Abstract
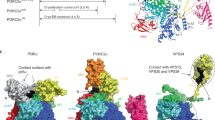
Lipid phosphate phosphatases (LPPs) catalyze the dephosphorylation of a broad range of bioactive lipid phosphates, including lysophosphatidic acid and sphingosine-1-phosphate, playing essential roles in embryonic vasculogenesis, cell differentiation and inflammation. Here we present the cryo-electron microscopic structure of human LPP1 as a tetramer with C4 symmetry. We capture the phosphohistidine intermediate state by using vanadate as a phosphate analog, where vanadate is coordinated by positively charged residues from three conserved motifs (C1, C2 and C3). Structural investigations of LPP1 variants with mutations in two catalytic histidine residues confirm that the histidine in the C2 motif facilitates phosphate bond cleavage. Enzymatic assays validate our structural observations. Additionally, a phosphatidylinositol 4,5-bisphosphate (PIP2) molecule was discovered in the LPP1 structure, underscoring a potential regulatory role for PIP2 in the catalytic activity of LPP1.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The atomic coordinates of LPP1-R, LPP1-C2M, LPP1-C3M, LPP1-V and LPP1-C3M (nanodisc) have been deposited in the Protein Data Bank (http://www.rcsb.org) under accession codes 9L0I, 9L0O, 9L0U, 9L0S and 9VL3. The corresponding electron microscopy map has been deposited in the Electron Microscopy Data Bank (https://www.ebi.ac.uk/pdbe/emdb/) under accession codes EMD-62715, EMD-62720, EMD-62723, EMD-62722 and EMD-65153. Data are available from the corresponding authors upon reasonable request. Source data are provided with this paper.
References
Tang, X. Y., Benesch, M. G. K. & Brindley, D. N. Lipid phosphate phosphatases and their roles in mammalian physiology and pathology. J. Lipid Res. 56, 2048–2060 (2015).
Jamal, Z., Martin, A., Gomez-Munoz, A. & Brindley, D. N. Plasma membrane fractions from rat liver contain a phosphatidate phosphohydrolase distinct from that in the endoplasmic reticulum and cytosol. J. Biol. Chem. 266, 2988–2996 (1991).
Zhang, N., Copeland, N. G., Gilbert, D. J., Jenkins, N. A. & Gridley, T. Cloning, expression, and chromosomal localization of a mouse gene homologous to the germ cell migration regulator wunen and to type 2 phosphatidic acid phosphatases. Genomics 63, 142–144 (2000).
Kai, M., Wada, I., Imai, S., Sakane, F. & Kanoh, H. Cloning and characterization of two human isozymes of Mg2+-independent phosphatidic acid phosphatase. J. Biol. Chem. 272, 24572–24578 (1997).
Hooks, S. B., Ragan, S. P. & Lynch, K. R. Identification of a novel human phosphatidic acid phosphatase type 2 isoform. FEBS Lett. 427, 188–192 (1998).
Roberts, R., Sciorra, V. A. & Morris, A. J. Human type 2 phosphatidic acid phosphohydrolases. Substrate specificity of the type 2a, 2b, and 2c enzymes and cell surface activity of the 2a isoform. J. Biol. Chem. 273, 22059–22067 (1998).
Ren, H. et al. Lipid phosphate phosphatase (LPP3) and vascular development. Biochim. Biophys. Acta 1831, 126–132 (2013).
Zhang, N., Sundberg, J. P. & Gridley, T. Mice mutant for Ppap2c, a homolog of the germ cell migration regulator wunen, are viable and fertile. Genesis 27, 137–140 (2000).
Escalante-Alcalde, D. et al. The lipid phosphatase LPP3 regulates extra-embryonic vasculogenesis and axis patterning. Development 130, 4623–4637 (2003).
Jia, Y. J., Kai, M., Wada, I., Sakane, F. & Kanoh, H. Differential localization of lipid phosphate phosphatases 1 and 3 to cell surface subdomains in polarized MDCK cells. FEBS Lett. 552, 240–246 (2003).
Ishikawa, T., Kai, M., Wada, I. & Kanoh, H. Cell surface activities of the human type 2b phosphatidic acid phosphatase. J. Biochem. 127, 645–651 (2000).
Geraldo, L. H. M. et al. Role of lysophosphatidic acid and its receptors in health and disease: novel therapeutic strategies. Signal Transduct. Target. Ther. 6, 45 (2021).
Bryan, A. M. & Del Poeta, M. Sphingosine-1-phosphate receptors and innate immunity. Cell Microbiol. 20, e12836 (2018).
Van Hoose, P. M. et al. Lipid phosphate phosphatase 3 in smooth muscle cells regulates angiotensin II-induced abdominal aortic aneurysm formation. Sci. Rep. 12, 5664 (2022).
Sanchez-Sanchez, R. et al. Lack of lipid phosphate phosphatase-3 in embryonic stem cells compromises neuronal differentiation and neurite outgrowth. Dev. Dyn. 241, 953–964 (2012).
Tripathi, H. et al. Myeloid-specific deletion of lipid Plpp3 (phosphate phosphatase 3) increases cardiac inflammation after myocardial infarction. Arterioscler. Thromb. Vasc. Biol. 43, 379–381 (2023).
Nema, R., Shrivastava, A. & Kumar, A. Prognostic role of lipid phosphate phosphatases in non-smoker, lung adenocarcinoma patients. Comput. Biol. Med. 129, 104141 (2021).
Tang, X. et al. Lipid phosphate phosphatase-2 promotes tumor growth through increased c-Myc expression. Theranostics 12, 5675–5690 (2022).
Benesch, M. G. K. et al. Decreased lipid phosphate phosphatase 1/3 and increased lipid phosphate phosphatase 2 expression in the human breast cancer tumor microenvironment promotes tumor progression and immune system evasion. Cancers (Basel) 15, 2299 (2023).
Tang, X. et al. Lipid phosphate phosphatase-1 expression in cancer cells attenuates tumor growth and metastasis in mice. J. Lipid Res. 55, 2389–2400 (2014).
Vishwakarma, S. et al. Downregulation of lipid phosphate phosphatase 3 correlates with tumor-infiltrating immune cells in oral cancer. Cureus 14, e23553 (2022).
Zeng, Y., Cao, S., Li, N., Tang, J. & Lin, G. Identification of key lipid metabolism-related genes in Alzheimer’s disease. Lipids Health Dis. 22, 155 (2023).
Smyth, S. S., Kraemer, M., Yang, L., Van Hoose, P. & Morris, A. J. Roles for lysophosphatidic acid signaling in vascular development and disease. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 1865, 158734 (2020).
Jose, A. & Kienesberger, P. C. Autotaxin-LPA-LPP3 axis in energy metabolism and metabolic disease. Int. J. Mol. Sci. 22, 9575 (2021).
Corbi, S. C. T. et al. Expression profile of genes potentially associated with adequate glycemic control in patients with type 2 diabetes mellitus. J. Diabetes Res. 2017, 2180819 (2017).
Tang, X., McMullen, T. P. W. & Brindley, D. N. Increasing the low lipid phosphate phosphatase 1 activity in breast cancer cells decreases transcription by AP-1 and expressions of matrix metalloproteinases and cyclin D1/D3. Theranostics 9, 6129–6142 (2019).
Stukey, J. & Carman, G. M. Identification of a novel phosphatase sequence motif. Protein Sci. 6, 469–472 (1997).
Barila, D. et al. The Dri 42 gene, whose expression is up-regulated during epithelial differentiation, encodes a novel endoplasmic reticulum resident transmembrane protein. J. Biol. Chem. 271, 29928–29936 (1996).
Messerschmidt, A. & Wever, R. X-ray structure of a vanadium-containing enzyme: chloroperoxidase from the fungus Curvularia inaequalis. Proc. Natl Acad. Sci. USA 93, 392–396 (1996).
Ishikawa, K., Mihara, Y., Gondoh, K., Suzuki, E. & Asano, Y. X-ray structures of a novel acid phosphatase from Escherichia blattae and its complex with the transition-state analog molybdate. EMBO J. 19, 2412–2423 (2000).
Sigal, Y. J., McDermott, M. I. & Morris, A. J. Integral membrane lipid phosphatases/phosphotransferases: common structure and diverse functions. Biochem. J. 387, 281–293 (2005).
Fan, J., Jiang, D., Zhao, Y., Liu, J. & Zhang, X. C. Crystal structure of lipid phosphatase Escherichia coli phosphatidylglycerophosphate phosphatase B. Proc. Natl Acad. Sci. USA 111, 7636–7640 (2014).
Long, J. S., Pyne, N. J. & Pyne, S. Lipid phosphate phosphatases form homo- and hetero-oligomers: catalytic competency, subcellular distribution and function. Biochem. J 411, 371–377 (2008).
Vardakou, M., Salmon, M., Faraldos, J. A. & O’Maille, P. E. Comparative analysis and validation of the malachite green assay for the high throughput biochemical characterization of terpene synthases. MethodsX 1, 187–196 (2014).
Zhang, Q. X., Pilquil, C. S., Dewald, J., Berthiaume, L. G. & Brindley, D. N. Identification of structurally important domains of lipid phosphate phosphatase-1: implications for its sites of action. Biochem. J. 345, 181–184 (2000).
Carman, G. M., Deems, R. A. & Dennis, E. A. Lipid signaling enzymes and surface dilution kinetics. J. Biol. Chem. 270, 18711–18714 (1995).
Tunyasuvunakool, K. et al. Highly accurate protein structure prediction for the human proteome. Nature 596, 590–596 (2021).
Simon, M. F. et al. Expression of ectolipid phosphate phosphohydrolases in 3T3F442A preadipocytes and adipocytes. Involvement in the control of lysophosphatidic acid production. J. Biol. Chem. 277, 23131–23136 (2002).
Xia, Z. et al. Structural insights into glucose-6-phosphate recognition and hydrolysis by human G6PC1. Proc. Natl Acad. Sci. USA 122, e2418316122 (2025).
Spiegel, S. & Milstien, S. Sphingosine-1-phosphate: an enigmatic signalling lipid. Nat. Rev. Mol. Cell Biol. 4, 397–407 (2003).
Shirzad-Wasei, N. et al. Rapid transfer of overexpressed integral membrane protein from the host membrane into soluble lipid nanodiscs without previous purification. Biol. Chem. 396, 903–915 (2015).
Ritchie, T. K. et al. Chapter Eleven - Reconstitution of membrane proteins in phospholipid bilayer nanodiscs. Methods Enzymol. 464, 211–231 (2009).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Grant, T. & Grigorieff, N. Measuring the optimal exposure for single particle cryo-EM using a 2.6 Å reconstruction of rotavirus VP6. eLife 4, e06980 (2015).
Punjani, A., Rubinstein, J. L., Fleet, D. J. & Brubaker, M. A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods 14, 290–296 (2017).
Wang, N. et al. Structural basis of human monocarboxylate transporter 1 inhibition by anti-cancer drug candidates. Cell 184, 370–383 (2021).
Rosenthal, P. B. & Henderson, R. Optimal determination of particle orientation, absolute hand, and contrast loss in single-particle electron cryomicroscopy. J. Mol. Biol. 333, 721–745 (2003).
Chen, S. et al. High-resolution noise substitution to measure overfitting and validate resolution in 3D structure determination by single particle electron cryomicroscopy. Ultramicroscopy 135, 24–35 (2013).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Acknowledgements
We are grateful to the Cryo-EM Center of the University of Science and Technology of China for EM facility support and to the staff members at the Cryo-EM Center for their technical support in cryo-EM data collection. This work was supported by the National Key R&D Program of China (2024YFA1307900 to H.Q.), the National Natural Science Foundation of China (32271241 to H.Q.), the Fundamental Research Funds for the Central Universities (WK9100000031 to H.Q.), the ‘Talent Fund Project of Biomedical Sciences and Health Laboratory of Anhui Province, University of Science and Technology of China’ (BJ9100000003 to H.Q.), start-up funding from the University of Science and Technology of China (KY9100000034 and KJ2070000082 to H.Q.), the Anhui Provincial Health and Wellness Scientific Research Projects (AHWJ2023BAc20032 to C.S.) and the Young Scientific and Technological Talent Development Program of Anhui Medical University Affiliated Hospital (2023cy022 to C.S.).
Author information
Authors and Affiliations
Contributions
H.Q. conceived the project and designed the experiments. M.Y. and C.S. performed cloning and protein purification. M.Y. prepared the cryo-EM samples and collected the data. H.Q. determined the structures. M.Y. performed the enzymatic assay. All authors contributed to the data analysis and preparation of the manuscript. H.Q., M.Y., Y.H. and C.S. prepared the figures, and H.Q. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Peer review
Peer review information
Nature Chemical Biology thanks David N. Brindley, Daohua Jiang and the other, anonymous, reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Purification and functional validation of human LPP1.
a, Representative size exclusion chromatography (SEC) profile of human LPP1 (hLPP1) solubilized with 0.005% LMNG. The peak fractions (15.5–17.5 ml) were concentrated for cryo-EM sample preparation. Inset: SDS‒PAGE analysis of the concentrated protein used for cryo-EM sample preparation. b, Schematic diagram of our enzymatic assay for hLPP1. The release of the phosphate group was detected using a modified malachite green-ammonium molybdate method, which forms a molybdophosphoric acid complex with absorption at 630 nm. This image was created with BioRender.com (https://BioRender.com/). c, Phosphate release catalyzed by wild-type (WT) and mutated LPP1 using phosphatidic acid (PA), lysophosphatidic acid (LPA), and sphingosine-1-phosphate (S1P) as substrates. Mutations at His171 and His223, essential catalytic residues, result in almost loss of catalytic activities. In this experiment, the reaction mixtures for all three substrates were incubated at 37 °C for 20 min. Data are presented as mean values ± s.d. of three independent experiments, each with two technical repeats (n=6). d, Initial rates of phosphate release, catalyzed by purified WT hLPP1, were measured at varying concentrations of PA and S1P. The enzyme was analyzed with the surface dilution kinetic model, and the apparent Km and Vmax values are indicated above the curves. Data are presented as means ± s.d. for three technical repeats (n=3). e, A representative micrograph and 2D class averages of cryo-EM samples of LPP1 in LMNG micelles. Similar images were obtained across all five datasets, resulting in the acquisition of over 5,000 micrographs. The box size for 2D averages is approximately 210 Å. The experiments in a, c, and d were repeated more than three times with similar results.
Extended Data Fig. 2 Flowchart for structural determination.
a, Flowchart of data processing. See Methods for details. b, c, Gold standard Fourier shell correlation (GSFSC) curves (b) and angular distribution maps (c) for the final reconstructions. d, Local resolution maps for the final reconstructions. The maps were calculated using cryoSPARC and generated with Chimera. The unit for resolutions is Å.
Extended Data Fig. 3 EM maps of representative structural elements in LPP1.
a, Membrane topology of LPP1 based on our structure. The C1, C2, and C3 motifs are distinctly colored brown, deep blue-green, and marine, respectively, and a glycosylation site is marked with an asterisk. This image was created with BioRender.com (https://BioRender.com/). b, EM maps of representative structural elements. The EM maps, contoured at 5σ, are generated based on the LPP1-R structure. c, Schematic elucidation of the coordination of PAE and PAI at the tetrameric interface. The residues from different protomers are distinguished by light blue and wheat words. The figures were prepared with LigPlot+. d, SEC profiles of enzymes with mutations related to tetramerization. e, Functional verification of the importance of the residues that engage in tetramerization using PA and S1P as substrates, respectively. Data are presented as the means ± s.d. of three independent experiments, each with two technical repeats (n=6). Significant differences were determined with one-way ANOVA with Tukey’s multiple comparisons tests. f, SDS-PAGE analysis of LPP1 variants in panel e and Fig. 2i. CB, Coomassie blue staining; WB, immunoblotting analysis with Strep antibody.
Extended Data Fig. 4 Isolation and functional validation of LPP heterooligomers.
a, Sequence alignment of human LPP1, LPP2, and LPP3 highlights the conservation of essential residues involved in tetramerization and the coordination of PAE, PAI, and PIP2 lipids. These residues are indicated by blue squares, purple circles, red circles, and red triangles, respectively. The C1, C2, and C3 loops are marked with brown, deep blue-green, and marine boxes, respectively. b, Structural overlay of the LPP1 structure with the Alphafold2-predicted models of LPP2 and LPP3. The Alphafold2-predicted models were downloaded from the UniProt database (https://www.uniprot.org/). c, Immunoblotting analysis demonstrated successful isolation of LPP1-LPP2, LPP1-LPP3, and LPP2-LPP3 heterooligomers. The representative blots shown here are from three independent experiments. Please refer to the Methods section for details. d, SEC analysis of these heterooligomers suggests that they exhibit similar oligomeric states. e, Functional assays confirmed that these LPP heterooligomers retain the ability to dephosphorylate LPA, PA, and S1P. In this experiment, the enzymatic systems were incubated at 37 °C for 20 min prior to quantification using the malachite green-ammonium molybdate method. Data are presented as the means ± s.d. of three independent experiments, each with two technical repeats (n=6).
Extended Data Fig. 5 Structural comparison of LPP1 structures and E. coli PgpB.
a, Two opposite views illustrating the superimposition of hLPP1 with E. coli PgpB (PDB code: 4PX7). The transmembrane segments TM2-6 of LPP1 align perfectly with those of PgpB. However, LPP1-TM1 is positioned opposite to that of PgpB. b, When superimposing hLPP1 with E. coli PgpB, PgpB-TM1 conflicts with the adjacent protomer in the LPP1 tetramer. c, SEC profiles of enzymes with mutations on essential residues involved in vanadate coordination. d, Comparison of the extra densities within the catalytic cavities of LPP1-C2M and LPP1-C3M. The extra densities are contoured at 3σ, 4σ, and 6σ, respectively. Notably, the phosphate head group is not connected with the remaining part when modeling LPA into the extra density in LPP1-C3M. This observation suggests that LPA molecules may have been hydrolyzed into PO4 and MAG in LPP1-C3M. e, EM maps for PO4 and MAG molecules in the LPP1-C3M structure. The extra densities are contoured at 5σ. f, SEC profiles of enzymes with mutations related to the coordination of MAG. g, Functional verification of the essential residues involved in MAG coordination using PA and S1P as substrates. Bottom, SDS-PAGE analysis of these LPP1 variants. CB, Coomassie blue; WB, immunoblotting analysis with Strep antibody. Data are presented as the mean ± s.d. of three independent experiments, each with two technical repeats (n=6). Significant differences were determined with one-way ANOVA with Tukey’s multiple comparisons tests.
Extended Data Fig. 6 LMNG-like densities in the hydrophobic pocket.
a, Functional verification of His31 and Lys161 residues using LPA, PA and S1P as substrates. Data are presented as the means ± s.d. of three independent experiments, each with two technical repeats (n=6). Significant differences were determined with one-way ANOVA with Tukey’s multiple comparisons tests. b, SEC profiles of enzymes with H31A and K161A mutations. c, Some extra densities are observed within a hydrophobic pocket in the vicinity of the catalytic cavity in all four structures. The catalytic and hydrophobic cavities are marked with yellow and red ellipses, respectively. The extra densities in the pocket are contoured at 4σ. Notably, these extra densities in LPP1-C3M align well with an LMNG molecule. d, EM maps of the LMNG molecule are contoured at 3σ. The LMNG molecules are shown as white sticks and balls. e, SEC profiles of LPP1-C3M reconstituted into nanodiscs. The peak fractions were concentrated for cryo-EM sample preparation. Inset: SDS‒PAGE analysis of the concentrated protein used for cryo-EM sample preparation. f, Map overlay of LPP1-C3M structures in detergent and nanodisc environments. Insets: Zoomed-in views to show the extra densities at the catalytic cavities of LPP1-C3M in detergent and nanodisc environments. Both MAG and LMNG molecules are absent in the LPP1-C3M structure in the nanodisc.
Extended Data Fig. 7 PIP2 binding is required for LPP1 function.
a, EM maps of PIP2 molecules in these four structures. The densities are contoured at 5σ in the LPP1-R, LPP1-C2M, and LPP1-C3M structures, and at 3σ in the LPP1-C3M structure. The PIP2 molecules are represented as black sticks and balls. b, SEC profiles of enzymes with mutations on essential residues involved in PIP2 coordination. c, Functional verification of essential residues involved in PIP2 coordination using PA and S1P as substrates. Data are presented as the means ± s.d. of three independent experiments, each with two technical repeats (n=6). Significant differences were determined with one-way ANOVA with Tukey’s multiple comparisons tests. d, SDS-PAGE analysis of LPP1 variants in panel c and Fig. 6e. CB, Coomassie blue staining; WB, immunoblotting analysis with Strep antibody. The representative blots shown here are from more than three independent experiments. e, Mechanistic model of LPP1-mediated dephosphorylation of LPA. LPP1 catalyzes the dephosphorylation of LPA via a ‘ping-pong’ mechanism. Step 1: the C2 histidine (His171) facilitates the cleavage of the phosphate bond, and the C3 histidine acts as a nucleophile to form a phosphohistidine intermediate with the cleaved phosphate. Subsequently, the first product, MAG, is released. Step 2: a water molecule enters the catalytic cavity and hydrolyzes the phosphohistidine intermediate, resulting in the release of the 2nd product, inorganic phosphate. The structures we present here likely capture the major intermediate states of Step 1, from substrate binding to the formation of the phosphohistidine intermediate.
Extended Data Fig. 8 S1P hydrolysis is sensitive to interprotomer coupling.
a, Some conserved residues in the C1, C2, and C3 loops participate in tetramerization and coordinating PAE molecules. In LPP1-A, the catalytic cavity is marked with a dotted circle, and the interfaces with LPP1-B and LPP1-D are indicated with black dotted lines. b, The chemical structures of LPA and S1P. The major differences are indicated with red dotted ellipses. c, The SEC profiles for the purification of the LPP1-WT&H223A chimera. Inset: Immunoblotting analysis demonstrates successful isolation of WT&H223A chimera. These experiments were conducted at least three times and the results shown here are the representative of those trials. d, Functional examination of the ability of the WT&H223A chimera to hydrolyze LPA, PA, and S1P. The WT&H223A chimera exhibits normal activities to hydrolyze LPA and PA but an obvious reduction in the dephosphorylation of S1P. These biochemical characterizations suggest that S1P hydrolysis is coupled between adjacent protomers, whereas LPA and PA hydrolysis are more independent for each protomer. WT+H223A, a mixture of WT and H223A enzymes after purification. In this experiment, 0.01 µg of enzymes was used to dephosphorylate LPA and PA at 37 °C for 10 min, whereas 0.03 µg of enzymes was employed to hydrolyze S1P at 37 °C for 20 min due to the relatively lower activity of the enzymes. Data are presented as the means ± s.d. of three independent experiments, each with two technical repeats (n=6).
Extended Data Fig. 9 Structural comparison between LPP1-C3M and G6PC1.
a, Two perpendicular views illustrate the structural superimposition of LPP1-C3M and G6PC1 (PDB: 9J7U). Inset: Zoomed-in views highlighting key essential elements within the catalytic cavities. b, Separate views displaying the electrostatic surface potentials of LPP1-C3M and G6PC1. The middle section presents zoomed-in views of the catalytic cavities of LPP1-C3M and G6PC1. The clashes between G6P and the LPP1-C3M cavity are indicated. The G6PC1 cavity lacks sufficient space to accommodate the hydrophobic tails of the LPP1 substrates. c, Functional assays indicate that LPP1 exhibits low activity in dephosphorylation of glucose 6-phosphate (G6P). Data are presented as the means ± s.d. of three independent experiments, each with two technical repeats (n=6).
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1 and 2
Supplementary Data 1 (download XLSX )
The source data used to determine the Km and Vmax values are listed in Supplementary Table 1.
Supplementary Data 2 (download XLSX )
The sequences of primers used in this study.
Supplementary Video 1 (download MP4 )
The conformational changes of the catalytic cavity. The movie was generated based on the structures of LPP1-R, LPP1-C2M, LPP1-C3M and LPP1-V. To ensure seamless transition in the movie, the alanine substitutions at histidine residues in the C2M (H171A) and C3M (H223A) structures were reverted to histidine.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download PDF )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Unprocessed western blots and gels.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 3 (download PDF )
Unprocessed western blots and gels.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download PDF )
Unprocessed western blots.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 5 (download PDF )
Unprocessed western blots and gels.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download PDF )
Unprocessed western blots and gels.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 8 (download PDF )
Unprocessed western blots.
Source Data Extended Data Fig. 9 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, M., Sun, C., He, Y. et al. Structural basis for the catalytic mechanism of human lipid phosphate phosphatases. Nat Chem Biol (2026). https://doi.org/10.1038/s41589-025-02121-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41589-025-02121-w