Abstract
In 2016, India introduced Rotavac (G9P[11]), an indigenous oral rotavirus vaccine administered at 6, 10 and 14 weeks of age through the Universal Immunization Program. Evaluating its effectiveness under routine programmatic conditions is critical, given the variable performance of rotavirus vaccines in low- and middle-income countries. Here we assessed Rotavac’s real-world effectiveness and impact across 31 hospitals in 9 states between 2016 and 2020 using a test-negative case–control design. Overall, 24,624 children were enrolled in surveillance (62% male and 38% female). Of 8,372 children aged 6–59 months eligible for effectiveness analysis (1,790 rotavirus-positive cases and 5,437 rotavirus-negative controls), 6,646 received 3 doses and 581 were unvaccinated. The adjusted vaccine effectiveness of 3 doses against severe rotavirus gastroenteritis was 54% (95% confidence interval (CI) 45% to 62%), with 1,574 vaccinated cases versus 5,072 vaccinated controls. Among children aged 6–23 months (1,486 vaccinated cases and 4,595 vaccinated controls), genotype-specific adjusted vaccine effectiveness was 51% (95% CI 36% to 62%) for G3P[8], 81% (95% CI 73% to 87%) for G1P[8] and 64% (95% CI 21% to 83%) for G1P[6]. Following vaccine introduction, rotavirus positivity among hospitalized children declined from 40% to 20%. These findings confirm that Rotavac provides substantial protection against severe rotavirus disease, including nonvaccine strains, and performs comparably to internationally licensed vaccines in similar settings.
Similar content being viewed by others
Main
Annually, 128,500 deaths occurring among children younger than 5 years are attributed to rotavirus, thus placing a high demand on healthcare systems1. Two live oral rotavirus vaccines, the monovalent human strain based Rotarix (GlaxoSmithKline Biologicals) and the pentavalent bovine-human reassortant strains containing RotaTeq (Merck and Co.), have been licensed and used internationally from 2006 onward and have successfully demonstrated a reduction in disease burden2. However, the vaccine performance in high mortality countries has been suboptimal3. Nonetheless, it has been estimated that approximately 28,900 deaths among children younger than 5 years were prevented globally in 2016 due to the use of these two vaccines1. Despite the availability of live-attenuated, oral rotavirus vaccines since 2006, rotavirus continues to be the most common cause of severe acute gastroenteritis (AGE) in young children in low- and middle-income countries (LMICs) with peak incidence among children aged 4–23 months4.
India is estimated to account for one-fifth of the global rotavirus attributed deaths, and sentinel-hospital-based surveillance during the period of 2012–2016 showed that approximately 37% gastroenteritis hospitalizations in children were due to rotavirus5.
In 2014, an indigenously manufactured rotavirus vaccine (Rotavac, Bharat Biotech) based on a single naturally occurring bovine-human reassortant strain, was licensed in India following clinical trials showing an efficacy of 54% against severe rotavirus gastroenteritis in children followed up for 24 months. The vaccine specifically targets group A rotavirus, which accounts for over 90% of all rotaviral gastroenteritis cases. The efficacy was comparable to that of Rotarix and RotaTeq in low-income settings6. The National Technical Advisory Group on Immunization (NTAGI) recommended the phased introduction of Rotavac in the Indian national vaccine program, beginning with four states, from April 2016 onward7.
In making their recommendations, the NTAGI emphasized the need for further monitoring of the effectiveness and impact of Rotavac. Further monitoring was suggested because vaccine performance can differ for conditions between routine programmatic use and clinical trials, especially given the variable performance of rotavirus vaccines in different populations3. In India, as in other LMICs, the oral polio vaccine (OPV) showed substantially diminished efficacy8. In addition, Rotavac is an oral, monovalent vaccine based on a unique, naturally attenuated neonatal rotavirus strain, G9P[11]. This strain has antigens that differ from those recently circulating in hospitalized Indian children; thus, examining vaccine protection against a range of strains is important5,9. Lastly, demonstrating the real-world impact of vaccination in reducing the burden of gastroenteritis hospitalizations provides useful evidence regarding the value of vaccination and can also help examine its potential indirect benefits in age groups not directly targeted by vaccination.
Here, we report data from a multicentric, hospital-based, observational study on the effectiveness and impact of a completed series of Rotavac against laboratory-confirmed rotavirus diarrheal admissions in Indian children. Vaccine effectiveness, assessed using a test-negative design, was stratified by age groups, nutritional status and against major circulating genotypes. Vaccine impact was assessed in a subset of hospitals with prevaccination surveillance data and compared with the postvaccination period data.
Results
Rotavirus vaccine effectiveness
A map of India with the details of states that were part of the surveillance are shown in Extended Data Fig. 1. Between January 2016 and January 2020, a total of 32,690 children were admitted to the surveillance network hospitals for AGE as shown in Fig. 1. The enrollment was following a detailed protocol as given in Extended Data Fig. 2. Of these, 24,624 children who fulfilled the criterion for inclusion were enrolled. Among the enrolled children, 62% were males and 38% were females. After excluding ineligible subjects, a total of 2,021 rotavirus-positive cases and 6,351 rotavirus-negative controls were included in the vaccine effectiveness analysis. Enrollment by site is described in Extended Data Table 1, and the characteristics of the cases and controls in Extended Data Table 2. The comparison of background characteristics among vaccinated and unvaccinated children is provided in Extended Data Table 3. The median age at hospital admission and sex of the cases and controls were not significantly different. This was similar among vaccinated and unvaccinated children as well. The covariates that were significantly different between the cases and controls included household size, education level of the mother, source of water supply in the household, asset score (measuring socioeconomic status), height for age Z (HAZ)-scores (measuring nutritional status), state of residence and maternal age. Among eligible children, positivity rates of rotavirus decreased from 50% in January to December 2016 to 23% in January 2020, and rotavirus vaccination coverage for three doses of the vaccine steadily increased from 50% in 2016 to 96% in January 2020. In Universal Immunization Program (UIP), OPV, pentavalent and rotavirus vaccine are coadministered at 6, 10, 14 weeks of age. During the study, 91%, 85%, and 77% of age-eligible children received their first, second and third doses of OPV and rotavirus vaccination on the same day (Extended Data Table 4). Extended Data Table 4 presents the difference in number of days between administration of OPV and rotavirus vaccine for the same child based on their vaccination card details. Extended Data Fig. 3 shows the difference in timeliness of an eligible child receiving each dose of pentavalent vaccine and rotavirus vaccine. A distinct time gap is observed on the basis of recommended time period of vaccine versus actually obtaining the vaccine in UIP. The time gap is more pronounced for the second and third doses of these vaccines.
A flow diagram outlining the data collection process for study surveillance, presenting the layout of participants from initial screening to eligibility assessment and inclusion in final analysis and providing details of exclusion stepwise.
In children aged 6–59 months, the adjusted vaccine effectiveness (aVE) for a completed series of Rotavac was 54% (95% confidence interval (CI) 45% to 62%), with similar vaccine effectiveness estimates in the first and second years of life (Table 1). The common circulating genotypes were G3P[8] (49%), G1P[8] (12%), G2P[4] (10%), G1P[6] (4%) and G9P[4] (3%), with 17% of rotavirus-positive samples containing mixed G and P types. Strain-specific aVE for three completed doses of the vaccine was 81% (95% CI 73% to 87%), 51% (95% CI 36% to 62%) and 32% (95% CI −23% to 62%) against G1P[8], G3P[8], and G2P[4], respectively (Table 1). The model-building approach for the unconditional logistic regression follows a stepwise selection method, incorporating both forward selection and backward elimination. Covariates were added one by one based on statistical significance, while also considering their clinical relevance. Model comparison and variable retention decisions were guided by the Bayesian Information Criterion to ensure model parsimony. Adjusting for the HAZ score, Vesikari score, site of enrollment, month and year of birth, month and year of admission, and socioeconomic status indicators did not significantly alter the effect estimates. The final analysis model does not include vaccine effectiveness based on sex of the child, as there was no significant association during preliminary analysis. Inclusion of sex variable into the final model did not improve the model strength. The final model retained covariates that were both statistically significant and contributed to the best overall goodness of fit.
The vaccine effectiveness estimates for the completed vaccination series among eligible children stratified by their age group and nutritional status (indicated by HAZ scores) revealed a vaccine effectiveness of 46% (95% CI 13% to 66%) among stunted children compared with that of 64% (95% CI 51% to 73%) among nonstunted children during the first year of life. The vaccine effectiveness estimates for eligible children stratified by dose of the vaccine and severity of AGE are included in Extended Data Table 5.
To check the robustness of the effectiveness estimates, we conducted a sensitivity analysis and results are included in Table 2. As the results remain stable across different models, the findings are not method dependent, and the inclusion of the E value suggests the strength of unmeasured confounders. The calculated E value is 3.77 for the main model, which shows that an unmeasured confounder would need to increase the odds of both being vaccinated and getting infected by at least 3.77× each to fully explain away the observed effect.
Rotavirus vaccine impact
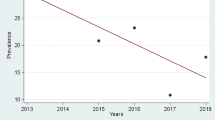
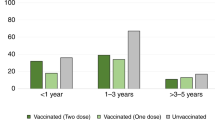
In the five hospitals with rotavirus surveillance data from September 2012 to June 2020, a total of 4,163 patients with AGE were enrolled from September 2012 to April 2016. Of these, 1,656 (39.8%) tested positive for rotavirus. Moreover, during the postvaccination period (May 2016 to June 2020), of a total of 4,336 children with AGE that were enrolled, 874 (21.4%) tested positive for rotavirus (Fig. 2). The rotavirus vaccination coverage that was 3.1% in September 2012 and attributable to the private sector, rose to 90.9% in June 2020. By site, declines in rotavirus detection rates were observed after vaccine introduction at each of the five sites, with progressive declines in later years with increasing vaccine coverage. The reduction in rotavirus positivity rate was 51.2% and 54.2% in infancy and among children aged 12–23 months, respectively (Fig. 3). A substantial reduction in the rate in the 24–59 month age group (ranging from 43.5% in a 24–35-month-old group to 43.1% in a 48–59-month-old group) was also noted. The vaccination coverage for these children in the postvaccination period ranged from 5.8% in the 48–59-month age group to 71.7% in the first year of life (Fig. 3). Extended Data Table 6 helps to understand the reduction rates in positivity and vaccination coverage trends across different age groups during the prevaccination and postvaccination periods and to quantify the proportion standard error and confidence limits of these estimates (Extended Data Table 6). In the interrupted time series model, the rotavirus positivity dropped by 41% showing a strong immediate effect, which is statistically significant (Table 3). The decline of rotavirus positivity as time progresses after the vaccination suggest a small but significant long-term reduction (Table 2).
A comparison of rotavirus positivity during prevaccination period and postvaccination period from five selected study sites. The bars indicate the number of rotavirus-positive cases (N = 2,530) and rotavirus-negative cases (N = 5,744) among children under 5 years admitted with AGE in the study settings by month and year of surveillance. The blue colored solid line shows the percentage of rotavirus-positives among children enrolled in the surveillance by month and year of surveillance. The blue dotted line shows the percentage of children who had received at least a single dose of rotavirus vaccine at the time of enrollment into the study surveillance. The orange straight line shows the time of rotavirus vaccine introduction into the UIP in India. The data during the prevaccine period from 2012 to 2016 were collected as a part of National Rotavirus Surveillance Network5,35. The postvaccine data were collected as a part of present study during the time period 2016–202036.
The percentage reduction in rotavirus positivity among children under 5 years after the introduction of rotavirus vaccination. The green bars show the percentage of children under 5 years of age who tested positive for rotavirus gastroenteritis during the prevaccination period stratified by their age groups (N = 4,163). The blue bars show the percentage of children under 5 years of age who tested positive for rotavirus gastroenteritis during the postvaccination period stratified by their age groups (N = 4,336). The dotted line shows the coverage of rotavirus vaccine during the study period of 2016–2020 for each age group. The data during the prevaccine period were collected as a part of National Rotavirus Surveillance Network (2012–2016) using similar protocol5,35. Postvaccine data were collected as a part of present study (2016–2020)36. The details are further elaborated in Extended Data Table 6.
Discussion
The effectiveness of the routine use of indigenous Rotavac vaccine in the national immunization program was 54% (95% CI 45% to 62%); this was reassuringly similar to the efficacy of 54% (95% CI 35% to 67%) reported during the phase 3 vaccine trial6. Further, the effectiveness was sustained in the first 2 years of life, when the burden of rotavirus is greatest1. The effectiveness of the vaccine was high against the predominant circulating strains, G1P[8] and G3P[8], which are different G and P types from G9P[11], further emphasizing that the monovalent vaccine offers good protection against heterologous strains. By introducing the vaccine, the number of rotavirus hospitalizations was also notably reduced at the sentinel sites, with a decrease in rotavirus positivity rates to less than half of that observed in the prevaccination period (Fig. 3), further corroborating the substantial public health impact of this vaccine.
Available literature on rotavirus vaccines used in immunization programs and the importance of vaccine introduction in all high-burden settings were summarized in a review in the year 20212. The performance of rotavirus vaccines since their introduction in 2006, with a focus on variations in effectiveness by socioeconomic status, age of vaccinated children and strain of rotavirus has been discussed in multiple literature reviews and meta-analyses with a recent update in the year 20223. Previous studies have consistently reported that the effectiveness of vaccines made by multinational manufacturers is poorer in LMICs (53%, 95% CI 36% to 62%) than in high-income settings10. The effectiveness of Rotavac in India was comparable with that of other internationally licensed vaccines (Rotarix and RotaTeq) in LMICs11,12,13,14,15,16,17,18,19,20,21,22,23,24 (Extended Data Table 7). The effectiveness of the vaccine in preventing rotaviral AGE of any level of severity among children less than 2 years of age in Bangladesh was 41.4% (95% CI 23.2% to 55.2%), with higher effectiveness observed during the first than the later years of life (45.2%, 95% CI 26.3% to 59.3%)25. The waning of vaccine effectiveness during the second year of life has been reported in several studies from LMICs; however, in Africa, the waning may be variable26. The vaccine effectiveness estimates from Malawi 3 years after the programmatic introduction of the vaccine showed estimates of 70.6% (95% CI 33.6% to 87.0%) in the first year of life and 31.7% (95% CI −140.6% to 80.6%) in the second year13. A study on the effectiveness of a full series of Rotarix vaccine among children aged 4–59 months from Botswana showed a vaccine effectiveness of 54% (95% CI 23% to 73%)4.
When stratified by the children’s age group and nutritional status (indicated as stunted or nonstunted using the HAZ scores), the vaccine effectiveness estimates in Africa differ and the difference is more pronounced in the first than in the later years of life. Between stunted and nonstunted children, in Zimbabwe27, the full series of Rotarix vaccine among children aged 6–11 months showed a vaccine effectiveness of 45% versus 71%, respectively, but not in older children. In Malawi13, for the full series of Rotarix vaccine among eligible children, the vaccine effectiveness was 28% versus 78%, respectively. In Mozambique28, the vaccine effectiveness for stunted versus nonstunted children was 14% and 59%, respectively, while in Botswana16 and Kenya23, among stunted children, the vaccine provided no such protection. In our study, we noted a difference in vaccine effectiveness estimates but with overlapping confidence intervals. Before the vaccine introduction in India, Rheingans et al. had concluded that by reducing socioeconomic and geographic disparities in Indian states, the impact of rotavirus vaccine could increase, saving an additional 10,000 children’s lives each year29.
The effectiveness of the neonatal vaccine of G9P[11] against the major circulating strains demonstrates the heterologous protection offered by rotavirus vaccines and is consistent with studies on other rotavirus vaccines in LMICs9,10,12,13,14,15,19,21. The study was powered to examine strain-specific vaccine effectiveness and demonstrated effectiveness against G1P[8], G3P[8] and G2P[4]. The major currently circulating strains of rotavirus in the developing world are G1P[8], G1P[6] and G3P[8]30. A systematic review by Leshem et al in 2014 has shown that Rotarix and RotaTeq provide heterologous protection without changes in the circulating strain31. Rotavirus vaccines work less effectively in preventing severe gastroenteritis in LMICs than in more developed countries, and this phenomenon has been reported both in clinical trials and effectiveness studies32. The reasons for the lower effectiveness are attributed to the damaged gut environments in children in LMICs that may interfere with response to the vaccine or other as yet unknown reasons30. The concomitant administration of rotavirus vaccine with OPV might also suppress vaccine rotavirus multiplication in the intestinal epithelium leading to a lower vaccine response. However, high coadministration rates prevented vaccine effectiveness evaluation in children who did not receive them concomitantly33.
Our study showed a significant reduction in rotaviral gastroenteritis among enrolled children in the postvaccination period compared with in the prevaccination period, adding to the limited findings from other countries2. A decline in rotavirus positivity rate among the cases was seen in all the five prevaccination surveillance sites over time, with increasing vaccine coverage, supporting the relationship with vaccine implementation. Similar declines were not observed in nonrotavirus AGE hospitalizations. The reduction in the number of admissions due to rotavirus AGE occurred in children less than 6 months of age, suggesting that even a partial vaccine series may offer some protection. Finally, and importantly, the decline in rotavirus positivity among 2–5 year old children, many of whom were age-ineligible to receive rotavirus vaccine, exceeded the expected based on low vaccine coverage in this age group and thus suggests indirect benefits due to reduced circulation of rotavirus in the population, as have been seen in high-income countries24.
In the current study, we observe a risk difference of −0.14, which highlights the fact that rotavirus positivity is less likely in the group that receives rotavirus vaccination. This indicates the substantial benefit of the vaccine in reducing the disease risk by 14% in the vaccinated population. The number needed to treat to prevent one additional case of severe rotaviral gastroenteritis is 7, this emphasizes the strong impact and protective effect of the vaccine in preventing disease.
Our study has important strengths in including recruitment at multiple hospitals in nine states, a large number of enrolled children after stringent verification of vaccination data and the availability of prior surveillance data at some sites for impact assessment. Limitations include the potential for selection bias in excluding children whose vaccination status could not be verified against health registries or national data. We addressed these limitations by using the test-negative design, which avoids the selective collection of vaccine cards of cases rather than controls since the study team was unaware of the rotavirus test result for each child. The inability to verify vaccination information because of poor or missing information in government records led to the exclusion of many children. However, retention of children with unverified vaccination information may have contributed to misclassification bias, resulting in lower than actual effectiveness estimates. The test-negative case–control design may introduce selection bias because health-seeking behavior may not accurately represent the broader population, potentially leading to an overestimation or underestimation of vaccine effectiveness. In addition, this design depends on the sensitivity and specificity of the diagnostic kits used, which can result in misclassification bias. The potential for residual confounding is also considerable, which may lead to biased vaccine effectiveness estimates. Moreover, the design assumes infection with a single pathogen, which may overlook the possibility of multiple or coinfections—a common issue in low-income settings. This study seeks to mitigate these limitations through strategic planning, the use of high-quality diagnostic tools and comprehensive data collection, ensuring that the findings are both accurate and generalizable. While estimating genotype specific vaccine effectiveness estimates, the limited sample sizes for specific genotypes have led to an increase in variability in the estimates. The genetic variability of circulating rotavirus strains might also be a reason for this variability, as different strains may exhibit distinct interactions with the vaccine, leading to variation in effectiveness. This calls for additional research and the need for larger sample sizes or more focused strain surveillance, which could improve the precision of these estimates in future studies.
There are limited impact assessment studies for newly introduced vaccines in LMICs. This study demonstrates that such studies are feasible and provide real-world evidence of the impact of rotavirus vaccination. A major concern regarding introducing a new vaccine is its safety and effectiveness. We recently reported the outcomes of a safety surveillance, which demonstrated that there was no association with intussusception34. Our study showed that Rotavac offers good protection against severe rotaviral gastroenteritis, including against genotypes excluded from the vaccine, in Indian children. Pre- and postintroduction data demonstrated a reduction in the overall rotavirus positivity rate in children hospitalized for gastroenteritis and in children not age-eligible for the vaccine, indicating an important public health impact. This affordable vaccine, which has been recently prequalified, is being rolled out in the Global Alliance for Vaccines and Immunization-eligible countries and will contribute to reducing global rotavirus burden.
Methods
Ethical approval
Ethical approval was obtained from Institutional Ethics Committee of Christian Medical College, Vellore, as well as from the institutional review boards of all participating hospitals. The ethics committees of all participating institutions approved the study as detailed in the published protocol36. Written informed consent was obtained from the parent or legal guardian of each enrolled child before data and sample collection. Any child who satisfied the study protocol were enrolled into the surveillance. All data were anonymized before analysis to ensure participant confidentiality. The 31 participating institutions are: KMCGGH, Kurnool, Andhra Pradesh, GGHRMC, Kakinada, Andhra Pradesh, KGHAMC, Vishakhapatnam, Andhra Pradesh, SVMC, Tirupati, Andhra Pradesh, RPGMC, Tanda, Himachal Pradesh, IGMC, Shimla, Himachal Pradesh, PGIMS, Rohtak, Haryana, SHKMGMC, Mewat, Haryana, BPSGMCW, Sonipat, Haryana, PGIMER, Chandigarh, Haryana, SVBPPGIP, Cuttack, Odisha, IMS, SUM, Bhubaneswar, Odisha, KIMS, Bhubaneswar, Odisha, Hi-Tech, Bhubaneswar, Odisha, MGMMC, Indore, Madhya Pradesh, SMSMC, Jaipur, Rajasthan, RNTMC, Udaipur, Rajasthan, SNMC, Jodhpur, Rajasthan, KKCTH, Chennai, Tamil Nadu, ICH, Chennai, Tamil Nadu, Christian Medical College (CMC), Vellore, Tamil Nadu, GVMC, Vellore, Tamil Nadu, Narayani, Vellore, Tamil Nadu, Nalam hospital, Vellore, Tamil Nadu, GRHMMC, Madurai, Tamil Nadu, BCH, Tezpur, Assam, GMC, Guwahati, Assam, KGMU, Lucknow, Uttar Pradesh, Mangla Hospital, Bijnor, Uttar Pradesh, IMS, BHU, Varanasi, Uttar Pradesh, BRDGMC, Gorakhpur, Uttar Pradesh.
Ethics and inclusion statement
Researchers from all 31 participating institutions were actively involved in the study design and responsible for obtaining ethical clearance from their respective institutional ethics committees, as well as implementing the study at their sites. Data ownership remains with the principal investigator at each institution, as outlined in formal memorandums of understanding, which were strictly adhered to. Many of the site investigators have previously published site-specific data to better characterize regional disease burden. All researchers from participating institutions are recognized as collaborators in the Rotavirus Vaccine Effectiveness and Impact Assessment Network and are included as coauthors of this paper. The data generated are unique to each site and offer valuable insights into the geographic variability in rotavirus disease burden across India. A supplement dedicated to this work was published in the Indian Journal of Pediatrics. The memorandums of understanding established before study initiation clearly defined the roles and responsibilities of each collaborator and, where feasible, included plans for capacity-building at participating institutions. No exceptions to standard ethical requirements were made, and approvals were obtained from each institution’s local ethics committee. As this was an observational study, regulations related to animal welfare, environmental protection and biorisk were not applicable. The study posed no risk of stigmatization, discrimination or personal harm to participants, and informed consent was obtained from parents or caregivers of all enrolled children. This research did not involve any transfer of biological materials, cultural artifacts or traditional knowledge outside the country. Relevant local and regional research has been appropriately cited. No health, safety or security risks to researchers were involved in conducting this study.
Study design
From January 2016 to January 2020, a prospective, active surveillance for rotavirus gastroenteritis was conducted in a network of 31 sentinel hospitals in rural and urban areas of the first nine Indian states to introduce Rotavac (Extended Data Fig. 1). This observational study uses long-term surveillance data from states that introduced rotavirus vaccine to estimate vaccine effectiveness using test negative case control study design and impact of the vaccine is assessed by comparing the rotavirus-positive and rotavirus-negative cases during the pre- and postvaccination periods. Details of the study are described in a published protocol36. A study flow diagram is shown in Extended Data Fig. 2. Briefly, information on all children admitted for gastroenteritis in each sentinel hospital was recorded in an admission logbook, with those aged 0–59 months eligible for this study. Rotaviral diarrhea distinctly exhibits a sex difference, often the ratio being 4:1 for males and females respectively in resource poor settings. Sex of the child plays a key role in analysis and results reported accordingly. The sex of the child enrolled in the study was determined on the basis of biological attribute. The sex of the child was recorded by the study personnel after examination of the child.
A detailed clinical history, sociodemographic information and vaccination history were collected. A stool sample of 5 ml was collected in a sterile screw-capped container and stored at each site at −20 °C until cold chain transported to the Wellcome Trust Research Laboratory, CMC, Vellore. The samples are tested using the Premier Rotaclone, (Meridian Biosciences) commercially available enzyme immunoassay (EIA). According to the manufacturer’s kit insert, the EIA kit used in the study, Premier Rotaclone, (Meridian Biosciences), has a sensitivity of >99% and specificity of 92–97%. However, two other studies have found the Premier Rotaclone to have a sensitivity of 76.8–80.7% and specificity of 100% (refs. 37,38). All EIA-positive samples were genotyped using published methods39,40. All the samples were genotyped using the WHO generic protocol for rotavirus surveillance. For EIA-positive samples that could not be genotyped, VP6 PCR was used to confirm for rotavirus positivity. A Sanger sequencing method was used to sequence the untyped samples and unusual rotavirus strains9,41.
Data management
Clinical and sociodemographic data captured on paper case report forms were collected from each site and sent periodically to CMC, Vellore, where the data were checked and entered into the Structured Query Language software database. The data were analyzed using licensed software’s Stata v.14.2 (StataCorp LLC) and Microsoft Excel v.16.78.
Statistical analysis
Rotavirus vaccine effectiveness
We used the test-negative case–control study design to estimate vaccine effectiveness in age-eligible children. The cases were defined as children with AGE who tested positive for rotavirus infection using EIA, whereas the controls had tested negative42. To define age-eligibility for rotavirus vaccination, only children born 6 weeks before the vaccine introduction date by state or after vaccine introduction were considered eligible. Because our objective was to determine the effectiveness of a full three-dose series of Rotavac, we restricted the vaccine effectiveness analysis to children 6 months of age and older given delays in vaccine dose administration in the Universal Immunization Program in India. Although, the three rotavirus vaccine doses are recommended at 6, 10 and 14 weeks, approximately 90% of eligible and vaccinated children in our study received all three doses of the vaccine by 27 weeks of age (Extended Data Fig. 3). A child was considered vaccinated with a particular dose if the vaccine was given 14 days before admission to the study hospital. The vaccination status of children was determined from a copy of the parent-retained immunization record. Incomplete or unavailable information was also recorded and confirmed with the government held immunization records at either the Health Sub-Centre, Primary Health Centre or the immunization website managed by the Ministry of Health and Family Welfare. Where confirmation of vaccination status was not possible, the child was excluded from the analysis.
To assess a vaccine effectiveness of 40%, we estimated that a minimum of 242 cases of rotavirus AGE were required with a control-to-case ratio of 2:1 and an expected vaccine coverage of 80% among controls36. Additional cases were enrolled to address the secondary objectives specified in the protocol36. Unconditional logistic regression was used to estimate odds ratios (OR) and 95% CIs. We calculated vaccine effectiveness as follows:
The OR was obtained from the regression models. The models built for estimating aVE included the admission hospital, educational status of the mother, assets owned by the family and age of the child in days at the time of hospital admission. Covariates were included in the model if their inclusion changed the OR associated with vaccination by more than 5%. Sex, birth month, birth year, year of hospital admission, hospital of admission and household characteristics were excluded from the final model. Stratified analyses were performed to assess aVE by age, nutritional status, state of residence, severity of diarrhea, dose of the vaccine and by circulating genotypes. An interaction term between vaccination status (completely vaccinated/unvaccinated) and age in months was included in the main model to assess the statistical significance of the resulting vaccine effectiveness. The full model (with the interaction term) was compared with a reduced model (without the interaction term) using a likelihood ratio test. The resulting P value from the likelihood ratio test was used to evaluate model fit. Inclusion of the interaction term of vaccination status and age of the child was not statistically significant and excluded from the final model. All reported aVE estimates were against admission to the hospital for acute rotavirus diarrhea.
To address potential confounding effects, we conducted a sensitivity analysis using various statistical techniques. The methods include an unmatched logistic regression, as adapted in the current study; matched regression analysis, where participants were matched based on relevant covariates before regression; propensity score matched regression, which matches participants based on propensity scores to reduce confounding; propensity score weighted regression, which weights observations based on propensity scores to balance covariates between the vaccine and nonvaccine groups; and estimation of the E value, which calculates the minimum strength of an unmeasured confounder needed to nullify the observed association. Estimation of the E value to measure residual confounding in the regression model was done using the formula
Comparing the main effectiveness estimates across these methods allows us to assess the stability of our findings and validate the statistical approach used in this study43,44.
Rotavirus vaccine impact
Of the 31 sentinel hospitals, 5 had conducted rotavirus surveillance as part of the National Rotavirus Surveillance Network from 2012 onward, based on a protocol similar to that in this study5,39,40. Thus, the assessment of the impact of the vaccine in the states of Andhra Pradesh, Himachal Pradesh, Haryana, Odisha and Tamil Nadu was enabled (Extended Data Fig. 1). We compared the rotavirus-positive and rotavirus-negative cases during the pre- and postvaccination periods. The age distribution and reduction in rates of rotavirus positivity before and after vaccine introduction were also analyzed. An interrupted time series analysis was conducted to demonstrate the effects of vaccine independently from pre-existing trends or other external influences45. This approach helps evaluate both the immediate and long-term impact of vaccine introduction.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
The data used in this study were collected through hospital-based surveillance conducted in early Rotavac-introducing regions of India between 2016 and 2020, using a uniform protocol previously published by the authors. The study includes newly generated data to assess vaccine effectiveness post introduction, as well as pre- and postvaccination data from five hospitals to evaluate vaccine impact. The data during prevaccination period of 2012–2016 were collected as a part of National Rotavirus Surveillance Network using similar protocol. While the corresponding authors retain ownership of the data, portions have been previously published as part of the broader surveillance network5,35. All the data generated and/or analyzed for the findings of this study are openly available via GitHub at https://github.com/drvenkatm/Rotavac_VE_India.git. The dataset includes all relevant variables used in the analysis and is accessible for public use.
Code availability
The code used for data analysis, model estimation and results generation is available via GitHub at https://github.com/drvenkatm/Rotavac_VE_India.git. The repository contains detailed instructions for reproducing the analysis, including dependencies and setup guidelines. All scripts are provided for open use and further research.
Change history
27 October 2025
A Correction to this paper has been published: https://doi.org/10.1038/s41591-025-04062-2
References
Troeger, C. et al. Rotavirus vaccination and the global burden of rotavirus diarrhea among children younger than 5 years. JAMA Pediatr. 172, 958–965 (2018).
Burnett, E., Parashar, U. D. & Tate, J. E. Real-world effectiveness of rotavirus vaccines, 2006–19: a literature review and meta-analysis. Lancet Glob. Health 8, e1195–e1202 (2020).
Varghese, T., Kang, G. & Steele, A. D. Understanding rotavirus vaccine efficacy and effectiveness in countries with high child mortality. Vaccines 10, 346 (2022).
Clark, A. et al. Estimating global, regional and national rotavirus deaths in children aged <5 years: current approaches, new analyses and proposed improvements. PLoS ONE 12, e0183392 (2017).
Kumar, C. G. et al. Epidemiology of rotavirus diarrhea among children less than 5 years hospitalized with acute gastroenteritis prior to rotavirus vaccine introduction in India. Vaccine 38, 8154–8160 (2020).
Bhandari, N. et al. Efficacy of a monovalent human-bovine (116E) rotavirus vaccine in Indian infants: a randomised, double-blind, placebo-controlled trial. Lancet 383, 2136–2143 (2014).
Malik, A. et al. Introducing rotavirus vaccine in the Universal Immunization Programme in India: from evidence to policy to implementation. Vaccine 37, 5817–5824 (2019).
John, T. J. India’s research contributions towards polio eradication (1965–2015). Indian Pediatr. 53, S38–S43 (2016).
Varghese, T. et al. Rotavirus strain distribution before and after introducing rotavirus vaccine in India. Pathogens 10, 416 (2021).
Ali, H. A. et al. Vaccine equity in low and middle income countries: a systematic review and meta-analysis. Int J. Equity Health 21, 82 (2022).
Patel, M. M. et al. Effectiveness of monovalent rotavirus vaccine in Bolivia: case–control study. BMJ 346, f3726 (2013).
Groome, M. J. et al. Effectiveness of monovalent human rotavirus vaccine against admission to hospital for acute rotavirus diarrhoea in South African children: a case–control study. Lancet Infect. Dis. 14, 1096–1104 (2014).
Cotes-Cantillo, K. et al. Effectiveness of the monovalent rotavirus vaccine in Colombia: a case–control study. Vaccine 32, 3035–3040 (2014).
Bar-Zeev, N. et al. Effectiveness of a monovalent rotavirus vaccine in infants in Malawi after programmatic roll-out: an observational and case–control study. Lancet Infect. Dis. 15, 422–428 (2015).
Bar-Zeev, N. et al. Population impact and effectiveness of monovalent rotavirus vaccination in urban Malawian children 3 years after vaccine introduction: ecological and case–control analyses. Clin. Infect. Dis. 62, S213–S219 (2016).
Gastañaduy, P. A. et al. Effectiveness of monovalent rotavirus vaccine after programmatic implementation in Botswana: a multisite prospective case–control study. Clin. Infect. Dis. 62, S161–S167 (2016).
Gastañaduy, P. A. et al. Effectiveness of monovalent and pentavalent rotavirus vaccines in Guatemala. Clin. Infect. Dis. 62, S121–S126 (2016).
Tate, J. E. et al. Effectiveness of pentavalent rotavirus vaccine under conditions of routine use in Rwanda. Clin. Infect. Dis. 62, S208–S212 (2016).
Armah, G. et al. Impact and effectiveness of monovalent rotavirus vaccine against severe rotavirus diarrhea in Ghana. Clin. Infect. Dis. 62, S200–S207 (2016).
Gheorghita, S. et al. Impact of rotavirus vaccine introduction and vaccine effectiveness in the republic of Moldova. Clin. Infect. Dis. 62, S140–S146 (2016).
Pringle, K. D. et al. Sustained effectiveness of rotavirus vaccine against very severe rotavirus disease through the second year of life, Bolivia 2013–2014. Clin. Infect. Dis. 62, S115–S120 (2016).
Abeid, K. A. et al. Monovalent rotavirus vaccine effectiveness and impact on rotavirus hospitalizations in Zanzibar, Tanzania: data from the first 3 years after introduction. J. Infect. Dis. 215, 183–191 (2017).
Khagayi, S. et al. Effectiveness of monovalent rotavirus vaccine against hospitalization with acute rotavirus gastroenteritis in Kenyan children. Clin. Infect. Dis. 70, 2298–2305 (2020).
Sahakyan, G. et al. Impact and effectiveness of monovalent rotavirus vaccine in Armenian Children. Clin. Infect. Dis. 62, S147–S154 (2016).
Zaman, K. et al. Effectiveness of a live oral human rotavirus vaccine after programmatic introduction in Bangladesh: a cluster-randomized trial. PLOS Med. 14, e1002282 (2017).
Rosettie, K. L. et al. Indirect rotavirus vaccine effectiveness for the prevention of rotavirus hospitalization: a systematic review and meta-analysis. Am. J. Tropical Med. Hyg. 98, 1197–1201 (2018).
Mujuru, H. A. et al. Cost estimates of diarrhea hospitalizations among children <5 years old in Zimbabwe. Vaccine 38, 6735–6740 (2020).
Chissaque, A. et al. Effectiveness of monovalent rotavirus vaccine in Mozambique, a country with a high burden of chronic malnutrition. Vaccines 10, 449 (2022).
Rheingans, R. et al. Estimated impact and cost-effectiveness of rotavirus vaccination in India: effects of geographic and economic disparities. Vaccine 32, A140–A150 (2014).
Antoni, S. et al. Rotavirus genotypes in children under five years hospitalized with diarrhea in low and middle-income countries: results from the WHO-coordinated Global Rotavirus Surveillance Network. PLOS Glob. Public Health 3, e0001358 (2023).
Leshem, E. et al. Distribution of rotavirus strains and strain-specific effectiveness of the rotavirus vaccine after its introduction: a systematic review and meta-analysis. Lancet Infect. Dis. 14, 847–856 (2014).
Parker, E. P. et al. Causes of impaired oral vaccine efficacy in developing countries. Future Microbiol. 13, 97–118 (2017).
Cowley, D. et al. Immunogenicity of four doses of oral poliovirus vaccine when co-administered with the human neonatal rotavirus vaccine (RV3-BB). Vaccine 37, 7233–7239 (2019).
Reddy, S. et al. Safety monitoring of ROTAVAC vaccine and etiological investigation of intussusception in India: study protocol. BMC Public Health 18, 898 (2018).
Giri, S. et al. Rotavirus gastroenteritis in Indian children < 5 years hospitalized for diarrhoea, 2012 to 2016. BMC Public Health 19, 69 (2019).
Nair, N. P. et al. Rotavirus vaccine impact assessment surveillance in India: protocol and methods. BMJ Open 9, e024840 (2019).
Gautam, R., Lyde, F., Esona, M. D., Quaye, O. & Bowen, M. D. Comparison of PremierTM Rotaclone®, ProSpecTTM, and RIDASCREEN® rotavirus enzyme immunoassay kits for detection of rotavirus antigen in stool specimens. J. Clin. Virol. 58, 292–294 (2013).
Lagare, A. et al. Diagnostic accuracy of VIKIA® Rota-Adeno and PremierTM Rotaclone® tests for the detection of rotavirus in Niger. BMC Res Notes 10, 505 (2017).
Kang, G. et al. Multicenter, hospital-based surveillance of rotavirus disease and strains among indian children aged <5 years. J. Infect. Dis. 200, S147–S153 (2009).
Babji, S. et al. Multi-center surveillance of rotavirus diarrhea in hospitalized children <5 years of age in India, 2009–2012. Vaccine 32, A10–A12 (2014).
Manual of Rotavirus Detection and Characterization Methods (World Health Organization, 2009, accessed 24 September 2025); https://apps.who.int/iris/bitstream/handle/10665/70122/WHO_IVB_08.17_eng.pdf?sequence=1
Tate, J. E. et al. Use of patients with diarrhea who test negative for rotavirus as controls to estimate rotavirus vaccine effectiveness through case–control studies. Clin. Infect. Dis. 62, S106–S114 (2016).
VanderWeele, T. J. & Ding, P. Sensitivity analysis in observational research: introducing the E-value. Ann. Intern Med 167, 268–274 (2017).
Austin, P. C. & Stuart, E. A. Moving towards best practice when using inverse probability of treatment weighting (IPTW) using the propensity score to estimate causal treatment effects in observational studies. Stat. Med 34, 3661–3679 (2015).
Hungerford, D. et al. Rotavirus vaccine impact and socioeconomic deprivation: an interrupted time-series analysis of gastrointestinal disease outcomes across primary and secondary care in the UK. BMC Med. 16, 10 (2018).
Acknowledgements
This study was funded by the Centers for Disease Control and Prevention Foundation, Atlanta, GA, USA (subcontract to Christian Medical College, Vellore grant no. MOA#871-15SC) and the Bill and Melinda Gates Foundation to the Translational Health Science and Technology Institute (grant no. OPP1165083). The grant was awarded to G.K. The authors recognize the invaluable contribution of staff from participating institutions and the parents of enrolled children.
Author information
Authors and Affiliations
Consortia
Contributions
G.K., U.D.P., J.E.T. and V.R.M. conceived and designed the study. N.P.N., S.N.R., R.A. and M.D.G. led the study implementation and coordinated and obtained the clinical data. G.K., S.G., P.H. and S.A.K. acquired the laboratory data. J.E.T., U.D.P., J.M., N.P.N., G.K., S.N.R., V.T. and V.R.M. analyzed the data. N.P.N. wrote the first draft. The Collaborators of the rotavirus vaccine effectiveness and impact assessment network coordinated study at their respective sites, obtained clinical data, and analyzed the data site wise. All authors critically reviewed drafts of the paper and gave final approval of the submitted version.
Corresponding author
Ethics declarations
Competing interests
N.P.N. is the recipient of a fellowship from the Indian Council of Medical Research, New Delhi. G.K. is now an employee of the Bill and Melinda Gates Foundation. The other authors declare no competing interests. The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention. The funders had no role in study design, collection, analysis and interpretation of data, writing of the report, or the decision to submit the paper for publication. The corresponding author had full access to all the data from the study and had final responsibility for the decision to submit for publication. No authors were affiliated with Bharat Biotech.
Peer review
Peer review information
Nature Medicine thanks Pranab Chatterjee, Khitam Muhsen and Nadim Sharif for their contribution to the peer review of this work. Primary Handling Editor: Lia Parkin, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Map of India showing the phased introduction by states of RotavacTM vaccine in the Universal Immunization Program in India from 2016 to 2018.
This map was generated using licensed version of ArcGIS® software to demonstrate the states in India that were part of the surveillance and introduction times of rotavirus vaccine in each of the participating states. Vaccine effectiveness surveillance data were collected from all nine states from January 2016 to January 2020. Data for assessing the impact of the vaccine were obtained from four states with pre-vaccination data from September 2012 to April 2016, post-vaccination data from May 2016 to June 2020, and from Tamil Nadu from 2018 to January 2020. A uniform protocol was followed throughout the surveillance period.
Extended Data Fig. 2 Study flow diagram.
Shows the protocol followed at all the study sites for enrolment and collection of data. EIA- enzyme immunoassay; MoHFW- Ministry of Health and Family Welfare, Government of India.
Extended Data Fig. 3 Timeliness of rotavirus and pentavalent vaccines among age-eligible children in the Universal Immunization Program in India.
Rotavirus vaccines and pentavalent vaccines are administered to children at 6, 10, and 14 weeks of age in the Universal Immunization Program in India. This figure shows the comparison between cumulative percentages of vaccinated children across timelines during the surveillance. A delay is observed in the completion of the series of both vaccines.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nair, N.P., Reddy, S.N., Giri, S. et al. Impact of the indigenous rotavirus vaccine Rotavac in the Universal Immunization Program in India during 2016–2020. Nat Med 31, 3871–3878 (2025). https://doi.org/10.1038/s41591-025-03998-9
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41591-025-03998-9