Abstract
The synergistic combination of two antimicrobial drugs is a promising therapeutic modality for many infectious diseases. However, systemic fungal infections still have a high mortality rate because of distinct in vivo distributions of the two drugs. Here we address this challenge by designing an antifungal polymer that forms micelles suitable for delivering a second antifungal agent to achieve temporal and spatial consistency of delivery. We show that the polymer, which mimics host defense peptides, exerts a synergistic effect with the antifungal amphotericin B (AmB). The AmB-encapsulated micelles (AmBmicelles) greatly reduce the toxicity of AmB through slow release and expand its therapeutic window in vivo. AmBmicelles can selectively target fungal pathogens through charge interactions with the fungal membrane. In mouse models of systemic candidiasis and cryptococcal meningitis, AmBmicelles increase the survival rate by 67–100% compared to the state-of-the-art drug AmBisome or AmBisome and 5-flucytosine combination, suggesting that the strategy may be effective in combating drug-resistant fungal infections including meningitis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data needed to evaluate the conclusions are available within the article or its Supplementary Information. Any additional data are available from the corresponding author. Source data are provided with this paper.
References
GBD 2021 Antimicrobial Resistance Collaborators Global burden of bacterial antimicrobial resistance 1990–2021: a systematic analysis with forecasts to 2050. Lancet 404, 1199–1226 (2024).
Denning, D. W. Global incidence and mortality of severe fungal disease. Lancet Infect. Dis. 24, e428–e438 (2024).
Zhang, F. et al. A marine microbiome antifungal targets urgent-threat drug-resistant fungi. Science 370, 974–978 (2020).
Fisher, M. C. et al. Tackling the emerging threat of antifungal resistance to human health. Nat. Rev. Microbiol. 20, 557–571 (2022).
Hancock, R. E. W. & Sahl, H.-G. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 24, 1551–1557 (2006).
Homer, J. A., Johnson, R. M., Koelln, R. A., Moorhouse, A. D. & Moses, J. E. Strategic re-engineering of antibiotics. Nat. Rev. Bioeng. 3, 213–229 (2025).
Fisher, M. C., Hawkins, N. J., Sanglard, D. & Gurr, S. J. Worldwide emergence of resistance to antifungal drugs challenges human health and food security. Science 360, 739–742 (2018).
Ho, C. S. et al. Antimicrobial resistance: a concise update. Lancet Microbe 6, 100947 (2025).
Xiong, M. et al. Helical antimicrobial polypeptides with radial amphiphilicity. Proc. Natl Acad. Sci. USA 112, 13155–13160 (2015).
Li, X. et al. Supramolecular antibacterial materials for combatting antibiotic resistance. Adv. Mater. 31, e1805092 (2019).
Kong, H. et al. Development of aromatic-linked diamino acid antimicrobial peptide mimics with low hemolytic toxicity and excellent activity against methicillin-resistant Staphylococcus aureus (MRSA). J. Med. Chem. 66, 7756–7771 (2023).
Ghosh, C., Sarkar, P., Issa, R. & Haldar, J. Alternatives to conventional antibiotics in the era of antimicrobial resistance. Trends Microbiol. 27, 323–338 (2019).
Lehar, J. et al. Synergistic drug combinations tend to improve therapeutically relevant selectivity. Nat. Biotechnol. 27, 659–666 (2009).
Brochado, A. R. et al. Species-specific activity of antibacterial drug combinations. Nature 559, 259–263 (2018).
Si, Z., Pethe, K. & Chan-Park, M. B. Chemical basis of combination therapy to combat antibiotic resistance. JACS Au 3, 276–292 (2023).
Ding, X. et al. A macromolecule reversing antibiotic resistance phenotype and repurposing drugs as potent antibiotics. Adv. Sci. 7, 2001374 (2020).
Si, Z. et al. A glycosylated cationic block poly(β-peptide) reverses intrinsic antibiotic resistance in all ESKAPE Gram-negative bacteria. Angew. Chem. Int. Ed. 59, 6819–6826 (2020).
Gong, Y., Wang, H. & Sun, J. AMP-mimetic antimicrobial polymer-involved synergic therapy with various coagents for improved efficiency. Biomacromolecules 25, 4619–4638 (2024).
Schmidt, N. W. et al. Engineering persister-specific antibiotics with synergistic antimicrobial functions. ACS Nano 8, 8786–8793 (2014).
Bai, H. et al. A supramolecular antibiotic switch for antibacterial regulation. Angew. Chem. Int. Ed. 54, 13208–13213 (2015).
Liu, L. et al. Self-assembled cationic peptide nanoparticles as an efficient antimicrobial agent. Nat. Nanotechnol. 4, 457–463 (2009).
Jiang, W. et al. Peptide-mimicking poly(2-oxazoline)s possessing potent antifungal activity and BBB penetrating property to treat invasive infections and meningitis. J. Am. Chem. Soc. 145, 25753–25765 (2023).
Zhou, M. et al. A dual-targeting antifungal is effective against multidrug-resistant human fungal pathogens. Nat. Microbiol 9, 1325–1339 (2024).
Zhang, X. et al. Development of lipo-γ-AA peptides as potent antifungal agents. J. Med. Chem. 65, 8029–8039 (2022).
Liu, F. et al. Synergistic wall digestion and cuproptosis against fungal infections using lywallzyme-induced self-assembly of metal-phenolic nanoflowers. Nat. Commun. 15, 9004 (2024).
Chen, L. et al. Brain glucose induces tolerance of Cryptococcus neoformans to amphotericin B during meningitis. Nat. Microbiol 9, 346–358 (2024).
Maji, A. et al. Tuning sterol extraction kinetics yields a renal-sparing polyene antifungal. Nature 623, 1079–1085 (2023).
Bennett, J. E. et al. A comparison of amphotericin B alone and combined with flucytosine in the treatment of cryptoccal meningitis. N. Engl. J. Med. 301, 126–131 (1979).
Beck, A., Goetsch, L., Dumontet, C. & Corvaia, N. Strategies and challenges for the next generation of antibody–drug conjugates. Nat. Rev. Drug Discov. 16, 315–337 (2017).
Li, X., et al. Multifunctional nanoparticle-mediated combining therapy for human diseases. Signal Transduct. Target. Ther. 9, 1 (2024).
Detappe, A. et al. Molecular bottlebrush prodrugs as mono- and triplex combination therapies for multiple myeloma. Nat. Nanotechnol. 18, 184–192 (2023).
Sofias, A. M. & Lammers, T. Multidrug nanomedicine. Nat. Nanotechnol. 18, 104–106 (2023).
Cheng, T. et al. Green tea catechin-based complex micelles combined with doxorubicin to overcome cardiotoxicity and multidrug resistance. Theranostics 6, 1277–1292 (2016).
Lancet, J. E. et al. CPX-351 versus 7+3 cytarabine and daunorubicin chemotherapy in older adults with newly diagnosed high-risk or secondary acute myeloid leukaemia: 5-year results of a randomised, open-label, multicentre, phase 3 trial. Lancet Haematol. 8, e481–e491 (2021).
Lancet, J. E. et al. CPX-351 (cytarabine and daunorubicin) liposome for injection versus conventional cytarabine plus daunorubicin in older patients with newly diagnosed secondary acute myeloid leukemia. J. Clin. Oncol. 36, 2684–2692 (2018).
Li, Y. et al. Co-delivery of precisely prescribed multi-prodrug combination by an engineered nanocarrier enables efficient individualized cancer chemotherapy. Adv. Mater. 34, e2110490 (2022).
Zhang, R. X., Wong, H. L., Xue, H. Y., Eoh, J. Y. & Wu, X. Y. Nanomedicine of synergistic drug combinations for cancer therapy—strategies and perspectives. J. Control. Release 240, 489–503 (2016).
Zhou, M. et al. Poly(2-oxazoline)-based functional peptide mimics: eradicating MRSA infections and persisters while alleviating antimicrobial resistance. Angew. Chem. Int. Ed. 59, 6412–6419 (2020).
Tauhardt, L., Kempe, K., Gottschaldt, M. & Schubert, U. S. Poly(2-oxazoline) functionalized surfaces: from modification to application. Chem. Soc. Rev. 42, 7998–8011 (2013).
Lorson, T. et al. Poly(2-oxazoline)s based biomaterials: a comprehensive and critical update. Biomaterials 178, 204–280 (2018).
Zhu, Y., Yang, B., Chen, S. & Du, J. Polymer vesicles: mechanism, preparation, application, and responsive behavior. Prog. Polym. Sci. 64, 1–22 (2017).
Cui, H., Webber, M. J. & Stupp, S. I. Self-assembly of peptide amphiphiles: from molecules to nanostructures to biomaterials. Biopolymers 94, 1–18 (2010).
Zhang, Z., Ai, S., Yang, Z. & Li, X. Peptide-based supramolecular hydrogels for local drug delivery. Adv. Drug Deliv. Rev. 174, 482–503 (2021).
Walker, L. et al. The viscoelastic properties of the fungal cell wall allow traffic of AmBisome as intact liposome vesicles. mBio 9, e02383-17 (2018).
Colombo, R. & Rich, J. R. The therapeutic window of antibody drug conjugates: a dogma in need of revision. Cancer Cell 40, 1255–1263 (2022).
Yongvongsoontorn, N. et al. Carrier-enhanced anticancer efficacy of sunitinib-loaded green tea-based micellar nanocomplex beyond tumor-targeted delivery. ACS Nano 13, 7591–7602 (2019).
Wang, C. H., Wang, W. T. & Hsiue, G. H. Development of polyion complex micelles for encapsulating and delivering amphotericin B. Biomaterials 30, 3352–3358 (2009).
Laconde, G., Amblard, M. & Martinez, J. Synthesis of α-amino acid N-carboxyanhydrides. Org. Lett. 23, 6412–6416 (2021).
Dos Reis, T. F., et al. A host defense peptide mimetic, brilacidin, potentiates caspofungin antifungal activity against human pathogenic fungi. Nat. Commun. 14, 2052 (2023).
Davis, S. A. et al. Nontoxic antimicrobials that evade drug resistance. Nat. Chem. Biol. 11, 481–487 (2015).
Wilcock, B. C., Endo, M. M., Uno, B. E. & Burke, M. D. C2′-OH of amphotericin B plays an important role in binding the primary sterol of human cells but not yeast cells. J. Am. Chem. Soc. 135, 8488–8491 (2013).
Liu, Y. -g, et al. NLRP3 inflammasome activation mediates radiation-induced pyroptosis in bone marrow-derived macrophages. Cell Death Dis. 8, e2579 (2017).
Wiradharma, N., Tong, Y. W. & Yang, Y. Y. Self-assembled oligopeptide nanostructures for co-delivery of drug and gene with synergistic therapeutic effect. Biomaterials 30, 3100–3109 (2009).
Jiang, W. et al. Short guanidinium-functionalized poly(2-oxazoline)s displaying potent therapeutic efficacy on drug-resistant fungal infections. Angew. Chem. Int. Ed. 61, e202200778 (2022).
Mukherjee, J., Pirofski, L. A., Scharff, M. D. & Casadevall, A. Antibody-mediated protection in mice with lethal intracerebral Cryptococcus neoformans infection. Proc. Natl Acad. Sci. USA 90, 3636–3640 (1993).
Jafari, H. S. et al. Effects of antifungal therapy on inflammation, sterilization, and histology in experimental Candida albicans meningitis. Antimicrob. Agents Chemother. 38, 83–89 (1994).
Acknowledgements
This research was supported by the Chinese Academy of Sciences Project for Young Scientists in Basic Research (YSBR-111, to R.L.), the National Natural Science Foundation of China (T2325010, to R.L.; 22475069 and 52203162, to Y.W.; 22305082, to M.Z.), the National Key Research and Development Program of China (2022YFC2303100, to R.L.), the Shanghai Frontiers Science Center of Optogenetic Techniques for Cell Metabolism (Shanghai Municipal Education Commission) and the Open Research Fund of State Key Laboratory of Polymer Physics and Chemistry (Changchun Institute of Applied Chemistry, Chinese Academy of Sciences). We thank Research Center of Analysis and Test of East China University of Science and Technology and the Analysis and Testing Center of School of Chemical Engineering at East China University of Science and Technology for support with the characterization. We thank the staff members of the Integrated Laser Microscopy System at the National Facility for Protein Science in Shanghai, Zhangjiang Lab for providing technical support and assistance in data collection and analysis.
Author information
Authors and Affiliations
Contributions
R.L. directed the whole project and conceptualized the study. R.L., M.Z. and L.L. proposed the strategy, designed the experiments, evaluated the data and wrote the paper. L.L. performed the majority of the experiments. Q.Z. contributed to the preparation of antifungal micelles. L.L., X.X., J.Z. and J.X. contributed to the in vitro antifungal study. L.L., Z.C., X.X.and J.X. contributed to the in vivo study. M.Z. and Y.W. contributed to the data analysis and discussion of the results. All authors proofread the paper.
Corresponding author
Ethics declarations
Competing interests
R.L. and M.Z. are coinventors on a patent application covering the reported synthesis of poly(2-oxazoline)s and their antifungal application. R.L. and L.L. are also coinventors on a patent application covering the reported application of poly(2-oxazoline)s in drug delivery. The other authors declare no competing interests.
Peer review
Peer review information
Nature Biotechnology thanks Sankaran Thayumanavan and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
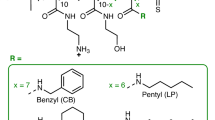
Extended Data Fig. 1 Structural characterization and reproducibility of block copolymers PGlyX-b-PNapY.
(a) Structural of poly(2-oxazoline)s block copolymers. (b) Summary of GPC characterization. Mn, number average molecular weight. Ð is the dispersity index. DP is the obtained degree of polymerization. (c) 1H NMR spectra of block poly(2-oxazoline)s in DMSO-D6, 400 MHz. (d) GPC traces of PGly6-b-PNap14 from 6 batches at the side chain NH-Boc protected stage. (e) Summary of GPC characterization. (f) The MIC values of PGly6-b-PNap14 from 6 batches.
Extended Data Fig. 2 The particle size and critical micelle concentration of polymer micelles.
(a) DLS measurement of the Size distribution of polymer micelles is indicated by spline fits. (b) The CMC of polymer micelles.
Extended Data Fig. 3 The synergistic effect of polymer micelles and AmB.
(a-k) The synergistic effect of polymer micelles and AmB against C. albicans, C. neoformans, C. glabrata and C. parapsilosis. The blue zone represents fungal growth visible to the naked eye, and the white zone represents complete fungal growth inhibition invisible to the naked eye. The star symbol represents the position of FICI value.
Extended Data Fig. 4 The fungal pathogen targeting ability of micelles.
(a-c) Distribution of AmBisome liposomes and micelles in a cells and fungi coculture mixture. (d-e) Distribution of micelles in a coculture mixture of Human umbilical vein endothelial cells (HUVEC, primary cells) and fungi, and Bone marrow-derived macrophages (BMDM, primary cells) and fungi.
Extended Data Fig. 5 The characterization and pathogen targeting studies of a series of polymeric micelles.
(a-b) Summary of GPC characterization. (c) GPC traces of of (PGlyaPEOzb)6-b-PNapY from 6 batches at the side chain NH-Boc protected stage. (d) DLS measurements and TEM images of micelles assembled from polymer (PGlyaPEOzb)6-b-PNapY. The scale bars in TEM images all represent 200 nm. (e-f) Distribution of AmBisome liposomes and micelles in a mammalian cells (NIH/3T3) and fungi (C. albicans) coculture mixture.
Extended Data Fig. 6 The loading capacity of polymer micelles.
(a) The water solubility of AmB and AmBmicelles. (b-c) MIC and FICI values of AmB in AmBmicelles and the mixture of AmB and micelles. (d) Normalized DLS measurements of the Size distribution of AmB and AmBmicelles are indicated by spline fits. (e) UV-vis spectra of AmB in H2O, AmB in DMSO, micelles and AmBmicelles. (f) The stability of AmBmicelles over two weeks at 4 oC. (g) The release kinetics of AmB in vitro at 37 oC. (h) The emission wavelength of nile red and nile red encapsulated in micelles in water under 559 nm excitation.
Extended Data Fig. 7 Pharmacokinetics and in vivo distribution of micelles and AmB in AmBmicelles.
(a) Structure of PGly4-b-PNap16-FITC. (b) Normalized DLS measurements of the Size distribution of AmBmicelles and AmBmicelles-FITC are indicated by spline fits. (c-d) Pharmacokinetics curve of micelles and AmB. (e-g) Distribution of micelles and AmB in different organs. The concentration ratios of micelles and AmB in different organs are listed at the top of the graph. The data are expressed as mean±s.d. n = 3 biologically independent animals.
Extended Data Fig. 8 The in vivo immunogenicity and toxicity of micelles and AmBmicelles.
(a) Cytokine profiling for serum after daily intravenous injection of micelles, AmBmicelles and saline, respectively, for 4 consecutive days. The mouse cytokine array coordinates are shown in Table S1. (b) Gray values of cytokine and chemokine expression. (c) The concentration of C3a in serum after daily intravenous injection of micelles, AmBmicelles and saline, respectively, for 4 consecutive days (n = 4 biologically independent animals). Data are presented as mean values ± s.d. Statistical analysis was conducted using one-way ANOVA with multiple comparisons using Tukey’s post-test.
Extended Data Fig. 9 The therapeutic efficacy of micelles and AmBmicelles in systemic candidiasis infection.
(a) Schematic diagram of an in vivo antifungal study. Survival ratio (b) and fungal cell burden in different organs (c) of mice with systemic fungal infection after treatment with saline (200 μL), micelles (15 mg/kg), and micelles (30 mg/kg), respectively. (n = 6 biologically independent animals). Data are presented as mean values ± s.d. Statistical analysis was conducted using one-way ANOVA with multiple comparisons using Tukey’s post hoc test. (d-e) Survival ratio and fungal cell burden in different organs of mice with systemic fungal infection after treatment with Saline (200 μL), AmB@PIC micelles (0.5 mg/kg), AmBisome (0.5 mg/kg, 1 mg/kg, 2 mg/kg, 4 mg/kg) and AmBmicelles (0.5 mg/kg) (n = 6 biologically independent animals). The concentration indicated is the concentration of AmB encapsulated by micelles. Data are presented as mean values ± s.d. Statistical analysis was conducted using one-way ANOVA with multiple comparisons using Tukey’s post hoc test.
Extended Data Fig. 10 Brain tissue slices and fungal cell burden in the brains of mice infected with fungal meningitis.
(a) Representative histological images from Grocott hexamine-silver stain of early treatment models of meningitis mouse brain at 15 and 22 days after infection treated with AmBmicelles (1 mg/kg AmB), AmBisome + 5FC (1 mg/kg AmB and 75 mg/kg 5FC), and Saline, respectively. Black arrows indicate the vesicular lesions in brain. (b) The fungal cell burden in mouse brains at 1 and 3 days after infection. (n = 6 biologically independent animals). Data are presented as mean values ± s.d. Statistical analysis was conducted using two tailed Student’s t test.
Supplementary information
Supplementary Information (download PDF )
Materials and instrumentation and Supplementary Figs. 1–15 and Table 1.
Source data
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 9 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 10 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, L., Zhou, M., Xiao, X. et al. Effective combinatorial antifungal therapy using a host defense peptide mimic that self-assembles into delivery micelles. Nat Biotechnol (2026). https://doi.org/10.1038/s41587-025-02930-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41587-025-02930-3