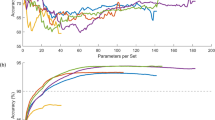
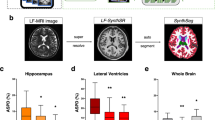
Abstract
With elderly populations increasing in many countries, rates of Alzheimer disease and related dementias (ADRD) are expected to rise worldwide in the coming years. Low-income and middle-income countries, where barriers to health care are most pronounced and research representation is limited, are predicted to experience the greatest increases in ADRD prevalence. Access to advanced diagnostic and research tools, such as neuroimaging, is severely restricted in these regions, but low-field MRI is emerging as a promising, accessible alternative to conventional imaging. By reducing infrastructure, cost and siting requirements, low-field MRI offers a potential pathway to expand access to dementia-relevant imaging beyond specialized centres. In this article, we summarize key structural imaging biomarkers in ADRD and review the current literature supporting the use of low-field MRI in the ADRD field. We highlight the utility of low-field MRI for the assessment of regional atrophy and cerebrovascular lesion burden and discuss emerging diffusion-based markers. We also consider challenges and future directions, offering insights to advance equitable access to diagnostic imaging, guide research priorities and support global implementation of low-field MRI in ADRD care and investigation.
Key points
-
The rapid rise in dementia prevalence in low-income and middle-income countries, where advanced neuroimaging access is scarce, highlights the urgent need for low-field MRI to close diagnostic and research gaps.
-
Studies demonstrate reliable detection of core Alzheimer disease markers, including hippocampal atrophy and white matter hyperintensities, on portable low-field systems, with promising correspondence to conventional high-field MRI findings.
-
Despite low resolution and signal-to-noise ratios, innovations such as segmentation algorithms and super-resolution methods extend the capability of low-field MRI, although validation across diverse populations is still required.
-
Low-field MRI offers transformative potential for global dementia research and care, but existing barriers must be addressed to enable scalable, sustainable worldwide implementation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Alladi, S. & Hachinski, V. World dementia: one approach does not fit all. Neurology 91, 264–270 (2018).
Mayeda, E. R., Glymour, M. M., Quesenberry, C. P. & Whitmer, R. A. Inequalities in dementia incidence between six racial and ethnic groups over 14 years. Alzheimers Dement. 12, 216–224 (2016).
Pham, T. M. et al. Trends in dementia diagnosis rates in UK ethnic groups: analysis of UK primary care data. Clin. Epidemiol. 10, 949–960 (2018).
Nichols, E. et al. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health 7, e105–e125 (2022).
Livingston, G. et al. Dementia prevention, intervention, and care: 2024 report of the Lancet standing Commission. Lancet 404, 572–628 (2024).
Fleming, K. A. et al. The Lancet Commission on diagnostics: transforming access to diagnostics. Lancet 398, 1997–2050 (2021).
Howitt, P. et al. Technologies for global health. Lancet 380, 507–535 (2012).
McGlinchey, E. et al. Biomarkers of neurodegeneration across the Global South. Lancet Healthy Longev. 5, 100616 (2024).
Paradela, R. S. et al. Population attributable fractions for risk factors for dementia in seven Latin American countries: an analysis using cross-sectional survey data. Lancet Glob. Health 12, e1600–e1610 (2024).
Frisoni, G. B., Fox, N. C., Jack, C. R. Jr, Scheltens, P. & Thompson, P. M. The clinical use of structural MRI in Alzheimer disease. Nat. Rev. Neurol. 6, 67–77 (2010).
Risacher, S. L. & Apostolova, L. G. Neuroimaging in dementia. Continuum 29, 219–254 (2023).
Wankhede, N. L. et al. Neuroimaging biomarkers: a critical review of their role in neurological disease diagnosis and management. Brain Disord. 18, 100222 (2025).
Haller, S., Jäger, H. R., Vernooij, M. W. & Barkhof, F. Neuroimaging in dementia: more than typical Alzheimer disease. Radiology 308, e230173 (2023).
Sims, J. R. et al. Donanemab in early symptomatic Alzheimer disease: the TRAILBLAZER-ALZ 2 randomized clinical trial. JAMA 330, 512–527 (2023).
Van Dyck, C. H. et al. Lecanemab in early Alzheimer’s disease. N. Engl. J. Med. 388, 9–21 (2023).
Sperling, R. et al. Amyloid-related imaging abnormalities in patients with Alzheimer’s disease treated with bapineuzumab: a retrospective analysis. Lancet Neurol. 11, 241–249 (2012).
Belder, C. R., Schott, J. M. & Fox, N. C. Preparing for disease-modifying therapies in Alzheimer’s disease. Lancet Neurol. 22, 782–783 (2023).
Akinyemi, R. O., Owolabi, M. O., Okubadejo, N., Ogunniyi, A. & Kalaria, R. N. The African dementia consortium. Lancet Neurol. 22, 28–29 (2023).
Ibanez, A. et al. The multi-partner consortium to expand dementia research in Latin America (ReDLat): driving multicentric research and implementation science. Front. Neurol. 12, 631722 (2021).
Udeh-Momoh, C. T. et al. A multi-national collaboration to assess the feasibility and sustainability of implementing multimodal brain health promotion strategies in Sub-Saharan Africa (the AFRICA-FINGERS project). Alzheimers Dement. 19, e079009 (2023).
Feigin, V. L. et al. Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 18, 459–480 (2019).
Anazodo, U. C., Adewole, M. & Dako, F. AI for population and global health in radiology. Radiol. Artif. Intell. 4, e220107 (2022).
Qin, C. et al. Sustainable low-field cardiovascular magnetic resonance in changing healthcare systems. Eur. Heart J. Cardiovasc. Imaging 23, e246–e260 (2022).
Wiese, L. A. K. et al. Global rural health disparities in Alzheimer’s disease and related dementias: state of the science. Alzheimers Dement. 19, 4204–4225 (2023).
World Health Organization. Global Atlas of Medical Devices 2022 https://www.who.int/publications/i/item/9789240062207 (WHO, 2022).
World Health Organization. Global Atlas of Medical Devices https://www.who.int/teams/health-product-policy-and-standards/assistive-and-medical-technology/medical-devices/global-atlas-of-medical-devices (WHO, 2017).
Jones, D. K. et al. Low field, high impact: democratizing MRI for clinical and research innovation. BJR Open 7, tzaf022 (2025).
Bhat, S. S. et al. Low-field MRI of stroke: challenges and opportunities. J. Magn. Reson. Imaging 54, 372–390 (2021).
Arnold, T. C., Freeman, C. W., Litt, B. & Stein, J. M. Low-field MRI: clinical promise and challenges. J. Magn. Reson. Imaging 57, 25–44 (2023).
Ogbole, G. I., Adeyomoye, A. O., Badu-Peprah, A., Mensah, Y. & Nzeh, D. A. Survey of magnetic resonance imaging availability in West Africa. Pan Afr. Med. J. 30, 240 (2018).
Jalloul, M. et al. MRI scarcity in low-and middle-income countries. NMR Biomed. 36, e5022 (2023).
Klein, H.-M. Clinical Low Field Strength Magnetic Resonance Imaging: A Practical Guide to Accessible MRI (Springer, 2015).
Malkin, R. & von Oldenburg Beer, K. Diffusion of novel healthcare technologies to resource poor settings. Ann. Biomed. Eng. 41, 1841–1850 (2013).
Perry, L. & Malkin, R. Effectiveness of medical equipment donations to improve health systems: how much medical equipment is broken in the developing world? Med. Biol. Eng. Comput. 49, 719–722 (2011).
Kimberly, W. T. et al. Brain imaging with portable low-field MRI. Nat. Rev. Bioeng. 1, 617–630 (2023).
Altaf, A. et al. Applications, limitations and advancements of ultra-low-field magnetic resonance imaging: a scoping review. Surg. Neurol. Int. 15, 218 (2024).
Marques, J. P., Simonis, F. F. & Webb, A. G. Low-field MRI: an MR physics perspective. J. Magn. Reson. Imaging 49, 1528–1542 (2019).
Nikpanah, M. et al. Low versus ultra-high field MRI: how to select your MRI fleet. Appl. Radiol. 52, 28–41 (2023).
Parasuram, N. R. et al. Future of neurology & technology: neuroimaging made accessible using low-field, portable MRI. Neurology 100, 1067–1071 (2023).
Kopanoglu, E. et al. MRI and implant safety at low-field and ultralow-field strengths. J. Magn. Reson. Imaging 63, 364–377 (2025).
O’Reilly, T., Teeuwisse, W. & Webb, A. Three-dimensional MRI in a homogenous 27 cm diameter bore Halbach array magnet. J. Magn. Reson. 307, 106578 (2019).
Liu, Y. et al. A low-cost and shielding-free ultra-low-field brain MRI scanner. Nat. Commun. 12, 7238 (2021).
Zhao, Y. et al. Whole-body magnetic resonance imaging at 0.05 Tesla. Science 384, eadm7168 (2024).
Obungoloch, J. et al. On-site construction of a point-of-care low-field MRI system in Africa. NMR Biomed. 36, e4917 (2023).
Padormo, F. et al. In vivo T1 mapping of neonatal brain tissue at 64 mT. Magn. Reson. Med. 89, 1016–1025 (2023).
Deoni, S. C. et al. Accessible pediatric neuroimaging using a low field strength MRI scanner. Neuroimage 238, 118273 (2021).
Abate, F. et al. UNITY: a low-field magnetic resonance neuroimaging initiative to characterize neurodevelopment in low and middle-income settings. Dev. Cogn. Neurosci. 69, 101397 (2024).
Arnold, T. C. et al. Sensitivity of portable low-field magnetic resonance imaging for multiple sclerosis lesions. Neuroimage Clin. 35, 103101 (2022).
Johnson, I. P. et al. Predicting white matter hyperintensity: leveraging portable MRI for accessible brain health screening. Am. J. Neuroradiol. 46, 1786–1792 (2025).
Okar, S. V. et al. High-field-blinded assessment of portable ultra-low-field brain MRI for multiple sclerosis. J. Neuroimaging 35, e70005 (2025).
Mazurek, M. H. et al. Portable, bedside, low-field magnetic resonance imaging for evaluation of intracerebral hemorrhage. Nat. Commun. 12, 5119 (2021).
Mazurek, M. H. et al. Detection of intracerebral hemorrhage using low-field, portable magnetic resonance imaging in patients with stroke. Stroke 54, 2832–2841 (2023).
Yuen, M. M. et al. Portable, low-field magnetic resonance imaging enables highly accessible and dynamic bedside evaluation of ischemic stroke. Sci. Adv. 8, eabm3952 (2022).
Sorby-Adams, A. J. et al. Portable, low-field magnetic resonance imaging for evaluation of Alzheimer’s disease. Nat. Commun. 15, 10488 (2024).
de Havenon, A. et al. Portable MRI for brain health: bridging clinical care and population surveillance. Preprint at SSRN https://ssrn.com/abstract=5681335 (2025).
Chetcuti, K. et al. Implementation of a low-field portable MRI scanner in a resource-constrained environment: our experience in Malawi. Am. J. Neuroradiol. 43, 670–674 (2022).
Cho, S.-M. et al. Assessing the SAfety and FEasibility of bedside portable low-field brain Magnetic Resonance Imaging in patients on ECMO (SAFE-MRI ECMO study): study protocol and first case series experience. Crit. Care 26, 119 (2022).
Turpin, J. et al. Portable magnetic resonance imaging for ICU patients. Crit. Care Explor. 2, e0306 (2020).
Hovis, G., Langdorf, M., Dang, E. & Chow, D. MRI at the bedside: a case report comparing fixed and portable magnetic resonance imaging for suspected stroke. Cureus 13, e16904 (2021).
Deoni, S. C. et al. Development of a mobile low-field MRI scanner. Sci. Rep. 12, 5690 (2022).
Deoni, S. C. et al. Neuroimaging and verbal memory assessment in healthy aging adults using a portable low-field MRI scanner and a web-based platform: results from a proof-of-concept population-based cross-section study. Brain Struct. Funct. 228, 493–509 (2023).
Webb, A. & O’Reilly, T. Tackling SNR at low-field: a review of hardware approaches for point-of-care systems. Magn. Reson. Mat. Phys. Biol. Med. 36, 375–393 (2023).
Schote, D. et al. Nexus: a versatile console for advanced low-field MRI. Magn. Reson. Med. 93, 2224–2238 (2025).
Gholam, J. et al. Diffusion tensor MRI and spherical-deconvolution-based tractography on an ultra-low field portable MRI system. Hum. Brain Mapp. 47, e70454 (2026).
Sorby-Adams, A. et al. Diffusion-weighted imaging fluid-attenuated inversion recovery mismatch on portable, low-field magnetic resonance imaging among acute stroke patients. Ann. Neurol. 96, 321–331 (2024).
Deoni, S. C., O’Muircheartaigh, J., Ljungberg, E., Huentelman, M. & Williams, S. C. Simultaneous high-resolution T2-weighted imaging and quantitative T 2 mapping at low magnetic field strengths using a multiple TE and multi-orientation acquisition approach. Magn. Reson. Med. 88, 1273–1281 (2022).
Sarracanie, M. & Salameh, N. Low-field MRI: how low can we go? A fresh view on an old debate. Front. Phys. 8, 172 (2020).
Islam, K. T. et al. Improving portable low-field MRI image quality through image-to-image translation using paired low-and high-field images. Sci. Rep. 13, 21183 (2023).
Lehtinen, J. et al. Noise2Noise: Learning image restoration without clean data. Preprint at https://doi.org/10.48550/arXiv.1803.04189 (2018).
Wu, Z. et al. Deep learning based MRI reconstruction with transformer. Comput. Methods Programs Biomed. 233, 107452 (2023).
Lin, H. et al. Low-field magnetic resonance image enhancement via stochastic image quality transfer. Med. Image Anal. 87, 102807 (2023).
Gopinath, K. et al. From low field to high value: robust cortical mapping from low-field MRI. Preprint at https://doi.org/10.48550/arXiv.2505.12228 (2025).
Man, C. et al. Deep learning enabled fast 3D brain MRI at 0.055 tesla. Sci. Adv. 9, eadi9327 (2023).
Ayde, R. et al. MRI at low field: a review of software solutions for improving SNR. NMR Biomed. 38, e5268 (2025).
Rao, B. N. & Reddy, D. L. S. Brain MRI noise reduction using convolutional autoencoder. In Artificial Intelligence and Data Science (ICAIDS 2021) (eds Kumar, A. et al.) https://doi.org/10.1007/978-3-031-21385-4_29 (Springer Nature, 2022).
Ji, Z. et al. Survey of hallucination in natural language generation. ACM Comput. Surv. 55, 1–38 (2023).
Harper, J. R. et al. Assessing the utility of low resolution brain imaging: treatment of infant hydrocephalus. Neuroimage Clin. 32, 102896 (2021).
Lepore, N. & Linguraru, M. G. Low Field Pediatric Brain Magnetic Resonance Image Segmentation and Quality Assurance: First MICCAI Challenge, LISA 2024, Held in Conjunction with MICCAI 2024, Marrakesh, Morocco, October 10, 2024, Proceedings (Springer Nature, 2025).
Billot, B. et al. SynthSeg: Segmentation of brain MRI scans of any contrast and resolution without retraining. Med. Image Anal. 86, 102789 (2023).
Váša, F. et al. Ultra-low-field brain MRI morphometry: test–retest reliability and correspondence to high-field MRI. Imaging Neurosci. 3, IMAG.a.930 (2025).
Laso, P. et al. Quantifying white matter hyperintensity and brain volumes in heterogeneous clinical and low-field portable MRI. Proc. IEEE Int. Symp. Biomed. Imaging https://doi.org/10.1109/isbi56570.2024.10635502 (2024).
Iglesias, J. E. et al. Quantitative brain morphometry of portable low-field-strength MRI using super-resolution machine learning. Radiology 306, e220522 (2022).
Oved, T. et al. Deep learning of personalized priors from past MRI scans enables fast, quality-enhanced point-of-care MRI with low-cost systems. Preprint at https://doi.org/10.48550/arXiv.2505.02470 (2025).
Evans, T. E. et al. Subregional volumes of the hippocampus in relation to cognitive function and risk of dementia. Neuroimage 178, 129–135 (2018).
Zotin, M. C. Z., Sveikata, L., Viswanathan, A. & Yilmaz, P. Cerebral small vessel disease and vascular cognitive impairment: from diagnosis to management. Curr. Opin. Neurol. 34, 246–257 (2021).
Mak, E. et al. Proximity to dementia onset and multi-modal neuroimaging changes: the prevent-dementia study. Neuroimage 229, 117749 (2021).
Tondelli, M. et al. Structural MRI changes detectable up to ten years before clinical Alzheimer’s disease. Neurobiol. Aging 33, 825.e825–825.e836 (2012).
Ayers, M. R., Svaldi, D. & Apostolova, L. G. Brain imaging in differential diagnosis of dementia. Pract. Neurol. https://practicalneurology.com/diseases-diagnoses/imaging-testing/brain-imaging-in-differential-diagnosis-of-dementia/31533/ (2019).
Vemuri, P. et al. Antemortem differential diagnosis of dementia pathology using structural MRI: differential-stand. Neuroimage 55, 522–531 (2011).
Chouliaras, L. & O’Brien, J. T. The use of neuroimaging techniques in the early and differential diagnosis of dementia. Mol. Psychiatry 28, 4084–4097 (2023).
Bakkour, A., Morris, J. C. & Dickerson, B. C. The cortical signature of prodromal AD: regional thinning predicts mild AD dementia. Neurology 72, 1048–1055 (2009).
Apostolova, L. G. et al. Three-dimensional gray matter atrophy mapping in mild cognitive impairment and mild Alzheimer disease. Arch. Neurol. 64, 1489–1495 (2007).
Singh, V. et al. Spatial patterns of cortical thinning in mild cognitive impairment and Alzheimer’s disease. Brain 129, 2885–2893 (2006).
Dickerson, B. C. et al. The cortical signature of Alzheimer’s disease: regionally specific cortical thinning relates to symptom severity in very mild to mild AD dementia and is detectable in asymptomatic amyloid-positive individuals. Cereb. Cortex 19, 497–510 (2009).
Woodworth, D. C. et al. Dementia is associated with medial temporal atrophy even after accounting for neuropathologies. Brain Commun. 4, fcac052 (2022).
Walters, M. E. et al. Differential MRI atrophy profiles and incident dementia: a cross-national comparison. J. Alzheimers Dis. 107, 1539–1554 (2025).
Scheltens, P., Pasquier, F., Weerts, J. G., Barkhof, F. & Leys, D. Qualitative assessment of cerebral atrophy on MRI: inter-and-intra-observer reproducibility in dementia and normal aging. Eur. Neurol. 37, 95 (1997).
Scheltens, P., Launer, L. J., Barkhof, F., Weinstein, H. C. & Van Gool, W. A. Visual assessment of medial temporal lobe atrophy on magnetic resonance imaging: interobserver reliability. J. Neurol. 242, 557–560 (1995).
Scheltens, P. et al. Atrophy of medial temporal lobes on MRI in “probable” Alzheimer’s disease and normal ageing: diagnostic value and neuropsychological correlates. J. Neurol. Neurosurg. Psychiatry 55, 967–972 (1992).
Duara, R. et al. Medial temporal lobe atrophy on MRI scans and the diagnosis of Alzheimer disease. Neurology 71, 1986–1992 (2008).
DeCarli, C. et al. Qualitative estimates of medial temporal atrophy as a predictor of progression from mild cognitive impairment to dementia. Arch. Neurol. 64, 108–115 (2007).
van der Velpen, I. F. et al. Subcortical brain structures and the risk of dementia in the Rotterdam Study. Alzheimers Dement. 19, 646–657 (2023).
Rao, Y. L. et al. Hippocampus and its involvement in Alzheimer’s disease: a review. 3 Biotech 12, 55 (2022).
Yang, W. & Yu, S. Synucleinopathies: common features and hippocampal manifestations. Cell. Mol. Life Sci. 74, 1485–1501 (2017).
Hill, D. L. et al. Coalition against major diseases/European Medicines Agency biomarker qualification of hippocampal volume for enrichment of clinical trials in predementia stages of Alzheimer’s disease. Alzheimers Dement. 10, 421–429. e423 (2014).
Bartsch, T. & Wulff, P. The hippocampus in aging and disease: from plasticity to vulnerability. Neuroscience 309, 1–16 (2015).
Zhang, J. et al. Hippocampal subfield volumes in mild cognitive impairment and Alzheimer’s disease: a systematic review and meta-analysis. Brain Imaging Behav. 17, 778–793 (2023).
Josephs, K. A. et al. Rates of hippocampal atrophy and presence of post-mortem TDP-43 in patients with Alzheimer’s disease: a longitudinal retrospective study. Lancet Neurol. 16, 917–924 (2017).
Nelson, P. T. et al. Limbic-predominant age-related TDP-43 encephalopathy (LATE): consensus working group report. Brain 142, 1503–1527 (2019).
Hanseeuw, B. J. et al. Association of pathologic and volumetric biomarker changes with cognitive decline in clinically normal adults. Neurology 101, e2533–e2544 (2023).
Hanseeuw, B. et al. Mild cognitive impairment: differential atrophy in the hippocampal subfields. Am. J. Neuroradiol. 32, 1658–1661 (2011).
De Flores, R., La Joie, R. & Chételat, G. Structural imaging of hippocampal subfields in healthy aging and Alzheimer’s disease. Neuroscience 309, 29–50 (2015).
Dawe, R. J. et al. Late-life cognitive decline is associated with hippocampal volume, above and beyond its associations with traditional neuropathologic indices. Alzheimers Dement. 16, 209–218 (2020).
Langnes, E. et al. Anterior and posterior hippocampus macro-and microstructure across the lifespan in relation to memory—a longitudinal study. Hippocampus 30, 678–692 (2020).
Iglesias, J. E. et al. SynthSR: a public AI tool to turn heterogeneous clinical brain scans into high-resolution T1-weighted images for 3D morphometry. Sci. Adv. 9, eadd3607 (2023).
Cooper, R. et al. Bridging the gap: improving correspondence between low-field and high-field magnetic resonance images in young people. Front. Neurol. 15, 1339223 (2024).
Lee, H. et al. Evaluating brain volume segmentation accuracy and reliability of FreeSurfer and Neurophet AQUA at variations in MRI magnetic field strengths. Sci. Rep. 14, 24513 (2024).
Buchanan, C. R. et al. Comparison of structural MRI brain measures between 1.5 and 3 T: data from the Lothian Birth Cohort 1936. Hum. Brain Mapp. 42, 3905–3921 (2021).
Di Perri, C. et al. White matter hyperintensities on 1.5 and 3 Tesla brain MRI in healthy individuals. J. Biomed. Graph. Comput. 3, 53 (2013).
Hsu, P., Marchetto, E., Sodickson, D. K., Johnson, P. M. & Veraart, J. Morphological brain analysis using ultra low-field MRI. Hum. Brain Mapp. 46, e70232 (2025).
Pretzsch, C. M. et al. Evaluating analytic strategies to obtain high-resolution, vertex-level measures of cortical neuroanatomy in children in low-and middle-income countries. Commun. Biol. 8, 918 (2025).
Wardlaw, J. M., Valdés Hernández, M. C. & Muñoz-Maniega, S. What are white matter hyperintensities made of? Relevance to vascular cognitive impairment. J. Am. Heart Assoc. 4, e001140 (2015).
Schmidt, R. et al. Heterogeneity in age-related white matter changes. Acta Neuropathol. 122, 171–185 (2011).
Carmichael, O. et al. Longitudinal changes in white matter disease and cognition in the first year of the Alzheimer disease neuroimaging initiative. Arch. Neurol. 67, 1370–1378 (2010).
Debette, S. & Markus, H. The clinical importance of white matter hyperintensities on brain magnetic resonance imaging: systematic review and meta-analysis. BMJ 341, c3666 (2010).
Prins, N. D. & Scheltens, P. White matter hyperintensities, cognitive impairment and dementia: an update. Nat. Rev. Neurol. 11, 157–165 (2015).
Chen, Y.-C. et al. Exploring the spectrum of subcortical hyperintensities and cognitive decline. J. Neuropsychiatry Clin. Neurosci. 30, 130–138 (2018).
Yoshita, M. et al. Extent and distribution of white matter hyperintensities in normal aging, MCI, and AD. Neurology 67, 2192–2198 (2006).
Keller, J. A. et al. White matter hyperintensity shape is associated with long-term dementia risk. Alzheimers Dement. 19, 5632–5641 (2023).
Garnier-Crussard, A., Cotton, F., Krolak-Salmon, P. & Chételat, G. White matter hyperintensities in Alzheimer’s disease: beyond vascular contribution. Alzheimers Dement. 19, 3738–3748 (2023).
Hotz, I. et al. Performance of three freely available methods for extracting white matter hyperintensities: FreeSurfer, UBO Detector, and BIANCA. Hum Brain Mapp. 43, 1481–1500 (2022).
Prasad, K., Wiryasaputra, L., Ng, A. & Kandiah, N. White matter disease independently predicts progression from mild cognitive impairment to Alzheimer’s disease in a clinic cohort. Dement. Geriatr. Cognit. Disord. 31, 431–434 (2011).
Yi, H.-A., Won, K. S., Chang, H. W. & Kim, H. W. Association between white matter lesions and cerebral Aβ burden. PLoS ONE 13, e0204313 (2018).
Manouvelou, S. et al. Differential diagnosis of behavioral variant and semantic variant of frontotemporal dementia using visual rating scales. Curr. Med. Imaging 16, 444–451 (2020).
Johnson, I. et al. Automated segmentation of white matter hyperintensities on portable low-field magnetic resonance imaging (P9-13.002). Neurology 104, 1610 (2025).
Ferguson, K. J. et al. Visual rating scales of white matter hyperintensities and atrophy: comparison of computed tomography and magnetic resonance imaging. J. Stroke Cerebrovasc. Dis. 27, 1815–1821 (2018).
Chen, Y. et al. Abnormal white matter changes in Alzheimer’s disease based on diffusion tensor imaging: a systematic review. Ageing Res. Rev. 87, 101911 (2023).
Cremers, L. G. et al. Structural disconnectivity and the risk of dementia in the general population. Neurology 95, e1528–e1537 (2020).
Benitez, A. et al. Greater diffusion restriction in white matter in preclinical Alzheimer disease. Ann. Neurol. 91, 864–877 (2022).
Racine, A. M. et al. Associations between white matter microstructure and amyloid burden in preclinical Alzheimer’s disease: a multimodal imaging investigation. Neuroimage Clin. 4, 604–614 (2014).
Weston, P. S., Simpson, I. J., Ryan, N. S., Ourselin, S. & Fox, N. C. Diffusion imaging changes in grey matter in Alzheimer’s disease: a potential marker of early neurodegeneration. Alzheimers Res. Ther. 7, 47 (2015).
Torso, M. et al. Detection of Alzheimer’s disease using cortical diffusion tensor imaging. Hum. Brain Mapp. 42, 967–977 (2021).
Nakaya, M. et al. Assessment of gray matter microstructural alterations in Alzheimer’s disease by free water imaging. J. Alzheimers Dis. 99, 1441–1453 (2024).
Spotorno, N. et al. Measures of cortical microstructure are linked to amyloid pathology in Alzheimer’s disease. Brain 146, 1602–1614 (2023).
O’Halloran, R. et al. Diffusion-weighted imaging at 0.064 T. In Proceedings of International Society in Magnetic Resonance Medicine, 43 (ISMRM, 2022).
Plumley, A. et al. Super-resolution diffusion tensor imaging at 64 mT. In ISMRM and ISMRT Annual Meeting and Exhibition Abstr. 3624 (ISMRM, 2023).
Gholam, J. et al. Undercooked PASTA: tractometry at low field and low SAR. In ISMRM and ISMRT Annual Meeting and Exhibition Abstr. 0315 (ISMRM, 2025).
Balaji, S. et al. Magnetization transfer imaging using non-balanced SSFP at ultra-low field. Magn. Reson. Med. 94, 602–614 (2025).
Pierpaoli, C. & Basser, P. J. Toward a quantitative assessment of diffusion anisotropy. Magn. Reson. Med. 36, 893–906 (1996).
Sperling, R. A. et al. Amyloid-related imaging abnormalities in amyloid-modifying therapeutic trials: recommendations from the Alzheimer’s Association Research Roundtable Workgroup. Alzheimers Dement. 7, 367–385 (2011).
Cummings, J. et al. Aducanumab: appropriate use recommendations update. J. Prev. Alzheimers Dis. 9, 221–230 (2022).
Cummings, J. et al. Aducanumab produced a clinically meaningful benefit in association with amyloid lowering. Alzheimers Res. Ther. 13, 98 (2021).
Hampel, H. et al. Amyloid-related imaging abnormalities (ARIA): radiological, biological and clinical characteristics. Brain 146, 4414–4424 (2023).
Cogswell, P. M. et al. Amyloid-related imaging abnormalities with emerging Alzheimer disease therapeutics: detection and reporting recommendations for clinical practice. Am. J. Neuroradiol. 43, E19–E35 (2022).
Sharifi, K. J. et al. The use of portable MRI in the detection and monitoring of amyloid-related imaging abnormalities. Alzheimers Dement. 21, e104467 (2025).
Okafor, J. P. N. et al. Advanced imaging modalities for ARIA detection and treatment efficacy monitoring in lecanemab therapy for Alzheimer’s disease: a collaborative prospective study. Alzheimers Dement. 20, e095167 (2024).
Bells, S. et al. “Tractometry” – comprehensive multi-modal quantitative assessment of white matter along specific tracts. In ISMRM 19th Annual Meeting and Exhibition Abstr. 0678 (ISMRM, 2011).
Sarracanie, M. et al. Low-cost high-performance MRI. Sci. Rep. 5, 15177 (2015).
Heiss, R., Nagel, A. M., Laun, F. B., Uder, M. & Bickelhaupt, S. Low-field magnetic resonance imaging: a new generation of breakthrough technology in clinical imaging. Invest. Radiol. 56, 726–733 (2021).
Kravchenko, D. et al. Low-field and portable MRI technology: advancements and innovations. Eur. Radiol. Exp. 9, 103 (2025).
Cho, A. MRI for all. Science 379, 748–751 (2023).
Rudisill, K. E., Mathur, N. & Kalyanpur, A. A teleradiology network for the improvement of healthcare and patient management in the developing countries of the African continent. Clin. Imaging 111, 110188 (2024).
Bermingham, S. L. The appropriate use of neuroimaging in the diagnostic work-up of dementia: an economic literature review and cost-effectiveness analysis. Ont. Health Technol. Assess. Ser. 14, 1 (2014).
Lugossy, A.-M., Malkin, R. & Teninty, B. Donations of Medical Imaging Equipment in Global Health. Radiol. Glob. Health: Strateg. Implement. Appl. 15, 65–71 (2025).
Malkin, R. A. Design of health care technologies for the developing world. Annu. Rev. Biomed. Eng. 9, 567–587 (2007).
Illemann, N. M. & Illemann, T. M. Mobile imaging trailers: a scoping review of CT and MRI modalities. Radiography 30, 431–439 (2024).
Shen, F. X. et al. Ethical, legal, and policy challenges in field-based neuroimaging research using emerging portable MRI technologies: guidance for investigators and for oversight. J. Law Biosci. 11, lsae008 (2024).
Wolf, S. M. & Illes, J. Far from home: managing incidental findings in field research with portable MRI. J. Law Med. Ethics 52, 805–815 (2024).
Winter, L. et al. Open-source magnetic resonance imaging: improving access, science, and education through global collaboration. NMR Biomed. 37, e5052 (2024).
Block, K. T. et al. MRI4ALL: a week-long hackathon for the development of an open-source ultra-low-field mri system. J. Magn. Reson. Imaging 62, 959–968 (2025).
Huang, S. et al. ezyMRI: how to build an MRI machine from scratch--experience from a four-day hackathon. Preprint at https://doi.org/10.48550/arXiv.2411.11365 (2024).
Ljungberg, E. et al. Characterization of portable ultra-low field MRI scanners for multi-center structural neuroimaging. Hum. Brain Mapp. 46, e70217 (2025).
Franke, K. & Gaser, C. Ten years of BrainAGE as a neuroimaging biomarker of brain aging: what insights have we gained? Front. Neurol. 10, 789 (2019).
Kaufmann, T. et al. Common brain disorders are associated with heritable patterns of apparent aging of the brain. Nat. Neurosci. 22, 1617–1623 (2019).
Biondo, F. et al. Brain-age is associated with progression to dementia in memory clinic patients. Neuroimage Clin. 36, 103175 (2022).
Biondo, F. et al. Brain-age in ultra-low-field MRI: how well does it work? Preprint at medRxiv https://doi.org/10.1101/2025.10.19.25338298 (2025).
Belder, C. R. et al. Brain volume change following anti-amyloid β immunotherapy for Alzheimer’s disease: amyloid-removal-related pseudo-atrophy. Lancet Neurol. 23, 1025–1034 (2024).
Jack, C. R. Jr et al. A/T/N: an unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology 87, 539–547 (2016).
Carbone, C. et al. Repeated brain MRI utility in identifying neurodegenerative disorders at the pre-dementia stage. Neurobiol. Aging 155, 35–43 (2025).
Chen, Y. A. et al. Tips and challenges for clinical use and interpretation of low field portable MRI in neuroimaging. Emerg. Radiol. 32, 279–289 (2025).
Zabinska, J., de Havenon, A. & Sheth, K. N. Recent advances in portable, low-field magnetic resonance imaging in cerebrovascular disease. Curr. Opin. Neurol. 38, 35–39 (2025).
Donald, K. A. et al. What is next in African neuroscience? eLife 11, e80488 (2022).
World Health Organization. Ageing and Health https://www.who.int/news-room/fact-sheets/detail/ageing-and-health (WHO, 2022).
Acknowledgements
T.E.E. is supported by the William H. Gates Sr. Fellowship from the Alzheimer’s Disease Data Initiative. A.S. is supported by the European Research Council (Horizon 2020 ERC Advanced PASMAR 101021218). H.H.H.A. is funded by a Hypatia Fellowship from the Radboud University Medical Center, project number R0007330. A.G.W. is supported by the European Research Council (Horizon 2020 ERC Advanced PASMAR 101021218) and the Dutch Science Foundation (NWO) Open Technology Grant 18981. D.M.C. is supported by an Alzheimer’s Society Dementia Research Leaders Fellowship (AS-DRL-23-005). D.K.J. is supported by Wellcome (Discovery Awards 227882/Z/23/Z and 317797/Z/24/Z) and the Gates Foundation (UNITY Programme). The authors thank C. Najac and B. Lena for providing example images from the 47 mT Halbach system. These images are from research funded by the NWO, Open Technology Grant 18981. The authors also thank J. Gholam for providing example images from the Hyperfine Swoop System. J. Gholam is supported by the Gates Foundation through the UNITY grant. The authors also thank M. Rosa-Grilo, M. Beament, C. Mummery, N. Fox and G. Parker for providing example images of individuals with ARIA-H and ARIA-E. These images are from research funded by the Alzheimer’s Society Heather Corrie Impact Fund (grant number 577 (AS-PG-21-045)), Biogen Idec UK, the National Institute for Health and Care Research University College London Hospitals Biomedical Research Centre (NIHR UCLH BRC) and the Rosetrees Trust (CF-2022-2\128). The authors also thank N. Vilor Tejedor for aiding in the production of Figs. 4 and 6. Lastly, the authors thank F. Váša and colleagues for the use of open-source data for the figures.
Author information
Authors and Affiliations
Contributions
T.E.E., J.H., A.S., D.M.C. and D.K.J. researched data for the article. T.E.E., J.H., A.G.W., J.H.C., H.H.H.A., D.M.C. and D.K.J. contributed substantially to discussion of the content. T.E.E., J.H., A.S., A.C., D.M.C. and D.K.J. wrote the article. All authors reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
D.M.C. has received consulting fees and travel support from Perceptive Imaging. The other authors declare no competing interests.
Peer review
Peer review information
Nature Reviews Neurology thanks František Váša, Godwin Ogbole and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
IAEA Medical Imaging and Nuclear Medicine Global Resources database (IMAGINE): https://www.iaea.org/resources/hhc/nuclear-medicine/databases/imagine
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Evans, T.E., Harper, J., Salehi, A. et al. The potential of low-field MRI for global dementia care. Nat Rev Neurol (2026). https://doi.org/10.1038/s41582-026-01199-7
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41582-026-01199-7