Abstract
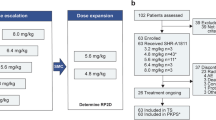
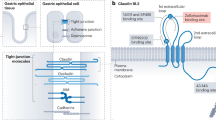
Claudin-18 isoform 2 (CLDN18.2), a tight junction protein expressed in non-malignant gastric epithelium and exposed on tumor cell surface during malignant transformation, is a promising therapeutic target for gastric and gastroesophageal junction (G/GEJ) cancers. SHR-A1904 is an antibody–drug conjugate comprising CLDN18.2-targeting monoclonal antibody, a DNA topoisomerase I inhibitor payload and a cleavable peptide-based linker. We conducted a first-in-human, three-stage, phase 1 study to evaluate SHR-A1904 in 95 previously treated patients with CLDN18.2-positive advanced G/GEJ cancer. In the dose-escalation stage (0.6–8.0 mg kg−1), dose-limiting toxicities were observed in two patients at 4.8 mg kg−1 (grade 3 febrile neutropenia and grade 3 increased blood bilirubin) and in one patient at 6.0 mg kg−1 (grade 3 gastric mucosal lesion). The maximum tolerated dose was not reached, and 6.0 mg kg−1 and 8.0 mg kg−1 were selected for pharmacokinetic and efficacy expansion. Treatment-emergent adverse events occurred in all 95 patients, most commonly anemia (72 (75.8%)), nausea (64 (67.4%)), hypoalbuminemia (61 (64.2%)) and decreased white blood cell count (56 (58.9%)). Additionally, 59 patients (62.1%) experienced drug-related grade 3 or higher adverse events. No treatment-related deaths were reported. Among response-evaluable patients, the confirmed objective response rate was 24.2% (95% confidence interval (CI), 11.1–42.3) at 6.0 mg kg−1 and 25.0% (95% CI, 12.1–42.2) at 8.0 mg kg−1. The median progression-free survival was 5.6 months (95% CI, 3.0–6.9) at 6.0 mg kg−1 and 5.8 months (95% CI, 3.0–8.6) at 8.0 mg kg−1. In conclusion, SHR-A1904 demonstrated a manageable safety profile and encouraging anti-tumor activity in patients with CLDN18.2-positive G/GEJ cancer, warranting further investigation. ClinicalTrials.gov identifier: NCT04877717.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
Data can be made available under restricted access due to proprietary reasons. The individual de-identified patient data generated during this study will be considered for sharing after the product and indication have been approved by major health authorities. Data may be requested 24 months after study completion. Qualified researchers should submit a proposal to the corresponding authors (R.-H.X., xurh@sysucc.org.cn; M.-Z.Q., qiumzh@sysucc.org.cn) outlining the reasons for requiring the data. The leading clinical site and sponsor will respond to requests in approximately 8 weeks. The sponsor will provide the data if the proposal is approved, provided that the requestor signs a data access agreement. Use of data must comply with the requirements of the Human Genetics Resources Administration of China and other country-specific or region-specific regulations. The study protocol and the statistical analysis plan are provided in the Supplementary Information.
Code availability
No custom code was used for data analysis in this study.
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263 (2024).
Diao, X. et al. Cancer situation in China: an analysis based on the global epidemiological data released in 2024. Cancer Commun. (Lond.) 45, 178–197 (2025).
Lordick, F. et al. Gastric cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann. Oncol. 33, 1005–1020 (2022).
NCCN Clinical Practice Guidelines. Gastric Cancer 2025 (version 2.2025–April 4, 2025); https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf
Shitara, K. et al. Trastuzumab deruxtecan (DS-8201a) in patients with advanced HER2-positive gastric cancer: a dose-expansion, phase 1 study. Lancet Oncol. 20, 827–836 (2019).
Shitara, K. et al. Trastuzumab deruxtecan in previously treated HER2-positive gastric cancer. N. Engl. J. Med. 382, 2419–2430 (2020).
Van Cutsem, E. et al. HER2 screening data from ToGA: targeting HER2 in gastric and gastroesophageal junction cancer. Gastric Cancer 18, 476–484 (2015).
Nakayama, I. et al. Claudin 18.2 as a novel therapeutic target. Nat. Rev. Clin. Oncol. 21, 354–369 (2024).
Sahin, U. et al. Claudin-18 splice variant 2 is a pan-cancer target suitable for therapeutic antibody development. Clin. Cancer Res. 14, 7624–7634 (2008).
Niimi, T. et al. claudin-18, a novel downstream target gene for the T/EBP/NKX2.1 homeodomain transcription factor, encodes lung- and stomach-specific isoforms through alternative splicing. Mol. Cell. Biol. 21, 7380–7390 (2001).
Rohde, C. et al. Comparison of Claudin 18.2 expression in primary tumors and lymph node metastases in Japanese patients with gastric adenocarcinoma. Jpn. J. Clin. Oncol. 49, 870–876 (2019).
Zhu, G. et al. Targeting CLDN18.2 by CD3 bispecific and ADC modalities for the treatments of gastric and pancreatic cancer. Sci. Rep. 9, 8420 (2019).
Pellino, A. et al. Association of CLDN18 protein expression with clinicopathological features and prognosis in advanced gastric and gastroesophageal junction adenocarcinomas. J. Pers. Med 11, 1095 (2021).
Xu, B. et al. Highly expressed Claudin18.2 as a potential therapeutic target in advanced gastric signet-ring cell carcinoma (SRCC). J. Gastrointest. Oncol. 11, 1431–1439 (2020).
Weng, W. et al. Decreased expression of claudin-18.2 in alpha-fetoprotein-producing gastric cancer compared to conventional gastric cancer. J. Gastrointest. Oncol. 13, 1035–1045 (2022).
Xu, G. et al. CMG901, a Claudin18.2-specific antibody-drug conjugate, for the treatment of solid tumors. Cell Rep. Med. 5, 101710 (2024).
Waters, R. et al. Retrospective study of claudin 18 isoform 2 prevalence and prognostic association in gastric and gastroesophageal junction adenocarcinoma. JCO Precis. Oncol. 8, e2300543 (2024).
Shitara, K. et al. Global prevalence of claudin 18 isoform 2 in tumors of patients with locally advanced unresectable or metastatic gastric or gastroesophageal junction adenocarcinoma. Gastric Cancer 27, 1058–1068 (2024).
Shah, M. A. et al. Zolbetuximab plus CAPOX in CLDN18.2-positive gastric or gastroesophageal junction adenocarcinoma: the randomized, phase 3 GLOW trial. Nat. Med. 29, 2133–2141 (2023).
Shitara, K. et al. Zolbetuximab in gastric or gastroesophageal junction adenocarcinoma. N. Engl. J. Med. 391, 1159–1162 (2024).
Ruan, D. Y. et al. Claudin 18.2-targeting antibody–drug conjugate CMG901 in patients with advanced gastric or gastro-oesophageal junction cancer (KYM901): a multicentre, open-label, single-arm, phase 1 trial. Lancet Oncol. 26, 227–238 (2025).
Sahin, U. et al. A phase I dose-escalation study of IMAB362 (Zolbetuximab) in patients with advanced gastric and gastro-oesophageal junction cancer. Eur. J. Cancer 100, 17–26 (2018).
Türeci, O. et al. A multicentre, phase IIa study of zolbetuximab as a single agent in patients with recurrent or refractory advanced adenocarcinoma of the stomach or lower oesophagus: the MONO study. Ann. Oncol. 30, 1487–1495 (2019).
Seiter, K. Toxicity of the topoisomerase I inhibitors. Expert Opin. Drug Saf. 4, 45–53 (2005).
Acknowledgements
This study was sponsored by Jiangsu Hengrui Pharmaceuticals Co., Ltd. This trial was designed by the authors in collaboration with the sponsor. The authors and sponsor were involved in data collection, analysis and interpretation; in guaranteeing the accuracy and completeness of the data; in writing of the report; and in the decision to submit the manuscript for publication. The sponsor had no right to veto publication or to control the decision regarding to which journal the manuscript was submitted. We are grateful to all patients, their families, the investigators, the site staff and the trial teams. Medical writing support was provided by H. Dong (medical writer at Hengrui) according to Good Publication Practice guidelines.
Author information
Authors and Affiliations
Contributions
R.-H.X. has full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: R.-H.X. Acquisition, analysis and interpretation of data: all authors. Drafting of the manuscript: D.-Y.R. and H.-X.W. Critical revision of the manuscript for important intellectual content: R.-H.X., M.-Z.Q., S.-X.L., W.-W.H., X.-J.L., Z.-X.N., Q.D., H.-L. Li, Z.-Y.P., H.-X.L., Y.-Q.Z., X.-Y.L., X.-Y.X., S.-R.C., Y.-G.D., J.Z., Z.L., H.-T.L., X.W., Y.Z., L.L., H.-L. Liu, P.-S.X., A.-L.S., R.-N.J., Y.-Q.L., X.-D.P., S.-C.W., A.-A.Y. and J.X. Statistical analysis: A.-A.Y. Administrative, technical or material support: R.-H.X., M.-Z.Q., D.-Y.R., H.-X.W., S.-C.W., A.-A.Y. and J.X. Supervision: R.-H.X. and M.-Z.Q.
Corresponding authors
Ethics declarations
Competing interests
R.-H.X. has served in a consulting or advisory role for Bristol Myers Squibb, Merck Serono, Roche, Astellas, AstraZeneca, Junshi, Hengrui, BeiGene and CPPC. S.-C.W., A.-A.Y. and J.X. are employed by Jiangsu Hengrui Pharmaceuticals Co., Ltd. The other authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks Sarbajit Mukherjee and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Ulrike Harjes, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Incidence of Vomiting and Nausea With or Without Prior Gastrectomy.
TEAE, treatment-emergent adverse event; TRAE, treatment-related adverse event.
Extended Data Fig. 2
Occurrence of Treatment-Related Nausea and Vomiting During Study.
Extended Data Fig. 3 Pharmacokinetic Profile.
(a) Mean serum concentration versus time curve of SHR-A1904 over cycles 1 to 3 at doses of 0.6, 1.2, 2.4, 3.6, 4.8, 6.0, and 8.0 mg/kg. (b) Mean serum concentration versus time curve of SHR-A1904, free payload, and total antibody over cycles 1 to 3 at dose of 6.0 and 8.0 mg/kg. Error bars indicate SD.
Extended Data Fig. 4
Kaplan-Meier Plots of Overall Survival in All Patients.
Extended Data Fig. 5 Correlation Between CLDN18.2 Expression and Anti-tumor activity of SHR-A1904.
CLDN18.2, claudin-18 isoform 2; ORR, objective response rate.
Supplementary information
Supplementary Information (download PDF )
Supporting preclinical data, Protocol and Statistical analysis plan.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ruan, DY., Wu, HX., Luo, SX. et al. The antibody–drug conjugate SHR-A1904 for targeting CLDN18.2 in advanced gastric or gastroesophageal junction cancer: a phase 1 trial. Nat Med 31, 3037–3046 (2025). https://doi.org/10.1038/s41591-025-03781-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41591-025-03781-w
This article is cited by
-
Antibody–drug conjugates in gastric cancer: clinical advances and resistance mechanisms
Gastric Cancer (2026)
-
Evolution of claudin18.2 therapies in gastroesophageal cancers
Nature Medicine (2025)