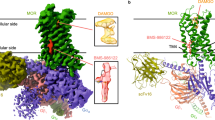
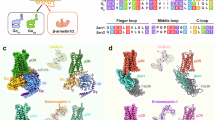
Abstract
Distinct ligands for the same G-protein-coupled receptor (GPCR) activate intracellular signalling partners to varying extents, but the molecular mechanisms that drive these differences remain elusive. Here, hypothesizing that such differences in signalling efficacy might be captured structurally in intermediate states under non-equilibrium conditions, we use a time-resolved cryo-electron-microscopy approach to visualize the GTP-induced activation of the Gαiβγ heterotrimer by the μ-opioid receptor bound to three ligands that show partial, full or super agonism on the receptor1. We resolve ensembles of conformational states along the G-protein activation pathway, including an intermediate state that enables us to visualize receptor dynamics as a function of bound ligand. The results reveal ligand-dependent differences in state occupancy and conformational stability, with higher ligand efficacy correlating with increased dynamics of the receptor’s transmembrane helices 5 and 6. Furthermore, we identify key differences between Gi and Gs in the mechanism of GTP-induced activation, which are likely to underlie the distinct activation kinetics of these G-protein types. Corroborated by molecular-dynamics simulations and single-molecule fluorescence assays, our findings provide a dynamic structural landscape of GPCR–G-protein interactions for ligands of various efficacies, and suggest that partial agonists produce a ‘kinetic trap’ during G-protein activation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
The atomic coordinates of the LFT-bound states have been deposited in the PDB under the accession codes 9ODE, 9ODF, 9ODG, 9ODH and 9ODI, and all associated maps have been deposited in the Electron Microscopy Data Bank (EMDB) under the accession codes EMD-70356, EMD-70357, EMD-70358, EMD-70359, EMD-70360, EMD-70361, EMD-70362 and EMD-70363. The atomic coordinates of the MP-bound states have been deposited in the PDB under accession codes 9ODJ, 9ODK and 9ODL, with maps deposited in the EMDB under EMD-70364, EMD-70365 and EMD-70366. The atomic coordinates of the DAMGO-bound states have been deposited in the PDB under the accession codes 9ODM, 9ODN, 9ODO and 9ODP, with maps deposited in the EMDB under the accession codes EMD-70369, EMD-70370, EMD-70371, EMD-70372, EMD-70373 and EMD-70374. MD trajectory data are available via Figshare at https://doi.org/10.6084/m9.figshare.30856892 (ref. 48). Coordinates of comparison structures were obtained through the PDB under the accession codes 7UL4 (ref. 14), 8GGF (ref. 10), 8GGB (ref. 10), 8GFV (ref. 10) and 1AZT (ref. 20). Cryo-EM maps were obtained through the EMDB under the accession codes EMD-29985 (ref. 10), EMD-40005 (ref. 10), EMD-29951(ref. 10) and EMD-29971 (ref. 10).
References
Qu, Q. et al. Insights into distinct signaling profiles of the μOR activated by diverse agonists. Nat. Chem. Biol. 19, 423–430 (2023).
Manglik, A. et al. Crystal structure of the μ-opioid receptor bound to a morphinan antagonist. Nature 485, 231–326 (2012).
Huang, W. et al. Structural insights into μ-opioid receptor activation. Nature 524, 315–321 (2015).
Manglik, A. et al. Structure-based discovery of opioid analygesics with reduced side effects. Nature 537, 185–190 (2016).
Spetea, M., Asim, M. F., Wolber, G. & Schmidhammer, H. The μ opioid receptor and ligand acting at the μ opioid receptor, as therapeutics and potential therapeutics. Curr. Pharmaceut. Des. 19, 7415–7434 (2013).
Spencer, M. R., Minino, A. M. & Warner, M. Drug overdose deaths in the United States, 2001–2021. NCHS Data Brief https://doi.org/10.15620/cdc:122556 (2022).
Van Eps, N. et al. Interaction of a G protein with an activated receptor opens the interdomain interfaces in the alpha subunit. Proc. Natl Acad. Sci. USA 108, 9420–9424 (2011).
Westfield, G. H. et al. Structural flexibility of the Gαs α-helical domain in the β2-adrenoreceptor Gs complex. Proc. Natl Acad. Sci. USA 108, 16086–16091 (2001).
Punjani, A. & Fleet, D. J. 3D variability analysis: resolving continuous flexibility and discrete heterogeneity from single particle cryo-EM. J. Struct. Biol. 213, 107702 (2021).
Papasergi-Scott, M. M. et al. Time-resolved cryo-EM of G-protein activation by a GPCR. Nature 629, 1182–1191 (2024).
Flock, T. et al. Selectivity determinants of GPCR-G-protein binding. Nature 545, 317–322 (2017).
Zhao, J. et al. Ligand efficacy modulates conformational dynamics of the μ-opioid receptor. Nature 629, 474–480 (2024).
Ballesteros, J. A. & Weinstein, H. Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G protein-coupled receptors. Methods Neurosci. 25, 366–428 (1995).
Robertson, M. J. et al. Structure determination of inactive-state GPCRs with a universal nanobody. Nat. Struct. Mol. Biol. 29, 1188–1195 (2022).
Batebi, H. et al. Mechanistic insights into G-protein coupling with an agonist-bound G-protein-coupled receptor. Nat. Struct. Mol. Biol. 31, 1692–1701 (2024).
Sun, D. et al. Probing Gαi1 protein activation at single amino acid resolution. Nat. Struct. Mol. Biol. 10, 686–694 (2015).
Kaya, A. I. et al. A conserved phenylalanine as a relay between the α5 helix and the GDP binding region of heterotrimeric Gi protein α subunit. J. Biol. Chem. 389, 24475–24487 (2014).
Knight, K. M. et al. A universal allosteric mechanism for G protein activation. Mol. Cell 81, 1384–1396 (2021).
Hammes, G. G., Chang, Y.-C. & Oas, T. G. Conformational selection or induced fit: a flux description of reaction mechanism. Proc. Natl Acad. Sci. USA 106, 13737–13741 (2009).
Sunahara, R. K., Tesmer, J. J., Gilman, A. G. & Sprang, S. R. Crystal structure of the adenylyl cyclase activator Gsα. Science 278, 1943–1947 (1997).
Jang, W., Lu, S., Xu, X., Wu, G. & Lambert, N. A. The role of G protein conformation in receptor–G protein selectivity. Nat. Chem. Biol. 19, 687–295 (2023).
Linder, M. E., Ewald, D. A., Miller, R. J. & Gilman, A. G. Purification and characterization of Goα and three types of Giα after expression in Escherichia coli. J. Biol. Chem. 265, 8243–8251 (1990).
Graziano, M. P., Freissmuth, M. & Gilman, A. G. Expression of Gsα in Escherichia coli: purification and properties of two forms of the protein. J. Biol. Chem. 264, 409–418 (1989).
Neto, J. A. et al. Biased versus partial agonism in the search for safer opioid analgesics. Molecules 25, 3870 (2020).
Peisley, A. & Skiniotis, G. 2D projection analysis of GPCR Complexes by negative stain electron microscopy. Methods Mol. Biol. 1335, 29–38 (2015).
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Kimanius, D., Dong, L., Sharov, G., Nakane, T. & Scheres, S. H. W. New tools for automated cryo-EM single-particle analysis in RELION-4.0. Biochem. J. 478, 4169–4185 (2021).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Rohou, A. & Grigorieff, N. CTFFIND4: fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2019).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Zivanov, J., Nakane, T. & Scheres, S. H. W. A Bayesian approach to beam-induced motion correction in cryo-EM single-particle analysis. IUCrJ 6, 5–17 (2019).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010).
Liebschner, D. et al. Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix. Acta Crystallogr. D 75, 861–877 (2019).
Robertson, M. J., van Zundert, G. C. P., Borrelli, K. & Skiniotis, G. GemSpot: a pipeline for robust modeling of ligands into cryo-EM maps. Structure 28, 707–716 (2020).
Lomize, M. A., Pogozheva, I. D., Joo, H., Mosberg, H. I. & Lomize, A. L. OPM database and PPM web server: resources for positioning of proteins in membranes. Nucleic Acids Res. 40, D370–D376 (2012).
Jorgensen, W. L., Chandrasekhar, J., Madura, J. D., Impey, R. W. & Klein, M. L. Comparison of simple potential functions for simulating liquid water. J. Chem. Phys. 79, 926–935 (1983).
Jo, S., Kim, T., Iyer, V. G. & Im, W. CHARMM-GUI: a web-based graphical user interface for CHARMM. J. Comput. Chem. 29, 1859–1865 (2008).
Phillips, J. C. et al. Scalable molecular dynamics on CPU and GPU architectures with NAMD. J. Chem. Phys. 153, 044130 (2020).
Huang, J. et al. CHARMM6m: an improved force field for folded and intrinsically disordered proteins. Nat. Methods 14, 71–73 (2017).
Klauda, J. B. et al. Update of the CHARMM all-atom additive force field for lipids: validation on six lipid types. J. Phys. Chem. B 114, 7830–7843 (2010).
Vanommeslaeghe, K. et al. CHARMM general force field: a force field for drug-like molecules compatible with the CHARMM all-atom additive biological force fields. J. Comput. Chem. 31, 671–690 (2010).
Humphrey, W., Dalke, A. & Schulten, K. VMD: visual molecular dynamics. J. Mol. Grap. 14, 33–38 (1996).
Goddard, T. D. et al. UCSF ChimeraX: meeting modern challenges in visualization and analysis. Protein Sci. 27, 14–25 (2018).
Gregorio, G. G. et al. Single-molecule analysis of ligand efficacy in β2AR–G-protein activation. Nature 547, 68–73 (2017).
Juette, M. F. et al. Single-molecule imaging of non-equilibrium molecular ensembles on the millisecond timescale. Nat. Methods 13, 341–344 (2016).
Fairhead, M., Krndija, D., Lowe, E. D. & Howarth, M. Plug-and-play pairing via defined divalent streptavidins. J. Mol. Biol. 426, 199–214 (2014).
Kiselev, R. et al. Parallel stopped-flow interrogation of diverse biological systems at the single-molecule scale. Nat. Methods 23, 78–87 (2026).
Robertson, M. J. & Skiniotis, G. Non-equilibrium snapshots of ligand efficacy at the μ-opioid receptor. figshare https://doi.org/10.6084/m9.figshare.30856892 (2026).
Acknowledgements
This work was supported by the St. Jude Children’s Research Hospital Collaborative Research Consortium on GPCRs (G.S. and S.C.B.), NIH R01 DA059978 (S.M.), NIH K99HL16140601 (M.M.P.-S.), NIH K99/R00 HD107581 (M.J.R.) and CPRIT award RR230042 (M.J.R.). M.J.R. is a CPRIT Scholar in Cancer Research. We thank N. Lambert, R. Sunahara, J. Javitch and I. Chen for discussions and critical reading of the manuscript; Z. Luo for figure and animation preparation; N. Casajuana Martin and M. Babu for providing additional computational resources and support; H. Prasad for generating the Gi1 expression construct; Y. Zhang for expressing and partially purifying Gi1 for single-molecule studies; and the St. Jude Children’s Research Hospital Single-Molecule Imaging Center for instrumentation.
Author information
Authors and Affiliations
Contributions
G.S. conceived and supervised the project. M.J.R. expressed and purified proteins, prepared complexes, prepared cryo-EM grids, collected, analysed and processed cryo-EM data, built and refined atomic models, designed and executed MD simulations, analysed data and prepared figures. M.M.P.-S. collected data for MP time points. M.C.P. expressed and purified G proteins. M.H. purified Δ7 MOR for biophysical studies under the supervision of R.K. A.M. labelled Δ7 MOR and performed single-molecule fluorescence and data analysis under the supervision of S.C.B. B.R.V. synthesized compounds under the supervision of S.M. M.J.R. and G.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
S.M. is a cofounder of Sparian Biosciences and has patents on mitragynine-based modulators. G.S. is a cofounder of and consultant for Deep Apple Therapeutics. S.C.B. holds an equity interest in Lumidyne Technologies.
Peer review
Peer review information
Nature thanks Henrik Dohlman, Maria Marti-Solano and Cheng Zhang for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Cryo-EM data processing workflow.
a, General processing workflow for the nucleotide-free datasets. b, General processing workflow for the GTP-bound datasets.
Extended Data Fig. 2 Cryo-EM maps of MOR–Gi bound to MP, DAMGO and LFT in the baseline, GTP-primed, G-ACT-1, G-ACT-2 and G-ACT-3 states.
a, Maps for the nucleotide-free state, each of the consensus refinements of the three main ensembles of states for the GTP-bound state, and the 3DVA-separated states of the G-ACT-2/3 ensemble with each of the three ligands. G-ACT-3* corresponds to G-ACT-3 for MP and DAMGO, and G-ACT-2′ for LFT. b, Additional maps generated in this work with local refinement and/or additional 3DVA/3D classification.
Extended Data Fig. 3 AHD motions in the nucleotide-free and GTP-bound Gi heterotrimer.
a, Comparison of the 3DVA analysis results for mode 0 and 1 of the nucleotide-free (left) and GTP-primed state (right) with each of the three ligands. b, AHD–RHD distances from nucleotide-free MD simulations where the AHD starts open (top, yellow gradient traces) or closed (bottom, magenta gradient traces). AHD–RHD distances from open and closed structures are drawn as straight lines. c, Comparison of the linker I region of Gi1 in the presence (purple) and absence (yellow–orange) of GTP. Maps are for the GTP-bound state (top right), consensus nucleotide-free (bottom left) and 3DVA-filtered for the presence of ordered linker (bottom right). d, Interactions between the AHD of Gα and Gβ in the GTP-bound, GTP-primed state.
Extended Data Fig. 4 Interactions between Gα and MOR TM5–TM6 in the G-ACT-3 state.
a, MP G-ACT-3 state model-map comparison highlighting the interface between TM5–ICL3–TM6 of MOR (aqua) and the C terminus-RHD of Gα (yellow). b, Overlay of MP G-ACT-3 with the structure of MOR–Mb6 complex (Mb6 in magenta, PDB: 7UL4). c, Residue frequency plot for the base of TM5 and TM6 across all human family A GPCRs. d, Analysis of the mean side-chain formal charge across all family A GPCRs at the base of TM5 and TM6. e, Analysis of the mean side-chain formal charge across all family A GPCRs subdivided by primary G-protein coupler. f, Time course of percentage of TM5–TM6 Gi interactions and ICL3 proximity interactions in the G-ACT-3 simulations with MP (top, purple traces) and LFT (bottom, green traces) over 2 microsecond MD simulations. Lines plotted are rolling averages of nine frames of MD simulation from five independent replicates.
Extended Data Fig. 5 Three-dimensional variability analysis of the G-ACT-1 and G-ACT-2/3 states.
a, Results of 3DVA for LFT MOR–Gi G-ACT-1 state particles with Gα coloured yellow for frame 0 and magenta for frame 19 of the principal component. b, Comparison of the coil portion of the switch II region for MP G-ACT-2 and G-ACT-3 highlighting the differences in G202 and G203. c, Comparison of the MP G-ACT-2 model with the map obtained from 3DVA filtering of the LFT G-ACT-2/3 particles, pooling the initial third of particles from the principal component. Arrow denotes the region of switch II near GTP that is more disordered in the map. d, Alignment on Gβγ of the structures modelled in the maps from LFT G-ACT-2/3 particles 3DVA analysis filtering, both the initial (3DVA 1) and final (3DVA 2) third of the particles. e, Comparison of the models for MP G-ACT-2, MP G-ACT-3 and the map obtained from 3DVA filtering of the LFT G-ACT-2/3 particles, pooling the final third of particles from the principal component. Green arrow highlights new map features and red arrow highlights where map features were lost compared to LFT G-ACT-2/3 3DVA 1. f, Alternative contour of an LFT G-ACT-2/3 map showing G protein and MOR/micelle separation.
Extended Data Fig. 6 Differences in Gi and Gs activation dynamics.
a, Comparison of the 3DVA analysis results for the first principal component of the nucleotide-free (left) and GTP-bound, predominant particle stack state (right) between MOR–Gi and β2AR–Gs. b, Alignment of Gi (yellow) and Gs (light blue) in an AHD-closed, GTP-bound state (G-ACT-2 for MOR–Gi, 3DVA frame 20 for β2AR–Gs). The inset is centred on the linker I region, highlighting the packing of αA and linker I against the rest of the AHD in Gi that is absent in Gs. c, Comparison of the sequences of the α1–linker I–αA sequences of Gi1 and Gs (short). d, Comparison of Gi (purple, magenta, yellow) and Gs (light blue) from roughly equivalent states in their cryo-EM trajectories for GTP-induced activation from their respective receptors.
Extended Data Fig. 7 Structural differences between Gi and Gs.
a, Comparison of the linker I region of Gs in the GTP-bound, cryo-EM frame 20 state (PDB: 8GGF) and GTP-analogue bound crystal structure (PDB: 1AZT). b, MD simulation AHD–RHD distance results for the adrenergic receptor–Gs complex and MOR–Gi complex with the linker I and Y69 swapped to match Gs, five independent trajectories each starting from either the AHD-closed or the AHD-open conformation. AHD–RHD distances for the open and closed conformations are shown as lines. c, Comparison of Gi in the G-ACT-2 state with Gs in the GTP-bound, cryo-EM frame 20 state (PDB: 8GGF, left) and GTP-analogue-bound crystal structure (PDB: 1AZT, right).
Extended Data Fig. 8 Interactions in the Gαi subunit during GTP-induced activation.
a, Analysis of the formation of new GTP–Gα contacts (white–green scale) and new Gα–Gα contacts in the key linker I, switch I (linker II), switch II and switch III regions (white–blue scale) as MOR/Gi GTP transitions from state GTP-primed, G-ACT-1, G-ACT-2 and G-ACT-3.
Extended Data Fig. 9 Two-dimensional class averages for the main MOR–Gi baseline and intermediate states.
a, Two-dimensional class averages of all of the states resolved in this manuscript. G-ACT-3* corresponds to G-ACT-3 for MP and DAMGO, and G-ACT-2′ for LFT.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs 1–3 and Supplementary Table 1.
Supplementary Video 1 (download MP4 )
Cartoon of the MOR–Gi complex undergoing GTP binding and transition through the states observed in this work.
Supplementary Video 2 (download MP4 )
3DVA maps derived from the MOR–Gi–MP G-ACT-2/3 particle stack.
Supplementary Video 3 (download MP4 )
3DVA maps derived from the MOR–Gi–DAMGO G-ACT-2/3 particle stack.
Supplementary Video 4 (download MP4 )
3DVA maps derived from the MOR–Gi–LFT G-ACT-2/3 particle stack.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Robertson, M.J., Modak, A., Papasergi-Scott, M.M. et al. Non-equilibrium snapshots of ligand efficacy at the μ-opioid receptor. Nature (2025). https://doi.org/10.1038/s41586-025-10056-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41586-025-10056-4