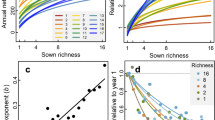
Abstract
The degree of synchronous versus compensatory dynamics among species is crucial for determining the stability of ecological communities. Although robust quantification of species synchrony requires long-term observations, empirical studies are often based on short time series. Here we explore the effects of time series length on species synchrony by combining spectral analysis, dynamical community models and empirical plant community data. Our theoretical analyses show that competition contributes to decreasing species synchrony over long timescales but causes increases in synchrony over short timescales. As a result, species synchrony tends to decrease with time series length. In model communities, species synchrony calculated from long time series decreases with species diversity and competition, whereas that calculated from short time series increases with diversity and competition. Empirical analyses of >2,000 time series of plant communities support these theoretical predictions. Our analyses demonstrate that both species synchrony itself and its relationship with species richness can exhibit opposite patterns, depending on the length of time series, challenging the implicit assumption in ecological studies that observational length should not qualitatively alter patterns of interest. Our findings help reconcile results from theoretical and empirical studies on synchrony and have implications for sampling design.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The analysed data and the codes to generate simulated data are available via figshare at https://figshare.com/s/52cd8c1a35f7cb8d801c (ref. 44).
Code availability
Numerical simulations were performed on Matlab2024b. Data analyses were performed on R (v.4.4.0) and linear mixed-effects models are analysed using R package lme4. Codes are available via figshare at https://figshare.com/s/52cd8c1a35f7cb8d801c (ref. 44).
Change history
09 July 2025
In the version of the article initially published, in the Acknowledgements, the National Key Research and Development Programme of China grant number was incorrect and has now been amended to 2022YFF0802103 in the HTML and PDF versions of the article.
References
Donohue, I. et al. Navigating the complexity of ecological stability. Ecol. Lett. 19, 1172–1185 (2016).
Shoemaker, L. G. et al. Integrating the underlying structure of stochasticity into community ecology. Ecology 101, e02922 (2020).
Gonzalez, A. & Loreau, M. The causes and consequences of compensatory dynamics in ecological communities. Annu. Rev. Ecol. Evol. Syst. 40, 393–414 (2009).
Loreau, M. et al. Biodiversity as insurance: from concept to measurement and application. Biol. Rev. 96, 2333–2354 (2021).
Ernest, S. & Brown, J. H. Homeostasis and compensation: the role of species and resources in ecosystem stability. Ecology 82, 2118–2132 (2001).
Bai, Y., Han, X., Wu, J., Chen, Z. & Li, L. Ecosystem stability and compensatory effects in the Inner Mongolia grassland. Nature 431, 181–184 (2004).
Valencia, E. et al. Synchrony matters more than species richness in plant community stability at a global scale. Proc. Natl Acad. Sci. USA 117, 24345–24351 (2020).
Tilman, D., Lehman, C. L. & Bristow, C. E. Diversity–stability relationships: statistical inevitability or ecological consequence? Am. Nat. 151, 277–282 (1998).
Yachi, S. & Loreau, M. Biodiversity and ecosystem productivity in a fluctuating environment: the insurance hypothesis. Proc. Natl Acad. Sci. USA 96, 1463–1468 (1999).
Hallett, L. M. et al. Biotic mechanisms of community stability shift along a precipitation gradient. Ecology 95, 1693–1700 (2014).
Ives, A. R., Gross, K. & Klug, J. L. Stability and variability in competitive communities. Science 286, 542–544 (1999).
Lehman, C. L. & Tilman, D. Biodiversity, stability, and productivity in competitive communities. Am. Nat. 156, 534–552 (2000).
Gross, K. et al. Species richness and the temporal stability of biomass production: a new analysis of recent biodiversity experiments. Am. Nat. 183, 1–12 (2014).
Loreau, M. & de Mazancourt, C. Species synchrony and its drivers: neutral and nonneutral community dynamics in fluctuating environments. Am. Nat. 172, E48–E66 (2008).
Loreau, M. & De Mazancourt, C. Biodiversity and ecosystem stability: a synthesis of underlying mechanisms. Ecol. Lett. 16, 106–115 (2013).
Zhao, L. et al. Biodiversity stabilizes plant communities through statistical-averaging effects rather than compensatory dynamics. Nat. Commun. 13, 7804 (2022).
De Mazancourt, C. et al. Predicting ecosystem stability from community composition and biodiversity. Ecol. Lett. 16, 617–625 (2013).
Zhao, L. et al. A new variance ratio metric to detect the timescale of compensatory dynamics. Ecosphere 11, e03114 (2020).
Shoemaker, L. G. et al. The long and the short of it: mechanisms of synchronous and compensatory dynamics across temporal scales. Ecology 103, e3650 (2022).
Houlahan, J. et al. Compensatory dynamics are rare in natural ecological communities. Proc. Natl Acad. Sci. USA 104, 3273–3277 (2007).
Davidson, J. L. & Shoemaker, L. G. Resistance and resilience to invasion is stronger in synchronous than compensatory communities. Ecology 104, e4162 (2023).
Yan, Y. et al. Mechanistic links between biodiversity effects on ecosystem functioning and stability in a multi‐site grassland experiment. J. Ecol. 109, 3370–3378 (2021).
Hautier, Y. et al. Eutrophication weakens stabilizing effects of diversity in natural grasslands. Nature 508, 521–525 (2014).
Craven, D. et al. Multiple facets of biodiversity drive the diversity–stability relationship. Nat. Ecol. Evol. 2, 1579–1587 (2018).
Schluter, D. A variance test for detecting species associations, with some example applications. Ecology 65, 998–1005 (1984).
Vasseur, D. A. & Gaedke, U. Spectral analysis unmasks synchronous and compensatory dynamics in plankton communities. Ecology 88, 2058–2071 (2007).
Brillinger, D. R. Time Series: Data Analysis and Theory (Society for Industrial and Applied Mathematics, 2001).
Luo, M. et al. The effects of dispersal on spatial synchrony in metapopulations differ by timescale. Oikos 130, 1762–1772 (2021).
Fagan, W. F., Lynch, H. J. & Noon, B. R. Pitfalls and challenges of estimating population growth rate from empirical data: consequences for allometric scaling relations. Oikos 119, 455–464 (2010).
Cortés, E. Perspectives on the intrinsic rate of population growth. Methods Ecol. Evol. 7, 1136–1145 (2016).
Pimm, S. L. & Redfearn, A. The variability of population densities. Nature 334, 613–614 (1988).
Inchausti, P. & Halley, J. The long-term temporal variability and spectral colour of animal populations. Evol. Ecol. Res. 4, 1033–1048 (2002).
Hallett, L. M. et al. codyn: an R package of community dynamics metrics. Methods Ecol. Evol. 7, 1146–1151 (2016).
Lepš, J. et al. Accounting for long‐term directional trends on year‐to‐year synchrony in species fluctuations. Ecography 42, 1728–1741 (2019).
Hoover, C. et al. Forest inventory and analysis data in action: examples from eastern national forests. Trees For. People 7, 100178 (2022).
Davies, S. J. et al. ForestGEO: understanding forest diversity and dynamics through a global observatory network. Biol. Conserv. 253, 108907 (2021).
Likens, G. E. The science of nature, the nature of science: long-term ecological studies at Hubbard Brook. Proc. Am. Philos. Soc. 143, 558–572 (1999).
Kominoski, J. S., Gaiser, E. E. & Baer, S. G. Advancing theories of ecosystem development through long-term ecological research. BioScience 68, 554–562 (2018).
Wang, S. et al. Biotic homogenization destabilizes ecosystem functioning by decreasing spatial asynchrony. Ecology 102, e03332 (2021).
Hastings, A. Timescales and the management of ecological systems. Proc. Natl Acad. Sci. USA 113, 14568–14573 (2016).
Reich, P. B., Hobbie, S. E., Lee, T. D. & Pastore, M. A. Unexpected reversal of C3 versus C4 grass response to elevated CO2 during a 20-year field experiment. Science 360, 317–320 (2018).
Shriver, R. K. et al. Transient population dynamics impede restoration and may promote ecosystem transformation after disturbance. Ecol. Lett. 22, 1357–1366 (2019).
Strogatz, S. Sync: The Emerging Science of Spontaneous Order (Penguin UK, 2004).
Luo, M. Code_spectral analysis_competition_synchrony. figshare https://figshare.com/s/52cd8c1a35f7cb8d801c (2025).
Acknowledgements
This work is supported by the National Key Research and Development Programme of China (2022YFF0802103) and the National Natural Science Foundation of China (32425036, 32588202) and is part of the Long-Term Ecological Research (LTER) Synchrony Synthesis Group funded by the National Science Foundation (US NSF) under grant no. DEB-1545288, through the LTER Network Communications Office and hosted at the National Center for Ecological Analysis and Synthesis. Cedar Creek data collection was supported by NSF LTER grant nos DEB-0620652, DEB-1234162 and DEB-1831944. D.R. was partly supported by US NCF grant nos 2023474 and 2414418 and the McDonnell and Humboldt foundations. L.S. was supported by NSF grant nos 2033292 and 2019528. P.B.R. was supported by NSF Long-Term Research in Environmental Biology (LTREB) grant nos DEB-1242531 and DEB-1753859; Ecosystem Sciences grant no. DEB-1120064; Biocomplexity grant no. DEB-0322057 and the ASCEND Biological Integration Institutes grant no. US NSF-DBI-2021898. M. Loreau was supported by the TULIP Laboratory of Excellence (ANR-10-LABX-41).
Author information
Authors and Affiliations
Contributions
S.W. designed the research. M. Luo and S.W. performed the research. M. Luo derived analytic solutions and analysed the model and data. M. Luo, L.M.H., D.C.R., L.G.S., L.Z. and S.W. contributed new reagents/analytic tools. P.B.R. and D.T. curated the field data. M. Luo and S.W. wrote the first draft of the paper. L.M.H., D.C.R., L.G.S., L.Z., L.J., M. Loreau, P.B.R. and D.T. contributed substantially to revision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Takehiro Sasaki, David Vasseur and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Effects of time series length on synchrony under different scenarios of environmental noises.
Effects of time series length on species synchrony in two-species symmetric Lotka-Volterra competition models under different scenarios of correlation in species environmental drivers (a) and temporal autocorrelation in environmental noise (b). Other parameters are same as in Fig. 2.
Extended Data Fig. 2 Effects of competition strength and species richness on species synchrony in short and long time series under different degrees of species correlation in environmental response.
Effects of competition strength (a,b) and species richness (c,d) on species synchrony in symmetric Lotka-Volterra competition models under different scenarios of correlation in species environmental response (ρ), in short (a,c) and long (b,d) time series. Other parameters are same as in Fig. 2.
Extended Data Fig. 3 Effects of competition strength and species richness on species synchrony in short and long time series under different degrees of temporal autocorrelation in environmental noise.
Effects of competition strength (a,b) and species richness (c,d) on species synchrony in two-species symmetric Lotka-Volterra competition models under different scenarios of temporal autocorrelation in environmental noise (q), in short (a,c) and long (b,d) time series. Other parameters are same as in Fig. 2.
Extended Data Fig. 4 Competition-driven synchrony over short timescales versus compensatory dynamics over long timescales.
(a) and (b) illustrates the responses of a two-species community to a pulse perturbation, with the blue and red curves representing the abundances of two species in a deterministic Lotka-Volterra competition model (that is without environmental noises; other parameters are same as for Fig. 2). Starting from equilibrium (both species at population size X* = 6.67), a perturbation occurs that suddenly increased (a) or decreased (b) the abundance of species 1. Taking (a) for example. Following the perturbation, the two species both decreased, leading to synchronous dynamics in the short term. These synchronous dynamics were followed by compensatory dynamics in the long term, where the species 2 begins to increase once the abundance of the species 1 recover to a certain level. (c) and (d) illustrates the responses of the community to continuous random perturbations (that is, environmental noises). In (c), the population dynamics of the two species are positively correlated at short terms (for example, grey areas) but negatively correlated at long terms. The numbers above the grey areas represented the correlation coefficients during the observational window. In (d), the population growth rates of the two species are positively correlated at both short and long terms.
Extended Data Fig. 5 Effects of time series length on two alternative synchrony metrics.
Effects of time series length on two alternative synchrony metrics: the mean pairwise correlation coefficients (a-c), and the LM synchrony metric developed by ref. 14 (b-d). (a,d) show the change in synchrony with increasing time series length. (b,c) show the relationship of synchrony with species richness, and (c,f) show the relationship of synchrony with competition strengths. In (a,d), competition strength = 0.5 and species richness = 2; in (b,e), species richness = 2; in (c,f), competition strength = 0.5. Other parameters are same as in Fig. 2.
Extended Data Fig. 6 The critical time series length for observing a negative regression slope between species diversity and synchrony.
Relationship between the critical time series length and the population growth rate, under different scenarios of experimental replicates (a) or competition strength (b). Based on our symmetric competition models, the critical time series length is determined as the minimum time series length with more than 80% chance of observing a negative regression slope between diversity and synchrony. In (a), a number of replicates (1 or 5) was simulated for each level of species richness (S = 2, 3, …, 10), and the synchrony-diversity relationships were constructed across all simulated communities under given time series length. In (b), results under two different competition strengths were presented. Other parameters are same as for Fig. 2.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–4 and Figs. 1–7.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Luo, M., Hallett, L.M., Reuman, D.C. et al. Short time series obscure compensatory dynamics in ecological communities. Nat Ecol Evol 9, 1405–1413 (2025). https://doi.org/10.1038/s41559-025-02757-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41559-025-02757-w