Abstract
Climate change has severe and wide-ranging health impacts, especially for vulnerable groups. Despite growing evidence of heat-associated adverse maternal and neonatal health outcomes, there remains a lack of synthesis quantifying associations and identifying specific risk periods. We systematically reviewed the literature on heat impacts on maternal, fetal and neonatal health and quantified impacts through meta-analyses. We found 198 studies across 66 countries, predominantly high income (63.3%) and temperate climate zones (40.1%), and 23 outcomes. Results showed increased odds of preterm birth of 1.04 (95% confidence interval (CI) = 1.03, 1.06; n = 12) per 1 °C increase in heat exposure and 1.26 (95% CI = 1.08, 1.47; n = 10) during heat waves. Similarly, high heat exposure increased the risk for stillbirths (odds ratio (OR) = 1.13 (95% CI = 0.95, 1.34; n = 9)), congenital anomalies (OR = 1.48 (95% CI = 1.16, 1.88; n = 6)) and gestational diabetes mellitus (OR = 1.28 (95% CI = 1.05, 1.74; n = 4)). The odds of any obstetric complication increased by 1.25 (95% CI = 1.09, 1.42; n = 11) during heat waves. Patterns in susceptibility windows varied by condition. The findings were limited by heterogeneity in exposure metrics and study designs. The systematic review demonstrated that escalating heat exposure poses a major threat to maternal and neonatal health, highlighting research priorities, guiding the selection and monitoring of heat-health indicators and emphasizing the need to prioritize maternal and neonatal health in national climate health programs.
Similar content being viewed by others
Main
The impacts of climate change on health are increasingly evident. Record high year-on-year temperatures pose major threats to vulnerable populations such as pregnant women and newborns1,2. Increases in temperature and extreme weather events are pushing both natural and human systems toward critical and irreversible tipping points3.
Anatomical and physiological changes during pregnancy alter thermoregulatory responses4,5. Increased cardiac output and plasma volume, coupled with heat generated by fetal metabolism and additional fat deposits, collectively heighten the vulnerability of pregnant women to heat exposure, especially in the later stages of pregnancy4,6,7. Increased vasodilation and sweating assist in dissipating heat and maintaining a thermal gradient for heat loss from the fetus, but these responses are dampened if the mother has hypertension, hemorrhage, sepsis or other underlying medical conditions4,5.
Heat-related risks during pregnancy include preterm birth8, stillbirth8,9, congenital anomalies10, small for gestational age neonates11, gestational diabetes mellitus6, premature rupture of membranes6, increased hypertensive disorders12 and cardiovascular events13 during labor. These conditions not only affect pregnancy outcomes but also have long-term implications for both the mother and the child14. However, knowledge gaps remain, particularly in the quantification of risk, the breadth of heat-sensitive outcomes, the critical periods for susceptibility, risk factors for heat vulnerability, heat thresholds and biological pathways6.
We aim to systematically review the literature and conduct meta-analyses to quantify the impacts of heat exposure on maternal, fetal and neonatal health globally, identify windows of susceptibility and grade the certainty of evidence. By integrating maternal, fetal and neonatal outcomes into a single systematic review, we provide a comprehensive evidence base that enhances the utility of our findings for policymakers and public health officials. Quantifying the current burden of heat-related health impacts allows researchers to project future health and economic burdens under different scenarios of climate change. Understanding which outcomes and populations are at risk, as well as the timing of risk can assist in establishing surveillance systems, developing targeted public health interventions, evaluating adaptation effectiveness and informing policy decisions that protect pregnant women and neonates from the escalating risks of heat. Any additional risk for adverse maternal and neonatal health outcomes from heat exposure will have major implications for public health and the socioeconomic conditions of affected individuals and communities.
Results
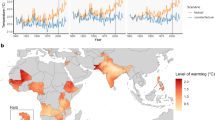
We identified 19,587 records for screening, of which 198 were included in this systematic review (Fig. 1). These studies span 66 countries and six continents (Table 1), with 84.9% published since 2010 (Extended Data Fig. 1). While the majority of studies focused on high-income countries (63.3%) and temperate climate zones (40.1%), evidence covers a range of climates and regions (Fig. 2). Only 3% of studies were from low-income countries. A summary-of-findings table and the 198 study references are available in Supplementary Table 1 and Supplementary Note 1.
Adapted Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart representing the study selection process from 2019 and 2023 searches. *2019 search identification as reflected in Chersich et al.8.
Distribution of 198 studies by country (a) and annual mean temperature (b).
Summary of effects
In total, 271 direction-of-effect estimates (positive, negative, not reported) and 221 effect estimates (risk ratios, odds ratios, hazard ratios) were extracted across 23 outcomes. The most studied outcomes were preterm birth (n = 84), accounting for approximately one-third of the total, low birth weight (n = 51), hypertensive disorders in pregnancy (n = 28), congenital anomalies (n = 22) and stillbirths (n = 19). Studies commonly used data from birth registers, hospital registers and existing cohorts. The direction of effect was predominantly harmful, with opposing evidence in some instances, especially low birth weight, hypertensive disorders in pregnancy and congenital anomalies, for which findings were more heterogeneous (Fig. 3).
The 271 direction-of-effect estimates as a result of heat exposure in pregnancy per outcome category using Global Alignment of Immunization safety Assessment in pregnancy (GAIA) preferred terminology (vote counting).
From the 221 effect estimates, 56 estimates quantified the impact of a temperature increase of 1 °C, 35 addressed the effects of heat wave exposure and 130 compared the health outcomes under conditions of high versus low heat exposure. Fig. 4 shows a summary of all exposure–outcome effect estimates. All effect estimates lie to the right of null, indicating increased risk, with the majority ranging between 1 and 1.5; demonstrating a consistent increase in risk across varied exposure–outcome metrics.
Forest plot representing a summary of effect estimates (n = 221) for each exposure–outcome group. Exposure–outcome groups are based on GAIA preferred terminology, heat exposure metrics and exposure lags. Summary effect estimates (SEE) are represented by the median odds and the interquartile range, while the effect estimates for the meta-analysis are represented by the average odds and a 95% CI. For all meta-analyses conducted, a random-effect model was used, and the I2 result is available as a percentage. MA, meta-analysis; *Basu et al.82 excluded due to duplication of data and effect estimate.
Maternal health
Heat exposure in pregnancy was associated with several adverse maternal outcomes. Hypertensive disorders of pregnancy, such as pre-eclampsia and gestational hypertension, were linked with heat exposure in 21 of the 28 studies. One study conducted in a Chinese cohort of over 2 million pregnancies showed consistent and significantly higher odds of hypertensive disorders of pregnancy with increasing heat exposure in the first and early second trimester, with a clear dose–response effect (OR = 1.16 (95% CI = 1.10, 1.22))15. Six studies found significant heat impacts on increased hypertensive disorders of pregnancy risk in the first 20 weeks, with studies predominantly investigating the month of conception15,16,17,18,19,20, while three studies with a negative association measured heat exposure after 20 weeks or in the month of childbirth and found a decrease in blood pressure with higher temperatures21,22,23.
All nine studies on gestational diabetes mellitus found a harmful association with heat exposure. A large study from Canada (n = 555,911) found that an increase in mean temperature of 10 °C in the past 30 days was associated with increased odds of gestational diabetes mellitus (OR = 1.06 (95% CI = 1.05, 1.07))24. In another large study in Taiwan (n = 371,131), even higher odds per 1 °C increase in heat exposure were reported (OR = 1.54 (95% CI = 1.48, 1.60)) when temperatures exceeded a threshold of 28 °C (ref. 25). Five of the nine studies found associations between heat exposure and increased gestational diabetes mellitus in the second trimester, around the time screening typically occurs24,26,27,28,29.
Heat exposure was associated with increased risk of all-cause antenatal bleeding (n = 3) and placental abruption (n = 2), with short-term effects particularly evident30,31,32. Rammah et al.31 found increased odds of placental abruption per 1 °C increase in apparent temperature at a 1-day lag (OR = 1.16 (95% CI = 1.03, 1.40)). An additional maternal outcome linked to heat exposure was prelabor rupture of membranes with an odds ratio of 1.63 at higher heat exposure (95% CI = 1.23, 2.16) (n = 5). Infections (n = 4), specifically group B streptococcal colonization33 and bacteriuria34, illustrated a harmful association with mean monthly temperature.
Mental health was assessed (n = 2) by screening for emotional stress, where a U-shaped association with temperature was demonstrated35, and, secondly, by admissions for any mental illness, where a positive, but non-significant association was found30.
Exposure to heat, and especially heat waves, was associated with increased all-cause maternal admissions from emergency visits and hospital admissions across all five studies examined. Two studies found evidence of a dose–response effect; more extreme and longer-lasting heat waves were associated with increasing odds of emergency visits and admissions30,36. Lastly, a positive but non-significant association was found between heat exposure and delivery by cesarean section (n = 1)37 and between increases in temperature and increased risk of cardiovascular events (including stroke and cardiac arrest) during the week before childbirth (n = 1)38.
Fetal and perinatal health
Heat exposure during pregnancy was associated with adverse fetal and perinatal outcomes, particularly stillbirths (n = 19), where harmful associations were found in over 10 million stillbirths. A meta-analysis of five USA-based studies indicated an increase in the odds of stillbirth for every 1 °C increase (OR = 1.14 (95% CI = 0.99, 1.32; I2 = 93%; n = 5)) (Extended Data Fig. 2a). This was similar to the increase in odds ratio in studies comparing high versus low heat exposure (OR = 1.13 (95% CI = 0.95, 1.34; I2 = 83%; n = 9)) (Extended Data Fig. 2b); however, there is asymmetry in the funnel plot, which may be suggestive of publication bias (Supplementary Note 2).
Congenital anomalies demonstrated a notable but more heterogeneous association with high temperatures, where five of the 22 effect estimates demonstrated a protective association with heat. The study with the largest sample size of over 2 million women and reporting 29,000 anomalies, conducted in the USA, found a dose–response effect with extreme heat (above the 95th percentile) associated with increased odds of total congenital anomalies (OR = 1.29 (95% CI = 1.21, 1.38))39. Most studies detected impacts of heat with exposure during the first few weeks of pregnancy40,41,42, while one study found no association when temperature was examined in the whole first trimester43. Meta-analysis assessing the impact of high versus low heat exposure on the odds of any congenital anomaly found an increased risk (OR = 1.48 (95% CI = 1.16, 1.88; I2 = 17%; n = 6)) (Extended Data Fig. 3), with a symmetrical funnel plot (Supplementary Note 2).
For spontaneous abortion (n = 7), despite most studies having indicated a harmful direction of effect, the evidence lacked consistency, with five studies yielding no statistically significant association for spontaneous abortion30,44,45,46,47, and one study exhibited an inverse U-shaped association48.
Additional findings included significant impacts of heat on conditions such as oligohydramnios, observed in two studies with relatively small sample sizes but with agreement on a short (up to 4 d) lagged effect49,50. Non-reassuring fetal status, encompassing outcomes like fetal hypoxia and fetal growth restriction, consistently presented harmful associations with heat (n = 5). In The Gambia, for example, increased fetal strain (heart rate greater than 160 beats per minute and increased umbilical artery resistance) was associated with a 1.12 increased odds for each 1 °C increase in heat exposure (95% CI = 1.03, 1.21)51. Relative risk (RR) of perinatal death increased by approximately 50% with exposure to temperature above the 95th percentile in Spain (RR = 1.53 (95% CI = 1.16, 2.02))52, while a study on the same topic in Sweden using data from 1800 to 1895 had similar findings, although this was not significant53. One study found reduced placental weight and volume with high temperature exposure in the third trimester54.
Neonatal health
Neonatal outcomes were affected by heat exposure, especially preterm birth, which is the most extensively studied condition identified, with 78 of the 84 effect estimates reporting a harmful direction of effect. A study with the largest sample size, analyzing 56 million USA births, identified a reduction in gestation with exposure to extreme heat days, with an estimated annual loss of 150,000 gestation days and 25,000 infants born earlier55. Overall, a compelling body of evidence across diverse geographical locations and populations established a dose–response relationship, in which risks for preterm birth rose in tandem with increases in heat exposure54,56,57,58,59,60.
In our meta-analysis (Fig. 5), the odds of a preterm birth increased by 4% (OR = 1.04 (95% CI = 1.03, 1.06; I2 = 85%; n = 12)) per 1 °C increase in temperature across all the study locations with an exposure lag of less than 4 weeks (short lag). While there is substantial heterogeneity across these studies, all the effect estimates had the same direction of effect and the funnel plot is symmetrical (Supplementary Note 2). Similarly, with heat wave exposure, the odds of preterm birth increased by 26% in our meta-analysis (OR = 1.26 (95% CI = 1.08, 1.47; I2 = 63%; n = 10)) (Fig. 5). When comparing high versus low heat exposure, the odds of preterm birth across all studies was 1.12 at higher heat exposure with a short lag (95% CI = 1.06, 1.18; I2 = 92%; n = 39) (Extended Data Fig. 4). There was substantial heterogeneity in this meta-analysis, with two studies that had a protective direction of effect and an asymmetrical funnel plot (Supplementary Note 2)
Meta-analysis of odds of preterm birth with heat exposure in the preceding 4 weeks. a–c, Per 1 °C increase in temperature (n = 12 studies) (a); heat wave (n = 10 studies) (b); subgroup meta-analysis at high versus low heat exposure, split by country income level (n = 39 studies) (c). Each line of the forest plot represents an odds, risk or hazard ratio from a study, represented by the vertical line, and the 95% CI, represented by the horizontal line. The overall OR and 95% CI, using a random-effect meta-analysis model, is represented by the diamond. The heterogeneity is illustrated by the I2 percentage at the bottom left of the figure. HK, Hartung–Knapp method.
When exploring windows of susceptibility, we saw varying evidence across the literature, possibly suggesting that more than one vulnerable exposure window may be present for risk of preterm birth. Studies of heat exposure with a longer lag period (an exposure to high temperatures more than 4 weeks prior) were larger in effect size than with those with short exposure61,62,63,64,65. Specifically, in the meta-analysis, the odds of preterm birth were 1.37 times higher (95% CI = 1.08, 1.74; I2 = 98%; n = 15) at higher heat exposure than those at the lower heat exposure for a long lag (Extended Data Fig. 5).
Subgroup analyses showed that the impacts of heat exposure on preterm birth risk varied by country income level, with the highest risk in low-income countries (OR = 1.61 (95% CI = 1.39, 1.86)) compared to upper–middle-income countries (OR = 1.10 (95% CI = 1.00, 1.21)) and high-income countries (OR = 1.11 (1.06, 1.15)) (Fig. 5) at high versus low heat exposures. For analysis by climate zone, most studies classified as multiple climates, yielding results that lacked actionable insights. Additionally, other subgroup analyses (mean temperature above or below 20 °C, latitude and carbon emissions) were underpowered, failing to detect any statistically significant differences.
In addition to studies presenting odds or risk, several studies quantified the number of preterm births associated with heat and preterm births attributed to climate change and quantified the economic burden. In Australia, an excess of 11 (95% CI = 9, 13) per 10,000 live-born infants were preterm due to immediate heat stress and an excess of 36 (95% CI = 29, 43) per 10,000 preterm births were due to cumulative (lag of 0–6 d) heat stress66. One attribution study and economic evaluation of heat wave-related preterm births due to anthropogenic climate change in China found an average of 4,609 (95% CI = 711, 6,110) preterm births attributed to climate change annually67. The total economic costs of human capital losses caused by anthropogenic climate change on preterm birth are expected to exceed $1 billion annually in China67.
Heat exposure was associated with increased odds of low birth weight; however, there was heterogeneity in this outcome, with six of the 51 effect estimates with a protective effect. The largest study, based on 34.7 million births in the USA68, found that every additional day with mean temperature between 26.7 and 32.2 °C in the preceding 9 months was associated with increased odds of very low birth weight by 0.008 per 1,000 (0.1% of mean, P < 0.5), particularly among Black and Hispanic mothers. Meta-analysis found odds of 1.29 (95% CI = 1.04, 1.59; I2 = 95%; n = 13) of low birth weight at higher versus lower heat exposure (Extended Data Fig. 6).
Additional neonatal outcomes assessed were small for gestational age neonates (n = 6), neonatal admissions (n = 4), neonatal morbidity (n = 6) (jaundice, respiratory distress syndrome, low Apgar scores) and neonatal death (n = 1), which were all adversely associated with heat exposures. Neonatal admissions were at higher odds with exposure to heat waves, from 1.33 (95% CI = 1.27 to 1.38) in Brazil to 1.43 (95% CI = 1.92, 1.88) in India36,69.
Composite outcomes
We derived five composite outcome groups from the data:
-
1.
Pregnancy-specific medical disorders: gestational diabetes mellitus, hypertensive disorders of pregnancy and cardiovascular disease related to pregnancy
-
2.
Obstetric complications: antenatal bleeding, preterm birth, prelabor rupture of membranes
-
3.
Pregnancy loss: spontaneous abortion, stillbirths
-
4.
Fetal growth effects: intrauterine growth restriction, small size for gestational age, low birth weight
-
5.
Healthcare system burden: cesarean section, maternal and neonatal admissions.
In a meta-analysis, the average odds of obstetric complications were 1.05 (95% CI = 1.03, 1.06; I2 = 92%; n = 14) for every 1 °C increase in temperature and 1.25 (95% CI = 1.09, 1.42; I2 = 59%; n = 11) with exposure to a heat wave (Extended Data Fig. 7). The other composite outcomes were not suitable for meta-analyses due to the extent of heterogeneity.
Certainty grading
Using an adapted approach to the International Panel on Climate Change (IPCC) confidence assessment, the evidence scores of type, quantity, quality and consistency across reported outcomes are shown in Extended Data Table 1. For quantity, the outcomes that have the most evidence are for preterm birth, low birth weight, hypertensive disorders of pregnancy, congenital anomalies and stillbirths. Most studies have consistent results, apart from the body of evidence on congenital anomalies and hypertensive disorders of pregnancy that scored poorly. Accounting for levels of agreement and evidence, the outcome with a very high certainty was preterm birth. Outcomes with high certainty were gestational diabetes mellitus, hypertensive disorders of pregnancy, stillbirths and neonatal admissions. Outcomes with limited evidence and low agreement that had very low confidence were cesarean section, placental outcome and neonatal death.
Discussion
This systematic review collated evidence from across the world on the harmful impacts of heat on maternal, fetal and neonatal health. Five critical outcomes, preterm births, low birth weight, hypertensive disorders of pregnancy, congenital anomalies and stillbirths, accounted for 75% of the evidence. Ten outcomes, however, including several major causes of maternal mortality, were represented by only one or two publications, constraining our ability to summarize the full extent of heat harms in these populations.
Maternal outcomes only account for a quarter of the body of literature in this review and are underrepresented in heat-health research. Evidence is, however, consistent for increased risk of hypertensive disorders in pregnancy (high confidence) and gestational diabetes mellitus (high confidence). Although few studies examined infectious disease outcomes, a large body of work outside of maternal and neonatal health suggests that heat-related exacerbations of infectious diseases may pose a profound threat1. Only two studies assessed the impact of heat on maternal mental health, a population at higher risk for mental illness.
Most studies on heat-related fetal outcomes had addressed the risk of stillbirth (high confidence) and congenital anomalies (medium confidence). A similar increased risk of stillbirth is reported by Chersich et al.8, with the inclusion of an additional 11 studies in this updated review. Our study findings also suggest potential heat impacts on non-reassuring fetal status (medium confidence) and spontaneous abortions (low confidence), which broadens the scope of inquiry into heat impacts on fetal health and provides insights into the underlying causal pathways that mediate adverse outcomes.
By far, the most evidence is available for adverse impacts on neonatal outcomes, particularly preterm birth (very high confidence) and low birth weight (medium confidence), both crucial determinants of child mortality, health and wellbeing over the life course. The evidence suggests a dose–response relationship for preterm birth. Our review also provides some evidence for heat impacts in understudied outcomes such as neonatal morbidity (medium confidence) and neonatal admissions (high confidence). Only one study found an association between heat in pregnancy and neonatal death. Evidence outside of this review suggests that heat exposure associated with early neonatal death (within the first 24 h) is more likely in neonates born after complications with prematurity and childbirth, which have both been associated with heat70.
While windows of vulnerability during pregnancy were identified for some outcomes, much remains unknown. For preterm birth, two periods of vulnerability were identified, whereas, for hypertension in pregnancy, the very early gestational period was identified as a critical period, likely related to the early placentation period16. Very short term lags were identified for hospital admission, antenatal bleeding and prelabor rupture of membranes, likely linked to their pathophysiological pathways. Gestational diabetes was strongly linked with heat during the second trimester. This may reflect the timing of screening for diabetes in pregnancy rather than a heightened period of vulnerability. This is one example of potential bias with opportunistic reanalyses of existing data. More detailed information on vulnerability windows in pregnancy and lag effects would help target protective interventions, especially timeous heat-health messaging for pregnant women. This is currently being evaluated in the HIGH Horizons project (https://www.high-horizons.eu/).
Previous systematic reviews on heat and maternal and neonatal health, such as those by Chersich et al.8, Veenema et al.13, Rekha et al.11 and Dalugoda et al.6, have established associations between heat exposure and adverse outcomes like preterm birth, stillbirth and congenital abnormalities, often highlighting variability in findings and a focus predominantly on higher-income countries. Our review expands on these studies by incorporating a wider geographic scope, analyzing rarer outcomes and quantifying risk. Quantifying these risks is essential for establishing surveillance systems to monitor heat-related morbidity, assessing intervention effectiveness, attributing risk to anthropogenic climate change with implications for loss and damage findings and modeling future health and economic impacts of climate change, exemplified by Zhang et al.67. Additionally, we employ a certainty grading, taking a step further to evaluate the robustness of the evidence, which enhances the reliability of findings for future research and policy action.
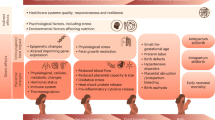
To explain the harmful impacts of heat during pregnancy, various pathophysiological mechanisms have been proposed, mostly based on animal studies9. Putative mechanisms include elevated maternal body temperatures (including from infection-related fever), maternal dehydration leading to electrolyte imbalances, endocrine system dysregulation, altered glucose metabolism and release of anti-diuretic hormone, oxytocin, adrenaline and stress hormones28,51,71,72. The upregulation of heat shock proteins may also play a role in promoting cytokine release and tissue inflammation, contributing to premature onset of labor and placental insufficiency73,74. A series of studies, funded by the Wellcome Trust, is underway to understand the biological mechanisms of heat vulnerability in pregnancy75.
The growing body of evidence allowed an initial exploration of composite outcomes, which are useful from a public health perspective as they may capture the overall burden of disease and allow for the quantification of impact on related disease processes (for example, conditions related to dehydration or fluid shifts) and in specific periods of pregnancy or childbirth. Understanding where the principal burdens lie will help inform decisions, for example, around whether to focus on home-based interventions in late pregnancy or on facility cooling.
Our review identified substantial research gaps, particularly in tropical climate zones and the Global South. Heat impacts on maternal and neonatal health involve a complex interplay of social and economic phenomena, alongside clinical and biological factors76. Women’s experiences are influenced by sociocultural norms, economic disparities, health service access and the built environment, all of which mediate the effects of climate change on health. These factors disproportionately affect the most vulnerable populations in low- and middle-income countries. Furthermore, the lack of evidence from the areas most affected complicates efforts to mobilize adequate resourcing or to target resources effectively. Data gaps may reflect differences in research funding and capacity but also in data quality and the availability of electronic health record data for reanalysis. Approaches such as systematically collating individual-level participant data from cohorts and trials77 and specific initiatives to extract data from paper-based records may be the only means of adequately addressing these gaps in the short term. These efforts, in turn, support the goal of establishing formal indicator systems that track burdens and progress with adaptation responses.
The systematic review has several limitations. The updated search was limited to PubMed and citation searching. Although the large majority of papers had been found in the initial search through this approach, some studies may still have been missed. We used the outcome definitions provided in each study, which could introduce bias due to varying definitions across locations. Additionally, we did not conduct a formal risk of bias assessment of the included studies, given that the original review noted a predominately high risk of bias ratings. We elected to rather use an IPCC tool to grade certainty.
Our systematic review is limited by considerable heterogeneity in heat exposure metrics, outcome measures and study designs, which complicated data synthesis and interpretation. Meta-analyses were limited to studies that provided directly comparable effect estimates. Furthermore, many meta-analyses had high I2 values and evidence suggestive of publication bias, which require cautious interpretation. Employing multiple synthesis methods in this review aimed to triangulate results to minimize biases inherent in each approach. Standardized methodologies and reporting guidelines in this field may enhance comparability and validity of findings.
In summary, this systematic review demonstrates clear and sizable relationships between heat exposure and multiple adverse maternal, fetal and neonatal health outcomes. The gaps in evidence on key outcomes and specific locations, however, and the lack of long-term systematic measurement of these harms signal a failure to plan and protect pregnant women, newborns and other vulnerable populations against climate change. Interventions to be prioritized now may include issuing timeous early warnings, intensive cooling and hydration initiatives during heat waves, behavior change awareness, cooling in health facilities and enhanced surveillance. More broadly, it will require the health sector and other sector leaders to exert real influence over health, urban planning, housing, transport and the energy sector to reduce heat exposure, to avert further climate collapse and to fulfill their duty of care to pregnant women and the next generation.
Methods
Study design
We conducted a systematic review following the 2020 PRISMA guidelines83. Included were all peer-reviewed epidemiological studies assessing the impact of heat during pregnancy on maternal and neonatal outcomes. This review forms part of a larger systematic mapping exercise that described the body of heat-health impacts and adaptation intervention literature84. We also include data from systematic reviews, conducted as part of the systematic mapping by Chersich et al.8 and Haghighi et al.10. The reviews are registered in PROSPERO under CRD42019140136, CRD42018118113 and CRD42020173519.
Search strategy and data extraction
We used a search strategy that was developed in MEDLINE (PubMed) and adapted it for Web of Science, the Science Citation Index Expanded, the Social Sciences Citation Index and the Arts and Humanities Citations Index. The systematic mapping search was conducted in September 2018 and updated in MEDLINE in 2019 and July 2023. The full search strategy can be found in Chersich et al.85 and Supplementary Note 3. We included all study designs apart from systematic reviews and qualitative studies. No date restrictions were applied, and no gray literature was included. The original review in 2018 involved independent, duplicate screening of titles and abstracts and data extraction from full-text articles. For the updated review, screening of titles and abstracts was done by a single reviewer, but study selection and data extraction were completed by two reviewers. EPPI-Reviewer version 4 software was used for reference management, screening and data extraction86. We extracted study citation identifiers, location, sample size, main study outcome and effect estimates. The study location was used to establish country-level gross domestic product (https://databank.worldbank.org/source/world-development-indicators) and Köppen–Geiger climate zones87.
Evidence synthesis
We used multiple synthesis methods to accommodate for heterogeneity, using SWiM guidelines88 and the Cochrane Handbook89. First, we grouped all the studies thematically by outcome type and population group, as per the WHO conceptual framework on extreme heat on maternal, newborn and child health (https://www.high-horizons.eu/conceptual-framework-on-extreme-heat-and-maternal-newborn-and-child-health/), using GAIA preferred terminology90. Vote counting illustrated the number of direction-of-effect measures, demonstrating benefit or harm per outcome group. Random-effect meta-analysis grouped studies based on heat exposure metrics and exposure lags.
Statistical analysis
Random-effect meta-analysis was selected to account for variability within and across studies and to describe the average exposure effect of heat. ORs, hazard ratios and risk ratios were considered comparable, as per the Cochrane Handbook. We selected three heat exposure metric groups as per Chersich et al.8: (1) odds of outcome per 1 °C increase in temperature, (2) odds of outcome during heat wave versus non-heat wave period (2 or more days above a predefined threshold, typically at or above the 90th percentile) and (3) odds of outcome at high versus low heat exposure (dichotomized thresholds). The third group reported combined effects without specific cutoff points due to variability, with high thresholds generally above the 75th, 90th or 99th percentile. Effect estimates were transformed when appropriate to meet these three groupings, for example, Fahrenheit converted to Celsius and per 10 °C increase calculated to per 1 °C increase. We further grouped preterm birth and stillbirths based on lag, where short-term lags account for exposure during the 4 weeks preceding the event and long-term lags account for exposure beyond that time. We calculated the standard error of the effect estimate using upper and lower CIs. When the upper- and lower-bound standard errors differed, we averaged the two and generated a new CI based on the estimated standard error. Heterogeneity was explored through subgroup analyses (country income level, latitude, mean temperature and climate zones) when random-effect meta-analyses were appropriate. We evaluated the meta-analyses and publication bias by interpreting a combination of the I2 value, the difference between fixed- and random-effect models, leave-one-out analyses, funnel plots and prediction intervals for outcomes with at least nine effect estimates. When meta-analysis was deemed inappropriate, the median OR and interquartile range were presented instead. All descriptive analyses and meta-analyses were conducted using R version 4.3.1 with the meta package 6.5-0 (ref. 91).
Certainty grading
We adapted the IPCC system of calibrated uncertainty language to evaluate evidence type, quantity, quality, consistency (ranked limited, medium or robust) and degree of agreement (low, medium or high) to express levels of confidence (from very low to very high) for each outcome92. We chose the IPCC grading system over GRADE and other similar tools due to its suitability for climate-related research. This approach is suitable for observational data, which might be undervalued by systems like GRADE that often rate such studies as low quality. For type, all observational data were ranked medium, above expert opinions or case studies and below randomized controlled trials. Quantity was assessed by the number of studies per outcome. Previous reviews have shown that nearly all observational studies are at high risk of bias; therefore, we did not assess quality or risk of bias in this study8,14,93. Consistency was measured by calculating the ratio of harmful to protective results. Agreement was determined by surveying four study authors and reviewers to apply their expert knowledge in the field to categorize each outcome as low, medium or high. Confidence statements were allocated based on evidence and agreement scores. The final agreement scores and confidence statements were reviewed and confirmed by consensus by D.P.L. and M.F.C.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
We conducted the searches on MEDLINE (PubMed), Web of Science, the Science Citation Index Expanded, the Social Sciences Citation Index and the Arts and Humanities Citations Index. Search results were uploaded to EPPI-Reviewer version 4 for deduplication and screening. Data supporting the findings of this study are available in the published literature, within the paper and in the Extended Data and the Supplementary Information.
Code availability
All descriptive analyses and meta-analyses were conducted using R version 4.3.1 with the meta package 6.5-0. There was no custom code developed for the study analyses.
References
Romanello, M. et al. The 2023 report of the Lancet Countdown on health and climate change: the imperative for a health-centred response in a world facing irreversible harms. Lancet 402, 2346–2394 (2023).
Chersich, M. F., Scorgie, F., Filippi, V. & Luchters, S. Increasing global temperatures threaten gains in maternal and newborn health in Africa: a review of impacts and an adaptation framework. Int. J. Gynaecol. Obstet. 160, 421–429 (2022).
Pörtner, H.-O. et al. (eds) IPCC, 2022: summary for policymakers. In Climate Change 2022: Impacts, Adaptation, and Vulnerability 3–33 (Cambridge University Press, 2022).
Samuels, L. et al. Physiological mechanisms of the impact of heat during pregnancy and the clinical implications: review of the evidence from an expert group meeting. Int. J. Biometeorol. 66, 1505–1513 (2022).
Dervis, S. et al. Heat loss responses at rest and during exercise in pregnancy: a scoping review. J. Therm. Biol. 99, 103011 (2021).
Dalugoda, Y., Kuppa, J., Phung, H., Rutherford, S. & Phung, D. Effect of elevated ambient temperature on maternal, foetal, and neonatal outcomes: a scoping review. Int. J. Environ. Res. Public Health 19, 1771 (2022).
Hall, M. E., George, E. M. & Granger, J. P. The heart during pregnancy. Rev. Esp. Cardiol. 64, 1045–1050 (2011).
Chersich, M. F. et al. Associations between high temperatures in pregnancy and risk of preterm birth, low birth weight, and stillbirths: systematic review and meta-analysis. BMJ 371, m3811 (2020).
Bonell, A. et al. An expert review of environmental heat exposure and stillbirth in the face of climate change: clinical implications and priority issues. BJOG 131, 623–631 (2023).
Haghighi, M. M. et al. Impacts of high environmental temperatures on congenital anomalies: a systematic review. Int. J. Environ. Res. Public Health 18, 4910 (2021).
Rekha, S., Nalini, S. J., Bhuvana, S., Kanmani, S. & Vidhya, V. A comprehensive review on hot ambient temperature and its impacts on adverse pregnancy outcomes. J. Mother Child 27, 10–20 (2023).
Mao, Y. et al. Associations between extreme temperature exposure and hypertensive disorders in pregnancy: a systematic review and meta-analysis. Hypertens. Pregnancy 42, 2288586 (2023).
Veenema, R. J., Hoepner, L. A. & Geer, L. A. Climate change-related environmental exposures and perinatal and maternal health outcomes in the U.S. Int. J. Environ. Res. Public Health 20, 1662 (2023).
Brink, N. et al. Impacts of heat exposure in utero on long-term health and social outcomes: a systematic review. BMC Pregnancy Childbirth 24, 344 (2024).
Xiong, T. et al. Association between ambient temperature and hypertensive disorders in pregnancy in China. Nat. Commun. 11, 2925 (2020).
Part, C. et al. Ambient temperature during pregnancy and risk of maternal hypertensive disorders: a time-to-event study in Johannesburg, South Africa. Environ. Res. 212, 113596 (2022).
Tran, T. C. et al. Are meteorological conditions within the first trimester of pregnancy associated with the risk of severe pre-eclampsia? Paediatr. Perinat. Epidemiol. 29, 261–270 (2015).
Nasiri, R., Ahmadi Shadmehri, A., Khajeh Ghiassi, P., Sarafraz Yazdi, M. & Mazloum Farsi Baf, M. Association of meteorological factors and seasonality with preeclampsia: a 5-year study in northeast of Iran. Clin. Exp. Hypertens. 36, 586–589 (2014).
Tam, W. H., Sahota, D. S., Lau, T. K., Li, C. Y. & Fung, T. Y. Seasonal variation in pre-eclamptic rate and its association with the ambient temperature and humidity in early pregnancy. Gynecol. Obstet. Invest. 66, 22–26 (2008).
Algert, C. S., Roberts, C. L., Shand, A. W., Morris, J. M. & Ford, J. B. Seasonal variation in pregnancy hypertension is correlated with sunlight intensity. Am. J. Obstet. Gynecol. 203, 215.e1–215.e5 (2010).
Immink, A., Scherjon, S., Wolterbeek, R. & Steyn, D. W. Seasonal influence on the admittance of pre-eclampsia patients in Tygerberg Hospital. Acta Obstet. Gynecol. Scand. 87, 36–42 (2008).
Melo, B., Amorim, M., Katz, L., Coutinho, I. & Figueiroa, J. N. Hypertension, pregnancy and weather: is seasonality involved? Rev. Assoc. Med. Bras. 60, 105–110 (2014).
Metoki, H. et al. Seasonal trends of blood pressure during pregnancy in Japan: the Babies and their Parents’ Longitudinal Observation in Suzuki Memorial Hospital in Intrauterine Period study. J. Hypertens. 26, 2406–2413 (2008).
Booth, G. L. et al. Influence of environmental temperature on risk of gestational diabetes. CMAJ 189, E682–E689 (2017).
Su, W. L., Lu, C. L., Martini, S., Hsu, Y. H. & Li, C. Y. A population-based study on the prevalence of gestational diabetes mellitus in association with temperature in Taiwan. Sci. Total Environ. 714, 136747 (2020).
Retnakaran, R. et al. Impact of daily incremental change in environmental temperature on beta cell function and the risk of gestational diabetes in pregnant women. Diabetologia 61, 2633–2642 (2018).
Zhang, H. et al. Assessing the effects of non-optimal temperature on risk of gestational diabetes mellitus in a cohort of pregnant women in Guangzhou, China. Environ. Int. 152, 106457 (2021).
Molina-Vega, M. et al. Relationship between environmental temperature and the diagnosis and treatment of gestational diabetes mellitus: an observational retrospective study. Sci. Total Environ. 744, 140994 (2020).
Teyton, A. et al. Examining the relationship between extreme temperature, microclimate indicators, and gestational diabetes mellitus in pregnant women living in Southern California. Environ. Epidemiol. 7, e252 (2023).
Qu, Y. et al. Ambient extreme heat exposure in summer and transitional months and emergency department visits and hospital admissions due to pregnancy complications. Sci. Total Environ. 777, 146134 (2021).
Rammah, A. et al. Temperature, placental abruption and stillbirth. Environ. Int. 131, 105067 (2019).
He, S., Kosatsky, T., Smargiassi, A., Bilodeau-Bertrand, M. & Auger, N. Heat and pregnancy-related emergencies: risk of placental abruption during hot weather. Environ. Int. 111, 295–300 (2018).
Dadvand, P., Basagana, X., Figueras, F., Sunyer, J. & Nieuwenhuijsen, M. J. Climate and group B streptococci colonisation during pregnancy: present implications and future concerns. BJOG 118, 1396–1400 (2011).
Minisha, F. et al. Bacteriuria in pregnancy varies with the ambiance: a retrospective observational study at a tertiary hospital in Doha, Qatar. J. Perinat. Med. 48, 46–52 (2019).
Lin, Y. et al. Association between temperature and maternal stress during pregnancy. Environ. Res. 158, 421–430 (2017).
Zhao, Q. et al. The association between heatwaves and risk of hospitalization in Brazil: a nationwide time series study between 2000 and 2015. PLoS Med. 16, e1002753 (2019).
Molina, S. The perils of climate change: in utero exposure to temperature variability and birth outcomes in the Andean region. Econ. Hum. Biol. 24, 111–124 (2017).
Ha, S. et al. Ambient temperature and risk of cardiovascular events at labor and delivery: a case-crossover study. Environ. Res. 159, 622–628 (2017).
Zhang, Y. et al. Ambient temperature and major structural anomalies: a retrospective study of over 2 million newborns. Sci. Total Environ. 882, 163613 (2023).
Davies, B. R. The seasonal conception of lethal congenital malformations. Arch. Med. Res. 31, 589–591 (2000).
Van Zutphen, A. R., Lin, S., Fletcher, B. A. & Hwang, S. A. A population-based case–control study of extreme summer temperature and birth defects. Environ. Health Perspect. 120, 1443–1449 (2012).
Auger, N., Siemiatycki, J., Bilodeau-Bertrand, M., Healy-Profitos, J. & Kosatsky, T. Ambient temperature and risk of preeclampsia: biased association? Paediatr. Perinat. Epidemiol. 31, 267–271 (2017).
Tikkanen, J. & Heinonen, O. P. Maternal hyperthermia during pregnancy and cardiovascular malformations in the offspring. Eur. J. Epidemiol. 7, 628–635 (1991).
Khodadadi, N., Dastoorpoor, M., Khanjani, N. & Ghasemi, A. Universal Thermal Climate Index (UTCI) and adverse pregnancy outcomes in Ahvaz, Iran. Reprod. Health 19, 33 (2022).
Dastoorpoor, M., Khanjani, N. & Khodadadi, N. Association between physiological equivalent temperature (PET) with adverse pregnancy outcomes in Ahvaz, southwest of Iran. BMC Pregnancy Childbirth 21, 415 (2021).
Bogan, M. et al. The effects of desert dust storms, air pollution, and temperature on morbidity due to spontaneous abortions and toxemia of pregnancy: 5-year analysis. Int. J. Biometeorol. 65, 1733–1739 (2021).
Bianchi-Demicheli, F. et al. Association between weather conditions and the incidence of emergency gynecological consultations. Gynecol. Obstet. Investig. 51, 55–59 (2001).
Sun, X. et al. Associations of ambient temperature exposure during pregnancy with the risk of miscarriage and the modification effects of greenness in Guangdong, China. Sci. Total Environ. 702, 134988 (2020).
Sciscione, A. C., Costigan, K. A. & Johnson, T. R. Increase in ambient temperature may explain decrease in amniotic fluid index. Am. J. Perinatol. 14, 249–251 (1997).
Luton, D., Alran, S., Fourchotte, V., Sibony, O. & Oury, J. F. Paris heat wave and oligohydramnios. Am. J. Obstet. Gynecol. 191, 2103–2105 (2004).
Bonell, A. et al. Environmental heat stress on maternal physiology and fetal blood flow in pregnant subsistence farmers in the Gambia, West Africa: an observational cohort study. Lancet Planet. Health 6, e968–e976 (2022).
Basagaña, X. et al. Heat waves and cause-specific mortality at all ages. Epidemiology 22, 765–772 (2011).
Schumann, B., Häggström Lundevaller, E. & Karlsson, L. Weather extremes and perinatal mortality—seasonal and ethnic differences in northern Sweden, 1800–1895. PLoS ONE 14, e0223538 (2019).
Wang, J. et al. Associations of maternal ambient temperature exposures during pregnancy with the placental weight, volume and PFR: a birth cohort study in Guangzhou, China. Environ. Int. 139, 105682 (2020).
Barreca, A. & Schaller, J. The impact of high ambient temperatures on delivery timing and gestational lengths. Nat. Clim. Change 10, 77–82 (2019).
Liu, X. et al. Associations of maternal ambient temperature exposures during pregnancy with the risk of preterm birth and the effect modification of birth order during the new baby boom: a birth cohort study in Guangzhou, China. Int. J. Hyg. Environ. Health 225, 113481 (2020).
Dadvand, P. et al. Climate extremes and the length of gestation. Environ. Health Perspect. 119, 1449–1453 (2011).
Wang, Q. et al. Effects of gestational ambient extreme temperature exposures on the risk of preterm birth in China: a sibling-matched study based on a multi-center prospective cohort. Sci. Total Environ. 887, 164135 (2023).
Wang, J., Williams, G., Guo, Y., Pan, X. & Tong, S. Maternal exposure to heatwave and preterm birth in Brisbane, Australia. BJOG 120, 1631–1641 (2013).
Yüzen, D. et al. Increased late preterm birth risk and altered uterine blood flow upon exposure to heat stress. EBioMedicine 93, 104651 (2023).
Wang, J., Tong, S., Williams, G. & Pan, X. Exposure to heat wave during pregnancy and adverse birth outcomes: an exploration of susceptible windows. Epidemiology 30, S115–S121 (2019).
Tapia, V. L. et al. Association between maximum temperature and PM2.5 with pregnancy outcomes in Lima, Peru. Environ. Epidemiol. 5, e179 (2021).
Zheng, X., Zhang, W., Lu, C., Norbäck, D. & Deng, Q. An epidemiological assessment of the effect of ambient temperature on the incidence of preterm births: identifying windows of susceptibility during pregnancy. J. Therm. Biol. 74, 201–207 (2018).
Giorgis-Allemand, L. et al. The influence of meteorological factors and atmospheric pollutants on the risk of preterm birth. Am. J. Epidemiol. 185, 247–258 (2017).
Wang, Y. Y. et al. Ambient temperature and the risk of preterm birth: a national birth cohort study in the mainland China. Environ. Int. 142, 105851 (2020).
Nyadanu, S. D., Tessema, G. A., Mullins, B. & Pereira, G. Prenatal acute thermophysiological stress and spontaneous preterm birth in Western Australia, 2000–2015: a space–time-stratified case-crossover analysis. Int. J. Hyg. Environ. Health 245, 114029 (2022).
Zhang, Y. et al. The burden of heatwave-related preterm births and associated human capital losses in China. Nat. Commun. 13, 7565 (2022).
Cil, G. & Kim, J. Extreme temperatures during pregnancy and adverse birth outcomes: evidence from 2009 to 2018 U.S. national birth data. Health Econ. 31, 1993–2024 (2022).
Kakkad, K., Barzaga, M. L., Wallenstein, S., Azhar, G. S. & Sheffield, P. E. Neonates in Ahmedabad, India, during the 2010 heat wave: a climate change adaptation study. J. Environ. Public Health 2014, 946875 (2014).
Dimitrova, A. et al. Temperature-related neonatal deaths attributable to climate change in 29 low- and middle-income countries. Nat. Commun. 15, 5504 (2024).
Limesand, S., Camacho, L., Kelly, A. & Antolic, A. Impact of thermal stress on placental function and fetal physiology. Anim. Reprod. 15, 886–898 (2018).
Pirhonen, J., Vähä-Eskeli, K., Seppänen, A., Vuorinen, J. & Erkkola, R. Does thermal stress decrease uterine blood flow in hypertensive pregnancies? Am. J. Perinatol. 11, 313–316 (1994).
Kanninen, T., Sisti, G. & Witkin, S. Induction of the 70 kDa heat shock protein stress response inhibits autophagy: possible consequences for pregnancy outcome. J. Matern. Fetal Neonatal Med. 29, 159–162 (2016).
MacPhee, D. & Miskiewicz, E. The potential functions of small heat shock proteins in the uterine musculature during pregnancy. Adv. Anat. Embryol. Cell Biol. 222, 95–116 (2017).
Biological vulnerability to extreme heat in maternal and child health. Wellcome Trust https://wellcome.org/grant-funding/schemes/biological-vulnerability-extreme-heat-maternal-and-child-health (2024).
Filippi, V., Chou, D., Barreix, M., Say, L. & the WHO Maternal Morbidity Working Group. A new conceptual framework for maternal morbidity. Int. J. Gynecol. Obstet. 141, 4–9 (2018).
Lakhoo, D. P. et al. Protocol of an individual participant data meta-analysis to quantify the impact of high ambient temperatures on maternal and child health in Africa (HE2AT IPD). BMJ Open 14, e077768 (2024).
Bakhtsiyarava, M. et al. Ambient temperature and term birthweight in Latin American cities. Environ. Int. 167, 107412 (2022).
Dieckmann, W. J. The geographic distribution and effect of climate on eclampsia, toxemia of pregnancy, hyperemesis gravidarum, and abruptio placentae. Am. J. Obstet. Gynecol. 36, 623–631 (1938).
Jensen, P. M. & Sørensen, M. Differences in human birth weight and corollary attributes as a result of temperature regime. Ann. Hum. Biol. 40, 385–395 (2013).
Wells, J. C. & Cole, T. J. Birth weight and environmental heat load: a between-population analysis. Am. J. Phys. Anthropol. 119, 276–282 (2002).
Basu, R., Chen, H., Li, D. K. & Avalos, L. A. The impact of maternal factors on the association between temperature and preterm delivery. Environ. Res. 154, 109–114 (2017).
Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372, n71 (2021).
Chersich, M. et al. Perspectives on the methods of a large systematic mapping of maternal health interventions. Global. Health 12, 51 (2016).
Chersich, M. et al. Mapping of research on maternal health interventions in low- and middle-income countries: a review of 2292 publications between 2000 and 2012. Global. Health 12, 52 (2016).
Thomas, J. et al. EPPI-Reviewer: Advanced Software for Systematic Reviews, Maps and Evidence Synthesis (EPPI-Centre, UCL Social Research Institute, University College London, 2022).
Beck, H. E. et al. Present and future Köppen–Geiger climate classification maps at 1-km resolution. Sci. Data 5, 180214 (2018).
Campbell, M. et al. Synthesis without meta-analysis (SWiM) in systematic reviews: reporting guideline. BMJ 368, l6890 (2020).
Higgins J. P. T. et al. (eds) Cochrane Handbook for Systematic Reviews of Interventions (Cochrane, 2023).
Bonhoeffer, J. et al. Global alignment of immunization safety assessment in pregnancy—the GAIA project. Vaccine 34, 5993–5997 (2016).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2020).
Mastrandrea, M. D. et al. Guidance Note for Lead Authors of the IPCC Fifth Assessment Report on Consistent Treatment of Uncertainties (ed. Intergovernmental Panel on Climate Change) (Intergovernmental Panel on Climate Change, 2010).
Lakhoo, D. P., Blake, H. A., Chersich, M. F., Nakstad, B. & Kovats, S. The effect of high and low ambient temperature on infant health: a systematic review. Int. J. Environ. Res. Public Health 19, 9109 (2022).
Acknowledgements
We acknowledge the contributions of D. Jackson and N. Roos, who reviewed this work as part of a project deliverable in HIGH Horizons. Similarly, we acknowledge V. Fillipi, who contributed to the literature search and data extraction. This project and the authors listed have received funding from the European Union’s Horizon Framework Programme under grant agreement no. 101057843: D.P.L., N.B., L.R., M.H.C., I.S., S.L., G.M., M.F.C. and C.B. Project partner LSHTM is funded by UKRI Innovate UK reference number 10038478. Furthermore, study authors are supported by the Fogarty International Center and the National Institute of Environmental Health Sciences and the OD/Office of Strategic Coordination of the National Institutes of Health under award number U54TW012083: D.P.L., N.B., I.S., S.L. and M.F.C. Lastly, this work was supported by the Wellcome Trust (227204/Z/23/Z): D.P.L., N.B., M.F.C., A.C., K.-G.T. and R.S. The content is solely the responsibility of the authors and does not necessarily represent the official views of the European Commission, the UKRI, the National Institutes of Health and the Wellcome Trust.
Author information
Authors and Affiliations
Consortia
Contributions
D.P.L.: conceptualization, methodology, validation, formal analysis, investigation, data curation, writing (original draft), writing (review and editing), visualization, project administration. N.B.: methodology, software, validation, formal analysis, investigation, data curation, writing (original draft), writing (review and editing), visualization. L.R.: methodology, software, validation, formal analysis, investigation, data curation, writing (original draft), writing (review and editing), visualization. M.H.C.: methodology, formal analysis, investigation, data curation, writing (original draft), writing (review and editing), visualization. M.D.P.: methodology, validation, investigation, writing (review and editing). M.M.H.: methodology, validation, formal analysis, investigation, writing (review and editing). A.W.: methodology, investigation, writing (original draft), writing (review and editing). I.S.: methodology, validation, investigation, writing (review and editing). S.L.: investigation, writing (review and editing). G.M.: investigation, writing (review and editing). M.F.C.: conceptualization, methodology, validation, formal analysis, investigation, writing (original draft), writing (review and editing), visualization, supervision, funding acquisition. Heat-Health Study Group: validation, investigation, writing (review and editing).
Corresponding author
Ethics declarations
Competing interests
D.P.L., N.B., M.H.C. and M.F.C. hold investments in the fossil fuel industry through their pension funds. The other authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks Claudia Hanson, Brenda Sequeira D’mello and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Ming Yang, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Publishing trends over time.
Number of publications per year (annualized for 2023).
Extended Data Fig. 2 Effects of heat exposure on stillbirth.
Meta-analysis representing odds of stillbirth with two heat exposure metrics a) per 1 °C increase in temperature (n = 5 studies) b) high versus low heat exposure (n = 9 studies). Each line of the forest plot represents an odds/risk/hazard ratio from a study, represented by the vertical line, and the 95% confidence interval, represented by the horizontal line. The weight each study carries in overall odds is represented by the grey shaded square. The overall odds ratio and 95% confidence interval, using a random effect meta-analysis model, is represented by the diamond. The heterogeneity is illustrated by the I2 percentage in the bottom left of the figure.
Extended Data Fig. 3 Effects of heat exposure on congenital anomalies.
Meta-analysis of odds of congenital anomalies at high versus low temperatures (n = 6 studies). Each line of the forest plot represents an odds/risk/hazard ratio from a study, represented by the vertical line, and the 95% confidence interval, represented by the horizontal line. The weight each study carries in overall odds is represented by the grey shaded square. The overall odds ratio and 95% confidence interval, using a random effect meta-analysis model, is represented by the diamond. The heterogeneity is illustrated by the I2 percentage in the bottom left of the figure.
Extended Data Fig. 4 Effects of high versus low heat exposure on preterm birth at a short lag.
Meta-analysis of odds of preterm birth at high versus low heat exposure at a short lag (n = 39 studies). Each line of the forest plot represents an odds/risk/hazard ratio from a study, represented by the vertical line, and the 95% confidence interval, represented by the horizontal line. The weight each study carries in overall odds is represented by the grey shaded square. The overall odds ratio and 95% confidence interval, using a random effect meta-analysis model, is represented by the diamond. The heterogeneity is illustrated by the I2 percentage in the bottom left of the figure.
Extended Data Fig. 5 Effects of heat exposure on preterm birth at a long lag.
Meta-analysis of odds of preterm birth at high versus low heat exposure at a long lag (n = 15 studies). Each line of the forest plot represents an odds/risk/hazard ratio from a study, represented by the vertical line, and the 95% confidence interval, represented by the horizontal line. The weight each study carries in overall odds is represented by the grey shaded square. The overall odds ratio and 95% confidence interval, using a random effect meta-analysis model, is represented by the diamond. The heterogeneity is illustrated by the I2 percentage in the bottom left of the figure.
Extended Data Fig. 6 Effects of heat exposure on low birth weight.
Meta-analysis of odds of low birth weight at high versus low heat exposures (n = 13 studies). Each line of the forest plot represents an odds/risk/hazard ratio from a study, represented by the vertical line, and the 95% confidence interval, represented by the horizontal line. The weight each study carries in overall odds is represented by the grey shaded square. The overall odds ratio and 95% confidence interval, using a random effect meta-analysis model, is represented by the diamond. The heterogeneity is illustrated by the I2 percentage in the bottom left of the figure.
Extended Data Fig. 7 Effects of heat exposure on obstetric complications.
Meta-analysis of odds of obstetric complications with two temperature metrics a) per 1 °C increase (n = 14 studies) b) heatwaves (n = 11 studies). Each line of the forest plot represents an odds/risk/hazard ratio from a study, represented by the vertical line, and the 95% confidence interval, represented by the horizontal line. The weight each study carries in overall odds is represented by the grey shaded square. The overall odds ratio and 95% confidence interval, using a random effect meta-analysis model, is represented by the diamond. The heterogeneity is illustrated by the I2 percentage in the bottom left of the figure.
Supplementary information
Supplementary Information (download PDF )
Supplementary Table 1 and Notes 1–4.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lakhoo, D.P., Brink, N., Radebe, L. et al. A systematic review and meta-analysis of heat exposure impacts on maternal, fetal and neonatal health. Nat Med 31, 684–694 (2025). https://doi.org/10.1038/s41591-024-03395-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41591-024-03395-8
This article is cited by
-
Disproportionate climate burden of rising temperatures on low birth weight in Pakistan
BMC Medicine (2026)
-
The role of housing instability in the association between ambient temperature and preterm birth
Environmental Health (2026)
-
Challenges in understanding the effect of climate extremes and climatic factors on maternal and infant health
Nature Health (2026)
-
The effect of ambient temperature in pregnancy on the risk of gestational diabetes mellitus: evidence from temperate region
International Journal of Biometeorology (2026)
-
Born Too Soon: Accelerating change to 2030 and beyond
Reproductive Health (2025)