Abstract
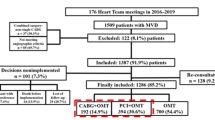
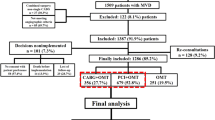
In patients with stable coronary artery disease (CAD), the long-term benefits of revascularization over medical therapy remain unclear. In the Fractional Flow Reserve versus Angiography for Multivessel Evaluation 2 trial, patients with hemodynamically significant stenoses (fractional flow reserve (FFR) ≤ 0.80) were randomized to receive FFR-guided percutaneous coronary intervention (PCI) plus medical therapy (n = 447) or medical therapy alone (n = 441). At 5 years, FFR-guided PCI reduced the risk of the primary composite outcome of time to death, myocardial infarction or urgent revascularization, largely because of fewer urgent revascularizations. We now report the long-term clinical outcomes from this trial. Sixteen hospitals, contributing 748 randomized patients (161 women, 21.5%), participated in the long-term follow-up. The primary composite outcome was analyzed hierarchically using the unstratified win ratio, which addressed differential missingness of data on nonfatal outcomes in deceased patients by prioritizing comparisons on time to death. At a median follow-up of 11.2 years, the primary endpoint occurred in 150 of 447 patients (33.6%) in the PCI group versus 182 of 441 (41.3%) in the medical therapy group. PCI was superior in 29.2% of comparisons, medical therapy in 23.3%, and the two groups were tied in 47.5%, resulting in a win ratio of 1.25 in favor of PCI (95% confidence interval (CI) 1.01–1.56, P = 0.043). The corresponding win difference was 5.9% (95% CI 0.2–11.6), and the number needed to treat was 17 (95% CI 9–500). Win ratios were 0.88 for all-cause death (95% CI 0.66–1.17), 1.50 for myocardial infarction (95% CI 0.98–2.31) and 4.57 for urgent revascularization (95% CI 2.53–8.24). During long-term follow-up, FFR-guided PCI in patients with stable CAD and hemodynamically significant stenoses reduced the composite of death, myocardial infarction or urgent revascularization, primarily because of fewer urgent revascularizations. These long-term findings reaffirm the efficacy of FFR-guided PCI over medical therapy in patients with stable CAD. ClinicalTrials.gov registration: NCT06159231.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The data underlying this study consist of clinical information from human participants. Participants provided informed consent for use of their data in this specific study but not for unrestricted public sharing. As such, open access to the dataset would compromise participant confidentiality and breach ethical and legal obligations under applicable data protection regulations. To balance participant privacy with scientific transparency, anonymized derived data will be made available upon reasonable request to qualified researchers with a methodologically sound proposal, subject to approval by the sponsoring institutions and, if required, relevant ethics committees. Requests for data should be sent to the corresponding authors, with a response expected within 4 weeks.
References
Tonino, P. A. L. et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N. Engl. J. Med. 360, 213–224 (2009).
De Bruyne, B. et al. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N. Engl. J. Med. 367, 991–1001 (2012).
De Bruyne, B. et al. Fractional flow reserve-guided PCI for stable coronary artery disease. N. Engl. J. Med. 371, 1208–1217 (2014).
Xaplanteris, P. et al. Five-year outcomes with PCI guided by fractional flow reserve. N. Engl. J. Med. 379, 250–259 (2018).
Al-Lamee, R. et al. Percutaneous coronary intervention in stable angina (ORBITA): a double-blind, randomised controlled trial. Lancet 391, 31–40 (2018).
Rajkumar, C. A. et al. A placebo-controlled trial of percutaneous coronary intervention for stable angina. N. Engl. J. Med. 389, 2319–2330 (2023).
Maron, D. J. et al. Initial invasive or conservative strategy for stable coronary disease. N. Engl. J. Med. 382, 1395–1407 (2020).
Al-Lamee, R. et al. Fractional flow reserve and instantaneous wave-free ratio as predictors of the placebo-controlled response to percutaneous coronary intervention in stable single-vessel coronary artery disease: physiology-stratified analysis of ORBITA. Circulation 138, 1780–1792 (2018).
Chaitman, B. R. et al. Myocardial infarction in the ISCHEMIA trial: impact of different definitions on incidence, prognosis, and treatment comparisons. Circulation 143, 790–804 (2021).
Johnson, N. P. et al. Prognostic value of fractional flow reserve: linking physiologic severity to clinical outcomes. J. Am. Coll. Cardiol. 64, 1641–1654 (2014).
Collet, C. et al. Influence of pathophysiologic patterns of coronary artery disease on immediate percutaneous coronary intervention outcomes. Circulation 150, 586–597 (2024).
Collet, C. et al. Measurement of hyperemic pullback pressure gradients to characterize patterns of coronary atherosclerosis. J. Am. Coll. Cardiol. 74, 1772–1784 (2019).
Cordoba, G., Schwartz, L., Woloshin, S., Bae, H. & Gøtzsche, P. C. Definition, reporting, and interpretation of composite outcomes in clinical trials: systematic review. BMJ 341, c3920 (2010).
Pocock, S. J., Ariti, C. A., Collier, T. J. & Wang, D. The win ratio: a new approach to the analysis of composite endpoints in clinical trials based on clinical priorities. Eur. Heart J. 33, 176–182 (2012).
Pocock, S. J., Gregson, J., Collier, T. J., Ferreira, J. P. & Stone, G. W. The win ratio in cardiology trials: lessons learnt, new developments, and wise future use. Eur. Heart J. 45, 4684–4699 (2024).
Gasparyan, S. B., Folkvaljon, F., Bengtsson, O., Buenconsejo, J. & Koch, G. G. Adjusted win ratio with stratification: calculation methods and interpretation. Stat. Methods Med. Res. 30, 580–611 (2021).
Beretta, L. & Santaniello, A. Nearest neighbor imputation algorithms: a critical evaluation. BMC Med. Inform. Decis. Mak. 16, 74 (2016).
Author information
Authors and Affiliations
Contributions
The study was conceptualized by B.D.B., N.H.J.P., W.F.F., C.C. and P.J. All authors were involved in data curation. Study methodology was developed by C.C., B.D.B. and P.J. Data analysis was performed by T.V.P., B.R.d.C., T. Mizukami and T. Mahendiran, under the supervision of B.D.B. and P.J. The original draft was written by C.C., T. Mahendiran, B.D.B. and P.J. All authors contributed to the review, editing and approval of the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
C.C. reports receiving research grants from Biosensor, Coroventis Research, Medis Medical Imaging, Pie Medical Imaging, CathWorks, Boston Scientific, Siemens, HeartFlow Inc. and Abbott Vascular and consultancy fees from HeartFlow Inc., OpSens, Abbott Vascular and Philips Volcano. T. Mahendiran is supported by a grant from the Swiss National Science Foundation (SNSF). W.F.F. has received institutional research support from Abbott Vascular, Boston Scientific and Medtronic; has a consulting relationship with CathWorks and Siemens; and has stock options with HeartFlow. N.H.J.P. has received institutional research grants from Abbott; has consulting relationships with and receives fees from Abbott and Coroventis; has equity in ASML Holding N.V., General Electric, HeartFlow and Philips; is a member of the Scientific Advisory Board of HeartFlow; and has patents pending in the field of the coronary microcirculation and aortic valve stenosis. B.D.B. has a consulting relationship with Boston Scientific, Abbott Vascular, CathWorks, Siemens and Coroventis Research; receives research grants from Abbott Vascular, Coroventis Research, CathWorks and Boston Scientific; and holds minor equities in Philips Volcano, Siemens, GE Healthcare, Edwards Lifesciences, HeartFlow, Sanofi and Celyad. The other authors declare no competing interests.
Peer review
Peer review information
Nature Medicine thanks Brian Ko and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Michael Basson, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Sensitivity analyses based on the incidence rate ratio.
Prespecified sensitivity analyses using Poisson regression with robust standard errors to estimate incidence rate ratios with 95% confidence intervals. In the model without data imputation, participants who dropped out or were lost to follow-up were censored at the time of the event or at the time of last available follow-up. IRR, incidence rate ratio. 95% CI, 95% confidence interval. PCI, percutaneous coronary intervention. PY, person-years. Two-sided P-values are from Wald tests of the regression coefficients in the Poisson regression models.
Extended Data Fig. 2 Post hoc sensitivity analyses based on the hazard ratio.
Post hoc sensitivity analyses using Cox proportional hazards models to estimate hazard ratios with 95% confidence intervals. In the model without data imputation, participants who dropped out or were lost to follow-up were censored at the time of the event or at the time of last available follow-up. HR, hazard ratio. 95% CI, 95% confidence interval. PCI, percutaneous coronary intervention. PY, person-years. Two-sided P-values are from Wald tests of the regression coefficients in the Cox proportional hazards models.
Extended Data Fig. 3 Time-to-event curves after multiple imputation.
Cumulative incidence curves for primary composite outcome (a) and its components (b) all-cause death, (c) myocardial infarction, and (d) urgent revascularization, after multiple imputation. IRR, incidence rate ratio. 95% CI, 95% confidence interval. PCI, percutaneous coronary intervention.
Extended Data Fig. 4 Summary of the main study findings.
The two-sided P-value for the win ratio was calculated using a Z test based on the Finkelstein and Schoenfeld method.
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1 and 2, study protocol and statistical analysis plan.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Collet, C., Mahendiran, T., Fearon, W.F. et al. Fractional flow reserve-guided percutaneous coronary intervention versus medical therapy for stable coronary artery disease: long-term results of the FAME 2 trial. Nat Med 32, 318–324 (2026). https://doi.org/10.1038/s41591-025-04132-5
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41591-025-04132-5