Abstract
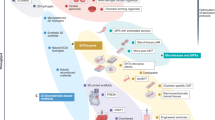
Heart rate is both an indicator and modulator of cardiovascular health. Prolonged elevation in heart rate or irregular heart rhythm can trigger the onset of cardiac dysfunction, a condition termed ‘tachycardia-induced cardiomyopathy’. While large animals have historically served as the primary model for studying this condition owing to their similar resting heart rates to humans, their use is limited by cost and throughput constraints. We recently developed the first engineered model of tachycardia-induced cardiomyopathy to overcome this technical bottleneck. Our model uses matured human engineered myocardium coupled with programmable electrical stimulation to emulate the pathophysiological changes in human heart rhythm. This in vitro model, capable of acutely and chronically modulating both beating rate and rhythm, recapitulated the clinical hallmarks of tachycardia-induced cardiomyopathy, and its utility was further validated via molecular comparisons against data from a canine model and human patients. Moreover, this model has improved the throughput and relevance to human genetics, enabling deep mechanistic explorations that were previously impossible. Here we present a comprehensive workflow detailing the fabrication and maturation of human engineered heart tissue, assembly of the electrical pacing system, functional analysis using open-source software and preparation for proteomic and transcriptomic analyses. This 5-week Protocol could be implemented by an experienced bench scientist with strong expertise in cell culture, ideally involving stem cell-derived cardiomyocytes. Given the broad implications of heart rhythm alterations in various cardiac conditions, this workflow can be employed with other biophysical and chemical cues to generate more complex and physiologically relevant cardiac models.
Key points
-
The programmable stimulation of mature engineered heart tissue (EHT) emulates diverse patterns of heartbeats associated with physiological and pathological changes, providing a platform to dissect the intricate role of heart rhythm on cardiac physiology at functional and molecular levels.
-
This procedure covers EHT fabrication and maturation; the construction of a cost-effective, portable and programmable pacing system; ready-to-use example code; the functional analysis of EHTs and sample preparation for proteomic and transcriptomic analysis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
All source data and design files are provided within the paper. RNA-seq data are available at the National Center for Biotechnology Information Gene Expression Omnibus repository, under accession number GSE242727. Source data are provided with this paper.
Code availability
Custom code is provided within the paper.
References
Fox, K. et al. Heart rate as a prognostic risk factor in patients with coronary artery disease and left-ventricular systolic dysfunction (BEAUTIFUL): a subgroup analysis of a randomised controlled trial. Lancet 372, 817–821 (2008).
Nanchen, D. et al. Resting heart rate and the risk of heart failure in healthy adults. Circ. Heart Fail. 6, 403–410 (2013).
Huizar, J. F., Ellenbogen, K. A., Tan, A. Y. & Kaszala, K. Arrhythmia-induced cardiomyopathy. J. Am. Coll. Cardiol. 73, 2328–2344 (2019).
Böhm, M. et al. Heart rate as a risk factor in chronic heart failure (SHIFT): the association between heart rate and outcomes in a randomised placebo-controlled trial. Lancet 376, 886–894 (2010).
Ling, L. et al. Irregular rhythm adversely influences calcium handling in ventricular myocardium. Circ. Heart Fail. 5, 786–793 (2012).
Slawik, J. et al. Irregular pacing of ventricular cardiomyocytes induces pro-fibrotic signalling involving paracrine effects of transforming growth factor beta and connective tissue growth factor. Eur. J. Heart Fail. 21, 482–491 (2019).
White, D. W. & Raven, P. B. Autonomic neural control of heart rate during dynamic exercise: revisited. J. Physiol. 592, 2491–2500 (2014).
Zouhal, H., Jacob, C., Delamarche, P. & Gratas-Delamarche, A. Catecholamines and the effects of exercise, training and gender. Sports Med. 38, 401–423 (2008).
Vega, R. B., Konhilas, J. P., Kelly, D. P. & Leinwand, L. A. Molecular mechanisms underlying cardiac adaptation to exercise. Cell Metab. 25, 1012–1026 (2017).
Rosen, K. M., Lau, S. H. & Damato, A. N. Simulation of atrial flutter by rapid coronary sinus pacing. Am. Heart J. 78, 635–642 (1969).
Powers, J. C. & Recchia, F. in Experimental Models of Cardiovascular Diseases: Methods and Protocols (ed. Ishikawa, K.) 309–325 (Springer, 2018).
Coleman, H. N. et al. Congestive heart failure following chronic tachycardia. Am. Heart J. 81, 790–798 (1971).
Hobai, I. A. & O’Rourke, B. Enhanced Ca2+-activated Na+–Ca2+ exchange activity in canine pacing-induced heart failure. Circ. Res. 87, 690–698 (2000).
Brixius, K., Wollmer, A., Bölck, B., Mehlhorn, U. & Schwinger, R. H. G. Ser16-, but not Thr17-phosphorylation of phospholamban influences frequency-dependent force generation in human myocardium. Pflug. Arch. 447, 150–157 (2003).
Hirt, M. N., Hansen, A. & Eschenhagen, T. Cardiac tissue engineering. Circ. Res. 114, 354–367 (2014).
Tu, C., Chao, B. S. & Wu, J. C. Strategies for improving the maturity of human induced pluripotent stem cell-derived cardiomyocytes. Circ. Res. 123, 512–514 (2018).
Strimaityte, D. et al. Contractility and calcium transient maturation in the human iPSC-derived cardiac microfibers. ACS Appl. Mater. Interfaces 14, 35376–35388 (2022).
Mannhardt, I. et al. Human engineered heart tissue: analysis of contractile force. Stem Cell Rep. 7, 29–42 (2016).
Kim, H., Kamm, R. D., Vunjak-Novakovic, G. & Wu, J. C. Progress in multicellular human cardiac organoids for clinical applications. Cell Stem Cell 29, 503–514 (2022).
Lewis-Israeli, Y. R. et al. Self-assembling human heart organoids for the modeling of cardiac development and congenital heart disease. Nat. Commun. 12, 5142 (2021).
Ng, W. H., Varghese, B., Jia, H. & Ren, X. Alliance of heart and endoderm: multilineage organoids to model co-development. Circ. Res. 132, 511–518 (2023).
Jiang, S., Feng, W., Chang, C. & Li, G. Modeling human heart development and congenital defects using organoids: how close are we? J. Cardiovasc. Dev. Dis. 9, 125 (2022).
Campostrini, G., Windt, L. M., van Meer, B. J., Bellin, M. & Mummery, C. L. Cardiac tissues from stem cells. Circ. Res. 128, 775–801 (2021).
Yang, H. et al. Transcriptome analysis of non human primate-induced pluripotent stem cell-derived cardiomyocytes in 2D monolayer culture vs. 3D engineered heart tissue. Cardiovasc. Res. 117, 2125–2136 (2021).
Querdel, E. et al. Human engineered heart tissue patches remuscularize the injured heart in a dose-dependent manner. Circulation 143, 1991–2006 (2021).
Maihemuti, W., Murata, K., Abulaiti, M., Minatoya, K. & Masumoto, H. Simultaneous electro-dynamic stimulation accelerates maturation of engineered cardiac tissues generated by human iPS cells. Biochem. Biophys. Res. Commun. 733, 150605 (2024).
Shen, S. et al. Physiological calcium combined with electrical pacing accelerates maturation of human engineered heart tissue. Stem Cell Rep. 17, 2037–2049 (2022).
Ronaldson-Bouchard, K. et al. Advanced maturation of human cardiac tissue grown from pluripotent stem cells. Nature 556, 239–243 (2018).
Hirt, M. N. et al. Functional improvement and maturation of rat and human engineered heart tissue by chronic electrical stimulation. J. Mol. Cell. Cardiol. 74, 151–161 (2014).
Ruan, J.-L. et al. Mechanical stress conditioning and electrical stimulation promote contractility and force maturation of induced pluripotent stem cell-derived human cardiac tissue. Circulation 134, 1557–1567 (2016).
Ronaldson-Bouchard, K. et al. Engineering of human cardiac muscle electromechanically matured to an adult-like phenotype. Nat. Protoc. 14, 2781–2817 (2019).
Ma, Z. et al. Contractile deficits in engineered cardiac microtissues as a result of MYBPC3 deficiency and mechanical overload. Nat. Biomed. Eng. 2, 955–967 (2018).
Wijnker, P. J. M. et al. Hypertrophic cardiomyopathy dysfunction mimicked in human engineered heart tissue and improved by sodium–glucose cotransporter 2 inhibitors. Cardiovasc. Res. 120, 301–317 (2024).
Bliley, J. M. et al. Dynamic loading of human engineered heart tissue enhances contractile function and drives a desmosome-linked disease phenotype. Sci. Transl. Med. 13, eabd1817 (2021).
Tu, C. et al. Tachycardia-induced metabolic rewiring as a driver of contractile dysfunction. Nat. Biomed. Eng. 8, 479–494 (2024).
Breckwoldt, K. et al. Differentiation of cardiomyocytes and generation of human engineered heart tissue. Nat. Protoc. 12, 1177–1197 (2017).
Tiburcy, M. et al. Defined engineered human myocardium with advanced maturation for applications in heart failure modeling and repair. Circulation 135, 1832–1847 (2017).
Yang, X. et al. Fatty acids enhance the maturation of cardiomyocytes derived from human pluripotent stem cells. Stem Cell Rep. 13, 657–668 (2019).
Parikh, S. S. et al. Thyroid and glucocorticoid hormones promote functional t-tubule development in human-induced pluripotent stem cell–derived cardiomyocytes. Circ. Res. 121, 1323–1330 (2017).
Wang, L. et al. Triiodothyronine and dexamethasone alter potassium channel expression and promote electrophysiological maturation of human-induced pluripotent stem cell-derived cardiomyocytes. J. Mol. Cell. Cardiol. 161, 130–138 (2021).
Tandon, N. et al. Electrical stimulation systems for cardiac tissue engineering. Nat. Protoc. 4, 155–173 (2009).
Qi, X. Y. et al. Cellular signaling underlying atrial tachycardia remodeling of L-type calcium current. Circ. Res. 103, 845–854 (2008).
Chen, Y.-J. et al. Effects of rapid atrial pacing on the arrhythmogenic activity of single cardiomyocytes from pulmonary veins. Circulation 104, 2849–2854 (2001).
Viero, C., Kraushaar, U., Ruppenthal, S., Kaestner, L. & Lipp, P. A primary culture system for sustained expression of a calcium sensor in preserved adult rat ventricular myocytes. Cell Calcium 43, 59–71 (2008).
Lim, C. C., Apstein, C. S., Colucci, W. S. & Liao, R. Impaired cell shortening and relengthening with increased pacing frequency are intrinsic to the senescent mouse cardiomyocyte. J. Mol. Cell. Cardiol. 32, 2075–2082 (2000).
Ritzer, A., Roeschl, T., Nay, S., Rudakova, E. & Volk, T. Rapid pacing decreases L-type Ca2+ current and alters Cacna1c isogene expression in primary cultured rat left ventricular myocytes. J. Membr. Biol. 256, 257–269 (2023).
Louch, W. E., Sheehan, K. A. & Wolska, B. M. Methods in cardiomyocyte isolation, culture, and gene transfer. J. Mol. Cell. Cardiol. 51, 288–298 (2011).
Wei, F., Pourrier, M., Strauss, D. G., Stockbridge, N. & Pang, L. Effects of electrical stimulation on hiPSC-CM responses to classic ion channel blockers. Toxicol. Sci. 174, 254–265 (2020).
Cui, C. et al. Structural and electrophysiological dysfunctions due to increased endoplasmic reticulum stress in a long-term pacing model using human induced pluripotent stem cell-derived ventricular cardiomyocytes. Stem Cell Res. Ther. 8, 109 (2017).
Thorpe, J. et al. Development of a robust induced pluripotent stem cell atrial cardiomyocyte differentiation protocol to model atrial arrhythmia. Stem Cell Res. Ther. 14, 183 (2023).
Sönmez, M. I. et al. Human induced pluripotent stem cell–derived atrial cardiomyocytes recapitulate contribution of the slowly activating delayed rectifier currents IKs to repolarization in the human atrium. EP Europace 26, euae140 (2024).
Geng, L. et al. Rapid electrical stimulation increased cardiac apoptosis through disturbance of calcium homeostasis and mitochondrial dysfunction in human induced pluripotent stem cell-derived cardiomyocytes. Cell. Physiol. Biochem. 47, 1167–1180 (2018).
Branco, M. A. et al. Transcriptomic analysis of 3D cardiac differentiation of human induced pluripotent stem cells reveals faster cardiomyocyte maturation compared to 2D culture. Sci. Rep. 9, 9229 (2019).
Shinbane, J. S. et al. Tachycardia-induced cardiomyopathy: a review of animal models and clinical studies. J. Am. Coll. Cardiol. 29, 709–715 (1997).
Patel, D., Stohlman, J., Dang, Q., Strauss, D. G. & Blinova, K. Assessment of proarrhythmic potential of drugs in optogenetically paced induced pluripotent stem cell-derived cardiomyocytes. Toxicol. Sci. 170, 167–179 (2019).
Quach, B., Krogh-Madsen, T., Entcheva, E. & Christini, D. J. Light-activated dynamic clamp using iPSC-derived cardiomyocytes. Biophys. J. 115, 2206–2217 (2018).
Lemme, M. et al. Chronic intermittent tachypacing by an optogenetic approach induces arrhythmia vulnerability in human engineered heart tissue. Cardiovasc. Res. 116, 1487–1499 (2020).
Lemoine, M. D. et al. Intermittent optogenetic tachypacing of atrial engineered heart tissue induces only limited electrical remodelling. J. Cardiovasc. Pharmacol. 77, 291 (2021).
Dwenger, M. et al. Chronic optical pacing conditioning of h-iPSC engineered cardiac tissues. J. Tissue Eng. 10, 2041731419841748 (2019).
Schwarzová, B. et al. Modulating cardiac physiology in engineered heart tissue with the bidirectional optogenetic tool BiPOLES. Pflug. Arch. 475, 1463–1477 (2023).
Monfredi, O., Dobrzynski, H., Mondal, T., Boyett, M. R. & Morris, G. M. The anatomy and physiology of the sinoatrial node—a contemporary review. Pacing Clin. Electrophysiol. 33, 1392–1406 (2010).
Min, S. et al. Versatile human cardiac tissues engineered with perfusable heart extracellular microenvironment for biomedical applications. Nat. Commun. 15, 2564 (2024).
Noor, N. et al. 3D printing of personalized thick and perfusable cardiac patches and hearts. Adv. Sci. 6, 1900344 (2019).
Sekine, H. et al. In vitro fabrication of functional three-dimensional tissues with perfusable blood vessels. Nat. Commun. 4, 1399 (2013).
Sharma, A. et al. Use of human induced pluripotent stem cell–derived cardiomyocytes to assess drug cardiotoxicity. Nat. Protoc. 13, 3018–3041 (2018).
Lian, X. et al. Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/β-catenin signaling under fully defined conditions. Nat. Protoc. 8, 162–175 (2013).
Windt, L. M. et al. Miniaturized engineered heart tissues from hiPSC-derived triple cell type co-cultures to study human cardiac function. Biochem. Biophys. Res. Commun. 681, 200–211 (2023).
Lock, R. I. et al. Macrophages enhance contractile force in iPSC-derived human engineered cardiac tissue. Cell Rep. 43, 114302 (2024).
Bhagwan, J. R. et al. Isogenic models of hypertrophic cardiomyopathy unveil differential phenotypes and mechanism-driven therapeutics. J. Mol. Cell. Cardiol. 145, 43–53 (2020).
O’Mahony, C., Elliott, P. & McKenna, W. Sudden cardiac death in hypertrophic cardiomyopathy. Circ. Arrhythm. Electrophysiol. 6, 443–451 (2013).
Nes, B. M., Janszky, I., Wisløff, U., Støylen, A. & Karlsen, T. Age-predicted maximal heart rate in healthy subjects: the HUNT fitness study. Scand. J. Med. Sci. Sports 23, 697–704 (2013).
Acknowledgements
C.T. discloses support for the research described in this study from the American Heart Association (AHA) (grant no. 20POST35080175) and the National Institutes of Health (NIH) (grant no. K99 HL164962). A.C. discloses support for the publication of this study from the NIH (grant no. F32HL173968) and AHA (grant no. 908136). S.M.N. discloses support for the publication of this study from the NIH (grant no. R01 HL162260). J.C.W. discloses support for the publication of this study from the NIH (grant nos. R01 HL176822, R01 HL163680, R01 HL141851, R01 HL141371, R01 HL113006, R01 HL130020, and U01 AI183953) and the National Aeronautics and Space Administration (grant no. 80ARC022CA003).
Author information
Authors and Affiliations
Contributions
C.T. and J.C.W. initiated and oversaw the entire study. C.T. designed and validated the pacing system and optimized the EHT fabrication method and stimulation protocol. C.T. collected all functional data. A.C. developed the method for EHT proteomic analysis and provided data. Y.L. performed transcriptomic analysis. C.T., A.C., Y.L., S.M.N. and J.C.W. wrote and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.C.W. is a cofounder and advisory board member of Greenstone Biosciences.
Peer review
Peer review information
Nature Protocols thanks Jamie Vandenberg and Aaron Baker for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key reference
Tu, C. et al. Nat. Biomed. Eng. 8, 479–494 (2024): https://doi.org/10.1038/s41551-023-01134-x
Extended data
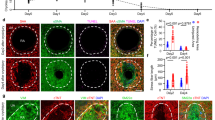
Extended Data Fig. 1 Confocal images of EHT.
Cryo-sections of EHTs were immunostained for cardiomyocyte marker (TNNT2, shown in green), nonmyocyte marker (vimentin, shown in red) and nuclei (DAPI, shown in blue). Scale bar, 50 μm.
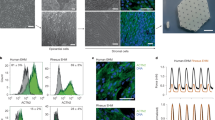
Extended Data Fig. 2 Assembly of the pacing device.
a, Custom-built H-bridge circuit on a breadboard with 830 points. b, The connection of the output wires to the H-bridge circuit. The alligator clips, shown in red and black, will be connected to the stimulation chamber. c, The power source (black adapter) connection to the H-bridge circuit. This power source determines the voltage of electrical stimulation. d, The connection of the Arduino microprocessor to the circuit. Pacing programs will be uploaded to and stored in the Arduino. Arduino requires an independent power source (power adapter in white). e, An overview of the completed pacing device. f, The Arduino and the breadboard are installed on a specialized holder (shown in blue).
Extended Data Fig. 3 Circuit diagram of the pacing system.
a, When both pins 9 and 12 are off, the circuit is open and there is no current flowing through the load (EHTs). b, When pin 9 is on, and pin 12 is off, the current passes the EHTs from left to right (shown in red). c, When pin 9 is off and pin 12 is on, current passes the EHTs from right to left (shown in blue).
Supplementary information
Supplementary Data 1
CAD design file of the stimulation chamber.
Supplementary Video 1
EHT beating without pacing.
Supplementary Video 2
EHT paced at 1.5 Hz.
Supplementary Video 3
EHT paced at 3 Hz.
Supplementary Video 4
EHT paced at 4 Hz.
Supplementary Video 5 (download MOV )
Demonstration of the pacing system using light-emitting diodes.
Supplementary Code 1 (download RTF )
Example Arduino code for different pacing programs.
Source data
Source Data Fig. 6 (download XLSX )
Contractility of EHTs being paced at different frequencies.
Source Data Fig. 8 (download XLSX )
Differentially expressed proteins in EHTs induced by sustained tachypacing.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tu, C., Caudal, A., Liu, Y. et al. Modeling heart rhythm using human engineered heart tissues. Nat Protoc 21, 827–850 (2026). https://doi.org/10.1038/s41596-025-01217-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41596-025-01217-w