al. Evolving populations in biofilms contain more persistent plasmids. Mol. Biol. Evol. 37, 1563–1576 (2020).
Yang, Y. et al. Structures of the RNA polymerase-σ54 reveal new and conserved regulatory strategies. Science 349, 882–885 (2015).
Whiteley, M., Diggle, S. P. & Greenberg, E. P. Progress in and promise of bacterial quorum sensing research. Nature 551, 313–320 (2017).
Wang, X. et al. Hot moment of N2O emissions in seasonally frozen peatlands. ISME J. 17, 792–802 (2023).
Bei, Q. et al. Extreme summers impact cropland and grassland soil microbiomes. ISME J. 17, 1589–1600 (2023).
Donhauser, J., Qi, W., Bergk-Pinto, B. & Frey, B. High temperatures enhance the microbial genetic potential to recycle C and N from necromass in high-mountain soils. Glob. Chang. Biol. 27, 1365–1386 (2021).
Levy-Booth, D. J. et al. Genomics and metatranscriptomics of biogeochemical cycling and degradation of lignin-derived aromatic compounds in thermal swamp sediment. ISME J. 15, 879–893 (2021).
ter Horst, A. M. et al. Minnesota peat viromes reveal terrestrial and aquatic niche partitioning for local and global viral populations. Microbiome 9, 233 (2021).
Dove, N. C., Torn, M. S., Hart, S. C. & Taş, N. Metabolic capabilities mute positive response to direct and indirect impacts of warming throughout the soil profile. Nat. Commun. 12, 2089 (2021).
Chen, S., Zhou, Y., Chen, Y. & Gu, J. Fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 34, i884–i890 (2018).
Yin, X. et al. ARGs-OAP v3.0: antibiotic-resistance gene database curation and analysis pipeline optimization. Engineering 27, 234–241 (2023).
Liu, B., Zheng, D., Zhou, S., Chen, L. & Yang, J. VFDB 2022: a general classification scheme for bacterial virulence factors. Nucleic Acids Res. 50, D912–D917 (2022).
Pärnänen, K. et al. Maternal gut and breast milk microbiota affect infant gut antibiotic resistome and mobile genetic elements. Nat. Commun. 9, 3891 (2018).
Buchfink, B., Reuter, K. & Drost, H. G. Sensitive protein alignments at tree-of-life scale using DIAMOND. Nat. Methods 18, 366–368 (2021).
Li, D., Liu, C. M., Luo, R., Sadakane, K. & Lam, T. W. MEGAHIT: an ultra-fast single-node solution for large and complex metagenomics assembly via succinct de Bruijn graph. Bioinformatics 31, 1674–1676 (2015).
West, P. T., Probst, A. J., Grigoriev, I. V., Thomas, B. C. & Banfield, J. F. Genome-reconstruction for eukaryotes from complex natural microbial communities. Genome Res. 28, 569–580 (2018).
Malik, A. A. et al. Defining trait-based microbial strategies with consequences for soil carbon cycling under climate change. ISME J. 14, 1–9 (2020).
Piton, G. et al. Life history strategies of soil bacterial communities across global terrestrial biomes. Nat. Microbiol. 8, 2093–2102 (2023).
Nayfach, S. & Pollard, K. S. Average genome size estimation improves comparative metagenomics and sheds light on the functional ecology of the human microbiome. Genome Biol. 16, 51 (2015).
Mikheenko, A., Prjibelski, A., Saveliev, V., Antipov, D. & Gurevich, A. Versatile genome assembly evaluation with QUAST-LG. Bioinformatics 34, i142–i150 (2018).
Pereira-Flores, E., Glöckner, F. O. & Fernandez-Guerra, A. Fast and accurate average genome size and 16S rRNA gene average copy number computation in metagenomic data. BMC Bioinform. 20, 453 (2019).
Kang, D. D., Froula, J., Egan, R. & Wang, Z. MetaBAT, an efficient tool for accurately reconstructing single genomes from complex microbial communities. PeerJ 3, e1165 (2015).
Wu, Y. W., Simmons, B. A. & Singer, S. W. MaxBin 2.0: an automated binning algorithm to recover genomes from multiple metagenomic datasets. Bioinformatics 32, 605–607 (2016).
Alneberg, J. et al. Binning metagenomic contigs by coverage and composition. Nat. Methods 11, 1144–1146 (2014).
Parks, D. H., Imelfort, M., Skennerton, C. T., Hugenholtz, P. & Tyson, G. W. CheckM: assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res. 25, 1043–1055 (2015).
Parks, D. H. et al. A standardized bacterial taxonomy based on genome phylogeny substantially revises the tree of life. Nat. Biotechnol. 36, 996 (2018).
Hyatt, D. et al. Prodigal: prokaryotic gene recognition and translation initiation site identification. BMC Bioinform. 11, 119 (2010).
Olm, M. R. et al. inStrain profiles population microdiversity from metagenomic data and sensitively detects shared microbial strains. Nat. Biotechnol. 39, 727–736 (2021).
Weissman, J. L., Hou, S. & Fuhrman, J. A. Estimating maximal microbial growth rates from cultures, metagenomes, and single cells via codon usage patterns. Proc. Natl Acad. Sci. USA 118, e2016810118 (2021).
Yang, K. et al. Mycorrhizal type regulates trade-offs between plant and soil carbon in forests. Nat. Clim. Chang. 14, 91–97 (2024).
Zhang, Z. et al. Global biogeography of microbes driving ocean ecological status under climate change. Nat. Commun. 15, 4657 (2024).
Lin, D. et al. Climate warming fuels the global antibiotic resistome by altering soil bacterial traits. figshare https://doi.org/10.6084/m9.figshare.28706258 (2025).
Lin, D. et al. Climate warming fuels the global antibiotic resistome by altering soil bacterial traits. GitHub https://github.com/DaLin-lab/Warming-ARGs (2025).
Söllinger, A. et al. Down-regulation of the bacterial protein biosynthesis machinery in response to weeks, years, and decades of soil warming. Sci. Adv. 8, eabm3230 (2022).
Acknowledgements
We acknowledge the funds of the National Natural Science Foundation of China (grant nos. 42222701 to D.Z. and 42090063 to Y.-G.Z.), J.P. was supported by the Catalan Government grant no. SGR·2021-1333, Youth Innovation Promotion Association, Chinese Academy of Sciences (grant no. 2023321 to D.Z.), Ningbo Yongjiang Talent Project (grant no. 2022A-163-G to D.Z.) and Ningbo S&T project (grant no. 2021-DST-004 to Y.-G.Z.).
Author information
Authors and Affiliations
Contributions
D.L., S.-Y.-D.Z. and D.Z. conceived and designed the research. D.L., S.-Y.-D.Z. and D.Z. performed the experiments. D.L., S.D., Z.Z., T.Z. and L.W. analysed the data and prepared the figures. D.L., S.D., Q.Z., S.-Y.-D.Z., D.W.G., D.T.T., D.Z., Y.-G.Z., J.P. and P.B.R. wrote and revised the paper. All authors read and approved the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Steven Djordjevic, Hang-Wei Hu, Carlos Guerra, Madhav Thakur and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Global distribution of ARG pollution at different latitudes.
The relationship between (a) ARGs, (b) risk index of ARGs, (c) VFGs and temperature in global soil at different latitudes. Linear regression model with a two-sided test was used for statistical analysis.
Extended Data Fig. 2 The role of climate variables in shaping the response of soil ARG pollution to warming.
The relative importance of climate variables in influencing the response of soil (a) ARGs, (b) risk index of ARGs and (c) VFGs to warming. The relative importances of climate variable was estimated by calculating the percentage increases in the mean squared error (MSE) of the variables in random forest models (n = 326). Significant differences are denoted as * (P < 0.05) and ** (P < 0.01).
Extended Data Fig. 3 The overall effect of artificial warming on VFGs and MGEs in soil.
The bars around the mean represent the 95% confidence interval. If the 95% confidence interval (CI) does not overlap with zero, the impact of warming on ARG-related variables is considered significant. Blue, red and gray indicate negative significant, positive significant and insignificant effects of warming on these variables, respectively (n = 150).
Extended Data Fig. 4 The relationships between the effect of warming and other factors.
(a) Relationships between effect sizes of warming on ARGs and the duration of warming treatment. We excluded three samples from Söllinger et al.97 with warming durations exceeding 56 years, as their treatment durations were considerably different from those of the other samples. (b) Relationships between effect sizes of warming on risk index of ARGs and soil depth. Linear regression model with a two-sided test was used for statistical analysis.
Extended Data Fig. 5 Features of bacterial community.
Comparison of the abundance of ARGs, MGEs and VFGs between (a) Proteobacteria (n = 77) or (b) bacteria (n = 997) positively affected by increasing temperatures and those negatively affected by warming. The number of ARGs, VFGs and MGEs is normalized using a linear scale, with all raw values standardized within the same range of 0-10. P values were calculated using two-tailed unpaired Student’s t test. The tops of the boxes represent the 75th percentile, the bottoms indicate the 25th percentile, and the center lines denote the median. The whiskers extend to the maximum and minimum non-outlier values.
Extended Data Fig. 6 Features of ‘high-risk’ bacteria.
Comparison of (a) maximum growth rates and (b) the number of ARGs, MGEs and VFGs between potential ‘high-risk’ bacteria (n = 84) positively affected by increasing temperatures and those negatively affected by warming. The number of ARGs, VFGs and MGEs is normalized using a linear scale, with all raw values standardized within the same range of 0-10. P values were calculated using two-tailed unpaired Student’s t test.
Extended Data Fig. 7 Composition of ‘high-risk’ bacteria.
Composition of (a) potential “high-risk” bacteria without MGEs and (b) potential ‘high-risk’ bacteria carrying MGEs (n = 84). The red and blue parts in the pie chart represent the proportion of potential “high-risk” bacteria increased and decreased under warming, respectively.
Extended Data Fig. 8 The co-occurrence patterns of ARGs, MGEs and VFGs on the contigs in the 84 potential ‘high-risk’ bacteria.
Potential ‘high-risk’ bacteria represent bacterial genotypes that undergo substantial changes (P < 0.05) in abundance with warming, as observed in meta-analysis, and also carrying ARGs and VFGs.
Extended Data Fig. 9 Gene abundance and expression in Escherichia coli.
(a) Abundance of ARGs and MGEs in Escherichia coli incubated at different temperatures (n = 12). Data are presented as mean values ± SD. (b) Comparison of gene expression differences in Escherichia coli incubated at different temperatures. P values were calculated using one-way ANOVA with LSD-test. Different lowercase letters indicate significant differences among the treatments at P < 0.05.
Extended Data Fig. 10 Future projections of changes in ARG abundance under three future climate scenarios.
‘Fold change in ARG abundance’ represents the change in ARG abundance of future climate scenarios relative to current conditions, with the values normalized using Log2 (n = 512).
Supplementary information
Supplementary Information (download PDF )
Supplementary Texts 1–4 and Figs. 1–4.
Supplementary Tables (download XLSX )
Supplementary Tables 1–12
Source data
Source Data Figs. 2–5, Extended Data Figs. 1–10 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, D., Du, S., Zhao, Z. et al. Climate warming fuels the global antibiotic resistome by altering soil bacterial traits. Nat Ecol Evol 9, 1512–1526 (2025). https://doi.org/10.1038/s41559-025-02740-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41559-025-02740-5
This article is cited by
-
Diet and environmental factors jointly drive the gut microbiome, resistome, and virulome of urban bats
npj Biofilms and Microbiomes (2026)
-
Increasing microbial risks under co-contamination: View from virulence factor genes
Soil Ecology Letters (2026)
-
Effects of predatory mite biocontrols on the dispersal of antibiotic resistomes and virulence factors in tea garden soils
Microbiome (2025)
-
Climate warming fuels the global antibiotic resistome by altering soil bacterial traits
Nature Ecology & Evolution (2025)
-
Warming threatens soil health
Nature Ecology & Evolution (2025)