Abstract
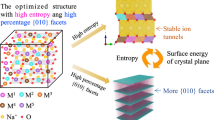
Gases are often by-products of battery materials during cell formation and degradation, affecting the cycle life and safety of rechargeable batteries. However, understanding gas-mediated (electro)-chemical reactions and nanoscale structural transformations during the synthesis of battery electrode materials remains challenging because of the lack of suitable characterization routes and the complexity of the interplay between thermodynamics and kinetics. Here we use operando synchrotron X-ray diffraction, in situ transmission X-ray microscopy and multiscale modelling to elucidate the reaction pathways and microstructural defect development of earth-abundant Mn-rich layered oxides as positive electrode materials for sodium-based batteries. In particular, we demonstrate the dominant role of CO2 over O2 and H2O(g) in modulating the competition between entropy and enthalpy during solid-state synthesis. Using Ni0.25Mn0.75CO3 as a model precursor, we reveal that CO2 generation favours the formation of entropy-driven metastable intermediates, suppresses closed pore/nanovoids formation and decreases chemical heterogeneity and residual lattice strain of Mn-rich layered oxides during the synthesis. This result motivates a fast-sintering strategy to promote CO2 release, which ultimately leads to improved chemo-mechanical and electrochemical stability of the Mn-rich positive electrodes when tested in non-aqueous Na metal coin cells.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data are available in the main text or the Supplementary Information.
References
Kumar, A. et al. Solid-state reaction synthesis of nanoscale materials: strategies and applications. Chem. Rev. 122, 12748–12863 (2022).
Kohlmann, H. Looking into the black box of solid-state synthesis. Eur. J. Inorg. Chem. 2019, 4174–4180 (2019).
de Nijs, B. et al. Entropy-driven formation of large icosahedral colloidal clusters by spherical confinement. Nat. Mater. 14, 56–60 (2015).
Fu, F. et al. Entropy and crystal-facet modulation of P2-type layered cathodes for long-lasting sodium-based batteries. Nat. Commun. 13, 2826 (2022).
Fermi, E. Thermodynamics. Reprint edn (Dover Publications, 2012).
Tajima, Y., Matsuo, T. & Suga, H. Phase transition in KOH-doped hexagonal ice. Nature 299, 810–812 (1982).
Jiang, H.-C., Wang, Z. & Balents, L. Identifying topological order by entanglement entropy. Nat. Phys. 8, 902–905 (2012).
Wang, B. et al. General synthesis of high-entropy alloy and ceramic nanoparticles in nanoseconds. Nat. Synth. 1, 138–146 (2022).
Rao, P. et al. Movable type printing method to synthesize high-entropy single-atom catalysts. Nat. Commun. 13, 5071 (2022).
Rost, C. M. et al. Entropy-stabilized oxides. Nat. Commun. 6, 8485 (2015).
Chamorro, J. R. & McQueen, T. M. Progress toward solid state synthesis by design. Acc. Chem. Res. 51, 2918–2925 (2018).
Bianchini, M. et al. The interplay between thermodynamics and kinetics in the solid-state synthesis of layered oxides. Nat. Mater. 19, 1088–1095 (2020).
Bai, J. et al. Kinetic pathways templated by low-temperature intermediates during solid-state synthesis of layered oxides. Chem. Mater. 32, 9906–9913 (2020).
Zuo, W. et al. Microstrain screening towards defect-less layered transition metal oxide cathodes. Nat. Nanotechnol. 19, 1644–1653 (2024).
Hu, E., Wang, X., Yu, X. & Yang, X. Q. Probing the complexities of structural changes in layered oxide cathode materials for Li-ion batteries during fast charge-discharge cycling and heating. Acc. Chem. Res. 51, 290–298 (2018).
Cui, Z. & Manthiram, A. Thermal stability and outgassing behaviors of high-nickel cathodes in lithium-ion batteries. Angew. Chem. Int. Ed. 62, e202307243 (2023).
Wu, B. et al. Unusual Li2O sublimation promotes single-crystal growth and sintering. Nat. Energy 10, 605–615 (2025).
Liu, X. et al. In situ observation of thermal-driven degradation and safety concerns of lithiated graphite anode. Nat. Commun. 12, 4235 (2021).
Park, H. et al. In situ multiscale probing of the synthesis of a Ni-rich layered oxide cathode reveals reaction heterogeneity driven by competing kinetic pathways. Nat. Chem. 14, 614–622 (2022).
Wang, D. et al. Synthetic control of kinetic reaction pathway and cationic ordering in high-Ni layered oxide cathodes. Adv. Mater. 29, 1606715 (2017).
Yabuuchi, N., Kubota, K., Dahbi, M. & Komaba, S. Research development on sodium-ion batteries. Chem. Rev. 114, 11636–11682 (2014).
Ortiz-Vitoriano, N., Drewett, N. E., Gonzalo, E. & Rojo, T. High performance manganese-based layered oxide cathodes: overcoming the challenges of sodium ion batteries. Energy Environ. Sci. 10, 1051–1074 (2017).
Ma, X., Chen, H. & Ceder, G. Electrochemical properties of monoclinic NaMnO2. J. Electrochem. Soc. 158, A1307 (2011).
Billaud, J. et al. Beta-NaMnO2: a high-performance cathode for sodium-ion batteries. J. Am. Chem. Soc. 136, 17243–17248 (2014).
Kumakura, S., Tahara, Y., Kubota, K., Chihara, K. & Komaba, S. Sodium and manganese stoichiometry of P2-type Na2/3MnO2. Angew. Chem. Int. Ed. 55, 12760–12763 (2016).
Liu, X. et al. P2-Na0.67AlxMn1−xO2: cost-effective, stable and high-rate sodium electrodes by suppressing phase transitions and enhancing Na+ mobility. Angew. Chem. Int. Ed. 58, 18086–18095 (2019).
Zuo, W. et al. Engineering Na+-layer spacings to stabilize Mn-based layered cathodes for sodium-ion batteries. Nat. Commun. 12, 4903 (2021).
Doeff, M. M., Richardson, T. J., Hollingsworth, J., Yuan, C.-W. & Gonzales, M. Synthesis and characterization of a copper-substituted manganese oxide with the Na0.44MnO2 structure. J. Power Sources 112, 294–297 (2002).
Li, X. L. et al. Boosting reversibility of Mn-based tunnel-structured cathode materials for sodium-ion batteries by magnesium substitution. Adv. Sci. 8, 2004448 (2021).
Zuo, W. & Yang, Y. Synthesis, structure, electrochemical mechanisms, and atmospheric stability of Mn-based layered oxide cathodes for sodium ion batteries. Acc. Mater. Res. 3, 709–720 (2022).
Liu, X., Fechler, N. & Antonietti, M. Salt melt synthesis of ceramics, semiconductors and carbon nanostructures. Chem. Soc. Rev. 42, 8237–8265 (2013).
Kaczmarek, A. M., Van Hecke, K. & Van Deun, R. Nano- and micro-sized rare-earth carbonates and their use as precursors and sacrificial templates for the synthesis of new innovative materials. Chem. Soc. Rev. 44, 2032–2059 (2015).
Zhang, J., Guo, X., Yao, S., Zhu, W. & Qiu, X. Tailored synthesis of Ni0.25Mn0.75CO3 spherical precursors for high capacity Li-rich cathode materials via a urea-based precipitation method. J. Power Sources 238, 245–250 (2013).
Wang, D., Belharouak, I., Koenig, G. M., Zhou, G. & Amine, K. Growth mechanism of Ni0.3Mn0.7CO3 precursor for high capacity Li-ion battery cathodes. J. Mater. Chem. 21, 9290 (2011).
Bleif, H. J. & Dachs, H. Cystalline modifications and structural phase transitions of NaOH and NaOD. Acta Crystallogr. A 38, 470–476 (1982).
Zhao, C. et al. Rational design of layered oxide materials for sodium-ion batteries. Science 370, 708–711 (2020).
Xiao, X., Xu, Z., Lin, F. & Lee, W. K. TXM-Sandbox: an open-source software for transmission X-ray microscopy data analysis. J. Synchrotron Radiat. 29, 266–275 (2022).
Zuo, W. et al. The stability of P2-layered sodium transition metal oxides in ambient atmospheres. Nat. Commun. 11, 3544 (2020).
Yang, Y. et al. Decoupling the air sensitivity of Na-layered oxides. Science 385, 744–752 (2024).
Somerville, J. W. et al. Nature of the ‘Z’-phase in layered Na-ion battery cathodes. Energy Environ. Sci. 12, 2223–2232 (2019).
Tang, Y. et al. Sustainable layered cathode with suppressed phase transition for long-life sodium-ion batteries. Nat. Sustain. 7, 348–359 (2024).
Lu, Z. & Dahn, J. R. In situ X-ray diffraction study of P2-Na2/3Ni1/3Mn2/3O2. J. Electrochem. Soc. 148, A1225 (2001).
Frith, J. T., Lacey, M. J. & Ulissi, U. A non-academic perspective on the future of lithium-based batteries. Nat. Commun. 14, 420 (2023).
Scurtu, R. G. et al. From small batteries to big claims. Nat. Nanotechnol. https://doi.org/10.1038/s41565-025-01906-3 (2025).
Momma, K. & Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Cryst. 44, 1272–1276 (2011).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 54, 11169 (1996).
Perdew, J. P. B. K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865 (1996).
Hammer, B. H. L. B. & Nørskov, J. K. Improved adsorption energetics within density-functional theory using revised Perdew-Burke-Ernzerhof functionals. Phys. Rev. B 59, 7413 (1999).
Blochl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Hong, J. et al. Metal-oxygen decoordination stabilizes anion redox in Li-rich oxides. Nat. Mater. 18, 256–265 (2019).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976).
Pack, J. D. & Monkhorst, H. J. ‘Special points for Brillouin-zone integrations’—a reply. Phys. Rev. B 16, 1748–1749 (1977).
Togo, A., Oba, F. & Tanaka, I. First-principles calculations of the ferroelastic transition between rutile-type and CaCl2-type SiO2 at high pressures. Phys. Rev. B 78, 134106 (2008).
Wang, V., Xu, N., Liu, J.-C., Tang, G. & Geng, W.-T. VASPKIT: a user-friendly interface facilitating high-throughput computing and analysis using VASP code. Comput. Phys. Commun. 267, 108033 (2021).
Chen, K. et al. Cobalt-free composite-structured cathodes with lithium-stoichiometry control for sustainable lithium-ion batteries. Nat. Commun. 15, 430 (2024).
Tayal, A. et al. In situ insights into cathode calcination for predictive synthesis: kinetic crystallization of LiNiO2 from hydroxides. Adv. Mater. 36, e2312027 (2024).
Wolfman, M. et al. The importance of surface oxygen for lithiation and morphology evolution during calcination of high-nickel NMC cathodes. Adv. Energy Mater. 12, 2102951 (2022).
Acknowledgements
The work conducted at Argonne National Laboratory was supported by the Office of Vehicle Technologies of the US Department of Energy through the Advanced Battery Materials Research (BMR) Program, including the LENS Consortium under contract number DE-AC02-06CH11357. Use of the Advanced Photon Source (APS, 17-BM and 12-BM) and the Centre for Nanoscale Materials, both Office of Science user facilities, was supported by the US Department of Energy, Office of Science and Office of Basic Energy Sciences, under contract number DE-AC02-06CH11357. This research used resources of the Argonne Leadership Computing Facility, a US Department of Energy (DOE) Office of Science user facility at Argonne National Laboratory and is based on research supported by the US DOE Office of Science-Advanced Scientific Computing Research Program, under contract number DE-AC02-06CH11357. This research used beamline 18-ID, 8-ID and 3-ID of the National Synchrotron Light Source II, a US DOE Office of Science user facility operated for the DOE Office of Science by Brookhaven National Laboratory under contract number DE-SC0012704. We thank N. Sunariwai and beamline 8-2 at Stanford Synchrotron Radiation Light source (SSRL) for the metal L-edge data collection. Use of the SSRL, SLAC National Accelerator Laboratory, was supported by the US Department of Energy, Office of Science, Office of Basic Energy Sciences under contract number DE-AC02-76SF00515.
Author information
Authors and Affiliations
Contributions
G.-L.X., W.Z. and K.A. conceived the idea and designed the experiments. G.-L.X. and K.A. initiated and supervised the project. W.Z. synthesized the positive electrode material. W.Z., X.J. and G.-L.X. carried out the lab characterizations, electrochemical tests and corresponding data analysis. F.R. carried out the DFT calculations. D.H. and Y.L. contributed to the STEM measurements. W.Z. and T.L. carried out the in situ and ex situ SXRD measurements and analysis with the assistance of W.X. W.Z., S.Z., X.J., D.H., G.W. and Y.L. contributed to the FIB-SEM, SEM and EDS experiment. W.Z. and X.X. conducted the in situ and ex situ TXM-XANES measurements and dataset analysis. W.Z. and Z.Y. conducted the ion beam experiment. W.Z. and Y.Q. conducted the TGA experiments and analysis. P.B. conducted the computational modelling of crystal growth. W.Z. and H.Y. conducted and analysed the XRF and nano-diffraction analysis at HXN beamline. W.Z. and G.-L.X. wrote the paper with input from all authors. All authors, including Y.S.C., Y.Y. and V.S., discussed the results and co-wrote and commented on the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Nanotechnology thanks Bernt Johannessen, Sunil Kumar and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Schematic illustration of a synthetic process and its influential factors.
Synthetic chemistry is a black box that connects precursors and products, governed by three critical parameters: thermodynamics, kinetics, and mechanics. Thermodynamics dictates the direction and spontaneity of chemical reactions, with enthalpy and entropy as the key parameters, directly or indirectly influenced by various synthetic conditions. For instance, enthalpy is primarily affected by the atmosphere, pressure, and phase state, while entropy is largely influenced by phase transitions, temperature, and heating/cooling rates. Kinetics controls the rate of reactions and can influence the reaction pathways, potentially leading to the formation of non-equilibrium metastable products, particularly when competing pathways with similar free energy values exist. Though mechanics does not directly determine the reaction direction or rate, it reflects the microstructures and defects of the product, which directly influence material properties such as mechanical strength, conductivity, and thermal stability. Understanding and controlling these three parameters is essential to the rational design and synthesis of advanced inorganic materials, enabling the optimization of microstrain, grain boundaries, and other mechanical properties that are critical for various applications.
Extended Data Fig. 2 Structures of Ni0.25Mn0.75CO3 precursor.
a, b. SEM micrographs and c. cross-sectional SEM and corresponding EDS mapping of Ni0.25Mn0.75CO3 precursor. (d) Powder XRD pattern of Ni0.25Mn0.75CO3. e, f. Structural illustration of Ni0.25Mn0.75CO3 along the crystallographic a-axis € and c-axis (f).
Extended Data Fig. 3 Precursor structure and its evolution during solid-state synthesis.
(a) In situ SXRD patterns and (b) corresponding structural evolutions of Ni0.25Mn0.75CO3-NaOH mixture during solid-state calcination. The wavelength for the in situ heating SXRD measurement is 0.24108 Å. SXRD patterns acquired under continuous heating with no holding at fixed temperatures.
Extended Data Fig. 4 Structural and chemical evolution of Ni0.25Mn0.75CO3-NaOH mixtures.
In situ heating SXRD of Ni0.25Mn0.75CO3-NaOH mixtures with the Na-Tm ratios of (a) 1.00 (λ = 0.24108 Å) and (b) 1.30 (λ = 0.45200 Å), which were calcined from 26.0 °C to 750 °C with heating ramp rate of 6 °C min−1.
Extended Data Fig. 5 Decoupling the contribution of thermodynamic parameters to synthetic reactions.
a. Reaction energy per formula unit of reaction 11 (Eq. 11 in the main text) at various temperatures. b. Entropy contribution to the Path 3 reaction 4 (Eq. 4 in the main text). c. Reaction energy for varying Na contents in O-type NaxNi0.25Mn0.75O2 at different temperatures. Increasing the Na content up to x = 0.5 enhances the thermodynamic favorability of forming layered oxides, whereas further increases lead to more positive reaction energies. The highest thermodynamically stable Na content in layered NaxNi0.25Mn0.75O2 is approximately 0.8. Excess Na in the precursor does not fully incorporate into the layered oxides. However, according to Le Chatelier principle, an increased Na-TM ratio in the precursor mixtures favor the direct transformation from carbonate to sodium layered oxides (path 2). This consideration aligns well with our operando SXRD findings.
Extended Data Fig. 6 Compact and clustered spheroid P2-Na0.67Ni0.25Mn0.75O2 particles.
(a) SEM, (b) cross-section SEM, and (c) STEM results of compact spheroid P2-Na0.67Ni0.25Mn0.75O2 particle; (d) SEM, (e) cross-section SEM, and (f) STEM results of porous clustered spheroid P2-Na0.67Ni0.25Mn0.75O2 particle.
Extended Data Fig. 7 Synchrotron powder XRD patterns of P2-type Na0.67Ni0.25Mn0.75O2 that were calcined at 950 °C for 20 h with different heating ramp rates of 0.5 °C min−1 (a), 1 °C min−1 (b), 5 °C min−1 (c), 10 °C min−1 (d), 20 °C min−1 (e). Wavelength = 0.45961 Å.
We systematically investigated the structural evolution of P2-type samples synthesized under various heating ramp rates by SXRD coupled with Rietveld refinement. All samples crystallize in a single-phase P2-type structure without detectable secondary phases or impurities. The refinement results reveal that lower ramping rates lead to higher microstrain values, indicating increased internal lattice distortion and defect concentration. Despite variations in thermal treatment, the refined lattice parameters (a ≈ 2.87 Å, c ≈ 11.18–11.19 Å) remain nearly constant across all conditions, suggesting comparable Na incorporation during calcination. These observations highlight that the ramping rate predominantly modulates microstructural uniformity rather than altering the final phase or stoichiometry.
Extended Data Fig. 8 Morphological evolution at slow heating ramping rate revealed by TXM-XANES.
This figure shows a more comprehensive morphological change at a slow heating ramp rate, as revealed by in situ heating TXM-XANES results. Rows depict different viewing planes (YZ, XZ, XY) and imaging slices (2D slice) at various temperatures ranging from 26.0 °C to 950 °C. Slow ramp heating reveals progressive pore development starting at 300 °C, and a highly interconnected pore network emerges by 750 °C.
Extended Data Fig. 9 Theoretical modeling of the impact of different reaction pathways on crystal growth.
Here, Path 2 is an enthalpy-driven path, corresponding to reaction 3 in the main text. Reaction 5 in the main text was leveraging to model the entropy-driven path (Path 3). a, b. Solid fraction (a) and specific surface area (b) evolutions based on Path 2 and Path 3. c, d. Microstructure of primary particles before and after calcination under Path 2 (c) and Path 3 (d).
Extended Data Fig. 10 Air-stability of P2-Na0.67Ni0.25Mn0.75O2 synthesized at different ramping rates.
(a) SXRD patterns of pristine and air-exposed samples (3 days in a constant-temperature oven at 30 °C, relative humidity (RH) = 68-74%) synthesized with different heating ramp rates. All samples retain the characteristic P2-phase reflections (marked by ∇). (b) Enlarged view of the 2θ = 8–19° region highlighting the emergence of new reflections indexed to NaHCO3 (red ticks) after air exposure, while no secondary phases such as NiO or Mn3O4 are detected. The appearance of NaHCO3 indicates limited surface reactions involving Na loss and carbonation under ambient moisture CO2, whereas the transition metal oxide framework remains intact. These results suggest that the P2-phase material exhibits good bulk air stability across various synthesis conditions. To evaluate surface degradation upon air exposure, we attempted Rietveld refinement of the NaHCO3 phase using synchrotron XRD patterns. The results indicate that the relative abundance of NaHCO3 is less than 2% compared to the dominant P2-Na0.67Ni0.25Mn0.75O2 phase, rendering precise quantitative refinement unreliable. Furthermore, the diffraction peak intensities of NaHCO3 show no appreciable difference among these three samples, suggesting comparable levels of surface reaction. This implies that the air stability of the three materials is essentially similar, despite distinct differences in particle morphology. Such stability can be attributed to two factors: first, the intrinsic structural robustness of P2-type Na0.67Ni0.25Mn0.75O2, which limits surface degradation regardless of morphological variation; and second, although samples synthesized under slower ramping rates exhibit fewer open pores, abundant nanoscale intergranular voids between primary particles still permit the diffusion of H2O and CO2 into the particle interior, allowing limited Na loss and surface carbonation.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–13, Tables 1–5 and Notes 1–6.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zuo, W., Ren, F., Barai, P. et al. Gas-mediated defect engineering in earth-abundant Mn-rich layered oxides for non-aqueous sodium-based batteries. Nat. Nanotechnol. 20, 1667–1677 (2025). https://doi.org/10.1038/s41565-025-01998-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41565-025-01998-x
This article is cited by
-
Advances in battery technologies for smart grids in 2025
Nature Reviews Clean Technology (2026)
-
Entropy-regulated solid-state synthesis of Na-based Mn-rich layered oxides
Nature Nanotechnology (2025)