Abstract
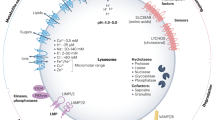
Metabolism unfolds within specific organelles in eukaryotic cells. Lysosomes are highly metabolically active organelles, and their metabolic states dynamically influence signal transduction, cellular homeostasis and organismal physiopathology. Despite the importance of lysosomal metabolism, a method for its in vivo measurement is currently lacking. Here we report a fluorescence-detected mid-infrared photothermal microscope (FILM) implemented with optical boxcar demodulation, artificial intelligence-assisted data denoising and spectral deconvolution, to map metabolic activity and composition of individual lysosomes in living cells and organisms. Using this method, we uncovered lipolysis and proteolysis heterogeneity across lysosomes within the same cell, as well as early-onset lysosomal dysfunction during organismal aging. In addition, we discovered organelle-level metabolic changes associated with diverse lysosomal storage diseases. This method holds the broad potential to profile metabolic fingerprints of individual organelles within their native context and quantitatively assess their dynamic changes under different physiological and pathological conditions, providing a high-resolution chemical cellular atlas.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data are available in this article and its Supplementary Information and via Figshare at https://doi.org/10.6084/m9.figshare.31302607 (ref. 56).
Code availability
Code supporting the current study, including SPEND denoising and spectral unmixing, can be accessed via GitHub at https://github.com/buchenglab under the GNU General Public License v3.0. The SPEND is also available via Zenodo at https://doi.org/10.5281/zenodo.19236367 (ref. 57).
References
DeBerardinis, R. J. & Thompson, C. B. Cellular metabolism and disease: what do metabolic outliers teach us?. Cell 148, 1132–1144 (2012).
Amorim, J. A. et al. Mitochondrial and metabolic dysfunction in ageing and age-related diseases. Nat. Rev. Endocrinol. 18, 243–258 (2022).
Abu-Remaileh, M. et al. Lysosomal metabolomics reveals V-ATPase- and mTOR-dependent regulation of amino acid efflux from lysosomes. Science 358, 807–813 (2017).
Laqtom, N. N. et al. CLN3 is required for the clearance of glycerophosphodiesters from lysosomes. Nature 609, 1005–1011 (2022).
Yu, Y. et al. Organelle proteomic profiling reveals lysosomal heterogeneity in association with longevity. eLife 13, e85214 (2024).
Chen, W. W., Freinkman, E., Wang, T., Birsoy, K. & Sabatini, D. M. Absolute quantification of matrix metabolites reveals the dynamics of mitochondrial metabolism. Cell 166, 1324–1337 (2016).
Chow, A., Toomre, D., Garrett, W. & Mellman, I. Dendritic cell maturation triggers retrograde MHC class II transport from lysosomes to the plasma membrane. Nature 418, 988–994 (2002).
Johnson, D. E., Ostrowski, P., Jaumouillé, V. & Grinstein, S. The position of lysosomes within the cell determines their luminal pH. J. Cell Biol. 212, 677–692 (2016).
Deng, D. et al. Quantitative profiling pH heterogeneity of acidic endolysosomal compartments using fluorescence lifetime imaging microscopy. Mol. Biol. Cell 36, br8 (2025).
Valm, A. M. et al. Applying systems-level spectral imaging and analysis to reveal the organelle interactome. Nature 546, 162–167 (2017).
Marvin, J. S. et al. iATPSnFR2: a high-dynamic-range fluorescent sensor for monitoring intracellular ATP. Proc. Natl Acad. Sci. USA 121, e2314604121 (2024).
Zhang, M. et al. Monitoring the dynamic regulation of the mitochondrial GTP-to-GDP ratio with a genetically encoded fluorescent biosensor. Angew. Chem. Int. Ed. Engl. 61, e202201266 (2022).
Liu, X., Shi, L., Zhao, Z., Shu, J. & Min, W. VIBRANT: spectral profiling for single-cell drug responses. Nat. Methods 21, 501–511 (2024).
Shi, L. et al. Mid-infrared metabolic imaging with vibrational probes. Nat. Methods 17, 844–851 (2020).
Zhang, D. et al. Depth-resolved mid-infrared photothermal imaging of living cells and organisms with submicrometer spatial resolution. Sci. Adv. 2, e1600521 (2016).
Fu, P. et al. Super-resolution imaging of non-fluorescent molecules by photothermal relaxation localization microscopy. Nat. Photonics 17, 330–337 (2023).
Tamamitsu, M. et al. Mid-infrared wide-field nanoscopy. Nat. Photonics 18, 738–743 (2024).
Yin, J. et al. Video-rate mid-infrared photothermal imaging by single-pulse photothermal detection per pixel. Sci. Adv. 9, eadg8814 (2023).
He, H. et al. Mapping enzyme activity in living systems by real-time mid-infrared photothermal imaging of nitrile chameleons. Nat. Methods 21, 342–352 (2024).
Xia, Q. et al. Click-free imaging of carbohydrate trafficking in live cells using an azido photothermal probe. Sci. Adv. 10, eadq0294 (2024).
Sakaguchi, R., Kiyonaka, S. & Mori, Y. Fluorescent sensors reveal subcellular thermal changes. Curr. Opin. Biotechnol. 31, 57–64 (2015).
Zhou, J., del Rosal, B., Jaque, D., Uchiyama, S. & Jin, D. Advances and challenges for fluorescence nanothermometry. Nat. Methods 17, 967–980 (2020).
Zhang, Y. et al. Fluorescence-detected mid-infrared photothermal microscopy. J. Am. Chem. Soc. 143, 11490–11499 (2021).
Li, M. et al. Fluorescence-detected mid-infrared photothermal microscopy. J. Am. Chem. Soc. 143, 10809–10815 (2021).
Prater, C. B. et al. Widefield super-resolution infrared spectroscopy and imaging of autofluorescent biological materials and photosynthetic microorganisms using fluorescence detected photothermal infrared (FL-PTIR). Appl. Spectrosc. 78, 1208–1219 (2024).
Folick, A. et al. Lysosomal signaling molecules regulate longevity in Caenorhabditis elegans. Science 347, 83–86 (2015).
Platt, F. M., d’Azzo, A., Davidson, B. L., Neufeld, E. F. & Tifft, C. J. Lysosomal storage diseases. Nat. Rev. Dis. Primers 4, 27 (2018).
Ballabio, A. & Bonifacino, J. S. Lysosomes as dynamic regulators of cell and organismal homeostasis. Nat. Rev. Mol. Cell Biol. 21, 101–118 (2020).
Settembre, C. & Perera, R. M. Lysosomes as coordinators of cellular catabolism, metabolic signalling and organ physiology. Nat. Rev. Mol. Cell Biol. 25, 223–245 (2024).
Ding, G. et al. Self-supervised elimination of non-independent noise in hyperspectral imaging. Newton 1, 100195 (2025).
Lin, H. et al. Microsecond fingerprint stimulated Raman spectroscopic imaging by ultrafast tuning and spatial-spectral learning. Nat. Commun. 12, 3052 (2021).
Lin, H. et al. Label-free nanoscopy of cell metabolism by ultrasensitive reweighted visible stimulated Raman scattering. Nat. Methods 22, 1040–1050 (2025).
Zhu, H. et al. Metabolomic profiling of single enlarged lysosomes. Nat. Methods 18, 788–798 (2021).
Jaumot, J., de Juan, A. & Tauler, R. MCR-ALS GUI 2.0: new features and applications. Chemometr. Intell. Lab. Syst. 140, 1–12 (2015).
Jamalpoor, A., Othman, A., Levtchenko, E. N., Masereeuw, R. & Janssen, M. J. Molecular mechanisms and treatment options of nephropathic cystinosis. Trends Mol. Med. 27, 673–686 (2021).
Savini, M. et al. Lysosome lipid signalling from the periphery to neurons regulates longevity. Nat. Cell Biol. 24, 906–916 (2022).
Yim, W. W. & Mizushima, N. Lysosome biology in autophagy. Cell Discov. 6, 6 (2020).
Byrnes, K. et al. Therapeutic regulation of autophagy in hepatic metabolism. Acta Pharm. Sin. B 12, 33–49 (2022).
Lloyd-Evans, E. et al. Niemann–Pick disease type C1 is a sphingosine storage disease that causes deregulation of lysosomal calcium. Nat. Med. 14, 1247–1255 (2008).
Samolis, P. D., Zhu, X. & Sander, M. Y. Time-resolved mid-infrared photothermal microscopy for imaging water-embedded axon bundles. Anal. Chem. 95, 16514–16521 (2023).
Zhang, L. et al. Spectral tracing of deuterium for imaging glucose metabolism. Nat. Biomed. Eng. 3, 402–413 (2019).
Chen, W. W. et al. Spectroscopic coherent Raman imaging of Caenorhabditis elegans reveals lipid particle diversity. Nat. Chem. Biol. 16, 1087–1095 (2020).
Tan, Y., Lin, H. & Cheng, J. X. Profiling single cancer cell metabolism via high-content SRS imaging with chemical sparsity. Sci. Adv. 9, eadg6061 (2023).
Bi, S. et al. Imaging metabolic flow of water in plants with isotope-traced stimulated Raman scattering microscopy. Adv. Sci. 11, e2407543 (2024).
Xiong, H. et al. Stimulated Raman excited fluorescence spectroscopy and imaging. Nat. Photonics 13, 412–417 (2019).
Whaley-Mayda, L., Guha, A., Penwell, S. B. & Tokmakoff, A. Fluorescence-encoded infrared vibrational spectroscopy with single-molecule sensitivity. J. Am. Chem. Soc. 143, 3060–3064 (2021).
Wang, H. et al. Bond-selective fluorescence imaging with single-molecule sensitivity. Nat. Photonics 17, 846–855 (2023).
Zhao, J. et al. Mid-infrared chemical imaging of intracellular tau fibrils using fluorescence-guided computational photothermal microscopy. Light Sci. Appl. 12, 147 (2023).
Guo, Z. et al. Structural mapping of protein aggregates in live cells modeling Huntington’s disease. Angew. Chem. Int. Ed. Engl. 63, e202408163 (2024).
Gros, F. & Muller, S. The role of lysosomes in metabolic and autoimmune diseases. Nat. Rev. Nephrol. 19, 366–383 (2023).
Shen, D. et al. Lipid storage disorders block lysosomal trafficking by inhibiting a TRP channel and lysosomal calcium release. Nat. Commun. 3, 731 (2012).
Yin, J. et al. Mid-infrared energy deposition spectroscopy. Phys. Rev. Lett. 134, 093804 (2025).
Stiernagle, T. Maintenance of C. elegans. In WormBook: The Online Review of C. elegans Biology (ed. Fay, D.) 1–11 (WormBook, 2006).
Rual, J. F. et al. Toward improving Caenorhabditis elegans phenome mapping with an ORFeome-based RNAi library. Genome Res. 14, 2162–2168 (2004).
Davis, O. B. et al. NPC1–mTORC1 signaling couples cholesterol sensing to organelle homeostasis and is a targetable pathway in Niemann–Pick type C. Dev. Cell 56, 260–276 (2021).
Ao, J. Data for ‘FILM: mapping organellar metabolism by mid-infrared photothermal modulated fluorescence’. Figshare https://doi.org/10.6084/m9.figshare.31302607 (2026).
Ao, J. SPEND code for ‘FILM: mapping organellar metabolism by mid-infrared photothermal modulated fluorescence’. Zenodo https://doi.org/10.5281/zenodo.19236367 (2026).
Acknowledgements
We thank L. Chantranupong and E. Ozsen (Boston University) for helpful discussions on lysosomal metabolism, R. Zoncu (University of California, Berkeley) for providing the HEK293 NPC1KO and HEK293 WT cell lines and D. Kim and S. Fischer (Boston Children’s Hospital) for lending us a stereo microscope. This work was supported by the National Institutes of Health (NIH) R35GM136223 (to J.-X.C.) and Howard Hughes Medical Institute (to M.C.W.). The authors thank the Visiting Scientist Program at HHMI Janelia Research Campus.
Author information
Authors and Affiliations
Contributions
J.A., J.Y., J.-X.C. and M.C.W. conceived the project. J.A. and J.Y. constructed the FILM system. M.C.W., Y.G. and M.S. prepared the C. elegans samples. J.A. prepared mammalian cell samples. B.W. prepared the S. flexneri sample. B.G. prepared the S. aureus sample. J.A. collected and analyzed data. H.L. and G.D. aided the SPEND and MCR–LASSO analysis. D.D., Q.X. and Z.G. contributed to the data analysis. B.L. contributed to the standards measurement during the revision. J.A., J.Y., J.-X.C. and M.C.W. wrote the paper with input and approval from all authors. J.-X.C. and M.C.W. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
J.-X.C. declares financial interest with Photothermal Spectroscopy Corp. at Santa Barbara. The other authors declare no competing interests.
Peer review
Peer review information
Nature Methods thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Nina Vogt, in collaboration with the Nature Methods team. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Optical boxcar strategy enhances the signal by shifting high odd-order harmonics to detected frequency.
a, Principle of the higher order harmonics shifting. b, FILM signal of Shigella flexneri expressing GFP at different duty cycle (n = 10 independent measurements). Statistical data are presented as mean ± s.d. Scale bar: 10 μm.
Extended Data Fig. 2 Optical boxcar strategy suppressed solvent background by harnessing the differential thermal dynamics between particles and water medium.
a, Photothermal dynamics of LipiRed labelled lipid droplets under different IR absorption peaks. Data are presented as mean ± s.d. The shaded area represents the standard deviation from three independent measurements, and the solid line indicates an exponential fit to the mean curve. b, The pulse pair serves as the gating windows to capture the time-resolved fluorescence signal, which is less sensitive to slow dynamic processes. Scale bar: 10 μm.
Extended Data Fig. 3 Spectral fidelity verification.
a, Spectral comparison of FILM and scattering-based MIP (Sc-MIP) with Rhodamine 6 G labelled S. aureus. b, Spectral comparison of FILM and ATR-FTIR with LysoSensor DND189 stained DMSO.
Extended Data Fig. 4 Head-to-head comparison of SPEND with BM4D and Noise2Void (N2V).
a, FILM images of lysosomes were acquired with IR at 1711 cm−1 and 1797 cm−1 before and after three denoising algorithms. b, the intensity profiles along the red dotted lines indicated in a. c, compares the raw, uncalibrated FILM spectra before and after three denoising algorithms. d, the quantification of image SNR and spectral SNR before and after three denoising algorithms (n = 13 lysosomes). In d, the boxes show the IQR, the centerlines indicate medians and the lines outside the boxes extend to 1.5 times the IQR. Scale bar: 10 μm.
Extended Data Fig. 5 Physiological validation of the 1587 and 1711 cm−1 features.
a, Quantification of the 1587 cm−1 intensity for individual lysosomes (n = 114 for L4440, n = 161 for ctns-1 derived from six to eight independent biological experiments; Two-sided two-sample t-test: *, P = 0.013). b, Quantification of the 1711 cm−1 peak intensity for individual lysosomes (n = 177 for WT, n = 200 for lipl-4 Tg derived from six to eight independent biological experiments; Two-sided two-sample t-test: ****, P = 2.14 × 10−52). c, Ratio of the 1711 cm−1 (free fatty acids) to 1741 cm−1 (lipid ester) peaks (n = 177 for WT, n = 200 for lipl-4 Tg derived from six to eight independent biological experiments; Two-sided two-sample t-test: ****, P = 3.11 × 10−18). Each point represents a single lysosome, with box plots indicating the median, interquartile range, and whiskers representing 1.5× IQR.
Extended Data Fig. 6 Visualization of two ratios and class discrimination.
a, Pixel-wise scatter plot of two calculated intensity ratios (200 × 200 pixels) (n = 3546 pixels for lysosomes, n = 32907 pixels for surrounding regions). b, The parallel set shows the relationship between two spectral ratios (1587 cm−1/1649 cm−1 and 1711 cm−1/1741 cm−1) and the class separation of lysosomes (red) and surrounding region (blue). The curves represent different data points from the corresponding classes. In a, the boxes show the IQR, the centerlines indicate medians and the lines outside the boxes extend to 1.5 times the IQR.
Extended Data Fig. 7 Correlations of lysosomal hydrolytic activity with lysosomal size and fluorescence intensity.
a, The correlation between lysosomal proteolytic activity and size is not notable with a Pearson coefficient of 0.48. b, The Pearson coefficient of lysosomal lipolytic activity and size is 0.44. c, The correlation between lysosomal proteolytic activity and fluorescence intensity is not notable with a Pearson coefficient of 0.49. d, The Pearson coefficient of lysosomal lipolytic activity and fluorescence intensity is 0.43.
Extended Data Fig. 8 Hydrolytic heterogeneity of lysosomes in mammalian cells.
a, Fluorescent and FILM images at 1587 and 1649 cm-1 of LysoSensor DND189 labelled HEK293T cells. b, Ratio-metric mapping of intensity ratios at 1587/1649 cm-1 and 1711/1741 cm-1. Scale bar: 10 μm. Representative results are shown from three independent experiments.
Extended Data Fig. 9 t-SNE–based classification of WT lysosomal spectra and identification of functional subtypes.
a, Within-individual heterogeneity. For each spectrum, the standard deviation (SD) across wavenumbers was calculated; Mean_SD denotes the average SD per worm (y-axis × 10-3, x-axis = Worm ID). b, t-SNE embedding (one dot per spectrum) colored by individual worm. Three regions are drawn: C1 (red), C2 (blue), C3 (green), corresponding to three clusters of ‘high proteolytic activity’, ‘high lipolytic activity’, and ‘high activity in both’. c, Cluster-specific quantitative analysis of activity ratios at 1587/1649 cm−1 (proteolytic activity) and 1711/1741 cm−1 (lipolytic activity) of lysosomes (n = 71 for C1 group, n = 211 for C2 group, n = 110 for C3 group; Two-sided two-sample t-test. Ratio of 1587 and 1649 cm−1: PC2 vs. C1 = 2.12 × 10−8, PC3 vs. C2 = 1.63 × 10−5, PC3 vs. C1 = 0.281; Ratio of 1711 and 1741 cm−1: PC2 vs. C1 = 5.06 × 10−4, PC3 vs. C2 = 0.995, PC3 vs. C1 = 0.005.); colors match panel b. d, Per-worm composition across regions. For each worm, the fraction of its spectra falling into C1/C2/C3 is shown as a stacked bar; colors match panels b-c. In c, the boxes show the IQR, the centerlines indicate medians and the lines outside the boxes extend to 1.5 times the IQR.
Extended Data Fig. 10 Hyperspectral FILM imaging of lipid droplet and mitochondria.
a, Fluorescence and FILM images at 1741 (ester C = O, on-resonance) and 1797 cm-1 (off-resonance) of LipiRed labelled HeLa cells. b, FILM spectral of lipid droplet marked by red circle and arrowhead in a. c, Fluorescence and FILM images at 1649 (Amide I, on-resonance) and 1797 cm-1 (off-resonance) of MitoTracker Green labelled HeLa cells. d, FILM spectral of mitochondria marked by red circle and arrowhead in c. Scale bar: 10 μm. Representative results are shown from three independent experiments.
Supplementary information
Supplementary Information (download PDF )
Supplementary Notes 1–7, Figs. 1–12 and Tables 1 and 2.
Supplementary Video 1 (download AVI )
Hyperspectral FILM imaging of lysosomes before and after SPEND processing, along with the corresponding raw, uncalibrated spectra.
Supplementary Video 2 (download AVI )
Fluorescence and hyperspectral FILM imaging, along with the corresponding raw, uncalibrated spectra of lysosomes in live C. elegans.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ao, J., Yin, J., Lin, H. et al. FILM: mapping organellar metabolism by mid-infrared photothermal-modulated fluorescence. Nat Methods (2026). https://doi.org/10.1038/s41592-026-03090-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41592-026-03090-1