Abstract
The high burden of dilated cardiomyopathy (DCM) in individuals of African descent remains incompletely explained. Here, to explore a genetic basis, we conducted a genome-wide association study in 1,802 DCM cases and 93,804 controls of African genetic ancestry (AFR). A nonsense variant (rs3211938:G) in CD36 was associated with increased risk of DCM. This variant, believed to be under positive selection due to a protective role in malaria resistance, is present in 17% of AFR individuals but <0.1% of European genetic ancestry (EUR) individuals. Homozygotes for the risk allele, who comprise ~1% of the AFR population, had approximately threefold higher odds of DCM. Among those without clinical cardiomyopathy, homozygotes exhibited an 8% absolute reduction in left ventricular ejection fraction. In AFR, the DCM population attributable fraction for the CD36 variant was 8.1%. This single variant accounted for approximately 20% of the excess DCM risk in individuals of AFR compared to those of EUR. Experiments in human induced pluripotent stem cell-derived cardiomyocytes demonstrated that CD36 loss of function impairs fatty acid uptake and disrupts cardiac metabolism and contractility. These findings implicate CD36 loss of function and suboptimal myocardial energetics as a prevalent cause of DCM in individuals of African descent.
Similar content being viewed by others
Main
Individuals of self-identified Black race are at disproportionate risk for dilated cardiomyopathy (DCM), a preeminent cause of heart failure with reduced ejection fraction (HFrEF) and the most common indication for cardiac transplantation1,2. A twofold increased odds of DCM has been reported among Black compared to white individuals, an observation not fully explained by differences in risk factor burden or socioeconomic factors such as access to care3,4,5. A distinct genetic basis for DCM in Black populations has been postulated, invoking the possibility of genetic polymorphisms specific to those with recent African genetic ancestry (AFR)6,7,8. While many rare and common genetic variants have now been implicated in the pathogenesis of DCM, their identification has relied on the study of populations composed largely of individuals of European genetic ancestry (EUR)9,10,11,12,13,14,15,16. Investigations for DCM-associated polymorphisms of particular relevance to African ancestral groups have been limited by the modest numbers of AFR participants in most cohorts with genetic data17,18. In this study, we leveraged the genetic diversity of the United States (US) Department of Veterans Affairs’ (VA) Million Veteran Program (MVP) biobank19 to examine the genetic determinants of DCM in AFR individuals. We ascertained the ancestry-specific prevalence of DCM in MVP and conducted a genome-wide association study (GWAS) of DCM in AFR participants. We characterized and functionally validated an AFR-specific genetic association and contextualized its contribution to the long-observed disparity in DCM risk between individuals of African and European descent. An overview of the study design can be found in Supplementary Fig. 1.
Results
Baseline characteristics
In the 95,606 MVP participants of AFR ancestry in this study, the mean age was 56.5 years, the majority were male (84.9%), and most had hypertension (69.9%). The prevalence of DCM was nearly twofold greater among individuals of AFR (1.9%) versus European (EUR) ancestry (1.0%, P < 0.001; Table 1). Of note, AFR participants also had a higher prevalence of hypertension, chronic kidney disease (CKD) and diabetes, while EUR participants had a higher prevalence of atrial fibrillation and coronary artery disease (CAD).
Association analyses, gene and variant prioritization
GWAS of DCM in MVP AFR participants (n = 1,802 cases) identified an association signal reaching genome-wide significance on chromosome 7 (Fig. 1a); the minor allele (C) of the lead variant rs3211916 was associated with increased risk of DCM (odds ratios (OR)C allele = 1.34 (1.22–1.48), P = 6.23 × 10−9). This intronic variant was located within the CD36 gene (Fig. 1b), and no meaningful test statistic inflation was detected (λ = 1.026; Supplementary Fig. 2). Conditional analysis adjusting for the lead variant did not show any other signals in the region (Supplementary Fig. 3), and the DCM association was robust to adjustment for cardiovascular risk factors, including hypertension, body mass index (BMI), diabetes, CAD, atrial fibrillation, CKD, total cholesterol, high-density lipoprotein (HDL) cholesterol, smoking status and alcohol intake (Supplementary Table 1). Of note, a second association signal on chromosome 10 reached genome-wide significance (lead variant rs3827681 within the ARHGAP22 gene); however, it did not replicate in external cohorts and was excluded from subsequent analyses (Supplementary Table 2). A full list of variants reaching the suggestive P-value threshold of P < 1 × 10−6 can be found in Supplementary Table 3, and the full GWAS summary statistics are available through dbGaP (https://www.ncbi.nlm.nih.gov/gap/) under the MVP accession (phs001672). Of the 217 previously reported SNPs from recent DCM GWAS11,14,15,16,20,21,22 (Supplementary Table 4) that overlapped with our AFR GWAS, 170 (78%) showed directionally consistent effects (binomial test P < 1 × 10−15), and 58 reached nominal significance (P < 0.05), supporting substantial cross-ancestral portability of known DCM risk loci.
a, Manhattan plot showing the association between common genetic variants and DCM across the genome in AFR individuals. b, Regional association plot of the CD36 locus, displaying variant-level associations and linkage disequilibrium with the lead SNP (rs3211916) based on the 1000 Genomes AFR population. c, Association of heterozygous (T/G) and homozygous (G/G) rs3211938 genotypes with DCM (versus the reference genotype (T/T)) in African ancestry participants of the VA MVP and PMBB, estimated using logistic regression adjusted for age, sex and the first ten principal components. Data are shown as ORs with 95% CIs. d, Associations of rs3211938 genotypes with cardiac MRI-derived left ventricular traits in African ancestry participants from the UKB, MESA and JHS. Associations were estimated using linear regression adjusted for age, sex and the first ten principal components. Data are presented as the mean difference relative to T/T homozygotes (per s.d. increase in trait) with 95% CIs. Only the meta-analysis results are shown; full results are provided in Supplementary Fig. 6. LVESVi, left ventricular end-systolic volume indexed for body surface area; LVEDVi, left ventricular end-diastolic volume indexed for body surface area; LVMi, left ventricular mass indexed for body surface area.
The association signal for DCM at chromosome 7 colocalized with regional summary statistics for CD36 gene expression in the left ventricle in data from the Genotype-Tissue Expression (GTEx) Project23 (Supplementary Fig. 4a, PPH4 = 0.94) and plasma CD36 levels in the Atherosclerosis Risk in Communities study24,25 (Supplementary Fig. 4b, PPH4 = 0.98). Additionally, rs3211916 was associated (P < 5 × 10−4) with expression of CD36 in several tissues, including left ventricle and atrial appendage, and was not associated with expression of genes beyond CD36 in GTEx (Supplementary Table 5), implicating CD36 as the most likely causal gene mediating DCM risk.
The 95% credible set was comprised of four common variants, including the lead signal rs3211916 (Supplementary Fig. 5 and Supplementary Table 6). Notably, left ventricular CD36 expression and plasma CD36 data highlighted another variant in the credible set, rs3211938, as the strongest variant in the region (Supplementary Fig. 4a,b). The G allele at rs3211938 is a nonsense variant resulting in the introduction of a premature stop codon (Y325Ter) within the second binding pocket of the CD36 protein and before its second transmembrane domain (Supplementary Fig. 6). This nonsense variant was in moderate-to-high linkage disequilibrium with the lead variant, rs3211916 (r2 = 0.64 in 1000 Genomes African ancestry, r2 = 0.65 in MVP AFR), and confers increased risk of DCM (ORG = 1.33 (1.20–1.48), P = 1.37 × 10−7), which was also robust to cardiovascular risk factor adjustment (Supplementary Table 1). The risk allele (G) of rs3211938 was specific to individuals of AFR ancestry with a frequency of 9% in AFR populations, but less than 0.1% in European ancestry populations, a consistent observation across the five study cohorts and various populations in dbSNP26 (Supplementary Table 7). Conditioning on the lead variant abrogated the association for rs3211938 (and vice versa), indicating that the two polymorphisms represent the same association signal for DCM (Supplementary Fig. 3). Due to its functional consequences, high LD with the lead variant, and strong effect estimate, rs3211938 was considered the likely causal variant at the region (hereby referred to as the ‘nonsense variant’) and served as the focus of downstream analyses.
Sex- and age-stratified associations
Given the preponderance of older, male participants in MVP, we examined whether the association between rs3211938 and DCM persists across ages and biological sexes (Supplementary Table 8). We observed a statistically significant association between the nonsense variant and DCM in participants above age 55 (n = 1,324 cases; ORG = 1.24 (1.09–1.42), P = 8.74 × 10−4), and an even stronger association in participants less than age 55, despite fewer cases (n = 478 cases; ORG = 1.56 (1.29–1.90), P = 4.72 × 10−6). The association was robust in the male-only stratum (n = 1,716 cases; ORG = 1.31 (1.18–1.47), P = 1.31 × 10−6) but also reached nominal significance among women-only despite a small sample size (n = 86 cases; ORG = 1.64 (1.08–2.51), P = 0.012). Of note, among AFR participants in MVP, women had a mean age of 48.4 compared to 58.0 for men, suggesting that the higher effect estimate observed in women may be related to the younger age of this population.
Replication of the association signal
We sought additional support for the association between rs3211938 and DCM in the AFR subsets of four independent cohorts. First, we examined the association of rs3211938 with DCM and the related phenotype of HFrEF in the Penn Medicine BioBank (PMBB)27. Using an additive model, the association of rs3211938 and DCM replicated in PMBB, and the effect size in PMBB (ORG = 1.37) was consistent with that in MVP (ORG = 1.33), with a meta-analyzed ORG of 1.33 (P = 5.22 × 10−9). Notably, analyses by genotype were suggestive of a nonlinear association between the number of G alleles at rs3211938 and DCM (that is, potential incomplete dominance), which was consistent across MVP and PMBB. As compared to those with the T/T genotype, heterozygotes (T/G) had an OR of 1.25 for DCM (Pmeta = 7.82 × 10−5) while risk allele homozygotes (G/G) had an OR of 2.75 (Pmeta = 2.37 × 10−9; Fig. 1c and Supplementary Table 9). Similarly, the association of rs3211938 with HFrEF was consistent across MVP and PMBB and suggestive of a nonlinear shape; heterozygotes had an OR of 1.15 (Pmeta = 2.48 × 10−6) while risk allele homozygotes had an OR of 2.05 (Pmeta = 4.85 × 10−12; Supplementary Fig. 7b and Supplementary Table 9). MVP participants with the TT, TG and GG genotypes did not differ appreciably with regard to mean age, biological sex composition, or prevalence of baseline clinical risk factors (Supplementary Table 10).
Validation of the association signal with cardiac MRI data
Next, using cardiac MRI data from UK Biobank (UKB)28, the Multi-Ethnic Study of Atherosclerosis (MESA)29 and the Jackson Heart Study (JHS)30, we tested the association of rs3211938 with relevant measures of left ventricular (LV) structure and function in those without overt cardiomyopathy. Risk allele homozygosity (that is, G/G genotype) was associated with differences consistent with a subclinical DCM phenotype, including an approximately 1 s.d. decrease in left ventricular ejection fraction (LVEF), 1 s.d. increase in body surface area-indexed values of left ventricular end-diastolic volume and left ventricular end-systolic volume, and more modest increases in body surface area-indexed left ventricular mass (P < 0.01 for all; Fig. 1d and Supplementary Fig. 7c) when compared to wild-type homozygotes (T/T). For LVEF, this corresponded to a 6.8%, 6.3% and 9.4% decrease in absolute values across UKB, MESA and JHS, respectively (and a 7.9% absolute decrease in LVEF across all three studies), for individuals with the G/G versus T/T genotype.
Phenome-wide association scan
To determine the broad phenotypic consequences of CD36 perturbation, we tested the association of rs3211938 with a spectrum of cardiovascular and noncardiovascular outcomes and traits in AFR participants from MVP and UKB. Consistent with the above link to LV dysfunction, the nonsense variant was positively associated with several phenotypic codes of relevance to cardiomyopathy and heart failure, such as those for ‘primary/intrinsic cardiomyopathy’, ‘hypertensive heart disease’ and ‘placement of cardiac defibrillator in situ’, the latter likely a marker of severe LV dysfunction. In addition, there were positive associations with metabolic outcomes such as ‘gout’, and inverse associations with thromboembolic outcomes such as ‘pulmonary embolism’ (Fig. 2a, Supplementary Table 11 and Supplementary Fig. 8). We also observed significant associations with HDL-cholesterol-related traits, several blood cell and platelet traits and increased levels of serum acetone (Fig. 2b and Supplementary Table 12).
a, Phenome-wide association results for binary traits (PheCodes) tested in up to 121,078 AFR ancestry participants of the VA MVP using logistic regression and adjusted for age, sex and ten principal components. Of 1,805 PheCodes tested, only those reaching Bonferroni-corrected significance threshold (P < 2.77 × 10−5) are shown. Results are presented as ORs with 95% CIs. b, Association of rs3211938 with continuous traits in AFR participants from MVP and UKB, tested using linear regression adjusted for age, sex and ten principal components. Traits were scaled to have a mean of zero and s.d. of 1. Of 247 traits tested, only those reaching Bonferroni-corrected significance (P < 2.02 × 10−4) are shown. Results are presented as MD per additional risk (G) allele (per s.d. increase in trait) with 95% CIs. †Estimates calculated in the UK Biobank population; all others from MVP. All analyses used an additive genetic model with the G allele at rs3211938 as the effect allele. MD, mean difference.
Assessments of population-wide impact on DCM
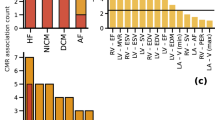
To assess the population-wide impact of rs3211938 on the risk for DCM, we focused on cases of DCM that occurred during the study enrollment period. Over a mean of 4.2 person-years, 829 incident DCM cases occurred, with rates of 91 and 59 per 100,000 person-years for MVP AFR and EUR individuals, respectively. We first estimated the population attributable fraction (PAF) of DCM due to clinical risk factors and rs3211938 in AFR participants. The highest PAF for DCM was for hypertension (30.4%). The PAF for rs3211938 alone (8.1%) was similar to that for other prominent clinical risk factors such as BMI (10.9%), diabetes mellitus (5.8%) and CKD (3.8%; Fig. 3 and Supplementary Table 13).
Bar plot showing the estimated PAF and 95% confidence intervals for clinical risk factors and rs3211938 in relation to DCM among African ancestry participants of the VA MVP. PAF values were truncated at a lower limit of zero, the minimum theoretical contribution to risk.
We then assessed the extent to which differences in DCM risk between AFR and EUR populations are accounted for by differences in cardiovascular risk factors and/or the presence of rs3211938. In an age-and-sex-adjusted model, AFR ancestry was associated with a 77% (HR = 1.77, 95% confidence interval (CI) = 1.52–2.06, P < 0.001) increase in risk for incident DCM as compared to EUR ancestry. Among clinical factors, adjustments for hypertension (HR = 1.65, 95% CI = 1.42–1.92), diabetes mellitus (HR = 1.70, 95% CI = 1.46–1.97), CKD (HR = 1.70, 95% CI = 1.46–1.97) or all three combined (HR = 1.58, 95% CI = 1.35–1.83) resulted in the greatest attenuation of risk. Notably, adjustment for rs3211938 alone attenuated the association between AFR ancestry and DCM by roughly one-fifth (HR = 1.61, 95% CI = 1.36–1.89), even when added to the three aforementioned clinical factors (HR = 1.43, 95% C I 1.21–1.69; Table 2 and Supplementary Table 14).
Experimental validation in hiPSC-CMs
As CD36 is a key transporter of fatty acids into cardiomyocytes (CMs), we hypothesized that its loss of function—the proposed consequence of rs3211938—impairs lipid uptake and compromises the energy supply required for contractility. To test this, we performed small-interfering RNA (siRNA)-mediated knockdown of CD36 in human-induced pluripotent stem cell-derived CMs (hiPSC-CMs), achieving a 76% reduction in CD36 mRNA expression (Fig. 4a). After 2 h of lipid starvation, CD36 knockdown led to a significant decrease in intracellular BODIPY FL C16 fluorescence (P < 0.0001), indicating reduced uptake of long-chain fatty acids (Fig. 4b). Since fatty acid oxidation is a major energy source for CMs, we next assessed mitochondrial function. CD36-deficient hiPSC-CMs exhibited impaired oxygen consumption rate (OCR) with palmitate as the sole fuel source, along with significantly reduced β-oxidation, as shown by lower etomoxir-sensitive OCR (Fig. 4c,d). Furthermore, CD36 knockdown impaired mitochondrial activity, evidenced by a marked reduction in maximal respiratory capacity, a key indicator of oxidative metabolic function (P = 0.0001; Fig. 4e,f).
a, Quantitative PCR showing CD36 mRNA expression in hiPSC-CMs transfected with siRNA targeting CD36 versus scrambled control siRNA, presented as relative fold change. **P < 0.01 (Mann–Whitney test; each dot represents an experimental replicate). b, Long-chain fatty acid uptake after 1-h incubation with BODIPY FL C16 in scrambled and CD36-siRNA-treated hiPSC-CMs. Data are shown as mean ± s.e.m.; n = 10 per group. ****P < 0.0001 (Mann–Whitney test). c, Averaged real-time OCR in hiPSC-CMs following CD36 knockdown or transfection with scrambled control, measured using seahorse extracellular flux analysis. After 1 h of starvation, cells were provided Pal as the sole substrate, followed by sequential treatments with Eto (β-oxidation inhibitor), Oligo (ATP synthase inhibitor), CCCP (respiratory uncoupler) and R/A (respiratory chain inhibitors). d, β-oxidation rate, calculated as OCR reduction following etomoxir treatment. Data are shown as mean ± s.e.m.; scrambled n = 11, CD36 siRNA n = 8. ****P < 0.0001 (Mann–Whitney test); dots represent experimental replicates. e, Comparison of averaged mitochondrial function using seahorse extracellular flux analyzer. f, Maximal respiratory capacity, defined as the difference between OCR following CCCP and rotenone/antimycin treatment in hiPSC-CMs treated with CD36 siRNA or scrambled control. Data are presented as mean ± s.e.m.; scrambled n = 9, CD36 siRNA n = 14. ***P < 0.0001 (Mann–Whitney test); dots represent experimental replicates. Pal, palmitate; Eto, etomoxir; Oligo, oligomycin; R/A, rotenone/antimycin.
Given that contractile dysfunction is a hallmark of DCM, we next investigated whether CD36-related metabolic disruption impairs force generation. Engineered cardiac microtissues were created by embedding CD36-siRNA or scrambled-control hiPSC-CMs alongside human cardiac fibroblasts in a 3D matrix (Fig. 5a). Microtissues comprising CD36-siRNA-treated iPSC-CMs exhibited a 53% reduction in contractile stress compared to control microtissues (Fig. 5b,c), supporting a model in which CD36 loss of function drives a cellular DCM phenotype via sequential disruption of energy metabolism and contractility (Fig. 5d).
a, Schematic of the EHT model used for assessing contractile function. b, Representative traces of contractile stress over time, normalized to the maximum value of scrambled control tissues. c, Quantification of contractile stress (force per unit area) in engineered cardiac microtissues derived from hiPSC-CMs transfected with scrambled or CD36 siRNA. Scrambled n = 15, CD36 siRNA n = 13; **P < 0.01 (Mann–Whitney test); center line, median; box limits, upper and lower quartiles; whiskers, 1.5× interquartile range; points, experimental replicates. d, Schematic summary of CD36 function in LCFA uptake and its role in energy generation required for CM contraction. The rs3211938 variant is highlighted in red, with red downward arrows indicating the cascade of disruptions caused by CD36 loss of function, ultimately contributing to a DCM phenotype. EHT, engineered heart tissue; LCFA, long-chain fatty acid.
Discussion
We identified a nonsense variant in CD36 associated with increased risk of DCM and left ventricular dysfunction across five independent cohorts. The variant was present in 17% of individuals of African ancestry but was nearly nonexistent in those of European ancestry. Roughly one-fifth of the excess risk for incident DCM in AFR populations, as compared to EUR, was attributable to this single genetic variant. Functional studies confirmed that CD36 loss of function disrupts fatty acid uptake and cardiac energy metabolism in hiPSC-CMs, leading to impaired contractility and a cellular DCM phenotype.
Several conclusions arise from our study findings. First, regional selective pressures on disease-relevant genetic variants might contribute to the differences in DCM prevalence observed across ancestries. Notably, CD36 is a scavenger receptor that may have a role in the sequestration of Plasmodium falciparum-infected red blood cells31,32. While earlier reports were inconsistent33,34,35, a more recent study has implicated the CD36 nonsense variant (rs3211938) in offering protection against malaria36. This suggests that, in malaria-endemic regions such as sub-Saharan Africa, the polymorphism may be positively selected despite its harmful effects on the myocardium, similar to the observed relationship between malaria and sickle cell trait. A similar phenomenon has been described for genetic polymorphisms within APOL1 that confer protection against Trypanosoma brucei (the cause of ‘African sleeping sickness’) but increase the risk for CKD, linking regional selection to another condition that disproportionately affects individuals of self-identified Black race37.
Second, differences in the genetic underpinnings of DCM between individuals of AFR and EUR ancestry have implications for both prognostication and mechanistic understanding of disease. The Eurocentric nature of most genetic association studies and resultant genetic predictors is well known38. Here the identification of a common, high-impact, genetic variant associated with DCM—not seen in prior studies of EUR participants9,10,11,12,13,14,15,16—affirms the importance of genomic analyses in non-European populations to improve estimations of genetic risk across ancestries and to offer new mechanistic insights into disease pathogenesis.
Third, rs3211938 mediates a considerable portion of DCM risk in the AFR population and does so independently of traditional clinical factors. Modifiable clinical factors remain a focus of heart failure prevention; in fact, we found that approximately 40% of DCM in the AFR population was attributable to hypertension and elevated BMI39. However, the association of rs3211938 with DCM was robust after adjusting for clinical factors. Moreover, its contribution to DCM risk in the AFR cohort was comparable to that of diabetes and CKD, underscoring its substantial impact as a population-level risk factor. Nevertheless, rs3211938 and all tested clinical factors accounted for less than half of the difference in DCM risk between AFR and EUR populations. Therefore, other (possibly genetic, social and structural) factors likely account for the remainder of the risk difference and require additional study.
Finally, the link between a common, loss-of-function variant in CD36 and DCM suggests that impaired myocardial energetics is a prevalent mechanism of LV dysfunction, particularly in the AFR population. CD36 is a multifunctional scavenger receptor for several ligands, including oxidized LDL cholesterol, thrombospondin and free fatty acids40. Prior studies have linked rs3211938 with a wide range of phenotypes, the majority of which were recapitulated in our PheWAS. Among traits of relevance to myocardial structure and function, rs3211938 has been associated with various electrocardiographic phenotypes, such as the TP segment, QT interval, P-wave duration and QRS interval. However, a mechanistic basis for these associations, predicated on CD36 function, has yet to be established41,42.
The normally functioning heart relies on fatty acid oxidation as a primary fuel source, while LV dysfunction is characterized by a reduction in myocardial fatty acid usage and proportionally greater reliance on ketone bodies43. In the myocardium, CD36 is responsible for a large proportion of cellular fatty acid uptake, as evidenced by Cd36 knockout mice, which experience 50–80% reductions in fatty acid transport compared to wild type44, and human studies that have found reduced cardiac fatty acid uptake in those with CD36 deficiency45. Conflicting experimental evidence exists around the direction of the association between CD36 and cardiomyopathy. In vivo models of CD36 loss-of-function have demonstrated both myocardial dysfunction due to inadequate fatty acid oxidation and improved myocardial function from reduced lipotoxicity—possibly depending on the presence of physiologic versus elevated circulating fatty acid levels46,47. In addition, the best therapeutic approach for modulating myocardial energetics has been debated as some studies have suggested improved markers of function in DCM with inhibitors of fatty acid oxidation (such as perhexilene)48. However, here we provide human genetic evidence that loss of CD36 function is associated with an increased risk of incident DCM. Furthermore, our in vitro modeling of CD36 loss-of-function in hiPSC-CMs supports that this association may be due to reduced fatty acid transport into the myocardium and consequent suboptimal substrate usage. In fact, our observation that carriers of rs3211938 manifest higher levels of serum ketones (acetone) suggests a subclinical shift in energy usage that may portend an elevated risk of cardiomyopathy. Furthermore, consistent with recent findings—and contrary to prevailing hypotheses— treatments that optimize cardiac energy metabolism by increasing fatty acid oxidation may provide a promising therapeutic strategy for patients with DCM and HFrEF that is orthogonal in strategy to current, guideline-based therapies49.
Our study has several limitations. First, cases of DCM were ascertained through electronic health record-based phenotyping from the VA and Penn Medicine Healthcare Systems, thereby carrying the potential for disease misclassification or under recognition, although this limitation would tend to bias associations toward the null. Second, our PAF and mediation analyses did not consider genetic variants beyond rs3211938—such as EUR-specific risk variants and established rare and common genetic variants (including polygenic scores) of relevance to DCM—that may contribute to the disparities in DCM risk seen across ancestries. Third, we did not conduct an exhaustive analysis of all nonsense mutations within CD36 via genome sequencing; instead, we were limited to variants that were directly genotyped or well-imputed. Fourth, the absolute numbers of homozygotes for rs3211938 in our imaging study cohorts were low, although consistent and statistically significant associations were observed across cohorts, even with these modest numbers. Finally, our in vitro assays in hiPSC-CMs used a complete knockdown of CD36 by siRNA to demonstrate a link between CD36 loss-of-function and DCM; future efforts will require introducing rs3211938 into hiPSC-CMs to fully validate the mechanism of action of this specific polymorphism.
In conclusion, we found a common, African ancestry-specific nonsense variant in CD36 associated with LV dysfunction and DCM across five cohorts. Further exploration of this association and underlying mechanisms may inform targeted screening strategies and therapeutic interventions and help reduce the excess burden of DCM among individuals of African descent.
Methods
Study populations
We evaluated study participants from MVP19 to compare the prevalence of DCM among AFR and EUR veterans. For the primary genetic association analyses, we studied 95,606 AFR participants (1,802 DCM cases) from MVP and pursued replication of genetic association results in 11,143 AFR participants (365 DCM cases) from PMBB27. We further validated findings using cardiac magnetic resonance imaging data from UKB28, MESA29 and JHS30 (Supplementary Fig. 1). All studies were approved by their respective institutional review board committees, and all participants provided informed consent. A full description of the contributing studies can be found in the Supplementary Note.
Clinical phenotypes
Disease phenotypes in MVP and PMBB were defined using hospital billing code data. The primary outcome was DCM, defined by the presence of International Classification of Diseases, Tenth Revision (ICD-10) billing code I42.0 (‘DCM’). Second, we evaluated genetic associations with the broader clinical phenotype of HFrEF in MVP, as defined previously50. We excluded those with other forms of cardiomyopathy and/or heart failure from the control group. Clinical cardiovascular risk factors in MVP—hypertension, BMI, diabetes, atrial fibrillation, CKD, total cholesterol, HDL cholesterol, smoking status and alcohol intake—were derived from the medical record (see Supplementary Note for additional details regarding phenotyping).
Genotyping, imputation and ancestry definition
Genotyping and quality control in MVP have been reported previously51,52 and are described in detail in the Supplementary Note. EUR and AFR genetic ancestry were approximated by projecting PCA loadings from the 1000 Genomes Project into MVP, then using a random forest classifier (Supplementary Note). Population-specific principal components (PCs) were computed using plink253. In MVP, rs3211938 was imputed and rs3211916 was genotyped (with only the few people with missing genotype data for this SNP imputed). Detailed information on genotyping and imputation for PMBB, UKB, MESA and JHS, as well as other genetic methods, can be found in the Supplementary Note.
Genome-wide association analysis and replication
A case–control genome-wide association analysis for DCM was performed in AFR participants from MVP using logistic regression with adjustment for age at study enrollment, biological sex and the first ten population-specific genetic PCs in plink2a53. Statistical significance was set at P < 5 × 10−8. In MVP, lead variants were also evaluated for association with DCM via logistic regression after adjusting for baseline clinical cardiovascular risk factors. We further examined the association of any lead signal with HFrEF in MVP, before pursuing replication in AFR participants from PMBB using the same logistic regression model and covariates.
Additional validation was performed by testing the association of any lead variant with cardiac MRI-derived measures of LV structure and function (LVEF, left ventricular end-diastolic volume, left ventricular end-systolic volume and indexed left ventricular mass) in UKB, MESA and JHS study participants without documentation of clinical heart failure, cardiomyopathy or CAD. Linear regression models were used, adjusting for age, biological sex and the first ten genetic PCs, and meta-analysis across studies was performed using fixed-effects and inverse-variance weighting in GWAMA54. Further information can be found in the Supplementary Note.
Gene and variant prioritization
Publicly available transcriptomic data (from GTEx23 version 8) and proteomic data (from the Atherosclerosis Risk in Communities study24,25) were used to identify the gene likely mediating the uncovered genetic signal. Conditional analyses were combined with assessments of CD36 expression in the left ventricle and CD36 levels in plasma to identify the common variant likely mediating the genetic association (Supplementary Note).
Phenome-wide association study (PheWAS)
We performed a PheWAS of the nonsense variant with 1,805 ICD code-based phenotypes (n = 121,078) and 49 clinical laboratory biomarkers (n = 117,702) in AFR individuals of MVP, as well as 30 laboratory biomarkers (n = 6,831) and 168 metabolites (n = 1,686) in AFR individuals of UKB. Logistic regression was used for the analysis of disease phenotypes, and linear regression for analyses of continuous biomarkers and metabolites, each adjusting for age at enrollment, biological sex and the first ten PCs in an additive model. Significance was defined as P < 2.8 × 10−5 (0.05/1,805) for the binary trait analysis and P < 2.0 × 0−4 (0.05/247) for continuous traits (more information is provided in the Supplementary Note).
Incident disease analysis
In AFR and EUR MVP participants free of baseline DCM, heart failure or coronary artery disease, we pursued time-to-event analyses to determine the risk of incident DCM. We estimated the PAF for DCM of the nonsense variant compared to clinical risk factors, the hazards of DCM in AFR compared to EUR ancestry participants and the extent to which adjusting for clinical factors and/or the functional genetic variant attenuates the observed ancestry-specific risk difference (Supplementary Note).
hiPSC-CM culture
Commercial Cellartis Human iPS Cell Line 22 (Takara Bio) was differentiated into CMs. The hiPSC line was maintained in StemFlex media (Life Technologies) and passaged using ReleSR (STEMCELL Technologies). When the hiPSC cultures reached 80% confluency, they were dissociated into a single-cell suspension using Accutase (STEMCELL Technologies). After 3 days, these single cells formed uniformly sized spheroids. The differentiation into ventricular CMs was initiated by modulating the Wnt/β-catenin signaling pathway. On day 17, beating spheroids were dissociated and cultured on flasks coated with fibronectin. Subsequently, the cells underwent a 6-day period of glucose starvation in a lactate purification medium consisting of DMEM without glucose or pyruvate, containing 1 M lactate, 200 μM ascorbic acid and 14 ng ml−1 sodium selenite. After purification, hiPSC-CMs were maintained in DMEM without glucose or pyruvate and supplemented with B27 supplements, nonessential amino acid, 2.5 mM glucose, 4 mM lactate, 0.4% concentrated lipid, 1× sodium pyruvate, 0.2 mM ascorbic acid, 0.5 mM L-carnitine, 5 mM taurine and 5 mM creatine. Unless otherwise specified, before functional analysis, cells were placed for 24 h in the same media lacking lactate and glucose to enhance lipid usage. Primary human cardiac fibroblast cells (Cell Systems, ACBRI 5118) were maintained in FGM-2 media (Lonza) and passaged with TrypLE at confluence.
CD36 knockdown
For gene knockdown, siRNA targeting CD36 (SR319610) and control, nontargeting scrambled sequence were purchased from OriGene. On day 30 of differentiation, transfection with either siCD36 or scrambled sequence was performed using Lipofectamine RNAiMAX Transfection Reagent (Thermo Fisher Scientific) as per the manufacturer’s instructions. Cells were used after 6 days of transfection.
qRT-PCR was performed to evaluate gene knockdown. Total RNA was extracted using the Zymo micro-kit (Zymo) according to the manufacturer’s instructions. cDNA was synthesized with iScript cDNA synthesis (Bio-Rad). Quantitative PCR was performed using SsoAdvanced Universal Probes Supermix (Bio-Rad) on a Bio-Rad CFX384 real-time system with probes from IDT for CD36 (Hs.PT.56a.3615957) and HPRT (Hs.PT.58v.45621572).
Long-chain fatty acid uptake
Three days after transfection, scrambled and siCD36 hiPSC-CMs were seeded at a density of 2 × 104 cells per well on a 384-well plate coated with fibronectin. The assessment of lipid uptake was carried out using BODIPY FL C16 (Thermo Fisher Scientific, D3821). Four days after seeding, the cells underwent a triple wash with Hanks’ balanced salt solution media and were subsequently preincubated in Hanks’ balanced salt solution for 2 h. Measurements were taken at 37 °C for 1 h after introducing the fluorescent fatty acid using a microplate fluorescence reader (Synergy H1) at 505/512 nm. Total protein lysate quantified by BCA was used for normalization.
Assessments of mitochondrial function
The Seahorse XFe96 extracellular flux analyzer was used to evaluate metabolic function. CMs were seeded onto fibronectin-coated plates at a density of 3 × 104 cells per XFe96 well. To compare the contribution of exogenous fatty acid to respiration after CD36 knockdown, cells were starved for an hour in Agilent Seahorse XF DMEM Basal Medium supplemented with 0.5 mM L-carnitine and 2 mM glutamine, after which 30 μl of palmitate–BSA was added before measurements. For analysis of mitochondrial function, the culture medium was replaced with Agilent Seahorse XF DMEM Basal Medium, supplemented with 2 mM glutamine, 5.5 mM glucose, 1 mM sodium pyruvate, 0.5 mM L-carnitine and 0.4% of concentrated lipid 1 h before and during the assay. During both tests, specific inhibitors were injected as indicated—etomoxir (100 μM), oligomycin (2.5 μM), CCCP (2 μM) and rotenone/antimycin A (2.5 μM). OCR values were normalized to the total protein content, quantified using the BCA method after cell lysis with RIPA buffer. Baseline OCR was defined as the average values measured from time points 1–4 during the experiments. Lipid β-oxidation was estimated as etomoxir-sensitive OCR and calculated by subtracting basal respiration to OCR after etomoxir addition. The maximal respiratory capacity was calculated as the difference in OCR between the uncoupler CCCP and rotenone/antimycin A.
Contractility assessment
Cardiac microtissues were generated as previously described55. PDMS microfabricated tissue gauge (µTUG) substrates, including fluorescent microbeads (Polysciences, Fluoresbrite 17147), were molded from SU-8 masters onto cantilevers. These μTUG substrates were treated with 0.2% Pluronic F127 for 30 min. Scrambled and siCD36 CMs were dissociated 3 days after transfection using STEMdiff Cardiomyocyte Dissociation Kit and mixed with stromal cells (human ventricular fibroblasts) to enable tissue compaction, with fibroblasts representing 6% of the total myocyte population. A suspension of ~1,200,000 cells combined with a reconstitution mixture of liquid-neutralized collagen I (2 mg ml−1; BD Biosciences) and human fibrinogen (0.5 mg ml−1; Sigma-Aldrich) was added to the substrate. Centrifugation was used to drive cells into micropatterned wells, and within 24 h, fibroblasts compacted the tissues. Force measurements were taken after 4 days of culture. On the day of the experiment, the µTUGs were incubated for 1 h in a glucose and lactate-deficient medium primarily reliant on lipids as the source of energy. Biphasic square electrical field stimulation of 1 ms was applied via two carbon electrodes positioned 2 cm apart on the sample sides, connected through platinum wires to a stimulator. Fluorescent microbead displacement on the cantilevers was monitored using the Spot Tracker plug-in in ImageJ, and the data were analyzed with a custom MATLAB script to calculate twitch force, static force and velocity. Contractile force and stress (force per unit area) generated by the engineered cardiac microtissues were quantified.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
GWAS summary statistics for MVP are available through dbGaP (https://www.ncbi.nlm.nih.gov/gap/) at study accession phs001672. Individual-level data for MESA and JHS are available through dbGaP at study accessions phs000209 and phs000286. Individual-level data for UKBB are available by application (www.ukbiobank.ac.uk).
Code availability
No custom code was developed for this study.
References
McNally, E. M. & Mestroni, L. Dilated cardiomyopathy: genetic determinants and mechanisms. Circ. Res. 121, 731–748 (2017).
Bibbins-Domingo, K. et al. Racial differences in incident heart failure among young adults. N. Engl. J. Med. 360, 1179–1190 (2009).
Coughlin, S. S., Labenberg, J. R. & Tefft, M. C. Black–white differences in idiopathic dilated cardiomyopathy: the Washington DC dilated Cardiomyopathy Study. Epidemiology 4, 165–172 (1993).
Chandra, A. et al. Race- and gender-based differences in cardiac structure and function and risk of heart failure. J. Am. Coll. Cardiol. 79, 355–368 (2022).
Dries, D. L. et al. Racial differences in the outcome of left ventricular dysfunction. N. Engl. J. Med. 340, 609–616 (1999).
Tishkoff, S. A. & Verrelli, B. C. Patterns of human genetic diversity: implications for human evolutionary history and disease. Annu. Rev. Genomics Hum. Genet. 4, 293–340 (2003).
Fu, W. et al. Analysis of 6,515 exomes reveals the recent origin of most human protein-coding variants. Nature 493, 216–220 (2013).
Haggerty, C. M. et al. Genomics-first evaluation of heart disease associated with titin-truncating variants. Circulation 140, 42–54 (2019).
Herman, D. S. et al. Truncations of titin causing dilated cardiomyopathy. N. Engl. J. Med. 366, 619–628 (2012).
Jordan, E. et al. Evidence-based assessment of genes in dilated cardiomyopathy. Circulation 144, 7–19 (2021).
Villard, E. et al. A genome-wide association study identifies two loci associated with heart failure due to dilated cardiomyopathy. Eur. Heart J. 32, 1065–1076 (2011).
Meder, B. et al. A genome-wide association study identifies 6p21 as novel risk locus for dilated cardiomyopathy. Eur. Heart J. 35, 1069–1077 (2014).
Esslinger, U. et al. Exome-wide association study reveals novel susceptibility genes to sporadic dilated cardiomyopathy. PLoS ONE 12, e0172995 (2017).
Aragam, K. G. et al. Phenotypic refinement of heart failure in a national biobank facilitates genetic discovery. Circulation 139, 489–501 (2019).
Tadros, R. et al. Shared genetic pathways contribute to risk of hypertrophic and dilated cardiomyopathies with opposite directions of effect. Nat. Genet. 53, 128–134 (2021).
Garnier, S. et al. Genome-wide association analysis in dilated cardiomyopathy reveals two new players in systolic heart failure on chromosomes 3p25.1 and 22q11.23. Eur. Heart J. 42, 2000–2011 (2021).
Need, A. C. & Goldstein, D. B. Next generation disparities in human genomics: concerns and remedies. Trends Genet. 25, 489–494 (2009).
Popejoy, A. B. & Fullerton, S. M. Genomics is failing on diversity. Nature 538, 161–164 (2016).
Gaziano, J. M. et al. Million Veteran Program: a mega-biobank to study genetic influences on health and disease. J. Clin. Epidemiol. 70, 214–223 (2016).
Xu, H. et al. A genome-wide association study of idiopathic dilated cardiomyopathy in African Americans. J. Pers. Med. 8, 11 (2018).
Jurgens, S. J. et al. Genome-wide association study reveals mechanisms underlying dilated cardiomyopathy and myocardial resilience. Nat. Genet. 56, 2636–2645 (2024).
Zheng, S. L. et al. Genome-wide association analysis provides insights into the molecular etiology of dilated cardiomyopathy. Nat. Genet. 56, 2646–2658 (2024).
GTEx Consortium The Genotype-Tissue Expression (GTEx) project. Nat. Genet. 45, 580–585 (2013).
The ARIC Investigators The Atherosclerosis Risk in Communities (ARIC) Study: design and objectives. Am. J. Epidemiol. 129, 687–702 (1989).
Zhang, J. et al. Plasma proteome analyses in individuals of European and African ancestry identify cis-pQTLs and models for proteome-wide association studies. Nat. Genet. 54, 593–602 (2022).
Phan, L. et al. The evolution of dbSNP: 25 years of impact in genomic research. Nucleic Acids Res. 53, D925–D931 (2025).
Park, J. et al. A genome-first approach to aggregating rare genetic variants in LMNA for association with electronic health record phenotypes. Genet. Med. 22, 102–111 (2020).
Bycroft, C. et al. The UK Biobank resource with deep phenotyping and genomic data. Nature 562, 203–209 (2018).
Bild, D. E. et al. Multi-Ethnic study of atherosclerosis: objectives and design. Am. J. Epidemiol. 156, 871–881 (2002).
Taylor, H. A. et al. Toward resolution of cardiovascular health disparities in African Americans: design and methods of the Jackson Heart Study. Ethn. Dis. 15, 4–17 (2005).
Ockenhouse, C. F., Tandon, N. N., Magowan, C., Jamieson, G. A. & Chulay, J. D. Identification of a platelet membrane glycoprotein as a falciparum malaria sequestration receptor. Science 243, 1469–1471 (1989).
Baruch, D. I., Ma, X. C., Pasloske, B., Howard, R. J. & Miller, L. H. CD36 peptides that block cytoadherence define the CD36 binding region for Plasmodium falciparum-infected erythrocytes. Blood 94, 2121–2127 (1999).
Aitman, T. J. et al. Malaria susceptibility and CD36 mutation. Nature 405, 1015–1016 (2000).
Fry, A. E. et al. Positive selection of a CD36 nonsense variant in sub-Saharan Africa, but no association with severe malaria phenotypes. Hum. Mol. Genet. 18, 2683–2692 (2009).
Pain, A. et al. A non-sense mutation in Cd36 gene is associated with protection from severe malaria. Lancet 357, 1502–1503 (2001).
Sepúlveda, N. et al. Malaria host candidate genes validated by association with current, recent, and historical measures of transmission intensity. J. Infect. Dis. 216, 45–54 (2017).
Genovese, G. et al. Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science 329, 841–845 (2010).
Martin, A. R. et al. Clinical use of current polygenic risk scores may exacerbate health disparities. Nat. Genet. 51, 584–591 (2019).
Hamo, C. E. et al. Heart failure risk associated with severity of modifiable heart failure risk factors: the ARIC Study. J. Am. Heart Assoc. 11, e021583 (2022).
Madan, N. et al. Functionalization of CD36 cardiovascular disease and expression associated variants by interdisciplinary high throughput analysis. PLoS Genet. 15, e1008287 (2019).
Baldassari, A. R. et al. Multi-ethnic genome-wide association study of decomposed cardioelectric phenotypes illustrates strategies to identify and characterize evidence of shared genetic effects for complex traits. Circ. Genom. Precis. Med. 13, e002680 (2020).
Young, W. J. et al. Genetic analyses of the electrocardiographic QT interval and its components identify additional loci and pathways. Nat. Commun. 13, 5144 (2022).
Murashige, D. et al. Comprehensive quantification of fuel use by the failing and nonfailing human heart. Science 370, 364–368 (2020).
Coburn, C. T. et al. Defective uptake and utilization of long chain fatty acids in muscle and adipose tissues of CD36 knockout mice. J. Biol. Chem. 275, 32523–32529 (2000).
Tanaka, T. et al. Defect in human myocardial long-chain fatty acid uptake is caused by FAT/CD36 mutations. J. Lipid Res. 42, 751–759 (2001).
Koonen, D. P. Y. et al. CD36 expression contributes to age-induced cardiomyopathy in mice. Circulation 116, 2139–2147 (2007).
Sung, M. M. et al. Cardiomyocyte-specific ablation of CD36 accelerates the progression from compensated cardiac hypertrophy to heart failure. Am. J. Physiol. Heart Circ. Physiol. 312, H552–H560 (2017).
Beadle, R. M. et al. Improvement in cardiac energetics by perhexiline in heart failure due to dilated cardiomyopathy. JACC Heart Fail. 3, 202–211 (2015).
Watson, W. D. et al. Retained metabolic flexibility of the failing human heart. Circulation 148, 109–123 (2023).
Joseph, J. et al. Genetic architecture of heart failure with preserved versus reduced ejection fraction. Nat. Commun. 13, 7753 (2022).
Klarin, D. et al. Genetics of blood lipids among ~300,000 multi-ethnic participants of the Million Veteran Program. Nat. Genet. 50, 1514–1523 (2018).
Hunter-Zinck, H. et al. Genotyping array design and data quality control in the Million Veteran Program. Am. J. Hum. Genet. 106, 535–548 (2020).
Chang, C. C. et al. Second-generation PLINK: rising to the challenge of larger and richer datasets. Gigascience 4, 7 (2015).
Mägi, R. & Morris, A. P. GWAMA: software for genome-wide association meta-analysis. BMC Bioinformatics 11, 288 (2010).
Hinson, J. T. et al. Titin mutations in iPS cells define sarcomere insufficiency as a cause of dilated cardiomyopathy. Science 349, 982–986 (2015).
Acknowledgements
This work would not be possible without the participation of our veteran partners who make up the MVP study cohort, and the tireless work of the MVP administrative, recruitment, local site and core teams. A full list of these MVP investigators can be found in the Supplementary Note. The views expressed in this manuscript are those of the authors and do not necessarily represent the views of the National Heart, Lung, and Blood Institute, the National Institutes of Health, the U.S. Department of Health and Human Services, the U.S. Department of Veterans Affairs or the U.S. Government.
This research is based on data from the MVP, Office of Research and Development, Veterans Health Administration, and was supported by awards I01-CX001737 (to L.S.P.) and I01-BX004821 (to P.W.F.W. and K.C.). Additional funding support came from the Department of Veterans Affairs (I01CX001922 to J.J.), the National Institutes of Health/National Heart, Lung, and Blood Institute (NIH/NHLBI; K08HL153937 to K.G.A.), and the American Heart Association (862032 to K.G.A.).
Whole-genome sequencing for the Trans-Omics in Precision Medicine (TOPMed) program was supported by the NHLBI. Whole-genome sequencing for ‘NHLBI TOPMed: MESA’ (phs001416.v1.p1) was performed at the Broad Institute of MIT and Harvard (3U54HG003067-13S1). Centralized read mapping and genotype calling, along with variant quality metrics and filtering, were provided by the TOPMed Informatics Research Center (3R01HL-117626-02S1). Phenotype harmonization, data management, sample-identity QC and general study coordination were provided by the TOPMed Data Coordinating Center (3R01HL-120393-02S1) and TOPMed MESA Multi-Omics (HHSN2682015000031/HSN26800004). The MESA projects are conducted and supported by the NHLBI in collaboration with MESA investigators. Support for the MESA projects is provided by the NHLBI in collaboration with MESA investigators. Support for MESA is provided by contracts 75N92020D00001, HHSN268201500003I, N01-HC-95159, 75N92020D00005, N01-HC-95160, 75N92020D00002, N01-HC-95161, 75N92020D00003, N01-HC-95162, 75N92020D00006, N01-HC-95163, 75N92020D00004, N01-HC-95164, 75N92020D00007, N01-HC-95165, N01-HC-95166, N01-HC-95167, N01-HC-95168, N01-HC-95169, UL1-TR-000040, UL1-TR-001079, UL1-TR-001420, UL1TR001881, DK063491 and R01HL105756. We thank the other investigators, the staff, and the participants of the MESA study for their valuable contributions. A full list of participating MESA investigators and institutes can be found at http://www.mesa-nhlbi.org. MESA is also supported in part by the NHLBI contract R01HL160793.
Molecular data for the TOPMed program were supported by the NHLBI. Genome sequencing for ‘NHLBI TOPMed: The Jackson Heart Study’ (phs000964.v1.p1) was performed at the Northwest Genomics Center (HHSN268201100037C). Core support, including centralized genomic read mapping and genotype calling, along with variant quality metrics and filtering, were provided by the TOPMed Informatics Research Center (3R01HL-117626-02S1; contract HHSN268201800002I). Core support, including phenotype harmonization, data management, sample-identity QC and general program coordination, were provided by the TOPMed Data Coordinating Center (R01HL-120393; U01HL-120393; contract HHSN268201800001I). We gratefully acknowledge the studies and participants who provided biological samples and data for TOPMed.
The Jackson Heart Study is supported and conducted in collaboration with Jackson State University (HHSN268201800013I), Tougaloo College (HHSN268201800014I), the Mississippi State Department of Health (HHSN268201800015I) and the University of Mississippi Medical Center (HHSN268201800010I, HHSN268201800011I and HHSN268201800012I) contracts from the NHLBI and the National Institute on Minority Health and Health Disparities. We also thank the staff and participants of the Jackson Heart Study.
Author information
Authors and Affiliations
Consortia
Contributions
J.E.H., L.G., Z.R.A.S., Y.V.S., P.T.E., J.J. and K.G.A. were involved in the conception or design of the current study. D.G., Y.V.S., K.C., L.S.P., J.J., P.W.F.W., D.J.R., S.S.R., J.I.R. and J.M.G. were involved in the conception or design of the participating studies. J.E.H., L.G., Z.R.A.S., X.G., V.K., R.L.J., L.M.R., K.J.B., B.C., A.C. and K.L.L. were involved in acquisition, analysis or interpretation of data. J.E.H., L.G. and K.G.A. have drafted the manuscript, and J.E.H., Z.R.A.S., P.T.E., S.M.D. and K.G.A. substantively revised it. M.G.L., Y.-I.M., J.P.P., N.R., R.R., S.S.S.V., B.A.V., A.V., J.Y., J.J.C., J.P.C., J.A.C.L., W.S.P., M.D.R., A.S., K.D.T., J.G.T. and C.J.O. provided critical revision of the manuscript for important intellectual content. All authors have approved the submitted version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
C.J.O. is an employee of the Novartis Institute of Biomedical Research. P.T.E. has received grant support from Bayer AG, Novo-Nordisk, Pfizer and Bristol Myers Squibb, and has served on advisory boards or consulted for Bayer AG, Quest Diagnostics, MyoKardia and Novartis. K.G.A. has received grant support from Sarepta Therapeutics, Bayer AG and Foresite Labs, and reports a research collaboration with the Novartis Institute for Biomedical Research. The other authors declare no competing interests.
Peer review
Peer review information
Nature Genetics thanks Neil Hanchard and Ntobeko Ntusi for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information (download PDF )
Supplementary Note, Supplementary Figs. 1–8 and Supplementary Tables 1, 2, 5, 8, 13 and 14.
Supplementary Tables 3, 4, 6, 7 and 9–12 (download XLSX )
Supplementary Tables 3, 4, 6, 7 and 9–12.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Huffman, J.E., Gaziano, L., Al Sayed, Z.R. et al. An African ancestry-specific nonsense variant in CD36 is associated with a higher risk of dilated cardiomyopathy. Nat Genet 57, 2682–2690 (2025). https://doi.org/10.1038/s41588-025-02372-2
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41588-025-02372-2