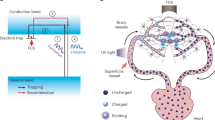
Abstract
Biological systems operate across distributed regions with fast, localized dynamics, yet existing biointerfaces fall short of providing both high spatiotemporal precision and the ability to dynamically target any region without disturbing surrounding tissue. Here we present an in vivo deep-tissue light source based on focused ultrasound scanning of mechanoluminescent nanotransducers circulating through the vasculature. We demonstrate the programmability of this approach in tissue-mimicking phantoms and the endogenous circulatory system of animals, where tunable spatial resolution and dynamic light patterning are achieved. We validate the functionality of the ultrasound-scanning light source in opsin-expressing neurons through electrophysiological recordings and immunostaining in both the brain and the spinal cord. We showcase dynamic three-dimensional brain targeting and temporally resolved behavioural control in freely moving animals via the ultrasound-scanning in vivo light source. This non-invasive deep-tissue light source offers a versatile strategy for body-wide optical interfacing.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The data that support the findings of this study are available within this article and its Supplementary Information. Source data are provided with this paper.
Code availability
The custom MATLAB code used in this study for spike sorting is available at https://github.com/ShanJiang1233/Jiang_2025 and is archived at Zenodo at https://doi.org/10.5281/zenodo.18609351 (ref. 65).
References
Schaffer, L. V. & Ideker, T. Mapping the multiscale structure of biological systems. Cell Syst. 12, 622–635 (2021).
Hosang, L. et al. The lung microbiome regulates brain autoimmunity. Nature 603, 138–144 (2022).
Simon, H. A. in Facets of Systems Science 457–476 (Springer, 1991).
Sahasrabudhe, A., Cea, C. & Anikeeva, P. Multifunctional bioelectronics for brain–body circuits. Nat. Rev. Bioeng 3, 465–484 (2025).
Hong, G. & Lieber, C. M. Novel electrode technologies for neural recordings. Nat. Rev. Neurosci. 20, 330–345 (2019).
Won, S. M., Cai, L., Gutruf, P. & Rogers, J. A. Wireless and battery-free technologies for neuroengineering. Nat. Biomed. Eng. 7, 405–423 (2023).
Shahriari, D., Rosenfeld, D. & Anikeeva, P. Emerging frontier of peripheral nerve and organ interfaces. Neuron 108, 270–285 (2020).
Zhu, X., Menozzi, L., Cho, S.-W. & Yao, J. High speed innovations in photoacoustic microscopy. npj Imaging 2, 46 (2024).
Hu, Z. et al. Airy-beam holographic sonogenetics for advancing neuromodulation precision and flexibility. Proc. Natl Acad. Sci. USA 121, e2402200121 (2024).
Hong, G., Antaris, A. L. & Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 1, 0010 (2017).
Yun, S. H. & Kwok, S. J. J. Light in diagnosis, therapy and surgery. Nat. Biomed. Eng. 1, 0008 (2017).
Darmani, G. et al. Non-invasive transcranial ultrasound stimulation for neuromodulation. Clin. Neurophysiol. 135, 51–73 (2022).
Guo, H. et al. Ultrasound produces extensive brain activation via a cochlear pathway. Neuron 98, 1020–1030.e4 (2018).
Szablowski, J. O., Lee-Gosselin, A., Lue, B., Malounda, D. & Shapiro, M. G. Acoustically targeted chemogenetics for the non-invasive control of neural circuits. Nat. Biomed. Eng 2, 475–484 (2018).
Ouyang, W. et al. A wireless and battery-less implant for multimodal closed-loop neuromodulation in small animals. Nat. Biomed. Eng. 7, 1252–1269 (2023).
Wang, W. et al. Ultrasound-induced cascade amplification in a mechanoluminescent nanotransducer for enhanced sono-optogenetic deep brain stimulation. ACS Nano 17, 24936–24946 (2023).
Jiang, Y. et al. Rational design of silicon structures for optically controlled multiscale biointerfaces. Nat. Biomed. Eng. 2, 508–521 (2018).
Chen, S. et al. Near-infrared deep brain stimulation via upconversion nanoparticle-mediated optogenetics. Science 359, 679–684 (2018).
Wu, X. et al. Tether-free photothermal deep-brain stimulation in freely behaving mice via wide-field illumination in the near-infrared-II window. Nat. Biomed. Eng. 6, 754–770 (2022).
Prominski, A. et al. Porosity-based heterojunctions enable leadless optoelectronic modulation of tissues. Nat. Mater. 21, 647–655 (2022).
Kim, Y. J. et al. Magnetoelectric nanodiscs enable wireless transgene-free neuromodulation. Nat. Nanotechnol. 20, 121–131 (2025).
Chen, R., Romero, G., Christiansen, M. G., Mohr, A. & Anikeeva, P. Wireless magnetothermal deep brain stimulation. Science 347, 1477–1480 (2015).
Sebesta, C. et al. Subsecond multichannel magnetic control of select neural circuits in freely moving flies. Nat. Mater. 21, 951–958 (2022).
Yang, Y. et al. Induction of a torpor-like hypothermic and hypometabolic state in rodents by ultrasound. Nat. Metab. 5, 789–803 (2023).
Kuang, X. et al. Self-enhancing sono-inks enable deep-penetration acoustic volumetric printing. Science 382, 1148–1155 (2023).
Bar-Zion, A. et al. Acoustically triggered mechanotherapy using genetically encoded gas vesicles. Nat. Nanotechnol. 16, 1403–1412 (2021).
Maresca, D. et al. Biomolecular ultrasound and sonogenetics. Annu. Rev. Chem. Biomol. Eng. 9, 229–252 (2018).
Hurt, R. C. et al. Genomically mined acoustic reporter genes for real-time in vivo monitoring of tumors and tumor-homing bacteria. Nat. Biotechnol. 41, 919–931 (2023).
Heiles, B. et al. Nonlinear sound-sheet microscopy: imaging opaque organs at the capillary and cellular scale. Science 388, eads1325 (2025).
Cadoni, S. et al. Ectopic expression of a mechanosensitive channel confers spatiotemporal resolution to ultrasound stimulations of neurons for visual restoration. Nat. Nanotechnol. 18, 667–676 (2023).
Yang, F. et al. Palette of rechargeable mechanoluminescent fluids produced by a biomineral-inspired suppressed dissolution approach. J. Am. Chem. Soc. 144, 18406–18418 (2022).
Wang, W. et al. H-bonded organic frameworks as ultrasound-programmable delivery platform. Nature 638, 401–410 (2025).
Kim, T. et al. Deep brain stimulation by blood–brain-barrier-crossing piezoelectric nanoparticles generating current and nitric oxide under focused ultrasound. Nat. Biomed. Eng. 7, 149–163 (2023).
Banishev, A. A. & Banishev, A. F. B. Photoluminescence features and mechanoluminescence mechanism inherent in composite materials based on a photopolymerizing resin and finely dispersed powders of SrAl2O4:(Eu2+,Dy3+) and Sr4Al14O25:(Eu2+,Dy3+,B) luminophores. Inorg. Mater. Appl. Res. 9, 484–489 (2018).
Kalita, J. M. & Chithambo, M. L. Probing the electron trap-depth distribution in Sr4Al14O25:Eu2+,Dy3+. J. Lumin. 265, 120245 (2024).
Stride, E. & Coussios, C. Nucleation, mapping and control of cavitation for drug delivery. Nat. Rev. Phys. 1, 495–509 (2019).
Klapoetke, N. C. et al. Independent optical excitation of distinct neural populations. Nat. Methods 11, 338–346 (2014).
Zhou, X. X., Fan, L. Z., Li, P., Shen, K. & Lin, M. Z. Optical control of cell signaling by single-chain photoswitchable kinases. Science 355, 836–842 (2017).
Xiong, B. et al. Precise cerebral vascular atlas in stereotaxic coordinates of whole mouse brain. Front. Neuroanat. 11, 128 (2017).
Jiang, S. et al. Spatially expandable fiber-based probes as a multifunctional deep brain interface. Nat. Commun. 11, 6115 (2020).
Yoo, S., Mittelstein, D. R., Hurt, R. C., Lacroix, J. & Shapiro, M. G. Focused ultrasound excites cortical neurons via mechanosensitive calcium accumulation and ion channel amplification. Nat. Commun. 13, 493 (2022).
Owen, S. F., Liu, M. H. & Kreitzer, A. C. Thermal constraints on in vivo optogenetic manipulations. Nat. Neurosci. 22, 1061–1065 (2019).
Sato, T., Shapiro, M. G. & Tsao, D. Y. Ultrasonic neuromodulation causes widespread cortical activation via an indirect auditory mechanism. Neuron 98, 1031–1041.e5 (2018).
Kubanek, J. et al. Ultrasound modulates ion channel currents. Sci. Rep. 6, 24170 (2016).
Sheng, M. & Greenberg, M. E. The regulation and function of c-fos and other immediate early genes in the nervous system. Neuron 4, 477–485 (1990).
Stujenske, J. M., Spellman, T. & Gordon, J. A. Modeling the spatiotemporal dynamics of light and heat propagation for in vivo optogenetics. Cell Rep. 12, 525–534 (2015).
Qian, H., Sheetz, M. P. & Elson, E. L. Single particle tracking. Analysis of diffusion and flow in two-dimensional systems. Biophys. J. 60, 910–921 (1991).
Kravitz, A. V. et al. Regulation of parkinsonian motor behaviours by optogenetic control of basal ganglia circuitry. Nature 466, 622–626 (2010).
Grimm, C. et al. Optogenetic activation of striatal D1R and D2R cells differentially engages downstream connected areas beyond the basal ganglia. Cell Rep. 37, 110161 (2021).
Chan, K. Y. et al. Engineered AAVs for efficient noninvasive gene delivery to the central and peripheral nervous systems. Nat. Neurosci. 20, 1172–1179 (2017).
Madisen, L. et al. A toolbox of Cre-dependent optogenetic transgenic mice for light-induced activation and silencing. Nat. Neurosci. 15, 793–802 (2012).
Lu, J. et al. Alcohol intake enhances glutamatergic transmission from D2 receptor-expressing afferents onto D1 receptor-expressing medium spiny neurons in the dorsomedial striatum. Neuropsychopharmacology 44, 1123–1131 (2019).
Cui, G. et al. Concurrent activation of striatal direct and indirect pathways during action initiation. Nature 494, 238–242 (2013).
Varin, C., Cornil, A., Houtteman, D., Bonnavion, P. & de Kerchove d’Exaerde, A. The respective activation and silencing of striatal direct and indirect pathway neurons support behavior encoding. Nat. Commun. 14, 4982 (2023).
Zhou, X. X. et al. A single-chain photoswitchable CRISPR-Cas9 architecture for light-inducible gene editing and transcription. ACS Chem. Biol. 13, 443–448 (2018).
Pulgarin, D. V. et al. Light-induced expression of gRNA allows for optogenetic gene editing of T lymphocytes in vivo. Nucleic Acids Res. 53, gkaf213 (2025).
Li, X., Lovell, J. F., Yoon, J. & Chen, X. Clinical development and potential of photothermal and photodynamic therapies for cancer. Nat. Rev. Clin. Oncol. 17, 657–674 (2020).
Obaid, G. et al. Engineering photodynamics for treatment, priming and imaging. Nat. Rev. Bioeng 2, 752–769 (2024).
Xu, C. et al. Nanoparticles with ultrasound-induced afterglow luminescence for tumour-specific theranostics. Nat. Biomed. Eng. 7, 298–312 (2023).
Menozzi, L. & Yao, J. Deep tissue photoacoustic imaging with light and sound. npj Imaging 2, 44 (2024).
Linsley, C. S. & Wu, B. M. Recent advances in light-responsive on-demand drug-delivery systems. Ther. Deliv. 8, 89–107 (2017).
Tsai, M.-F. et al. Near-infrared light-triggered drug release from ultraviolet- and redox-responsive polymersome encapsulated with core–shell upconversion nanoparticles for cancer therapy. ACS Appl. Bio Mater. 4, 3264–3275 (2021).
Jiang, S., Wu, X., Yang, F., Rommelfanger, N. J. & Hong, G. Activation of mechanoluminescent nanotransducers by focused ultrasound enables light delivery to deep-seated tissue in vivo. Nat. Protoc. 18, 3787–3820 (2023).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences (Routledge, 2013).
ShanJiang. ShanJiang1233/Jiang_2025: v1.0. Zenodo https://doi.org/10.5281/zenodo.18609351 (2026).
Acknowledgements
We thank A. Deniz Guler for donation of D1–cre mice and the University of Virginia School of Medicine Research Histology Core Facility for help with the preparation of histologic specimens. Part of the confocal microscopy imaging was performed at the Stanford Wu Tsai Neuroscience Microscopy Service. G.H. acknowledges three awards by NIH (5R00AG056636-04, 1R34NS127103-01 and R01NS126076-01), a National Science Foundation (NSF) CAREER Award (2045120), an NSF EAGER Award (2217582), a Rita Allen Foundation Scholars Award, a Beckman Technology Development Grant, a grant from the Focused Ultrasound Foundation, a gift from the Spinal Muscular Atrophy Foundation, a gift from the Pinetops Foundation, two seed grants from the Wu Tsai Neurosciences Institute, two seed grants from the Bio-X Initiative of Stanford University and a Synthetic Neurobiology Grant of Stanford University. H.S. acknowledges four awards by NIH (R01AG072430, R56 AG077720, R01AG085359 and R01NS123069). S.J. acknowledges support by the BRAIN Postdoctoral Fellowship from the University of Virginia. M.G.M. and N.J.R. acknowledge support by Bio-X Graduate Student Fellowships. M.G.M. and N.J.R. acknowledge support by the NSF Graduate Research Fellowships Program (award 1656518). X.W. acknowledges support by a Stanford Graduate Fellowship. X.C. acknowledges four awards by NIH (R01NS129834, R01DA059602, R01MH116904 and R01DA045664). Some illustrations were created with BioRender.com.
Author information
Authors and Affiliations
Contributions
S.J., X.C., J.D., H.S. and G.H. conceived and designed the project; S.J., M.G.M. and F.Y. synthesized MLNTs; F.Y. and H.C. performed the structure and morphology characterizations; S.J., F.Y. and N.J.R. performed the optical characterizations; S.J., M.G.M. and N.J.R. performed the pressure mapping; S.J. and X.W. performed imaging of the mechanoluminescence emission from the mouse; S.J. and M.G.M performed in vivo fibre photometry; S.J. performed the electrophysiological recording; S.J. performed the immunostaining and confocal microscopy imaging; S.J. and S.Z. performed the evaluation of recharging efficiency; Y.Z. and Q.Z. fabricated and validated the wearable transducer; S.J. designed the head-mounted system; S.J. and S.S.H. performed the behaviour assays; S.J., M.G.M., S.S.H. and L.C. performed the biocompatibility studies. S.J., F.Y., L.C., H.S. and G.H. analysed the data. All authors contributed to the writing of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Huiliang Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Pressure mapping of the FUS transducers operating at different frequencies.
a-d, Acoustic pressure fields in both the lateral (x–y plane at z0 = 0, i) and axial (y–z plane, at x0 = 0, ii) planes for a 0.65-MHz transducer (a), 1.5-MHz transducer (b), 3.3-MHz transducer (c), and 5.7-MHz transducer (d). In each panel, subpanels iii and iv display the corresponding spatial pressure profiles across the dashed lines in i and ii, respectively. FWHM was used to quantify the lateral resolution, while the depth of focus (DOF), defined as the −6 dB pressure width, was used to quantify the axial resolution.
Extended Data Fig. 2 Validating the efficacy of the ultrasound-scanning in vivo light source with c-Fos immunostaining.
a, Schematic illustration of the FUS-mediated light source in the M1 region. b, Representative immunostaining images of ChR2-YFP and c-Fos in the M1 region under different experimental conditions. c, Statistical analysis of the c-Fos cell density in the M1 region across different experimental groups. d, Schematic illustration of the FUS-mediated light source in the vDG. e, Representative immunostaining images of ChR2-YFP and c-Fos in the vDG under different experimental conditions. f, Statistical analysis of the c-Fos cell density in the vDG across different experimental groups. Scale bars represent 40 µm in b and 80 µm in e. All data are presented as mean ± s.d. with data points shown for n = 3 mice in each group. Statistical significance and P values are determined by ordinary one-way ANOVA: **P < 0.01, ****P < 0.0001.
Extended Data Fig. 3 Biocompatibility assessment of the ultrasound-scanning light source.
a, e, Representative immunostaining images of the M1 region 1 week (a) and 4 weeks (e) post-procedure under different experimental conditions. b, f, Statistical analysis of neuronal density 1 week (b) and 4 weeks (f) post-procedure. c, g, Statistical analysis of GFAP area 1 week (c) and 4 weeks (g) post-procedure. d, h, Statistical analysis of Iba1 area 1 week (d) and 4 weeks (h) post-procedure. All scale bars represent 50 µm. All data are presented as mean ± s.d. with data points shown for each animal from n = 4 mice in each group. Statistical significance and P values are determined by ordinary one-way ANOVA: P ≥ 0.05 (n.s.).
Supplementary information
Supplementary Information (download PDF )
Supplementary Video Legends 1–4, Note 1, Figs. 1–31, Table 1 and refs. 1–5.
Supplementary Video 1 (download AVI )
Dynamic ultrasound-mediated photostimulation of the left striatum in freely moving D1–Cre::ChR2–YFP mouse.
Supplementary Video2 (download AVI )
Dynamic ultrasound-mediated photostimulation of the right striatum in freely moving D1–Cre::ChR2–YFP mouse.
Supplementary Video3 (download AVI )
Dynamic ultrasound-mediated photostimulation of the left striatum in freely moving A2a–Cre::ChR2–YFP mouse.
Supplementary Video4 (download AVI )
Dynamic ultrasound-mediated photostimulation of the right striatum in freely moving A2a–Cre::ChR2–YFP mouse.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiang, S., Malinao, M.G., Yang, F. et al. An ultrasound-scanning in vivo light source. Nat. Mater. (2026). https://doi.org/10.1038/s41563-026-02556-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41563-026-02556-z